1 School of Physical Therapy and Graduate Institute of Rehabilitation Science, Chang Gung University, 333323 Taoyuan, Taiwan
2 Department of Neurosurgery, Chang Gung Memorial Hospital, Linkou, 333423 Taoyuan, Taiwan
3 Neuroscience Research Center, Chang Gung Memorial Hospital, Linkou, 333423 Taoyuan, Taiwan
4 Department of Electrical Engineering, National Taiwan University, 106319 Taipei, Taiwan
5 Department of Physical Medicine and Rehabilitation, Chang Gung Memorial Hospital, Linkou, 333423 Taoyuan, Taiwan
6 School of Biomedical Engineering, College of Biomedical Engineering, Taipei Medical University, 110301 Taipei, Taiwan
7 Division of Neurosurgery, Department of Surgery, Min-Sheng General Hospital, 330056 Taoyuan, Taiwan
8 Healthy Aging Research Center, Chang Gung University, 333323 Taoyuan, Taiwan
†These authors contributed equally.
Abstract
Parkinson’s disease (PD) is the second most common neurodegenerative disorder among the elderly. Although pharmacological therapies can alleviate symptoms, they often fail to provide sustained or complete symptom control, underscoring the need for alternative therapeutic strategies. Neuromodulation techniques, particularly cortical electrical stimulation (CES), have shown promise in modulating cortical plasticity. However, the therapeutic efficacy of CES in PD remains to be fully elucidated. In this study we investigated the long-term therapeutic potential of a novel CES protocol in a transgenic MitoPark mouse model of PD.
MitoPark mice received CES beginning at 8 weeks of age (one session per day, 2 days per week) for a total of 12 weeks. Motor function was assessed using a comprehensive behavioral battery, including beam walking, open-field, and gait performance tests. Neuroprotective effects were evaluated by quantifying dopaminergic neuronal survival and striatal fiber density using tyrosine hydroxylase (TH) immunohistochemistry.
Long-term CES treatment significantly ameliorated motor impairments in MitoPark mice, improving locomotor activity, gait coordination, and beam walking performance compared with sham controls. Immunohistochemical analyses further revealed enhanced survival of nigrostriatal dopaminergic neurons and fibers in the CES-treated group, indicating pronounced neuroprotective effects.
These findings demonstrate that early and sustained CES intervention mitigates motor deficits and enhances dopaminergic neuron survival in the MitoPark PD model. The results provide compelling preclinical evidence supporting CES as a potential adjunctive neuromodulatory therapy for Parkinson’s disease.
Keywords
- cortical electrical stimulation
- Parkinson’s disease
- MitoPark mouse model
- neuroprotection
- motor function
Parkinson’s disease (PD) is recognized as the second most prevalent neurodegenerative disorder, affecting millions of individuals worldwide [1, 2]. The progressive degeneration of dopamine-producing neurons in the substantia nigra pars compacta (SNc) is the primary pathological hallmark of PD, which disrupts the normal functioning of the basal ganglia-cortex circuits, resulting in a variety of motor and non-motor symptoms [3, 4, 5]. The mechanisms underlying inducing neurodegeneration in PD are associated with genomic, epigenetic, and environmental factors that cause dysregulation of neuronal mitochondrial function and oxidative stress. Notably, mitochondrial dysfunction plays a substantial role in contributing to dopaminergic (DA) neuronal death [6, 7, 8]. Both preclinical and clinical observations evidence that defects in the respiratory chain (complex I), increased mutations of mitochondrial deoxyribonucleic acid (DNA), abnormal mitochondrial calcium homeostasis, and increased oxidative stress are consequences of mitochondrial dysfunction that promote neuronal apoptosis in PD [7, 9, 10, 11]. In animal models, toxins such as 6-hydroxydopamine (6-OHDA) and N-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) are commonly used to induce selective degeneration of DA neurons by directly or indirectly disrupting mitochondrial function [12, 13]. However, toxin-induced models have limitations, including systemic toxicity and a more rapid progression of neurodegeneration compared to human PD [14, 15]. Recently, the development of the MitoPark mouse, a transgenic model involving targeted impairment of mitochondrial respiratory chain function in DA neurons, has provided a valuable tool for studying PD. These mice exhibit motor deficits between 14 and 20 weeks of age, gradual nigrostriatal dysfunction, and responsiveness to treatments, closely mimicking the clinical features of human PD [15, 16, 17]. The MitoPark model is well-suited for investigating the progression of PD and evaluating the therapeutic potential of novel interventions.
While pharmacologic therapies, particularly levodopa, have long been the standard treatment for PD, their long-term effectiveness is limited by complications such as dyskinesia and motor fluctuations [18, 19, 20]. This has created an urgent need to explore complementary therapeutic strategies. Neuromodulation, particularly brain stimulation techniques, has therefore gained increasing attention as a promising therapeutic avenue [21, 22, 23]. Deep brain stimulation (DBS) targeting the medial pallidum (GPi) or the subthalamic nucleus (STN) has demonstrated clinical efficacy in patients with advanced PD who no longer respond adequately to pharmacologic treatment [24, 25]. Cortical electrical stimulation (CES) has emerged as an alternative neuromodulation approach that targets cortical networks [26]. Previous studies have demonstrated that CES can exert relatively focal modulatory effects and alleviate symptoms across a range of neurological conditions, including chronic pain, epilepsy, ischemic stroke, traumatic brain injury, and PD [27, 28, 29, 30]. In addition to its symptomatic benefits, CES has been reported to facilitate activity-dependent neuroplasticity, which may contribute to neuroprotective and neuroregenerative processes under certain pathological conditions [31].
In the context of PD, CES delivers electrical fields epidurally to the M1, thereby modulating neuronal membrane potentials and cortical excitability. Rather than directly targeting subcortical nuclei, CES is proposed to influence basal ganglia function indirectly through established cortico-basal ganglia networks, including corticostriatal projections and hyperdirect cortico-subthalamic pathways [32, 33]. Through these interconnected circuits, modulation of motor cortical activity may alter downstream basal ganglia-thalamocortical dynamics that are disrupted in PD. Consistent with this network-level framework, a recent animal study reported that motor cortex CES using hyperpolarizing currents enhanced cortical unit activity and partially compensated for impaired striatal inhibition, resulting in improved locomotor performance in a Parkinsonian model. These findings suggest that CES may serve as a potential adjunct or alternative to conventional STN-DBS by modulating cortical-subcortical interactions rather than directly stimulating deep nuclei. Additional support for the therapeutic benefits of CES is provided by Kuo and colleagues [34], who demonstrated improvements in gait patterns, reduced akinesia, enhanced locomotor activity, and preservation of DA neurons following a 4-week CES intervention in a 6-OHDA rat model. Clinical studies have further reported that extradural or epidural motor cortex stimulation can lead to improvements in motor and, in some cases, cognitive symptoms in patients with PD, with a relatively favorable safety profile and without severe adverse events [35, 36, 37]. Although CES may not achieve the same magnitude of symptomatic relief as DBS in advanced disease stages, its comparatively lower invasiveness and safety profile suggest potential advantages for long-term or early-stage intervention in PD.
