1 Department of Psychology, Sapienza University of Rome, 00185 Rome, Italy
2 Body and Action Lab, Istituto di Ricovero e Cura a Carattere Scientifico (IRCCS) Fondazione Santa Lucia, 00179 Rome, Italy
3 Departmental Faculty of Medicine and Surgery, UniCamillus-Saint Camillus International University of Health Sciences, 00131 Rome, Italy
Abstract
Electrophysiological studies have played a crucial role for the present conceptualization of Insomnia Disorder (ID) as a 24-h disorder characterized by hyperarousal expressed during wakefulness and sleep. In this Opinion piece, we highlight novel findings and delineate relevant future directions in the field of electroencephalographic (EEG) assessment in ID. Prolonged home recordings are crucial to provide ecological assessment also considering night-to-night variability. High-density EEG allows the description of local frequency-specific electrophysiological alterations in ID. A multimodal approach, combining EEG with neuroimaging techniques and non-invasive brain stimulation, may be informative about the neurophysiological mechanisms underlying ID and guide the development of targeted therapeutic strategies. Also, we highlight the need for longitudinal studies in this field. Novel approaches to quantitative EEG are represented by the assessment of aperiodic components and genuine oscillatory events. Finally, emerging research avenues include the assessment of sleep EEG hallmarks (e.g., sleep spindles and K-complexes) beyond their mere quantification, the application of artificial intelligence for automated identification and subtyping of ID, and EEG-based functional connectivity.
Keywords
- insomnia
- EEG
- polysomnography
- local sleep
- hyperarousal
Nowadays, Insomnia Disorder (ID) is mainly conceptualized as a 24-h disorder characterized by cognitive, behavioural, emotional, and physiological hyperarousal expressed during both wakefulness and sleep [1]. Research on electrophysiological markers of arousal in individuals with ID has played a crucial role for this conceptualization of ID.
Extensive assessment using classical polysomnographic (PSG) measures has provided findings about altered sleep macrostructure in ID (i.e., reduced total sleep time, prolonged sleep latency, increased wake after sleep onset, reduced slow-wave sleep and rapid eye movement (REM) sleep) [2]. However, results have been substantially inconsistent, and PSG is not considered essential for the diagnosis of ID. On the other hand, advanced analyses of the electroencephalographic (EEG) signals can provide more specific physiological indexes of cortical arousal, potentially improving our knowledge about ID pathophysiology, phenotypization, and neural mechanisms underlying treatment efficacy.
At present, the EEG assessment has revealed markers of cortical hyperarousal during wakefulness, REM, and non-rapid eye movement (NREM) sleep in ID patients [1, 3], and both theoretical and clinical interest in this kind of research is growing. A recent systematic review and meta-analysis on EEG spectral analysis in ID found (a) greater beta power during wakefulness and sleep, (b) increased theta and gamma power in wakefulness and alpha and sigma power during REM sleep, and (c) increased theta-to-sigma power combined with reduced delta power during NREM sleep [4]. Among these findings, the reduced level of NREM delta power and the general increase of high-frequency EEG activity may represent signs of cortical hyperarousal, whereas greater theta activity during wakefulness has been interpreted as an expression of daytime sleepiness and some effects of hypnotic medications [4]. Considering that different studies alternatively report absolute or relative EEG spectral power, it would be relevant to understand which stage-specific solution is more sensitive to ID. At present, absolute and relative EEG power measures exhibit similar sensitivity in the detection of ID-related EEG spectral properties during wakefulness and REM sleep, while relative power appears more sensitive to ID markers during NREM sleep compared to absolute power [4]. This issue, however, needs further systematic investigation.
Quantitative EEG analyses have also been conducted to determine if electrophysiological markers of hyperarousal may be related to sleep state misperception (SSM), which is the tendency to overestimate wake periods during the night and underestimate total sleep time. Such phenomenon is common among individuals with ID, but its underlying mechanisms are still unclear. Mounting evidence suggests that EEG indexes of cortical hyperarousal are associated with SSM during the sleep onset process and in both REM and NREM sleep stages [5, 6, 7, 8]. Crucially, some of these indexes (i.e., higher fast frequencies during the sleep onset and decreased slow frequencies both during the sleep onset and the whole night) may be specific for the ID phenotype characterized by SSM, not for ID per se [8].
