1 Department of Neurology, First Affiliated Hospital of Harbin Medical University, Harbin Medical University, 150001 Harbin, Heilongjiang, China
2 Department of Neurology, Fourth Affiliated Hospital of Harbin Medical University, Harbin Medical University, 150001 Harbin, Heilongjiang, China
3 Key Laboratory of Acoustic, Optical and Electromagnetic Diagnosis and Treatment of Cardiovascular Diseases, 150001 Harbin, Heilongjiang, China
Abstract
Cerebral ischemia-reperfusion injury (CIRI) represents the most critical pathological event in the evolution of ischemic stroke (IS). Apoptosis is particularly important in CIRI pathophysiology. The interleukin-7 receptor (IL7R) is involved in various disease regulatory mechanisms; however, its specific role during CIRI remains unclear. We investigated the mechanistic function of IL7R in CIRI through a mouse model in vivo and through an astrocyte model in vitro.
C57BL/6 mice were randomly allocated to one of five groups: (1) sham; (2) transient middle cerebral artery occlusion (tMCAO); (3) tMCAO + IL7R treatment; (4) tMCAO + negative control (NC); or (5) tMCAO + IL7R + the phosphatidylinositol 3-kinase (PI3K) pathway inhibitor (LY294002) (n = 3–7 per group) to evaluate the role of IL7R in CIRI. The in vitro study groups were (1) control; (2) oxygen-glucose deprivation/reoxygenation (OGD/R); (3) OGD/R + IL7R; (4) OGD/R + NC; and (5) OGD/R + IL7R + LY294002 groups. After IL7R overexpression was induced, the resulting changes in infarct volume, neurological score, cell viability, and expression of apoptosis-related proteins were assessed.
IL7R overexpression significantly attenuated CIRI-induced apoptosis. In vivo, this intervention improved neurological function, alleviated cerebral edema, and decreased infarct volume in tMCAO mice. In vitro, after the overexpression of IL7R, flow cytometry analysis revealed a reduction in apoptosis rates post-OGD/R, whereas transmission electron microscopy revealed fewer morphological alterations associated with apoptosis. In addition, the level of Bcl-2-associated X protein (Bax) and cysteine-dependent aspartate-specific Protease-3 (caspase-3) were decreased, whereas that of B-cell lymphoma-2 (Bcl-2) was increased; these effects were reversed by LY294002.
Overexpression of IL7R was shown to alleviate CIRI by suppressing apoptosis. These findings indicate IL7R as a novel target for IS treatment.
Keywords
- apoptosis
- IL7R
- ischemic stroke
- PI3K/Akt
- reperfusion injury
Cerebrovascular diseases rank as one of the leading global causes of mortality [1]. Ischemic stroke (IS), an aggressive and widespread subtype of acute stroke [2]. After ischemia, the resulting restoration of blood supply can paradoxically worsen brain damage, a condition called cerebral ischemia–reperfusion injury (CIRI). CIRI is implicated in relatively high rates of mortality and disability, greatly influencing both the health of individuals and their quality of life [3, 4]. Given its high incidence, the underlying mechanisms of CIRI remain elusive. The induction of apoptosis is a key event in CIRI. The reactive oxygen species (ROS) are rapidly produced after ischemia initiates the processes of neuronal death, brain edema, and subsequent cellular damage [5]. During reperfusion, ROS and oxidative stress (OS) can even cause the apoptosis of cells, aggravating brain damage [6]. Hence, suppressing OS and inhibiting apoptosis are essential measures to improve CIRI outcomes, indicating the need to search for possible treatment targets.
Interleukin-7 (IL-7) is a cytokine of four antiparallel alpha-helices that bind
to cytokine receptors known as type I cytokine receptors. Stromal cells
constitute the primary source of IL-7. IL-7 is necessary for the ontogeny and
maintenance of lymphocytes [7], with a role in immune system homeostasis [8].
IL-7 acts by using the IL-7 receptor (IL7R), which is a heterodimeric protein
comprising an
Stroke, a prevalent neurological disorder with substantial global health implications, continues to demand innovative treatment solutions. The present study proposed an innovative therapeutic strategy for IS through the modulation of IL7R expression. Through in-vivo and in-vitro experiments, elevated IL7R expression mitigates CIRI was revealed through the suppression of apoptosis, which is mediated by PI3K/Akt signaling pathway activation. These results establish IL7R as a viable candidate for developing targeted interventions against CIRI.
The study design complied with the Animal Research: Reporting of In Vivo Experiments (ARRIVE) guidelines [20]. Male C57BL/6 mice, ranging in weight from 20 to 25 g, were used for this investigation. All Specific Pathogen Free (SPF)-grade mice were obtained from Liaoning Changsheng Biotechnology Co., Ltd. (Benxi, Liaoning, China) and were maintained by animal-care protocols established by the First Affiliated Hospital of Harbin Medical University. Throughout the study, the mice were housed under strictly regulated conditions with a 12/12-h photoperiod and an ambient temperature maintained at 23–25 °C. Under anesthesia, the mice were kept at a constant 37 °C. Mice were randomly assigned to the following groups: (1) sham; (2) transient middle cerebral artery occlusion/reperfusion (tMCAO/R); (3) tMCAO/R + negative control (NC); (4) tMCAO/R + IL7R; and (5) tMCAO/R + IL7R + the PI3K pathway inhibitor (LY294002). A total anesthetic and analgesia were administered during every surgical procedure. The detailed grouping criteria and the mice that died or bled are already provided in the Supplementary Table 1, Experiment 1–3.
Hanheng Biological Technology (Shanghai, China) Co., Ltd. supplied the AAV
serotype 9 expressing IL7R (AAV-IL7R; Titer: 1.5
LY294002 (S1737, Beyotime Biological Co., Ltd., Shanghai, China) was dissolved in phosphate-buffered saline containing 25% Dimethyl sulfoxide (DMSO, ST038, Beyotime Biological Co., Ltd.) at 50 mM. After dilution, 2 µL of the solution was administered by intracerebroventricular injection to each mouse 1 h before tMCAO induction [22]. LY294002 (a concentration of 10 µM) was supplemented to C8D1A cells (CL-0506, Procell Life Science&Technology Co., Ltd., Wuhan, China) 1 h before oxygen-glucose deprivation/reoxygenation (OGD/R) treatment [23].
The middle cerebral artery (MCA) was occluded in an ischemia model using line embolism. To summarize, anesthesia was induced with 1.25% avertin (0.02 mL/g, i.p.). When a mouse showed signs of awakening during surgery, an additional dose of 1/2 or 1/3 of the original anesthetic was administered. The mouse’s vital parameters were continuously tracked to ensure successful completion of the surgery. The procedure involved shaving and sterilizing the neck in preparation for a midline incision that would expose the common carotid artery (CCA), internal carotid artery (ICA), and external carotid artery (ECA). Using surgical thread, the proximal end of the CCA, the distal end of the ICA, and both ends of the ECA were ligated. To occlude the MCA successfully, a tiny incision was made between the ligatures of the ECA. A line embolism then progressed into the ICA through the carotid bifurcation until it reached a mark of approximately 1 cm. After the surgery was completed, the mouse was placed on a heating pad. The line embolism was removed, and the incision was sutured after 1 h of tMCAO. After the mouse regained consciousness, it was placed in an incubator with temperature control for a full day. The only difference in the protocol of the sham group was the absence of MCA occlusion. Reducing the wound size without exposure was the goal of keeping the neck incision small. To ensure the mouse’s comfort and rapid recovery, it was kept on a heating pad set at 37 °C from the time it was anesthetized until it regained consciousness [24]. The mice were randomly divided into each experimental group. The sham-operated mice underwent identical surgical exposure without MCA occlusion. The survival rate of the mice reached 90%, and the mice that died or bled were excluded.
To evaluate neurological impairments in the mice, the modified Longa score was used. Each mouse underwent neurological function testing, and a researcher who was blind to the aim of the experiment assessed the mice. The scoring system was as follows: 0, no abnormalities; 1, trunk turning to the ipsilateral side when the tail was grasped; 2, the contralateral forelimb showed incomplete extension; 3, turning toward the paralyzed side; 4, leaning toward the opposite side; 5, inability to walk independently, loss of consciousness, or death. If the score was between 1 and 4, the tMCAO model was deemed successful [25]. Mouse data were excluded from the study if (1) the mouse’s score was 0 or 5 at the time of euthanasia; (2) the mouse died before euthanasia; or (3) the mouse showed signs of subarachnoid or intraparenchymal hemorrhage.
