1 Cardiology Unit, ASST Fatebenefratelli Sacco - Luigi Sacco University Hospital, 20157 Milan, Italy
2 Department of Cardiology, Saint Martin Private Hospital Center, 14000 Caen, France
3 Division of Cardiology, Department of Medicine, Johns Hopkins University School of Medicine, Baltimore, Maryland, MD 21205, USA
† These authors contributed equally.
Academic Editor: Peter A. McCullough
Abstract
Acute coronary syndromes (ACS) might be complicated by atrioventricular (AV) and intraventricular (IV) blocks in a significant number of cases, and often represent a diagnostic and a therapeutic challenge. These conduction disturbances are predictors of adverse prognosis, with complete AV blocks presenting the most severe outcomes, showing an increased in-hospital mortality. With the advent of emergency percutaneous coronary intervention (PCI) and the end of the thrombolysis era, the incidence of both AV and IV blocks has surely decreased, but their prognosis in this setting still remains a matter of debate. The aim of this review is to evaluate the current knowledge on AV and IV blocks in the AMI setting with or without ST segment elevation.
Keywords
- Atrioventricular blocks
- Intraventricular blocks
- Acute coronary syndrome
- Acute myocardial infarction
- Review
Acute myocardial infarction (AMI) is a life-threatening condition that needs to be diagnosed without delay. In this clinical scenario, especially when a ST-segment elevation MI (STEMI) is suspected, a 12-lead electrocardiogram (ECG) needs to be collected within 10 min after the first medical contact. Indeed, the electrocardiographic evaluation is mandatory not only to analyze the ischemic findings, but also to evaluate the presence of different conduction disturbances that may arise in the context of acute coronary syndromes (ACS). Thus, AMIs could be complicated by atrioventricular (AV) and intraventricular (IV) blocks in a significant number of cases, and often represent a diagnostic and a therapeutic challenge. These conduction disturbances are predictors of adverse prognosis, with complete AV blocks presenting the most severe outcomes, showing an increased in-hospital mortality. With the advent of emergency percutaneous coronary intervention (PCI) and the end of the thrombolysis era, the incidence of conduction disturbances has surely decreased, but the true prognosis of AV and IV blocks still remains a matter of debate. Fig. 1 summarizes different AMI types in relation to the onset of AV and IV blocks. The aim of this review is to evaluate the current knowledge on AV and IV blocks in the AMI setting, with or without ST segment elevation.