Beyond quantitative EEG assessment, several studies have focused on the role of specific sleep EEG phenomena in ID. Alterations in sleep spindles — NREM hallmarks represented by oscillatory bursts of neural activity around 11–15 Hz involved in sleep stability- have been observed, despite findings are still inconsistent [9, 10, 11]. However, it is worth noting that such literature is limited and the spindle features investigated are heterogeneous. Concerning the K-complexes (KC) — isolated downstates during NREM sleep that contribute to sleep protection and arousal modulation during sleep — early studies found mixed results, mainly suggesting the absence of relevant KC alterations in ID [10, 12, 13, 14]. However, the few available studies were often conducted on a single anterior EEG derivation and focused on KC density. More recently, the investigation of both quantitative and morphological features of KCs in different midline cortical derivations in ID as well as their relationship with SSM and sleep architecture, showed a specific reduction of frontal KC amplitude and area under the curve in ID, without KC density alterations, associated with SSM [15]. This result can be interpreted in light of the concept of hyperarousal: the KC morphological reduction in ID may represent the cortical expression of impaired sleep-protection mechanism during NREM sleep associated with the 24-h hyperarousal characterizing ID. These findings point to sleep EEG hallmarks as a promising area of research in ID, albeit greater methodological homogeneity and wider attention to the phenomenological aspects of these EEG phenomena are still needed. Moreover, an extensive assessment of the temporal coordination between sleep EEG oscillations in ID would provide more information about possible ID-related alterations of sleep electrophysiological processes. The temporal coupling between NREM sleep oscillations is crucial for overnight information transfer [16], supports the efficiency of memory pathways [17], and may have a role in sleep continuity, subjective sleep depth, and sleep time misperception in ID. Anomalies in slow oscillations-spindles coupling are associated with memory impairment, brain atrophy, and neurodegeneration [18, 19]. A recent study found no ID-related changes in phase-amplitude coupling between sleep slow oscillations and spindle activity [20], but this issue should be further investigated.
Given the heterogeneous nature of ID, some studies have emphasized the need to identify different subcategories of insomnia, based on life history, affect, and personality traits [21]. One EEG aspect that would appear to define ID across its different phenomenology is the increased frequency of transition from stage N2 to stage N1 or wakefulness [22], independently of any conventional sleep parameter. In this view, the potential use of sleep EEG patterns to define possible ID subtypes represents an interesting area of research. A promising research avenue is focused on artificial intelligence (AI)-based methods to automatically identify and potentially subtype ID starting from EEG signals. Specifically, AI have the potential to enable analyses of the EEG complexity that would be impossible manually, extracting and integrating multiple EEG features (and other sources of information) to provide scores used to discriminate ID and its subtypes. Several AI-based methods have been used by different research groups to support ID diagnosis and classification [23, 24, 25, 26]. A recent study demonstrated the ability of a machine learning model applied on a robust combination of EEG-derived features to distinguish individuals with ID from individuals without ID diagnosis, also providing a quantitative “Insomnia EEG Score” (IES) [27]. The authors suggest that this model, trained on the largest ID dataset available, may provide the basis for an ID subtyping based on electrophysiological features. The scarcity and heterogeneity of large EEG databases of ID individuals, the difficult interpretability of complex composite scores, the absence of a standardized methodology, and the limited clinical validations of these measures represent the main challenges in this field.
Finally, recent studies have provided novel insights about the effect of pharmacological [28, 29] and non-pharmacological interventions [30, 31, 32] on sleep EEG measures. Moreover, baseline EEG may predict ID treatment outcome. Indeed, lower pre-treatment sleep spindles density during NREM sleep stages predicted lower improvement in self-reported sleep measures collected at 12-month follow-up [33]. These findings support the view that the assessment of cortical electrophysiology may be useful to understand the neural mechanisms underlying specific treatments, their side effects, and to predict and monitor their efficacy.