The mouse was euthanized, and its brain was removed and cut into 6 coronal
sections, each 1 mm thick, beginning posterior to the frontal lobe. After 30 min
of dark incubation in 2% TTC (G3005, Solarbio, Beijing, China) at 37
°C, the tissue sections were fixed in 4% paraformaldehyde (SI101-01,
Sevenbio, Beijing, China). We used ImageJ software (1.53k, National Institutes of
Health, Bethesda, MD, USA) to measure the infarct volume after imaging the brain
slices. The infarct volume measured by TTC staining was corrected for edema using
the formula: corrected infarct volume = volume of the contralateral hemisphere –
volume of the viable tissue in the ipsilateral hemisphere. The infarction volume
(%) = (contralateral hemisphere volume – ipsilateral hemisphere viable tissue
volume)/contralateral hemisphere volume
The brains were obtained without perfusion, immediately weighed (wet weight).
The tissue was dried in an oven for 12 h and re-weighed (dry weight), and the BWC
percentage was calculated as: [(wet weight – dry weight)/wet weight]
The mice’s brains were harvested 24 h after tMCAO. After the blood was completely removed by perfusion with 4 °C saline, the mice were perfused with 4% paraformaldehyde until they became stiff. Coronal sections (7 µm thick) prepared using a cryostat (NX50, Thermo Fisher Scientific, Waltham, MA, USA) underwent immunofluorescence staining. After 20 min permeabilization in 0.5% Triton X-100 (BL3087A, Biosharp, Jiangsu, China) at room temperature, sections were blocked with 10% goat serum (AR0009, BOSTER, Wuhan, China) for 1 h. Primary antibodies targeting IL7R (1:100, TD6362, Abmart, Shanghai, China), glial fibrillary acidic protein (GFAP, 1:100, MU184607, Abmart), allograft inflammatory factor 1 (IBA1, 1:1000, MU146067, Abmart), and neuron-specific nuclear protein (NEUN, 1:1000, M050661, Abmart) were applied overnight at 4 °C, followed by 1 h incubation with suitable fluorophore-conjugated secondary antibodies (ZF-0516, ZF-0512 ZSBIO, Beijing, China) at room temperature under light-protected conditions. Nuclear counterstaining was achieved via 4′,6-Diamidino-2-phenylindole (DAPI, BL739A, Biosharp). Fluorescence images were captured using a fluorescence microscopy platform (Nikon Eclipse Ti2, Tokyo, Japan).
RNA isolation was performed using distinct protocols: TRIzol reagent
(15596026CN, Invitrogen, Thermo Fisher Scientific) was used for brain tissue
samples, and total RNA extraction reagent (BSC51, Biozol, Hangzhou, China) was
used for C8D1A cells. After RNA isolation, genomic DNA extraction, and cDNA
synthesis were conducted according to the reverse transcription kit (FSQ301,
ToYoBo, Tokyo, Japan). The sequences of primers used were as follows:
IL7R-F, 5′-ACGATTACTTCAAAGGCTTCTGGAG-3′; IL7R-R,
5′-AATGGTGACACTTGGCAAGACAG-3′; Bcl-2-F,
5′-CTACGAGTGGGATGCTGGAGATG-3′; Bcl-2-R,
5′-GGTTGCTCTCAGGCTGGAAGG-3′; Bax-F,
5′-GGAGACACCTGAGCTGACCTTG-3′; Bax-R,
5′-GCTCCATATTGCTGTCCAGTTCAT-3′; Caspase3-F,
5′-GACTGGAAAGCCGAAACTCTTCAT-3′; Caspase-3-R,
5′-AGTCCCACTGTCTGTCTCAATG-3′;
At 24 h after ischemia/reperfusion (I/R) induction, protein samples were
obtained from the right hemisphere and C8D1A cells. Protein concentration was
quantified using a BCA assay kit (P0010S, Beyotime Biological Co., Ltd.).
Electrophoresis was conducted on sodium dodecyl sulfate polyacrylamide gel
electrophoresis (SDS-PAGE) gels (12.5% or 10%) loaded with 30 µg
of protein per sample. After 1 h of blocking with 5% skim milk at ambient
temperature, the membranes were subjected to primary antibodies overnight at 4
°C. The secondary antibodies used were goat anti-rabbit (S0001, 1:3000,
Affinity, Jiangsu, China) and goat anti-mouse (1:5000, ZB-2305, ZSBIO) for 1 h.
Three Tris Buffered Saline Tween (TBST) washes preceded chemiluminescent
detection using the BL520A ECL substrate (Biosharp). The primary antibodies used
included IL7R (1:1000, TD6362, Abmart), caspase-3 (1:1000, T40044, Abmart), Bax
(1:1000, T40051, Abmart), Bcl-2 (1:1000, T40056, Abmart), PI3K (1:1000, T40115,
Abmart), phosphorylated PI3K (1:1000, T40116, Abmart), Akt (1:1000, 4691, Cell
Signaling Technology, Danvers, MA, USA), and phosphorylated Akt (1:1000, 4058,
Cell Signaling Technology), with GAPDH (1:150,000, 60004-1, Proteintech, Wuhan,
China) and
The C8D1A (CL-0506, Procell Life Science & Technology Co., Ltd., Wuhan, China ) astrocyte cell line has undergone STR identification and mycoplasma testing, which confirmed that the quality of the C8D1A cell line is correct and there is no mycoplasma contamination. It was cultured in complete medium consisting of Dulbecco’s modified Eagle’s medium (DMEM, CM-0506, Procell Life Science&Technology Co., Ltd.) supplemented with 10% fetal bovine (CM-0506, Procell Life Science&Technology Co., Ltd.) serum and 1% penicillin/streptomycin [26]. The cell lines were authenticated using DNA short tandem repeat analysis. Cultured cells were placed in a tri-gas incubator with 5% CO2, 1% O2, and balanced N2 for 3 h after being cultured in glucose-free DMEM (NaHCO3, glutamine, sodium pyruvate, serum-free, pH: 7.2–7.4) to induce OGD/R [27]. The cells were then cultivated for 2, 6, 12, 24, or 48 h under normoxic conditions (95% air, 5% CO2) after restoration of complete medium. The cells that were used as controls were those that were kept under anoxic conditions. This study divided the cells into 5 groups: (1) control; (2) OGD/R; (3) OGD/R + NC; (4) OGD/R + IL7R; and (5) OGD/R + IL7R + LY294002.
A lentivirus with a titer of 108 TU/mL was generated by cloning the coding sequence of the target gene into the LV5 vector. Gemma Gene Co., Ltd. (Suzhou, Jiangsu, China) generated the lentiviral construct and oversaw its synthesis. To measure the success of transfection, a virus that did not express any GFP was used as a negative control [28]. Once the transfection efficiency surpassed 70%, stable cell line selection was initiated. After lentiviral transfection, stably transfected cells, also known as mixed clones, were selected with puromycin (G04010, Genepharma, Suzhou, China) as a selection factor. The cells were grown and expanded after selection. After that, RNA was taken from the overexpression group and the negative control group for RT-qPCR.
C8D1A astrocytes (4500 cells/well, 96-well plates) were subjected to transfection and OGD/R [29], then incubated with 10 µL CCK-8 (C0037, Beyotime Biological Co., Ltd.) for 1 h. Cell viability was quantified by absorbance at 450 nm on a microplate reader integrated with an optical density detection system (EPOCH, BioTek, Winnsboro, VT, USA).
The rate of neuronal cell apoptosis was measured using the Fc method. After induction of OGD/R and lentiviral transduction, the C8D1A cells were harvested. The cells were assessed by Fc from an Annexin V-APC/7-AAD Apoptosis Detection Kit (abs50008, Absin Biotechnology Co., Ltd., Shanghai, China) [30].
The C8D1A cell samples were fixed, embedded, dehydrated, and cut into thin slices (60–70 nm). An imaging TEM microscope (H-7700, Hitachi, Tokyo, Japan) was used to examine and photograph these slices.