This present study is conducted to investigate the therapeutic effects of CES by evaluating motor performance and DA neurodegeneration in the transgenic MitoPark mouse model of PD. Our findings are expected to provide critical insights into the efficacy of CES, emphasizing its potential as a promising intervention for PD.
This study utilized male MitoPark mice, a genetic model of PD that replicates many key characteristics of the condition, including progressive neurodegeneration. This model was applied to investigate the effects of CES, offering a platform to study the underlying mechanisms of Parkinsonism. The mice were housed in standard plastic cages with ad libitum access to food and water. The animals were kept under controlled conditions at a constant temperature of 24 °C with a 12-hour light-dark cycle. All experimental procedures were reviewed and approved by the Institutional Animal Care and Use Committee (IACUC) of Chang Gung University (CGU15-151), ensuring compliance with the Guide for Laboratory Animal Facilities and Care issued by the Council of Agriculture, Executive Yuan, Taiwan. All MitoPark transgenic mice were obtained from the National Center for Biomodels, where genotype verification was performed by polymerase chain reaction (PCR) prior to shipment.
Eight-week-old male MitoPark mice were randomized into two groups (n = 8 per group): the CES-treated group (MitoPark_CES (+)) and the sham-treated control group (MitoPark_CES (–)). The CES intervention was administered twice per week for 12 weeks. Behavioral assessments were conducted every two weeks to evaluate motor functions, including locomotor activity, gait, and coordination. At the end of the treatment period, immunohistochemistry (IHC) analysis was performed to quantify the survival of DA neurons in targeted brain regions. A schematic of the experimental design is presented in Fig. 1.
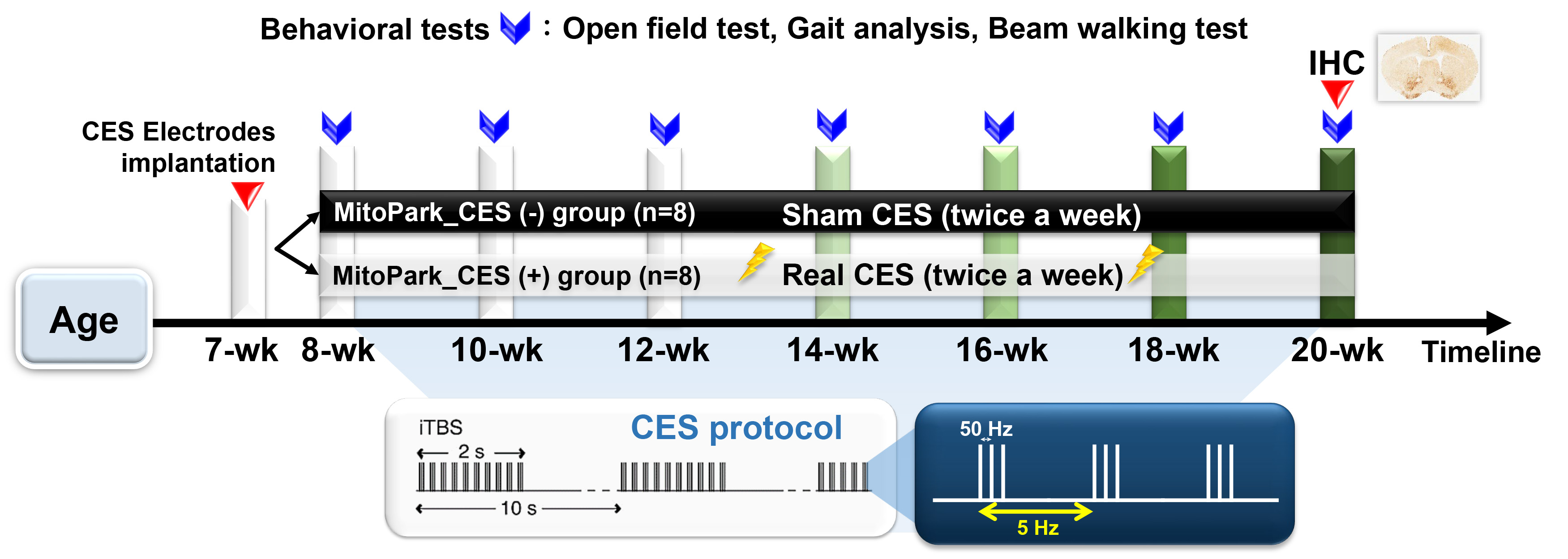 Fig. 1.
Fig. 1.
Experimental design for long-term CES treatment. CES treatment began when the MitoPark mice reached 8 weeks of age and was administered twice per week for 12 consecutive weeks. Behavioral assessments, including beam walking test, open field test, and gait analysis, and were conducted every two weeks to evaluate the effects of CES over time. At the end of the treatment period (at 20 weeks of age), TH-immunoreactivity was quantified to determine the neuroprotective effects of 12-week CES intervention on nigrostriatal DA neurons and fibers. CES, cortical electrical stimulation; TH, tyrosine hydroxylase; DA, dopaminergic; IHC, immunohistochemistry.
