1 College of Integrated Traditional Chinese and Western Medicine Clinical Medicine, Zhejiang Chinese Medical University, 310051 Hangzhou, Zhejiang, China
2 Department of Neurology, Tongde Hospital of Zhejiang Province, 310012 Hangzhou, Zhejiang, China
3 Department of Internal Medicine of Traditional Chinese Medicine, Tongde Hospital of Zhejiang Province, 310012 Hangzhou, Zhejiang, China
4 Department of Acupuncture, Tongde Hospital of Zhejiang Province, 310012 Hangzhou, Zhejiang, China
5 Department of Radiology, Tongde Hospital of Zhejiang Province, 310012 Hangzhou, Zhejiang, China
6 Department of Radiology, The Second Affiliated Hospital and Yuying Children’s Hospital, Wenzhou Medical University, 325027 Wenzhou, Zhejiang, China
7 Department of Psychiatry, Tongde Hospital of Zhejiang Province, 310012 Hangzhou, Zhejiang, China
Abstract
Although donepezil alleviates Alzheimer’s disease (AD) symptoms by raising acetylcholine levels, its impact on cholinergic pathways remains unclear. In this longitudinal, resting-state functional magnetic resonance imaging (rs-fMRI) study, we investigated donepezil-induced changes in cholinergic pathway networks in AD.
AD patients and healthy controls (HCs) were enrolled. AD patients received 24 weeks of donepezil treatment. Cognitive and emotional symptoms were assessed using the Mini-Mental State Examination (MMSE), Cornell Scale for Depression in Dementia (CSDD), and Neuropsychiatric Inventory (NPI) pre- and post-treatment. rs-fMRI was used to examine basal forebrain (BF) functional connectivity.
Sixteen AD patients and 16 HCs completed the study. Post-treatment MMSE scores improved, and NPI and CSDD scores decreased. Reduced BF functional connectivity in the left cerebellar lobule VI, post-treatment, was revealed by rs-fMRI. Compared with HCs, post-treatment AD patients showed lower BF functional connectivity in the right postcentral gyrus (PoG); pre-treatment patients exhibited higher BF functional connectivity in the left cerebellar lobule VI. Right PoG functional connectivity was negatively correlated with disease duration pre-treatment and positively correlated with MMSE post-treatment.
Donepezil improved clinical symptoms in AD by modulating the BF–PoG cholinergic pathway.
Keywords
- Alzheimer’s disease
- cholinergic
- donepezil
- fMRI
- functional connectivity
The cholinergic hypothesis suggests that the progressive degeneration of cholinergic neurons in the basal forebrain (BF) of Alzheimer’s disease (AD) patients is responsible for memory loss, disorientation, and behavioral and personality changes [1]. Donepezil, a cholinesterase inhibitor, increases acetylcholine levels and improves cognition in AD, but its precise effects on BF pathways remain unclear [2].
Insights into donepezil’s therapeutic mechanisms in Alzheimer’s disease have been gained through functional magnetic resonance imaging (fMRI) [3, 4, 5, 6]. After 12 weeks of treatment, Goveas et al. [3] found that cognitive improvements in mild AD patients were linked to specific patterns of hippocampal functional connectivity. They found that the inferior frontal gyrus, dorsolateral prefrontal cortex, and left parahippocampal gyrus were the key regions underlying cognitive improvement. Cheng et al. [4] also found that AD patients had lower regional homogeneity in the right gyrus rectus, right precentral gyrus, and left superior temporal gyrus after 24 weeks of donepezil treatment. Zheng et al. [5] reported that after 6 months of donepezil treatment, AD patients showed improved cognitive and non-cognitive symptoms, which were associated with the increased amplitude of low-frequency fluctuations in cerebellar and frontostriatal regions. White-matter imaging also showed enhanced connectivity in the nucleus accumbens shell and anterior limb of the internal capsule, correlating with acetylcholinesterase inhibitors (AChEIs) exposure [6].
Although those studies have provided valuable insights into the effects of donepezil on distributed brain networks, they have not specifically targeted the basal forebrain cholinergic system—the primary site of cholinergic neuron degeneration in AD and the direct pharmacological target of cholinesterase inhibitors. This gap is significant because understanding how donepezil modulates the functional architecture of its primary target system would provide more direct evidence of its therapeutic mechanism.
