1 Neurological Institute of Jiangxi Province, 330006 Nanchang, Jiangxi, China
2 Department of Neurology, Jiangxi Provincial People’s Hospital, The First Affiliated Hospital of Nanchang Medical College, and Xiangya Hospital of Central South University at Jiangxi, 330006 Nanchang, Jiangxi, China
3 Department of Psychology, Jiangxi Mental Hospital, Nanchang University, 330029 Nanchang, Jiangxi, China
†These authors contributed equally.
Abstract
In idiopathic rapid eye movement (REM) sleep behavior disorder (iRBD) patients, electroencephalogram (EEG) activities during REM sleep in some cortical regions are different from those in normal controls. This study aimed to examine inter-hemispheric asymmetry and intra-hemispheric EEG discrepancy in iRBD patients and normal controls during REM sleep.
Polysomnographic recordings were carried out on 15 iRBD patients and 15 normal controls. The inter-hemispheric asymmetry and intra-hemispheric differences of EEG activities in the iRBD patients were compared with those in the normal controls during REM sleep.
During REM sleep, most of the powers of theta, alpha, sigma, beta, and gamma waves in the right cerebral hemisphere were significantly greater than those in the left cerebral hemisphere in both the iRBD patients and normal controls. The inter-hemispheric asymmetry was significantly larger in the central and occipital regions and generally smaller in the frontal region in the iRBD patients compared with the normal controls. In the iRBD patients and normal controls, the powers of theta, alpha, beta, and gamma bands during tonic and phasic REM sleep were highest in the frontal cortical region, followed by the central cortical region, and lowest in the occipital cortical region; sigma power during phasic, but not tonic, REM sleep fully followed this rule.
In iRBD patients EEG activities are unevenly distributed, with an altered inter-hemispheric asymmetry that might be associated with changed bilateral neuronal differences compared with normal controls.
Keywords
- electroencephalography
- functional laterality
- power spectral analysis
- REM sleep behavior disorder
Rapid eye movement sleep behavior disorder (RBD) is a parasomnia with a loss of muscle atonia accompanied by dream enactment behaviors during rapid eye movement (REM) sleep [1]. RBD predominantly occurs in males, and the onset age in most patients is 40–70 years old [2]. Violent behaviors during dreams range from mild physical convulsions, minor limb jerking and twitching to complex violent movements such as hitting walls and kicking legs, which can cause serious physical injuries to the patients themselves and/or their bedmates during REM sleep [2, 3, 4].
REM sleep can be further divided into phasic and tonic REM phases based on the presence absence of REMs or not and during phasic REM sleep, motor-behavioral episodes in RBD patients happen more frequently than during tonic REM sleep [5].
A notable clinical form of RBD with a relationship to REM sleep is idiopathic RBD (iRBD), which refers to RBD that occurs without any other obviously associated neurological diseases or triggers, such as the use of antidepressants [6]. It has been reported that approximately 80% iRBD patients develop neurodegenerative diseases within 10 years [7]. iRBD is also considered a strong predictor and an early sign for neurodegenerative alpha-synucleinopathies, such as Lewy body dementia (DLB), Parkinson’s disease (PD) and multiple system atrophy [8, 9], although the pathophysiologic mechanisms underlying iRBD are still unclear. iRBD patients might have impairments in memory, executive function, and visuospatial ability [10, 11, 12, 13], and up to 50% of iRBD patients have mild cognitive impairments (MCI) [11].
Compared to normal controls, iRBD patients show a higher metabolism in the premotor cortex and hippocampus and a lower metabolism in the occipital region [14], suggesting that a discrepancy in electroencephalogram (EEG) activities between different cortical regions within the same hemisphere may be exhibited in the iRBD patients. Inter-hemispheric differences in EEG activities in iRBD patients might differ from those in normal controls during REM sleep. Furthermore, the cerebral hemispheres are not structurally or functionally identical in both sides [15, 16, 17]. During REM sleep, for example, EEG activity in the right side is more active than in the left [15, 18]. In addition, it has been reported that the difference in alpha power between the left and right frontal cortical regions (frontal alpha asymmetry, FAA) might predict the level of anger in dream during REM sleep, and that people with a greater alpha power in the right frontal cortical region might be less able to control intense emotional states in dream, such as anger [19]. Since iRBD patients are often reported to have violent dreams, it is possible that iRBD patients might have a greater alpha asymmetry in EEG activities than normal controls. A previous study has shown that the brain networks of iRBD patients during the eyes-open resting state display higher topological properties in the left hemisphere when analyzed at the hemispheric level [20].
Since these various findings suggest both intra-hemispheric and inter-hemispheric EEG differences during REM sleep might be dissimilar between iRBD patients and normal controls, the study aimed to compare inter-hemispheric asymmetry and intra-hemispheric discrepancy in EEG activities during phasic and tonic REM sleep between iRBD patients and normal controls.
A total of 40 subjects, including 20 iRBD patients and 20 normal controls, were recruited and clinical examined at the Jiangxi Provincial People’s Hospital. A total of 5 iRBD patients and 5 controls were eliminated due to failure to meet the inclusion criteria or having conditions in the exclusion criteria (see below). At the end, a total of 30 participants were enrolled in this study, including 15 iRBD patients and 15 normal controls.
The patients were recruited if they were 30–70 years old males diagnosed with
iRBD based on the criteria defined in the International Classification of Sleep
Disorders, Third Edition (ICSD-3) [1], and age-matched male normal controls were
also included. Inclusion criteria included: (1) no insomnia, bruxism, narcolepsy,
restless leg syndrome and any other sleep disorders; (2) no intake of anxiolytic,
antidepressant or psychiatric medications within a month prior to
polysomnographic (PSG) examination. The subjects were not allowed to take
stimulants such as caffeinated beverages (e.g., tea and coffee) within 24 hours
prior to the experiments. The patients and normal controls with one or more the
following conditions were excluded: (1) apnea hypopnea index (AHI)
All iRBD patients were newly diagnosed during this assessment period and had not previously received any treatment for iRBD. The study included four patients with hypertension (Stage I), and three patients with well-controlled type II diabetes mellitus without complications. Patients with type II diabetes mellitus were primarily treated with metformin and none of the patients were taking medications known to significantly affect sleep architecture or EEG activity.
