1 Department of Neurology, The Second Affiliated Hospital of Harbin Medical University, 150086 Harbin, Heilongjiang, China
2 Department of Neurology, The Fourth Affiliated Hospital of Harbin Medical University, 150001 Harbin, Heilongjiang, China
Abstract
Alzheimer’s disease (AD) is a degenerative condition affecting the central nervous system and is the primary cause of dementia. Current therapies for AD are ineffective. Although brain regeneration via stem cell transplantation has therapeutic potential, suitable sources are limited. Hair follicle stem cells (HFSCs) are multi-potent cells and can differentiate into mesodermal and ectodermal lineages, and proliferate for extended periods. Nerve growth factor (NGF) is a neurotrophin that is vital for neuronal development and survival, and the regulation of apoptosis in neurodegenerative disorders. However, using HFSCs to treat AD has not been extensively investigated. Herein, we evaluated the therapeutic effects of HFSCs and the synergistic effect of NGF and HFSCs on AD.
A rat model of AD was established by intrahippocampal injection of amyloid β-protein 1–42 (Aβ1–42). After 14 days, HFSCs and HFSCs overexpressing NGF were injected into the hippocampus of AD rats for therapy. The cognitive function of the treated AD rats was tested using the Morris water maze test. Congo red staining, immunohistochemistry, and enzyme-linked immunosorbent assay (ELISA) were used to detect deposition, as well as soluble Aβ1–40 and Aβ1–42 levels. Additionally, western blotting was used to assess tau protein, the phosphoinositide-3 kinase (PI3K)/protein kinase B/glycogen synthase kinase-3β (Akt/GSK-3β) pathway, and the levels of synapse proteins.
HFSCs and HFSCs/NGF transplantation not only significantly reduced Aβ deposition but also inhibited GSK-3β activity and reduced tau protein hyperphosphorylation by stimulating the PI3K/Akt signaling pathway. Moreover, HFSC and HFSC/NGF transplantation led to significant overexpression of the synaptophysin (SYP) and postsynaptic density protein 95 (PSD95) in the hippocampus of AD rats.
HFSCs and NGF-modified HFSCs may become a promising treatment option for AD.
Keywords
- Alzheimer’s disease
- cognitive dysfunction
- hair-follicle stem cells
- nerve growth factor
- PI3K-Akt pathway
Alzheimer’s disease (AD) is a degenerative neurological condition that worsens gradually over time. It is characterized by memory loss and cognitive decline and has rapidly emerged as one of the most expensive, fatal, and burdensome diseases [1]. According to Alzheimer’s Disease International, the worldwide occurrence of dementia in 2016 was approximately 50 million, with AD accounting for 60%–80% of the cases [2]. The World Health Organization has predicted that the global number of individuals affected by dementia will increase to 139 million by 2050, from 55 million in 2019, due to the aging of the population [3, 4]. The percentage of deaths attributable to AD in the United States is increasing, in contrast to declining overall mortality rates from stroke and cardiovascular diseases. AD was officially the 6th most prevalent cause of mortality in the U.S. in 2019 and the seventh most frequent cause of death in 2020 and 2021 [5].
AD is distinguished pathologically by senile plaques, which are clumps of
amyloid
Before lecanemab was approved as a treatment, patients with AD had access to only two classes of medication: (a) cholinesterase inhibitors, including donepezil, rivastigmine, and galantamine, which are used to treat individuals with mild, moderate, and severe AD dementia [9]; and (b) memantine, which is a medication with two activities—it serves as a noncompetitive antagonist of N-methyl-D-aspartate receptors and as an agonist of dopamine. Memantine manifests in individuals with moderate to severe AD [10]. On 7 January 2023, lecanemab was granted rapid approval by the U.S Food and Drug Administration for AD management, marking a significant advancement in the treatment of this disease [11]. Stem cell transplantation is one of the most cutting-edge and promising experimental treatments for AD. Stem cells replace cells and use various methods to control inflammatory or damaged environments [12, 13, 14]. Hair follicle stem cells (HFSCs) are thought to be an excellent cell type for the treatment of neurological illnesses. Mouse hair follicles harbor HFSCs that express the neural progenitor marker nestin rather than the keratin cytokeratin [15]. McKenzie et al. [16] demonstrated that HFSC transplantation contributes to myelin-sheath development in the sciatic nerve in mice that have a genetic myelin basic protein deficit. Moreover, HFSCs first migrate to the brain and then to the fore-head, where they give rise to various neural phenotypes, including immature neurons and well-differentiated astrocytes in rodents model [17, 18, 19]. However, additional investigations are necessary to clarify the functions and mechanisms of HFSCs in AD.
Nerve growth factor (NGF) is categorized as a neurotrophin. Rita Levi-Montalcini
first discovered it in mouse sarcoma in the early 1980s [20]. The peripheral
nervous system, immunological cells, sensory afferents,
sympathetic-efferent-innervated peripheral nerves, and the central nervous system
(CNS) all express NGF [21]. In the CNS, certain areas, such as the cortex and
hippocampus, which are targeted by cholinergic neurons, have an elevated level of
NGF expression [22]. NGF is vital for neuron development [23], survival [24], and
the regulation of apoptosis in mammals, including humans [25]. NGF starts
signaling by binding to two distinct cell-surface receptors [26]: tropomyosin
receptor kinase A (TrkA) and p75 neurotrophin receptor (p75 NTR), which are
lower-affinity receptors. TrkA-mediated pathways are key for the growth and
metabolism of neurons [27]. When NGF binds to the TrkA receptor, its intrinsic
tyrosine kinase activity catalyzes TrkA’s autophosphorylation, which activates
three pathways: phospholipase C-
This investigation assessed the potential therapeutic benefits of HFSCs and NGF-modified HFSCs, and explored the underlying mechanisms, in a rat model of AD, in order to help develop new approaches for improving AD treatment.
Hair follicles were extracted by mechanical dissection after enzymatic digestion
[15]. To isolate the hair follicles, hair-follicle pads of male Sprague-Dawley (SD)
rats (21–26 g; purchased from the Animal Experiment Center of the Second Affiliated Hospital of Harbin Medical University) were dissected and digested with 0.1% collagenase (17018029,
Gibco, Thermo Fisher Scientific, Waltham, MA, USA) in Dulbecco’s modified Eagle’s
medium (DMEM) (11965092, Gibco, Thermo Fisher Scientific). The hair follicles
were then carefully extracted from the pads and placed into 12-well tissue
culture dishes (Beyotime, Shanghai, China) for cultivation. The hair follicles
were cultivated in DMEM/F12 (11320033, Gibco, Thermo Fisher Scientific) medium
with 1% penicillin and streptomycin (15140122, Gibco, Thermo Fisher Scientific)
and 10% fetal bovine serum (0025, ScienCell Research Laboratories, Santiago, CA,
USA) at 37 °C in a humidified conditions with 5% CO2. Cell growth
was monitored under a microscope, and half-medium alterations were conducted
every 2–3 days. Primary cultures were first passaged after 14 days of
cultivation, followed by subsequent passages every 3–5 days. Upon reaching
85–90% confluence, cells were dissociated via Accutase™
(A1110501, Gibco, Thermo Fisher Scientific), a neural stem-cell-specific cell
dissociation reagent, during passage. The third-generation HFSCs were cultivated
(3
The lentivirus vector was PLJM1-EGFP-puro (FH1717, Fenghui Biological Company,
Changsha, Hunan, China). The third passage of cultured HFSCs was co-cultured with
either NGF-overexpressing lentivirus (Gene: Lv-Rat-NGF-OE,
NM_001277055) or blank lentivirus at a multiplicity of infection of 80
for 72 h. Specifically, 16 µL of virus stock with a titer of 1
We performed surface-marker detection of the cell line using flow cytometry.