A key cholinergic source is the nucleus basalis of Meynert (NBM) in the BF, the atrophy of which has been shown to be correlated with AD severity [7, 8, 9]. Grothe et al. [8] found cholinergic BF atrophy rates were higher than ageing-related global brain shrinkage rates in AD patients. Schumacher et al. [9] reported higher mean diffusivity in NBM-originating white-matter tracts; this was linked to increased dementia risk and global cognitive impairment. Those structural findings suggested that the NBM and its projection network are critically compromised in AD, raising the question of whether pharmacological intervention with donepezil can modulate the functional integrity of this system. However, to date, the specific impact of donepezil treatment on functional connectivity within NBM-centered cholinergic networks remains poorly understood.
The present longitudinal, resting-state fMRI (rs-fMRI) study investigated donepezil-induced changes in cholinergic pathway networks in AD. Using BF subregions as regions of interest, we tested the hypothesis that donepezil treatment modulates functional connectivity within BF-centered networks. Specifically, based on previous evidence [3, 4, 5, 6, 7, 8, 9], we predicted increased functional connectivity between the NBM and prefrontal/cingulate regions, and that these changes were correlated with cognitive improvements.
Sixteen AD patients were recruited from Tongde Hospital of Zhejiang Province.
Patients aged 65–80 years met the criteria for probable AD according to National
Institute on Aging-Alzheimer’s Association guidelines [10] and were in the mild
stage (Clinical Dementia Rating [CDR] = 1, Mini-Mental State Examination [MMSE]
Patients were assessed using the MMSE, the
Neuropsychiatric Inventory (NPI) [11], and the Cornell Scale for Depression in
Dementia (CSDD) [12] at baseline and after 24 weeks of donepezil treatment.
Depression was indicated by CSDD
MRI scans were performed on a 3T scanner (MAGNETOM Verio, Siemens Healthcare,
Erlangen, Germany). Resting-state functional images were acquired using a
gradient echo-planar imaging sequence (repetition time (TR)/echo time (TE) =
2000/30 ms; slice thickness = 4.8 mm; field of view = 200
Data preprocessing was conducted using SPM12 (Wellcome Centre for Human
Neuroimaging, University College London, London, UK) and DPABI 7.2 (Institute of
Psychology, Chinese Academy of Sciences, Beijing, China). After discarding the
first 10 volumes, functional images underwent slice-timing and motion correction
(all participants: mean framewise displacement (FD)
Functional connectivity analysis was performed using four BF seeds per participant, defined according to the Julich-Brain Atlas [15] (Fig. 1, Ref. [16]): bilateral cholinergic cell group (Ch)123 (medial septum/diagonal bands) and bilateral Ch4 (NBM). Whole-brain connectivity maps were generated for each seed using Pearson correlation, followed by Fisher’s r-to-z transformation to z scores for normalization.
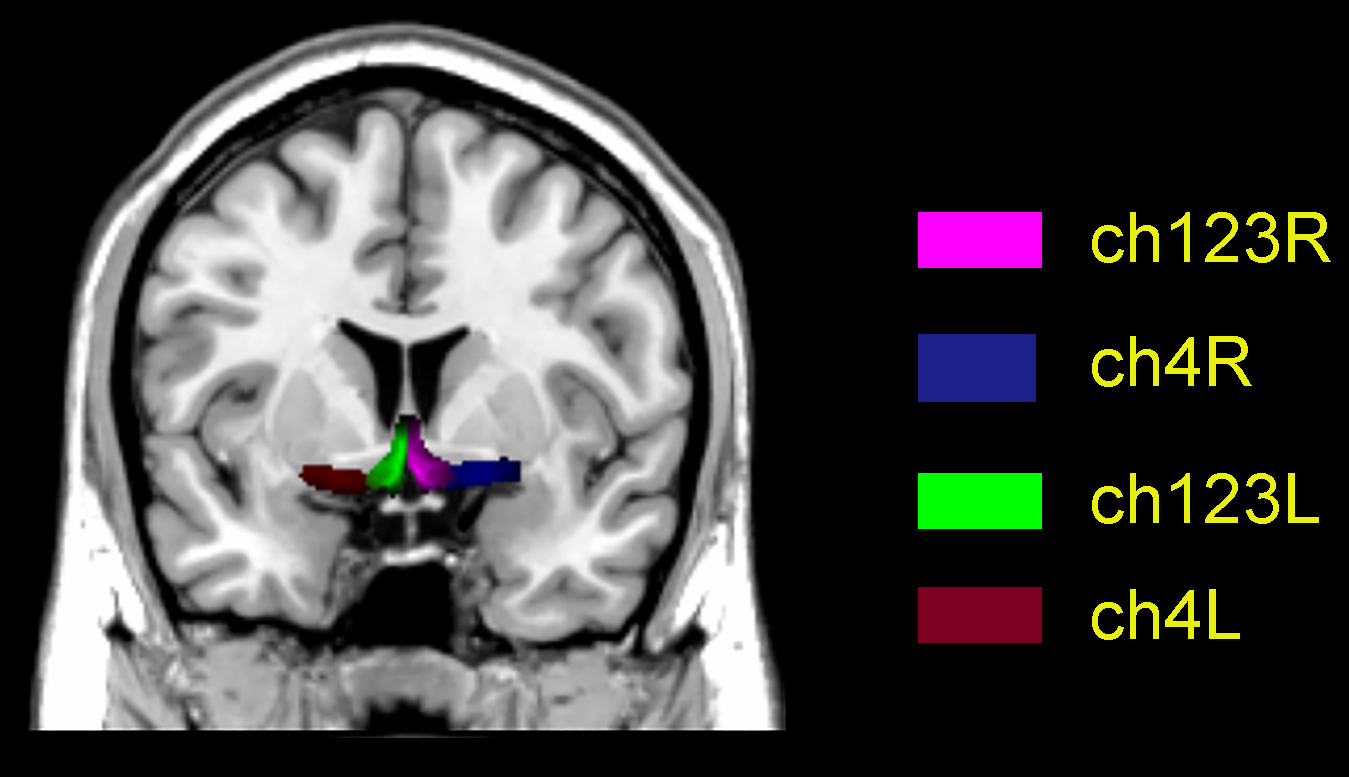 Fig. 1.
