1 The Third School of Clinical Medicine (School of Rehabilitation Medicine), Zhejiang Chinese Medical University, Key Laboratory of Acupuncture and Neurology of Zhejiang Province, 310053 Hangzhou, Zhejiang, China
2 Department of Acupuncture and Moxibustion, Third Affiliated Hospital of Zhejiang Chinese Medical University, 310009 Hangzhou, Zhejiang, China
†These authors contributed equally.
Abstract
The perineuronal net (PNN) is an important extracellular environment around parvalbumin interneuron (PV IN) in the spinal cord. Chondroitin sulfate proteoglycan (CSPG) serves as a key factor mediating PNN effects on the spinal cord, primarily formed by covalently linked chondroitin sulfate glycosaminoglycan (CS-GAG) chains and diverse core proteins. Extensive research suggests that degradation of CS-GAG following nerve injury may contribute to severe spinal cord damage. Inhibiting CS-GAG degradation could enhance PNN stability and plasticity, thereby promoting recovery from nerve injury. Electroacupuncture (EA) intervention demonstrates significant neuroprotective effects, facilitating restoration of spinal cord nerve function and axonal regeneration. This study aims to observe the changes in CS-GAG and the expression of PV IN after spinal cord injury (SCI) in rats and explore the effect.
An SCI model was established in Sprague–Dawley rats using an Infinite Horizon (IH) impactor, and EA was applied to the Jia-ji acupoints (EX-B2). The Basso–Beattie–Bresnahan (BBB) score of SCI rats was evaluated, and electromyography (EMG) of the gastrocnemius muscle of the hind limbs was performed. The protein expression levels of CS-GAG and glutamic acid decarboxylase (GAD) were detected using western blotting, and perineuronal nets (PNN) and PV IN were observed using immunofluorescence (IF). Fiber-optic calcium imaging was used to detect and analyze PV IN activity. Adeno-associated virus containing carbohydrate sulfotransferase 11 (Chst11) was injected into T9 and T10 spinal cord spaces using a microneedle, and changes in CS-GAG in the spinal cord of SCI rats before and after EA intervention were observed.
CS-GAG and GAD expression levels were significantly decreased after SCI and PNN stability was reduced. Chondroitinase ABC (ChABC) treatment increased PV IN activity and GAD expression. EA effectively promoted an increase in CS-GAG and GAD, improved PNN stability and PV IN activity, and reversed the inhibitory effect of Chst11, thereby facilitating the rehabilitation of rats with SCI.
The mechanisms and effects of EA on SCI repair were investigated. The results revealed that EA can regulate the recovery of PNN structure and function via CS-GAG and GAD, improve PV IN activity, and reverse the inhibitory effect of Chst11 to promote SCI rehabilitation in rats.
Keywords
- spinal cord injury
- electroacupuncture
- parvalbumin interneuron
- chondroitin sulfate glycosaminoglycan
- glutamic acid decarboxylase
With the rapid development of the global economy and the increasingly severe aging process of the population, the incidence of spinal cord injury (SCI) remains high [1], and its disability rate has become the second leading cause of paralysis [2], placing a heavy burden on the social healthcare system. The pathological mechanisms of SCI are complex and diverse, and effective treatments for SCI are still lacking.
The perineuronal net (PNN) is an important extracellular environment around parvalbumin interneurons (PV IN) in the spinal cord [3]. It plays an important role in neuroprotection and the maintenance of neuronal plasticity and stability. PNN is a network structure within the extracellular matrix (ECM) that contains chondroitin sulfate proteoglycan (CSPG) [4]. Research has demonstrated that CSPG is an important factor in the action of PNN on the spinal cord [5], which is primarily formed by covalent bonds between chondroitin sulfate glycosaminoglycan (CS-GAG) chains and various core proteins. CS-GAG is sulfated in different ways depending on the chondroitin sulfotransferase [6, 7], with two main types being chondroitin 4-sulfation (C4S) and chondroitin 6-sulfation (C6S). Carbohydrate sulfotransferase-11 (Chst11)/chondroitin-4-sulfotransferase-1 (C4ST-1), an enzyme widely distributed in the central nervous system (CNS), plays an important role in CS4-O-sulfation and the amount of CS synthesis [8]. C4ST-1 reduction accelerates neural axon regeneration in zebrafish after SCI [9]. One study has presented that the ratio of C4S/C6S reflects the development and structural stability of PNN to a certain extent [10]. Meanwhile, the C4S/C6S ratio determines maturation of PV IN and regulates the plasticity of PNN [10]. One study has demonstrated that the absence of CS reduces the spike reaction of PV IN wrapped in PNN [11], and the maturation of PV IN promotes the development of PNN and the sulfation mode of CS-GAG [11]. Research suggests that the negative charge carried by CS-GAG plays a crucial role in maintaining the plasticity and stability of neurons, and the degradation of CS-GAG after nerve injury may cause severe damage to the spinal cord [5]. Therefore, if CS-GAG degradation is prevented, it enhances the stability and plasticity of PNN to a certain extent, allowing neuronal damage to be repaired.
