1 Pharmacy Department, The First Affiliated Hospital of Bengbu Medical University, 233000 Bengbu, Anhui, China
2 School of Pharmacy, Bengbu Medical University, 233030 Bengbu, Anhui, China
†These authors contributed equally.
Abstract
Epilepsy is a central nervous system disorder characterized by abnormal neuronal discharges in the brain. The purpose of this study was to investigate the protective effects of a caspase-1 inhibitor on glial hyperactivation and neuronal apoptosis in epilepsy.
A pilocarpine-induced status epilepticus (SE) mouse model was established. Belnacasan (VX765), a caspase-1 inhibitor, was administered intraperitoneally. ELISA was used to detect inflammatory cytokines interleukin (IL)-1β, IL-6, and tumor necrosis factor-alpha (TNF-α) in peripheral blood. Immunohistochemistry and transmission electron microscopy were used to evaluate glial activation and neuronal damage in the hippocampus. Western blotting was performed to detect caspase-1 and Gasdermin D N-terminal (GSDMD-N) expression. In vitro, primary glial cells were stimulated with lipopolysaccharide (LPS), and the effects of glial conditioned medium on HT22 neuronal apoptosis were assessed using cell counting kit-8 (CCK-8) and flow cytometry.
In vivo experiments showed that, as epilepsy progressed, the levels of pro-inflammatory cytokines IL-1β and IL-6 in peripheral blood were significantly increased, consistent with findings in patients with epilepsy. At 21 days after epilepsy induction, the numbers of hyperactivated microglia and astrocytes increased significantly and exhibited activation-related features such as organelle swelling, whereas neuronal numbers were markedly reduced and displayed cytological features of apoptosis. VX765 significantly alleviated seizure frequency and severity in epileptic mice and attenuated peripheral blood levels of IL-1β and IL-6, hippocampal caspase-1 activity, glial hyperactivation, and neuronal apoptosis. In vitro experiments demonstrated that glial conditioned medium (CMG) promoted apoptosis of HT22 neurons by regulating Bcl-2-associated X protein (Bax) and B-cell lymphoma 2 (Bcl-2) expression, whereas VX765 alleviated HT22 neuronal apoptosis by inhibiting the secretion of inflammatory factors from glial cells.
These results indicate that inhibiting glial cell hyperactivation and neuroinflammation via caspase-1 inhibition may represent a potential therapeutic strategy for epilepsy.
Keywords
- epilepsy
- glial hyperactivation
- caspase-1
- VX765
- neuroinflammation
Epilepsy is a neurological disorder characterized by abnormal neuronal activity that affects approximately 70 million individuals worldwide [1]. The overall prevalence of epilepsy in China is approximately 1.68 per 1000 individuals [2]. Epilepsy is often accompanied by psychological, cognitive, and behavioral disorders and is associated with an increased risk of disability and accidental injury [3]. A previous study has reported that seizures are accompanied by elevated levels of inflammatory cytokines and activation of astrocytes and microglia [4]. Neuron–glial interactions are essential for maintaining synaptic homeostasis and plasticity. In addition, perivascular microglia, macrophages, and astrocytes are closely associated with cerebral microvasculature and lymphatic function [5]. However, the pathological mechanisms linking glial cell activation (astrocytes or microglia) to neuronal apoptosis during epilepsy remain poorly understood.
Glial cells account for more than 90% of cells in the brain, and their primary
functions include nourishing, supporting, and protecting neurons [6]. Local or
systemic inflammatory responses can lead to aberrant neural connectivity and
excessive activation of glial cells. Astrocytes and microglia can transition from
a resting state to an activated state in response to injury and stress, thereby
maintaining brain homeostasis and limiting tissue damage [7, 8]. When normal
feedback mechanisms fail to resolve inflammation, glial cell–mediated
inflammatory processes may promote the onset and progression of epilepsy [9].
After seizures, levels of interleukin (IL)-1
The current antiepileptic drugs are mainly Na+ and Ca2+ ion channel blockers. These drugs primarily control seizure symptoms. However, they do not prevent the development of epilepsy and are often associated with drug resistance or adverse effects [14]. Belnacasan (VX765) is an oral prodrug and a caspase-1 inhibitor capable of penetrating the blood brain barrier (BBB) [15]. This study aimed to elucidate changes in pro-inflammatory factors in peripheral blood during epilepsy progression and to investigate the effects of VX765 on microglial and astrocytic activation, glial hyperactivation, and neuronal apoptosis. In addition, the effects of glial cell–conditioned medium on neuronal injury and the protective role of VX765 against neuronal apoptosis were examined in vitro. Together, these findings help clarify the role of neuroinflammation regulation as a potential therapeutic strategy for epilepsy.
Male C57BL/6J mice (12 weeks old, weighing 25
The animal experiments described in this study were conducted in strict accordance with the ARRIVE guidelines and the National Institutes of Health (NIH) Guide for the Care and Use of Laboratory Animals (8th edition, 2011). All efforts were made to minimize animal suffering and reduce the number of animals used. In accordance with the requirements of the Animal Ethics Committee of Bengbu Medical University, the experimental mice were humanely euthanized by cervical dislocation after anesthesia at the end of the study.
First, anisodamine (1 mg/kg, HY-N0584, MedChemExpress (MCE), Monmouth Junction, NJ, USA) was administered intraperitoneally to mice to reduce peripheral cholinergic effects. After 30 min, pilocarpine (300 mg/kg, P129614, Aladdin, Shanghai, China) was given to induce status epilepticus (SE) [16]. The animals in the control (Ctrl) group were given the same amount of drug solvent.
We first treated the mice with VX765 (HY‑13205, MedChemExpress (MCE)) or carbamazepine (CBZ, HY‑B0246, MedChemExpress (MCE)) once daily for 4 days via intraperitoneal injection, and then induced the epilepsy model with pilocarpine as described above. The experiment was initially divided into nine groups, nine mice in each group, including a control group (Ctrl group, saline 1 mL/kg), epilepsy groups at 3, 7, 14, and 21 days, VX765 group (200 mg/kg, once a day for 4 days), VX765+SE group, CBZ group (14 mg/kg, once a day for 4 days), and CBZ+SE. The above administrations involved intraperitoneal injection (i.p.). The occurrence and frequency of spontaneous seizures were recorded on video. Mice were intraperitoneally injected with 2% pentobarbital sodium (40 mg/kg, P0225, Tongshan, Shanghai, China) to ensure that the animals were generally anesthetized. After the anesthetic effect was satisfied, the mice were euthanized, and subsequent experiments were conducted.
To determine the effects of VX765 on the intensity of SE, the epileptic seizure grades were evaluated using the Racine Scale. The rating method of epileptic seizures involved the following: Grade I, ear and face convulsions; Grade II, nod; Grade III, anterior limb clonus or posterior limb tilt; Grade IV, bilateral forelimb clonic seizures or standing; and Grade V, repeated tonic clonic seizures or falls. SE was defined as an epileptic seizure lasting more than 5 min. A mouse model with recurrent epileptic seizures for more than 60 min without death was defined as a successful model. If the first administration reached grade IV or above, and the mouse died after the end of administration, it was defined as sudden death, and was eliminated.
An enzyme-linked immunosorbent assay (ELISA) kit (20210326, Jiancheng
Bioengineering Institute, Nanjing, Jiangsu, China) was used to analyze the levels
of inflammatory cytokines (IL-1
Six left hippocampal tissues were homogenized and the protein supernatant was obtained. Caspase-1 enzyme activity in the mouse hippocampal tissue was analyzed using a caspase-1 activity assay kit (C1101, Beyotime). Briefly, the caspase-1-containing tissue supernatant was incubated with the substrate, Ac-YVAD pNA, to generate pNA, and the mixture was incubated at 37 °C for 1–2 h. The activity was then detected at 405 nm using a microplate reader, with each sample analyzed in triplicate.