Fig. 1.
Basal forebrain subregion map on a Montreal Neurological Institute (MNI) template. Ch123L/R denote left/right medial septum/diagonal bands; Ch4L/R denote left/right nucleus basalis of Meynert. Ch, cholinergic cell group. [16].
To examine the effects of whole brain volume on functional connectivity (FC) analyses, T1-weighted images were analyzed with the voxel-based morphometry (VBM8, version r435, http://dbm.neuro.uni-jena.de/) toolbox in SPM8 (released April 2009, http://www.fil.ion.ucl.ac.uk/spm). T1-weighted images were normalized to MNI space using Diffeomorphic Anatomical Registration through Exponentiated Lie Algebra for each subject. The resulting images were then segmented into gray matter (GM), WM and cerebrospinal fluid (CSF). Allowing for individual differences in brain size, the segmented images were modulated using non-linear deformation. Individual GM volume (GMV) of the whole brain was calculated.
Demographic and clinical data were analyzed using SPSS 15.0 (IBM Corp., Armonk,
NY, USA). Age and education were compared with the Mann-Whitney U test, and sex
was compared with the Chi-square test. For functional connectivity analysis,
voxelwise one-way analysis of covariance (ANCOVA) was performed on BF
functional connectivity maps across the Post-Treatment AD, Pre-Treatment AD, and
HCs, controlling for mean head motion, age, sex and GM maps. Significant clusters
(voxel-level p
Mean BF functional connectivity values were extracted from significant clusters
and correlated with clinical variables in AD patients pre- and post-treatment
using Pearson correlation (p
A total of 32 participants (16 HCs, 16 AD patients) completed the study. No
significant differences were observed in age, sex, or education between groups
(p
After the 24-week treatment, the AD group’s MMSE scores had increased
significantly (t = 2.423, p
| AD group | HCs group | p | ||
| Sex, n (M/F) | 16 (8/8) | 16 (7/9) | 2 | 0.157 |
| Age, years | 65.20 (8.10) | 69.10 (4.50) | 1.49 | 0.135 |
| Education, years | 9.10 (2.00) | 8.30 (2.10) | −0.50 | 0.613 |
| MMSE | 19.70 (2.60)* | 28.31 (0.95) | −14.15 | |
| MMSE (24 weeks) | 20.10 (2.50)* | 28.56 (0.63) | −13.78 | |
| NPI | 4.56 (2.90)** | 0.15 (0.36) | 13.45 | |
| NPI (24 weeks) | 1.38 (1.20)** | 0.06 (0.25) | 4.39 | |
| CSDD | 3.25 (2.40)*** | 1.56 (0.81) | 2.80 | 0.014 |
| CSDD (24 weeks) | 0.75 (0.71)*** | 0.31 (0.48) | 1.70 | 0.110 |
Data are presented as means (standard deviations). The Chi-square test was used
to compare sex, and two-sample t-tests were used to compare age and
neuropsychological data. * Comparison of MMSE score post- vs. pre-treatment
(t = 2.423, p
Post-treatment, AD patients displayed decreased BF functional connectivity in the left cerebellar lobule VI. When compared to HCs, post-treatment AD patients showed reduced BF functional connectivity in the right postcentral gyrus (PoG). Furthermore, pre-treatment AD patients exhibited elevated BF functional connectivity in the left cerebellar lobule VI than did HCs (Table 2).