Surgical preparation involved anesthetizing 7-week-old mice with intraperitoneal
injection of Zoletil 50 (50 mg/kg, Vibac, Carros, France)/xylazine (10 mg/kg,
Rompun, Bayer, Barmen, Germany). Once deep anesthesia was confirmed, animals were
stabilized in a stereotaxic frame (Model 940, David Kopf Instruments, Tujunga,
CA, USA). Following a 2-cm midline scalp incision to expose the skull, stainless
steel epidural screw electrodes (1.6 mm pole diameter, 0–80
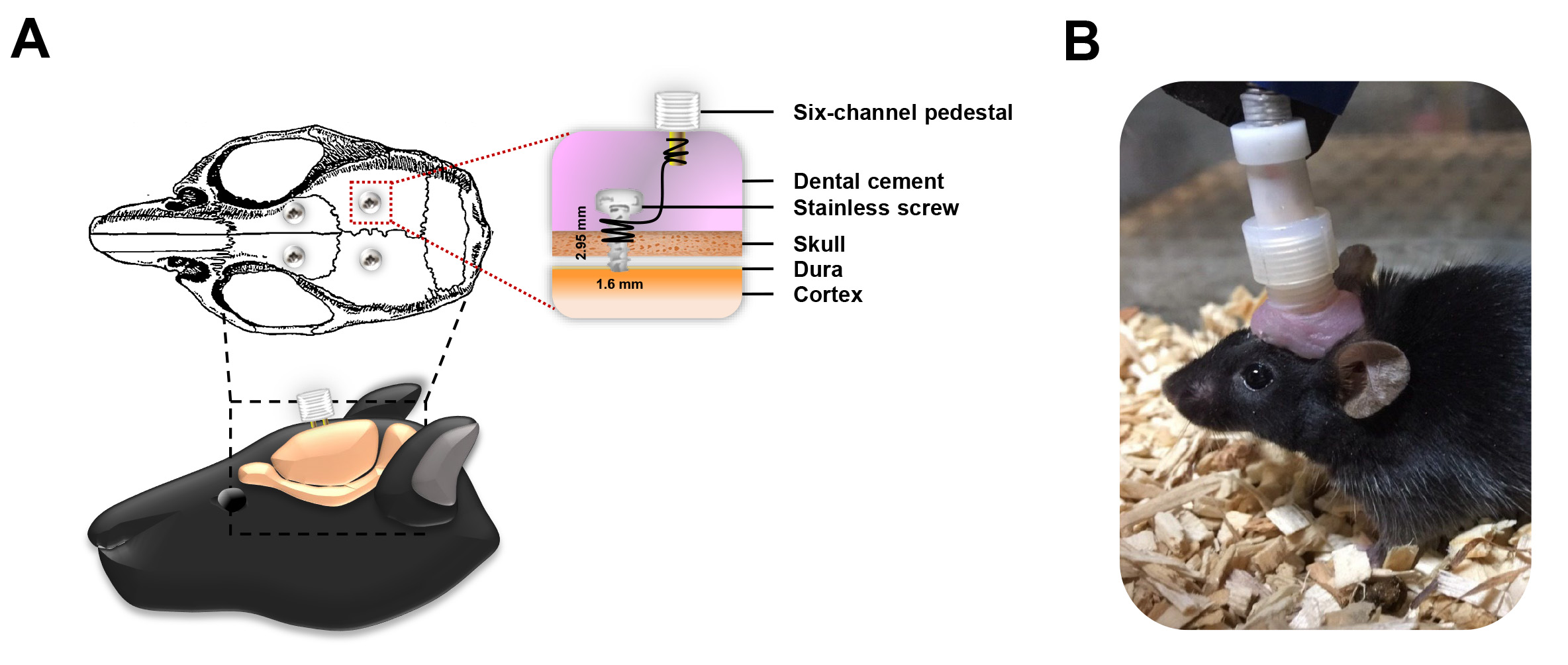 Fig. 2.
Fig. 2.
Placement and assembly of the CES electrode system. (A) Electrodes were bilaterally implanted over the M1 regions (forelimb and hindlimb), wired to a six-channel pedestal and secured using dental acrylic. (B) For each treatment session, the head electrode pedestal functioned as the interface for the stimulator connector, ensuring consistent electrical coupling for stimulation delivery.
In the MitoPark_CES (+) group, CES was delivered using an intermittent theta-burst stimulation (iTBS) protocol, following established parameters [38]. Electrical stimulation was applied to the M1 using charge-balanced biphasic square-wave pulses, designed to ensure net zero charge transfer per pulse and to minimize the risk of electrochemical tissue injury. Each biphasic pulse consisted of symmetrical cathodic and anodic phases with equal amplitude and duration, resulting in the absence of a net direct current component. The iTBS protocol consisted of triplets of biphasic pulses delivered at a frequency of 50 Hz, with each triplet repeated every 200 ms. Each iTBS train lasted 2 seconds and was followed by a 10-second inter-train interval. A total of 20 trains were delivered per session, resulting in 600 pulses (approximately 230 s) per session [39, 40]. CES sessions were administered twice per week for 12 consecutive weeks.
To standardize the intervention, the intensity of CES was administered at 80% of the resting motor threshold (RMT), with the threshold defined by the minimum output required to elicit a visible motor response in the forelimb. The motor threshold was determined individually for each animal, and stimulation intensity was adjusted accordingly to maintain a consistent relative stimulation level across subjects. In the MitoPark_CES (–) group, mice underwent identical surgical implantation, handling, and experimental procedures as the MitoPark_CES (+) group, but no electrical stimulation was delivered during the corresponding time points.
Motor performance was assessed to evaluate the effects of CES, including a battery of three behavioral assessments in the following order: the beam walking test, the open field test, and gait analysis. All behavioral tests were conducted biweekly by a trained examiner who was blinded to the treatment assignments. Furthermore, to determine potential neuroprotective effects of CES on DA neurons and fibers, IHC analysis was performed upon completion of the study at 20–21 weeks.
The beam walking test is a well-established tool for evaluating motor coordination and balance in rodents [41]. The test setup consisted of a Plexiglas beam (120 cm long, 1 cm wide) supported by two vertical stands (30 cm high) with a square home box (10 cm3) positioned at one end of the beam. An aversive stimulus, created using a 60-watt incandescent bulb, was applied at the starting point to encourage the mice to traverse the beam. Before surgery, the mice were trained for at least five successful trials to cross the beam toward the home box. All assessment trials were video-recorded for later analysis. The average time required to traverse the beam across five trials, without falling, was calculated to evaluate performance.
The open field test was employed to assess both anxiety levels and locomotor
activity [42]. The test was conducted in a black Plexiglas arena (40
Gait patterns were evaluated using a specialized walking track equipped with a
video-based system to measure spatiotemporal gait parameters [43, 44]. The setup
included a Plexiglas chamber (40 cm long
The recorded images were processed using a threshold-based image processing technique to delineate paw prints accurately. MATLAB software (version 9.6, R2019a; MathWorks, Natick, MA, USA) was utilized to extract spatial coordinates from the paws, enabling the computation of comprehensive gait indices. From these footprints, we analyzed key spatial parameters (such as step/stride length and step width) and temporal parameters (including walking speed and stance/swing phase duration). A custom-designed image processing program was used for semi-automatic identification of all spatiotemporal data to ensure consistency and accuracy.
