1 Department of Rehabilitation Medicine, The First Affiliated Hospital With Nanjing Medical University, 210029 Nanjing, Jiangsu, China
2 School of Rehabilitation Medicine, Nanjing Medical University, 210029 Nanjing, Jiangsu, China
3 Department of Rehabilitation Medicine, Changzhou De’an Hospital, 213002 Changzhou, Jiangsu, China
†These authors contributed equally.
Abstract
The cortex and cerebellum have a closely connected closed-loop circuit. Intermittent theta burst stimulation (iTBS) targeting the cerebellum has shown promise in improving balance function and inducing neuroplasticity. This study investigates whether cerebellar iTBS can elicit cortical responses.
One hundred healthy volunteers were randomly assigned to a real or sham iTBS stimulation group. Functional near-infrared spectroscopy (fNIRS) measured cortical activation during resting, walking, and unilateral support tasks.
During the unilateral support task, graph theory analysis revealed significant changes in brain network properties, suggesting a deviation from optimal small-world organization and reduced global integration. No significant changes were observed during the walking and resting-state tasks.
These findings suggest that cerebellar iTBS can modulate cortical activity, though further studies are needed to confirm its clinical effects.
ChiCTR2300077916, https://www.chictr.org.cn/showproj.html?proj=207394.
Keywords
- cerebellum
- cerebral cortex
- intermittent theta burst stimulation
- functional near-infrared spectroscopy
Closed-loop circuits make a close connection between the cerebellum and the cerebral cortex [1]. The cerebellum can modulate the cortex’s contralateral motor and non-motor areas via the cerebello-dentato-thalamo-cortical pathway to affect motor, cognitive, or other performance [2, 3]. Therefore, the cerebellum has thus been investigated over the years as a critical modulator of the activity of the cerebral cortex [4].
Different methods have explored the connections between the cerebral cortex and the cerebellum. In animal experiments, Kelly and Strick [5] injected transneuronal tracers into different regions of the cerebellum in macaque monkeys and discovered projections from the cerebellum to the M1 and prefrontal cortex (PFC). Imaging evidence from Krienen and Buckner [6] used functional magnetic resonance imaging (fMRI) to perform functional connectivity analysis between the cerebellum and seed points in the motor cortex, dorsolateral PFC, medial PFC, and anterior PFC. As a result, they constructed a provisional map of cerebellar topography based on functional correlations with the frontal cortex.
Non-invasive brain stimulation (NIBS) techniques have also been applied to explore this connection. Although cerebellum-brain inhibition (CBI) provides important evidence for cerebellar–cortical interactions [7, 8], it mainly reflects changes in M1 excitability and offers limited insight into task-related cortical dynamics across multiple brain regions, suggesting an inhibitory effect of the cerebellar hemisphere on the cerebral cortex. At the same time, in a study by Koch et al. [9], intermittent theta burst stimulation (iTBS) was applied to the uninjured cerebellar hemisphere to address persistent balance issues in patients with chronic supratentorial stroke. This intervention resulted in significant improvements in balance function and was accompanied by cortical remodeling in the posterior parietal cortex (PPC). These findings suggest that cerebellar NIBS may influence cortical plasticity by modulating cerebellar–cortical pathways. However, whether cerebellar NIBS can directly and reliably affect cerebral cortical activity remains unclear. This uncertainty largely stems from the limitations of CBI paradigms based on motor-evoked potentials (MEPs). MEPs primarily reflect the excitability of the corticospinal tract, which integrates contributions from multiple subcortical and cortical structures, including the cerebellum, thalamus, basal ganglia, and brainstem. Moreover, hemispheric cerebellar transcranial magnetic stimulation (TMS) may also influence adjacent brainstem regions through current spread. As a result, changes in MEP amplitude cannot be unequivocally attributed to modulation of M1 or broader cortical regions. Consequently, conventional TMS-based approaches provide only indirect and spatially limited evidence of cortical involvement, with observations largely restricted to M1. Nevertheless, some studies have begun using magnetic resonance imaging and electroencephalography (EEG) to reveal changes in the cerebral cortex following cerebellar stimulation [10, 11]. Due to equipment limitations, these observed changes have been based on the resting state. While resting-state analysis provides indirect evidence for balance function, real-time observation during balance tasks might offer more direct insights.
Functional near-infrared spectroscopy (fNIRS), like other brain imaging tools, can observe cortical changes at rest and can also conduct real-time monitoring during motor tasks. Red light frequencies between 700 and 900 nm were used by fNIRS, capable of detecting oxyhemoglobin (Oxy-Hb) and deoxyhemoglobin (Dxy-Hb) [12]. Compared with fMRI and EEG, fNIRS shows greater robustness to movement-related interference, which makes it particularly suitable for studying complex motor tasks [13]. Based on previous experiments [14], we conducted the resting-state data collection. By leveraging fNIRS’s resistance to motion artifacts, we also performed real-time data collection during balance and walking tasks, allowing us to observe changes more directly related to balance regulation. The regions of interest (ROIs) we selected are well-aligned with the balance. The premotor cortex (PMC) and supplementary motor area (SMA) play a crucial role in both anticipatory and compensatory postural adjustments [15]. The PFC has also been shown to be involved in complex balance tasks, while the sensorimotor cortex (SMC) is responsible for executing movements and contributing to balance regulation [16, 17]. Considering the cerebellar hemisphere’s deeper anatomical location, we used a double-cone coil with a greater depth range for stimulation [18, 19].
Therefore, the main objective of our experiment is to observe whether a single session of cerebellar iTBS can induce changes in the cerebral cortex. These changes are monitored not only at resting tasks but also in real-time during walking and unilateral support tasks, which are closely related to balance.
A total of 100 healthy volunteers (52 in the real stimulation group)
participated in this study. Participants were randomly assigned to two groups
using a coin-randomization method: one group received real stimulation, while the
other received sham stimulation. Six participants (4 in the real stimulation
group) were excluded due to weak fNIRS signal collection, and fifteen (8 in the
real stimulation group) participants did not complete all tests due to scheduling
conflicts. Ultimately, 40 participants (age: 23.77
Resting-state tasks are now widely used in research. In addition to this, we designed tasks related to balance. During walking, the body constantly needs to adjust balance to maintain stability and prevent falls. Furthermore, single-leg support further increases the difficulty of maintaining balance. Thus, each participant completed the following three tasks: (1) Resting state: sitting in a relaxed position on a chair for 6 minutes; (2) Walking task: walking at a preferred speed for 30 seconds, followed by 40 seconds of rest, repeated 3 times; (3) Unilateral support task: raising their leg onto a step and maintaining balance on the right leg for 6 minutes. The three tasks were performed on separate days, with a 7-day washout period between any two tasks [10]. The order of the tasks was randomized using a dice-rolling method. For each session, one task was performed first, followed by either real or sham iTBS intervention, depending on the group, and then the task was immediately repeated. Throughout all tasks, fNIRS was used to collect signal data. The procedure is illustrated in Fig. 1.