Three whole brains from each group were prepared for immunohistochemical staining. After PBS perfusion, precooled 4% paraformaldehyde (BL539A, Biosharp, Guangzhou, China) was used for perfusion until the mice were stiff. Then, the brain was decapitated, and soaked in 4% paraformaldehyde solution for 24 h, followed by dehydration in 20%–30% sucrose solution for 7 days. After blocking endogenous peroxidase with 3% H2O2, serum was added. Then, primary antibody [rabbit antiGFAP (glial fibrillary acidic protein, 1:500, Catalog No. GB11096, Servicebio, Wuhan, Hubei, China), rabbit antiIBA1 (ionized calciumbinding adapter molecule 1, 1:500, Catalog No. GTX635363, GeneTex, Irvine, CA, USA), and mouse antiNeuN (neuronal nuclei, 1:500, Catalog No. ab279296, Abcam, Cambridge, UK)] was added and incubated at 4 °C overnight, followed by washing three times with PBS, every 5 min. The sample was then incubated in secondary antibody (BL003A, Biosharp) for 1 hour at room temperature and washed three times with PBS every 5 min. Then, 3,3 N-diaminobenzidine tetrahydrochloride (DAB, Catalog No. DAB-0031, Biosharp) was added, followed by observation using a microscope, to determine the termination time according to the color. Three slices were randomly taken from each group, and the positive areas and cell numbers of the mouse hippocampus were statistically analyzed using Case Viewer software (2.4.0.119028, Servicebio, Wuhan, Hubei, China).
Three right hippocampal tissues were taken after perfusion and transmission electron microscopy analyses. The hippocampal tissues of mice were quickly preserved at 4 °C and the fixative solution and the sampling apparatus were precooled to 4 °C. The hippocampal tissue specimens were cut into small 1-mm pieces and placed in the electron microscope fixation solution (HY-DY3001, MedChemExpress, Monmouth Junction, NJ, USA). After fixation at 4 °C for 2 h, 0.1 M sodium dimethyl arsenate buffer was used for rinsing, three times. Alcohol gradient dehydration was then conducted, with a dehydration time of 10 min for each concentration of alcohol, to a final concentration of 100%. The tissues were then embedded in epoxy resin (14300, Electron Microscopy Sciences, Hatfield, PA, USA) and sectioned. Ultrathin sections were stained with uranyl acetate (22400, Electron Microscopy Sciences) and lead citrate (17800, Electron Microscopy Sciences), and then detected with an H-7500 transmission electron microscope (Hitachi, Tokyo, Japan).
Three right hippocampal tissues were used for western blotting. Hippocampal
tissue was fully homogenized using a tissue grinder (Cat. No. MagMixA, Magen
Biotechnology Co., Ltd., Guangzhou, Guangdong, China) at 60 Hz for a total time
of 60 seconds. The sample was then centrifuged at 12,000 rpm for 20 min at 4 °C,
followed by the collection of the supernatant. The protein was then denatured at
95 °C for 5 min. The protein samples were then resolved using Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis (SDS-PAGE, E8042,
Amresco/Shanghai Jinshan Biotechnology Co., Ltd., Shanghai, China). After
electrophoresis, the proteins were transferred to nitrocellulose membranes. A
rapid blocking solution (P0252, Beyotime) was used for blocking for 1 hour at
room temperature. The membrane was then incubated with the primary antibody at 4
°C overnight. The primary antibodies included rabbit anti-GSDMD (1:1000, G7422, Merck,
Sigma-Aldrich, Darmstadt, Germany), mouse anti caspase-1 (AG-20B-0048, AdipoGen Life Sciences,
San Diego, CA, USA), B-cell lymphoma 2 (Bcl-2, BL-0062-1, BioSharp), Bcl-2-associated X protein (Bax, BL-0056-1, BioSharp), and
mouse anti
Fifteen C57BL/6J mice within 72 h of birth were disinfected twice with alcohol, decapitated, and the hippocampus removed. The brain skin and skull were cut along the sagittal suture of the mouse brain, and the pia mater and blood vessels were stripped, leaving the hippocampus. The hippocampus specimens were washed with PBS, then placed in a centrifuge tube, followed by the addition of an appropriate amount of 0.25% trypsin solution (G4004, Servicebio), homogenized with a tissue grinder, mixed with the homogenate, and digested in a 37 °C, 5% CO2 incubator (51030285, Thermo Fisher Scientific, Waltham, MA, USA) for 15–20 min. Prepared Dulbecco’s Modified Eagle Medium/Nutrient Mixture F-12 (DMEM-F12) medium (G4611, Servicebio) was used to terminate the digestion, followed by filtration through a 200-mesh screen, centrifugation at 1000 rpm for 5 min at 4 °C, and then the supernatant discarded. The cell pellet was resuspended with DMEM-F12, inoculated into a tissue culture flask, and placed in an incubator. After 24 h, the DMEM-F12 medium was changed, and then changed every three days.
The purified glial cells were seeded in a large dish or plate for subsequent
experiments, 2
The effect of glial supernatant on neuronal function was detected using the
CCK-8 cell proliferation assay and flow cytometry. HT22 cells were seeded in
96-well plates at a density of 1
Flow cytometry was used to detect the effects of inflammatory glial cell
supernatant on neurons, 6 well plates, 2
Prism 7.0 software (GraphPad, San Diego, CA, USA) was used to analyze the data.
All data are expressed as the mean
Several studies have reported that seizures are accompanied by cerebral
neuroinflammation. The epilepsy model was induced with pilocarpine, and
spontaneous epileptic seizures occurred on the 0th day, the 3rd day, the 7th day,
the 14th day, and the 21st day respectively (Fig. 1A). The results showed that
there was no statistically significant difference in seizure number from 0 to 21
days after the epilepsy model was established (Fig. 1B). The severity of the
mouse epilepsy model reached its peak on the 21st day (Fig. 1C). To verify
whether pilocarpine induced the epileptic mouse model with inflammation,
IL-1
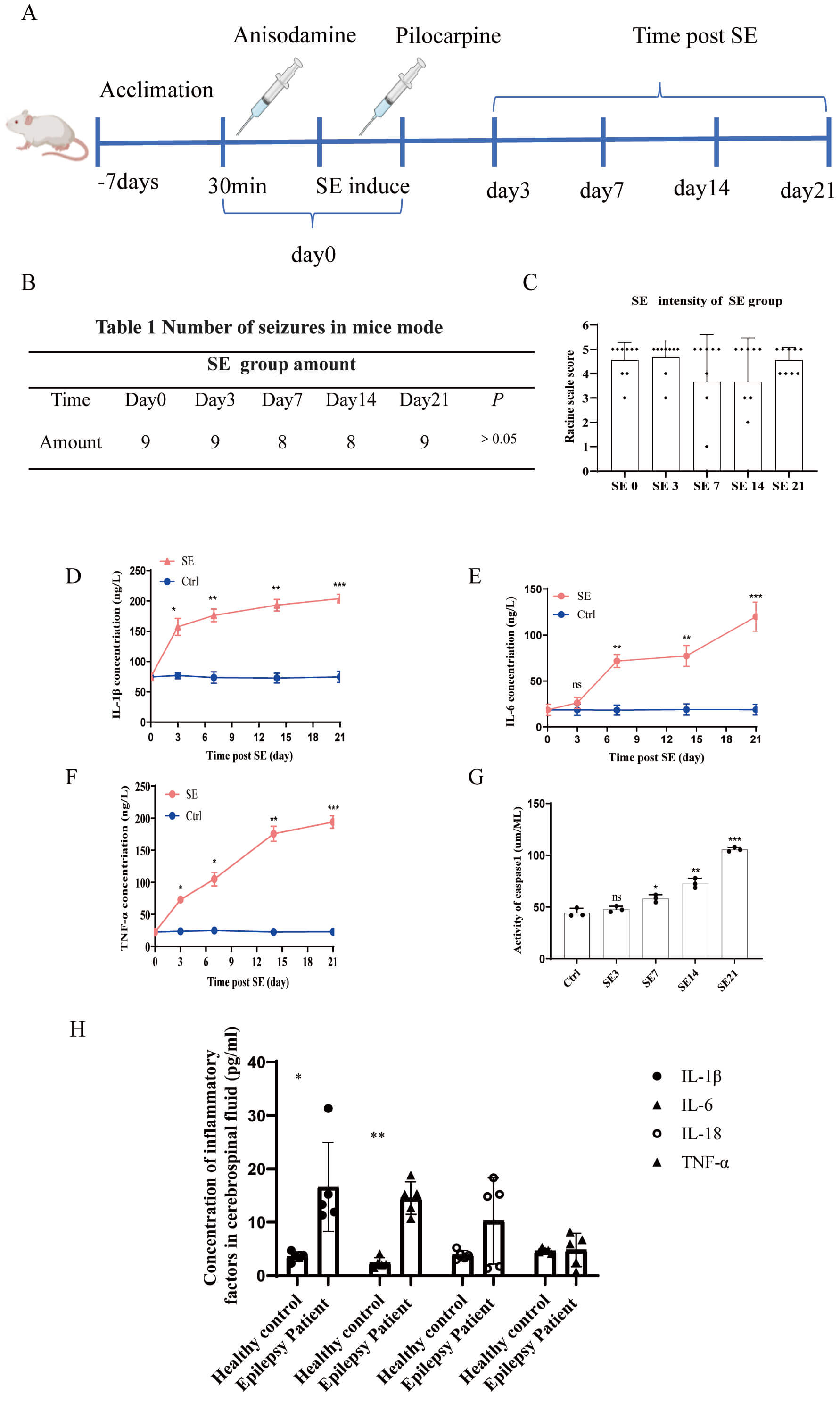 Fig. 1.