In line with this view, several studies have investigated the modulation of cortical electrophysiology in ID using non-invasive brain stimulation (NIBS) techniques [34, 35] to provide a better understanding of ID neural mechanisms and produce potential clinical applications. Multiple clinical trials showed that repetitive transcranial magnetic stimulation (rTMS) significantly improves subjective and objective sleep quality in ID, thanks to its ability to modulate cortical excitability and neural plasticity, potentially reducing hyperarousal [36]. Furthermore, transcranial alternating current stimulation (tACS) has the potential to entrain neural oscillations relevant to sleep, enhancing the probability to increase sleep propensity [37]. Different tACS protocols have been used in ID to successfully mitigate self-reported nocturnal symptoms [38] and investigate hyperarousal from a novel perspective, demonstrating the role of increased brain-heart interplay (BHI) in the pathophysiology of ID [39]. Also, recent studies were focused on the efficacy of personalized, state-dependent closed-loop tACS protocols in normalizing sleep in individuals with ID, thanks to the possibility to synchronize the stimulation frequency with the predominant individualized neural oscillation frequency specific to each state [40], and the possibility to modulate brain connectivity [41]. In this view, the possibility to provide personalized EEG-based strategies for improving treatment outcomes through the application of tACS protocol should be considered in future studies. Although these techniques could represent promising non-pharmacological treatments for ID, the heterogeneity of the stimulation protocols used, and the small sample sizes require caution in generalizing the results. Nonetheless, this area has offered new insights regarding the electrophysiological mechanisms underlying ID and its treatment.
Overall, EEG assessment in ID has been fundamental for the conceptualization of the role of hyperarousal in ID, and the literature in this area is growing, progressively allowing a better understanding of ID pathophysiology, phenotypes, symptoms, and treatment. However, further efforts are needed in this field (Fig. 1). First, starting from the consolidated view of sleep as a local phenomenon, more data are required regarding the topography of electrophysiological findings in ID. In this view, high-density EEG studies are needed. Also, a multimodal approach, combining EEG with other neuroimaging techniques, may be particularly informative about the neurophysiological mechanisms underlying ID, facilitating the detection of specific biomarkers and guiding the development of targeted therapeutic strategies. Nevertheless, this approach is still underutilized in this field. Additionally, the assessment of regional EEG power should be more frequency-specific, and the evaluation of specific sleep EEG hallmarks like sleep spindles and KC in ID should be addressed to describe not only their occurrence but also their phenomenology (e.g., duration, morphology), reciprocal interaction (e.g., temporal coupling), overnight changes and relationship with diurnal symptoms. Considering the night-to-night variability of ID symptomatology, home recordings over prolonged periods would provide more accurate and ecological measures compared to a single laboratory recording. Also, longitudinal studies would be relevant to understand if EEG peculiarities may be considered predisposing factors for ID. In this view, the possibility to use wearable devices and deep learning pipelines, combining simplified EEG recordings with other sources (e.g., accelerometry and heart rate) for extended periods and analyzed with machine learning methods, may provide a relevant approach for scalable screening, longitudinal monitoring, ID subtyping, and mechanism testing. The quantitative analysis of EEG signals, like the assessment of genuine oscillatory events would improve our knowledge about the meaning of specific neural alterations in ID. In particular, the aperiodic component of the EEG power spectrum, reflecting the balance between excitatory and inhibitory neural activity [42], may provide insightful information about the neural correlates of ID. A recent study found a reduced aperiodic exponent, considered an index of an impaired inhibitory neural activity, during resting-state EEG recordings in ID individuals compared to healthy controls [43], promoting this measure as a promising tool for a deeper understanding of the neural basis of the concept of hyperarousal in ID. Finally, our knowledge about EEG-based connectivity in ID should be improved. Indeed, fMRI studies suggest an association between ID symptomatology and impaired intra- and inter-hemispheric interactions in several resting-state networks, supporting the hypothesis that ID is characterized by altered organization of the brain functional connectome, resulting in cognitive, emotional, and sleep-related alterations [44]. However, EEG-based connectivity studies are scarce and heterogeneous [44]. A recent study using source-localization EEG showed significant hyperconnectivity in the gamma frequency band, especially in the brain regions involved in cognitive and sensory processing [45], suggesting an association with greater sleep fragmentation. Li and coworkers [46] assessed the coherence across multiple EEG frequency bands in ID during wakefulness and sleep stages. Individuals with ID exhibited changes in alpha and delta band connectivity during wakefulness, mainly between anterior and posterior regions, and sleep stage-specific connectivity alterations, with increased alpha connectivity in NREM sleep and enhanced delta connectivity in phasic and tonic REM sleep. Interestingly, the pattern of connectivity changes in ID had a mediator role in the relationship between sleep disruption and emotional regulation [46]. Interventional studies found that EEG-derived connectivity measures predicted the therapeutic effect [47] and showed post-treatment changes [48, 49, 50] when rTMS was applied in ID. Finally, a recent work suggests that EEG-derived functional connectivity measures may predict the therapeutic effects of sleep-aiding music in ID [51]. In this view, a larger use of sleep and wake EEG recordings for the assessment of brain connectivity would also provide a fundamental step forward in this field.