Electroencephalographic (O1-A2, O2-A1, C3-A2, C4-A1, F3-A2, F4-A1), electrooculographic (EOG), and electrocardiographic (ECG) activities, as well as electromyographic (EMG) activities from bilateral mylohyoid, masseter, tibialis anterior and gastrocnemius muscles, as well as flexor and extensor radialis were simultaneously recorded with the Grael 4K PSG:EEG recording system (Compumedics Limited, Abbotsford, Victoria, Australia) as previously reported [21, 22]. In addition, the participants’ respiration was also recorded via a nasal pressure-monitoring cannula, an airflow thermistor, as well as thoracic and abdominal piezoelectric belts. Peripheral vascular oxygen saturation (SpO2) was recorded with a pulse oximetry. In addition, body position was monitored by body position sensors, and snoring was recorded by snore sensors. All participants were monitored with audio-visual recorders during PSG recordings.
All PSG data were initially analyzed using the Pro Fusion PSG 4 Software (Compumedics Limited, Abbotsford, Victoria, Australia) based on the standards developed by the American Academy of Sleep Medicine [23], and then analyzed with the Brainstorm program (Version 2, GNU GPLv2, McGill University, Montreal, QC, Canada). The EEG signals were digitized and filtered with a low- and a high-pass filter set at 70 Hz and 0.1 Hz, respectively [4], and a 50 Hz notch filter.
REM sleep was divided further into phasic and tonic REM phases based on the
presence or absence of REMs, which were described as irregular, sharply peaked
and conjugate eye movements recorded in the EOG derivations, with an initial
deflection lasting shorter than 0.5 seconds [4, 23]. Phasic REM epochs were
defined as a 4-second segment with
Phasic and tonic REM sleep in the iRBD patients were further categorized according to the presence or absence of excessive transient muscle activity (ETMA), which was defined as EMG bursts lasting 0.1–0.5 seconds and with amplitudes at no less than two times of the EMG activities with muscle atonia, or the lowest EMG amplitude during non-rapid eye movement (NREM) sleep if no muscle atonia was present during REM sleep [23]. In the iRBD patients and normal controls, 50 4-second phasic epochs (25 with and 25 without ETMA) and 50 4-second tonic epochs (25 with and 25 without ETMA) were randomly selected per participant. Power spectral analysis of the epochs of EEG signals was carried out using the Welch’s method with 1-second windows and 50% overlap [4].
The ratio of duration of REM sleep without atonia (RSWA) to the total duration of REM sleep. A higher percentage of REM sleep showing increased muscle tone (tonic) or excessive transient activity (phasic) was used to indicate more severe pathology. The correlation of EEG measures with the ratio of RSWA/REM was analyzed.
The absolute and relative power of five specific EEG frequency bands specifically theta (4–8 Hz), alpha (8–12 Hz), sigma (12–15 Hz), and beta (15–30 Hz) [4], as well as gamma (30–48 Hz) were calculated based on linked mastoid reference (A1 + A2)/2. The 30–48 Hz gamma band, rather than the 30–50 Hz range as used in the previous study [4], was analyzed to avoid including alternating current (50 Hz) noise. Delta waves during REM sleep were not analyzed due to possible contamination with EOG signals.
Inter-hemispheric EEG asymmetry was defined as the difference in EEG power of the same waves during REM sleep in the frontal, central and occipital regions between left and right side, and expressed as inter-hemispheric EEG asymmetry score (INTER-EEG-A-Score), which was calculated by using the following formula:
INTER-EEG-A-Score = (Power Right – Power left)/(Power Right + Power left) [25, 26].
In contrast, intra-hemispheric discrepancy referred to the difference in the power of certain EEG waves between any two cortical regions in the same cortical hemisphere.
Sample size was estimated based on the results of the preliminary data from four
iRBD patients and four normal controls with G*Power (version 3.1.9.7,
http://www.gpower.hhu.de/;
Prior to any statistical analysis, normality for all variables was examined with
Shapiro-Wilk tests. Normally distributed data were reported as mean
No significant difference (p
| iRBD patients | Normal controls | p | ||
| Sex | 15 Males | 15 Males | ||
| Age (years) | 40 (35–56) | 42 (38–51) | NS | |
| Total sleep time (min) | 421.33 |
428.30 |
NS | |
| WASO (min) | 70.73 |
37.67 |
||
| Sleep efficiency (%) | 83.07 |
90.47 |
||
| Sleep stage (%) | ||||
| N1 | 12.11 |
9.23 |
NS | |
| N2 | 53.87 |
52.70 |
NS | |
| N3 | 15.80 |
18.52 |
NS | |
| REM | 18.22 |
19.55 |
NS | |
| Sleep stage (min) | ||||
| N1 | 51.17 |
38.47 |
NS | |
| N2 | 227.23 |
225.27 |
NS | |
| N3 | 66.97 |
80.20 |
NS | |
| REM | 75.97 |
84.37 |
NS | |
| Microarousal index (events/h) | 14.40 (11.10–20.80) | 10.70 (6.40–15.50) | NS | |
| SB index (events/h) | 0.80 (0.40–1.90) | 0.80 (0.40–3.40) | NS | |
| PLMI (events/h) | 1.70 (0.00–9.90) | 4.90 (0.50–10.10) | NS | |
| AHI (events/h) | 6.11 |
7.93 |
NS | |
Normally distributed variables are reported as mean
AHI, Apnea hypopnea index; N1–N3, Non-rapid eye movement sleep stage 1–3; NS, not significant; PLMI, Periodic limb movement index; REM, rapid eye movement sleep; SB, sleep bruxism; WASO, wake after sleep onset; iRBD, idiopathic rapid eye movement sleep behavior disorder.
EEG powers of theta, alpha, sigma, beta and gamma waves in the right frontal,
central and occipital regions were significantly greater (p
| Tonic REM sleep | With ETMA in iRBD patients | Without ETMA in iRBD patients | Normal controls | ||||
| Left | Right | Left | Right | Left | Right | ||
| Theta | |||||||
| Frontal | 17.07 |
17.29 |
16.89 |
16.98 |
16.06 |
16.45 | |
| Central | 16.00 |
16.55 |
15.99 |
16.41 |
15.34 |
15.52 | |
| Occipital | 14.05 |
14.05 |
14.22 |
14.22 |
13.50 |
13.77 | |
| Alpha | |||||||
| Frontal | 15.12 |
15.30 |
14.86 |
15.13 |
14.24 |
14.77 | |
| Central | 14.67 |
15.07 |
14.65 |
14.99 |
14.19 |
14.58 | |
| Occipital | 13.99 |
14.2 |
14.11 |
14.24 |
13.62 |
13.77 | |
| Sigma | |||||||
| Frontal | 11.71 |
11.92 |
11.59 |
11.91 |
11.38 |
11.91 | |
| Central | 11.42 |
11.76 |
11.54 |
11.82 |
11.33 |
11.80 | |
| Occipital | 10.61 |
10.83 |
10.66 |
10.92 |
10.75 |
10.81 | |
| Beta | |||||||
| Frontal | 17.84 |
18.06 |
17.62 |
17.74 |
17.26 |
17.66 | |
| Central | 16.62 |
17.09 |
16.56 |
16.94 |
16.78 |
17.08 | |
| Occipital | 14.14 |
14.44 |
14.10 |
14.38 |
14.75 |
14.84 | |
| Gamma | |||||||
| Frontal | 14.59 |
14.97 |
14.21 |
14.47 |
13.81 |
14.06 | |
| Central | 13.32 |
13.61 |
13.08 |
13.29 |
12.99 |
13.07 | |
| Occipital | 11.01 |
11.23 |
10.84 |
11.09 |
11.12 |
11.20 | |
All values were shown as mean
* p
§§§ p
# p
EEG, electroencephalogram; ETMA, excessive transient muscle activity; dB, decibel.