| Brain regions | Voxels | BA | MNI coordinates | F/T | p | |||
| x | y | z | ||||||
| ANCOVA | ||||||||
| Cerebellar_VI_L | 25 | 37 | −30 | −57 | 27 | 11.93 | ||
| Postcentral_R | 51 | 5 | 12 | −54 | 75 | 15.16 | ||
| t2 vs. t1 | ||||||||
| Cerebellar_VI_L | 27 | 37 | −42 | −57 | −33 | −4.63 | ||
| t1 vs. HCs | ||||||||
| Cerebellar_VI_L | 33 | 37 | −30 | −60 | −24 | 4.22 | ||
| t2 vs. HCs | ||||||||
| Postcentral_R | 186 | 5 | 12 | −54 | 75 | −6.53 | ||
ANCOVA, analysis of covariance; BA, brodmann area; t1, pre-treatment; t2, post-treatment.
A significant negative correlation was found between right PoG functional connectivity values and disease duration in pre-treatment AD patients (r = –0.64, p = 0.008). Conversely, in post-treatment AD patients, right PoG functional connectivity values showed a significant positive correlation with MMSE scores (r = 0.60, p = 0.014).
Using rs-fMRI and functional connectivity analysis, this study examined changes in BF networks in AD patients pre- vs. post-treatment with donepezil. Compared to baseline, post-treatment patients showed decreased BF functional connectivity in the left cerebellar lobule VI. Relative to HCs, treated patients exhibited lower BF functional connectivity in the right PoG and higher functional connectivity in the left cerebellum.
Studies have consistently implicated the PoG in AD. Diffusion imaging showed
altered microstructure in the PoG, precentral gyrus, and superior temporal gyrus
of AD patients [17]. Non-memory agnosia in AD has been correlated with
episodic/semantic memory and gray matter volumes in regions including the
anterior cingulate cortex, precentral gyrus, superior frontal gyrus, PoG, and
lingual gyrus [18]. Network analyses have further revealed centrality loss in the
precuneus, precentral gyrus, and PoG during AD progression [19]. Longitudinal
studies have shown higher
Meta-analyses confirmed significant cerebellar atrophy in AD patients, which was
correlated with cognitive decline [24]. Notably, early-onset AD patients
exhibited substantial amyloid-beta (A
The observed functional connectivity changes between the NBM and the cerebellum/PoG in the present study warrant discussion. This was initially unexpected because basal forebrain cholinergic neurons do not provide direct monosynaptic input to these regions. However, we should clarify that functional connectivity is not the same as direct anatomical connectivity. Functional connectivity reflects statistical dependencies that can arise through polysynaptic pathways, intermediary regions, or common modulatory inputs. This perspective is supported by growing evidence implicating the cerebellum in cognition—including attention, executive function, and working memory—beyond its traditional motor role [28]. The connections of the cerebellum with associative cortical regions provide a substrate for its cognitive contributions. Thus, altered NBM-cerebellar functional connectivity may have reflected modulation of cerebello-cerebral circuits via indirect pathways (e.g., brainstem, thalamus, or cholinergically-innervated cortical regions). Similarly, PoG connectivity changes were likely to involve multi-synaptic mechanisms. Anatomically, cholinergic input to the sensory cortex originates from the NBM, whereas cerebellar cholinergic afferents arise from the pedunculopontine tegmental nucleus (PPN), a key regulator of movement, gait, and postural control [29]. The PPN may therefore serve as a functional interface linking cerebellar motor circuits with basal forebrain cognitive networks [30]. Notably, gait and balance impairments are recognized as early manifestations of AD and correlate with cognitive decline [31, 32]. Donepezil has been demonstrated to improve gait in AD patients [33]. Our findings suggested that donepezil alleviated AD symptoms, in part, by modulating cholinergic pathways involved in motor integration. The observed connectivity changes involving the cerebellum and PoG may have reflected enhanced sensorimotor integration, a process fundamental to both motor function and cognition. The mirror neuron system (MNS), which includes the PoG and receives cerebellar modulation via inhibitory interneurons, is central to this integration, enabling the updating of internal sensorimotor models based on sensory feedback [34, 35]. Cholinergic modulation influences MNS activity [35], and our findings of increased NBM-cerebellum and altered PoG connectivity may have represented enhanced cholinergic drive to these circuits. This interpretation aligns with evidence that donepezil improves gait in AD patients [33], a function heavily dependent on intact sensorimotor integration, and suggests that treatment benefits may extend beyond cognition to the real-time calibration of action-perception loops. Collectively, these findings indicated the distributed effects of cholinergic enhancement beyond direct projection targets. Previous literature is limited, with only one study reporting NBM-cerebellum connectivity in healthy controls [34]. Future studies using effective connectivity methods (e.g., dynamic causal modeling) or molecular imaging could help clarify the pathways underlying these network-level changes and test the hypothesis of PPN-mediated connectivity.