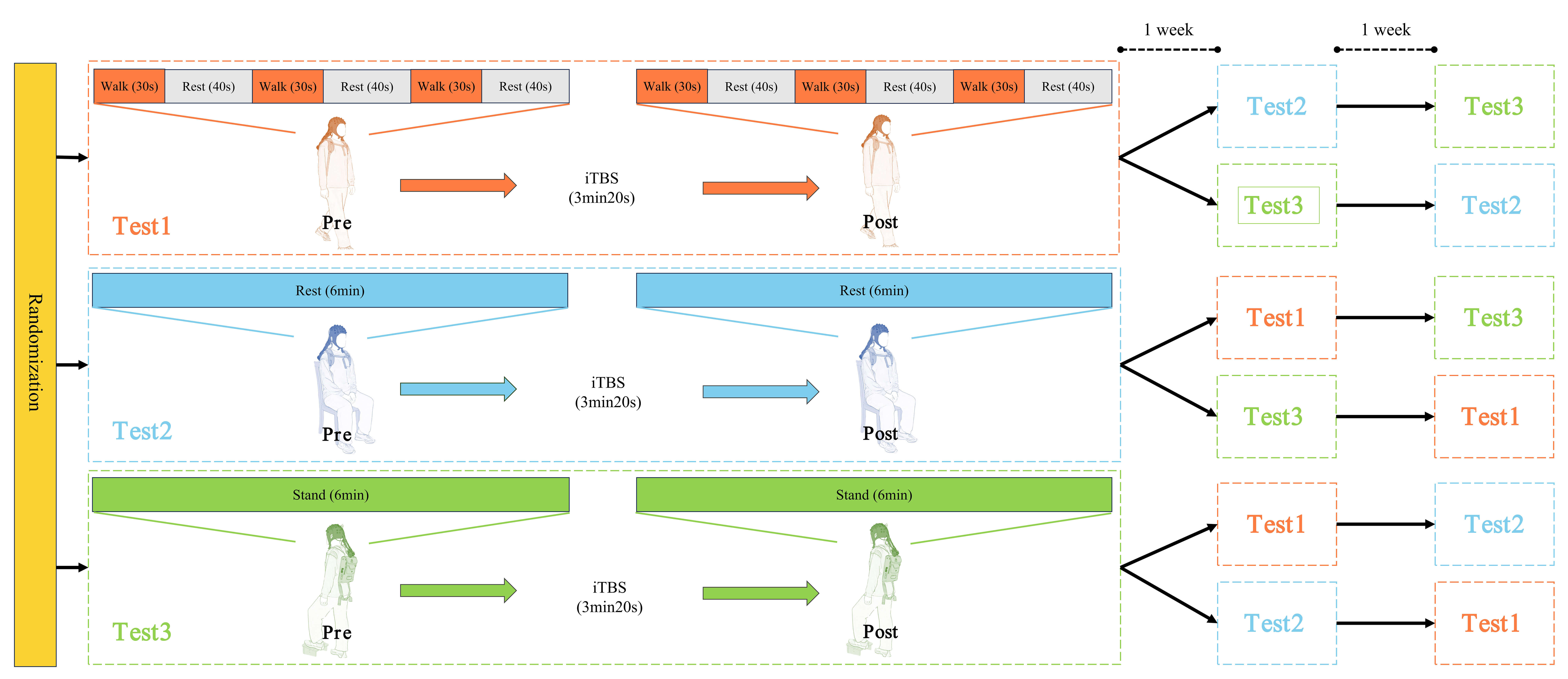 Fig. 1.
Fig. 1.
The diagram of procedures. iTBS, intermittent theta burst stimulation.
Cerebellar stimulation was delivered using a double-cone coil (Magneuro R480, Vishee Medical,
Nanjing, Jiangsu, China) connected to a 6-T magnetic stimulator, with stimulation
applied to the right cerebellar hemisphere in a single iTBS session. The coil
consisted of two conical windings with an outer diameter of 215 mm, an inner
diameter of 140 mm, and an inter-coil angle of 105°. The stimulation
site was defined anatomically as a point located 3 cm lateral to the midline and
1 cm inferior to the inion, corresponding to the external occipital protuberance.
The center of the coil was located on the target point. During the iTBS
application, the stimulator output intensity was set to 80% of the active motor
threshold (AMT) [20]. The AMT was defined as the lowest intensity that produced a
MEP of
The NirSmart fNIRS device (NirSmart-3000A, Danyang Huichuang Medical Equipment Co., Ltd., Zhenjiang, Jiangsu, China) was used in this study. Hemodynamic activity in the prefrontal, temporal, and parietal cortices during walking was recorded using near-infrared light at wavelengths of 730 and 850 nm, with a sampling rate of 11 Hz. A total of 46 measurement channels were arranged across the cortical surface, consisting of 19 light sources and 27 detectors. The three-dimensional positions of all optodes were acquired using an electromagnetic digitization system (Patriot, Polhemus, Colchester, VT, USA; https://polhemus.cn/motion-tracking/all-trackers/patriot/). Channel locations were subsequently transformed into Montreal Neurological Institute (MNI) space and mapped onto a standard MNI brain template. Each channel was assigned to a functional region according to the Brodmann area with the highest proportional overlap. The distance between adjacent optodes was 3 cm. Further details regarding channel positions and the division of ROIs are provided in Fig. 2 and Table 1.