Data analysis was conducted with GraphPad Prism version 8.0 software (GraphPad
Software, San Diego, CA, USA), and the experimental results are expressed as the
means
To explore the involvement of IL7R in CIRI, we conducted temporal expression
profiling of this receptor in the right cerebral hemisphere after tMCAO. Compared
with controls, IL7R expression was significantly down-regulated following I/R
injury. WB analysis revealed a time-dependent reduction in the IL7R protein
level, with the lowest expression observed at 24 h postinjury (Fig. 1A). A marked
reduction in IL7R expression was detected at 24 h post-tMCAO (p
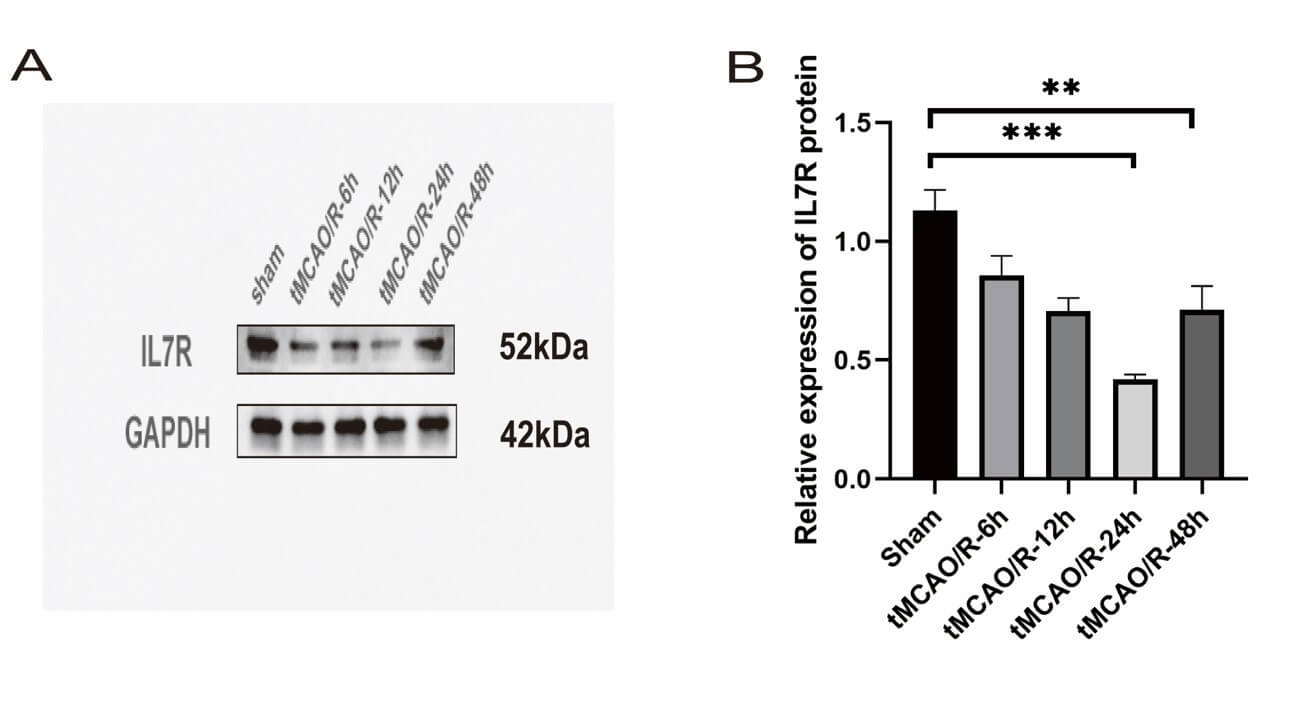 Fig. 1.
Fig. 1.
IL7R protein levels are suppressed at multiple post-tMCAO time
points. (A) IL7R protein levels were detected by WB (n = 7 per group).
(B) The quantitative analysis of IL7R protein in different groups. Data are shown
as mean
In normal brain tissue supplied by the middle cerebral artery of the mice, we
performed double IF staining of IL7R with specific markers for astrocytes (GFAP),
microglia (IBA1), and neurons (NEUN) (Fig. 2A). IL7R was colocalized with
astrocytes, and IL7R was expressed in astrocytes. IL7R was hardly expressed in
microglia (Fig. 2B). IL7R was rarely expressed in neurons (Fig. 2C). The Pearson
correlation coefficients for these three groups of data were calculated with
ImageJ software (Fig. 2D); |r|
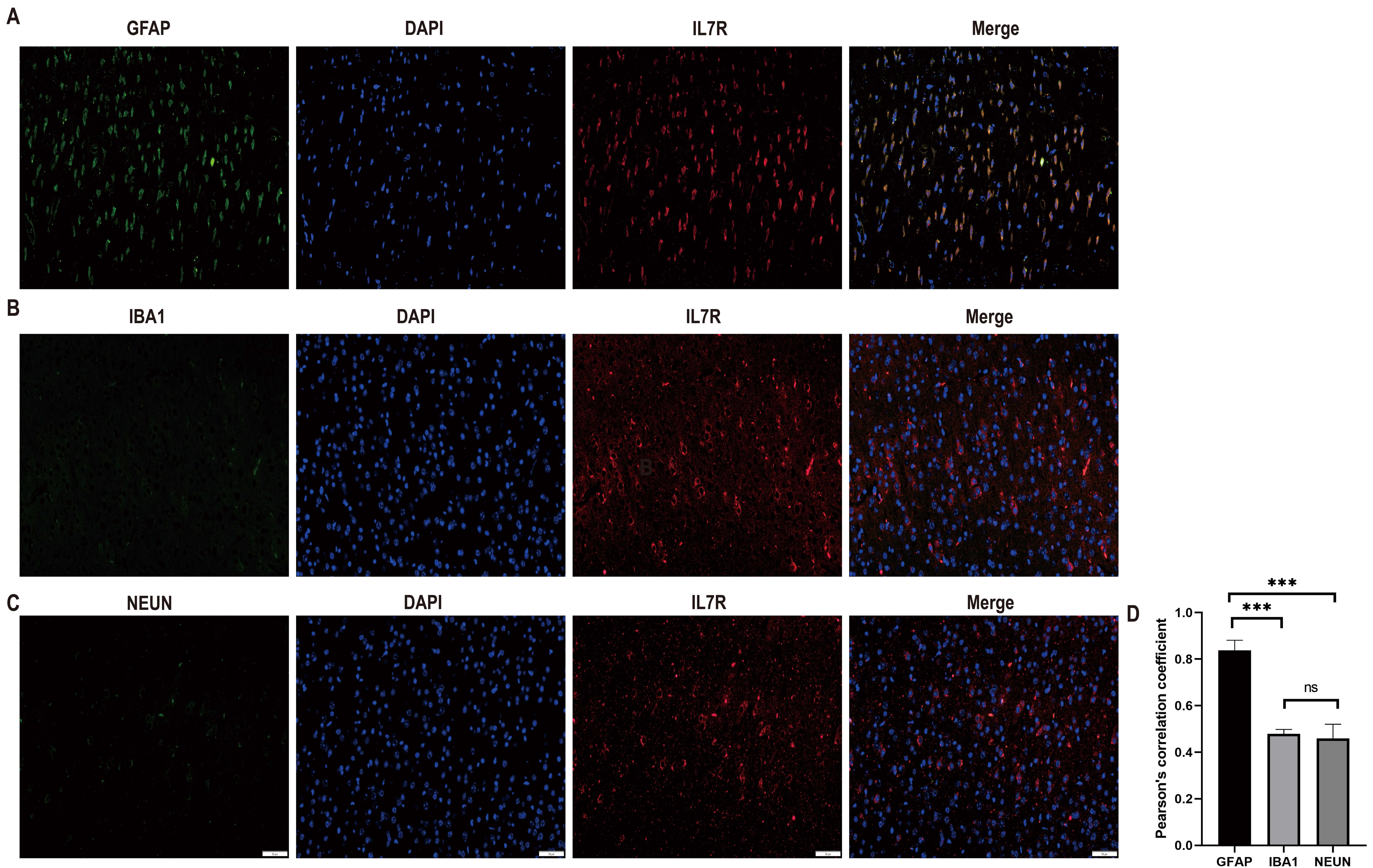 Fig. 2.
Fig. 2.
IF double–staining of IL7R, GFAP, IBA1, and NEUN in mouse brain
tissue. (A) Representative images of double-IF staining showing the
colocalization of IL7R (red)/GFAP (green)/DAPI (blue). (B) Representative images
of double-IF staining showing the colocalization of IL7R (red)/IBA1 (green)/DAPI
(blue). (C) Representative images of double-IF staining showing the
colocalization of IL7R (red)/NEUN (green)/DAPI (blue). (Scale bar = 50 µm for Fig. 2A–C).