Fig. 1.
Pilocarpine-induced neuroinflammation in the peripheral serum of
the epileptic mouse model. (A) The pilocarpine-induced epilepsy method on days
3, 7, 14, and 21. (B) Table of seizure numbers of mice in the SE group at
different times. (C) Bar graph of seizure intensities of SE mice in the SE group
at different times (n = 9/group). (D–F) Contents of inflammatory factors on days
3,7,14, and 21 in the epileptic mouse model. (G) Concentrations of activated
caspase-1 on days 3, 7, 14, and 21 in the hippocampal areas of the epileptic
mouse model. (H) Histogram of inflammatory factors in the cerebrospinal fluid of
healthy and epileptic patients (n = 5/group). ns indicates no statistical
significance. *p
Hippocampal sclerosis, a neuropathological hallmark of temporal lobe epilepsy, is characterized by neuronal loss, gliosis, and synaptic reorganization. Because seizures in epileptic mice were accompanied by a significant increase of inflammatory factors and activated caspase-1 in the hippocampus, we further detected the changes in glial cells and neurons in murine brain tissue. Three mice from each group were selected for immunohistochemistry and electron microscopy assays. The results showed that GFAP-labeled positive cells started to activate during the early stage of SE, and the hyperactivation increased significantly on 7 days after SE, with the largest hyperactivation on the 21st day (Fig. 2A,D). IBA1-labeled positive cells showed no significant activation on day 3, while the activation was significantly increased on days 7 and 21 (Fig. 2B,E). Neuronal nuclei antigen (NEUN)-labeled positive cells continued to decrease on days 3, 7, and 21 after SE (Fig. 2C,F). The results showed that astrocytes and microglia in the hippocampal region of the brain after SE were swollen and rounded, while the cell membranes were ruptured, with death mainly from pyroptosis. However, death was mainly due to apoptosis, because cell bodies of neurons were atrophic with an intact cell envelope (Fig. 2G). These results showed that seizures were accompanied by hyperactivation of glial cells and apoptosis of neurons.
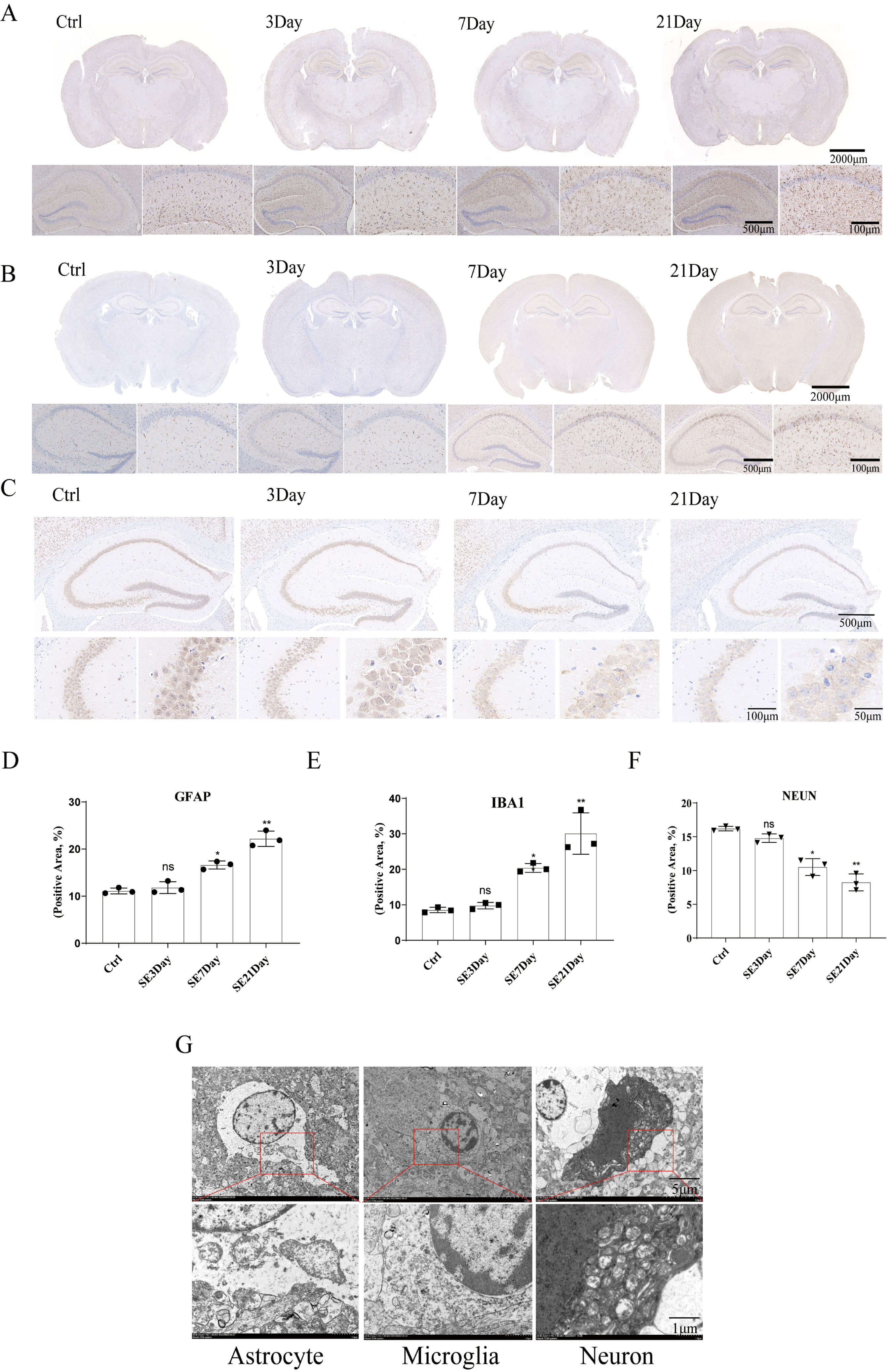 Fig. 2.