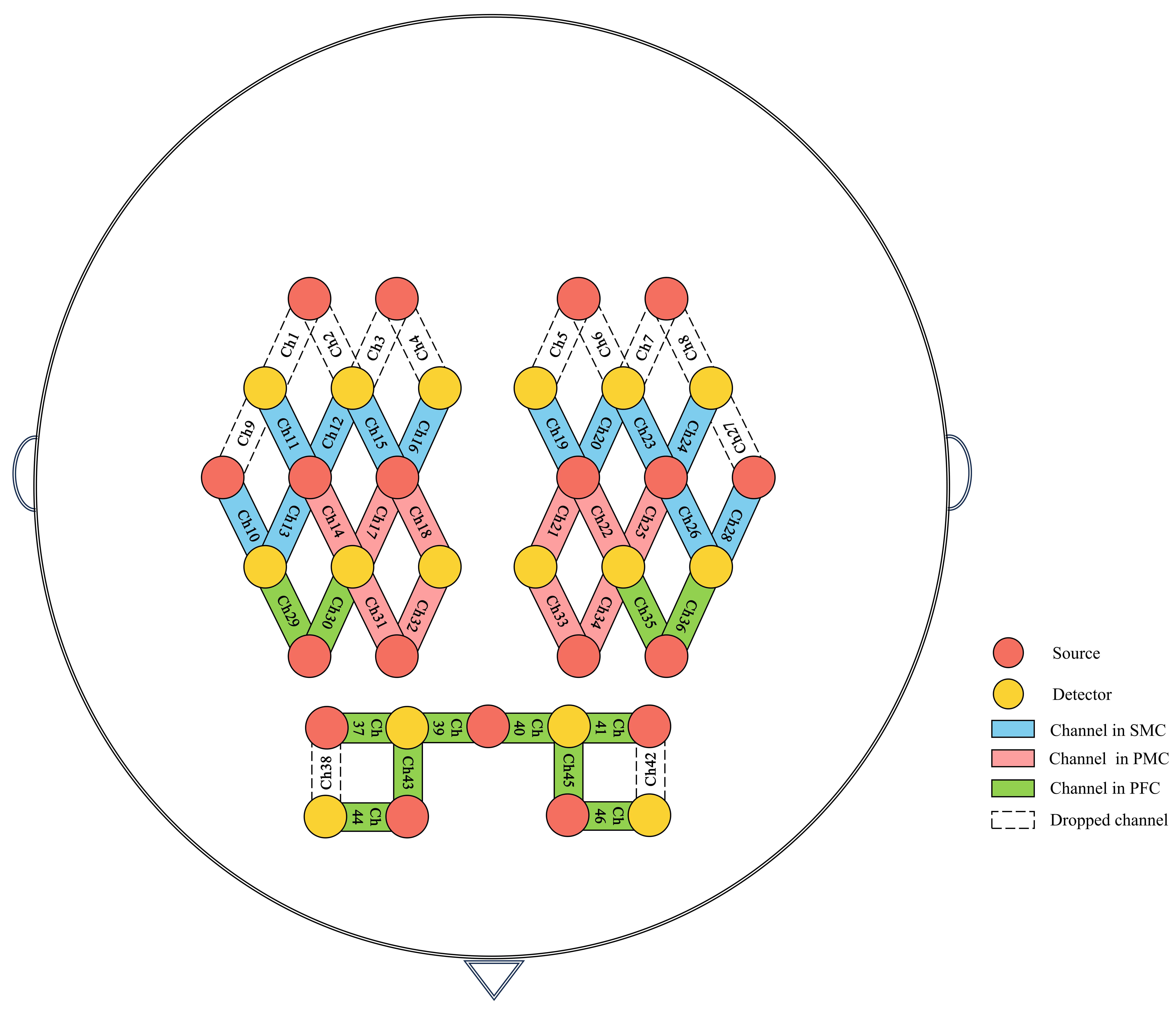 Fig. 2.
Fig. 2.
The attribution of channels and division of 6 ROIs. Pink channels belong to the SMC, yellow channels belong to PMC/SMA, and green channels belong to the PFC. ROIs, regions of interest; SMC, sensorimotor cortex; PMC, premotor cortex; SMA, supplementary motor area; PFC, prefrontal cortex; ch, channel.
| ROI | Brodmann area | Channel |
| R-SMC | 1 | 11 |
| R-SMC | 3 | 12 |
| R-SMC | 4 | 10, 13, 15, 16 |
| L-SMC | 1 | 24 |
| L-SMC | 3 | 23 |
| L-SMC | 4 | 19, 26, 28 |
| R-PMC/R-SMA | 6 | 14, 17, 18, 31, 32 |
| L-PMC/L-SMA | 6 | 21, 22, 25, 33, 24 |
| R-PFC | 9 | 29, 30 |
| R-PFC | 10 | 39, 43, 44 |
| R-PFC | 46 | 37 |
| L-PFC | 9 | 35, 36 |
| L-PFC | 10 | 40, 45, 46 |
| L-PFC | 46 | 41 |
R-, right; L-, left.
We implemented a series of preprocessing steps on the fNIRS data using Homer2 (Homer2_UI v1.5, Massachusetts General Hospital/Harvard Medical School, Charlestown, MA, USA). Raw fNIRS signals were first converted into optical density data. Motion-related artifacts were then corrected using a cubic spline interpolation approach, with thresholds set at 6.0 for standard deviation and 0.5 for signal amplitude. To reduce physiological drift and high-frequency noise while retaining task-relevant hemodynamic components, the data were band-pass filtered between 0.01 and 0.1 Hz. The processed optical density signals were subsequently transformed into changes in hemoglobin concentration using the modified Beer–Lambert law, with a differential path length factor of 6 [22, 23].
For the walking task, 30 seconds time window of changes in Oxy-Hb
(
For the resting-state and unilateral support tasks, we performed functional connectivity (FC) analysis and graph theory analysis. FC in brain imaging refers to the temporal correlation between spatially remote neural structures [25]. The 34 channels were grouped into six ROIs, and the Oxy-Hb values from all channels within each ROI were averaged. These ROI-averaged Oxy-Hb values were then used to calculate the Pearson correlation coefficients between each pair of ROIs, which served as the FC values. Graph theory analysis is a data-driven approach that models the brain as a graph composed of nodes and edges. By calculating global and local network topological properties, it reveals changes in the brain’s network topology [26].