| Phasic REM sleep | With ETMA in iRBD patients | Without ETMA in iRBD patients | Normal controls | ||||
| Left | Right | Left | Right | Left | Right | ||
| Theta | |||||||
| Frontal | 18.90 |
19.06 |
18.27 |
18.55 |
17.96 |
18.16 | |
| Central | 17.60 |
18.01 |
16.92 |
17.37 |
16.50 |
16.55 | |
| Occipital | 14.84 |
14.88 |
14.57 |
14.59 |
13.98 |
14.49 | |
| Alpha | |||||||
| Frontal | 16.22 |
16.49 |
15.68 |
16.00 |
15.41 |
15.74 | |
| Central | 15.31 |
15.66 |
14.80 |
15.22 |
14.69 |
14.97 | |
| Occipital | 14.42 |
14.54 |
14.09 |
14.30 |
13.85 |
14.20 | |
| Sigma | |||||||
| Frontal | 12.00 |
12.18 |
11.98 |
12.18 |
11.81 |
12.26 | |
| Central | 11.42 |
11.60 |
11.47 |
11.76 |
11.40 |
11.66 | |
| Occipital | 10.49 |
10.68 |
10.56 |
10.77 |
10.29 |
10.42 | |
| Beta | |||||||
| Frontal | 17.79 |
17.82 |
17.82 |
17.89 |
17.56 |
17.98 | |
| Central | 16.45 |
16.75 |
16.55 |
16.88 |
16.59 |
16.86 | |
| Occipital | 14.27 |
14.62 |
14.20 |
14.62 |
14.84 |
14.97 | |
| Gamma | |||||||
| Frontal | 14.48 |
14.74 |
14.44 |
14.65 |
14.27 |
14.38 | |
| Central | 13.21 |
13.42 |
13.20 |
13.40 |
13.18 |
13.26 | |
| Occipital | 11.39 |
11.66 |
11.13 |
11.42 |
11.67 |
11.87 | |
All values were shown as mean
*** p
§§§ p
# p
During tonic REM sleep (regardless of ETMA) in the iRBD patients and normal
controls, the powers of theta, beta, and gamma waves in the frontal region were
significantly greater than those in the ipsilateral central (p
During phasic REM sleep (regardless of ETMA) in the iRBD patients and normal
controls, the powers of all five EEG frequency bands analysed in the frontal
region were significantly greater than those in the ipsilateral central
(p
In the frontal region, during tonic REM sleep regardless of ETMA,
inter-hemispheric EEG asymmetry scores of theta, alpha, sigma and beta waves were
significantly smaller in the iRBD patients compared to normal controls (p
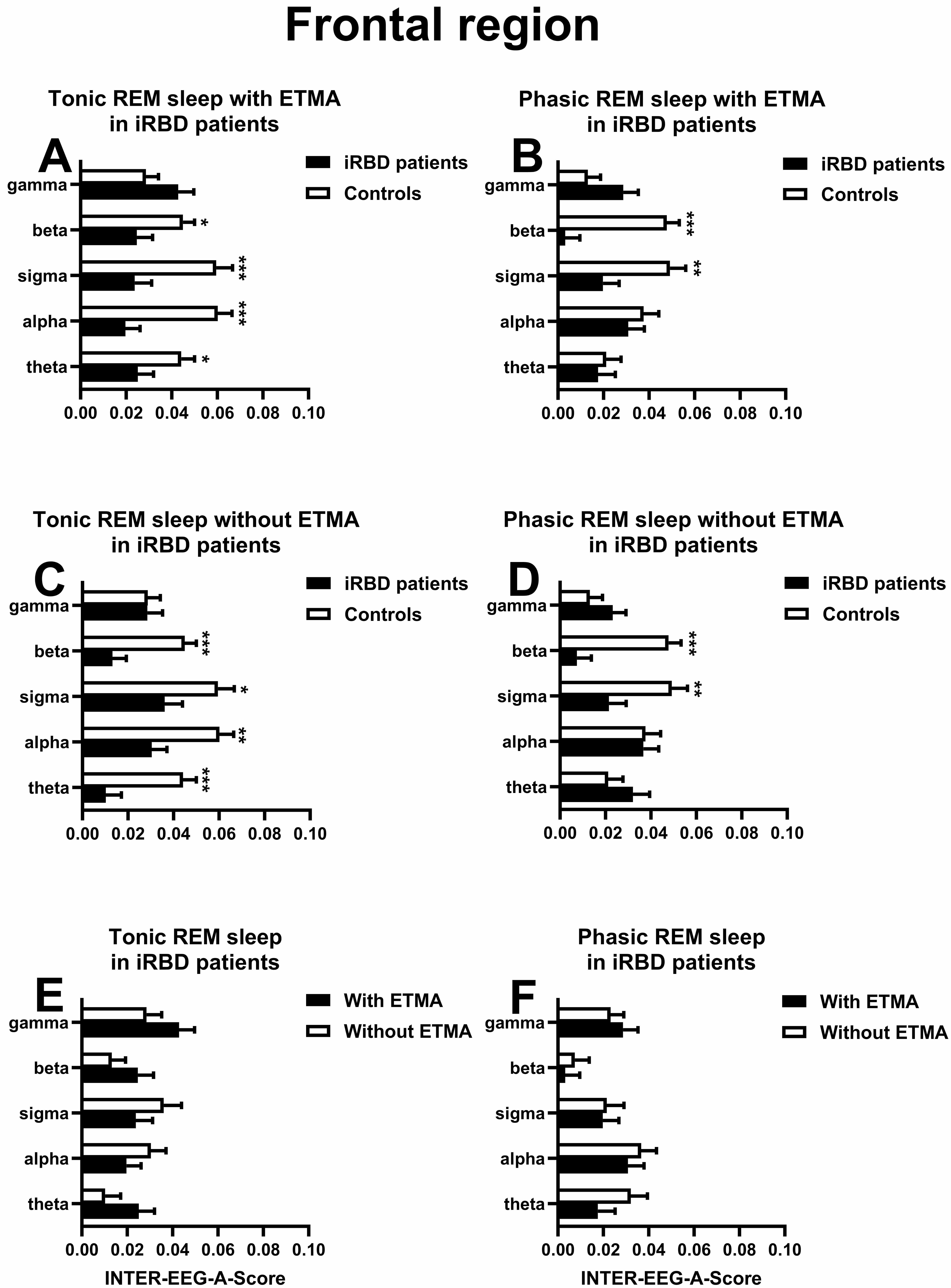 Fig. 1.