Electroacupuncture (EA) at the jia-ji acupoints is a reliable and effective clinical method for safely improving the peripheral microenvironment after SCI. EA intervention has a significant neuroprotective effect [12], promoting the recovery of spinal cord nerve function and axonal regeneration [13, 14]. Our previous research has confirmed that EA promotes the proliferation and differentiation of neural stem cells, oligodendrocyte precursor cells, and oligodendrocytes, improves myelin sheath formation after SCI, promotes the recovery of SCI via the upregulation of PNN by inhibition of Sema3A, and plays a vital role in promoting the recovery of motor function after SCI [13, 15, 16]. Current studies have focused on the regulation of PNN components by EA. Consequently, this study aimed to elucidate whether EA regulation of CS-GAG after SCI promotes the restoration of PNN stability and facilitates the repair of SCI.
Healthy adult male SD rats (8 weeks of age, 200–220 g) were purchased from Shanghai Sipur-Bike Laboratory Animal Co., LTD (Shanghai, China, Animal license No.: SCXK(Shanghai) 2018-0006), and were raised in Laboratory Animal Center of Zhejiang Chinese Medicine University and approved by the China Laboratory Animal Management Evaluation and Accreditation Association (AAALAC, Animal license No.: SYXK(Zhejiang) 2018-0012). The rats were kept in controlled conditions and had free access to water and food. All animal experiments were conducted in accordance with all relevant animal testing and research ethics regulations and in accordance with the animal protocol approved by the Animal Ethics Committee of Zhejiang University of Chinese Medicine (ZSLL, 2017183).
A total of 144 rats were included in this study. All rats were randomly assigned to each group. (1) In the Sham group (n = 8), only the T10 vertebral plate was removed without any other intervention. (2,3,4) The SCI (n = 8), SCI-1d (n = 8) and SCI-7d (n = 8) groups were subjected to SCI model preparation without intervention. (5,6) SCI + phosphate buffer saline (PBS) (n = 8) and SCI + Chondroitinase ABC (ChABC) (Sigma-Aldrich, C3667, St. Louis, MO, USA) (n = 8) groups received 14 days of PBS and ChABC injections, respectively, based on the SCI model preparation. (7,8) After preparing the SCI model, the SCI + ShamEA (n = 8) (sham acupuncture control group, with needles hung off the skin without other interventions) and SCI + EA (n = 8) groups received acupuncture without electrical stimulation and acupuncture with electrical stimulation interventions for 14 days, starting on the second day. (9,10) SCI + Vehicle (Veh) (n = 8) and SCI + Chst11 (n = 8) groups were injected with empty vector control virus and Chst11 overexpression virus, respectively, and SCI models were prepared. (11) The SCI + Chst11 + EA (n = 8) group was added to the SCI + Chst11 group and included a 14-day EA intervention. The above section comprises a total of 88 rats, divided into 11 groups. Due to the unique nature of qRT-PCR experiments, this section is designated separately, involving a total of 56 rats. Groups included: Sham (n = 8), SCI (n = 8), SCI + PBS (n = 8), SCI + ChABC (n = 8), SCI + Veh (n = 8), SCI + Chst11 (n = 8), and SCI + Chst11 + EA (n = 8).
Dissolve ChABC or PBS in 1% fetal bovine serum (Sigma-Aldrich, F0193) to prepare a solution at a concentration of 5 U/mL [17]. Prepare fresh daily. Starting from day 1 after SCI model establishment, deliver ChABC or PBS to the rat’s spinal cord via intrathecal injection for 14 days [18].