Global properties of the brain network were computed using the GRETNA V2.0
software package (Imaging Connectomics Lab, Beijing Normal University, Beijing,
China). The sparsity range was set between 0.12 and 0.40, with an interval of
0.01 and an iteration of 100 times. We used the built-in function of the GRETNA
toolbox (gretna_get_rmax) to estimate the lower bound of sparsity based on the
34 used channels, which was determined to be 0.12, while the upper bound was set
according to a previous study [27]. The area under the curve for each network
matrix was calculated. The global properties analyzed included: (1) Global
network efficiency metrics, specifically global efficiency (Eglob) and local
efficiency (Eloc); (2) Small-world parameters, including clustering coefficient
(Cp), characteristic path length (Lp), and normalized small-worldness
(
Our fNIRS data were mainly analyzed using MATLAB R2015b (The MathWorks, Inc., Natick, MA, USA). The mixed analysis of variance (ANOVA) was applied to all results. First, regarding normality, the results of cortical activation and global properties network metrics were generally consistent with a normal distribution, with only a small number of brain regions showing mild deviations from normality. Given that the sample size in each group exceeded 30 participants, the statistical analyses are considered robust to moderate violations of the normality assumption. Second, for FC measures, more than half of the connections initially exhibited non-normal distributions. Therefore, Fisher’s z transformation was applied to the FC data prior to statistical analysis. After transformation, normality was assessed using the Shapiro–Wilk test, and the vast majority of FC values were found to conform to the normality assumption. In addition, homogeneity of variance was evaluated using Levene’s test, and the results indicated that the assumption of equal variances was generally satisfied across all measures. When multiple comparisons were made, the Bonferroni method was used for correction, adjusting the significance p-value to 0.05/n (where n is the number of comparisons). When significant main effects were found, further tests were conducted on the difference indicators. Paired t-tests were used within groups, and independent samples t-tests were used between groups, with the Bonferroni method applied to correct for multiple comparisons.
After a single session of iTBS effects on cortical activation, significant
statistical differences were observed in several ROIs. Due to 6 ROIs, the
significance level for the mixed ANOVA was set at 0.05/6. The results of
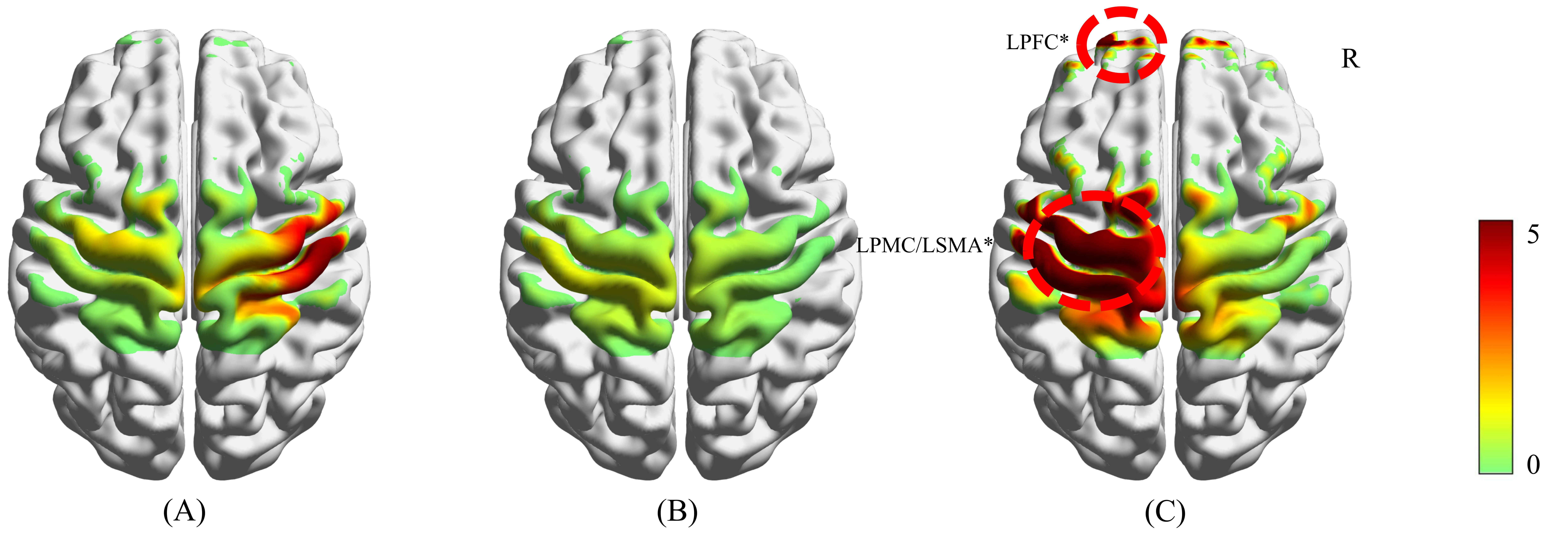 Fig. 3.
Fig. 3.