Single-cell suspensions of third-passage HFSCs (4-week in vitro culture)
were prepared, counted, and aliquoted (
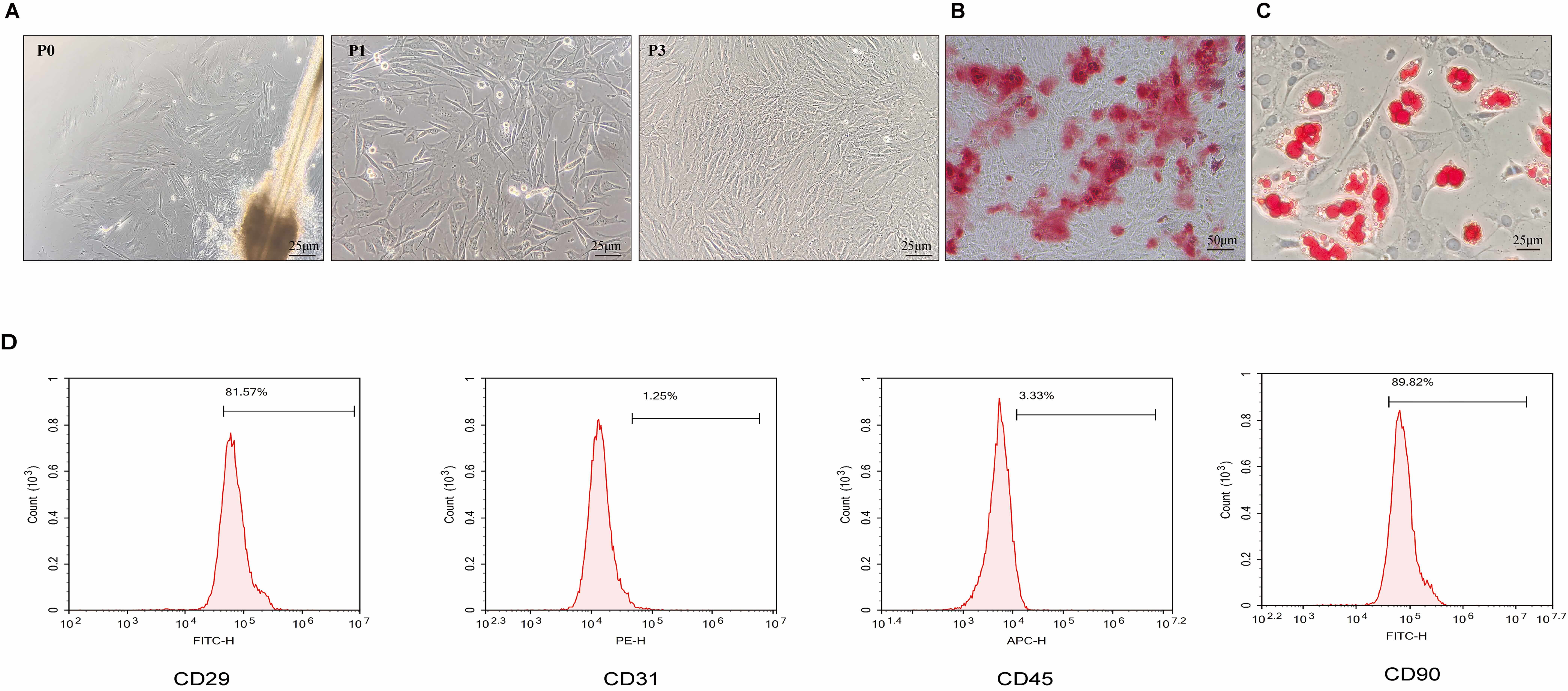 Fig. 1.
Fig. 1.
Results of isolation, culture, and identification of HFSCs from the bulge region of hair follicles and the ability of HFSCs to poly-differentiate. Optical microscopy of an intact hair follicle, microdissected and extracted from the hair follicle in the bulge region for primary cell culture. (A) HFSCs in primary, P1, and P3 were under an optical microscope. After 14 days of culture, osteogenic and adipogenic differentiation was confirmed by (B) Alizarin red and (C) Oil Red O staining. (D) Flow cytometry detection of HFSCs surfaces positive markers CD29, CD90, negative markers CD31, CD45. (A,C) scale bar = 25 µm, (B) scale bar = 50 µm. Abbreviations: HFSCs, hair follicle stem cells; FITC-H, fluorescein isothiocyanate-height; PE-H, phycoerythrin-height; APC-H, allophycocyanin-height; CD, cluster of differentiation; P, passage.
As directed by the manufacturer [32], the growth medium was exchanged for an osteogenic or adipogenic differentiation medium after a 24-h incubation. Three days were given for a shift in the osteogenic and adipogenic differentiation media while the cells were cultivated in an appropriate environment at 37 °C with 5% CO2. Alizarin red (Y027377, Beyotime) and Oil Red O (Y264669, Beyotime) were used to confirm osteogenic and adipogenic differentiation after 14 days of culture.
Flow cytometry was applied in order to detect cell-surface markers. The
3rd-generation HFSCs cell suspension was made by digesting the cells with
trypsin (0.25%, 25200056, Thermo Fisher Scientific), then rinsing two times with phosphate buffered saline (PBS), and finally
adjusting to 1
The Animal Experiment Center of the Second Affiliated Hospital of Harbin Medical
University provided clean-grade, male, SD rats (6–8 weeks old,
200–210 g) [License No. SCXK (Hei) 2023-001]. All rat experiments were conducted
as per the principles outlined in the “Guide for the Care and Use of Laboratory
Animals” published by the Ministry of Science and Technology of the People’s
Republic of China. Sixty were randomly allocated into four groups (n =
15/gp): Sham+PBS, A
After being anesthetized, rats were subjected to transcardial perfusion with 0.9% physiological saline via thoracic cavity exposure. Brains were subsequently dissected, immersed in 4% paraformaldehyde (70-F0001; MultiSciences) for fixation, then PBS was utilized for rinsing. After that, the tissues were processed through gradient dehydration, paraffin-embedded, and sliced at a thickness of 40 µm. Then, slices were deparaffinized, followed by gradient rehydration and stained with Congo red dye for 10–20 min, then differentiated with alkaline ethanol differentiation solution for several seconds, and washed with water, then counterstained with hematoxylin (H3136; Sigma-Aldrich) for 2 min and rinsed with running water for 1–2 min. After that, the slices were dipped in ascending grades of ethanol. Finally, they were cleared with xylene (214736; Sigma-Aldrich) and sealed with neutral resin (G8590; Solarbio, Beijing, China). The slides were photographed under an optical microscope (ECLIPSE Ti2-U, Nikon).