Animal deaths and experimental dropouts inevitably occurred during this study, and this section provides an explanation. (1) Two rats in the Sham group died from postoperative infection complications. A rat sustained an accidental injury to its right hind limb during electromyography (EMG) amplitude measurement, which was not recorded. (2) One rat in the SCI group died from infection caused by bladder rupture during assisted urination due to excessive residual urine postoperatively. Additionally, one rat was lost during calcium imaging due to fiber optic detachment. (3) Two rats in the SCI + PBS group died from postoperative complications and bladder rupture. One rat was not recorded during calcium imaging due to fiber detachment. (4) Two rats in the SCI + ChABC group died from postoperative complications. Two rats were not recorded during calcium imaging due to fiber detachment. (5) In the SCI + ShamEA group, one rat failed to obtain amplitude measurements during EMG recording due to accidental injury to the right hindlimb muscles. (6) One rat in the SCI + EA group died from infection induced by postoperative bladder rupture. One rat failed to obtain amplitude measurements during EMG recording due to accidental injury to the right hindlimb muscles. (7) In the SCI + Veh, SCI + Chst11, and SCI + Chst11 + EA groups, two rats each (six total) died from bladder rupture during postoperative assisted urination. Among these, one rat in the SCI + Veh group could not be measured during qRT-PCR due to technical error. Due to technical errors, one rat each in the SCI + Chst11 and SCI + Chst11 + EA groups failed to yield data for the Chst11 gene loading lane, resulting in one dropout per group (total of 2). All animals removed from the groups were euthanized (Painless euthanasia was performed by administering 100–200 mg/kg of pentobarbital sodium solution via intraperitoneal injection to induce respiratory arrest in rats.) and promptly transported to the animal center for disposal by designated personnel.
Healthy adult male SD rats were deeply anesthetized with 3% pentobarbital
sodium solution (Sigma-Aldrich, P3761) (2 mL/kg) and placed on a constant
temperature operating table at 37 °C. The T10 cone was exposed, and the
lamina was removed to expose the T10 spinal cord. An acute SCI model was created
by impinging on the T10 spinal cord with a force of 200 kDynes using the IH Impactor
0400 SCI Impinger (Precision Systems and Instrumentation, LLC, IH-400, Lexington,
KY, USA). The following signs indicated the successful modeling: (1) body
spasmodic tremor, (2) spasmodic movement of the tail, and (3) intradural
congestion or hematoma. The Basso–Beattie–Bresnahan (BBB) score was assessed on
the day after modeling. Rats with scores
The EA was administered at the T9–T11 Jia-ji acupoints (EX-B2), located beside the spinous processes on the back, starting on the first day after surgery. The procedure was as follows: a disposable sterile stainless steel acupuncture needle (0.18 mm in diameter; Beijing Zhongyantaihe Medical Instrument Co., Ltd., V518215, Beijing, China) was inserted 1.5 mm lateral to the midline of the spinous process to a depth of 4–5 mm, until the needle tip contacted the lamina, and connected to the HANS200A electroacupuncture instrument (Nanjing Jisheng Medical Technology Co., Ltd., HANS200A, Nanjing, China). The parameters were set as follows: frequency, 2/100 Hz; current intensity, 1 mA; once daily for 20 min. The EA treatment period was 14 days.
As previously described [19], the BBB hind limb movement test was scored on a scale of 0–21 (0 = no visible hind limb movement; 21 = normal movement). Hind limb movement of rats with SCI was tested in an open field, including hind limb joint movement, weight support, footstep, coordination, paw position, fine paw movement, and trunk and tail control. BBB testing was performed on the first day after establishing the model. Each rat was placed in an open field and evaluated by two experimenters who were unaware of the experimental group for 3 min. One of the experimenters recorded the scores, and all rats were evaluated before establishing the SCI model to ensure baseline differences. Each score was averaged to obtain the final score. For specific scoring criteria, refer to Table 1.