The present study had several limitations. First, the sample size in this study was relatively small, which represented a key limitation. The modest sample size increased the risk of type II errors (missing true effects) and potential overestimation of observed effects, and limited the generalizability of our findings. Second, this study did not include biomarker confirmation for AD, such as CSF biomarkers or PET imaging due to hospital condition limitations and the difficulty in obtaining cerebrospinal fluid samples, we would add plasma biomarkers to future sample collection. Third, we acknowledge a methodological limitation concerning the duration of our rs-fMRI acquisition. Each scan lasted 6 min, which, although consistent with many clinical studies involving AD patients, was shorter than the durations recommended for optimal test-retest reliability of functional connectivity estimates. Our 6-min protocol was necessitated by practical considerations related to patient tolerability and motion control in an elderly AD cohort. Nonetheless, we recognize that this may have increased variability in our connectivity estimates. Future studies should incorporate longer scan durations or multi-session acquisitions to enhance reliability, particularly if individual-level analyses are pursued. Fourth, the clinical symptoms are mainly assessed using the MMSE, but without evaluations of olfaction, sleep, and other aspects, it cannot fully reflect the efficacy of the drug and its connection to brain neural mechanisms.
It is worth noting that AD progression and treatment responses vary considerably among individuals. Longitudinal studies are essential to determine whether the observed connectivity improvements persist over time and how they relate to cognitive outcomes. Also, AD is a heterogeneous disease due to the presence of different genetic phenotypes, which likely contributed to the variable therapeutic responses. Future investigations should therefore stratify patients based on genetic markers (e.g., apolipoprotein E (APOE) genotype), phenotypic characteristics, and clinical variables, to better understand the determinants of treatment response and to facilitate the development of more personalized therapeutic approaches.
This exploratory study based on real-world clinical data has uncovered the neural mechanisms of donepezil intervention in Alzheimer’s disease, which to some extent deepens our understanding of both disease pathophysiology and therapeutic effects. These findings also provide a direction for future research. However, further studies with more rigorous designs—including randomized controlled trials, biomarker-based patient stratification, and multimodal neuroimaging—are still needed to validate and extend these findings. In particular, given recent evidence suggesting that the cerebellum may serve as a synergistic therapeutic target in other neurodegenerative conditions such as frontotemporal dementia (FTD) [36], future studies could explore whether combining cholinergic therapy with cerebellar neuromodulation yields enhanced clinical benefits in AD.
In the present study, we compared BF functional connectivity among AD patients at baseline and after donepezil treatment, as well as with HCs. The results revealed intervention-related functional alterations in the right PoG and left cerebellar lobule VI. BF–PoG connectivity may serve as a potential neuroimaging marker for evaluating the therapeutic effect of donepezil treatment, though its utility as a specific treatment target requires further validation.
The datasets used and analysed during the current study are available from the corresponding author upon reasonable request.
YG and FW contributed to the acquisition of the data and drafted the manuscript. ZZ, HL, and BC collected the data, XL and ZG conceived, analyzed the data and revised the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This study was performed in accordance with the principles of the Declaration of Helsinki. Approval was granted by the ethics committee of Tongde Hospital of Zhejiang Province (approval no. 2017-11-12). Written informed consent was obtained from all participants.
We thank Sarina Iwabuchi, PhD, from Liwen Bianji (Edanz) for editing the language of a draft of this manuscript.
This research was supported by the General Project of the Department of Science and Technology of Zhejiang Province (2017KY109 and 2020358406 to XL), the General Project of the Department of Science and Technology of Zhejiang Province (2018KY031 and 2024KY873 to ZG), and the National Leading Medical Specialty Development Project-Department of Geriatrics, Tongde Hospital of Zhejiang Province (Project Number: [2024]90662), and Zhejiang Provincial Alliance of Traditional Chinese Medicine Advantage Specialty for Geriatric Diseases (Project Number: [2024]10).
The authors declare no conflicts of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