F-values of mixed analysis of variance (ANOVA) of
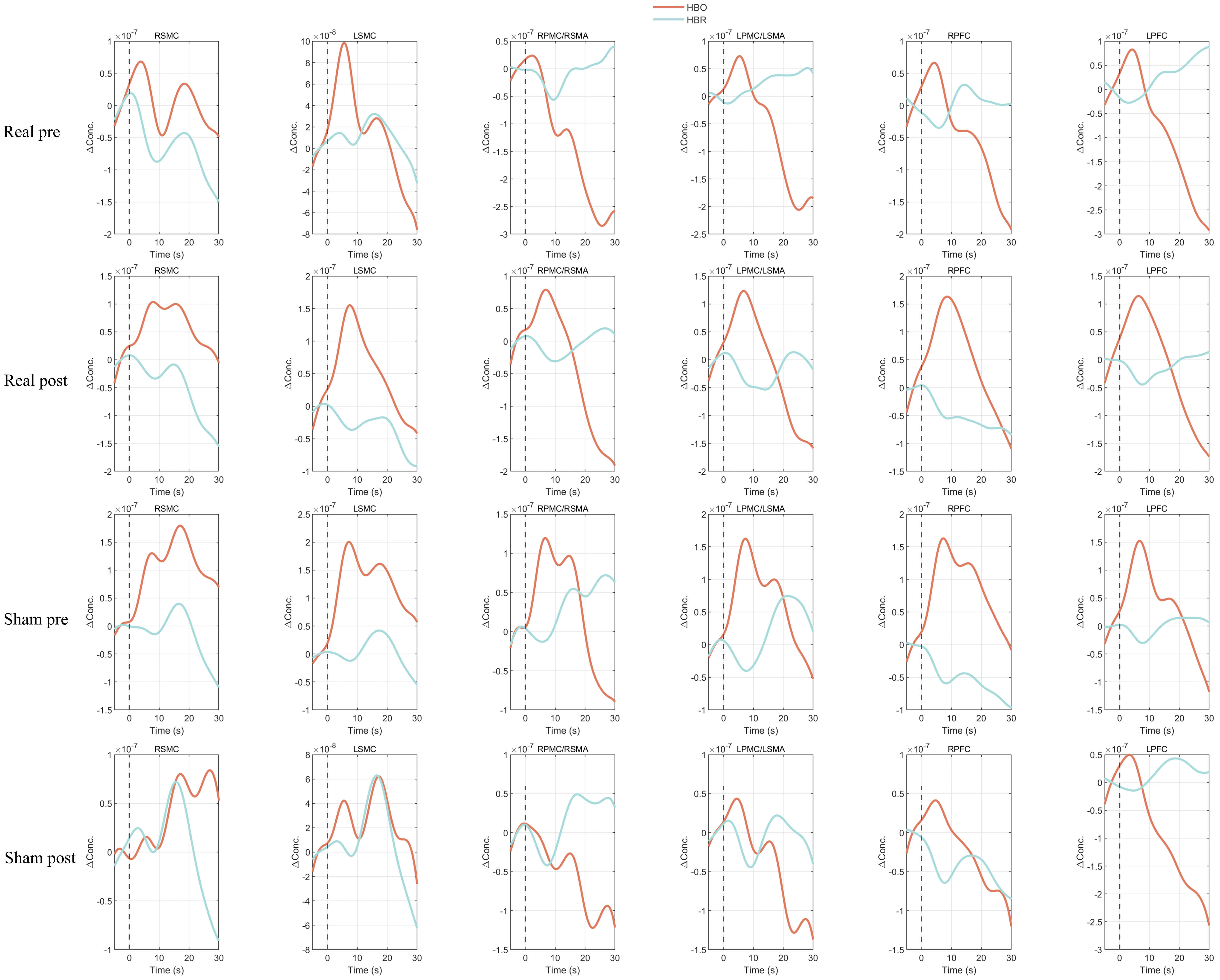 Fig. 4.
Fig. 4.
| ROI | Pre (n = 40) | Post (n = 39) | t | p | |
| LPMC/LSMA | Real | –0.011 |
–0.004 |
–1.218 | 0.231 |
| Sham | 0.006 |
–0.012 |
2.573 | 0.014 | |
| t | –1.381 | 0.661 | |||
| p | 0.171 | 0.511 | |||
| LPFC | Real | –0.018 |
–0.002 |
–2.427 | 0.020 |
| Sham | 0.002 |
–0.014 |
2.412 | 0.021 | |
| t | –1.541 | 1.162 | |||
| p | 0.127 | 0.249 |
No significant differences were observed following either real or sham iTBS intervention from the FC and graph theory perspectives. This suggests that neither real nor sham stimulation led to measurable changes during the resting task. The details are shown in Fig. 5, Fig. 6, and the Supplementary Table 3 and Table 4.
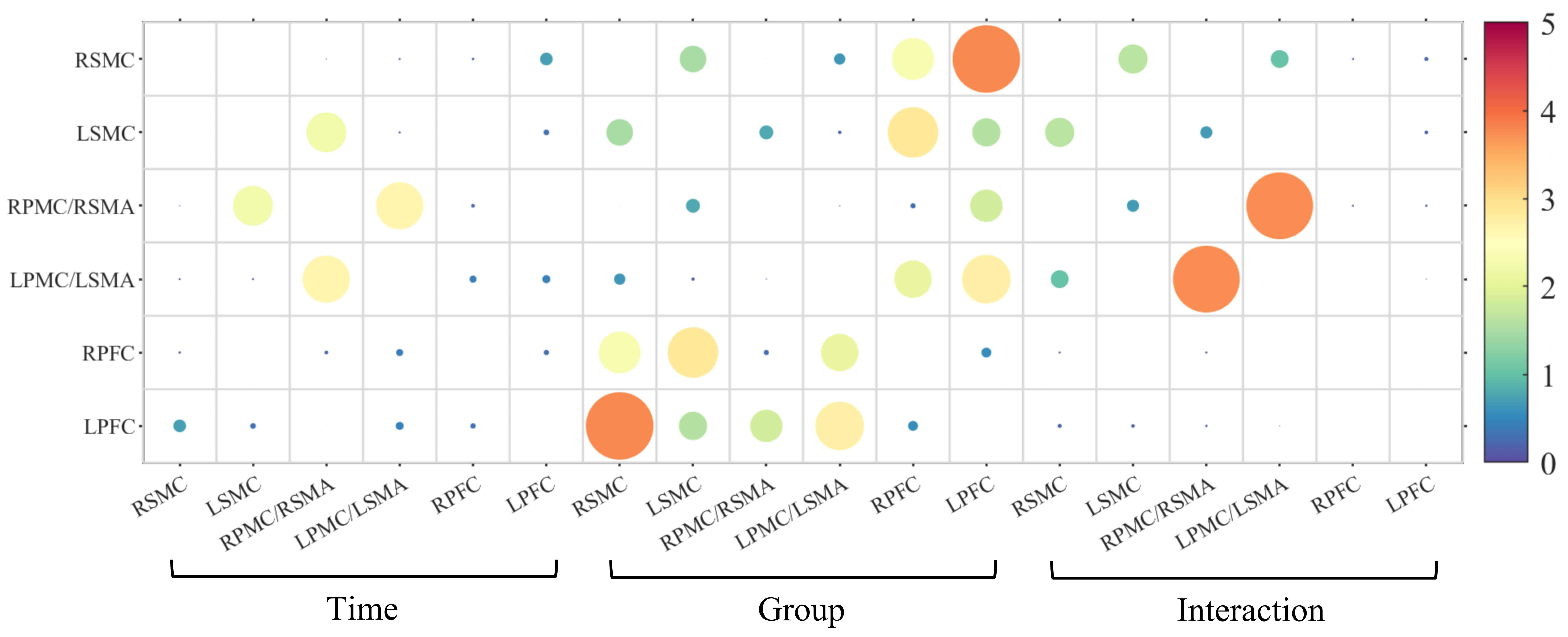 Fig. 5.
Fig. 5.