To minimize potential confounding factors and allow appropriate comparison across experimental conditions, brains were harvested several hours (approximately 6–8 h) after completion of behavioral assessments to evaluate DA neuronal integrity in the SN and caudate putamen (CPu). Tyrosine hydroxylase (TH) immunohistochemistry, a commonly used marker of dopamine-producing neurons, was employed for histological analysis. For immunohistochemistry, mice were deeply anesthetized with 5% isoflurane (Attane, cat. no. 08547, Panion & BF Biotech Inc., New Taipei City, Taiwan). Transcardial perfusion was performed using saline followed by 4% paraformaldehyde (PFA; cat. no. 15710, Electron Microscopy Sciences, Halfield, PA, USA). Brains were post-fixed in 4% PFA and cryoprotected in 30% sucrose at 4 °C until fully equilibrated. Frozen brains were coronally sectioned at a thickness of 30 µm using a cryostat (Leica CM3050 S, Miami, FL, USA). Sections encompassing the SN and CPu were collected based on stereotaxic coordinates relative to the bregma. To ensure systematic sampling while minimizing section-to-section bias, serial sections were collected and one section was selected at regular, predefined intervals along the rostrocaudal axis, yielding a total of 10 evenly spaced sections per animal for each region of interest. Histological analyses were performed in four animals per group. Free-floating sections were treated with 0.3% hydrogen peroxide (H₂O₂; cat. no. 31642, Honeywell Fluka, Seelze, Germany) in phosphate-buffered saline (PBS; cat. no. P4417, Sigma-Aldrich, St. Louis, MO, USA) for 10 min to quench endogenous peroxidase activity and subsequently blocked with 10% goat serum (cat. no. G9023, Sigma-Aldrich, St. Louis, MO, USA) for 1 h at room temperature to reduce non-specific antibody binding. Sections were incubated with a rabbit anti-tyrosine hydroxylase (TH) primary antibody (1:1000; AB152, Millipore, Burlington, MA, USA) for 1 h at room temperature, followed by incubation with a biotinylated rabbit secondary antibody (1:200; MP-7401, Vector Laboratories, Burlingame, CA, USA) for 1 h. Immunoreactivity was visualized using 3,3′-diaminobenzidine (DAB; SK-4105, Vector Laboratories) for 3–5 min. After staining, sections were mounted on glass slides and digitized at high resolution using an Aperio CS2 slide scanner (Leica Biosystems, Buffalo Grove, IL, USA). Acquired images were transformed into 8-bit grayscale for quantitative analysis. The TH-positive neuronal density in the SN was determined using ImageJ software (version 1.54g, National Institutes of Health, Bethesda, MD, USA) via an automated particle-counting algorithm following standardized thresholds and background correction procedures. Quantification of TH-positive neurons in the substantia nigra was performed independently by two investigators blinded to experimental conditions to minimize observer bias. Values represent the average number of TH-positive neurons per section within the unilateral substantia nigra, derived from systematically sampled coronal sections, and were used for group-level statistical analysis. For the caudate putamen (CPu), TH-positive fiber density was assessed by measuring optical density after subtraction of non-specific background signal. Optical density measurements were obtained separately for each hemisphere and averaged across sections and animals within each group.
All statistical analyses were performed using SPSS version 25.0 (IBM Corp.,
Armonk, NY, USA) according to a predefined analysis plan. Data are expressed as
mean
A two-way repeated-measures ANOVA revealed significant main effects of group
F(1, 14) = 6.877, p = 0.020, but no significant main effect of time
F(6, 84) = 1.809, p = 0.107 and no time
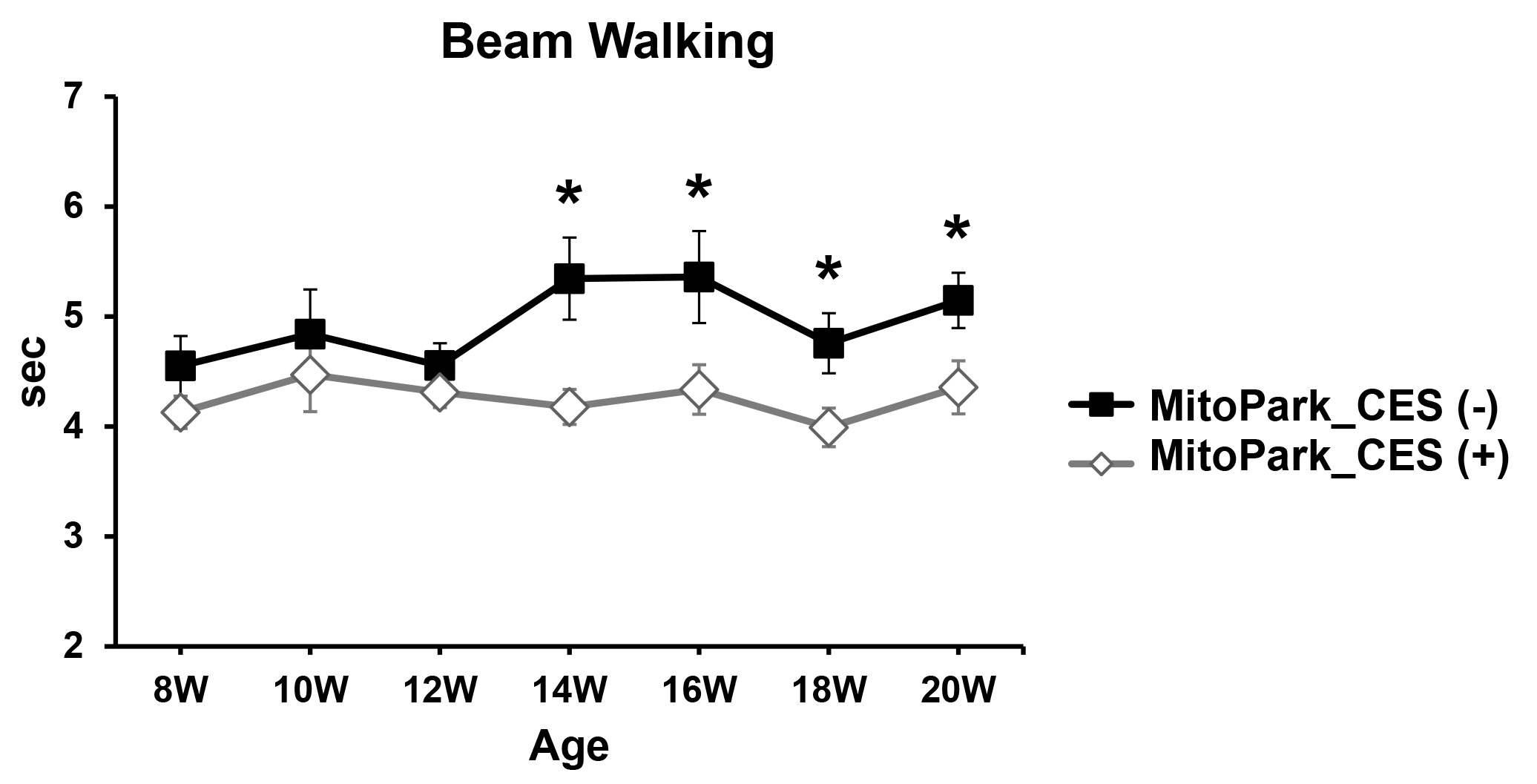 Fig. 3.
Fig. 3.
Effects of CES on motor balance in MitoPark mice.
Time-course analysis of beam walking performance in CES-treated and sham-treated
MitoPark mice over the 12-week intervention period (8 to 20 weeks of age).
