1 College of Acupuncture, Moxibustion, Tuina and Rehabilitation, Hunan University of Chinese Medicine, 410208 Changsha, Hunan, China
2 Department of Traditional Chinese Medicine, The Second Affiliated Hospital, University of South China, 421001 Hengyang, Hunan, China
3 Hengyang Medical College, University of South China, 421001 Hengyang, Hunan, China
Abstract
Although pressing intervention is widely used clinically to alleviate pain associated with chronic myofascial trigger points (MTrPs), the mechanisms by which it modulates pain via sensory nerves remain unclear. This study aimed to investigate the effects of pressing intervention on transient receptor potential vanilloid 1 (TRPV1) channels in sensory nerves and to explore its potential analgesic mechanisms.
Twenty-six male Sprague-Dawley rats were randomly divided into a blank group (n = 6) and a model establishment group (n = 20). Chronic MTrPs were induced in the model group by blunt strike combined with eccentric exercise. Eighteen successfully prepared were randomized into model, press, and press + capsaicin (TRPV1 agonist) groups (n = 6 per group). The press group received local pressing at MTrPs every two days for seven sessions, while the press + capsaicin group received intraperitoneal capsaicin prior to pressing. Pressure pain threshold (PPT) and soft tissue tension (STT), with STT quantified by the displacement at a loading force of 0.2 kg (D0.2), were measured before and after intervention. After treatment, the MTrPs tissue and the L2–L4 dorsal root ganglia (DRG) were collected for analysis. Skeletal muscle morphology was observed by hematoxylin-eosin (HE) staining. Inflammatory mediators in MTrPs tissue were measured by enzyme-linked immunosorbent assay (ELISA). TRPV1 protein expression in DRG was detected by Western blot. Immunofluorescence was used to detect TRPV1 on CGRP⁺ fibers in MTrPs and to quantify TRPV1+/c-Fos+ cells in DRG.
Compared with the blank group, the model group showed reduced pain threshold, increased soft tissue tension, disorganized myocytes, inflammatory infiltration, elevated TRPV1 in nerve endings, increased interleukin-1β (IL-1β), prostaglandin E2 (PGE2), calcitonin gene-related peptide (CGRP), substance P (SP), decreased interleukin-10 (IL-10), and upregulated TRPV1 and TRPV1+/c-Fos+ cells in DRG (p < 0.01 or p < 0.05). Pressing reversed these changes, restored the pain threshold, soft tissue tension, and myocyte morphology, reduced TRPV1 and pro-inflammatory mediators, increased IL-10, and downregulated TRPV1 and TRPV1+/c-Fos+ cells in DRG (p < 0.01 or p < 0.05). These effects were partially blocked by capsaicin, as the press + capsaicin group exhibited reversed effects compared with pressing alone (p < 0.01 or p < 0.05).
Pressing intervention increases the mechanical pain threshold and improves soft tissue tension in rats with MTrPs. The underlying mechanism may be associated with alleviating local inflammation, modulating the TRPV1 channel in unmyelinated C-type sensory nerve fibers, and inhibiting TRPV1 expression, thereby reducing sensory nerve excitability.
Keywords
- pressure
- myofascial pain syndromes
- nociceptors
- TRPV1
- capsaicin
- dorsal root ganglia
Myofascial trigger points (MTrPs) are palpable hypersensitive nodules within taut bands of skeletal muscle, pathologically characterized by persistent contraction of sarcomeres [1]. Clinically, they manifest as spontaneous pain and motor dysfunction, often accompanied by autonomic symptoms such as anxiety and depression [2, 3]. Epidemiological studies indicate that approximately 85% of adults are affected by MTrPs, and 95% of chronic pain patients present with MTrPs [4], making them an important cause of musculoskeletal pain [5, 6].
Persistent sarcomere contraction and sterile inflammation are considered major
pathological bases of MTrPs formation [7]. Studies have shown that in active
MTrPs, proinflammatory mediators such as interleukin-1
The transient receptor potential vanilloid 1 (TRPV1) channel is a multimodal,
non-selective cation channel widely expressed in the peripheral nervous system of
rodents and humans, predominantly in small-diameter sensory neurons of the dorsal
root ganglion (DRG) and trigeminal ganglion, particularly in unmyelinated
peptidergic C fibers. C fibers are unmyelinated sensory fibers that detect and
convey nociceptive signals. TRPV1 is susceptible to can be activated by noxious
heat (
As a key molecule mediating nociceptive transmission and maintaining inflammatory states in sensory neurons, the precise role of TRPV1 in the pathogenesis of MTrPs remains to be fully elucidated. Previous studies have shown that local pressing effectively alleviates MTrP-related mechanical pain, improves soft tissue tension, and attenuates local inflammation [18, 19]. Accordingly, the present study sought to investigate the deactivation mechanism of pressing on MTrPs from the perspective of TRPV1 channel regulation in unmyelinated C-type sensory fibers.
Twenty-six specific pathogen-free (SPF) male Sprague-Dawley rats, aged 10–12 weeks and weighing 250–280 g, were provided by Hunan Silaike Jingda Experimental Animal Co., Ltd. (Animal Production License No.: SCXK (Xiang) 2024-0009, Changsha, Hunan, China). The animals were housed in the Experimental Animal Center of Hunan University of Chinese Medicine, where they were grouped three per cage and maintained under standardized conditions: Ambient temperature controlled between 24–26 °C, humidity 50–70%, and a 12-hour light-dark cycle. All animals had access to food and water ad libitum. All procedures strictly followed the relevant animal ethics guidelines and were approved by the Laboratory Animal Ethics Committee of Hunan University of Chinese Medicine (Ethics Approval No.: LL2021102004).
Isoflurane (R510-22-10, Ruiward Life Technology Co., Ltd, Shenzhen, Guangdong,
China); Capsaicin (HY-10448R, MedChemExpress, Shanghai, China); Paraformaldehyde
(G1101-500ML, Servicebio, Wuhan, Hubei, China); hematoxylin-eosin (HE)
Staining Kit (B1003, Baiqiandu, Wuhan, Hubei, China); RIPA Lysis Buffer (CW2333S,
CWBio, Taizhou, Jiangsu, China); BCA Protein Assay Kit (E-BC-K318-M, Elabscience,
Wuhan, Hubei, China); TRPV1 antibody (ab305299, Abcam, Cambridge, UK; western blot (WB): 1:1000;
immunofluorescence (IF): 1:100); c-Fos antibody (GB11069, Servicebio, Wuhan, Hubei, China, 1:500);
CGRP antibody (ab283568, Abcam, Cambridge, UK, 1:200); GAPDH antibody
(60004-1-Ig, Proteintech, Wuhan, Hubei, China, 1:50,000); Goat Anti-Rabbit IgG
(511203, ZEN-Bioscience, Chengdu, Sichuan, China; WB: 1:5000; IF: 1:400); Goat
Anti-Mouse IgG (511103, ZEN-Bioscience, Chengdu, Sichuan, China, 1:400); enzyme-linked immunosorbent assay (ELISA)
Kits for IL-1
Respiratory anesthesia machine (R550IE, Ruiward Life Technology Co., Ltd, Shenzhen, Guangdong, China); Treadmill (C100, Yusheng, Jinhua, Zhejiang, China); Press therapy instrument (Patent No. ZL202210366733.1, Self-developed device, Changsha, Hunan, China); Mechanical pressure pain threshold tester (ZS-miniTR, Zhongshi Dichuang, Beijing, China); Soft tissue tension tester (JZL-Ⅲ, Mingtong Shiji, Tianjin, China); Tissue embedding machine (JB-P5, Junjie, Wuhan, Hubei, China); Rotary microtome (RM2016, Laica, Shanghai, China); Tissue floating bath (KD-P, Kedi, Jinhua, Zhejiang, China); Multifunctional microplate reader (Cytation 5, BioTek, Winooski, VT, USA); Gel imaging system (Universal Hood II, Bio-Rad, Hercules, CA, USA); fluorescence microscope (MF43-N, Mshot, GuangZhou, Guangdong, China).
