1 Baylor Heart and Vascular Institute, Dallas, TX 75226, USA
2 Baylor University Medical Center, Dallas, TX 75226, USA
3 Baylor Jack and Jane Hamilton Heart and Vascular Hospital, Dallas, TX 75226, USA
Abstract
Great attention has been paid to endothelial dysfunction (ED) in coronavirus
disease 2019 (COVID-19). There is growing evidence to suggest that the
angiotensin converting enzyme 2 receptor (ACE2 receptor) is expressed on
endothelial cells (ECs) in the lung, heart, kidney, and intestine, particularly
in systemic vessels (small and large arteries, veins, venules, and capillaries).
Upon viral infection of ECs by severe acute respiratory syndrome coronarvirus 2
(SARS-CoV-2), ECs become activated and dysfunctional. As a result of endothelial
activation and ED, the levels of pro-inflammatory cytokines (interleukin -1,
interleukin-6 (IL-6), and tumor necrosis factor-
Keywords
- COVID-19
- coagulation
- cytokines
- endothelial dysfunction
- SARS-CoV-2
- von Willebrand factor
- thrombosis
Generally, endothelial dysfunctioned (ED) involves functional phenotype modifications in order to regulate hemostasis and thrombosis and control inflammatory reactions within blood vessels (Gimbrone and García-Cardeña, 2016). In regard to an acute pro-inflammatory response, endothelial activation, particularly in small vessels, is divided into two types: type I endothelial activation (stimulation) and type 2 endothelial activation, which involve effector proteins including class-II major histocompatibility antigens, E-selectin, vascular cell adhesion molecule-1 (VCAM-1), tissue factor, interleukin-8 (IL-8) and monocyte chemotractant protein-1 (MCP-1) (Gimbrone and García-Cardeña, 2016). These proteins are detectable in the microvasculature at sites of inflammation.
From a pathological perspective, ED consists of type 1 and type II endothelial activation, as well as endothelial cell injury (e.g., endothelial cell apoptosis and necrosis), as demonstrated by pathological findings in animal studies (Zhang et al., 2012). The following terms “endothelial activation”, “endothelial dysfunction”, “endothelial damage” and “endothelial injury” may be used interchangeably (Zhang et al., 2017).
The sequence of cellular events leading to ED may be described as (1) reversible type I endothelial activation (endothelial stimulation), which represents an immediate event of ED that release pro-stored proteins vWF, P-electin, thrombin, histamine, etc; (2) reversible type II endothelial activation (over hours or days), which synthesizes new proteins such as CRP, fibrinogen, vWF, tissue factor, E-selectin, intercellular adhesion molecule-1 (ICAM-1), vascular cell adhesion molecule-1 (VCAM-1), IL-1, and MCP-1; (3) irreversible endothelial apoptosis, which leads to detachment of ECs by anoikis (a form of programmed cell death, as manifested by their detaching from the surrounding extracellular matrix); and (4) irreversible endothelial necrosis. In both endothelial apoptosis and necrosis, increased plasma levels of thrombomudulin and vWF further lead to endothelial damage (Fig. 1) (Zhang et al., 2010). Furthermore, ED may be associated with vascular EC injury in the development of vasculitis and thrombosis.