The immersion of paraffin slices was conducted in 0.3% H2O2 for 30
min. Next, normal goat serum (c0265, Beyotime) was utilized to block the slices
for 30 min at 25 °C, then an overnight incubation with primary mouse
anti-
The rats underwent a navigation test in a water maze (the water temperature was
controlled at 25
Behavioral testing and image quantification were conducted blindly. All animals were ear-tagged for identification. Group allocation was done by a different person; both the operators performing behavioral tests and the personnel collating data were unaware of the groupings, thus reducing bias.
Rats from every group were euthanized and the hippocampus was dissected for
measurement of the protein levels of A
The hippocampus was homogenized with a tissue extraction buffer that included
protease inhibitors. This was accompanied by centrifugation at 12,000 rpm at a
temperature of 4 °C for 10 min. The resulting supernatants were gathered and
assessed for protein concentration with BCA protein assay kits (ab102536, Abcam).
The protein samples, weighing 40 µg, were separated using 10% or
12.5% SDS-polyacrylamide gel electrophoresis (P0012A, Beyotime) at 100 V for
90–120 min. Then they were moved to a polyvinylidene fluoride (PVDF) membrane
(88520, Thermo Fisher Scientific) using a current of 200 mA at 4 °C for 120 min.
The PVDF membrane was preincubated with 3% nonfat milk in Tris-buffered saline
with 0.1% Tween-20 (TBS-T) at 25 °C for 60 min to block nonspecific
binding sites. The incubation of PVDF membranes was conducted overnight at 4 °C
with primary antibodies targeting protein kinase B (Akt) (1:1000; 4685, Cell
Signaling Technology, Danvers, MA, USA), phospho-Akt (Thr308) (1:1000; 13038,
Cell Signaling Technology), glycogen synthase kinase (GSK)-3
NGF levels in the untreated HFSCs, the HFSCs/Vector, and the HFSCs/NGF groups were quantified by qRT-PCR assay. In brief, total RNA isolation from the respective HFSCs was conducted via TriZol reagent (15596026CN, Invitrogen, Carlsbad, CA, USA). Reverse transcription was conducted via the PrimeScript RT reagent kit (RR037A, TaKaRa, Dalian, Shandong, China). Quantitative PCR was conducted via the SYBR PrimeScript RT-PCR Kit II (RR820A, Merck KGaA, Darmstadt, Germany) on an LC480 real-time PCR system (Influence Bio, Shanghai, China). The sequence of each primer is shown in Table 1.
| Gene | Primer sequence |
| rat NGF F | ACTCTGTCCCTGAAGCCCACTG |
| rat NGF R | TGTTGCGGGTCTGCCCTGTC |
| rat |
GGAGATTACTGCCCTGGCTCCTAGC |
| rat |
GGCCGGACTCATCGTACTCCTGCTT |
qRT-PCR, quantitative real-time reverse transcription PCR; NGF, nerve growth factor.
Data are reported as the mean
HFSCs’ isolation from the upper lips of male SD rats was conducted; hair-follicle pads were cultivated until the 3rd generation (Fig. 1A). After inducing the osteogenic differentiation, HFSCs differentiated into osteoblasts, confirmed by positive Alizarin red staining (Fig. 1B). Moreover, HFSCs differentiated into adipocytes, as manifested by the presence of lipid droplets with orange coloration upon staining with Oil Red O dye (Fig. 1C). These steps illustrated the multidirectional differentiation capability of HFSCs. Flow-cytometry analysis revealed that the cell surface markers of HFSCs, such as CD29 and CD90, were overexpressed, while the endothelial cell-surface marker CD31 and the hematopoietic cell-surface marker CD45 were suppressed (Fig. 1D).
Fig. 2A illustrates the viral vector utilized in this investigation. The level of NGF mRNA expression was quantified by using PCR in the control, the HFSCs/Vector, and the HFSCs/NGF groups (Fig. 2B). The expression levels of NGF in these groups were assessed with WB analysis (Fig. 2C,D, the original WB images can be found in the Supplementary Material). These outcomes verified the HFSCs/NGF’s successful construction.
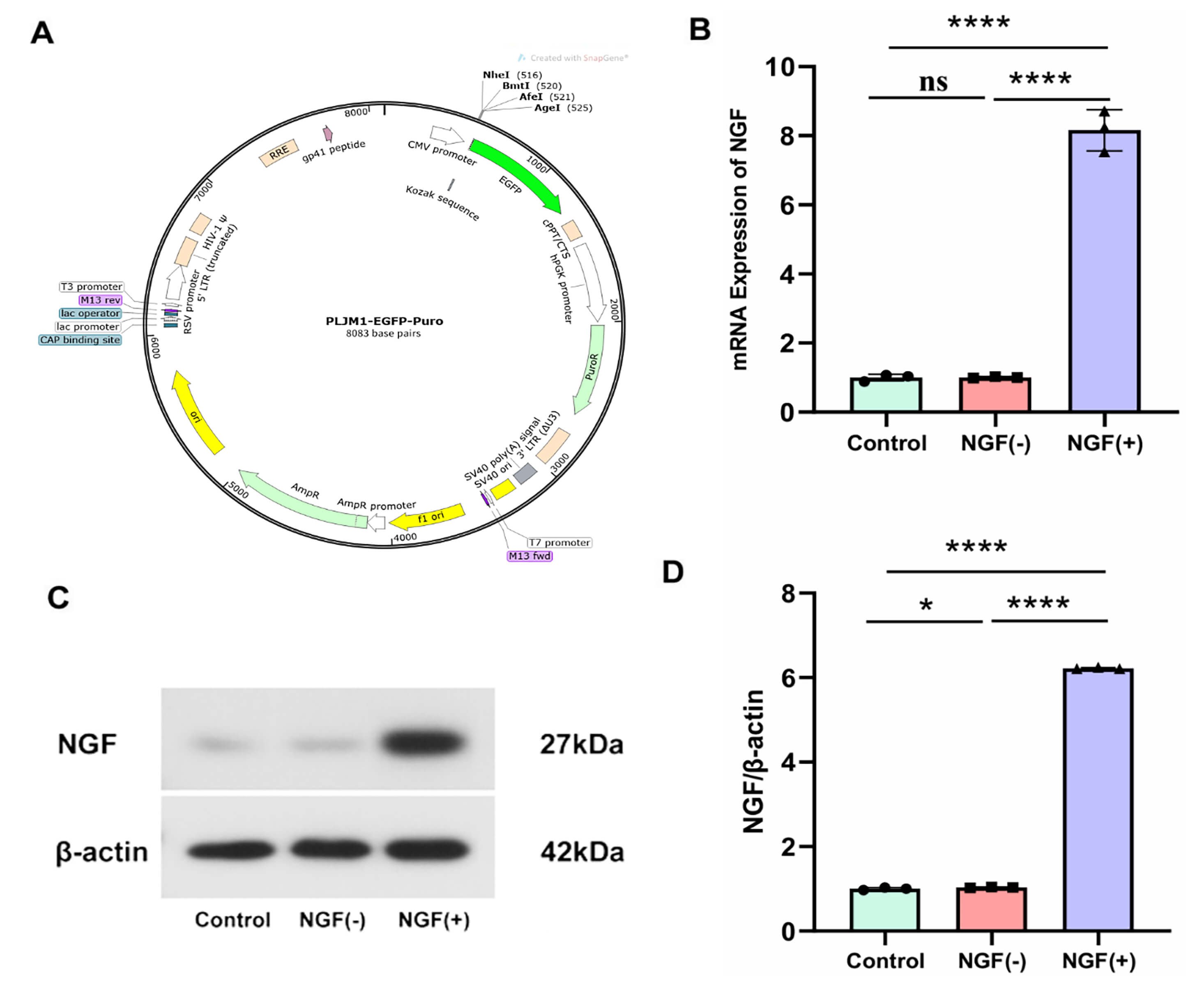 Fig. 2.