| Score | The ability of Lower limb motor |
| 0 | There is no visible hindlimb (HL) movement |
| 1 | Light movement of one or both joints, usually hip and/or knee |
| 2 | Broad movement of one joint or joint and slight movement of the other |
| 3 | Extensive movement of the two joints |
| 4 | Light movement of three joints |
| 5 | Light movement of two joints and wide movement of the third |
| 6 | Broad movement of the two joints and light movement of the third |
| 7 | The extensive movement of all three joints of HL |
| 8 | The ball of the foot without weight support or without weight support |
| 9 | The soles of the feet occasionally bear the weight of the ground (for example, when stationary), frequent or consistent load-bearing movements of the dorsal claw, without the soles of the feet supporting the movement |
| 10 | Paw surface occasionally moves with load bearing without Forelimb-hindlimb (FL-HL) coordination |
| 11 | Paw surface has more load bearing movement and no FL-HL coordination |
| 12 | More load bearing movement and occasional FL-HL coordination on paw surface |
| 13 | Common paw bearing movement and frequent FL-HL coordination |
| 14 | Continuous palm-surface bearing movement with consistent FL-HL coordination, or common palm-surface movement, continuous fore-hind limb coordination, and occasionally dorsal claw movement |
| 15 | Continuous paw and palm bearing movement and consistent FL-HL coordination, no or occasional ground grasping movement in the forward motion of the forelimbs, and the position of the main claw parallel to the body at the initial contact |
| 16 | In the gait, the continuous paw landing and the coordinated movement of the front and rear limbs are common in the process of grasping the ground. The main claw position is parallel to the body at initial contact, and rotates after load transfer |
| 17 | In the gait, the continuous paw landing and the coordinated movement of the front and rear limbs are common in the process of grasping the ground. The main claw position is parallel to the body at initial contact and load transfer |
| 18 | In the gait, the continuous paw touches the ground in a coordinated manner with the front and rear limbs. In the process of progress, the continuous paw grasps the ground. The position of the main paw is parallel to the body at the initial contact |
| 19 | In the gait, the continuous paw touches the ground in a coordinated manner with the front and rear limbs. The continuous paw grasps the ground in the process of advancing. The position of the main paw is parallel to the body at the initial contact and load transfer |
| 20 | The position of the main claw is parallel to the body during initial contact and weight transfer. The trunk is unstable and the tail kept cocking up |
| 21 | The position of the main claw is parallel to the body at the initial contact and load transfer, and the trunk is stable and the tail kept cocking up |
The rats were bilaterally injected into the spinal cord with 500 nL of
rAAV-CMV-Chst11-Flag-WPREs, AAV2/9 (BrainVTA Co., Ltd., PT-0634, Wuhan, China); a
negative control rAAV-CMV-Flag-WPREs, AAV2/9 (BrainVTA Co., Ltd., PT-0634); or a
combination of rAAV-CAG-FLEX-jGCaMP7f-WPRE-SV40pa, AAV2/9 (BrainVTA Co., Ltd.,
PT-1422) and rAAV-PV-CRE-bGHpa, AAV2/9 (BrainVTA Co., Ltd., PT-0275). The
procedure was as follows: after exposing the spinal cord by removing the skin and
connective tissue between the T9 and T10 vertebral plates with forceps, the virus
was injected into the spinal cord under a microscope. Using the spinal vessels
along the midline as anatomical landmarks, the injection site was positioned 1 mm
lateral and 0.3 mm from the median vessels, with the needle inserted at a
10° angle. After injection, the needle tip was left in the spinal cord
for 5 min to prevent viral backflow. The injection was performed at a final titer
of 5.00
The rats (n = 3 per group) were euthanized with 3% pentobarbital (Sigma-Aldrich, P3761) (100–200 mg/kg), followed by transmyocardial perfusion with saline, and the T10 segmental spinal cord was obtained, and immersed the spinal cord in a 4% paraformaldehyde solution (Sigma-Aldrich, 16005). The T10 spinal cord was cut into 12-µm-thick transverse sections using a Frozen Slicer (Thermo Scientific CryoStar™ NX70, 957010K, Thermo Fisher Scientific, Waltham, MA, USA), with each rat containing at least five consecutive sections. All slides were closed for 1 h at 37 °C in a water bath with TBST containing 10% normal goat serum, followed by overnight incubation at 4 °C with the primary antibody. Rabbit monoclonal anti-parvalbumin antibody (1:100, ab181086, Abcam, Cambridge, UK) and wisteria floribunda lectin (WFA) (1:100, B-1355-2, Vector Laboratories, Newark, California, USA) were used for incubation. The next day, the slices were rinsed with tris-buffered saline with tween-20 (TBST) and reacted with the corresponding secondary antibody mixture (1:200, Alexa Fluor® 555 Streptavidin Coupler and 1:300, Alexa Fluor® 488 Goat anti-rabbit IgG, Thermo Fisher Scientific). Fluorescence images were obtained using a Zeiss structured illuminated optical slice microscope (Axio Imager M2; Zeiss AG, Jena, Germany). All stained sections were observed and analyzed using a blinded method. Non-experimental personnel counted and statistically analyzed the number of cells in each section using Adobe Photoshop 13.0 (Adobe Systems, 13.0, San Jose, CA, USA).