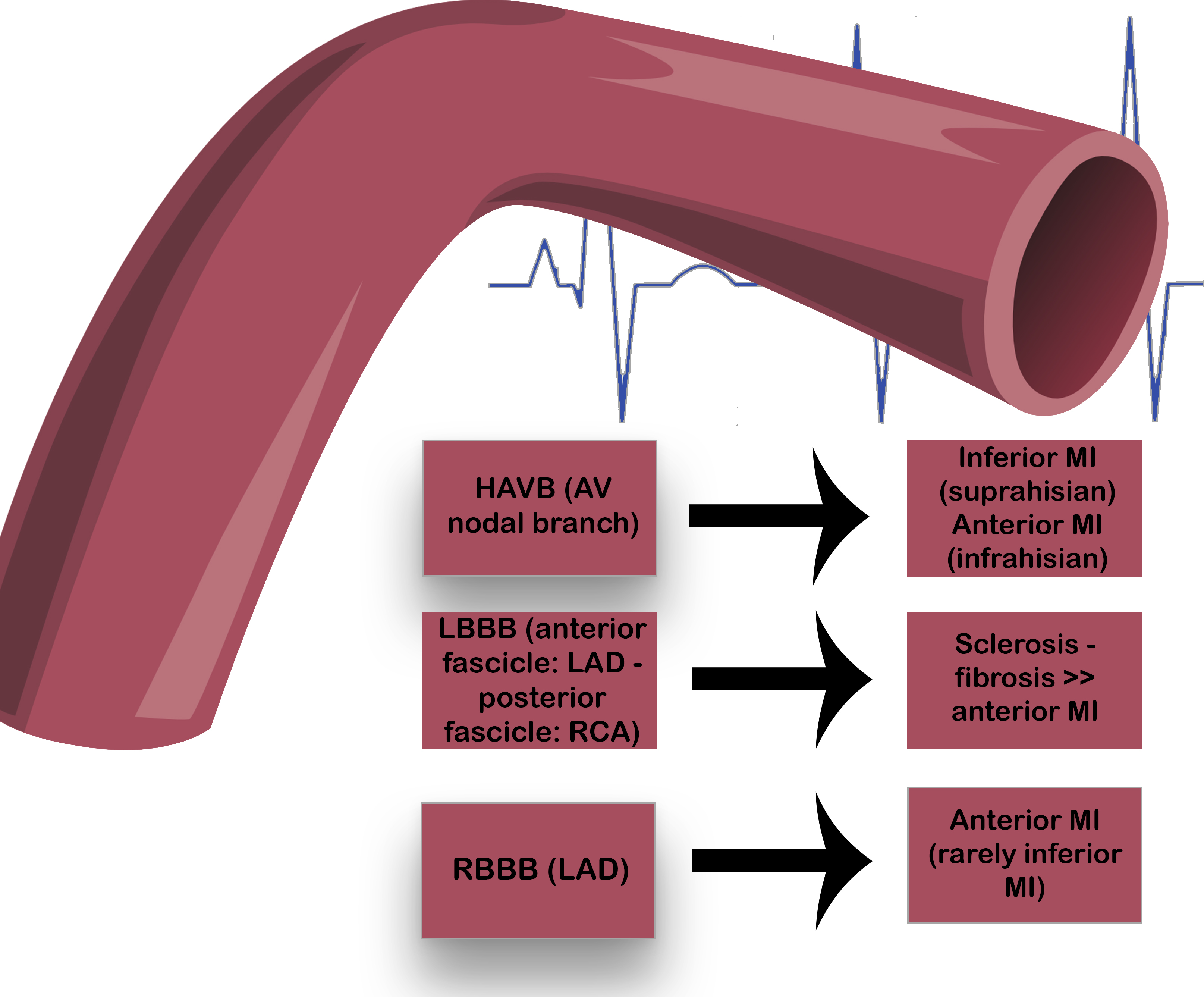 Fig. 1.
Fig. 1.Atrioventricular and intraventricular blocks in the AMI
setting.
Ischemia of the AV nodal branch may result in HAVB, with suprahisian blocks in
case of inferior MI or infrahisian blocks in case of anterior MI. Bundle branches
ischemia may derive from LAD or RCA involvement, resulting in LBBB or RBBB.
Abbreviations: AV, atrioventricular; HAVB, high-degree atrioventricular
block; LBBB, left bundle branch block; LAD, left anterior descending; MI,
myocardial infarction; RBBB, right bundle branch block; RCA, right coronary
artery.
High-degree atrioventricular block (HAVB), defined as second-degree type 2 or third-degree AV block, is an alarming finding in patients with AMI, especially in patients with STEMI, that is generally characterized by an occlusion of the culprit artery [1, 2]. Specifically, inferior wall MI, deriving from a thrombotic occlusion of the dominant coronary artery, can result in a supra-hisian AV block in 90% of patients, while patients presenting with an anterior MI usually develop infra-hisian HAVB, involving both bundle branches, below the AV node. Developing AV blocks in the setting of AMI is specifically related to an involvement of the AV nodal branch, that supplies the AV node. This branch might show significant variations in its origin: in 85–90% of individuals it arises from the right coronary artery (RCA), either from the proximal posterolateral branch (77%), or from the distal posterolateral branch (2%), or directly from the distal RCA (10% of cases) [3, 4]. In a lower number of patients (about 6–7%) it may originate from the posterior descending artery (branch of the RCA or of the left coronary artery, LCA) or from the distal circumflex branch of the LCA (approximately 3–4%) [3]. It should be specified that in cases of codominance (balanced coronary system, 10–20% of individuals), both the RCA and the left circumflex artery supply the posterior descending artery, and thereby the risk of inferior MI resulting in HAVBs is lower than in patients with right dominance (80–90%). Finally, in about 2–3% of people, the AV nodal branch may arise from both the RCA and the left circumflex artery [5]. Indeed, a dual blood supply to the AV node has often been postulated, since it has been reported that patients with inferior MI and left anterior descending (LAD) artery obstruction may have a significantly higher risk of developing complete AV block than patients with inferior MI without LAD obstruction [6].