Fig. 2.
Successful construction of NGF over-expressed HFSCs. (A) The
structure of the lentivirus vector with NGF over-expressing. (B) The NGF
mRNA level was quantified with Real-time PCR. (C,D) The NGF expression levels
were estimated through western blot analysis. Data analysis was conducted by
one-way ANOVA with Tukey’s post-hoc test (B,D), *p
A
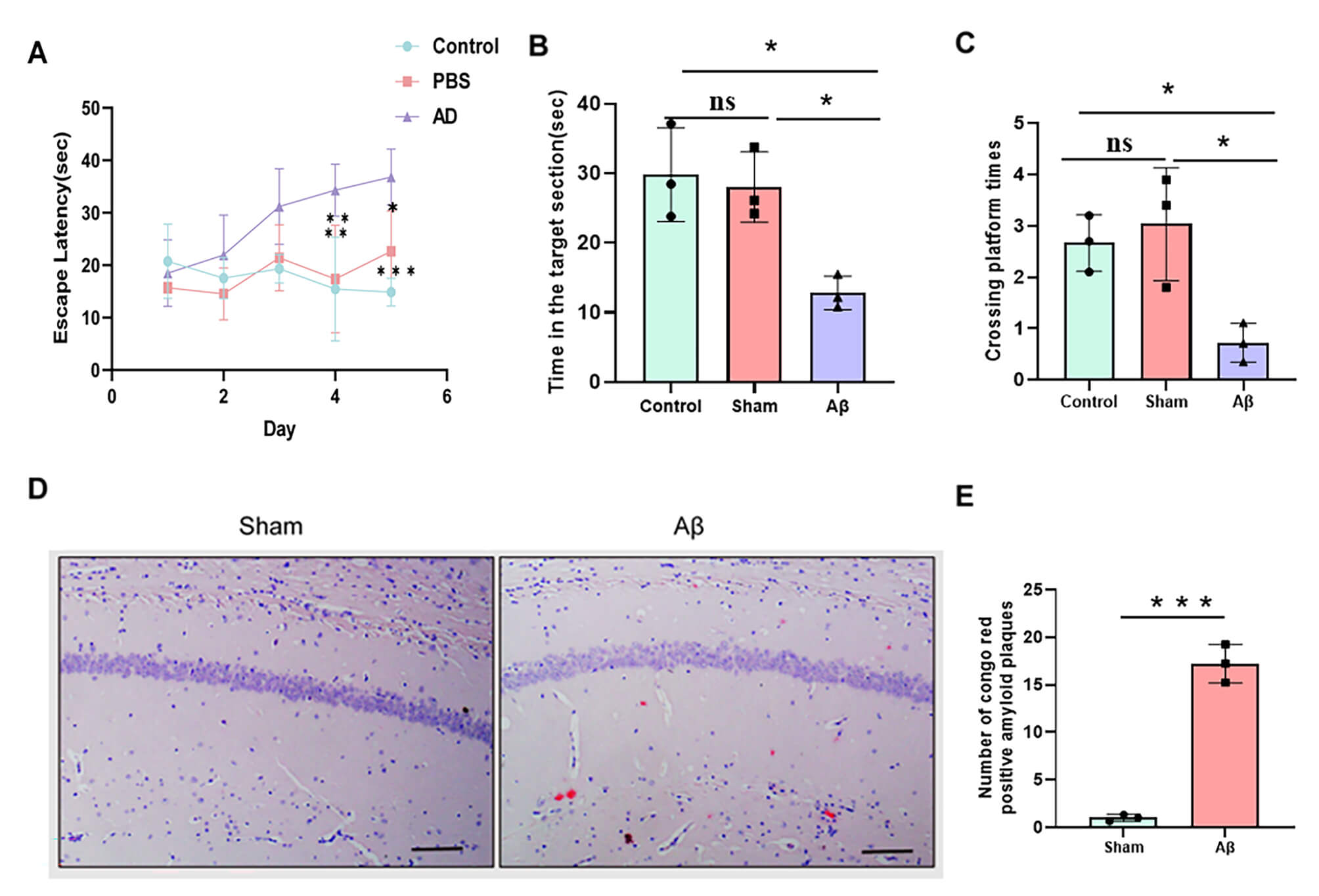 Fig. 3.
Fig. 3.
AD rat model results. (A) The latency for all groups to escape
in the location-navigation exam. (B,C) In the spatial probe test, the number of
platform crossings and the time in the target section in each group. (D)
A
To investigate the potential of transplanted HFSCs to restore behavioral deficits in AD rats, Morris water maze tests were performed 14 days post-cell transplantation. During the five-day training period, HFSC-transplanted rats exhibited a gradual increase in their ability to find a concealed platform (Fig. 4). Rats in the HFSCs/NGF transplanted group exhibited significantly better performance than did those in the HFSCs transplanted group (Fig. 4A). Throughout the spatial probe test, the frequency of crossing the eliminated platform was higher (Fig. 4B), and the duration in the target quadrant was longer in the HFSCs group and HFSCs/NGF group than in the AD group (Fig. 4C). Moreover, a significant difference in tracks of the spatial probe test on the 6th day was observed among the four groups (Fig. 4D); the rats transplanted with HFSCs and HFSCs/NGF improved their approaches for reaching the platform. In summary, these results indicated that transplantation of HFSCs and HFSCs/NGF reduced cognitive dysfunction in AD rats.