After seven days of acclimatization, 26 rats were randomly allocated into two groups: the blank group (n = 6) and the model group (n = 20). The model group underwent MTrPs induction while the blank group was maintained under standard conditions. Eighteen successfully modeled rats were then randomized into three groups (n = 6 per group): The model group (no intervention), the press group (receiving local press therapy), and the press + capsaicin group (receiving intraperitoneal capsaicin prior to press therapy).
The rat MTrPs model was established using a combination of blunt striking and eccentric exercise [20]. The modeling process consisted of an adaptation phase (1 week), a modeling phase (8 weeks), and a recovery phase (4 weeks) (Fig. 1).
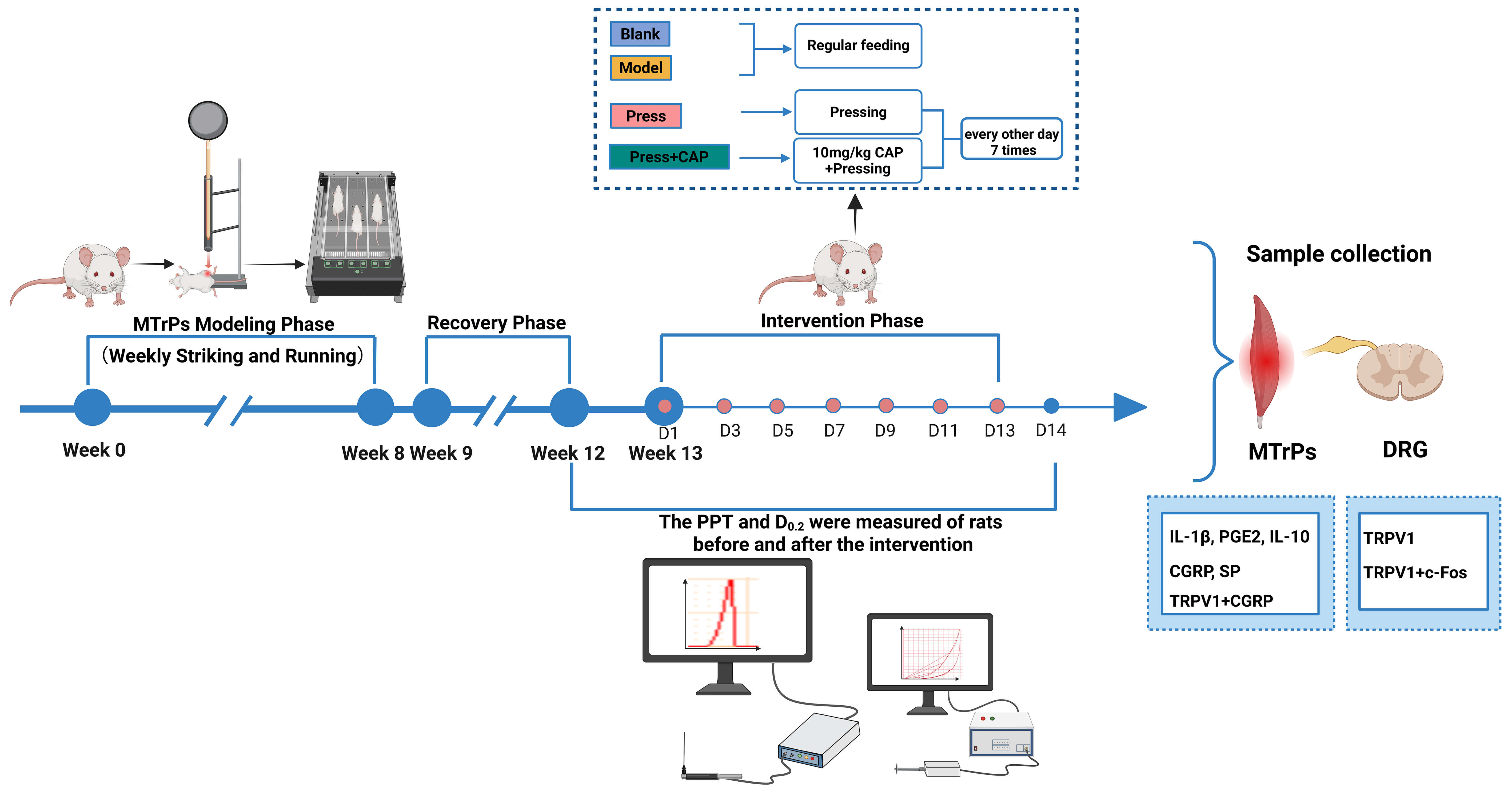 Fig. 1.
Fig. 1.
Study flowchart. MTrPS, myofascial trigger points; CAP,
capsaicin; PPT, pressure pain threshold; D0.2, displacement at a loading
force of 0.2 kg; DRG, dorsal root ganglia; IL-1
(1) Adaptation phase: Rats in the model group underwent progressive adaptive training on a motorized treadmill (0° incline, 16 m/min, 15 min/session, once every two days, three sessions in total) to acclimate to the exercise environment and reduce stress responses.
(2) Modeling phase: Each week began with a blunt strike on day one, followed by eccentric exercise on day two. On day one, following anesthesia with 3% isoflurane, and the projection area of the left vastus medialis (corresponding to the medial portion of the quadriceps muscle group on the anterior thigh) was identified by palpation and then marked according to a rat anatomical atlas. The marked site was secured on the striking platform beneath a blunt wooden rod, which was released from a 20 cm to deliver the strike impact. On day two, rats underwent eccentric exercise (–16° incline, 16 m/min, 90 min). Mechanical, auditory, or electrical stimuli were applied as needed to ensure completion of the 90 min eccentric running. Detailed procedures are provided in the Supplementary Material 1. At all other times, rats were maintained under standard feeding conditions.
(3) Recovery phase: All animals were housed in a regular environment and were not exposed to any form of intervention.
(4) Evaluation of the model: Experienced investigators palpated the same marked projection area of the left vastus medialis, which had been identified and marked according to the rat anatomical atlas. Positive criteria were defined as: Presence of a palpable taut band or nodule, elevated soft tissue tension, significantly reduced mechanical pain threshold, and a twitch response induced by electrode insertion. Successful modeling was determined based on a comprehensive assessment of these criteria [21].
After modeling, the blank and model groups were maintained under routine conditions without any intervention. Rats in the press group were anesthetized with 3% isoflurane, fixed in a supine position on a self-developed press therapy instrument platform, and received local vertical pressing over the marked left MTrPs site using a 0.5 cm diameter press head. The device was capable of simulating manual therapeutic manipulation and ensured that the rats received mechanical pressing stimulation with uniform frequency and force. The intervention parameters were based on previous studies [22, 23]: Applied force of 242 g, frequency of 10 presses/min, and duration of 7.5 min per session were administered once every two days for a total of seven sessions. The press + capsaicin group received an intraperitoneal injection of the TRPV1 agonist (capsaicin, 10 mg/kg) 30 minutes before each pressing intervention [24], with the pressing procedure identical to the press group (Fig. 1).