(D) The Pearson correlation coefficients for these three groups (n = 3
per group). Data are shown as mean
AAV-IL7R was injected into the lateral ventricle of mice to induce IL7R
overexpression. In mouse brain sections of the MCA supply area treated with
AAV-GFP (AAV-ZsGreen, 1.3
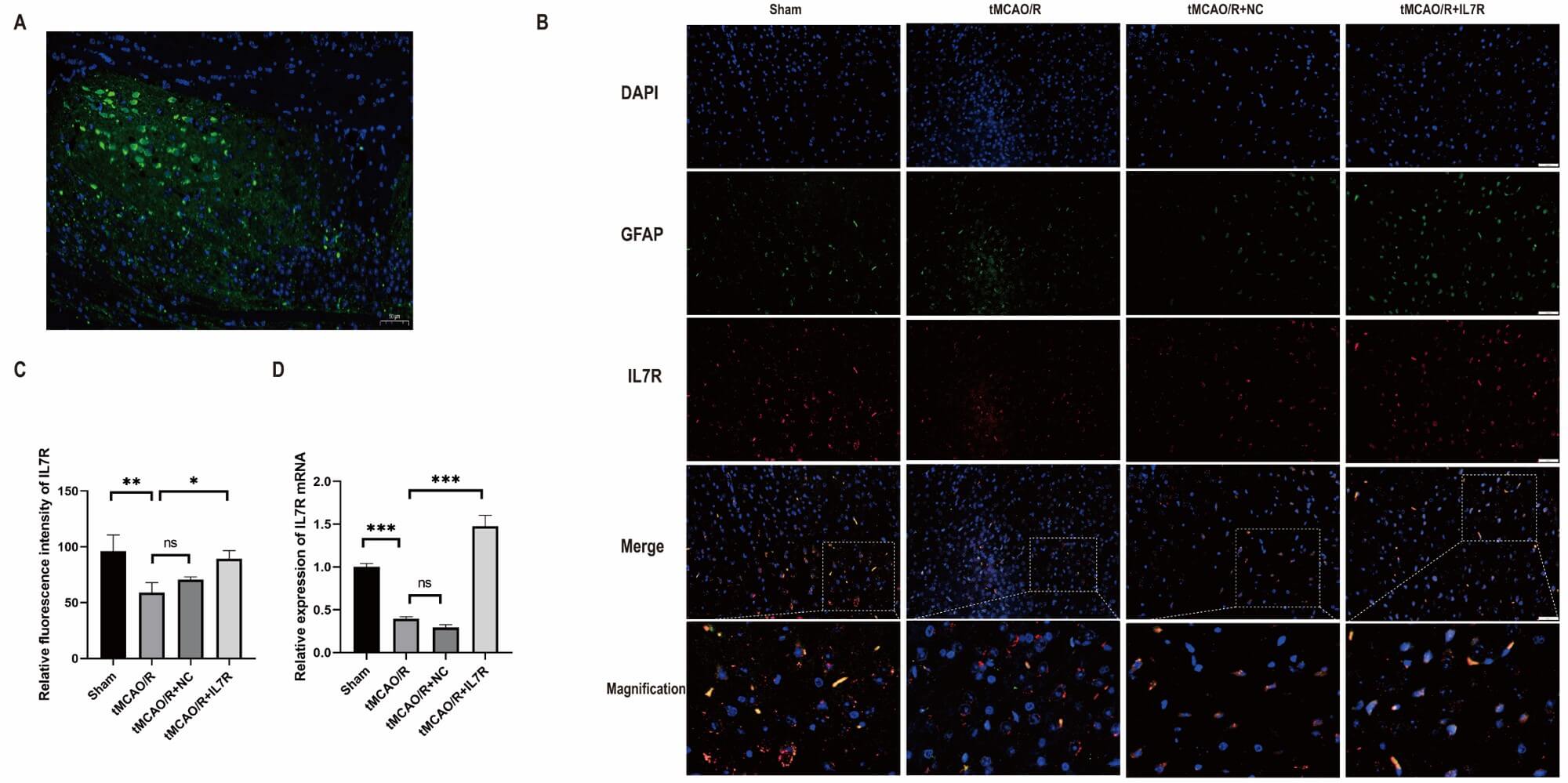 Fig. 3.
Fig. 3.
Verification of successful overexpression of IL7R in tMCAO/R
mouse brain tissue. (A) Representative IF image of AAV-GFP-transfected brain
sections from mice. (Scale bar = 50 µm). (B) Representative images of
double-IF staining showing the colocalization of IL7R (red)/GFAP (green)/DAPI
(blue). (Scale bar = 50 µm). (C) Statistical results of IF staining for
IL7R in the groups (n = 3 per group). (D) Relative mRNA levels of
IL7R in the sham, tMCAO/R, tMCAO/R + NC, and tMCAO/R + IL7R groups
(n = 3 per group). Data are shown as mean
In our previous studies, we observed minimal IL7R expression at 24 h post-tMCAO
(Fig. 1A), which prompted us to harvest brain tissues at this time point. To
determine whether IL7R overexpression mitigates CIRI, we conducted TTC staining
(Fig. 4A). Mice treated with IL7R-AAV presented smaller cerebral infarction
volumes (Fig. 4A,B, p
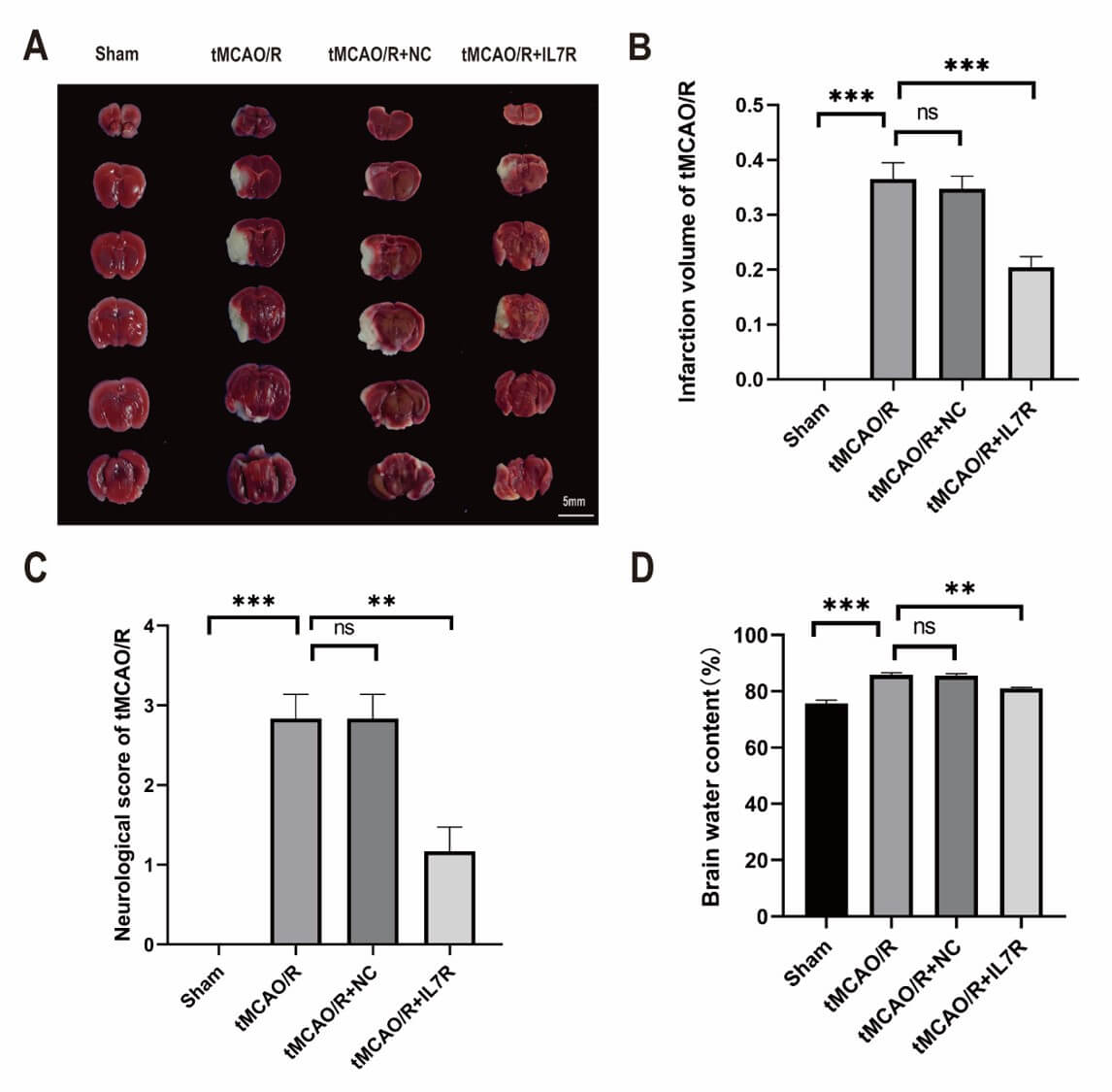 Fig. 4.