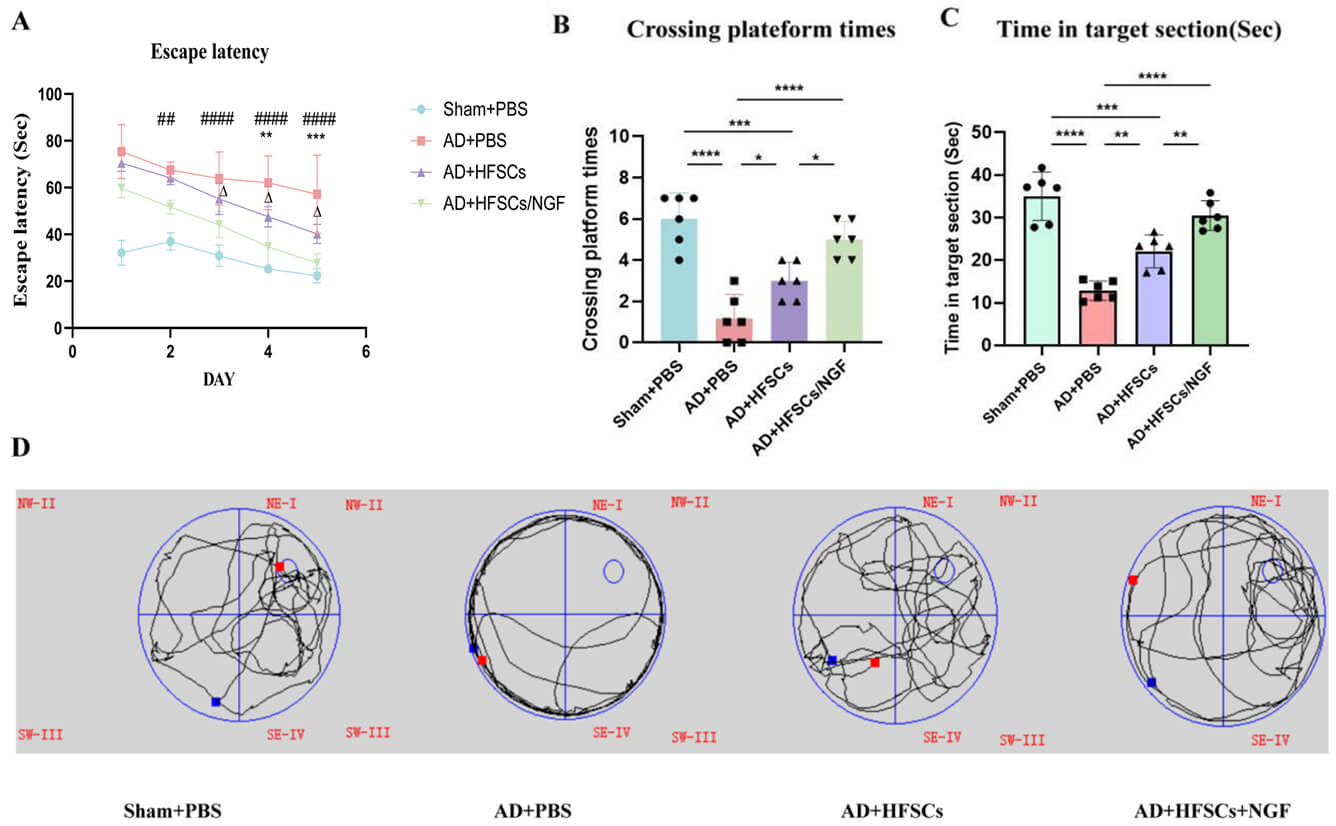 Fig. 4.
Fig. 4.
HFSCs and HFSCs/NGF transplantation improved the learning and
memory impairment. (A) The latency for each group to escape in the
location-navigation test, in the spatial-probe test, (B) the number of platform
crossing and (C) time in the target section. (D) Illustrative tracks of the
spatial probe test on day 6. When comparing with the AD rats, the rats
transplanted with HFSCs and HFSCs/NGF showed significantly improved learning and
memory. Data were assessed by two-way ANOVA (A), and analyzed by one-way ANOVA
(B,C), AD+PBS group vs. AD+HFSCs group, *p
To investigate the effects of HFSCs and HFSCs/NGF transplantation on the
production of toxic amyloid plaques, immunohistochemistry was conducted using
6E10 antibodies in the brains of AD rats (Fig. 5A). Quantification of stained
plaques in the hippocampus revealed significant variations between the HFSCs and
the HFSCs/NGF groups (Fig. 5B). Protein levels were measured in four rat groups
using A
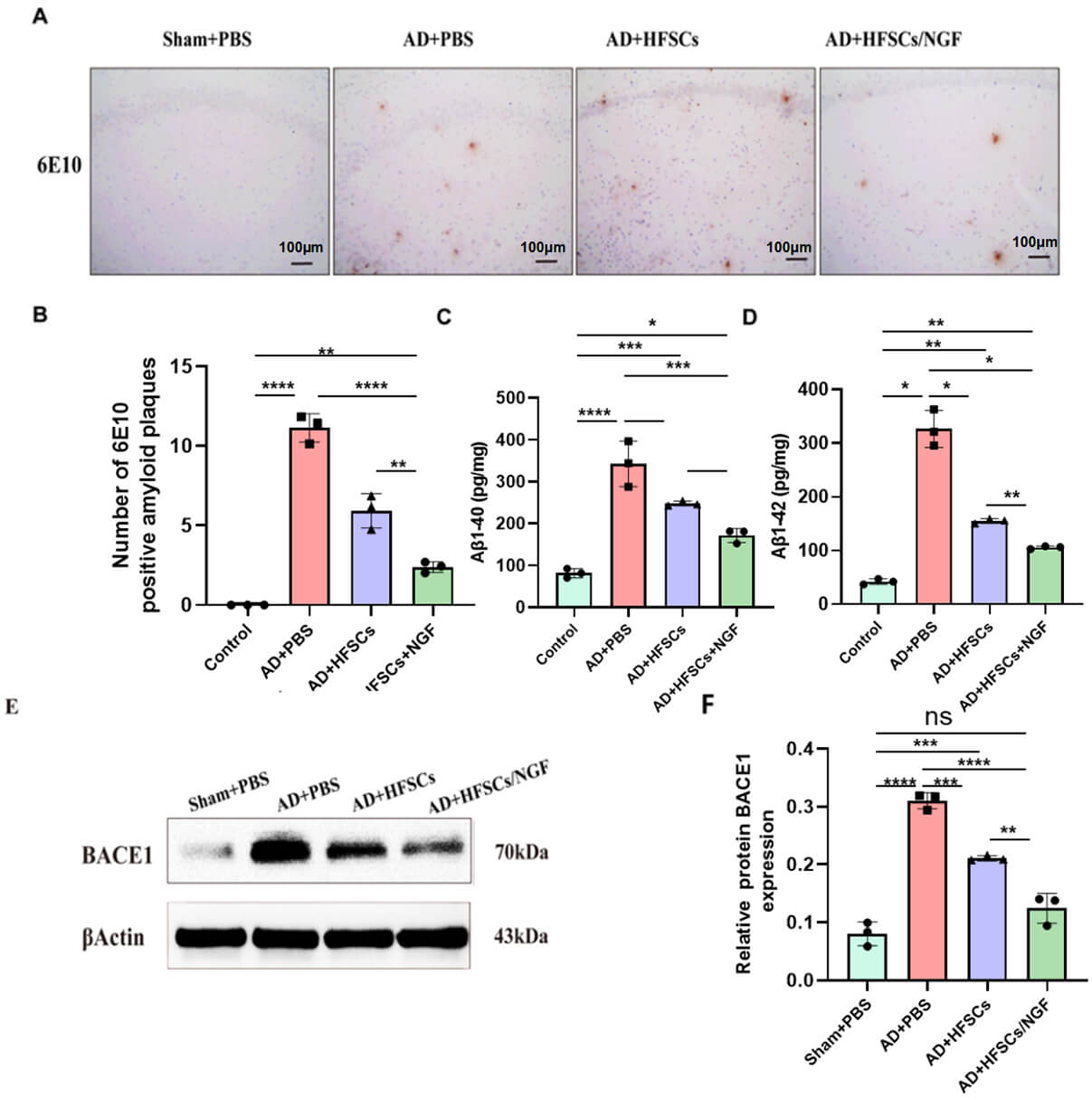 Fig. 5.
Fig. 5.