Besides a pure ischemic damage of the AV node, additional mechanisms may result in transient AV block during inferior MI, such as the cardiodepressor Bezold—Jarisch reflex. Indeed, experimental and early clinical reports have shown that this parasympathetic response may arise suddenly after the occlusion and the recanalization of the RCA [7, 8]. Later studies have reported how this reflex response has progressively been deemed as an indicator of successful thrombolysis [9, 10, 11]. Although the exact mechanisms underlying this neurologic phenomenon remain partially unknown, a stimulation of the vagal afferents in response to sympathetic overactivity inducing bradycardia, hypotension and vasodilatation, mostly due to early coronary artery reperfusion, has been identified as the major determinant of this reflex [11].
Several reports have shown how that the incidence of HAVB may range from 3% to
14% in different clinical settings, and especially in inferior or anterior STEMI
patients, more than in non-STEMI cases [12, 13, 14, 15, 16, 17, 18, 19]. Although the incidence of HAVB
has decreased after the thrombolytic era from 5–7% to 3% [12], specific risk
factors such as older age, hypertension, diabetes, female sex, worse Killip class
at presentation and tobacco use, still represent independent predictors that may
predispose to AVB in this setting [20]. More importantly, as described in a
meta-analysis from Meine et al. [21], HAVB represented a major predictor
of mortality in the thrombolytic era especially when considering in-hospital
mortality as the endpoint (nearly 3 to 5-fold higher), while instead their
occurrence does not seem to impact long-term mortality. More recently, Auffret
et al. [22] highlighted how, although patients with HAVB had a higher
mortality rate than patients without it, HAVB was not an independent predictor of
in-hospital mortality. This finding might be likely related to the progress that
has been made in managing AMI during the last decade. Nevertheless, in another
study conducted during PCI era, including also NSTEMI patients, patients with
HAVB showed eight-fold higher mortality rates, especially when considering
patients with anterior MI [23]. Moreover, in this study, there was no significant
difference in mortality at one-year follow-up between patients with or without
HAVB [23]. A recent Danish registry including 2073 STEMI patients treated with
primary PCI had shown how, although the incidence has been reduced compared with
earlier reports, HAVB remains a severe prognostic marker in the AMI setting
[hazard ratio = 3.14 95% confidence interval (CI), 2.04–4.84, P
These findings have been further confirmed by Singh et al. [25],
analyzing the Global Registry of Acute Coronary Events (GRACE), including a total
of 59229 patients with ACS between 1999 and 2007 and providing what seems to be a
robust evidence on this topic. In particular, a total of 2.9% of patients had
developed HAVB at any point during hospitalization, 22.7% of whom dying during
in-hospital admission [adjusted odds ratio = 4.2, 95% confidence interval (CI),
3.6–4.9, P
| Authors | Years | Location | Study type | Population | Inclusion criteria | Prevalence | Mortality |
| Feigl et al. [29] | 1972–1982 | Israel | Prospective | n = 288 | HAVB | 14% | NA |
| Gang et al. [24] | 2012 | Denmark | Retrospective | n = 2073 | HAVB | 3.2% | 37% (in-hospital) |
| Nguyen e et al, [12] | 1975–2005 | U.S. | Retrospective | n = 13663 | CAVB | 4% | 43.2% (in-hospital) |
| Singh et al. [25] | 1999–2007 | International | Retrospective | n = 59229 | HAVB | 2.9% | 22.7% (in-hospital) |
| Hashmi et al. [30] | 2015–2016 | Pakistan | Prospective | n = 179 | CAVB | 7.3% | NA |
| Meine et al. [20] | 2005 | International (GUSTO-I-IIb-III, and ASSENT-II trials) | Prospective | n = 75993 | HAVB | 6.9% | 23% (in-hospital) |
| Aguiar Rosa et al. [23] | 2005–2015 | Portugal | Retrospective | n = 4779 | CAVB | 1.9% | 28% (in-hospital) |
| Auffret et al, [22] | 2006–2013 | France | Retrospective | n = 6662 | HAVB | 3.5% | 18.1% (on admission) |
| 28.6% (in-hospital) | |||||||
| Aplin et al, [13] | 1990–1992 | Denmark (TRACE trial) | Prospective | n = 6657 | CAVB | 5% | 31.6% (30-days) |