Two-way repeated-measures ANOVA revealed a significant main effect of group,
indicating overall shorter beam traversal times in CES-treated mice compared with
sham-treated controls across the testing period. Data are expressed as mean
In the open field test, CES-treated mice showed an increase in locomotor activity compared with control mice over the 12-week observation period during 10-min sessions (Fig. 4).
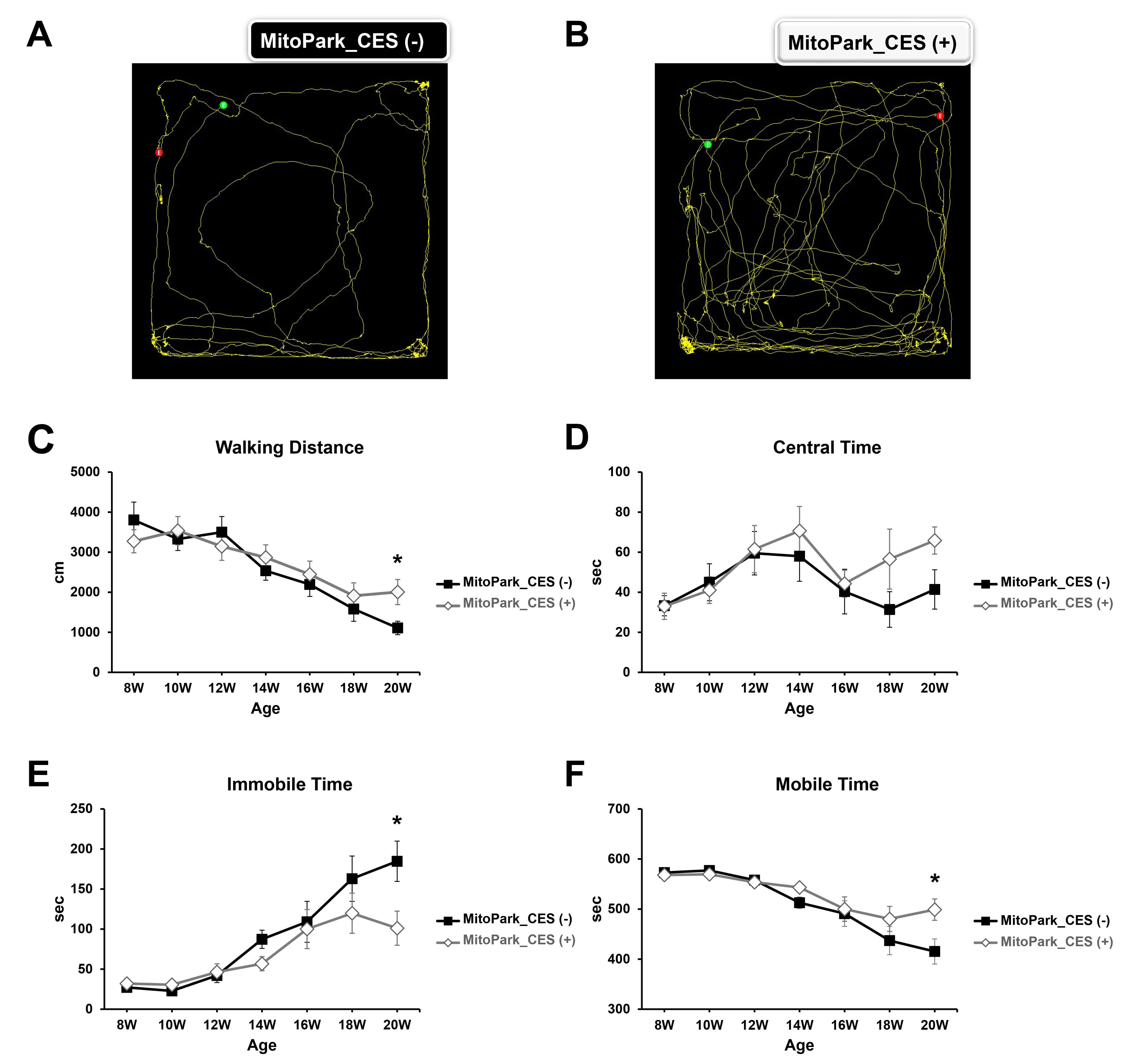 Fig. 4.
Fig. 4.
Effects of cortical electrical stimulation (CES) on
locomotor activity assessed using the open field test. (A) and (B) show
representative movement traces recorded at 20 weeks of age, following completion
of the 12-week CES intervention, from individual sham-treated and CES-treated
MitoPark mice, respectively. (C–F) Quantitative analyses of open field
performance, including (C) total walking distance, (D) time spent in the center
area, (E) immobile time, and (F) mobile time, measured longitudinally from 8 to
20 weeks of age. Two-way repeated-measures ANOVA revealed significant time
For total distance traveled, two-way repeated-measures ANOVA revealed a
significant main effect of time F(6, 84) = 29.583, p
Regarding anxiety-related behavior, analysis of the time spent in the central
area revealed a significant main effect of time F(4.747, 66.46) = 3.385,
p = 0.01, whereas no significant main effects of group (F(1, 14) =
1.188, p = 0.294 or time
Analysis of immobility time demonstrated a significant main effect of time
F(3.920, 54.885) = 22.928, p
For mobile time, two-way repeated-measures ANOVA showed significant main effects
of time F(3.922, 54.907) = 22.956, p
Gait patterns were evaluated by measuring walking speed, stride length, step length, stance phase time, swing phase time, and step width (Fig. 5).