HFSCs and HFSCs/NGF transplantation declined the level of
A
Another important pathology of AD is tau hyperphosphorylation. To verify the consequences of HFSCs and HFSCs/NGF on tau hyperphosphorylation, the phosphorylation levels of tau 5 and other tau sites were quantified by using western blot analysis. The outcomes illustrated that HFSCs and HFSCs/NGF transplantation inhibited tau 5 and tau phosphorylation at Ser202/Thr205, Thr231, S396, and S404 sites in the AD rat hippocampus (Fig. 6, the original WB images can be found in the Supplementary Material).
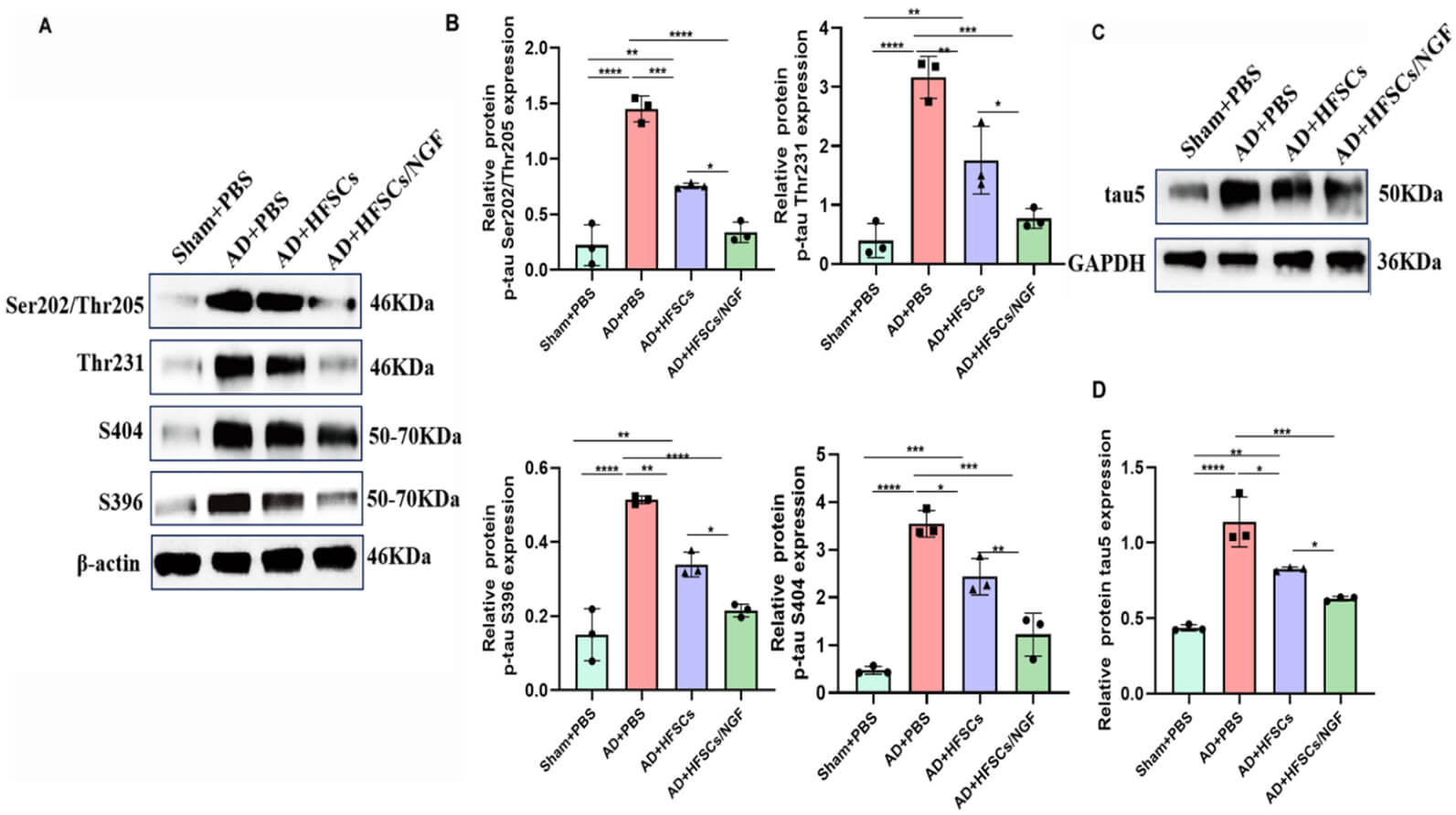 Fig. 6.
Fig. 6.
HFSCs and HFSCs/NGF transplantation reduced tau
hyperphosphorylation. (A) The tau phosphorylation site levels were quantified by
western blot analysis, showing that HFSCs and HFSCs/NGF transplantation inhibited
tau phosphorylation at Ser202/Thr205, Thr231, S396, and S404 sites in the
hippocampus of AD rats. (B) The relative quantification of Ser202/Thr205, Thr231,
S396, and S404 sites protein expression. (C) The tau 5 concentration was assessed
by western blot analysis in the hippocampus. (D) The relative quantification of
tau5 protein expression. One-way ANOVA with Tukey’s post-hoc test was utilized to
analyze the data (B,D), *p
Proteins of the PI3K/Akt/GSK3
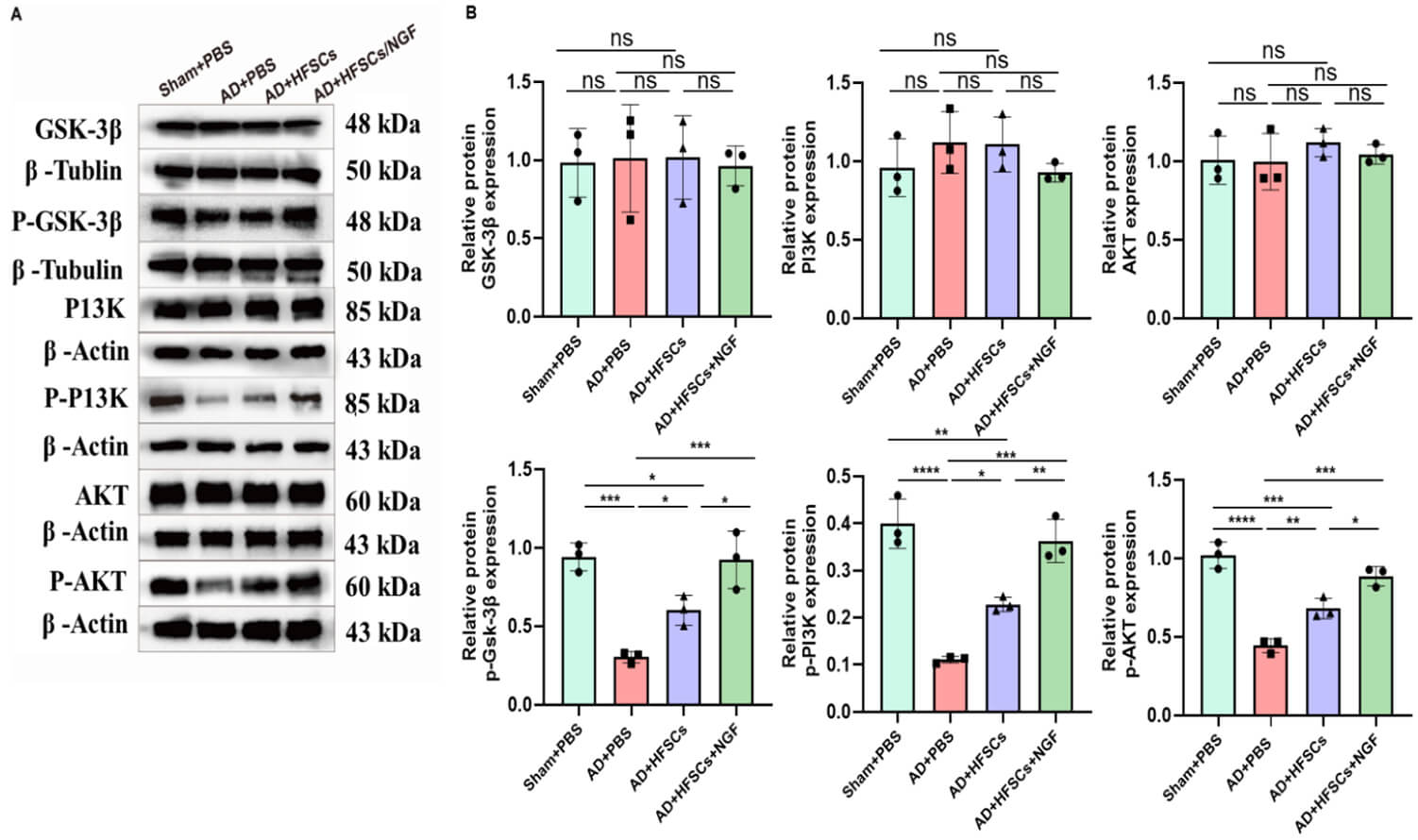 Fig. 7.