Fig. 1.
INTER-EEG-A Scores in the frontal regions in the iRBD patients
and normal controls during REM sleep. (A–D) INTER-EEG-A Scores in the frontal
regions during tonic (A,B) and phasic (C,D) REM sleep in the iRBD patients and
normal controls. (E,F) Inter-hemispheric EEG asymmetry Score (INTER-EEG-A-Score) in the frontal regions during tonic and
phasic REM sleep with and without excessive transient muscle activity (ETMA) in the iRBD patients. Data are reported as
mean
In the central region, inter-hemispheric EEG asymmetry scores of beta and gamma
waves during tonic REM sleep with ETMA, not during tonic sleep without ETMA, were
significantly greater in the iRBD patients than those in normal controls
(p
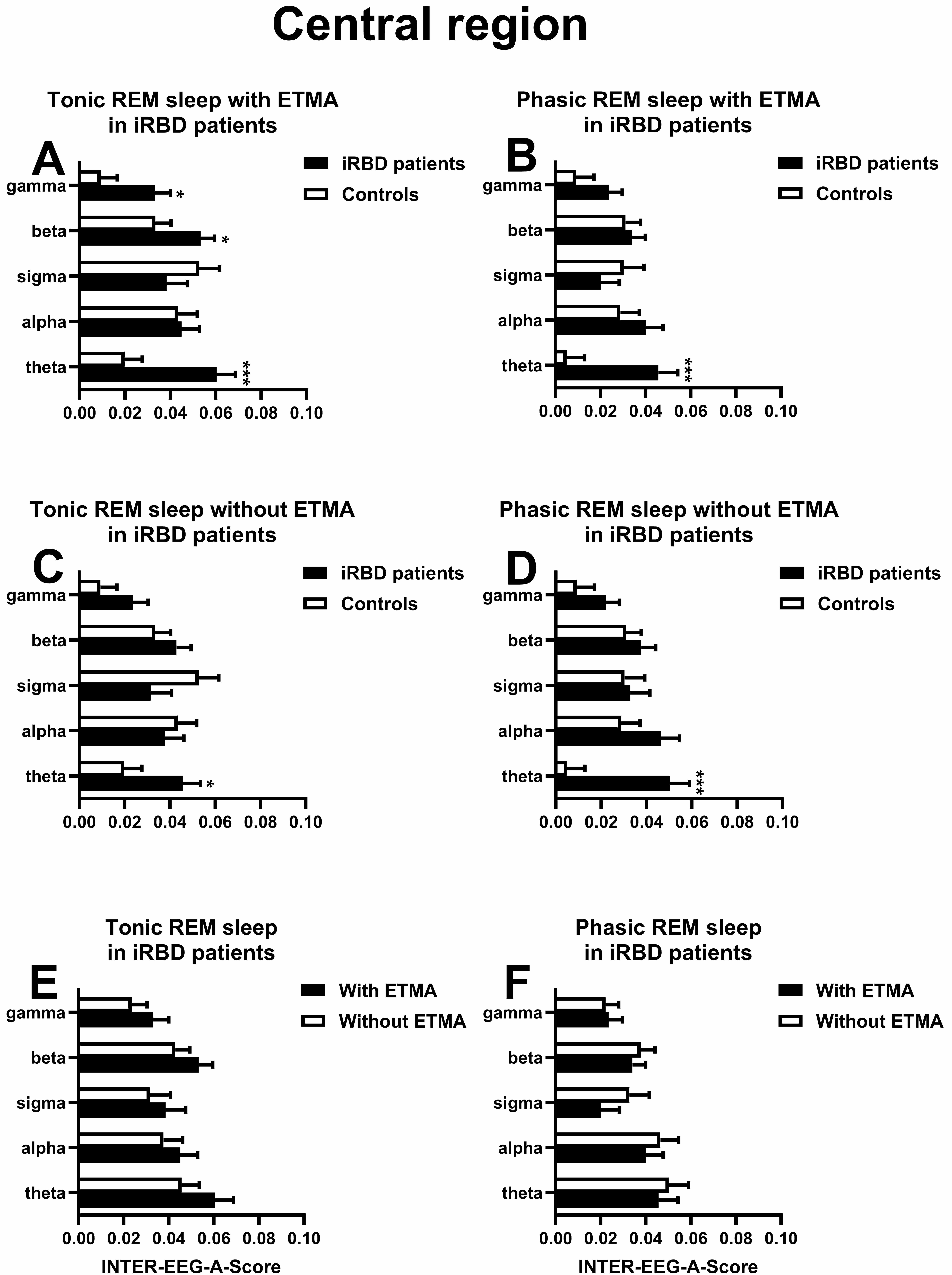 Fig. 2.
Fig. 2.
EEG powers and INTER-EEG-A Scores in the central regions in the
iRBD patients and normal controls during REM sleep. (A–D) INTER-EEG-A Scores in
the central regions during tonic (A,B) and phasic (C,D) REM sleep in the iRBD
patients and normal controls. (E,F) INTER-EEG-A Scores in the central regions
during tonic and phasic REM sleep with and without ETMA in the iRBD patients.