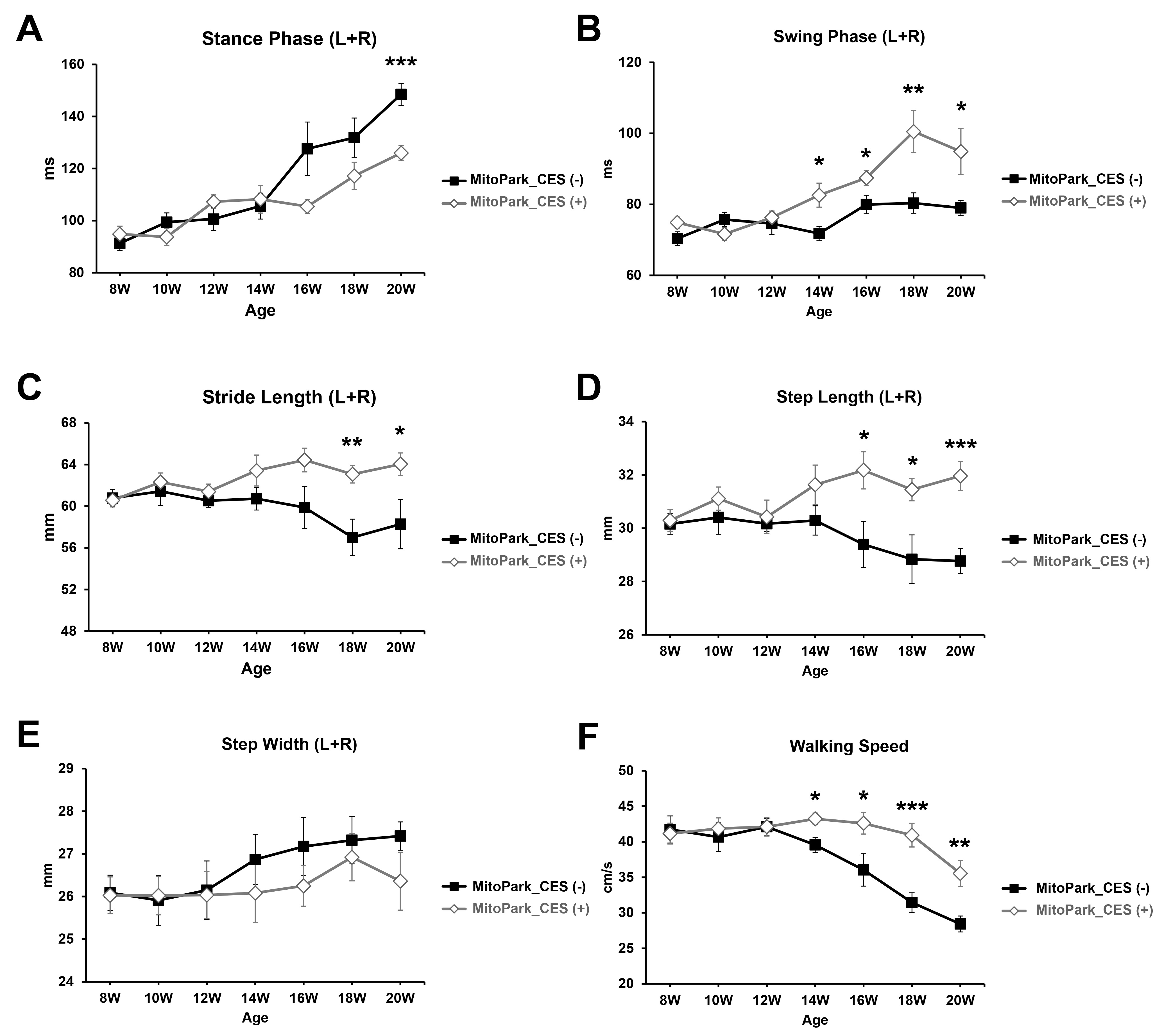 Fig. 5.
Fig. 5.
Longitudinal effects of cortical electrical stimulation
(CES) on gait parameters in MitoPark mice. Time-course analyses of gait
performance, including (A) stance phase duration, (B) swing phase duration, (C)
stride length, (D) step length, (E) step width, and (F) walking speed, in
CES-treated and sham-treated MitoPark mice over the 12-week intervention period
(8 to 20 weeks of age). Two-way repeated-measures ANOVA revealed significant time
For stance phase duration, two-way repeated-measures ANOVA revealed significant
main effects of time F(6, 84) = 24.497, p
Regarding swing phase time, two-way repeated-measures ANOVA revealed significant
main effects of group F(1, 14) = 13.090, p = 0.003, time
F(4.520, 63.279) = 8.887, p
For stride length, two-way repeated-measures ANOVA revealed a significant main
effect of group F(1, 14) =14.918, p = 0.002, indicating overall longer
stride lengths in CES-treated mice compared with controls. No significant main
effect of time F(5.447, 76.264) = 0.772, p = 0.583 or time
Analysis of step length demonstrated significant main effects of group
F(1, 14) = 26.464, p
For step width, no significant main effects of group F(1, 14) = 1.510, p = 0.239, main effects of time (F(6, 84) = 1.445, p = 0.207), or interactions F(6, 84) = 0.382, p = 0.889 were detected, suggesting that CES treatment did not alter lateral paw placement.
Finally, for walking speed, two-way repeated-measures ANOVA revealed significant
main effects of group F(1, 14) = 13.090, p = 0.003, time F(6,
84) = 13.129, p
Quantitative analysis of DA neurons and fibers was performed in the SN and CPu following CES intervention using TH immunostaining (Fig. 6). Independent-samples t-test analysis revealed a significantly higher number of TH-positive neurons in the SN of the MitoPark_CES (+) group compared with the MitoPark_CES (–) group t(6) = 3.821, p = 0.009, indicating greater preservation of DA neuronal cell bodies. In contrast, no significant difference was observed in TH-positive fiber density in the CPu between the two groups t(6) = 0.856, p = 0.425, suggesting that CES treatment did not significantly alter striatal DA fiber density. Together, these findings indicate that CES is associated with preservation of DA neurons in the SN, whereas its effects on striatal DA fibers appear limited under the present experimental conditions.
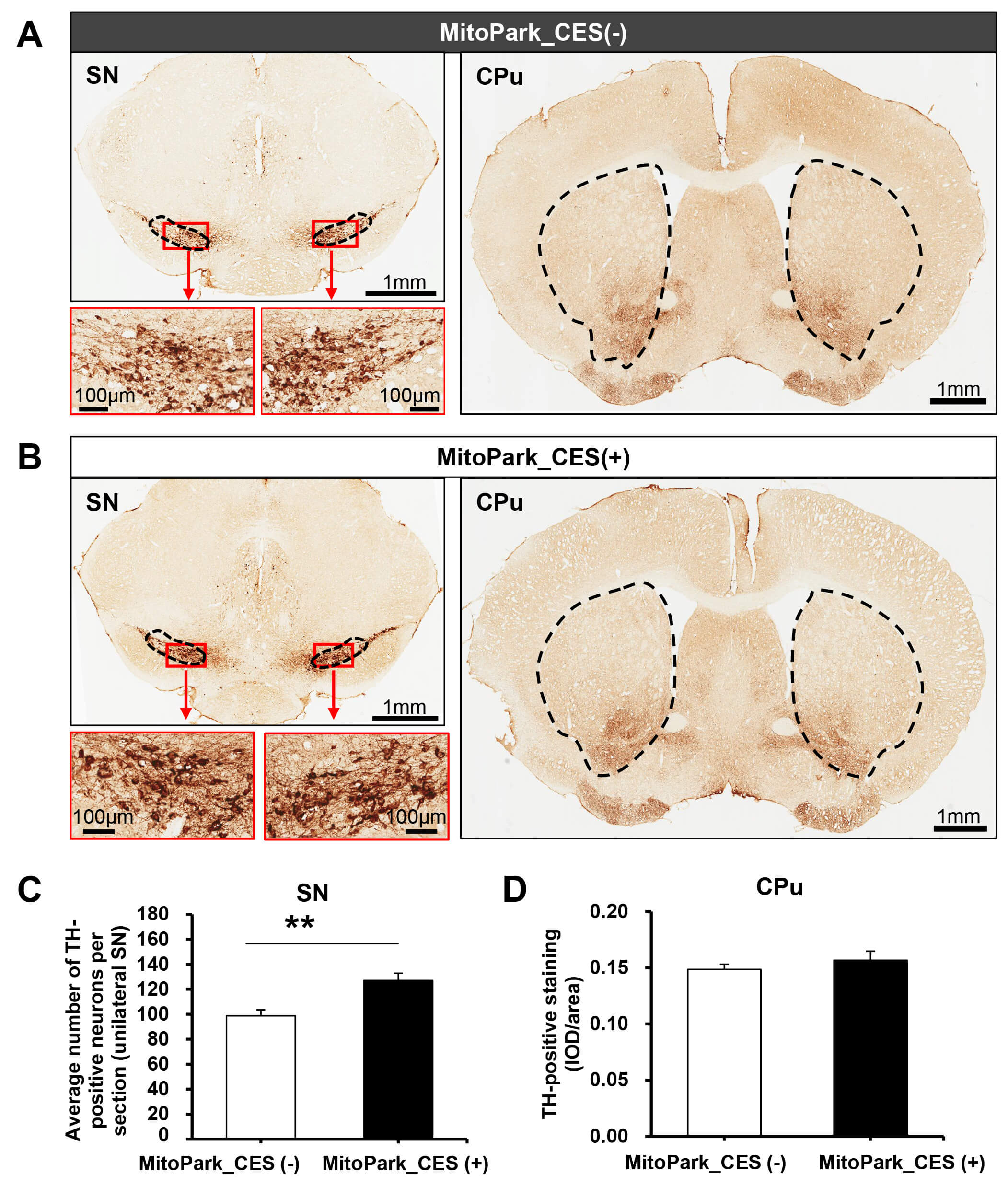 Fig. 6.