The PPT was assessed in all groups following model establishment and post-intervention. Rats were fixed in the supine position with the medial left hind limb exposed. An experienced experimenter palpated and marked the MTrPs site. The corresponding anatomical region on the same side was examined in the blank group. After the rats adapted and remained calm, vertical pressure was applied to the marked area with the probe for measurement. Local tissue tremor, muscle fasciculation, or paw withdrawal was considered a positive endpoint, and the corresponding pressure value was recorded [19]. Each rat was tested once every 10 minutes for a total of three trials, and the mean of the multiple readings was used as the definitive PPT.
The STT, quantified as the displacement at a loading force of 0.2 kg (D0.2), was assessed in all groups following model establishment and post-intervention. The fixation and positioning of rats were the same as for PPT testing. Vertical pressure was applied to the marked site with a soft tissue tension tester, with loading and unloading each maintained for three seconds to generate a load-displacement curve [19, 25]. Each rat was tested once every 10 minutes for three trials, and the mean value was recorded as the final tension.
After the final PPT and STT measurements, rats were euthanized. The detailed procedure was as follows: the animals were placed in an anesthesia chamber, and once they lost consciousness, the concentration of isoflurane was increased to 5% and maintained for more than 5 minutes until respiration ceased, pupillary dilation occurred, and the corneal reflex disappeared. Subsequently, the left medial vastus muscle (trigger point region or the corresponding site in the blank group) and L2–L4 DRG were collected. Six muscle specimens per group were each split into two portions: One fixed in 4% paraformaldehyde for HE staining or immunofluorescence, and the other snap-frozen in liquid nitrogen and preserved at –80 °C for ELISA. DRG tissues from three rats were per group were fixed for immunofluorescence, and the remaining three were snap-frozen for Western blot analyses.
Tissue samples from the trigger points were fixed in 4% paraformaldehyde for 48 h, processed by dehydration, through a graded ethanol dehydration series, embedded in paraffin, and sectioned into 4 µm slices. These slices were subsequently HE stained, dehydrated, and mounted using neutral resin. Morphological features of skeletal muscle cells were assessed by light microscopy.
Equal masses of frozen trigger point tissue were weighed, minced, and
homogenized using a cryogenic grinder. The homogenate was kept on ice, then
centrifuged, and the supernatant subsequently collected for analysis. ELISA kits
were equilibrated to room temperature (20 °C). Standard solutions were
prepared via serial dilution. All procedures, including sample addition, washing,
and incubation, were performed following the manufacturer’s protocol. The
absorbance was then measured at 450 nm using a multimode microplate reader.
Concentrations of IL-1
Frozen DRG samples were lysed and homogenized, and the protein concentrations were determined using a BCA assay kit. Denatured protein samples were prepared. Equal amounts of denatured protein samples were sequentially subjected to electrophoresis (160 V, 50 min), membrane transfer (300 mA, 30 min), and blocking (1 h at room temperature). Membranes were incubated with primary antibodies (TRPV1 1:1000; GAPDH 1:50,000) at 4 °C overnight, followed by incubation with an HRP-linked secondary antibody (1:5000) for 1 h at room temperature, with TBST washes three times after each incubation. Protein bands were visualized using ECL chemiluminescence. Band intensities were analyzed using ImageJ software (1.54P, NIH, Bethesda, MD, USA), and relative TRPV1 expression was calculated as the ratio of the target protein to the internal reference protein.
After fixation, trigger point tissues and DRGs were paraffin-embedded, sectioned, and subjected to dewaxing, antigen retrieval, and washing. Sections were then incubated with 3% hydrogen peroxide for 25 min and blocked with 10% goat serum for 30 min, followed by overnight incubation at 4 °C with primary antibodies against TRPV1 (1:100), CGRP (1:200), or c-Fos (1:500). After washing, HRP-conjugated secondary antibodies (goat anti-rabbit or goat anti-mouse, 1:400) were applied for 50 min at room temperature, and signal amplification was performed using Tyramide Signal Amplification (TSA) reagent for 20 min. Nuclei were counterstained with 4′,6-Diamidino-2-phenylindole dihydrochloride (DAPI), and sections were mounted with antifade medium. Fluorescent images were acquired using a fluorescence microscope, with three random fields selected per section. The colocalized fluorescence area of CGRP with TRPV1 and the number of TRPV1/c-Fos double-positive cells were quantified using ImageJ software.
Data were analyzed with SPSS 26.0 (IBM Corp., Armonk, NY, USA), and continuous
variables are displayed as mean
The results are shown in Fig. 2 and indicate that, before the intervention, the
model, press, and press + capsaicin groups all significantly
reduced mechanical pain thresholds compared with the blank group (p
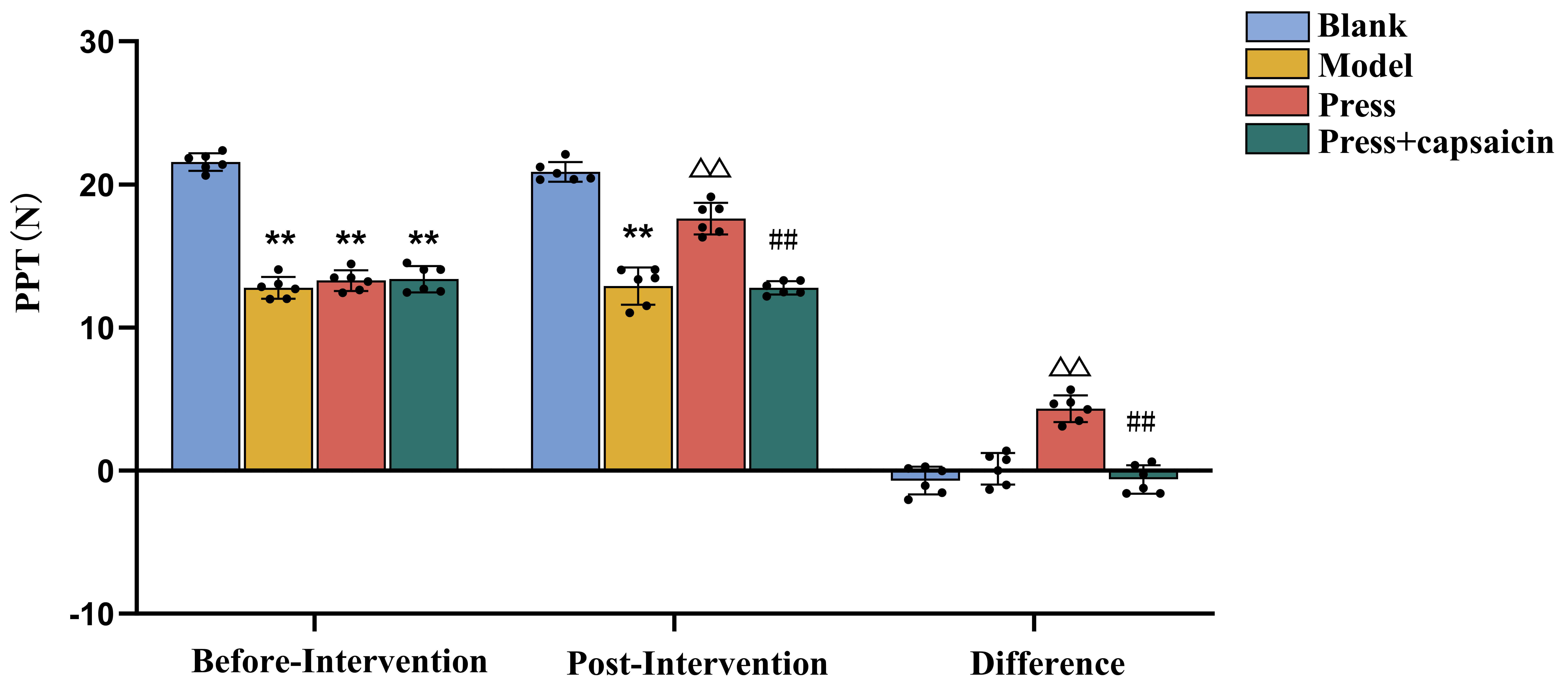 Fig. 2.