Fig. 2.
The activation of astrocytes and microglia, and reduction of
neurons in the SE mouse brain induced by pilocarpine. (A,D)
Immunohistochemistry. The scale bars are 2000 µm, 500 µm,
and 100 µm, respectively. IHC images and bar graphs of the number of
activated astrocytes of the SE mouse hippocampus on days 3, 7, and 21 in the
control and epilepsy groups. (B,E) IHC images and bar graphs of the number of
activated microglia of the SE mouse hippocampus on days 3, 7, and 21 in the
control and epilepsy groups. The scale bars are 2000 µm, 500 µm, and
100 µm, respectively. (C,F) IHC images and bar graphs of the reduced
numbers of neurons of the mouse hippocampus on days 3, 7, and 21 in the control
and epilepsy groups. The scale bars are 500 µm, 100 µm, and 50
µm, respectively. (G) Activation of astrocytes and neuronal apoptosis in
the SE mouse brain (n = 3/group). The scale bars are 5 µm and 1 µm. ns
indicates not statistically significant. *p
Epileptic mice were intraperitoneally injected with VX765 or the positive control drug CBZ, both once a day for a total of four times, and then the epilepsy model was induced with pilocarpine (Fig. 3A). The results showed that all nine mice in the SE group had seizures, with seizure levels ranging from 4–5. Both the VX765+SE and CBZ+SE groups had one mouse seizure and seizure levels of grades 2 and 1, respectively (Fig. 3B,C). These results showed that both VX765 and carbamazepine had good anti-epileptic effects, suppressing the intensities of seizures.
 Fig. 3.
Fig. 3.
VX765 suppressed the seizure intensity of the SE mouse. (A) VX765 administration and the pilocarpine-induced epilepsy method. (B) Table of seizure numbers of mice in each group. (C) Bar graph of seizure intensities of SE mice in each group (n = 9/group). CBZ, carbamazepine.
VX765 is a specific inhibitor of caspase-1 and has powerful anti-inflammatory
functions under various pathological conditions [18]. ELISA results showed that
the levels of IL-1
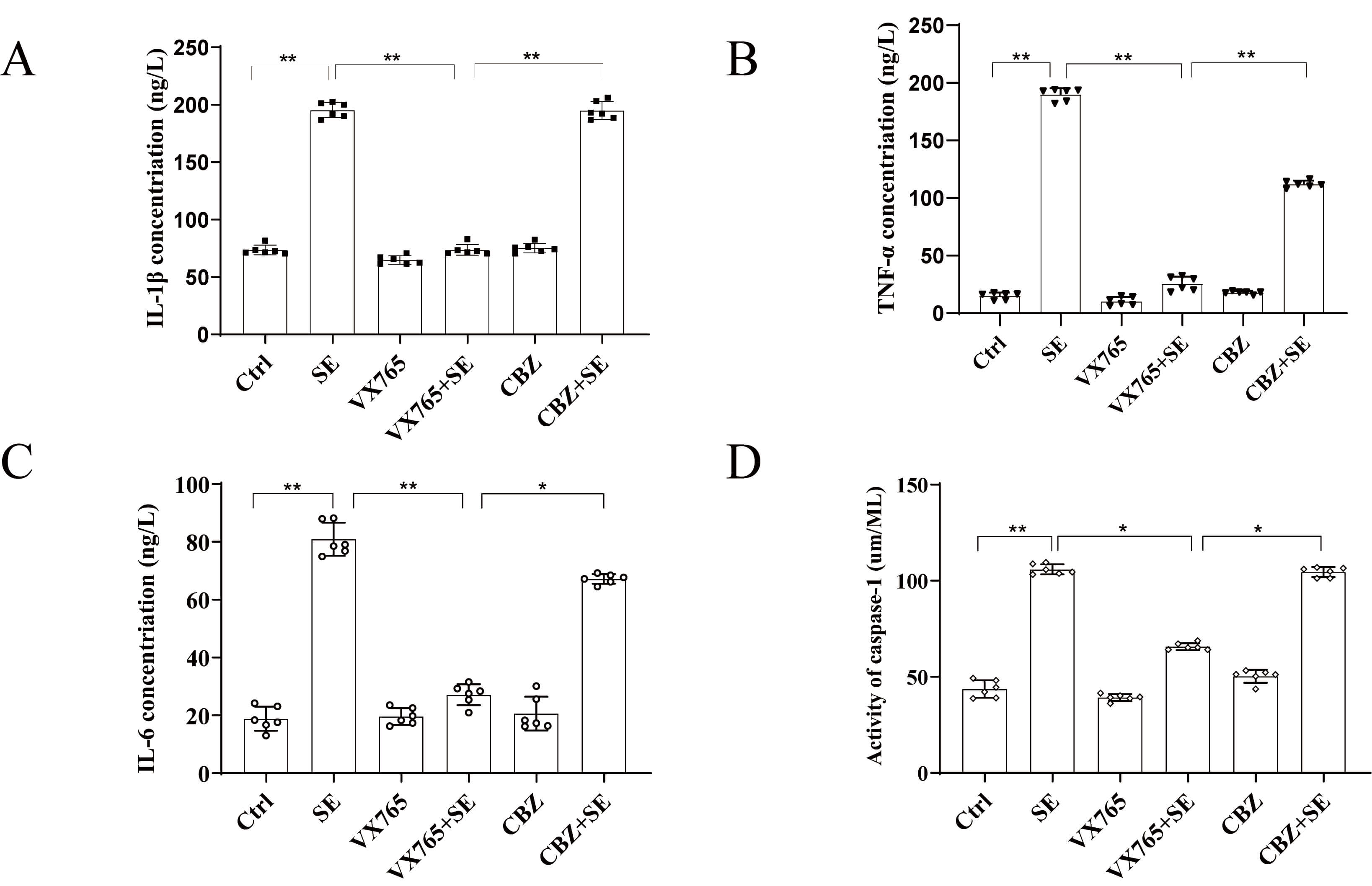 Fig. 4.
Fig. 4.
VX765 reduced neuroinflammation in the hippocampal region of the
epileptic mouse model induced by pilocarpine. (A–C) Contents of IL-1
Astrocytes not only play a role in nutritional support for neurons, but also induce neuroinflammation. The immunohistochemistry results showed that the number of GFAP-labeled positive cells in the SE group was significantly higher than in the Ctrl group, and the number of GFAP-labeled positive cells in the VX765+SE group was significantly lower than in the SE group (Fig. 5A,C). The number of astrocytes in the hippocampus of the Ctrl group mice did not significantly increase, and the cell bodies did not swell. Compared with the Ctrl group, the number of astrocytes in the hippocampal region of the SE group was significantly increased, the cells significantly swelled, and cytoplasmic nuclear glycosome depolymerization was observed. Mitochondria, endoplasmic reticulum, and other organelles also showed swelling to varying degrees (Fig. 5B). The VX765 group was similar to the Ctrl group, with the cells having no obvious change. Compared with the SE group, the extent of astrocyte pyroptosis in the VX765+SE group decreased. Together, these results indicated that VX765 inhibited the hyperactivation of astrocytes.