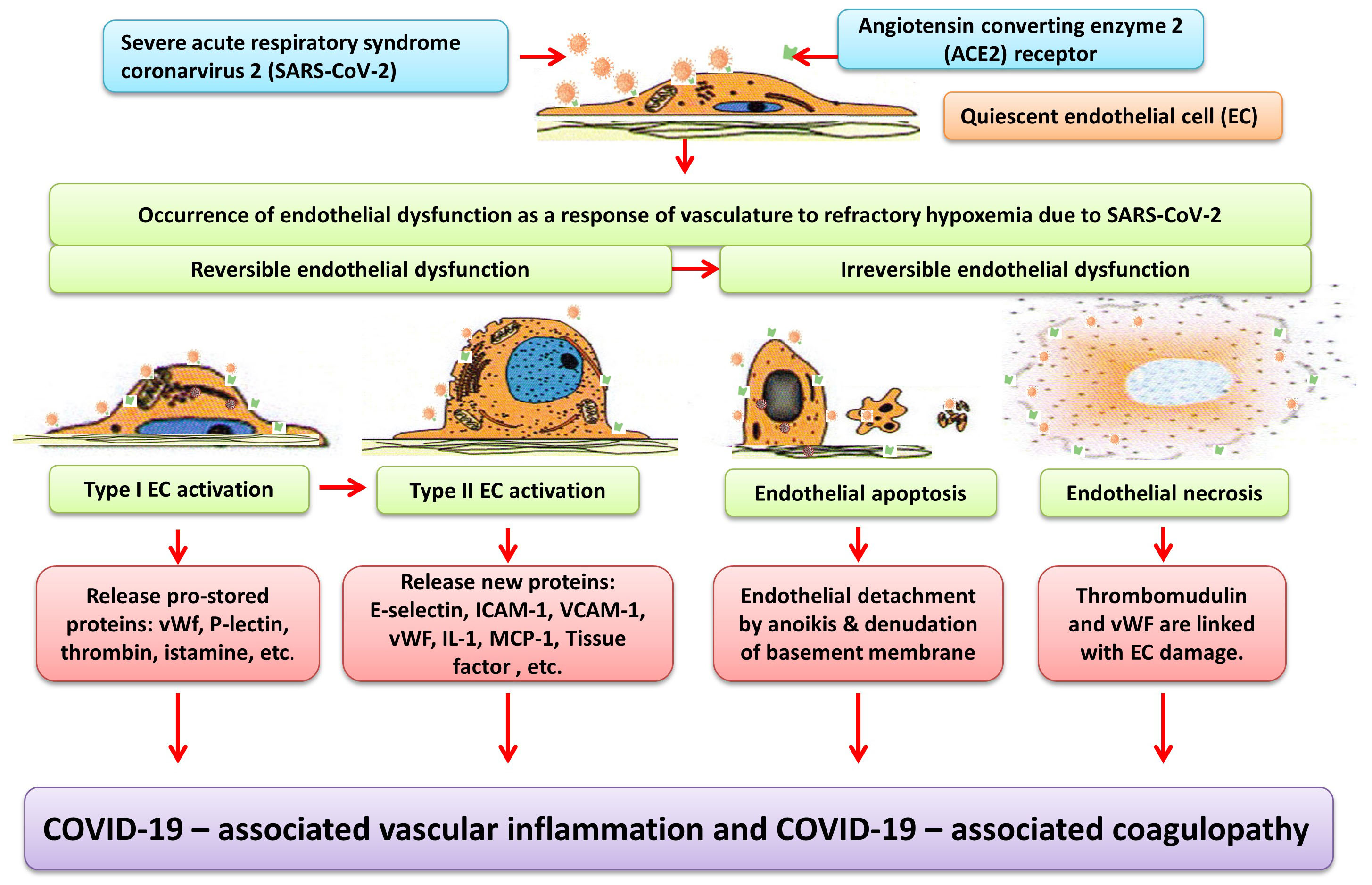 Fig. 1.
Fig. 1.Schematic diagram depicting endothelial dysfunction (ED) contributing to COVID-19-associated vascular inflammation and coagulopathy. The angiotensin converting enzyme 2 (ACE2) receptor is widely expressed on endothelial cells in the lung, heart, kidney, and intestine, allowing endothelial cells (EC) to be infected by severe acute respiratory syndrome coronarvirus 2 (SARS-CoV-2). The sequence of cellular events leading to ED begins (as early as) immediately for type I EC activation, followed by type II EC activation, EC apoptosis, and EC necrosis. Type I EC activation does not require de novo protein synthesis by means of immediately releasing pro-stored proteins such as von Willebrand factor (vWF), P-selectin, thrombin, and histamine. Conversely, type II EC activation requires de novo protein synthesis by means of releasing new proteins such as vWF, Tissue factor, fibrinogen, E-selectin, intercellular adhesion molecule-1 (ICAM-1), vascular cell adhesion molecule-1 (VCAM-1), interleukin-1 (IL-1), monocyte chemoattractant protein-1, (MCP-1), and C-reactive protein. Endothelial apoptosis results in endothelial detachment by anoikis & denudation of basement membrane, whereas endothelial necrosis results in further release of thrombomudulin and vWF. Finally, leukocyte adhesion molecules, pro-inflammatory cytokines (IL-1, IL-6, and TNF), chemokines (MCP-1), together with pro-coagulant molecules contribute to COVID-19 –associated inflammation and coagulopathy.