Fig. 6.
Effects of cortical electrical stimulation (CES) on dopaminergic
neurons and fibers in MitoPark mice. (A) Representative tyrosine hydroxylase
(TH)–immunostained coronal sections of the substantia nigra (SN) region from
sham-treated MitoPark mice (MitoPark_CES (–)). The dashed outline indicates the
SN. Red boxes denote regions shown at higher magnification below. Scale bar = 1 mm
(main images) and 100 μm (insets). (B) Corresponding TH-immunostained sections from CES-treated MitoPark mice
(MitoPark_CES (+)). Higher-magnification insets illustrate TH-positive neurons
within the SN. Scale bar = 1 mm (main images) and 100 μm (insets).
In both (A) and (B), representative TH-positive fiber staining in
the caudate putamen (CPu) is shown on the right, with dashed outlines indicating
the analyzed CPu regions. (C) Quantification of TH-positive neurons in the SN,
expressed as the average number of TH-positive neurons per section within the
unilateral SN, revealed a significantly higher value in CES-treated mice compared
with sham-treated mice. (D) Quantification of TH-positive fiber density in the
CPu, expressed as integrated optical density (IOD) normalized to area, showed no
significant difference between the two groups. Data are presented as mean
While studies investigating the therapeutic mechanisms of CES in PD remain limited, our findings contribute valuable insights into this field. This study aimed to explore the therapeutic potential of 12-week CES applied to the motor cortex in MitoPark mice by assessing motor functions through behavioral tests and DA neuron preservation using IHC technique. Our results demonstrate that CES significantly improved motor performances, including dynamic balance, locomotor activity, and gait patterns. Moreover, CES-treated MitoPark mice exhibited a higher number of TH-positive neurons in the SN compared to sham-treated mice, suggesting that CES exerts neuroprotective effects by promoting DA neuron survival. These findings align with prior studies reporting that long-term CES improved motor deficits and preserved DA neurons in the 6-OHDA rat model of PD [34].
CES has garnered significant attention in neuroscience and neurorehabilitation for its ability to modulate neuronal activity. As a minimally invasive alternative to DBS, CES provides a safer option for individuals with PD who are ineligible for DBS [28]. Clinical studies have demonstrated that CES can reduce medication requirements, improve motor symptoms, particularly axial symptoms, and enhance quality of life for PD patients [36, 37, 45, 46, 47, 48, 49]. CES has also been associated with improvements in cognitive functions, including memory and executive processes [37]. These findings underscore the therapeutic potential of CES in addressing both motor and non-motor symptoms of PD. In this study, we employed an iTBS protocol, a form of repetitive transcranial magnetic stimulation (rTMS) known to promote long-term cortical plasticity [38, 50, 51]. iTBS has shown efficacy in enhancing motor function, particularly gait, and alleviating depression in PD patients [52]. Preclinical studies have demonstrated that iTBS can improve corticostriatal plasticity, preserve DA neurons, and enhance motor and cognitive functions, including gait, locomotion, and memory [40, 53, 54, 55, 56, 57, 58]. Integrating iTBS with CES in our study provides novel insights into the potential benefits of this paradigm for PD treatment. Stimulation of the M1 in this study was guided by neuroimaging evidence implicating M1 dysfunction in PD pathophysiology [59]. Impairments in the cortico-basal ganglia-thalamocortical network, including M1, are associated with motor deficits in PD. Dopamine depletion disrupts corticostriatal plasticity, increasing inhibition of motor thalamus nuclei and reducing cortical excitability, which collectively contribute to abnormal motor output [60, 61, 62]. Abnormal firing patterns in this network are thought to underlie motor symptoms such as bradykinesia and rigidity [63, 64]. Modulating M1 activity through CES can restore the balance between excitatory and inhibitory inputs, akin to other neuromodulation techniques like rTMS and transcranial direct current stimulation (tDCS). These interventions also enhance motor-evoked potentials (MEPs), a well-established biomarker of cortical plasticity linked to motor improvements [65, 66, 67]. In our study, CES-treated MitoPark mice exhibited significant improvements across several behavioral tests, including the beam walking test, open field test, and gait analysis. These results highlight CES as a promising therapeutic approach for alleviating motor deficits in PD and support its potential role in neurorehabilitation.