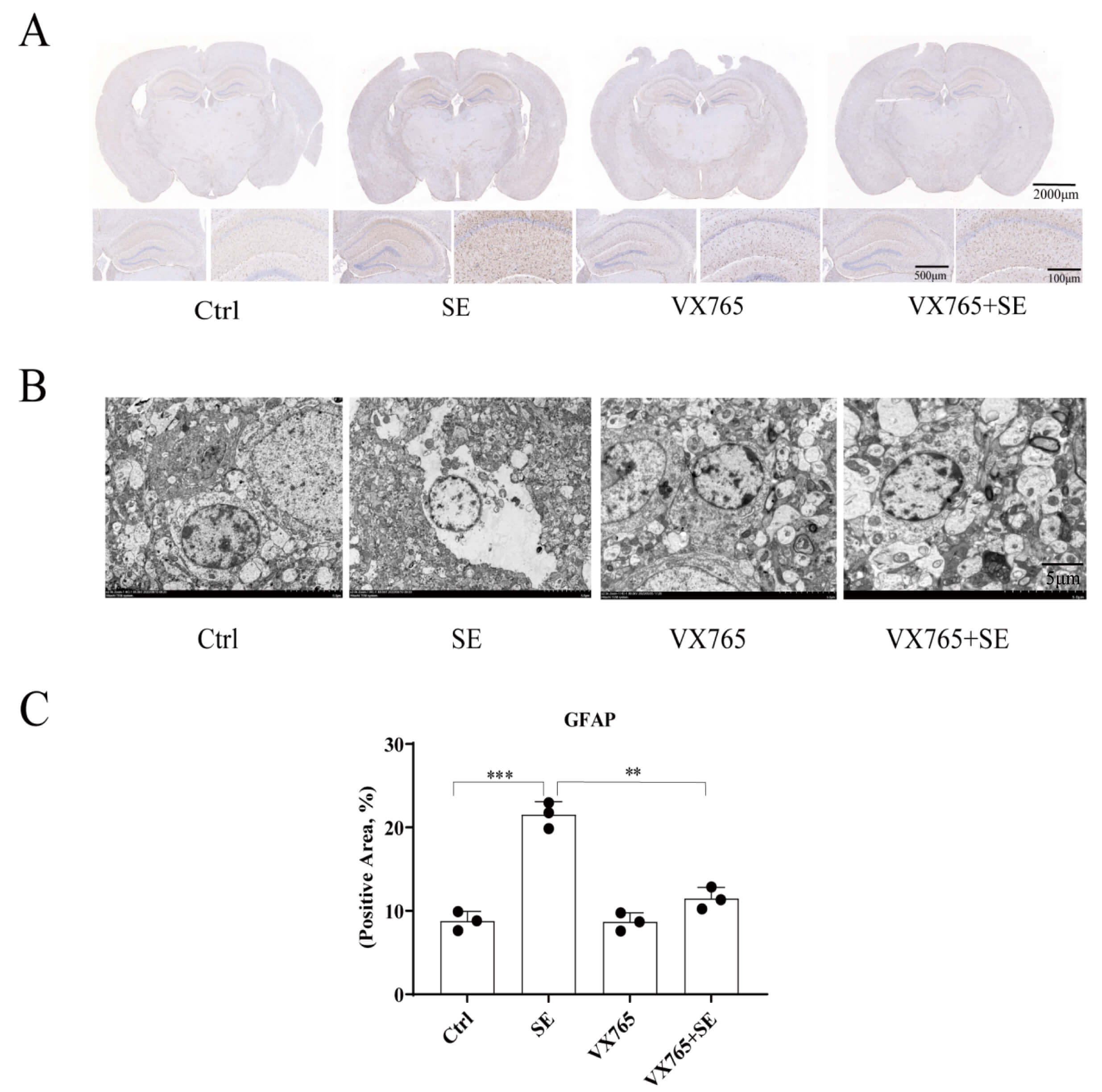 Fig. 5.
Fig. 5.
VX765 reduced activation of astrocytes in the hippocampus of the
epileptic mouse model. (A) Immunohistochemistry images of GFAP-stained positive
cells. The scale bars are 2000 µm, 500 µm, and 100 µm,
respectively. (B) Transmission electron microscopy images of astrocytes. The
scale bars are 5 µm. (C) Histogram of the number of GFAP positive cells (n
= 3/group). **p
As the first immune defense line of the nervous system, microglia mediate the secretion of inflammatory factors after proliferation and activation, and play an important role in the development of epilepsy. The results showed that compared with the Ctrl group, the number of IBA1-labeled positive cells was significantly increased in the SE group, but not in the VX765 group. Compared with the SE group, the number of positive IBA1 markers in the VX765+SE group decreased (Fig. 6A,C). The results of electron microscopy showed that compared with the Ctrl group, microglia in the SE group swelled, surrounded the injured neurons, and there were a large number of lipid droplet vacuoles in the activated microglia, which could engulf the damaged apoptotic neurons. Compared with the SE group, the activated microglia in the VX765+SE group were significantly reduced (Fig. 6B). These results indicated that VX765 treatment inhibited the hyperactivation of microglia.
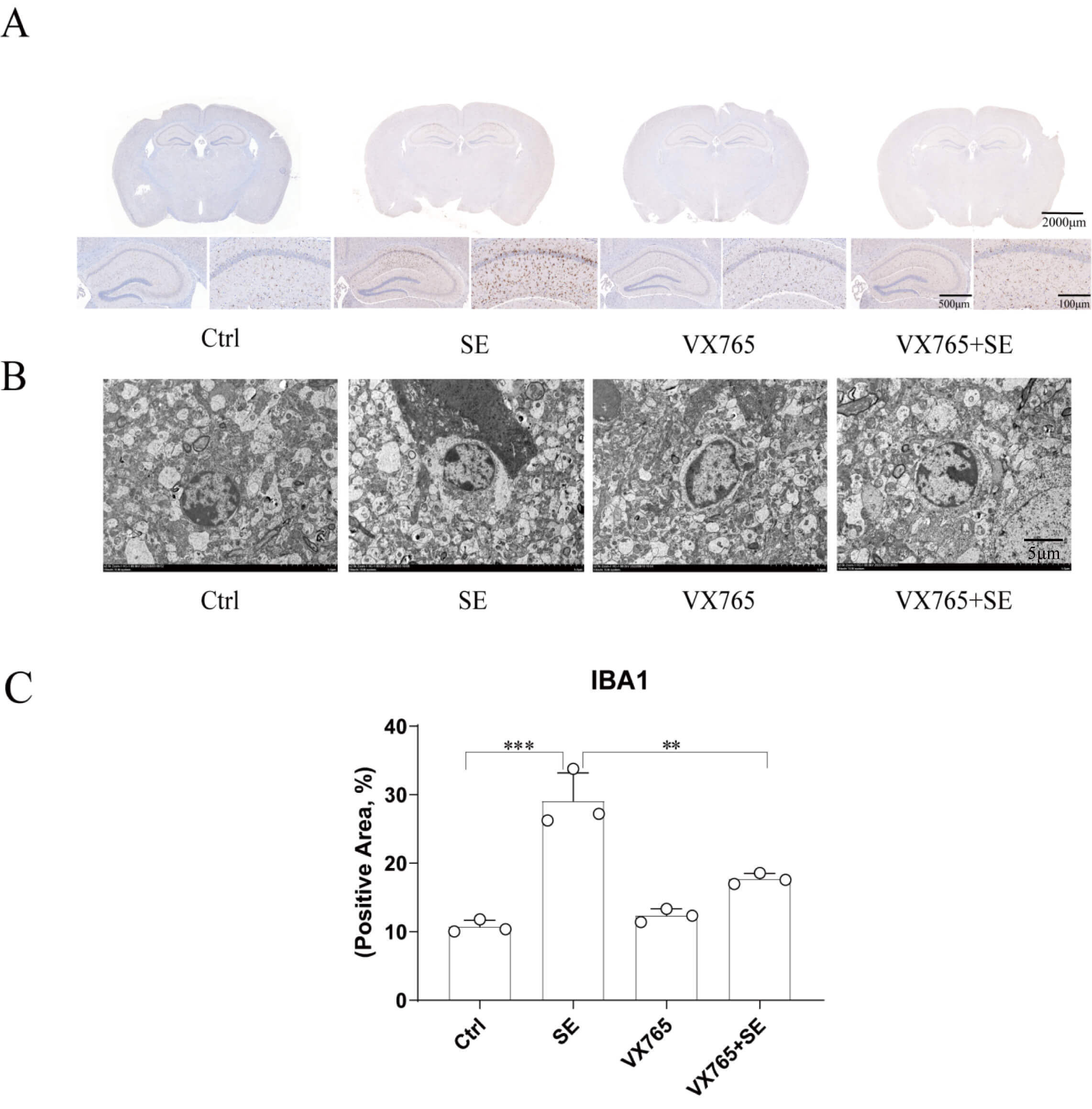 Fig. 6.