Functional connectivity (FC) of F-values in mixed ANOVA for the resting task.
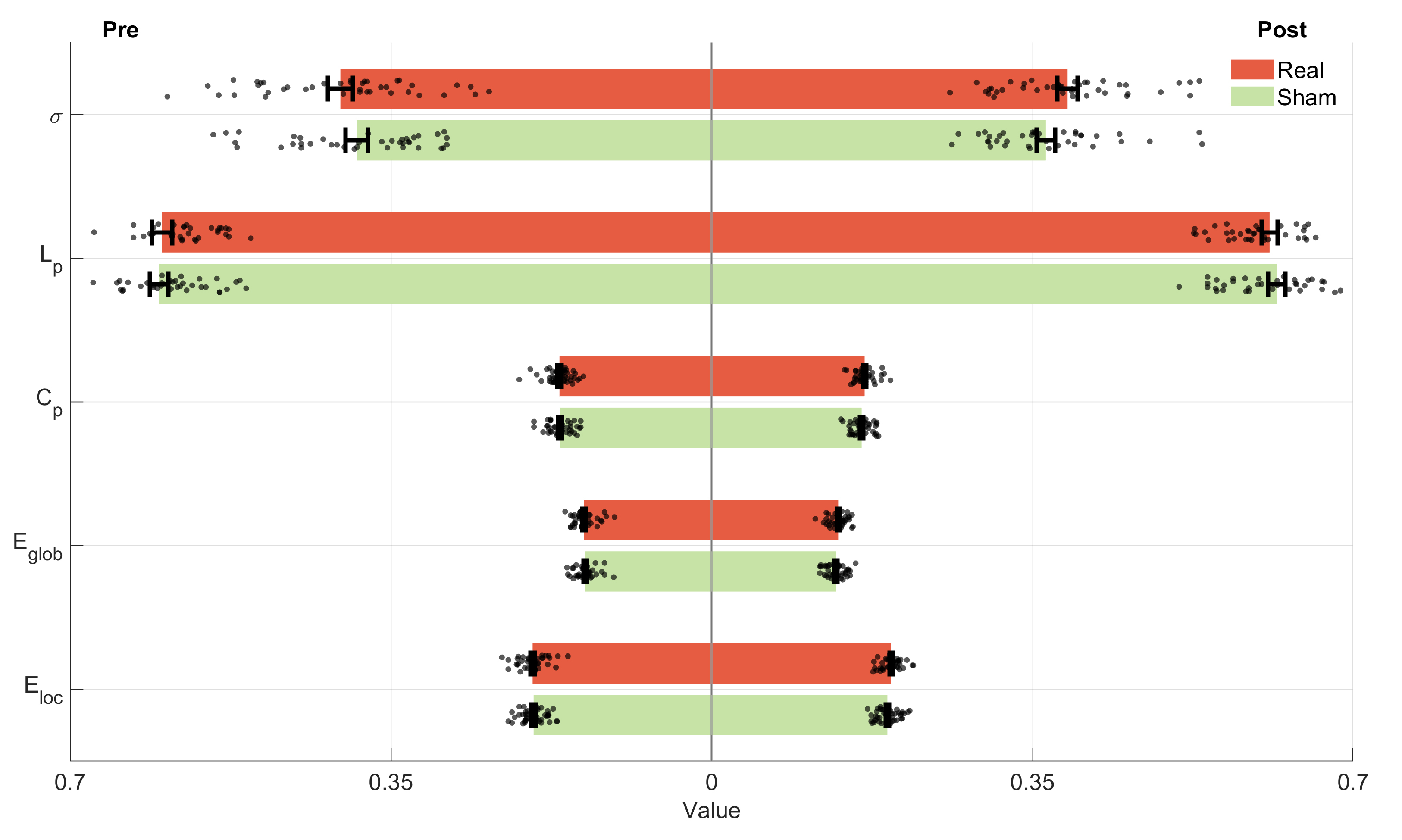 Fig. 6.
Fig. 6.
The graph theory metrics of pre- and post- iTBS for the resting task.
The FC analysis results showed no significant time, group, or interaction
effects between the two groups of subjects (see Fig. 7 and Supplementary Materials Table 5). Positive results were
observed only after real iTBS when analyzed using graph theory, and the
significance level for the mixed ANOVA was set at 0.05. The graph theory analysis
results (see Fig. 8 and Supplementary Materials Table 6) revealed significant time effects in the two groups for Lp
(F(1,77) = 5.220, p = 0.025,
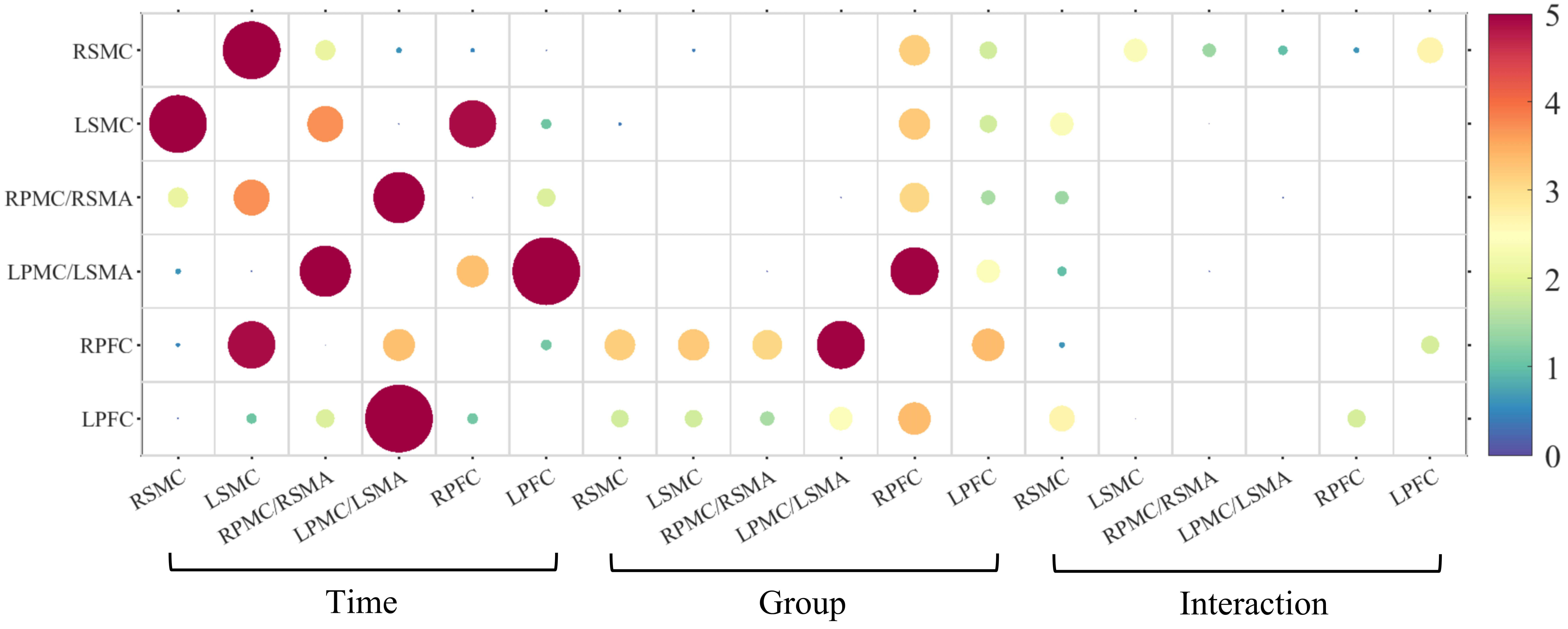 Fig. 7.