The MitoPark mouse model was particularly relevant for this study, as it closely replicates the gradual progression of PD. This model exhibits key features such as levodopa responsiveness, adult-onset neurodegeneration, and behavioral impairments, including reduced locomotor activity and cognitive deficits starting at 8 weeks of age, followed by axonal degeneration in the striatum at 14 weeks and DA neuron loss at 20 weeks [15, 68, 69]. Unlike toxin-based models (6-OHDA and MPTP), which induce acute DA depletion, the MitoPark model mirrors the slow, progressive nature of PD, making it an ideal platform for testing therapeutic strategies and investigating underlying disease mechanisms. Importantly, our decision to initiate the intervention at 8 weeks was based on evidence that this stage marks the onset of neurophysiological and behavioral changes, such as impaired spatial learning, reduced locomotion, and altered firing patterns in DA neurons [15, 70]. These early-stage alterations provide a critical window to reverse or slow down disease progression, and our findings offer valuable insights into the early therapeutic effects of CES. By initiating treatment before severe impairments develop, we aim to enhance the translational relevance of this study and identify effective intervention windows for PD. In addition to the motor and neuropathological features described above, the general phenotype and body weight trajectory of MitoPark mice have been well characterized in previous studies [71, 72, 73, 74]. MitoPark mice typically exhibit normal development during early postnatal stages, followed by progressive motor impairment and a gradual decline in body weight at later stages of disease progression, particularly around 16–20 weeks of age, which coincides with substantial nigrostriatal dopaminergic degeneration [73, 74]. These phenotypic changes have been consistently reported across independent cohorts and are considered hallmarks of the MitoPark model. Accordingly, although longitudinal body weight measurements were not included as predefined outcome measures in the present study, the observed behavioral impairments and disease progression in our cohort are consistent with the established natural history of this model.
Our neuroprotective findings revealed that sham-treated mice experienced significant DA neuron loss in the SN, which correlated with motor impairments, highlighting the link between SN neurodegeneration and motor dysfunction [71]. However, both groups showed a similar extent of TH-positive fiber loss in the striatum, indicating that CES did not significantly affect striatal fiber preservation. This discrepancy may suggest that DA terminal degeneration in the striatum precedes neuronal loss in the SN [72, 73]. While CES demonstrated significant neuroprotection in the SN, this effect was not observed in the striatum. One possible explanation for this discrepancy is that CES primarily benefits the soma of DA neurons in the SN, while its effects may not sufficiently extend to their axonal projections or terminals in the striatum. This hypothesis aligns with the known vulnerability of DA axons and terminals, which often precedes neuronal loss in the SN during the progression of PD [74, 75]. Additionally, CES-induced neuroprotective mechanisms, such as the release of neurotrophic factors (e.g., BDNF and GDNF), and the modulation of neuroinflammation and mitochondrial functions, may predominantly support the survival and function of neuronal cell bodies in the SN, but may not directly enhance DA release or preserve terminal integrity in the striatum [76, 77, 78]. In contrast, previous studies in a 6-OHDA rat model of PD demonstrated that CES preserved both TH-positive neuron counts in the SN and TH-positive fibers in the striatum [34]. This discrepancy might be attributed to differences in the models used. The 6-OHDA model involves acute DA depletion, whereas the MitoPark model reflects a more gradual, mitochondrial dysfunction-driven neurodegeneration, which may render DA terminals in the striatum more susceptible to progressive damage. Moreover, the unique neurodegenerative dynamics of the MitoPark model could highlight limitations in the spatial reach or efficacy of CES in protecting distal DA projections. These findings emphasize the need for future studies to investigate the mechanisms underlying CES-induced neuroprotection across the entire nigrostriatal pathway. Accordingly, the histological findings in the present study should be interpreted as supportive and hypothesis-consistent evidence rather than definitive proof of dopaminergic neuroprotection. Assessments of axonal integrity, DA release, and synaptic function in the striatum, alongside molecular analyses of neurotrophic factor expression and neuroinflammatory markers, would provide valuable insights. Additionally, exploring alternative CES parameters or combined approaches, such as CES with targeted neurotrophic delivery, may enhance its efficacy in protecting both the soma and axonal terminals of DA neurons. Such studies would contribute to optimizing CES as a therapeutic strategy for PD by addressing its full therapeutic potential along the nigrostriatal axis.
Neuroinflammation plays a significant role in DA neurodegeneration in PD models,
including MitoPark mice [79]. Chronic neuroinflammation, driven by persistent
microglial activation and elevated pro-inflammatory cytokines (e.g., IL-6,
IL-1
Several limitations of the present study should be acknowledged. First, the
sample size was limited by the availability and breeding demands of the MitoPark
transgenic mouse model. Although this model closely recapitulates the progressive
nature of Parkinsonian pathology, the modest cohort size restricts statistical
power and generalizability, and the present findings should therefore be
interpreted as preliminary. Second, CES effects were evaluated exclusively in
Parkinsonian mice, without inclusion of non-pathological controls, which limits
conclusions regarding baseline safety and potential off-target effects. Future
studies incorporating healthy control animals will be important to further
establish the safety profile of CES. Third, histological analyses were primarily
focused on nigrostriatal dopaminergic pathology and did not include dedicated
assessments of stimulation-associated cortical alterations, such as local
gliosis, inflammatory responses, or electrode trajectories at the implantation
site. Although electrode placement was guided by established stereotaxic
coordinates targeting the primary motor cortex, direct histological confirmation
of electrode tracks and long-term implant stability was not performed, and these
factors may contribute to inter-individual variability in treatment responses. In
addition, dopaminergic integrity was assessed using tyrosine hydroxylase
immunolabeling, which does not distinguish dopaminergic from noradrenergic
fibers. While noradrenergic innervation of the striatum is relatively sparse
compared with dopaminergic projections and tissues were processed in parallel
under identical conditions, a minor contribution to the measured TH-positive
fiber signal cannot be completely excluded. Moreover,
In conclusion, this study provides compelling evidence that CES is a promising therapeutic strategy for alleviating motor deficits and preserving DA neurons in the MitoPark mouse model of PD. The results highlight the relevance of CES as a non-invasive neuromodulation approach with potential translational applications for human PD patients. These findings contribute to the growing body of literature on non-invasive therapies for neurodegenerative diseases and provide a foundation for future research exploring CES as a viable treatment option for PD.
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
T-HH and C-WK contributed to the conceptualization of the study. Methodology was developed by C-WK, K-TC, C-WP, H-LL, TXDN, and T-HH. Investigation, data curation, formal analysis, and visualization were performed by C-WK and T-HH. The original draft was prepared by TXDN, C-WK, and T-HH. C-KW, T-HH, C-WP, K-TC, M-YC, and H-LL contributed to the analysis and interpretation of data, writing, review, and editing. Funding acquisition was supported by T-HH, M-YC, H-LL and K-TC, with project administration and supervision provided by T-HH. All authors contributed to the revision and editorial improvement of the manuscript, read and approved the final version, and agree to be accountable for all aspects of the work.
All experimental procedures were reviewed and approved by the Institutional Animal Care and Use Committee (IACUC) of Chang Gung University (Approval No. CGU15-151). All animal experiments were conducted in accordance with the Guide for the Care and Use of Laboratory Animals and the regulations for laboratory animal facilities and care issued by the Council of Agriculture, Executive Yuan, Taiwan.
Not applicable.
This work was supported by the National Science and Technology Council (NSTC 113-2314-B-182-050-MY3, and NSTC114-2321-B002-028) and Chang Gung Medical Foundation, Taiwan (CORPD1P0041 and CMRPD1N0302, CMRPD1P0282).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