Fig. 7.
HFSCs and HFSCs/NGF transplantation inhibited the activity of
GSK3
Of all the pathological changes that occur in the brains of individuals with AD, synaptic loss is most strictly related to cognitive decline [34]. Therefore, we analyzed the SYP and PSD-95 levels in the hippocampus of the four groups by immunohistochemical staining and western blotting (Fig. 8). The SYP and PSD-95 levels were significantly lower in AD+PBS rats than in Sham+PBS rats. However, after HFSCs and HFSCs/NGF transplantation, the level of SYP rose significantly (Fig. 8A,B, the original WB images can be found in the Supplementary Material). The PSD-95 level displayed a significant elevation in the HFSCs/NGF group, but no significant difference was shown between the AD+PBS and the AD+HFSCs groups (Fig. 8C,D).
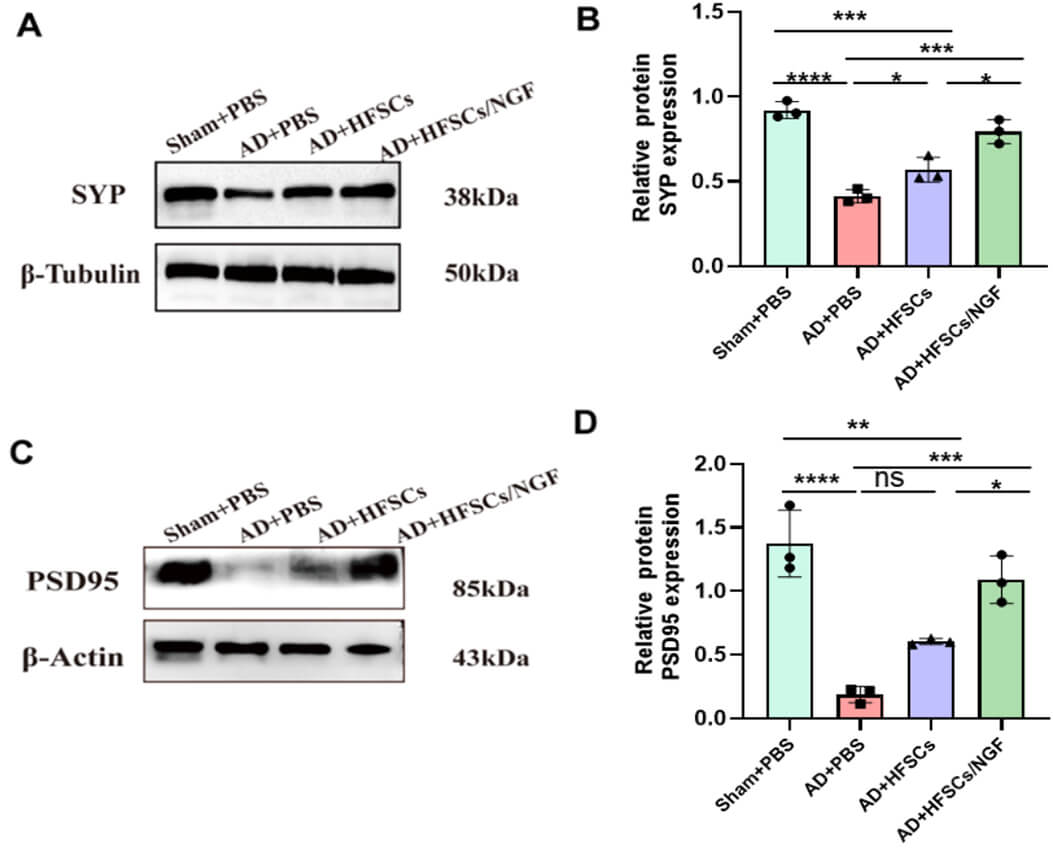 Fig. 8.
Fig. 8.
HFSCs transplantation increased the level of synaptophysin and
PSD-95 expression. (A) The SYP protein level was assessed by western blot
analysis in the hippocampus of three groups. (B) The relative quantification of
protein expression. (C) The PSD-95 protein level was assessed by western blot
analysis in the hippocampus of three groups. (D) The relative quantification of
protein expression. Data were analyzed by one-way ANOVA with Tukey’s post-hoc
test (B,D), *p
AD is characterized by the presence of amyloid plaques and fibrillary tangles,
along with massive neuronal death and synaptic disruption in various brain
regions. Stem cell transplantation is one approach for treating AD. However, the
present study is the first attempt to treat AD model rats using HFSCs. Due to
advancements in genetic engineering, transplantation of transgenic stem cells
overexpressing specific factors can significantly enhance the reparative
potential of native stem cells. This potential restoration is attributed to
several mechanisms, including improved survival and differentiation capabilities
of the transplanted cells, apoptosis suppression, reduction in infarct volume,
and enhancement of neovascularization or functional recovery [35]. Herein, we
explored the potential mechanism of HFSCs and NGF-modified HFSCs on AD-like
behaviors in an A
A
Another common pathological change associated with AD is excessive tau protein
phosphorylation of NFTs in neurons. Notably, both A
Phosphorylation of GSK3
Synaptic proteins are crucial molecules that constitute the synaptic structure
and are closely related to neurotransmitter secretion and synaptic plasticity
[42]. Therefore, the expression levels of synaptic proteins can serve as
molecular markers of synaptic plasticity, particularly long-term-potentiation
capacity. Synaptic proteins include presynaptic and postsynaptic-membrane
proteins. A study has shown that soluble A
NGF is necessary for neuronal growth, survival, inhibition of apoptosis,
supporting stem cell survival, multiplication, differentiation, and enhancing
memory in AD rat models [47]. Furthermore, PI3K is important for NGF-stimulated
TrkA internalization [27]. Owing to NGF stimulation, the PI3K/Akt pathway
participates in enhancing cholinergic gene expression, neurite elongation [48],
and cell survival [49]. The activated Akt enzyme phosphorylates and inhibits its
downstream target, GSK3
HFSCs have drawn much attention because of their convenient accessibility, strong proliferation, and wide-ranging potential for differentiation [55, 56]. Consequently, they have excellent neural differentiation and neurotrophic abilities [18, 57]. In spinal-cord-injury rat models, transplanted HFSCs survive and proliferate within the injured rat spinal cord. These cells can differentiate into glial cell lineages (including astrocytes and oligodendrocytes) and neurons. Oligodendrocytes contribute to myelin repair around intact and demyelinated axons. Furthermore, hair-follicle stem cells express neurotrophic factors and other nutritional substances that collectively facilitate nerve repair and promote functional recovery [58]. In rats with ischemic stroke, transplantation of HFSCs significantly inhibited the activation of microglia to suppress inflammatory responses, enhanced the integrity of the blood-brain barrier, and reduced cerebral edema [59].