| Harpaz et al, [14] | 1992–1996 | Israel | Prospective | n = 3300 | CAVB | 3.7% | 21% (7-days) |
| 29% (30-days) | |||||||
| 35% (1-year) | |||||||
| Spencer et al. [15] | 1975–1997 | USA | Retrospective | n = 9082 | CAVB | 5% | 46.8% (in-hospital) |
| Simons et al. [16] | 1990–1993 | International (GUSTO-1 trial) | Prospective | n = 40898 | CAVB | 8.3% | 21% (in-hospital) |
| Goldberg et al. [17] | 1975–1988 | USA | Retrospective | n = 4762 | CAVB | 5.8% | 48.9% (in-hospital) |
| Hreybe et al. [18] | 1996–2003 | USA | Retrospective | n = 21807 | CAVB | 2.5% | NA |
| Clemmensen et al. [19] | 1985–1988 | International (TAMI trials) | Prospective | n = 373 | CAVB | 13% | 20% (in-hospital) |
Several studies have reported the presence of a significant interaction between the cardiovascular system and Coronavirus disease (COVID-19), often sustained by macro- and microthrombosis, as well as a direct cardiac damage, that may lead to significant myocardial injury in different ways [31, 32, 33, 34, 35, 36, 37, 38]. As highlighted in different reports, ACS and cardiac bradyarrhythmias and tachyarrhythmias are commonly reported complications in COVID-19 hospitalized patients, often impairing prognosis [39, 40, 41, 42, 43, 44, 45]. Specifically, an interesting case series from Dagher et al. [46] described 4 cases of COVID-19 patients, without previous history of arrhythmia, who developed a transient HAVB during the course of their hospitalization, not requiring permanent pacing. The authors hypothesized that a high systemic inflammatory burden, a direct viral injury to the myocardium, or a drug-related damage (e.g., hydroxychloroquine, which has however proven to be safe, though not effective [47, 48, 49]) could have induced HAVB in these patients, even if the exact underlying mechanism remained to be postulated. Another case of an infra-hisian block, otherwise requiring permanent PM implantation, has been recently reported [50]. Also in this case, a direct myocardial damage was hypothesized as the most likely cause. Indeed, as far as this can be reasonable, this panel of authors would suggest that also a transient vascular damage and/or a supply/demand imbalance resulting in myocardial ischemia, which is now regarded as the most important mechanism of cardiovascular damage in COVID-19, even in the absence of an overt AMI, may have played a significant role in these findings. However, specific data on HAVB and type 2 MI have not been investigated so far in other settings. Surely, further studies are needed to better characterize arrhythmias in COVID-19 patients and their potential links with myocardial ischemia, as well as specific studies focused on type 2 MI and its implication on transient HAVB, due do a consistent and durable supply/demand imbalance.
Intraventricular (IV) conduction disturbances in the setting of ACS represent a significant diagnostic challenge, since the ECG interpretation becomes often difficult and may lead to a late recognition of a potentially life-threatening condition. Intraventricular conduction blocks, in addition to the extent of ST-segment deviations, are regarded to be related with a more severe ischemic damage and a more rapid progression of myocardial necrosis than ST-segment deviations alone [51, 52]. When considering the vascularization of the conduction system below the AV node, it should be noted that, as for the left bundle branch, the LAD and its septal branches supply blood to the anterior fascicle, and the posterior fascicle is perfused by the RCA, whereas the right bundle branch is entirely supplied LAD and/or its proximal septal branches [53]. Due to its dual blood supply, a left bundle branch block (LBBB) is caused mainly by sclerosis and fibrosis, more than by a massive myocardial infarction, while RBBB suggests a proximal occlusion of LAD, expressing a wider infarct size in anterior MI, thus being usually associated with a higher mortality rate (see after) [54, 55]. Moreover, in a minor report, some authors have suggested that RBBB could occur not only in anterior MI, but also during inferior MI, in relation to a concurrent right ventricular enlargement, thus representing an independent predictor of adverse outcome during hospitalization regardless of infarction location in their cohort [56].