Fig. 2.
Local PPT at trigger points before and after
intervention in each group, based on assessments a mechanical pressure pain
threshold device (n = 6 per group). **p
The results are given in Fig. 3. The D0.2 value was inversely correlated
with soft tissue tension, a higher D0.2 value indicated lower soft tissue
tension. Before intervention: Compared with the blank group, the model, press,
and press + capsaicin groups all showed significantly reduced D0.2 values
(p
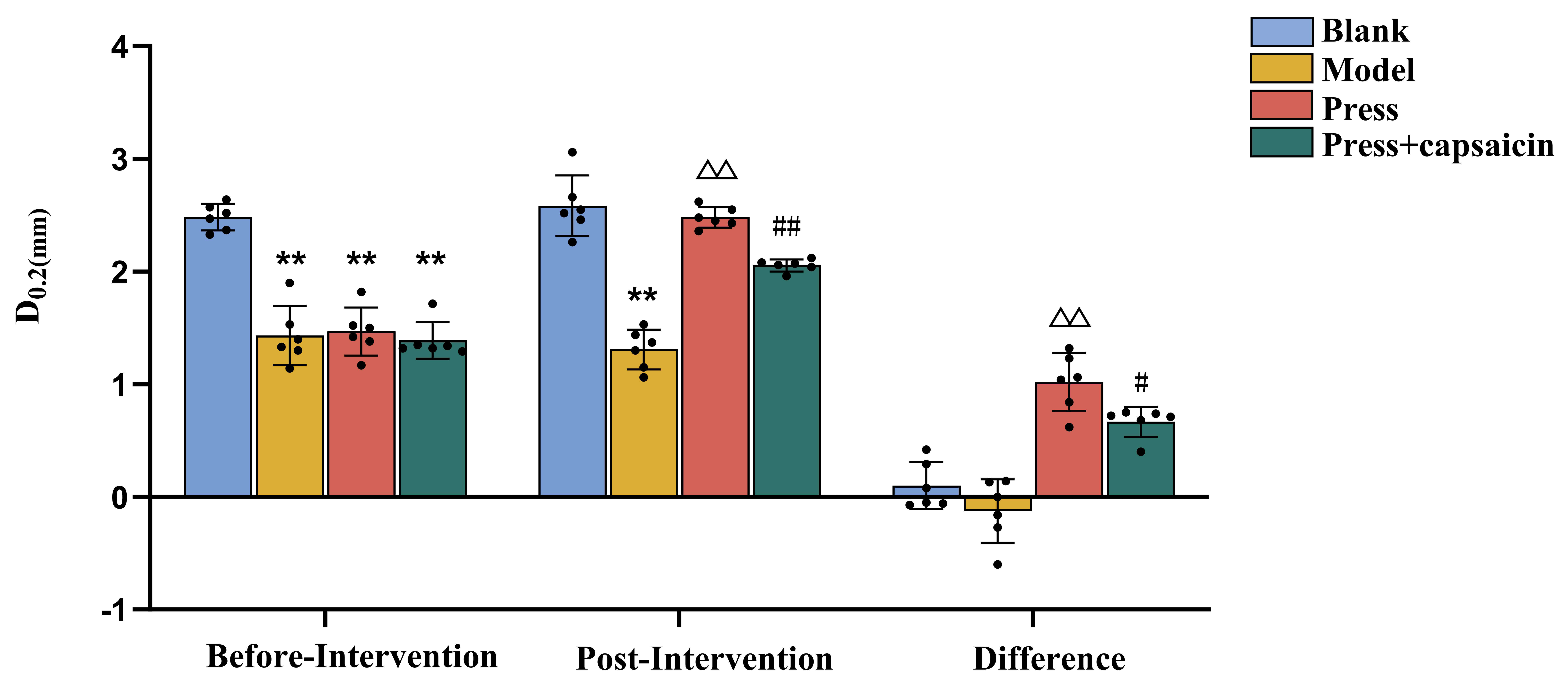 Fig. 3.
Fig. 3.
Local STT D0.2 at trigger points before and after
intervention in each group determined by the soft tissue tension tester (n = 6
per group). **p
Results are given in Fig. 4. In the blank group, skeletal muscle cells were polygonal with clear boundaries and uniform size, nuclei aligned along the sarcolemma, cells tightly and regularly arranged across the field, and cytoplasm evenly stained. In the model group, muscle cells were significantly enlarged and mostly round, with some nuclei displaced centrally, disorganized and loosely arranged cells, deeply stained cytoplasm in some areas. In addition, inflammatory cells were identified based on their morphological features, including nuclear shape, chromatin density, cytoplasmic volume, and staining intensity, revealing extensive inflammatory cell infiltration in the tissue. In the press group, muscle cell morphology was largely restored to regular polygons, cytoplasm was evenly stained, nuclei returned to peripheral alignment, cells were orderly arranged, and inflammatory infiltration was reduced. In the press + capsaicin group, the morphology resembled the model group, with round, deeply stained muscle cells, loose arrangement, and increased inflammatory cell infiltration. These results suggest that press intervention helps restore muscle cell morphology at MTrPs and mitigates inflammation, whereas this effect is weakened by capsaicin stimulation.