Fig. 6.
VX765 reduces the activation of microglia in the hippocampus of
the epileptic mouse model. (A) Immunohistochemical image of IBA1-positive cells.
The scale bars are 2000 µm, 500 µm, and 100 µm, respectively.
(B) Transmission electron microscopy images of microglia. The scale bars are 5
µm. (C) Histogram of IBA1-positive cells (n = 3/group). **p
Recurrent seizures are accompanied by abnormal firing and reduction of neurons. The results showed that compared with the Ctrl group, neuronal cells in the hippocampus of the SE group were significantly lost, while there was no significant change in the VX765 group, when compared with the Ctrl group. Compared with the SE group, the neuron loss in the VX765+SE group was significantly reduced (Fig. 7A,C). The results of electron microscopy showed that compared with the Ctrl group, neurons in SE group were pyknotic, the electron densities of cytoplasm and processes increased, and the chromatin in the nucleus was highly condensed, indicating that neuronal cells underwent apoptosis. There was no significant change in neurons in the VX765 group. The apoptosis of neurons in the VX765+SE group was significantly lower than that in the SE group (Fig. 7B). Together, these results indicated that VX765 had a neuroprotective effect.
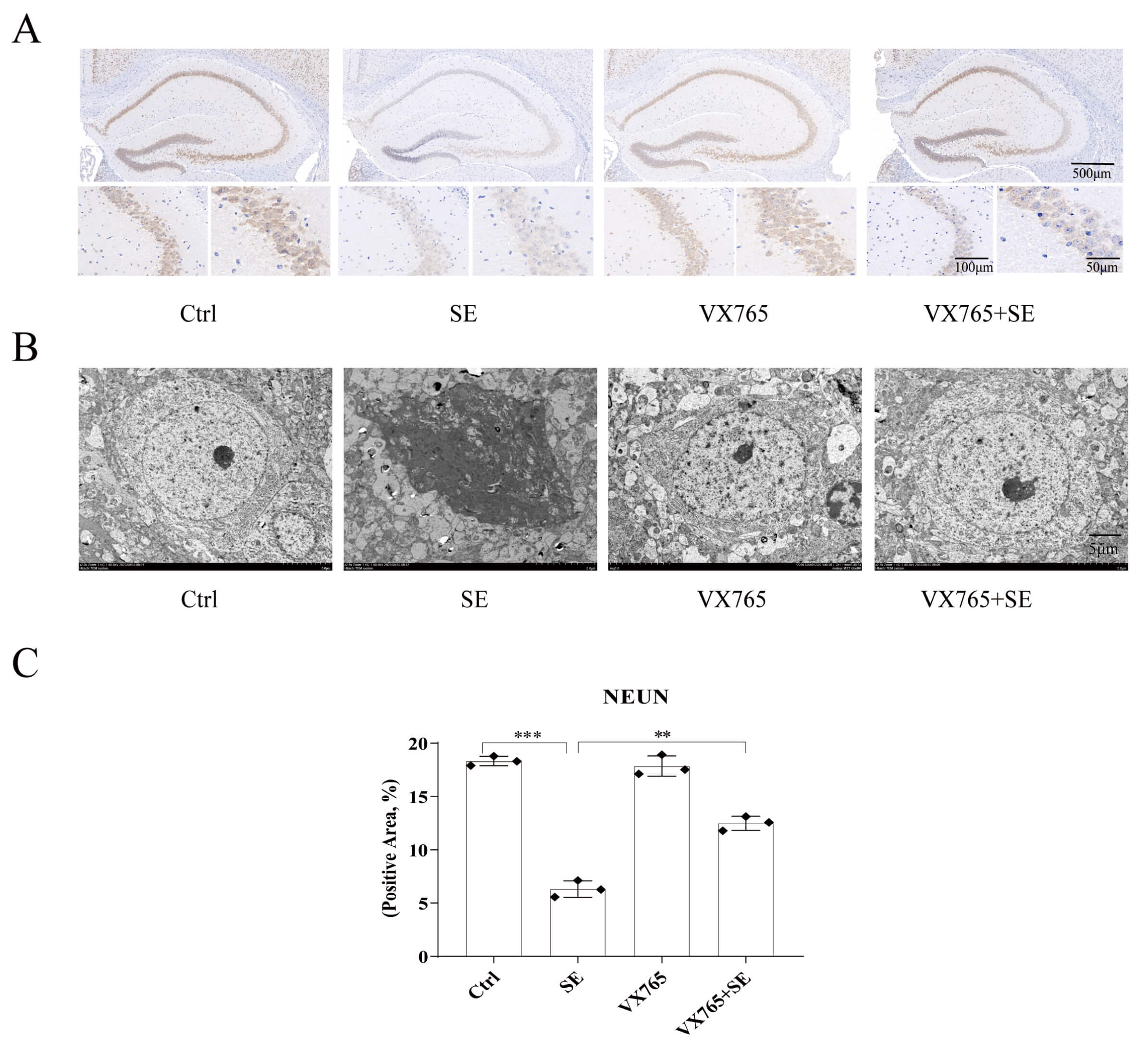 Fig. 7.
Fig. 7.
VX765 reduces damage of neurons in the hippocampus of the
epileptic mouse model. (A) Immunohistochemical image of NEUN staining of
positive cells. The scale bars are 500 µm, 100 µm, and 50 µm,
respectively. (B) Transmission electron microscopy images of neurons. The scale
bars are 5 µm. (C) Histogram of NEUN positive cells (n = 3/group).
**p
To determine the effect of VX765 on glial hyperactivation, the supernatant of the hippocampal tissue homogenate was used to extract proteins, and the expressions of caspase-1 and Gasdermin D N-terminal (GSDMD-N)were detected. The results showed that compared with the Ctrl group, the expressions of caspase-1 and GSDMD-N were significantly increased in the SE group, while there was no significant change in the VX765 group, when compared with the Ctrl group. The expressions of caspase-1 and GSDMD-N in the VX765+SE group were significantly lower than those in the SE group (Fig. 8A,B; The original western blotting images can be found in the Supplementary Materials). These results further indicate that VX765 can alleviate the excessive activation of nerve cells in the hippocampal region of the SE mice.