Fig. 7.
FC of F-values in mixed ANOVA for the unilateral support task.
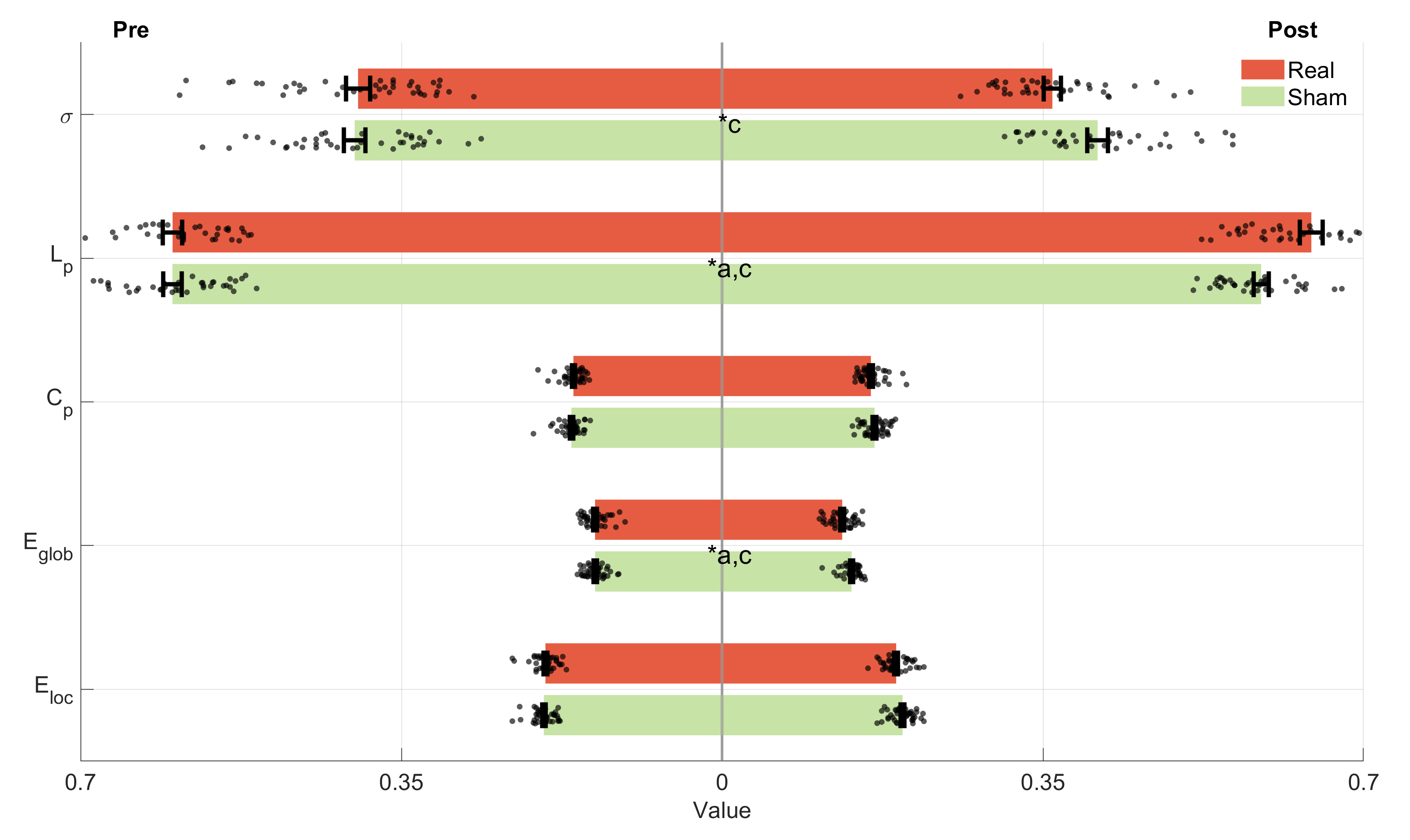 Fig. 8.
Fig. 8.
The graph theory metrics of pre- and post- iTBS for the unilateral support task. *a, intervention factor with a significant difference, *b, group factor with a significant difference; *c, interaction factor with a significant difference. Cp, clustering coefficient; Eloc, local efficiency.
| Pre (n = 40) | Post (n = 39) | t | p | ||
| σ | Real | 0.397 |
0.360 |
3.004 | 0.005* |
| Sham | 0.401 |
0.410 |
–0.684 | 0.498 | |
| t | –0.211 | –3.400 | |||
| p | 0.833 | 0.001* | |||
| Lp | Real | 0.600 |
0.643 |
–4.340 | |
| Sham | 0.600 |
0.588 |
1.207 | 0.235 | |
| t | –0.008 | 3.652 | |||
| p | 0.993 | ||||
| Eglob | Real | 0.139 |
0.131 |
4.761 | |
| Sham | 0.139 |
0.141 |
–1.381 | 0.175 | |
| t | 0.050 | –3.900 | |||
| p | 0.960 |
* with significant difference. Lp, characteristic path length; σ, normalized small-worldness; Eglob, global efficiency.