Data are reported as mean
In the occipital region, inter-hemispheric EEG asymmetry scores of beta waves
during tonic and phasic REM sleep (regardless of ETMA) were significantly greater in the iRBD patients than those in normal controls (p
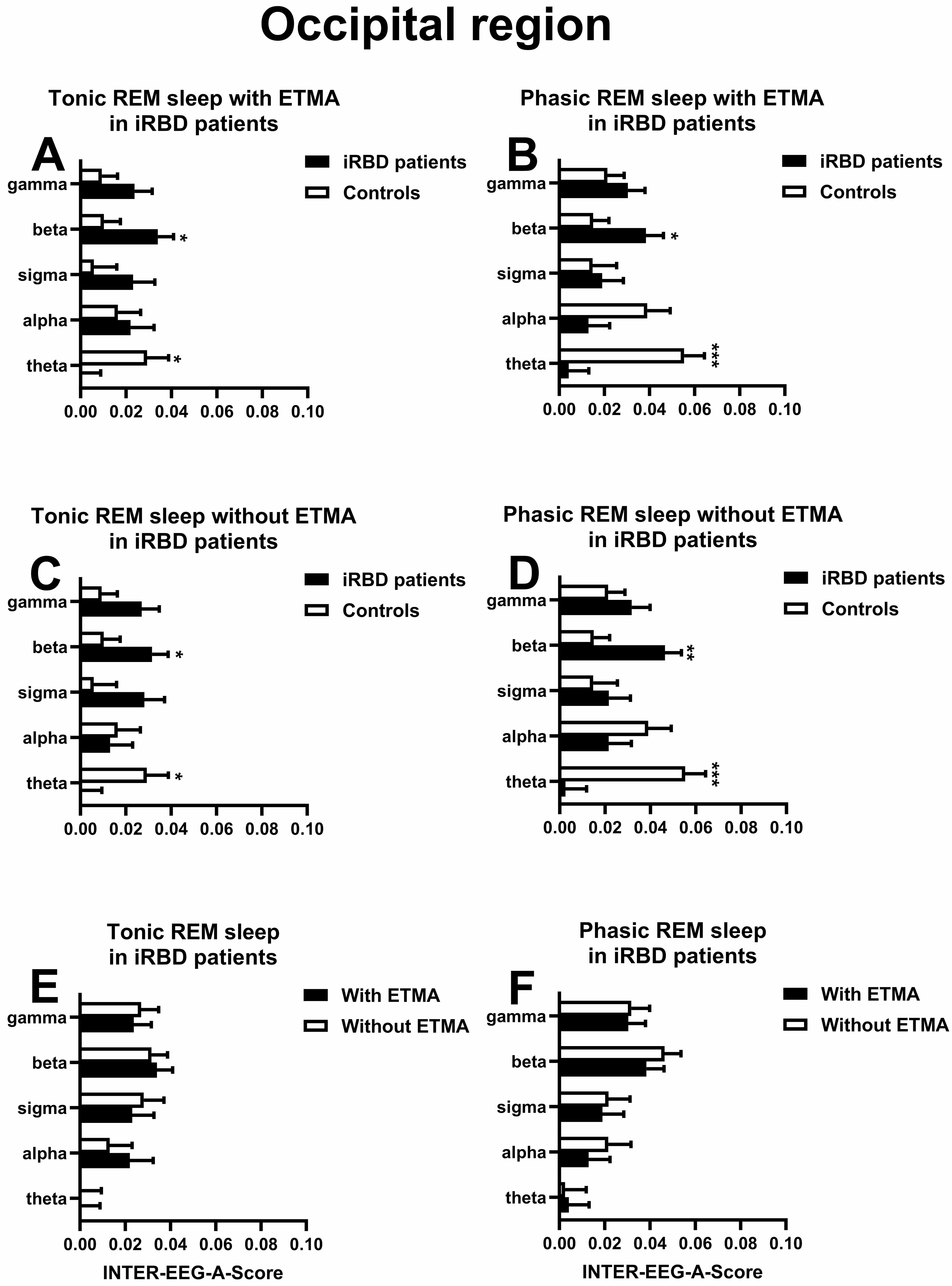 Fig. 3.
Fig. 3.
EEG powers and INTER-EEG-A Scores in the occipital regions in
the iRBD patients and normal controls during REM sleep. (A–D) INTER-EEG-A
Scores in the occipital regions during tonic (A,B) and phasic (C,D) REM sleep in
the iRBD patients and normal controls. (E,F) INTER-EEG-A Scores in the occipital
regions during tonic and phasic REM sleep with and without ETMA in the iRBD
patients. Data are reported as mean
In addition, no significant difference was found in inter-hemispheric EEG
asymmetry scores of all five frequency bands in the frontal, central, and
occipital regions between REM sleep with and without ETMA in the iRBD patients
(p
As reported in Table 4, during tonic REM sleep with ETMA in the frontal cortical
region, the iRBD patients exhibited a significantly greater EEG power in theta,
alpha, sigma, beta and gamma waves in the left side, and theta, alpha, beta, and
gamma waves in the right side than that in the normal controls (p
| Tonic REM sleep | iRBD patients | Controls | p* | p§ | p# | ||
| With ETMA | Without ETMA | ||||||
| Left frontal region (F3) | |||||||
| Theta | 17.07 |
16.89 |
16.06 |
NS | |||
| Alpha | 15.12 |
14.86 |
14.24 |
||||
| Sigma | 11.71 |
11.59 |
11.38 |
NS | NS | ||
| Beta | 17.84 |
17.62 |
17.26 |
||||
| Gamma | 14.59 |
14.21 |
13.81 |
||||
| Right frontal region (F4) | |||||||
| Theta | 17.29 |
16.98 |
16.45 |
||||
| Alpha | 15.30 |
15.13 |
14.77 |
NS | |||
| Sigma | 11.92 |
11.91 |
11.91 |
NS | NS | NS | |
| Beta | 18.06 |
17.74 |
17.66 |
NS | |||
| Gamma | 14.97 |
14.47 |
14.06 |
||||
| Left central region (C3) | |||||||
| Theta | 16 |
15.99 |
15.34 |
NS | |||
| Alpha | 14.67 |
14.65 |
14.19 |
NS | |||
| Sigma | 11.42 |
11.54 |
11.33 |
NS | NS | NS | |
| Beta | 16.62 |
16.56 |
16.78 |
NS | NS | NS | |
| Gamma | 13.32 |
13.08 |
12.99 |
NS | |||
| Right central region (C4) | |||||||
| Theta | 16.55 |
16.41 |
15.52 |
NS | |||
| Alpha | 15.07 |
14.99 |
14.58 |
NS | |||
| Sigma | 11.76 |
11.82 |
11.80 |
NS | NS | NS | |
| Beta | 17.09 |
16.94 |
17.08 |
NS | NS | NS | |
| Gamma | 13.61 |
13.29 |
13.07 |
NS | |||
| Left occipital region (O1) | |||||||
| Theta | 14.05 |
14.22 |
13.50 |
NS | |||
| Alpha | 13.99 |
14.11 |
13.62 |
NS | NS | ||
| Sigma | 10.61 |
10.66 |
10.75 |
NS | NS | NS | |
| Beta | 14.14 |
14.10 |
14.75 |
NS | |||
| Gamma | 11.01 |
10.84 |
11.12 |
NS | |||
| Right occipital region (O2) | |||||||
| Theta | 14.05 |
14.22 |
13.77 |
NS | NS | ||
| Alpha | 14.20 |
14.24 |
13.77 |
NS | |||
| Sigma | 10.83 |
10.92 |
10.81 |
NS | NS | NS | |
| Beta | 14.44 |
14.38 |
14.84 |
NS | |||
| Gamma | 11.23 |
11.09 |
11.20 |
NS | NS | ||
| Phasic REM sleep | |||||||
| Left frontal region (F3) | |||||||
| Theta | 18.90 |
18.27 |
17.96 |
NS | |||
| Alpha | 16.22 |
15.68 |
15.41 |
NS | |||
| Sigma | 12.00 |
11.98 |
11.81 |
NS | NS | NS | |
| Beta | 17.79 |
17.82 |
17.56 |
NS | NS | ||
| Gamma | 14.48 |
14.44 |
14.27 |
NS | NS | NS | |
| Right frontal region (F4) | |||||||
| Theta | 19.06 |
18.55 |
18.16 |
||||
| Alpha | 16.49 |
16.00 |
15.74 |
NS | |||
| Sigma | 12.18 |
12.18 |
12.26 |
NS | NS | NS | |
| Beta | 17.82 |
17.89 |
17.98 |
NS | NS | NS | |
| Gamma | 14.74 |
14.65 |
14.38 |
NS | |||
| Left central region (C3) | |||||||
| Theta | 17.60 |
16.92 |
16.50 |
||||
| Alpha | 15.31 |
14.8 |
14.69 |
NS | |||
| Sigma | 11.42 |
11.47 |
11.40 |
NS | NS | NS | |
| Beta | 16.45 |
16.55 |
16.59 |
NS | NS | NS | |
| Gamma | 13.21 |
13.2 |
13.18 |
NS | NS | NS | |
| Right central region (C4) | |||||||
| Theta | 18.01 |
17.37 |
16.55 |
||||
| Alpha | 15.66 |
15.22 |
14.97 |
NS | |||
| Sigma | 11.60 |
11.76 |
11.66 |
NS | NS | NS | |
| Beta | 16.75 |
16.88 |
16.86 |
NS | NS | NS | |
| Gamma | 13.42 |
13.4 |
13.26 |
NS | NS | NS | |
| Left occipital region (O1) | |||||||
| Theta | 14.84 |
14.57 |
13.98 |
||||
| Alpha | 14.42 |
14.09 |
13.85 |
NS | |||
| Sigma | 10.49 |
10.56 |