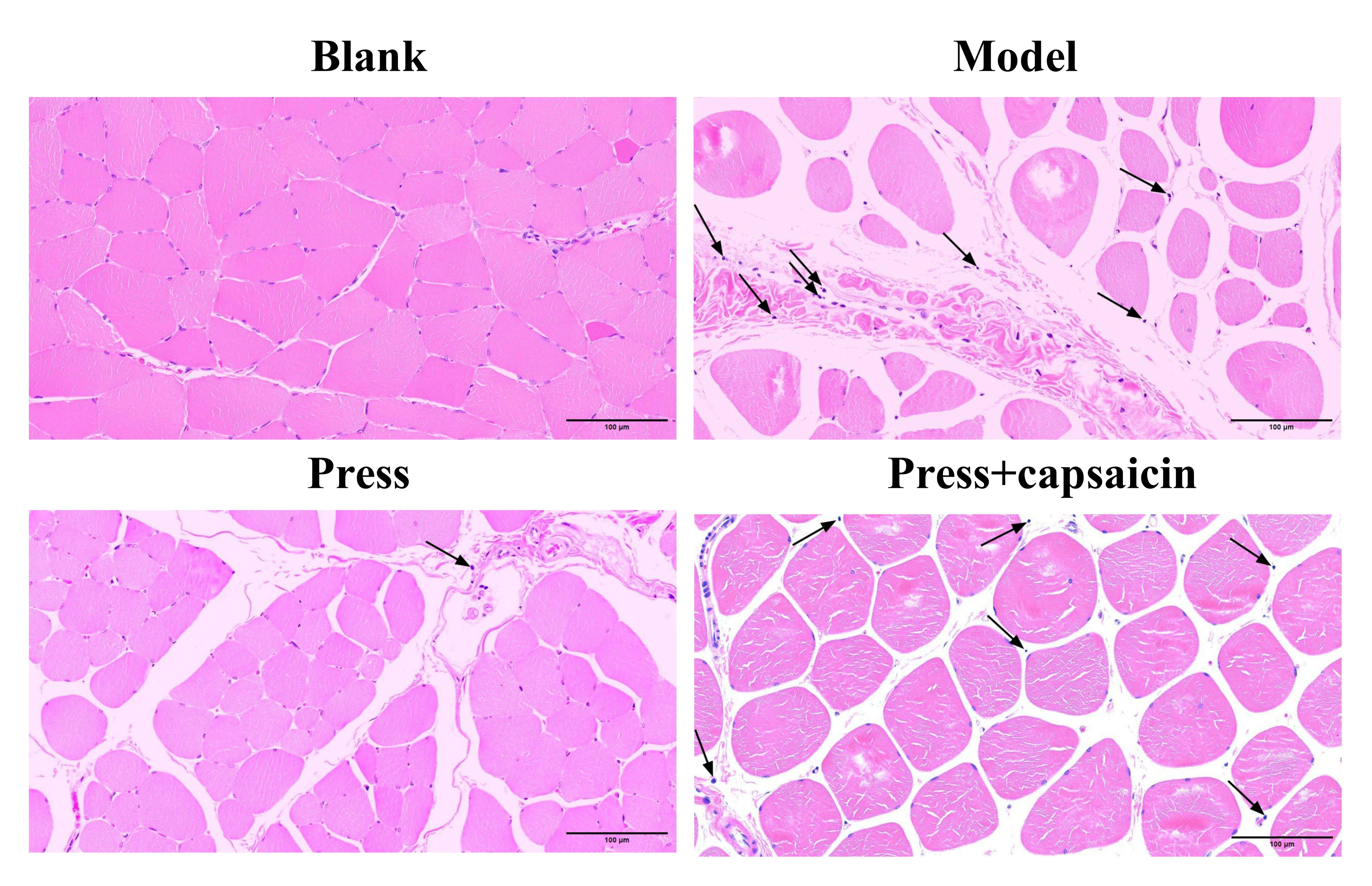 Fig. 4.
Fig. 4.
HE-stained skeletal muscle cell morphology images. The
microscope has a magnification power of
Results are given in Fig. 5. Compared with the blank group, the model group
showed markedly elevated levels of IL-1
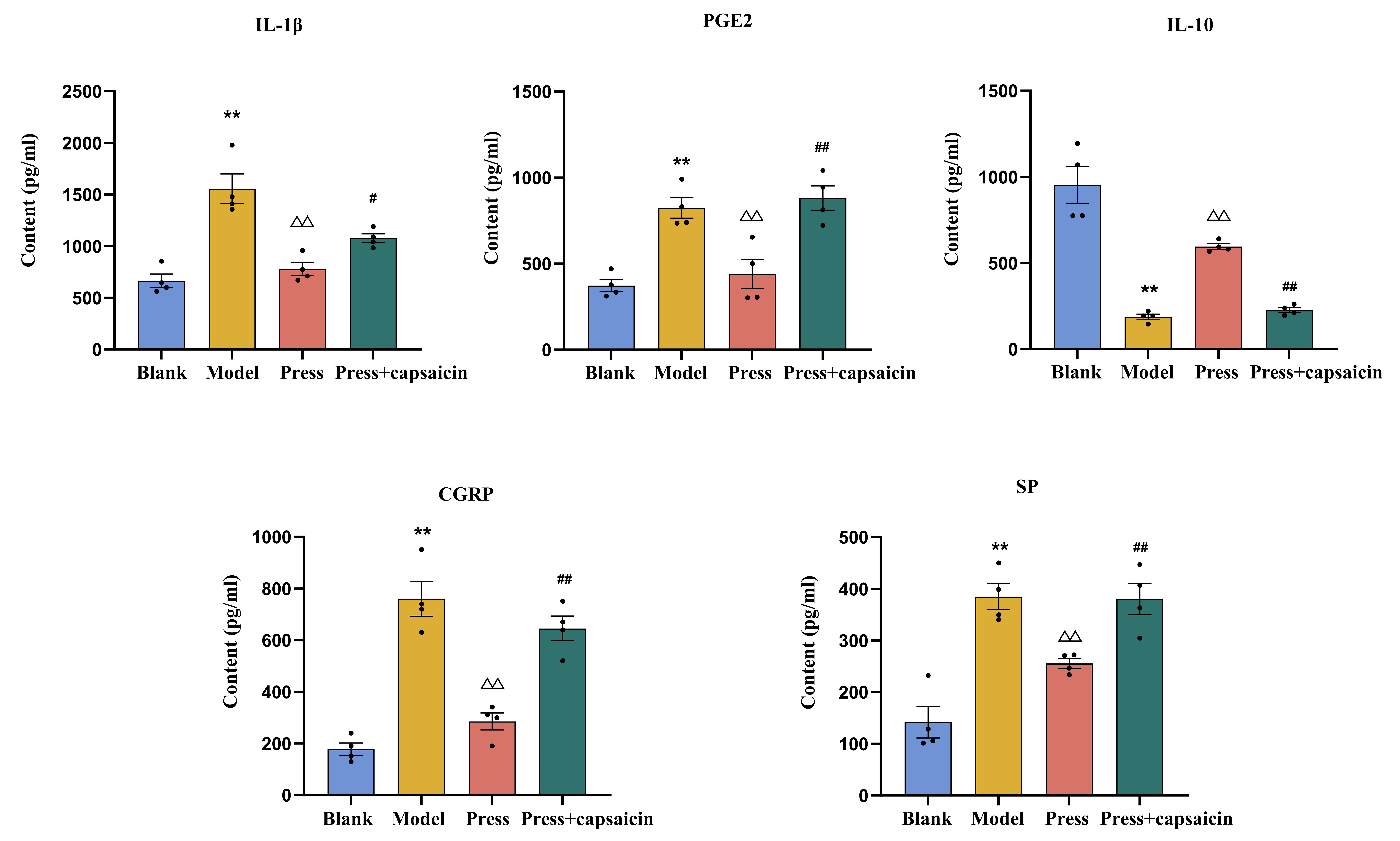 Fig. 5.
Fig. 5.