Changes in QRS duration and pattern usually reflect severe ischemia in ACS, and when ventricular conduction blocks occur, they generally define a high-risk this scenario, although the evidence is inconclusive. Indeed, few studies suggested that patients presenting with bundle branch block (BBB) did not show higher in-hospital mortality [57] (although reflecting high-risk clinical characteristics), whereas in other studies, BBB—and especially right bundle branch block (RBBB)—has been linked with major adverse cardiovascular events (MACEs) during follow-up [58, 59]. Particular attention should be paid to the new-onset of a permanent BBB which appeared to be independently associated with a higher 30-day and 7-year all-cause mortality in different reports [60, 61]. Interestingly, even a transient ventricular conduction block has shown to be a strong independent predictor of 1-year mortality, maybe because developing a BBB (especially a RBBB) usually indicates a wider ischemia in this scenario [51]. Table 2 (Ref. [26, 56, 61, 62, 63]) summarizes the main studies focused on the prevalence and related outcomes of RBBB and LBBB) in the AMI setting.
| Authors | Years | Location | Study type | Population | Inclusion criteria | Prevalence | Mortality |
| Melgarejo-Moreno et al. [62] | 1992–1994 | Spain | Retrospective | n = 1238 | RBBB | 10.9% (37.8% new-onset, 34.2% old, 28.1% indeterminate) | - Early mortality: 43.1% (new-onset) - 15.5% (old) - 15.3% (indeterminate) |
| - 1 year mortality: 58.8% (new-onset) - 35.5% (old) - 23% (indeterminate) | |||||||
| Iwasaki et al. [56] | 1997–2006 | Japan | Retrospective | n = 1265 | RBBB | 11.50% (68.3% new-onset, 13.8% old, 17.9% indeterminate) | 20.6% (in-hospital) |
| Tolppanen et al. [51] | 2005–2012 | International | Prospective | n = 199 | RBBB or LBBB | 50% (37% transient) | 68% (1-year)—this study included only patients with cardiogenic shock |
| Neumann et al. [63] | 2007–2008 | International | Prospective | n = 4067 | RBBB or LBBB | NA | 10.7% (1-year - RBBB) |
| 7% (1-year - LBBB) | |||||||
| Vivas et al. [61] | 2004–2006 | Spain | Retrospective | n = 913 | RBBB or LBBB | RBBB: 13% (77% new-onset or indeterminate, 23% old) | RBBB: 17% (in-hospital) |
| LBBB: 2% (62% new-onset or indeterminate, 38% old) | LBBB: 29% (in-hospital) |
Recognition of MI in the context of a LBBB has always been a challenge in the
emergency department (ED), since the ST-segment deviation is one of the
fundamental features of LBBB. Over the years, many criteria have been proposed to
help clinicians making a correct diagnosis in this setting, with the most
well-known being the Sgarbossa criteria. The so-called Sgarbossa rule was first
described in 1996 and it is still often used in the ED in its original version
showing a high specificity, although a low sensitivity must be taken into account
[64]. According to the original Sgarbossa rule, at least 3 points of the