Previous work outlined a panel of soluble mediators as biomarkers of endothelial
activation in drug-induced vascular injury (Zhang et al., 2010). Vascular
inflammation is mediated by endothelial adhesion molecules: E-selectin
(endothelial–leukocyte adhesion molecule 1), immunoglobulin gene superfamily
(ICAM-1, VCAM-1, etc.), pro-inflammatory cytokines (TNF-
It is worth noting that P-selectin is released from Weibel-Palade bodies of
activated ECs’ cytoplasm and also from a-granules of activated platelets. The vWF
and vWF pro-peptides are also released from Weibel-Palade bodies of activated ECs
as well as platelets, megakaryocytes, and plasma. Furthermore, thrombomodulin is
released from activated ECs as well as monocytes, macrophages, and leukocytes
(Zhang et al., 2010). Therefore, the activities of both pro- and
anti-coagulant molecules are involved in activated ECs, platelets, and
megakaryocytes. Furthermore, it is suggested that when thrombomodulin is
suppressed, IL-
Indeed, increased levels of pro-inflammatory cytokines (e.g., IL-2R, IL-6,
TNF-
Escher et al. (2020) reported a 72-year-old previously healthy male patient who presented with acute respiratory distress syndrome. Continual increases of D-dimer (a marker of fibrin degradation) were observed on day 4, day 11, and day 21. On day 21, the patient had significant elevation of vWF, with vWF: antigen and vWF: activity, as well as increased Factor VIII clotting activity. The profile of increased vWF family likely represents reversible type I endothelial activation (endothelial stimulation) and type II cell activation, as described in the preceding section. The authors therefore suggest that endothelial activation is likely mediated by the ACE2 receptor.
A pathological study of COVID-19 by Varga et al. (2020) identified viral inclusion bodies in a peritubular space and viral particles in ECs of the glomerular capillary loops by using electron microscopy. The characteristic appearance of aggregates of viral particles is that of a dense circular surface and lucid center. In addition, an immunohistochemical staining of caspase 3 revealed apoptosis of ECs and apoptotic bodies in both small intestine and lung sections. Further, post-mortem histology displayed an accumulation of inflammatory cells associated with endothelium in the heart, small bowel, and lymphocytic endotheliitis in the lung, heart, kidney, liver, and small intestine. The study illustrates a prominent feature of ED occurring in COVID-19. First, ECs can directly be infected by SARS-CoV-2 because the ACE2 receptor is expressed widely in ECs. Second, after direct viral infection of ECs, ED leads to diffuse vascular inflammation (endotheliitis). Third, over time, reversible ED (endothelial activation) progresses to irreversible ED (endothelial apoptosis and necrosis). Ultimately, ED induces vascular dysfunction of different organs.
Ackermann et al. (2020) utilized immunohistochemical staining to discover a
significantly larger average number of ACE2-positive ECs per field of view (0.55
COVID-19-induced endotheliitis may be attributed to endothelial adhesion
molecules (selectins, ICAM-1, VCAM-1), pro-inflammatory cytokines (IL-6, IL-2
receptor, and TNF-
There is evidence to support increased levels of ED-released mediators not only induce ED, but also induce a prothrombotic state. It is noteworthy that micro- and macrovascular thrombosis both are present in venous and arterial circulations, and that venous thrombotic events (e.g., pulmonary embolism) are associated with elevated D-dimer in COVID-19 patients. Klok et al. (2020) reported a 31% incidence of thrombotic complications for patients in the intensive care unit with COVID-19 infections. Tang et al. (2020) reported abnormal coagulation parameters, including significantly higher levels of D-dimer and fibrin degradation product, longer prothrombin time and activated partial thromboplastin time in the non-survivors with COVID-19. Léonard-Lorant et al. (2020) demonstrated that patients with pulmonary embolus had higher D-dimer levels than those without. Upon autopsy, Fox et al. (2020) discovered that thrombotic microangiopathy in the small vessels and capillaries of the lungs contributed to death in patients with severe COVID-19. As in others’ findings, D-dimer levels were markedly elevated (Fox et al., 2020). Pulmonary microthrombi were characterized by the appearance of small and firm thrombus, multi foci and within small pulmonary artery and venule of the cut section of peripheral parenchyma (Fox et al., 2020). Thrombosed small vessels were immunostained with vWF; however, CD61 immunostains also showed additional fibrin, platelet thrombus, and positive stained megakaryocytes (Fox et al., 2020). These findings suggest that pulmonary thrombosis is involved in other cellular events, platelets, and megakaryocytes. The mechanisms for pulmonary thrombosis are likely manifold in COVID-19 patients; however, it is presumed that ED is an initial major event, which can further trigger activation of platelets and megakaryocytes, thereby causing an abnormal anticoagulant status. Ackermann et al. (2020) discovered widespread thrombosis with microangiopathy, which is characterized by multiple fibrinous microthrombi, in the alveolar capillaries and postcapillary venules. Finally, a review by Becker (2020) provides a timely examination of COVID-19-associated coagulopathy, highlighting distinctions from disseminated intravascular coagulopathy and thrombotic microangiopathy and reiterates that COVID-19-associated coagulopathy is marked by elevated D-dimer levels in the peripheral blood, indicating a breakdown of fibrin polymers by plasmin.