Comparison of the levels of IL-1
Results are given in Fig. 6. The percentage of TRPV1+/CGRP+ positive
area was expressed as the area of overlapped TRPV1+/CGRP+ fluorescence
divided by the total area of the cropped image. Compared with the blank group,
the model group exhibited a significant increase in the co-localized fluorescent
area of TRPV1 and CGRP (p
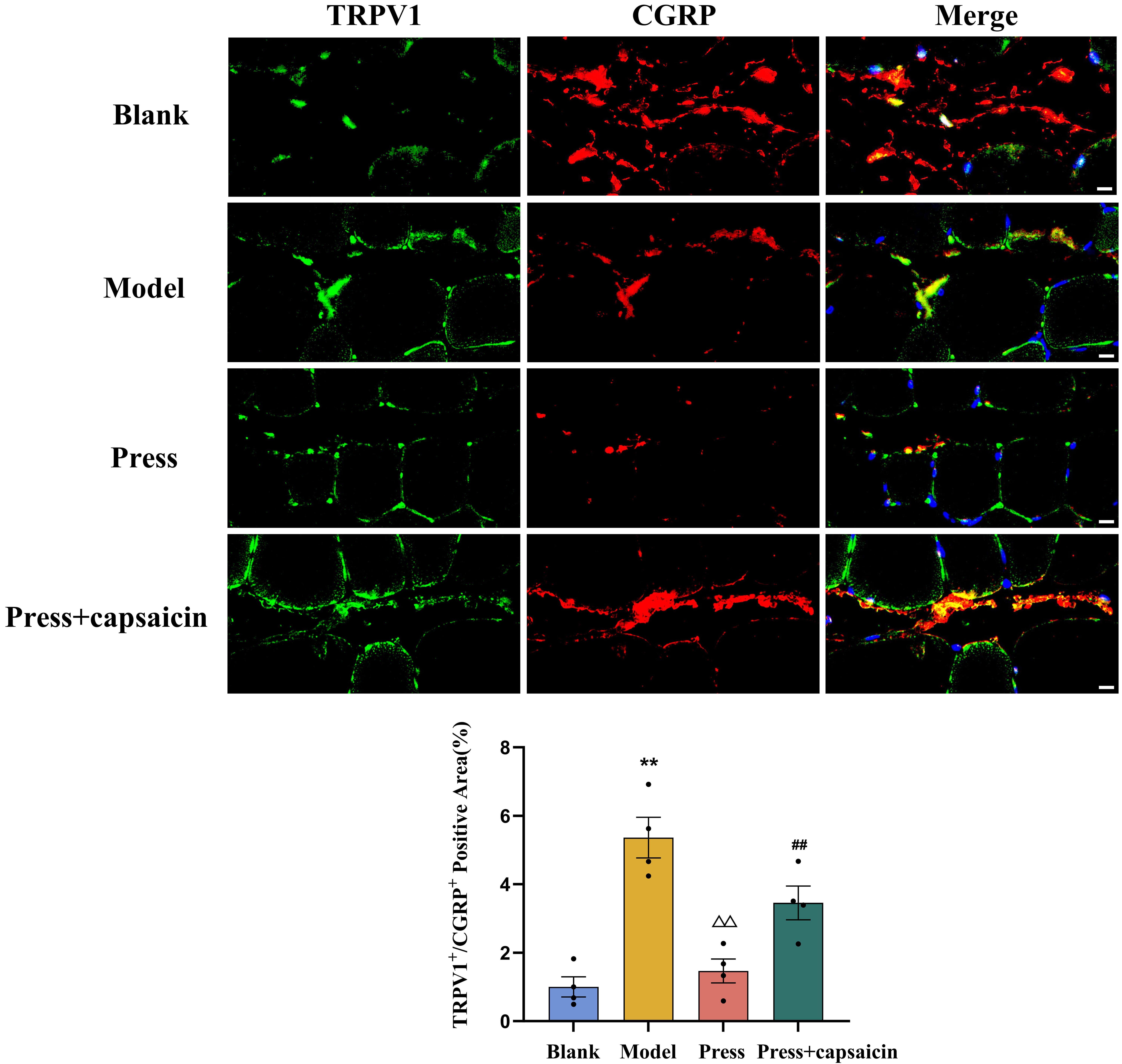 Fig. 6.
Fig. 6.
Immunofluorescence co-localization of TRPV1 and CGRP at
MTrPs in each group (n = 4 per group). TRPV1
(transient receptor potential vanilloid 1) and CGRP (calcitonin gene-related
peptide, a marker for C-type sensory nerve fibers). TRPV1 (green), CGRP (red).
Scale bar: 10 µm. **p
Results are given in Fig. 7. For all original WB figures in Fig. 7, see Supplementary Material 2. Compared with the blank group, the model group
showed a significant increase in TRPV1 expression (p
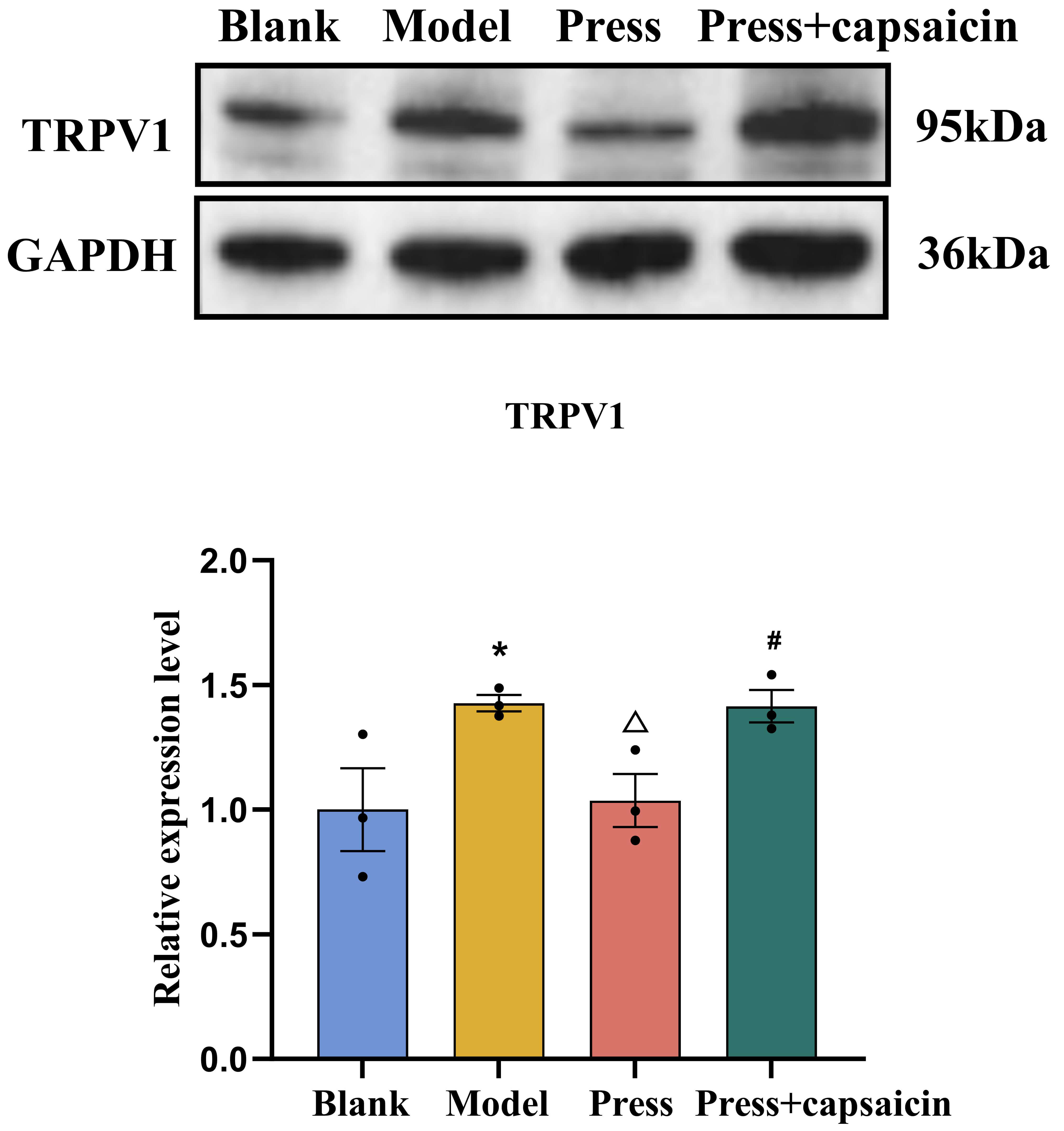 Fig. 7.