following criteria are needed to diagnose MI in the presence of LBBB: (1)
ST-segment elevation of 1 mm concordant with the QRS in at least 1 lead = 5
points; (2) ST-segment depression of 1 mm in any leads V1–V3 = 3 points; (3)
discordant ST-segment elevation in any lead
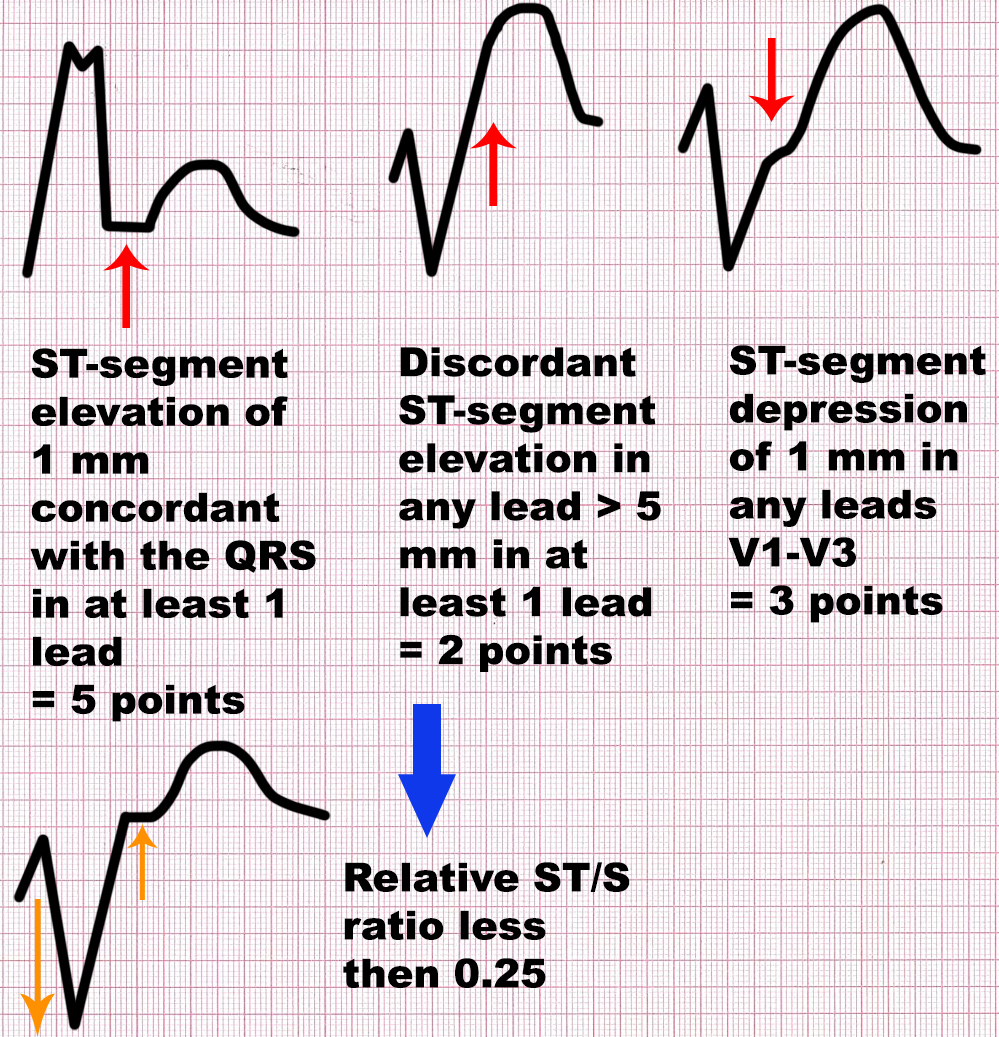 Fig. 2.
Fig. 2.Sgarbossa and modified Sgarbossa criteria. At least 3 points of
the following criteria are needed to diagnose MI in the presence of LBBB,
according to the original Sgarbossa rule: (1) ST-segment elevation of 1 mm
concordant with the QRS in at least 1 lead = 5 points; (2) ST-segment depression
of 1 mm in any leads V1–V3 = 3 points; (3) discordant ST-segment elevation in
any lead
The exact prevalence of patients presenting with RBBB in the setting of a MI is still not clear, representing about 3.1% of patients presenting to the ED with AMI [63]. The prognosis of these patients presenting with RBBB during MI is still debated, but as previously mentioned, patients with MI and RBBB generally show poor prognosis [69]. Since also in the presence of RBBB (and not only in the LBBB scenario) it might be difficult to detect transmural ischemia, potentially confounding a STEMI diagnosis, the 2017 ESC guidelines for the management of STEMI patients recommend considering an emergency activation of the catheterization laboratory when persistent ischemic symptoms occur in the presence of RBBB. A distinction should be made between permanent and transient new-onset RBBB, as well as with new-onset and previous RBBB. Indeed, in a recent review, transient RBBB appeared to be associated with a lower risk of short-term mortality, whereas patients with new-onset RBBB had higher risk when compared to those being admitted with a previous RBBB [70].
HAVB in the AMI setting has become less frequent in the PCI era, but there is still an association with an increased in-hospital mortality, although long-term mortality appears to be not affected. IV blocks represent a relevant diagnostic challenge, and specific attention should be paid to clinical presentation in order to avoid inadequate treatment delay. Mortality of patients with RBBB seems to be higher, being usually associated to a larger extent of the ischemic myocardial damage.
MS, FS, CG, and GBF conceived of this review. MS, FS, CG and AG structured and organized this review. MS, CG, FS, MD, LZ, revised the literature and synthesized study data. MS, FS, and CG wrote the original draft of this paper. MD and LZ updated this review by analyzing the latest published studies and reports. MS, FS, CG and AG organized study tables and images. AG and GBF revised and edited the original draft of this paper. GBF supervised the entire work as senior author. All authors have read and approved the submitted version.
Not applicable.
The authors would like to thank all the peer reviewers for their opinions and suggestions.
This study received no external funding.
The authors declare no conflict of interest.