There have been autopsy findings to suggest that COVID-19 is associated with vascular inflammation and coagulopathy. Pulmonary pathology from 23 autopsies of COVID-19 decedents was characterized by acute interstitial pneumonia with diffuse alveolar damage (Buja et al., 2020). Another notable feature in that examination was fibrin-rich thrombi within capillaries and small blood vessels, as well as large pulmonary thromboemboli (Buja et al., 2020). Further, thrombotic microangiopathy and pulmonary thromboembolism in the lungs, along with endotheliitis and microvascular dysfunction in the cardiovascular system, and virus induced pro-coagulant state and coagulopathy and predisposition of deep vein thrombosis and pulmonary thromboembolism in the hematologic system were observed (Buja et al., 2020). In another autopsy report in the United States, Magro et al. (2020) noted a pattern of tissue damage consistent with complement-mediated microvascular injury in the lung and/or skin of 5 individuals with severe COVID-19. Their results highlight the role of complement activation and microvascular thrombosis in cases of persistent, severe COVID-19. Microthrombi, attributed to ED, within small lung arteries and prostate were reported in Germany (Wichmann et al., 2020). Glomerular capillary microthrombi were observed in both kidneys from a patient with COVID-19 in Japan, indicating early signs of disseminated intravascular coagulation (Adachi et al., 2020). In addition, fibrinous thrombi in alveolar arterioles were reported at autopsy in Brazil (Nunes Duarte-Neto et al., 2020). The authors suspected that virus-induced EC dysfunction may have been involved, but it was not determined at the time of publication (Nunes Duarte-Neto et al., 2020). An examination in Italy considered post-mortem wedge liver biopsies from 48 patients who died from severe pulmonary COVID-19 and respiratory failure. The authors found partial or complete luminal thrombosis of portal and sinusoidal vessels, indicating that not only the lungs, but also the endothelial layer of blood vessels were targeted by the virus (Sonzogni et al., 2020).
In non-autopsy studies, coagulopathy is a common abnormality in patients with COVID-19. For example, a case of acute portal vein thrombosis, which is accompanied by a pro-thrombotic profile of increased levels of vWF, D-dimer, and fibrinogen in addition to increased levels of IL-6 and C-reactive protein, was reported in Italy. That case was characterized by sepsis-related hypotension and portal vein thrombosis relevant to impairment of arterial and venous hepatic blood flow; the patient’s symptoms rapidly improved following anticoagulation (La Mura et al., 2020). In France, 69% of anticoagulated patients with severe COVID-19 illness had venous thromboembolism (Llitjos et al., 2020).
ECs may serve as antigen-presenting cells. By expressing major
histocompatibility complex (MHC) class I and MHC II antigens, ECs contribute to
innate and adaptive immune responses. However, the number of T cells activated by
ECs is far fewer than those activated by professional antigen-presenting cells.
Cytokine-induced endothelial activation and cell adhesion in the endothelium, as
well as cytokine-increased MHC class I or II molecules in the endothelium have
been studied in this context. Cytokines, such as the interferon family, TNF
family, IL-1, and IL-2 increase MHC class I or II antigen expression in the
endothelium. For example, TNF can enhance IFN-
ED and endothelial activation likely co-determine an uncontrolled immune response in COVID-19 (Teuwen et al., 2020). However, many questions remain to be determined. Among these questions are (1) to what extent this subtype of capillary ECs is involved in the immune response against SARS- CoV-2 infection (Teuwen et al., 2020) (2) how ECs act synergistically with other antigen-presenting cells to actively participate in innate and adaptive immune responses; and (3) how much ECs stimulate proliferation of CD4 and CD 8 lymphocytes in COVID-19. Further investigation is needed to prove the effect of ECs in immune response.
Evidence is emerging to suggest that ED is a contributing factor to vasculopathy and coagulopathy in COVID-19. The vascular endothelium and the various manifestations of its dysfunction in the context of COVID-19 have been considered to be cardinal features. Further studies regarding the role of ED in COVID-19 may have important clinical implications for possible therapeutic interventions.
JZ designed and drafted this work. KMT and PMC critically revised this work. All authors gave final approval.
This work was partially funded by the Baylor Health Care System Foundation.
The authors declare no conflicts of interest statement.