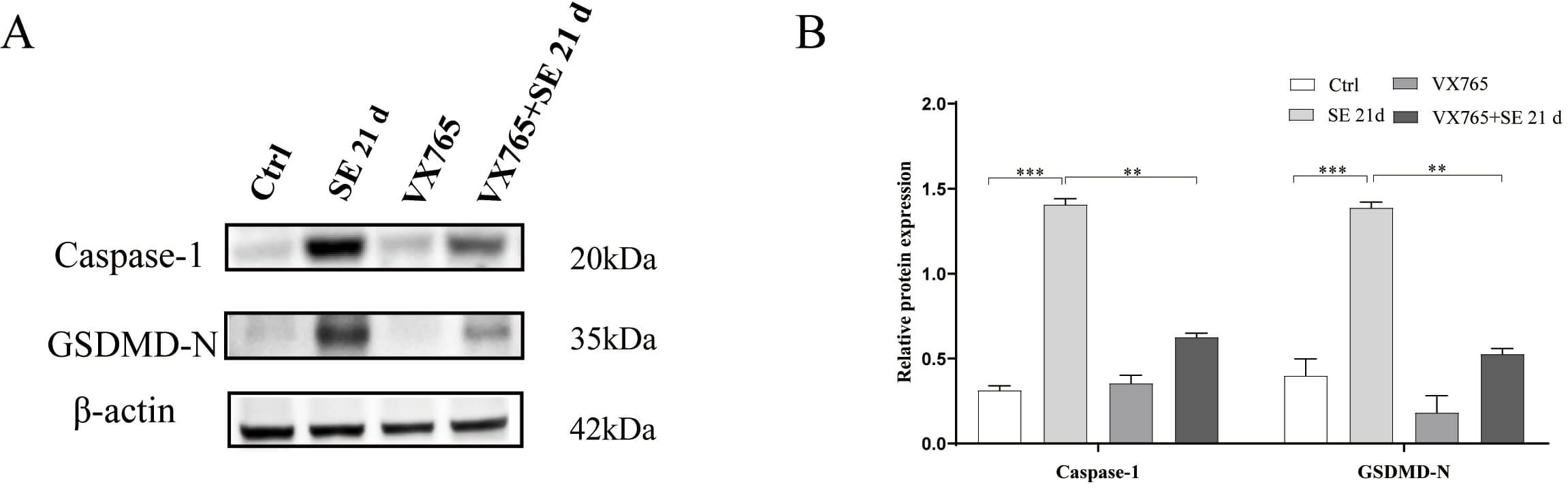 Fig. 8.
Fig. 8.
VX765 reduces the expressions of caspase-1 and GSDMD-N, in the
hippocampus of the epileptic mouse model. (A) Expression bands (western
blotting) of caspase-1 and GSDMD-N proteins in the hippocampus. (B) Histogram of
caspase-1 and GSDMD-N protein expressions. **p
To further characterize its mechanism, we conducted cell experiments involving cultured primary glia and hippocampal neuronal cells (HT22), and verified that VX765 exerted neuronal protection by inhibiting glial hyperactivation. The glia-neuron co-culture model was prepared according to Fig. 9A. Compared to the Ctrl group, the apoptosis of HT22 cells in the glial conditioned medium (CMG) group increased significantly, and the apoptosis of HT22 in the VX765+CMG group was significantly decreased compared with the CMG group (Fig. 9B,C). The CCK-8 results showed that when HT22 was stimulated with LPS, no neuronal death occurred, and there was no statistical difference among the four groups. When HT22 was stimulated with the CMG, compared with the Ctrl group, the survival of HT22 in the CMG group was significantly reduced, and the survival of HT22 in the CMG+VX765 group was significantly improved, when compared with that of the CMG group (Fig. 9D,E). Western blotting results showed that when compared with the Ctrl group, Bax expression in the CMG group increased significantly, whereas Bcl-2 expression decreased (Fig. 9F,G; The original western blotting images can be found in the Supplementary Materials). Together, the results showed that LPS induced glial cells to secrete an inflammatory factor rich supernatant, which may have induced neuronal apoptosis. VX765 may have protected neurons by regulating glial hyperactivation, reducing the secretion of inflammatory factors, and inhibiting neuronal apoptosis (Fig. 10).
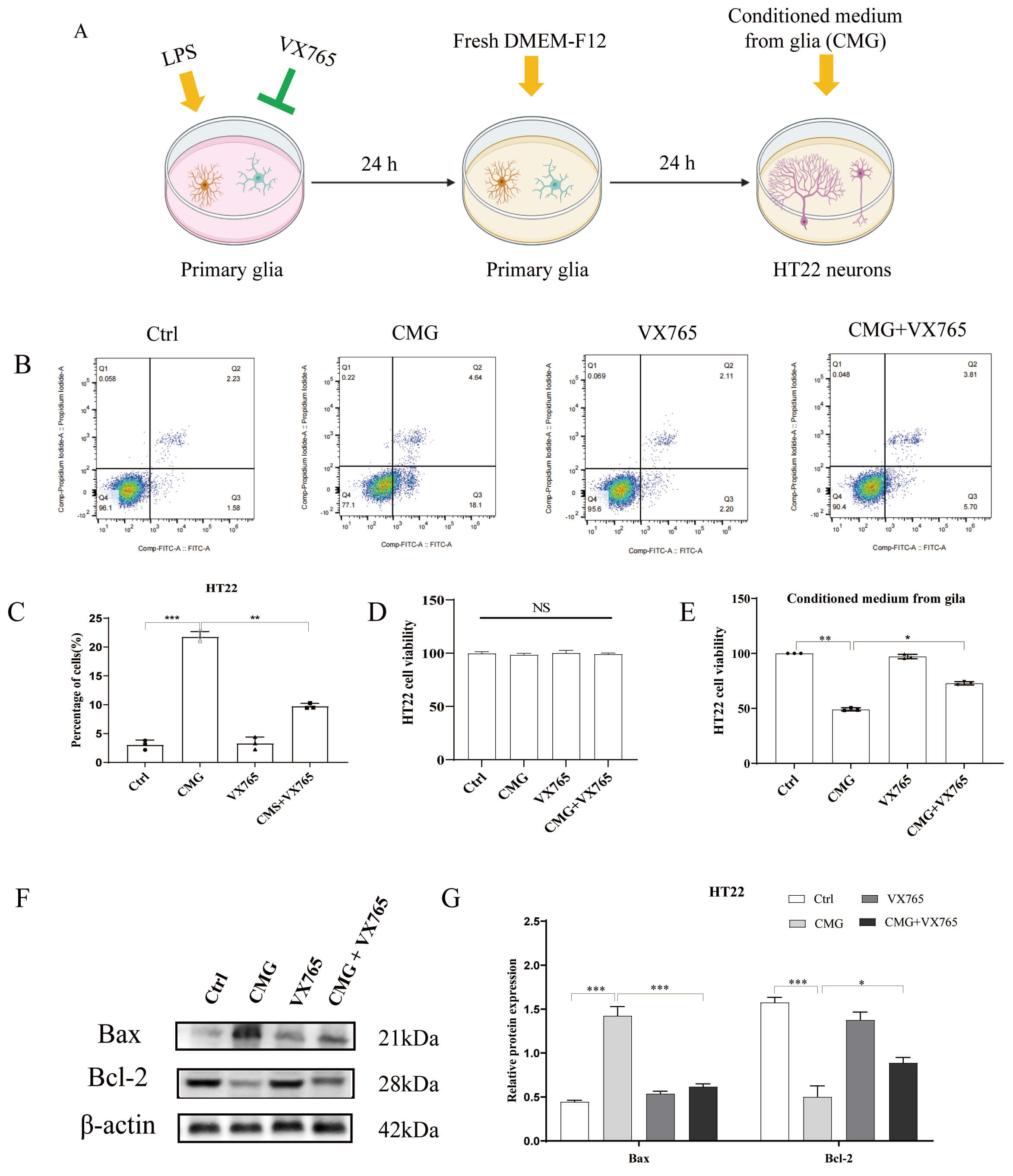 Fig. 9.
Fig. 9.
VX765 improves the survival of HT22 cells. (A) Methodology of
the cell experimental model. The yellow arrows indicate the addition of the above
mentioned drugs or culture medium to the culture dish, while the green arrow
represents the addition of VX765 to the culture dish to inhibit caspase-1,
thereby suppressing the secretion of inflammatory factors. (B) Flow cytometry
results of apoptosis of HT22 cells. (C) Histogram of the apoptosis of HT22 cells.