Fig. 4.
Overexpression of IL7R enhanced cerebral protection by reducing
infarct volume, BWC, and neurological deficit scores in vivo. (A) TTC
staining of brain tissues after tMCAO/R. (Scale bar = 5 mm). (B) Statistical analysis of the TTC
staining results (n = 6 per group). (C) Neurological deficit scores of
the mice post-tMCAO/R (n = 6 per group). (D) BWCs in the cerebral
hemisphere were measured using the wet and dry weight method (n = 6 per
group). Data are shown as mean
Apoptosis is a critical process in CIRI. Therefore, we investigated the effects
of IL7R on apoptosis. The RT-qPCR data revealed substantial elevation in the mRNA
levels of caspase-3 (Fig. 5C, p
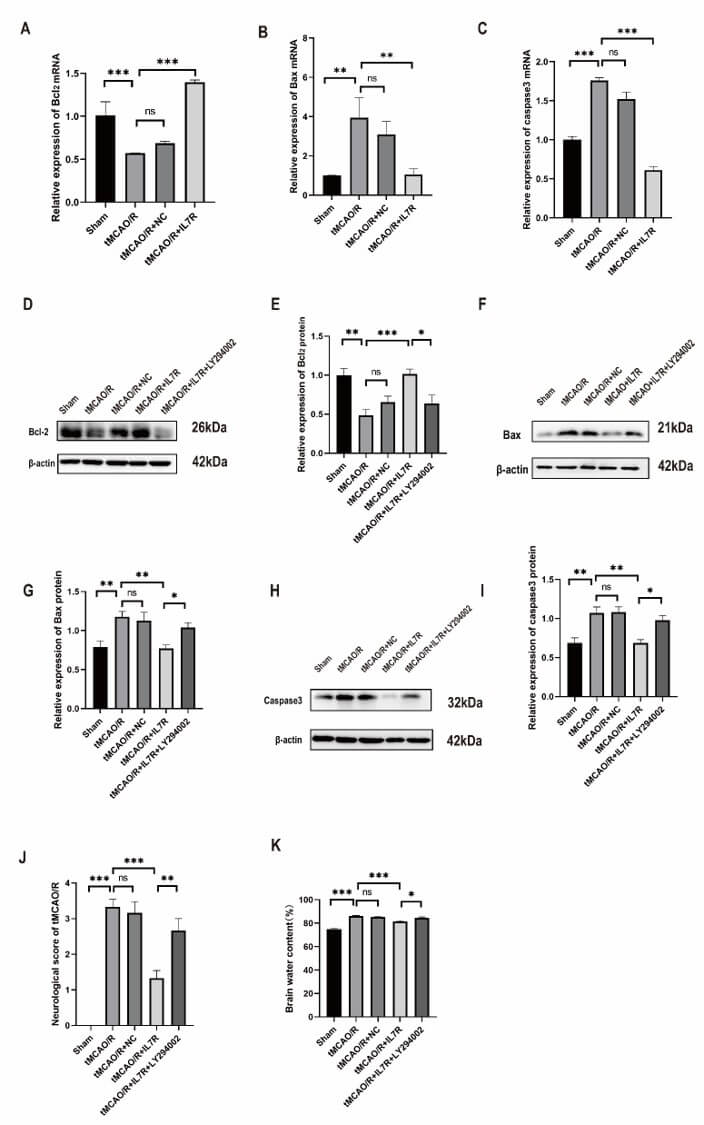 Fig. 5.
Fig. 5.
The overexpression of IL7R improved the cerebral-protective
effects by reducing apoptosis after tMCAO in vivo. (A–C) Relative mRNA
levels of (A) Bcl-2, (B) Bax, and (C) caspase-3 in the
sham, tMCAO/R, tMCAO/R + NC, and tMCAO/R + IL7R groups (n = 3 per
group). (D–I) WB and quantitative analysis of Bcl-2, Bax, and caspase-3 protein
levels in the sham, tMCAO/R, tMCAO/R + NC, tMCAO/R + IL7R, and tMCAO/R + IL7R +
LY294002 groups (n = 7 per group). (J) Neurological deficit scores of
each group of mice (n = 6 per group). (K) BWC of each group of mice
(n = 6 per group). Data are shown as mean
IF staining for IL7R and GFAP in brain tissue revealed that IL7R and GFAP
expression were reduced after tMCAO, whereas GFAP expression was increased upon
IL7R overexpression. These results confirmed that IL7R was colocalized with
astrocytes (Fig. 3B), leading us to select the C8D1A cell line for further
in vitro validation. Cell viability was assessed in vitro at
various time points during OGD. Cell viability significantly decreased as the
duration of OGD increased. To achieve a cell survival rate of approximately 50%,
OGD was set to 3 h (Fig. 6A). In the OGD/R model, IL7R mRNA levels were
markedly down-regulated by RT-qPCR, particularly after 24 h of OGD/R (Fig. 6B,
p
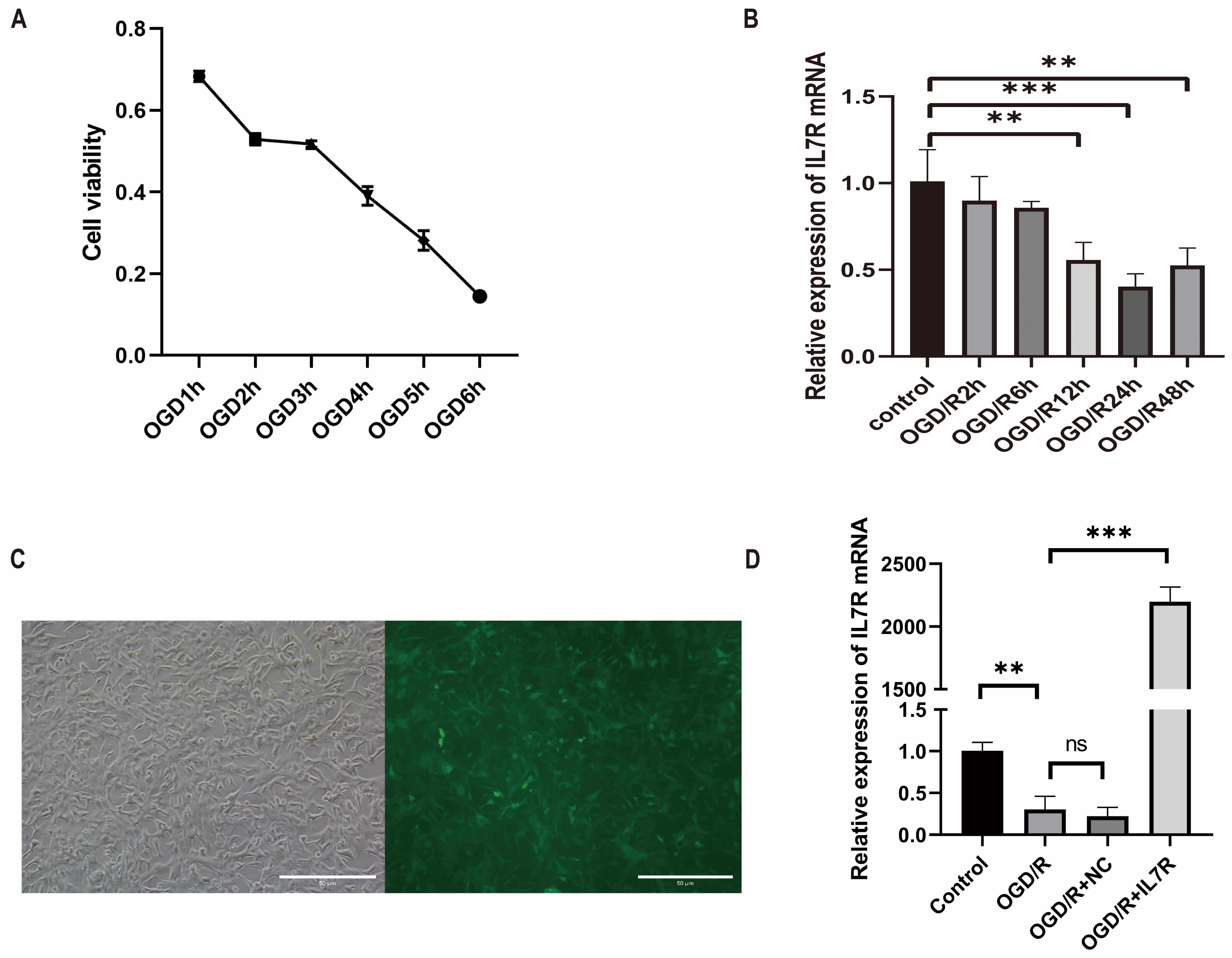 Fig. 6.