Fig. 7.
TRPV1 protein expression in DRG of each group (n = 3
per group). *p
Results are given in Fig. 8. Compared with the blank group, the number of
TRPV1/c-Fos double-labeled positive cells in the DRG was markedly increased in
the model group (p
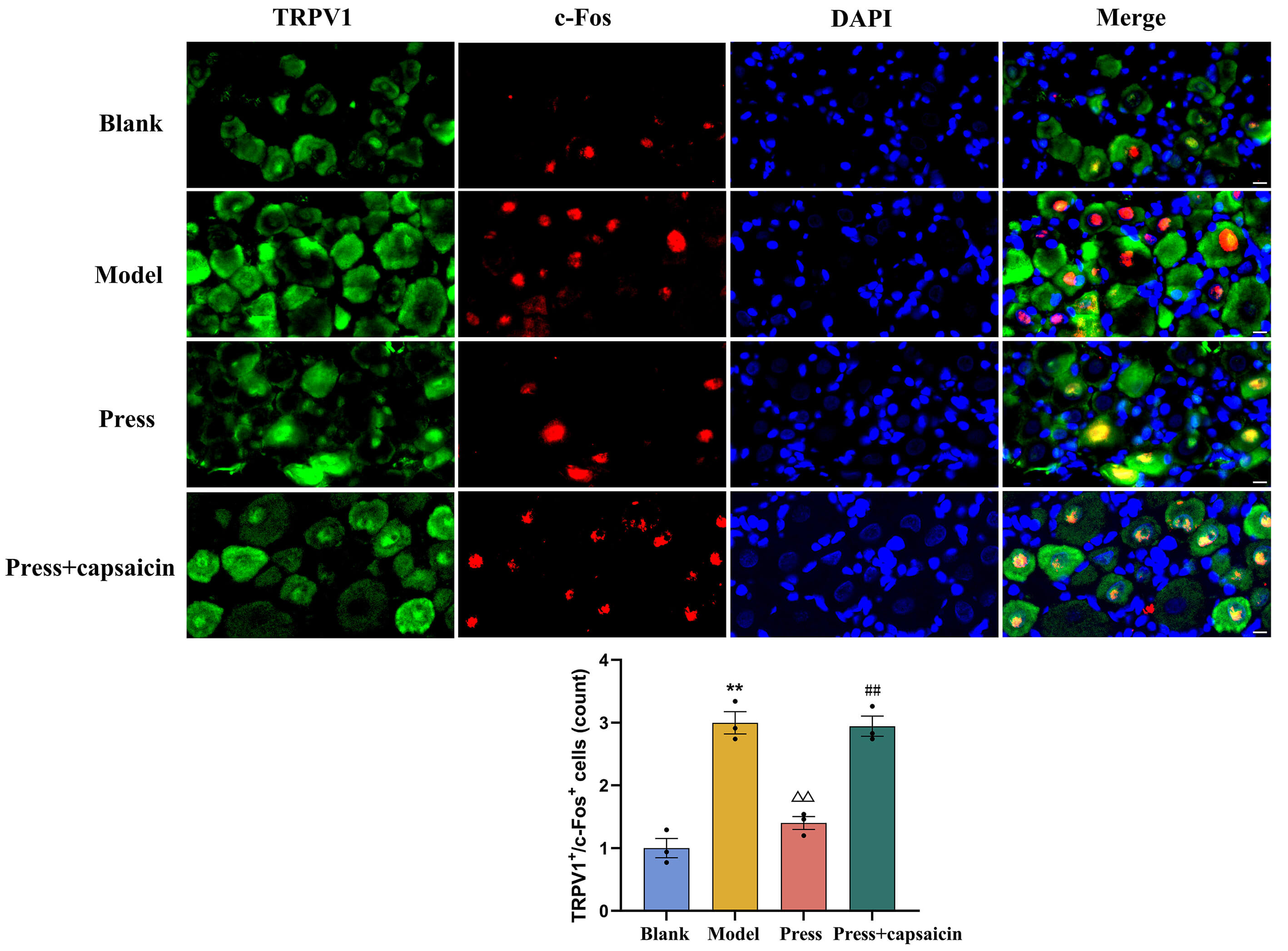 Fig. 8.
Fig. 8.
Immunofluorescence co-localization of TRPV1 and c-Fos
in DRG of each group (n = 3 per group). TRPV1 (green), c-Fos (red), and DAPI
(blue). Scale bar: 10 µm. **p
The pathogenesis of MTrPs is multifactorial and remains incompletely understood, but accumulating evidence highlights the involvement of sustained sarcomere contraction, local ischemia and hypoxia, aseptic inflammation, and sensitization of sensory nerves [26, 27]. Among these factors, aseptic inflammation is a vital driver behind MTrPs formation [28]. Accumulation of inflammatory mediators causes pathological changes in muscle fiber adhesion resembling “taut bands”, as well as activates peripheral nociceptors, thereby inducing hyperalgesia. In this study, pressure pain threshold testing, tissue tension measurement, and histomorphological observation by HE staining were performed to evaluate and characterize local muscle function at rat MTrPs. Results demonstrated that, following modeling, rats exhibited significantly decreased mechanical pain thresholds, increased soft tissue tension, and disorganized muscle cell arrangement with inflammatory cell infiltration. Such findings indicate that the muscle was in a state of sustained high tension and contraction, which is consistent with the histological characteristics of MTrPs [21]. Pressing intervention significantly increased pain thresholds, reduced muscle tension, and promoted the restoration of muscle cell morphology, thus suggesting that mechanical stimulation can restore muscle homeostasis with MTrPs. However, these beneficial effects were partially suppressed when TRPV1 was activated by capsaicin in the press + capsaicin group, implying that TRPV1 may mediate the analgesic effects of pressing on MTrPs.
TRPV1, a nociceptor predominantly expressed in sensory neurons, responds to
diverse noxious stimuli and mediates pain, while also participating in the
regulation of inflammation and neuronal sensitization [15, 29]. Although recent
studies have highlighted TRPV1’s involvement in musculoskeletal pain
sensitization [29] its role in MTrPs-related pain remains to be elucidated.