(D,E) CCK-8 cell proliferation assay results of HT22 cells. NS means Normal
Saline (0.9% NaCl). (F) Expressions of the apoptosis-related proteins. (G)
Quantification plot of the protein results. *p
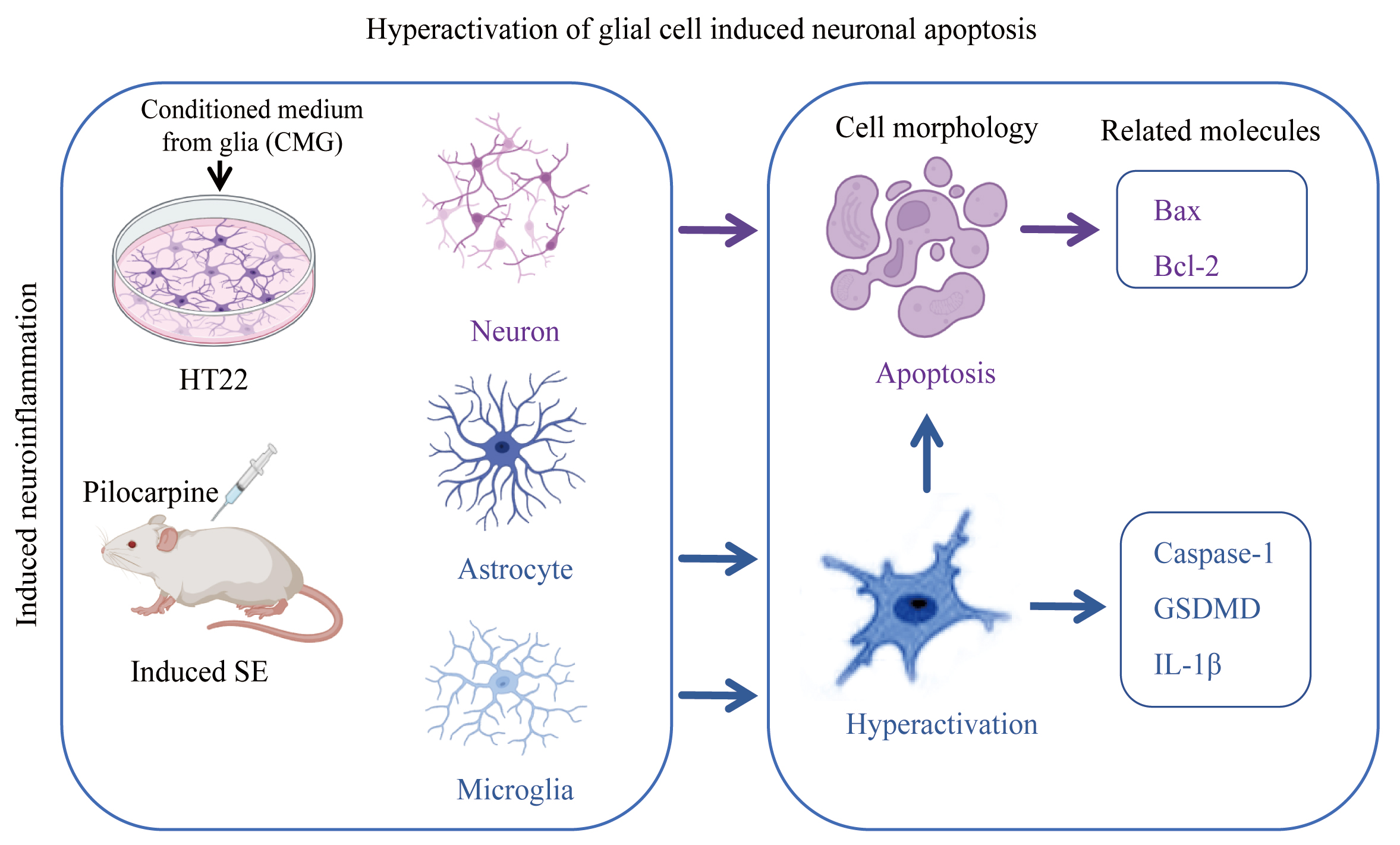 Fig. 10.
Fig. 10.
Pilocarpine induced the frequency of epilepsy in mice, promoted the excessive release of glial inflammatory factors, and resulted in neuronal apoptosis. The blue arrows indicate the effects of glial cells, and the purple arrows indicate the effects of neurons. Bax, Bcl-2-associated X protein; Bcl-2, B-cell lymphoma 2.
The main mechanisms by which glial cells promote the development of seizures and
epilepsy include increased excitability and inflammation. Uncontrolled
glial-mediated immune responses can cause sustained inflammatory changes and
promote epileptogenesis [19]. The inflammatory state after astrocyte changes may
be the main cause of mesial temporal lobe epilepsy with sclerosis, which is
usually described as upregulation of GFAP [20]. Astrocytes in spontaneously
epileptic mice became activated, accompanied by increased expression of glial
fibrillary acidic protein [21]. This is also consistent with our results,
hyperactivation of the number of microglia and astrocytes increased significantly
and showed pyroptosis features such as organelle swelling, while the number of
neurons decreased significantly and showed cytological features of apoptosis.
Dimethyl fumarate significantly reduced inflammatory factors such as IL-18 and
IL-1
By inhibiting neuroinflammation, increasing evidence shows that VX765 is
beneficial to a variety of neurological diseases. VX765 attenuated the High
Mobility Group Box 1/Toll-like Receptor 4/Nuclear Factor Kappa B
(HMGB1/TLR4/NF-
Glial cells are closely related to a variety of neuronal functions, such as,
activated microglia promote the hyperactivation of astrocytes. However, when
microglia are overactivated, they will release excessive inflammatory factors,
causing neurotoxicity [29]. Activated microglia will secrete IL-1, and the
released IL-1 will further stimulate hyperactivation of microglia, eventually
leading to neuronal damage. IL-1
The pilocarpine-induced epilepsy model exhibited increased inflammatory factors and glial hyperactivation. VX765 treatment reduced glial hyperactivation and neuronal loss, attenuating epileptic progression. Its neuroprotective mechanism may involve inhibiting glial inflammatory factor release and reducing neuronal apoptosis. In vivo, VX765 concurrently suppressed glial activation and neuronal apoptosis. In vitro, VX765 alleviated neuronal apoptosis induced by glial inflammatory factors, suggesting that its neuroprotective effects may be mediated through the regulation of glial activation and inflammatory responses. It should be noted that the in vitro model cannot fully replicate the in vivo microenvironment, providing only preliminary insights. In this study, VX765 was administered prophylactically, which differs from clinical practice where treatment begins after unpredictable seizures. Future studies should evaluate its efficacy when given after epilepsy establishment to better assess translational relevance. Future studies using advanced techniques like co‑culture systems or live imaging could further elucidate the temporal and causal relationships between glial activation and neuronal injury in epilepsy.
The data that support the findings of this study are available from the corresponding author upon reasonable request.
HC finished the animal study and was responsible for the in vitro study, wrote the manuscript. XZ finished the animal study, were responsible for the in vitro study. XX analyzed data and constructed the graphs. ZL analyzed data and revised the manuscript. HL designed the study. YS designed the study, analyzed data and constructed the graphs, revised the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
All animal experimental protocols were reviewed and approved by the Institutional Animal Care and Use Committee of Bengbu Medical University (Approval No. 2021-105). The animal experiments described in this study were conducted in strict accordance with the ARRIVE guidelines and the National Institutes of Health (NIH) Guide for the Care and Use of Laboratory Animals (8th edition, 2011).
Written informed consent was obtained from all participants before their involvement in this study. The consent process was conducted in accordance with the ethical standards of the Declaration of Helsinki and was reviewed and approved by the Medical Research Ethics Committee of Bengbu Medical University (Approval No. 440 of 2025). The consent forms clearly explained the purpose, procedures, potential risks, and benefits of the study, and participants were informed of their right to withdraw at any time without penalty.
Not applicable.
This work was supported by the Natural Science Foundation of China (82104152), Cultivation program for middle and young teachers in universities (YQYB2025016).
The authors declare no conflicts of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/JIN49777.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.