Fig. 6.
Generation of an OGD/R model and an IL7R overexpression model in
mouse brain astrocytes. (A) Cell viability analysis via CCK-8 assays after
various durations of OGD (1, 2, 3, 4, 5, or 6 h), followed by 24 h of
reoxygenation (n = 3 per group). (B) IL7R expression was
significantly downregulated in OGD/R-exposed C8D1A cells. RT-qPCR analysis
confirmed the significant decreasing in IL7R mRNA levels at 24 h of
OGD/R (n = 3 per group). (C) Representative IF images of LV-IL7R and
GFP-transfected C8D1A cells under normal and fluorescence microscopy. (Scale bar = 50 µm). (D)
Relative mRNA levels and quantitative analysis of IL7R (n = 3
per group). Data are shown as mean
Fc assessment was employed to determine the relationship between IL7R and
apoptosis in vitro. The results revealed that C8D1A cells exposed to
OGD/R exhibited a pronounced elevation in apoptotic rate, indicating substantial
cellular damage. However, IL7R overexpression significantly reduced apoptosis
(Fig. 7A,B; p
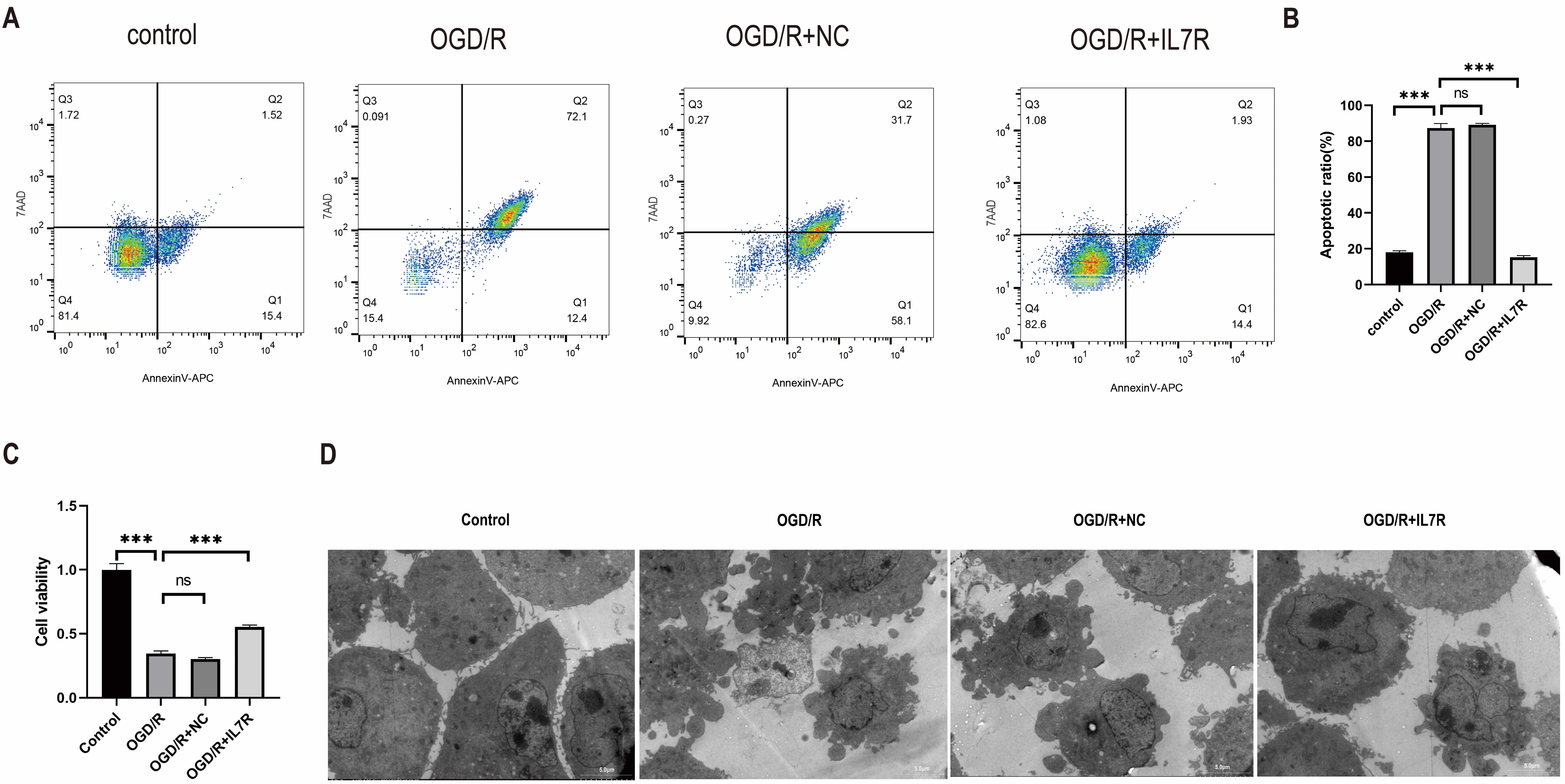 Fig. 7.
Fig. 7.
IL7R overexpression reduced apoptosis after OGD/R in
vitro. (A,B) Fc and quantitative analysis of apoptosis rates in C8D1A cells
across different groups (n = 3 per group). (C) Cell viability analysis
with the CCK-8 assay (n = 3 per group). (D) Representative TEM images of
C8D1A cells (Scale bar = 5.0 µm). Data are shown as mean
To investigate the mechanism of IL7R-mediated neuroprotection via PI3K/Akt
signaling in CIRI, pharmacological inhibition was implemented using LY294002,
preceding IL7R overexpression and OGD/R induction. The RT-qPCR data revealed a
marked reduction in Bcl-2, in parallel with elevated Bax and
caspase-3 expression, in the OGD/R model. The overexpression of
IL7R counteracted these alterations, resulting in significant
enhancement of the Bcl-2 mRNA level and concurrent suppression of the
Bax and caspase-3 mRNAs. Conversely, pharmacological PI3K
inhibition with LY294002 abolished the antiapoptotic effects mediated by
IL7R overexpression (Fig. 8A–C, p
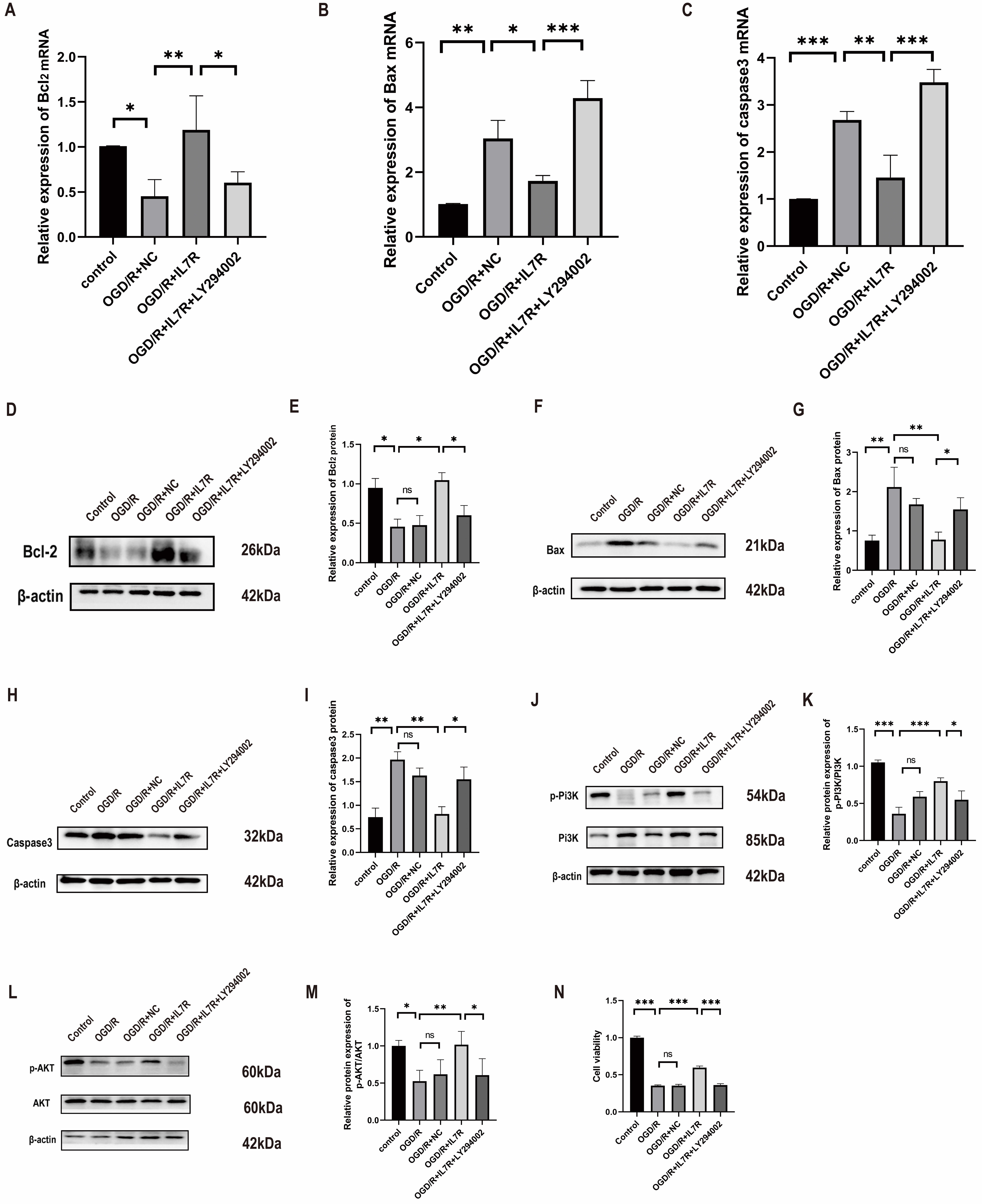 Fig. 8.