10.29 |
NS | NS | NS | |
| Beta | 14.27 |
14.20 |
14.84 |
NS | |||
| Gamma | 11.39 |
11.13 |
11.67 |
||||
| Right occipital region (O2) | |||||||
| Theta | 14.88 |
14.59 |
14.49 |
NS | |||
| Alpha | 14.54 |
14.3 |
14.20 |
NS | NS | NS | |
| Sigma | 10.68 |
10.77 |
10.42 |
NS | NS | ||
| Beta | 14.62 |
14.62 |
14.97 |
NS | |||
| Gamma | 11.66 |
11.42 |
11.87 |
NS | |||
p*: REM sleep with ETMA in iRBD patients vs Normal controls.
p§: REM sleep without ETMA in iRBD patients vs Normal controls.
p#: REM sleep with ETMA in iRBD patients vs REM sleep without ETMA in iRBD patients. NS, not significant.
During tonic REM sleep with ETMA in the central cortical region, the iRBD
patients exhibited a significantly greater EEG power in theta, alpha and gamma
waves in the left and right side than that in the normal controls (p
During tonic REM sleep with ETMA in the occipital cortical region, the iRBD
patients exhibited a significantly greater EEG power in theta and beta waves in
the left side, and alpha, beta, and theta waves in the right side than that in
the normal controls (p
In addition, during tonic REM sleep with ETMA, the iRBD patients exhibited a
significantly greater (p
As shown in Table 4, during phasic REM sleep with ETMA in the frontal cortical
region, the iRBD patients exhibited a significantly greater EEG power in theta,
alpha waves in the left and right sides, gamma waves in the right side compared
to the normal controls (p
During phasic REM sleep with ETMA in the central cortical region, the iRBD
patients exhibited a significantly greater EEG power in theta and alpha waves in
the left and right sides than that in the normal controls (p
During phasic REM sleep with ETMA in the occipital cortical region, the iRBD
patients exhibited a significantly great EEG power in theta and beta waves in
bilateral sides, and alpha and gamma waves in the left side than that in the
normal controls (p
In addition, during phasic REM sleep with ETMA, the iRBD patients showed a
significantly greater (p
No significant correlation was found between EEG activity asymmetry of theta,
alpha, sigma, beta, and gamma waves in frontal, central and occipital regions
during REM sleep (with and without ETMA) with the ratio of RSWA duration/REM
sleep duration (p
iRBD is a common sleep movement disorder with an abnormal increase in muscle tone accompanied by dream enactment behaviors during REM sleep in patients without any neurological disease. In this study, the authors systematically investigated differences in EEG activities between bilateral cerebral hemispheres and between different cortical regions in the same hemisphere during REM sleep, and found there were intra-hemispheric discrepancy and inter-hemispheric EEG asymmetry during both tonic and phasic REM sleep in the iRBD patients and normal controls. In addition, compared to the normal controls, the iRBD patients exhibited a significantly greater inter-hemispheric asymmetry in EEG activities in the central and occipital regions, whereas the frontal region showed a significantly smaller asymmetry. These findings of the altered inter-hemispheric asymmetry in EEG activities of iRBD patients might reflect changes in the bilateral neuronal differences that normally exist in the left and right hemispheres.
Tonic and phasic REM sleep occurs alternatively during REM sleep and might have different functions. Phasic REM sleep is mainly involved in the processing of internal motor, sensory and corticothalamic network information, promoting sleep stability and emotional regulation, and human subjects are not sensitive to changes in the surrounding environment during phasic REM sleep [5, 27]. By contrast, tonic REM sleep is associated with a higher alertness to the surroundings and a lower threshold of awakening to protect the body from potential external dangers [27]. In addition, the differences between tonic and phasic REM sleep are also reflected in alterations in memory and sensory processing [5, 27]. The memory and sensory processing functions of individuals are weakened to a large extent during phasic REM sleep, but partially recovered during tonic REM sleep [5, 27].