Recent investigations have indicated that HFSCs could secrete various
neuroprotective agents [60], which may explain the progressive improvement in the
performance of rats treated with HFSCs. Moreover, Tang [59] has shown that HFSCs
could regulate astrocyte stimulation through paracrine cytokines (transforming growth factor-
Overall, our data have indicated that HFSCs and NGF/HFSCs injections are a
promising therapy for restoring the behavioral decline that is a characteristic
symptom of AD. Our research results supported the concept that neuroprotection of
HFSCs and NGF/HFSCs is exerted by their capacity to reduce A
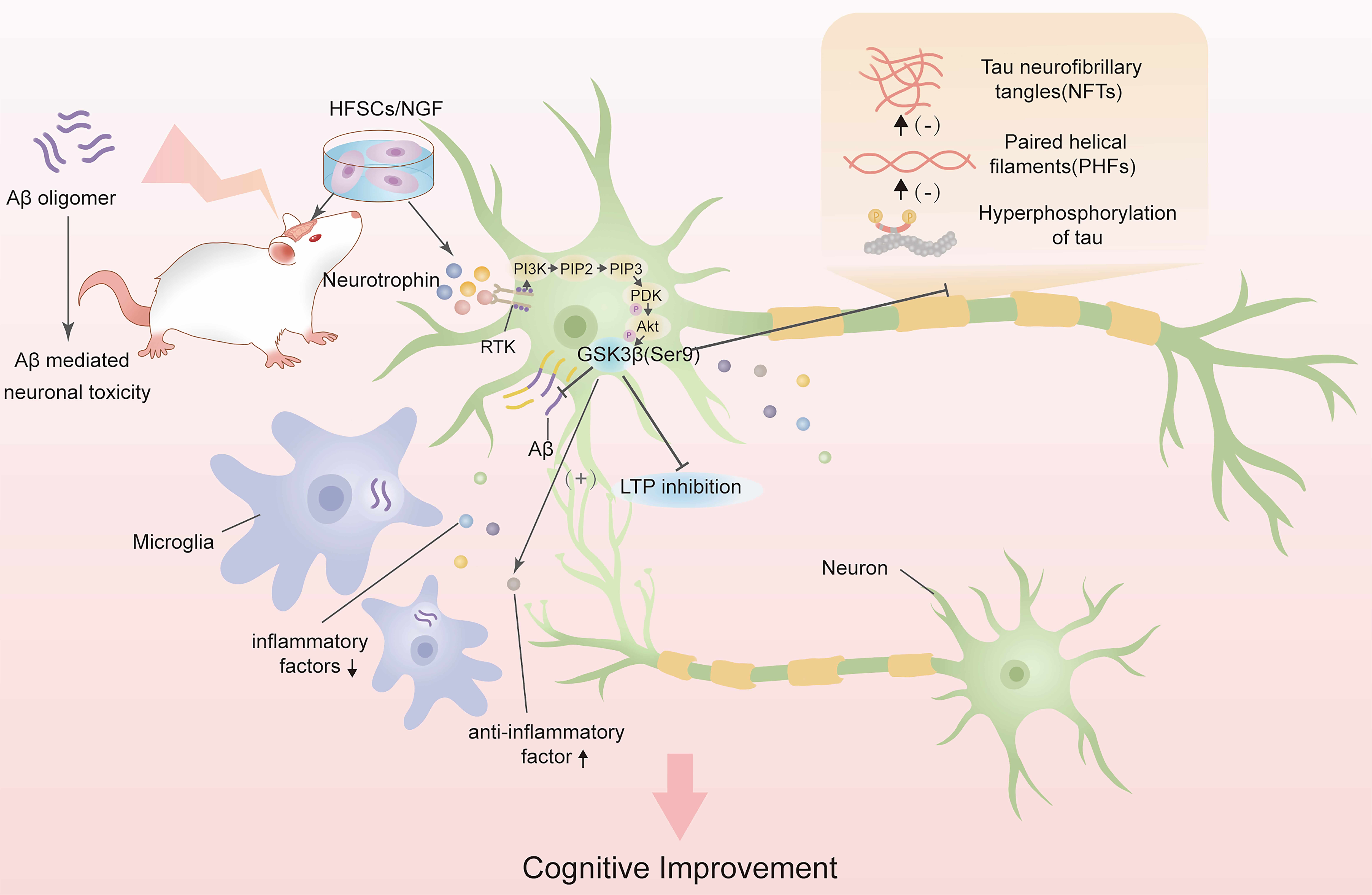 Fig. 9.
Fig. 9.
Overview diagram of the mechanism of HFSCs and HFSCs/NGF
treatment in AD rats. Dots: Signaling molecules (e.g., neurotrophic/inflammatory
factors). Arrows: Regulatory direction; “
Although our investigation offers proof for the application of HFSCs in the
therapy of AD rats, we acknowledge that further research about HFSCs in the
treatment of AD is necessary. The present study had some limitations, including
the need to clarify the in vivo expression, localization, and
differentiation of NGF-overexpressing HFSCs in the brain; the need to evaluate
the neuroinflammatory response induced during A
In this study, we discovered a new therapeutic effect of HFSCs that could serve
as a new treatment for AD. The utilization of HFSCs decreased A
The datasets generated during the current study are available from the corresponding authors upon reasonable requests.
DY and LL: conception and design, experiments execution, collection and assembly of data, data analysis and interpretation, manuscript writing; JF: conception and design, supervised the study, revised the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
All animal experiments were conducted in strict adherence to the principles outlined in the “Guide for the Care and Use of Laboratory Animals” published by the Ministry of Science and Technology of the People’s Republic of China. The experiment was approved by the Ethics Committee of the Second Affiliated Hospital of Harbin Medical University, with the approval number: SYDW2022-078.
We would like to thank Dr. Haitong Dou from the department of Neurology, The Second Affiliated Hospital of Harbin Medical University, for her guidance in cultivating cells.
This research received no external funding.
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/JIN43410.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.