Muscle injury triggers the secretion of proinflammatory mediators including
IL-1
Previous research has shown significant upregulation of TRPV1 across different
pain conditions, such as arthritis, colitis, and chronic inflammatory pain
[40, 41, 42]. In line with these reports, our finding revealed increased
proinflammatory mediators (IL-1
The DRG, where cell bodies of peripheral sensory neurons reside, serves a pivotal node in nociceptive pain perception, processing, and transmission. Sustained activation of TRPV1 in the DRG can lower action potential thresholds, increase neuronal firing frequency, induce long-term potentiation in the spinal dorsal horn, and drive central sensitization, ultimately contributing to persistent pain [44, 45]. As an immediate early gene, c-Fos serves widely as a marker of neuronal activation. In this study, elevated expression of TRPV1 and increased TRPV1/c-Fos co-localization were observed in DRG neurons following modeling, indicating enhanced neuronal excitability. Pressing intervention reduced TRPV1 protein levels and decreased the number of TRPV1/c-Fos double-positive neurons, thereby inhibiting neuronal hyperactivation. However, these effects were partly reversed by capsaicin treatment, further supporting TRPV1 as a critical peripheral target mediating the analgesic effects of pressing.
Studies have shown that massage promotes tissue repair through mechanisms associated with immune regulation, particularly by facilitating M2 macrophage polarization and reduction of pro-inflammatory cytokine release from M1 macrophages [46]. Given that TRPV1 is highly sensitive to the inflammatory microenvironment, the present findings suggest that massage may exert analgesic effects by improving local inflammatory status and microcirculation, thereby indirectly suppressing the expression and activation of TRPV1 channels in sensory nerves (Fig. 9).
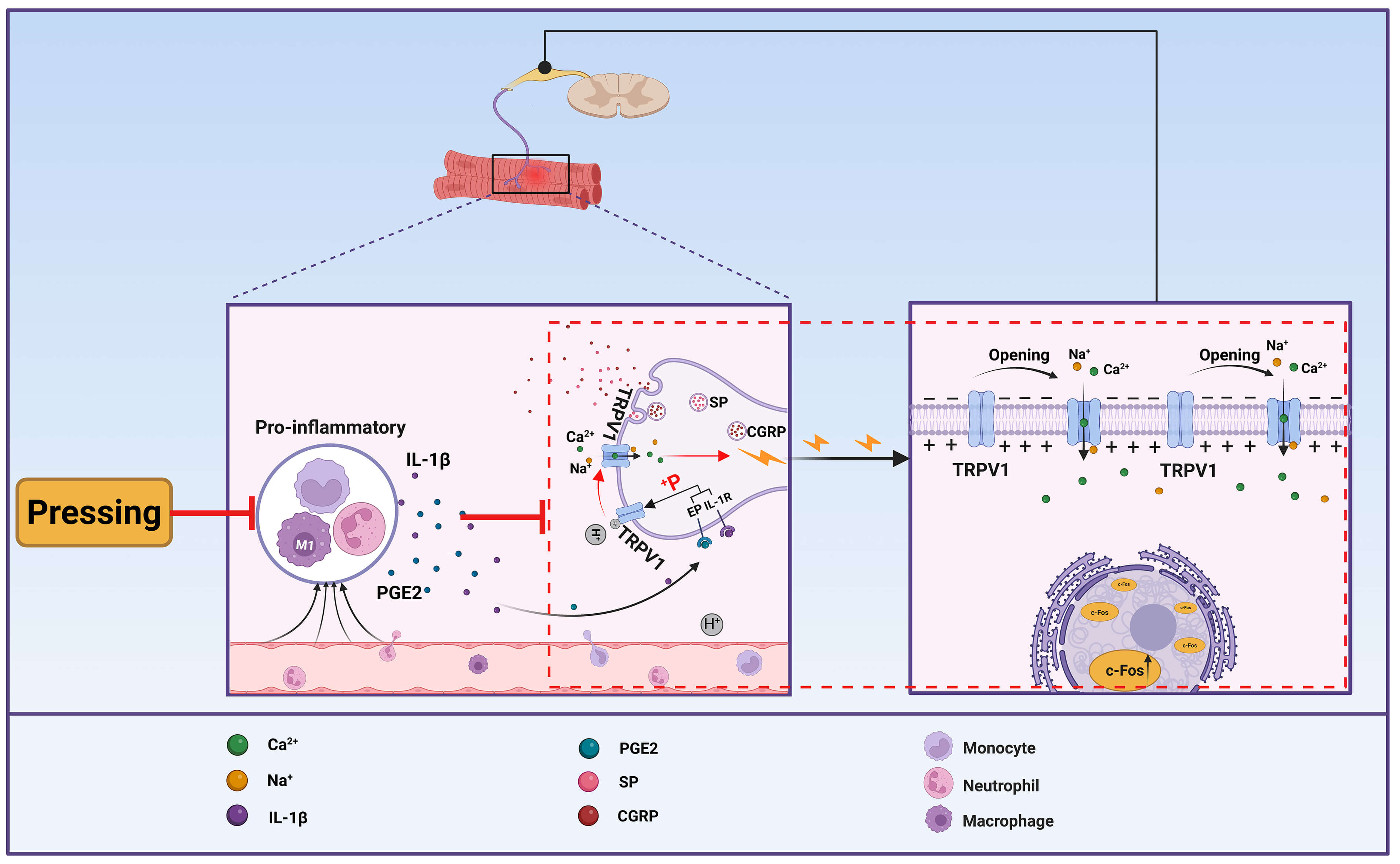 Fig. 9.
Fig. 9.
Schematic diagram of a mechanism by which pressing alleviates pain in MTrPs. The figure was created using Biorender (https://www.biorender.com).
In summary, pressing alleviates pain and reduces local inflammation in MTrPs model rats, an effect that may involve modulation of TRPV1 channel activity in unmyelinated C-fiber sensory neurons. This provides mechanistic evidence that pressing mediates analgesic effects via regulation of sensory neuron ion channels, offering the potential to reduce analgesic drug use and optimize clinical multimodal pain management strategies. However, this study has several limitations. The precise regulatory relationship between pressing, inflammatory mediators, and TRPV1 remains to be elucidated, and the mechanistic depth of the current work is limited. Although the model is trauma-induced, it reproduces key pathophysiological features of clinical MTrPs, supporting the translational relevance of the reported findings.
This study focused on the potential neural mechanisms by which pressing alleviates local inflammation and pain at MTrPs. Findings suggest that pressing may relieve trigger point pain by modulating the inflammatory microenvironment and downregulating TRPV1 activity in C-fibers. These results support the hypothesis that sensory neuronal TRPV1 is a key therapeutic target mediating the analgesic effects of pressing, providing important mechanistic support for pressing as a non-pharmacological treatment strategy.
The datasets used and analyzed during the current study are available from the corresponding author upon reasonable request.
WL and XXK conceived and designed the study. YQZ, LYT, XYL, and FL carried out all experimental procedures. YQZ and LYT performed statistical analyses of the collected data. YQZ drafted the original manuscript. JSL and XXK provided critical insights into data interpretation and revised the manuscript. LYT offered technical assistance throughout the experiments. WL and JSL provided guidance throughout the study, oversaw its implementation, and secured research funding. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
All animal procedures were approved by the Laboratory Animal Ethics Committee of Hunan University of Chinese Medicine (Ethics Approval No.: LL2021102004) and were conducted in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
We sincerely thank the Laboratory of the College of Acupuncture, Moxibustion, Tuina and Rehabilitation, Hunan University of Chinese Medicine, for providing experimental facilities and equipment. We are also deeply grateful to Dr. Quanrui Jiang (Guangxi University of Chinese Medicine) for his invaluable guidance on revising the manuscript.
This work was supported by National Natural Science Foundation of China (No.82374613, No.82405597, No.82174526, No.82274676, No.82474669).
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/JIN46853.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.