Dream-enactment behaviors during phasic REM sleep, in RBD patients are directionally coherent with saccadic eye movements, implying active visual scanning of dream imagery [28, 29]. The previous study has shown that pre-representative behavior (i.e., the 60 s prior to dream enactment behaviors) was related to an increased delta power in the left frontal cortex and gamma power in the right frontal and occipital cortex, and enhanced beta-band functional connectivity compared with background (i.e., a 60 s segment with the least submental muscle EMG activity) [30].
In this study, theta and alpha power in the frontal, central and occipital regions, and gamma power in the occipital region were found to be significantly greater during phasic REM sleep with ETMA than during phasic REM sleep without ETMA (Table 4). Gamma-band synchronization has been reported to play a crucial role in the transmission of visual information and in visuomotor integration [31, 32]. In iRBD patients, phasic REM is associated with abnormal excitation of the motor cortex and altered high-frequency (beta/gamma) connectivity [4]. By contrast, during tonic REM sleep with ETMA, beta power in the frontal cortical region and gamma power in the frontal, central and occipital cortical regions were found to be significantly greater than those during tonic REM sleep without ETMA (Table 4). In comparison to tonic and phasic REM sleep without ETMA, the EEG power changes observed during REM sleep with ETMA likely reflect a pathological overactivation of sensorimotor and associative networks, which may drive the occurrence of dream enactment behaviors [4].
In the current study, theta and alpha powers in the iRBD patients were generally
greater than those in the normal controls during tonic REM sleep regardless of
ETMA, except for no significant difference in alpha power in left occipital
region in the iRBD during tonic REM with ETMA compared to the normal controls
(Table 4). This feature is similar to previous findings in patients with
obstructive sleep apnea, showing that percentage of total sleep time with
SpO2
Sigma band activity has been reported to show relatively little variation during REM sleep [37], which aligns with the finding that sigma power did not differ significantly in most cortical regions (Table 4). Compared to the normal controls, beta and gamma power during phasic REM sleep and beta power during tonic REM sleep in the iRBD patients were significantly smaller in the occipital region. In contrast, beta and gamma power in the frontal region during tonic REM sleep and gamma power in the central region during tonic REM sleep with ETMA were significantly greater compared to the normal controls (Table 4). Cholinergic neurons in the brainstem suppress specific low-frequency oscillations, while high-frequency oscillations are related to increased acetylcholine release from the thalamus and cortex [38]. Rats with basal forebrain cholinergic lesions showed significantly decreased high-frequency EEG activities, particularly in the gamma band, compared to sham controls [39]. The widespread changes in beta and gamma power that are observed in iRBD patients likely reflects impaired functional integration within large-scale cortical networks, and may indicate early dysfunction of cholinergic system in the basal forebrain, which is critical for maintaining high-frequency oscillatory activity during REM sleep.
The nucleus basalis of Meynert (NBM), a primary source of cholinergic projections to the neocortex [40], receives inputs from many brainstem nuclei, such as the laterodorsal tegmental nucleus, the locus coeruleus and the pedunculopontine tegmental nucleus [41, 42], and the functional connection between the NBM and the cerebral cortex has been used to evaluate the basal forebrain cholinergic system [43]. Studies have found that NBM projects to the occipital region of the brain, and that the functional connection between the NBM and the occipital region in iRBD patients is weaker than in normal controls [44]. Although the pathophysiologic mechanisms underlying iRBD are not fully clear, the pathogenesis of iRBD is considered to be associated with damages to the cholinergic neurons in the laterodorsal tegmental nuclei [45], which causes dysfunction in the thalamus, given that the primary sources of cholinergic projections to the thalamus come from the pedunculopontine tegmental nucleus and the laterodorsal tegmental nucleus [46].
The current study revealed that the EEG powers of almost all analyzed EEG waves in the right cerebral hemisphere were significantly greater than those in the left cerebral hemisphere in the iRBD patients and normal controls during tonic and phasic REM sleep (Tables 2,3). This is consistent with the previous study showing structural or functional differences in bilateral cerebral hemispheres, and more involvement of the right cerebral hemisphere in maintaining alertness than the left [15], as maintenance of a certain degree of vigilance at night is necessary [18].
Hemispheric specialization, particularly in the prefrontal cortex, supports efficient cognitive processing by lateralizing functions such as attention, executive control, and emotional regulation [47]. In the frontal region, iRBD patients showed significantly reduced inter-hemispheric asymmetry in theta, sigma, and beta bands during tonic REM sleep regardless of ETMA, and in sigma and beta waves during phasic REM sleep regardless of ETMA (Fig. 1). Reduced frontal asymmetry may indicate a loss of hemispheric specialization. Under comparable cognitive task conditions, older adults typically show reduced lateralization of prefrontal activity relative to younger adults [47]. In contrast, elevated asymmetry in the central regions may indicate lateralized cortical hyperexcitability.
In the central region, the iRBD patients in the current study exhibited increased asymmetry in theta, beta, and gamma bands during tonic REM sleep with ETMA, and in the theta band during tonic REM sleep without ETMA, and in theta band during phasic REM sleep regardless of ETMA. This suggests that in the central region, the right hemisphere became relatively more active than the left in iRBD patients. In addition, the presence of ETMA appears to exacerbate the asymmetry in theta and beta bands during tonic REM sleep. Although iRBD patients do not present with clinical motor symptoms, this pattern may reflect early-stage lateralized neurodegeneration. In the PD patients, symptom laterality is associated with sleep EEG asymmetry, particularly with greater slow-wave power observed in the hemisphere contralateral to the left, but not the right, dominant motor symptoms side [48]. Enhanced central asymmetry in iRBD patients might indicate early unilateral alterations in brainstem or basal ganglia circuits. However, the inter-hemispheric EEG asymmetry scores in the iRBD patients showed no significant difference between REM sleep regardless of ETMA in frontal, central and occipital cortical regions (Figs. 1,2,3). The findings indicate that inter-hemispheric imbalance may not be related to the limb movements.
In the occipital region, iRBD patients showed increased asymmetry in sigma and beta bands during tonic and phasic REM sleep with or without ETMA. As a central hub for cognitive processes, the thalamus regulates communication in the cerebral cortex, playing a critical role in memory, attention, and executive functions [49]. The disturbance of thalamo-occipital functional connection is related to cognitive deficits in various neurological diseases [50] and iRBD patients [51], and significantly weaker functional connection between NBM and the left occipital region in iRBD patients [40, 52].