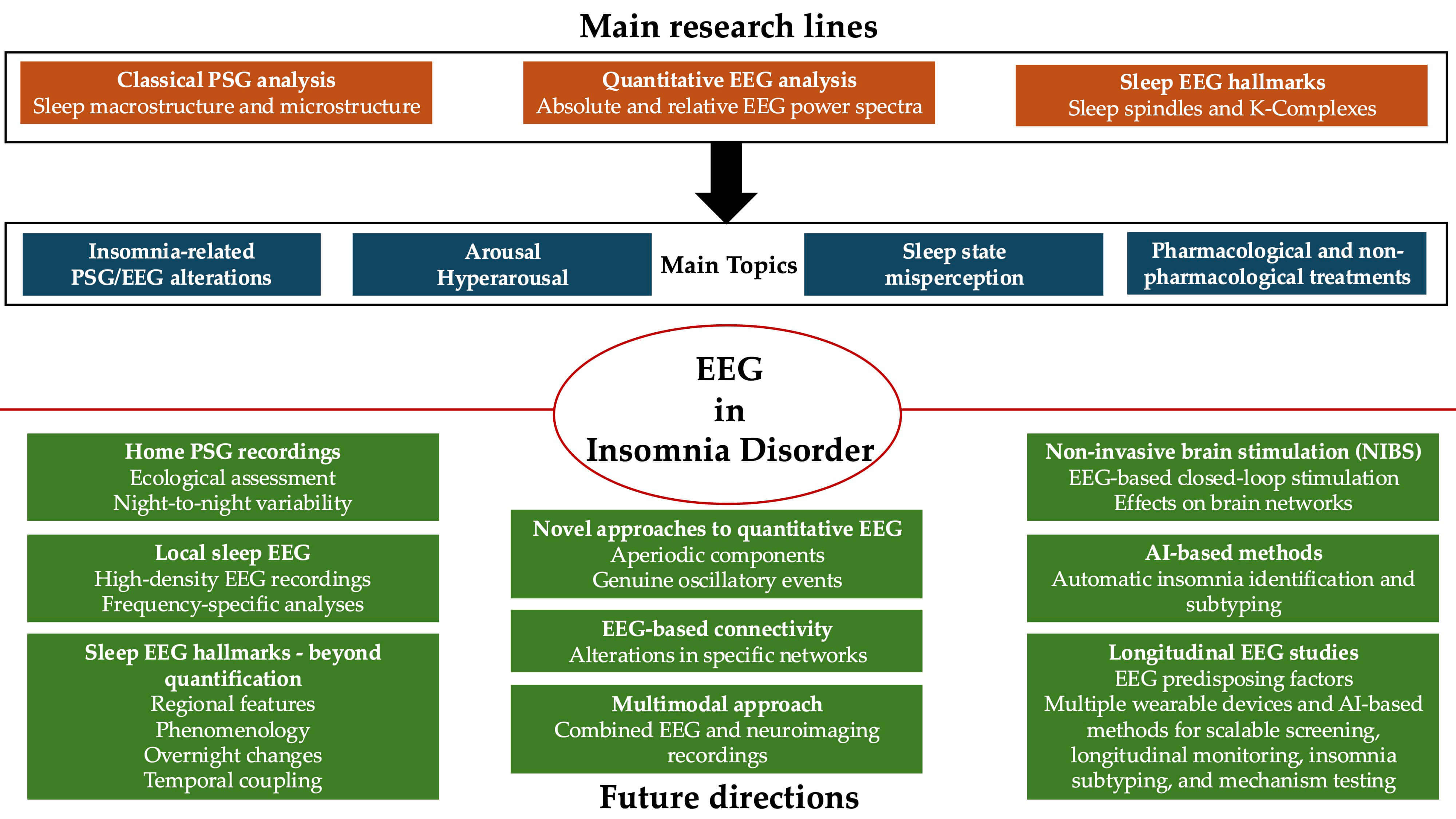 Fig. 1.
Fig. 1.
Main research lines, topics of interest, and future directions in the field of electroencephalographic (EEG) assessment in Insomnia Disorder. PSG, polysomnography.
MG, VA, and LDG wrote the manuscript and conducted a literature review. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest. Luigi De Gennaro is serving as one of the Editorial Board members of this journal. We declare that Luigi De Gennaro had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Bettina Platt.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