Fig. 8.
LY294002 reversed the ability of IL7R to inhibit apoptosis after
CIRI. (A–C) Relative mRNA levels and quantitative analysis of Bcl-2,
Bax, and caspase-3 (n = 3 per group). (D–M)
Representative WB images and quantitative analysis of Bcl-2 (D,E), Bax (F,G),
caspase-3 (H,I), p-PI3K/PI3K (J,K), and p-Akt/Akt (L,M) protein levels in C8D1A
cells of different groups (n = 3 per group). (N) Cell viability of C8D1A
cells in different groups (n = 5 per group). Data are shown as mean
Our results observed that IL7R level was downregulated in the cerebral I/R model. Furthermore, IL7R overexpression protected against CIRI. Our results demonstrated that IL7R overexpression reduced cerebral edema, conferred better neurological outcome, and limited both infarct expansion and apoptosis after I/R. The overexpression of IL7R led to decreased Bax and caspase-3 expression and elevated Bcl-2 levels. LY294002 largely reversed IL7R-mediated protection against CIRI, confirming that PI3K/Akt signaling partially drives the beneficial effect (Fig. 9).
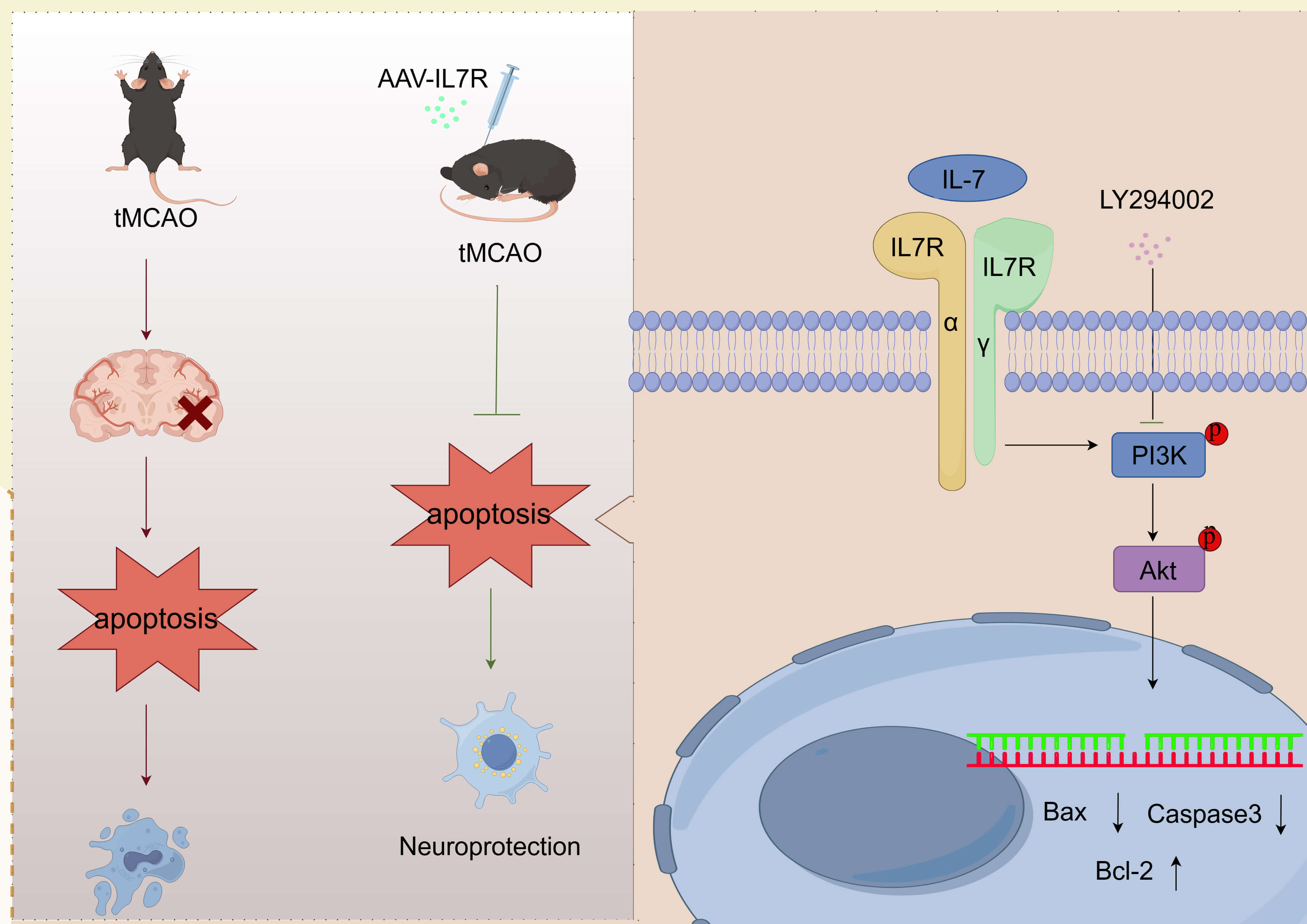 Fig. 9.
Fig. 9.
The mechanism by which IL7R may attenuate astrocyte apoptosis through PI3K/Akt pathway activation after cerebral I/R. Figdraw provided the material for drawing (https://www.figdraw.com/). I/R, ischemia/reperfusion.
Globally, IS ranks among the foremost causes of long-term disability and death. Vascular recanalization therapy has become widely applied clinically, with early reperfusion considered to be the most effective treatment for IS [31]. However, reperfusion itself can trigger additional pathological mechanisms that worsen brain damage [32]. IL-7 is a crucial homeostatic cytokine that supports the persistence and viability of T-cell and other lymphoid populations [33]. IL-7 is associated with inflammation, autoimmune conditions, neoplastic diseases, and various biological processes [34]. IL-7 transcripts are detectable in both lymphoid and non-lymphoid tissues, which are essential for biological barriers [35]. IL7R is likewise indispensable for the ontogeny and continued survival of multiple immune-cell lineages and is involved in diseases linked to lymphatic dysfunction [36]. Furthermore, IL7R is involved in neurodegenerative-disease pathology, including Parkinson’s disease and multiple sclerosis [37, 38]. The IL-7/IL7R signaling pathway influences inflammation after intracerebral hemorrhage and may be a new immunotherapy target in the pathophysiological mechanism of intracerebral hemorrhage treatment [39]. The reduced expression of IL7R has been correlated with delayed myelination [40]. IL-7 also has trophic properties in the developing brain, suggesting its potential as a growth factor for neurons of physiological value [41]. These observations suggest a potential neuroprotective function for IL7R. The result of a porcine femoral artery I/R study indicated that dynamic changes occurred in gene expression after I/R. These changes may be closely related to T-cell activation. During T-cell activation, increased IL7R potentially orchestrates I/R injury [42]. In the myocardial I/R model, IL-7 intensified cardiac injury by modulating macrophage infiltration and polarization. Controlling the level of IL-7 may help alleviate myocardial I/R injury [43]. However, the role of IL7R in CIRI has yet to be explored. We found that IL7R expression was downregulated after IS and inversely correlated with infarct size and neurological deficits, indicating that IL7R may be a critical player in CIRI and a potential therapeutic target. However, the exact molecular basis of IL7R action requires further exploration.