This study aimed to investigate whether cerebellar iTBS induces a cortical response, thereby validating the feasibility of a closed-loop cerebellar-cerebral cortex circuit using NIBS techniques. Significant differences in graph theory metrics were observed before and after the unilateral support task, while no significant differences were found during the walking and resting-state tasks. In contrast, the sham stimulation group exhibited no differences across any of the tasks.
During the walking task, we found significant interaction factors in LPFC and
LPMC/LSMA. However, further simple effect analysis of these two brain regions
revealed no significant differences. This pattern likely reflects diverging
trends between groups, which is sufficient to produce a significant interaction
term but insufficient to yield statistically significant simple effects under
conservative multiple-comparison correction. From the results, we observed that
the real stimulation group showed an increasing trend in
An interesting phenomenon observed during walking, regardless of whether the stimulation was real or sham, was that participants exhibited smaller changes in cortical blood flow compared to the resting state. It is well known that the centers controlling walking are primarily governed at lower levels of the neuroaxis, including brainstem regions, spinal pattern-generating circuits, and cerebellar circuits. These structures generate patterns of intermuscular and interlimb coordination, promoting the automation of walking and minimizing reliance on cortical control [33]. Disruption of these structures can lead to significant consumption of cortical resources, as demonstrated in stroke and Parkinson’s disease patients [35]. In the study by Koenraadt et al. [34], it was found that participants exhibited decreased activation in the SMA and PFC during normal walking compared to resting states. However, during precision stepping, activation in the SMA and PFC increased compared to resting states. When walking requires more control, the activation of motor control-related cortices, such as the PFC and SMA, also correspondingly increases. In our results, the significant interaction factor in activation was particularly evident in the LPFC and LPMC/LSMA, further suggesting that the changes in activation during normal walking compared to resting states may be closely related to walking automation. Additionally, even though we emphasized maintaining natural conditions during data collection, it remains uncertain whether the collected resting state truly represents rest. The observer effect also plays a significant role in this context. This factor may also lead to participants exhibiting higher activation during resting states.
In the balance task, we designed a unilateral support task to engage
participants in postural control. We observed an increase in
For the resting-state results, we did not observe any positive outcomes at the levels of FC or graph theory metrics. One possible reason for this could be the relatively low spatial resolution of fNIRS, which may have limited our ability to detect changes in FC within the observed brain regions. Wang et al. [11] investigated changes in swallowing-related brain regions using resting-state fMRI after a single session of iTBS on the cerebellar hemisphere. They found changes in the middle temporal gyrus, precuneus, and superior frontal gyrus that could not be detected by fNIRS. Additionally, it is not only our study that failed to find significant results in the resting state. Thomson et al. [43] also reported similar findings. They suggested that the hemodynamic response induced by sub-threshold repetitive TMS in a single trial is generally very mild and may be masked when averaging epochs, which presents a challenge for fNIRS-based measurements. Regarding FC during the single-leg support task, no significant differences were observed. Currently, no studies are exploring whether cerebellar iTBS can induce changes in functional connectivity between brain regions. However, we speculate that there may be functional changes between the cerebellum and the cerebral cortex, which warrant further investigation using fMRI in future research.
Our study still has certain limitations. First, there are inherent limitations of near-infrared spectroscopy, which cannot detect subcortical structures that also participate in the cerebellar-cortical circuitry. Second, this study only intervened with healthy individuals, and no research has confirmed that cerebellar NIBS improves balance function in healthy people. We assessed the balance function (Berg Balance Scale) of all participants before and after the intervention and found both full scores without any differences, so we did not mention this result. We speculated that the scale we selected has low sensitivity among healthy individuals. Future experiments should use more precise balance measures for further observation. Third, leg dominance was not formally assessed, and all participants performed the unilateral support task using the right leg. Although this approach ensured task standardization, future studies should consider evaluating lower-limb dominance and counterbalancing the supporting leg to examine potential laterality effects. Fourth, we did not perform within-task time-course analyses (e.g., early vs. late epochs) because subdividing the recording may reduce the robustness of connectivity and graph metrics and would require concurrent behavioral/physiological fatigue measures for reliable interpretation. Furthermore, in recent years, fNIRS systems equipped with short-separation channels have been increasingly adopted, as they provide important advantages for improving signal specificity and reducing motion-related and systemic physiological artifacts. The incorporation of such hardware designs facilitates more accurate separation of cortical hemodynamic responses from superficial and non-neuronal signals. Future studies may therefore consider employing fNIRS systems with short-separation channels to further enhance data quality during dynamic motor tasks.
This study provides evidence that cerebellar iTBS can induce cortical responses, supporting the hypothesis of a cerebellar-cerebral cortex circuit involved in balance regulation. The changes in brain network properties during the unilateral support task indicate that iTBS may disrupt the efficient balance of brain networks in healthy individuals. However, no significant effects were observed during the walking and resting tasks, which may be due to the limitations of fNIRS in detecting subtle functional connectivity changes. Overall, these findings contribute to our understanding of the neural mechanisms underlying cerebellar NIBS and its potential therapeutic applications in improving balance. Further research is required to explore the clinical relevance of these findings.
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
CG and AG contributed equally to this work. CG, AG, TW, and SZ conceived and designed the study. CG, AG, QW, JS, CK, and YS performed the research, including participant recruitment, assessment, and data collection. CG and AG analyzed and interpreted the data and drafted the manuscript. QW, JS, CK, and YS contributed to data curation and provided critical feedback on the manuscript. TW and SZ supervised the study, provided important intellectual input, and critically revised the manuscript for important intellectual content. TW and SZ also took primary responsibility for the overall coordination of the study and communication during the research and publication process. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This study was approved by the Changzhou De’an Hospital Human Ethics Committee (CZDALL-2023-013) and registered with the China Clinical Trial Registration Center (ChiCTR2300077916) on November 23, 2023. The research was conducted in accordance with the principles embodied in the Declaration of Helsinki and in accordance with local statutory requirements. Prior to participation, all participants provided written informed consent and received financial compensation.
We are sincerely grateful to all the researchers (excluding the co-authors) and participants for their valuable contributions to this study.
This work was supported by the National Natural Science Foundation of China (NSFC) (Grant No. 82302882) and the Key Medical Discipline Project of Jiangsu Province (No. XK201110).
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/JIN47653.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.