In this study, it was found there were no significant differences in EEG
activity asymmetry of theta, alpha, sigma, beta, and gamma waves in the frontal,
central and occipital regions between REM sleep with ETMA and without ETMA. In
addition, no significant correlation was found between EEG activity asymmetry of
these waves in all cortical regions during REM sleep (with and without ETMA) with
the ratio of RSWA duration/REM sleep duration (p
In the iRBD patients and normal controls during tonic and phasic REM sleep, it was found that the EEG powers of theta, beta, and gamma bands in the frontal region were significantly greater than those in the central and occipital regions, and those in the central region were significantly greater than in the occipital region (Tables 2,3). The unique physiological functions of REM sleep, such as consolidation of emotional and procedural motor memory [53], might be taking place in the frontal cortex [54, 55], which might result in more active EEG activity in the frontal region than other brain regions, as shown in the current study. During REM sleep, rhythmic waves of neuronal activities in the frontal region are dominated by theta and beta waves [56], and theta and beta waves in the anterior cingulate cortex and dorsolateral prefrontal cortex are also very active [56].
Theta waves were initially found in the hippocampus during spontaneous activity (e.g., walking or exploratory sniffing) and during REM sleep [53], but could also be recorded in many other cortical and subcortical structures [57]. The theta activity in the prefrontal lobe and its interaction with the hippocampus and amygdala during REM sleep may be important for the consolidation of emotional memory [56]. In addition, the theta rhythm during human REM sleep was more phasic than the tonic theta oscillations in rodents [58, 59]. During REM sleep, beta waves have also been found to be active in the frontal and central regions [56]. This is in agreement with findings in the current study showing that EEG power of theta and beta waves in the frontal and central regions were significantly greater than those in the occipital region during tonic and phasic REM sleep.
Gamma waves are related to emotional processing, consolidation of memory, and dream recall [53, 60, 61], and may be a potential marker of the suppression of central adrenergic neurons participating in the encoding of emotionally salient events and activation in the amygdala-hippocampal system during REM sleep [61]. The amygdala is an emotional center of the brain, and the interaction between the amygdala and the hippocampus can regulate emotional recalls [60]. The inhibition of central adrenergic neurons during REM sleep plays a crucial role in emotional regulation, such as decreasing emotional intensity and defusing affective experiences [62].
Gamma as well as beta waves in the frontal region are associated with memory and emotional regulation [63, 64], and may be interchangeable during memory regulation, because the EEG activities can switch from beta to gamma waves in a very short time [65]. It was shown in a previous study that no precise boundary occurs between beta and gamma waves, and that these waves might fluctuate simultaneously, such as increased EEG activities of both beta and gamma waves during cognitive processes involving memory [66]. It was shown in the current study that the EEG powers of theta, beta and gamma waves in the frontal region consistently were significantly greater than in the ipsilateral central and occipital cortical regions during tonic and phasic REM sleep (Tables 2,3). In addition, in the central region, these frequency bands were also significantly greater than in the ipsilateral occipital region.
The function of alpha and sigma waves during REM sleep is still not clear. Alpha
and sigma waves mainly exist during NREM sleep, and high alpha activities may
represent active preparation of the cerebral cortex for complex information
processing, while sigma waves are associated with non-emotional memory
consolidation during NREM sleep [67]. During tonic and phasic REM sleep, alpha
and sigma powers in the frontal region were significantly greater than those in
the central or occipital region, and those in central region were also
significantly greater than in occipital region in the iRBD patients and normal
controls, forming a descending gradient (frontal
In short, in both iRBD patients and normal controls, interhemispheric asymmetry of EEG activities exists and EEG activities of theta, beta and gamma waves are unevenly distributed across different cortical regions of the same hemispheres during tonic and phasic REM sleep. In iRBD patients, inter-hemispheric asymmetry in EEG activities is significantly greater, which might reflect increased differences in neuronal activities between the left and right hemispheres.
Although EEG activities were shown to be unevenly distributed across different cortical regions, and enlarged inter-hemispheric asymmetry in EEG activities in the iRBD patients, this study has some limitations. First of all, these included the selection of only male subjects. The rationale for this was because iRBD was more likely to occur in males than in females, and iRBD symptoms in males are more severe [2]. Therefore, gender differences in EEG activities during REM sleep could not be examined in this study. Second, the age range of the patients and controls was relatively narrow and the number of patients and controls was relatively small. Third, the cognitive functions of the iRBD patients were not systematically examined and the relationship between EEG activities during REM sleep and cognitive functions could not be investigated in this study. Fourth, patients with mental disorders have been excluded from this study and depressive comorbidity has been reported to be relatively common in iRBD [69]. Therefore, our findings may not be fully generalizable to iRBD populations with comorbid depression, and further studies in such populations are warranted. In addition, future studies could include more iRBD patients and controls, including females with a wider range of ages, and the cognitive functions of the iRBD patients will be systematically tested and long-term follow-up will be conducted.
Since the current study is cross-sectional study without follow-up data, EEG asymmetry in iRBD patients cannot serve as a predictive marker of disease progression or reflect the causal relationship of EEG asymmetry with the occurrence of iRBD.
In iRBD patients, EEG activities are unevenly distributed, with an altered inter-hemispheric asymmetry that might be associated with changed bilateral neuronal differences compared to normal controls.
The data used to support the findings of this study are available from the corresponding author upon reasonable request.
ZZ, QL, XZ and DY contributed to the conceptualization and study design; ZZ, QL, QO, MW, CG, FY, and ZX acquired the data; ZZ, QL, XZ, and MW analyzed the data; ZZ, QL and DY drafted the original manuscript, reviewed and edited the final version. All authors have participated in drafting, revising, or critically reviewing the article, and approved the final version for publication. All authors agreed to be accountable for all aspects of the work.
The experimental protocol was reviewed and approved by the local Ethical Committee at the Jiangxi Provincial People’s Hospital (No. 2020102) according to the ethical principles of the Declaration of Helsinki. Informed consent was obtained from all the subjects before participation.
We are grateful to Dr. Barry Sessle of the University of Toronto for his help in revising this article, and we also gratefully acknowledge the contributions of all participants who took part in this study.
This research was supported by the Jiangxi Provincial People’s Hospital Grant (No. 2019-009), Jiangxi Provincial Overseas High-level Talent Project (No. 20242BCE50018) and Jiangxi Province Key Laboratory of Neurology Grant (No. 2024SSY06081).
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/JIN47930.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