Previous studies have shown that certain protein-coding genes orchestrate apoptosis control; whether IL7R participates in a regulatory network with these genes remains to be explored. Future research can explore the specific mechanisms of these proteins in different disease states. Silent Information Regulator1 (SIRT1) suppresses apoptotic signaling, curbs neuro-inflammation by reducing pro-inflammatory cytokine release, and preserves mitochondrial integrity, thereby conferring broad neuroprotection against CIRI [44, 45, 46]. These pleiotropic effects make SIRT1 a promising therapeutic target in stroke. Emerging evidence has suggested that SIRT1 may also govern IL7R expression. In NSCLC, SIRT1 modulated IL7R levels on immune cells, shaping tumor prognosis [47], and loss of SIRT1 paralleled down-regulated IL7R and subsequent T-cell dysfunction in COVID-19 [48]. Although a direct SIRT1–IL7R axis has not yet been validated in CIRI, their complementary cytoprotective and immunomodulatory roles imply that an interaction could influence ischemic outcome. Future studies should therefore determine whether SIRT1 regulates IL7R after stroke and whether co-targeting both molecules might augment recovery. Furthermore, small extracellular vesicles derived from mesenchymal stromal cells have recently been shown to markedly enhance neural recovery and brain remodeling in both young and aged rats after permanent distal MCAO [49], underscoring their therapeutic promise for elderly stroke patients. Estrogen additionally confers cerebrovascular protection, emphasizing that advanced age and female sex are critical determinants of CIRI severity and outcome. However, our present work did not stratify animals by age or sex, a limitation that will be addressed in follow-up studies designed to dissect how these variables modulate CIRI pathobiology and to translate the findings into sex- and age-tailored strategies for human stroke prevention and rehabilitation.
The pathogenesis of IS is multifaceted and involves disturbances in energy metabolism, inflammatory responses, and apoptosis [50, 51]. Inflammation and apoptosis induced by the activation of astrocytes are central pathological mechanisms in CIRI. The neuroprotective effects observed during CIRI are due to the inhibition of astrocyte apoptosis [52]. The study has revealed that CIRI has protective effects against neuronal damage via the inhibition of apoptosis [14]. Apoptosis constitutes the main driver of neuronal death after ischemia. Therefore, attenuating apoptosis is essential for improving the outcome of CIRI [53]. IL7R has been shown to regulate apoptosis in other diseases. Liu and colleagues [18] demonstrated that IL-7/IL7R signaling suppresses apoptosis in NSCLC cells by modulating Bcl-2 and Bax through the p53 pathway. IL7R also triggers the Janus kinase1 (JAK1)/STAT5 pathway to increase Bcl-2 expression in pancreatic-duct adenocarcinoma [54], as IL7R has been implicated in demyelination and the process of oligodendrocyte apoptosis [37]. These results collectively emphasize the antiapoptotic effects of IL7R in various diseases. However, the specific relationship between IL7R and apoptosis in CIRI had not previously been explored. Therefore, we actively investigated whether IL7R functions in regulating apoptosis in CIRI. Our study explored whether IL7R regulated apoptosis in CIRI; results revealed that IL7R does protect against apoptosis in CIRI. Specifically, the overexpression of IL7R resulted in a decreased infarct volume and enhanced neurological outcomes (Fig. 4), decreased level of proapoptotic proteins, and a reduced rate of apoptosis while increasing the level of the antiapoptotic protein (Fig. 5). We found that IL7R can mediate neuroprotection by exerting antiapoptotic effects, which aligns with earlier reports. Our findings confirmed that IL7R protects against CIRI through antiapoptotic mechanisms and provide a foundation for the development of IL7R-based therapies for ischemic brain diseases.
The PI3K/Akt signaling cascade is central to the pathobiology of CIRI and has well-documented neuroprotective characteristics [55]. Catalpol administration enhanced the neurovascular unit in an IS model by activating the PI3K/Akt [56]. Colin has antiapoptotic effects on cardiomyocytes by regulating the PI3K/Akt [57]. Resveratrol administration alleviated apoptosis in stroke models through the PI3K/Akt pathway [58]. IL7R affects hematopoiesis and immune responses through the JAK-STAT and PI3K/Akt pathways [12]. IL-7 activates the autophagy process in lung cancer cells through the PI3K/Akt [17]. Our data revealed that IL7R, through the PI3K/Akt pathway, is involved in the modulation of apoptosis. To substantiate the pathway-specific neuroprotection hypothesis, we used pharmacological PI3K inhibition with LY294002 and effectively attenuated the IL7R-mediated protective effects (Fig. 5). Although gene knockout or co-immunoprecipitation methods can increase the specificity of the pathway, LY294002 has been widely recognized for this application [59]. These cumulative results establish PI3K/Akt signaling as an essential mediator of the therapeutic potential of IL7R in CIRI management; however, whether the mitogen-activated protein kinase (MAPK), JAK-STAT, and other specific regulatory mechanisms are involved in this process requires further exploration in the future.
Our study had several limitations. First, the animal sample in some experiments was not large, which was a decision made to minimize animal use, according to NIH recommendations. Although this approach enhanced the ethical standard of the research, it limited the statistical power for detecting more subtle effects. Second, for the experiments involving the PI3K inhibitor LY294002, a separate vehicle-control group was not included. Instead, comparisons were made against the standard model control group. Although the effects of the solvent DMSO were expected to be minimal based on common practice and previous literature, the absence of a dedicated control group meant that we could not entirely rule out its potential contribution to the observed effects. It is noteworthy that the assessment of neurological function in this study was conducted using the modified Longa score, a well-validated tool for global deficit evaluation. Future studies incorporating more specialized tests for motor coordination, balance, and strength will be valuable in order to delineate the specific facets of functional recovery influenced by IL7R overexpression. In addition, the precise contribution of the IL-7 ligand remains undefined; the upstream and the downstream regulatory networks controlled by IL7R require further exploration. Consequently, clinical translation of IL7R-based interventions will demand extensive additional investigation.
Overall, this study provided new insights and potential therapeutic avenues for treating CIRI. Our observations suggested that the overexpression of IL7R diminishes apoptosis levels by modulating the interactions of proteins associated with apoptosis, thereby mitigating CIRI, probably consequent to PI3K/Akt activation. Nevertheless, additional research is essential to elucidate, comprehensively, the precise pathway mechanisms associated with molecules both upstream and downstream of IL7R.
Collectively, our experimental data demonstrate that IL7R administration confers neuroprotection against cerebral damage and apoptotic processes through stimulation of the PI3K/Akt. Our results identify IL7R as a candidate target for CIRI therapy. Although its precise molecular actions remain to be fully defined, the approach holds clear translational promise and offers a fresh direction for developing effective CIRI interventions.
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
SY conceived the study, performed the experiments, analyzed the data, drew the graphs, and wrote the manuscript. QS, WL, and DM participated in some animal model experiments. ZS participated in the conception process and guided and revised the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Strict adherence to the U.S. National Institutes of Health (NIH) criteria for the care and use of animals was maintained throughout the investigation and the research was approved by the Institutional Animal Ethics Committee of the First Affiliated Hospital of Harbin Medical University (Ethics No. 2023124). Efforts were made to minimize the number of mice used and to alleviate their pain as much as possible. The procedure was strictly followed by the guidelines approved by the Institutional Animal Care and Use Committee, with the guiding principles of minimizing pain, ensuring rapid death, and respecting animal life.
We are grateful for the help provided by the teachers and students of the Central Laboratory of the First Affiliated Hospital of Harbin Medical University during the experiment. Figdraw provided the material for drawing (https://www.figdraw.com/).
This study was supported by the Graduate Research Allowance of the First Affiliated Hospital of Harbin Medical University.
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/JIN46638.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.