Rats were euthanized with 3% pentobarbital (Sigma-Aldrich, P3761) (100–200
mg/kg), followed by transmyocardial perfusion with saline, and the T10 segmental
spinal cord was obtained. On day 14 after SCI model preparation, rats were
euthanized, and the T10 segmental spinal cord was rapidly removed. According to
the manufacturer’s instructions, bicinchoninic acid
assay (BCA; Pierce ™ BCA, 23227, Thermo Fisher Scientific) was
used for protein concentration determination, with 20 mg protein per lane. To
detect CS protein content, 5 U/mL of ChABC was added to the protein supernatant
and incubated in a shaking bed at 37 °C for 8 h. Protein samples were separated on
a 4%–15% SDS-polyacrylamide gel electrophoresis (SDS-PAGE) gel and
electrophoretically transferred to a polyvinylidene fluoride (PVDF) membrane
(Merck Millipore, Inc., IPVH00010, Billerica, MA, USA). The membrane was closed
with 5% skim milk (Becton, Dickinson and Company, 232100, Franklin Lakes, NJ,
USA) at room temperature for 1 h, and then incubated overnight at 4 °C in a
diluted primary antibody in a closed buffer:
The rats were euthanized with 3% pentobarbital (100–200 mg/kg), followed by
trans-myocardial perfusion with saline. Then, the T10 segmental spinal cord was
obtained. A 0.5 cm segment of spinal cord tissue was placed in TRIzol reagent
(Invitrogen, 15596018CN, Thermo Fisher Scientific) to extract total RNA. The
extracted total RNA was reverse-transcribed using a transcription kit (Takara
Biomedical Technology (Beijing) Co., Ltd., RR047A, Beijing, China) according to
the manufacturer’s protocol. The target gene was mixed with SYBR Green (Solarbio,
MQ10101S, Monad Biotech Co., Ltd., Beijing, China) reagent, and qRT-PCR was
performed using LightCycler® 480 Instrument II (Roche,
05015243001, Basel, Switzerland). Gene expression was calculated using the
2-ΔΔCT method, and the final data were normalized to
| Primers | Forward | Reverse | Amplicon size (bp) |
| Chst11 | GTA CGA GAC GCT GGA GGA GGA C | CGG TAG TTC GGG TGG ACT TTG C | 120 |
| Chst3 | CCT CGG AGC AGT TTG AGA AGT GG | ATG ATG GAG CGG TTG GTG AGT G | 129 |
qRT-PCR, quantitative real-time polymerase chain reaction; Chst, carbohydrate sulfotransferase.
Four weeks prior to modeling, rAAV-CAG-FLEX-jGCaMP7f-WPRE-SV40pa virus was bilaterally injected into the T9-T10 spinal cord junction using a Hamilton (Hamilton, 202630, Bonaduz, Switzerland) syringe. The injection flow rate was 100 nL/min, with a total injection volume of approximately 500 nL. The viral delivery sites and transfection coverage are shown in Supplementary Fig. 1. Immediately after successful model establishment, proceed with fiber implantation following these steps: The T10 spinal cord segment was fully exposed, and a 0.01 mm-thick perforated titanium plate (Xi’an Saite Metal Materials Development Co., Ltd., Xi’an, China) was placed on the spinal cord. The plate was secured to the surrounding soft tissue and vertebral body using non-absorbable surgical sutures at the four corners. All procedures were performed using a microscope. A fiber-optic probe (200 µm in diameter and 2 mm in length, Thinker Tech Nanjing Bioscience Inc, Nanjing, Jiangsu, China), mounted on a stereotaxic instrument (RWD, 68804, RWD Life Science, Shenzhen, China) with a fiber-optic holder, was positioned adjacent to the central canal of the spinal gray matter and inserted to a depth of 1.5 mm. The assembly was secured with dental cement (Shanghai New Century Dental Materials Co., Ltd., 20173170702, Shanghai, China) and adhesive (Single Bond Universal, 3 M ESPE, St. Paul, MN, USA) and covered with toner (Macklin, C805116, Shanghai, China) to prevent light interference. GCaMP7f fluorescence was excited at two wavelengths: 470 nm for the calcium-dependent signal and 405 nm for the baseline signal. The amplitude-modulated excitation light was reflected by a dichroic mirror and coupled to an optical fiber, enabling carrier wave fiber calcium imaging photometric recording. The recorded data were imported into MATLAB (MathWorks, R2018a, Natick, MA, USA) for analysis. Calcium activity was recorded from the first 2 s of the external stimulus to 10 s after the stimulus to capture changes in calcium signaling shortly after sensory induction (by pinching the foot).
Nicolet EDX: Viking QUEST equipment (Nicolet EDX: Viking QUEST, Natus Neurology, Madison, WI, USA) was used to acquire EMG data from the right lower limb gastrocnemius muscle of rats. The rats were placed on a special rat sleeve, and the hair of the gastrocnemius muscle was shaved using a hair-shaving machine. The rats were tested in an emotionally calm state. During detection, recording electrodes were inserted into the gastrocnemius muscle of the right lower limb of the rats, and EMG signals with a frequency range of 20–500 Hz were collected 20–25 times consecutively. The gastrocnemius potential was detected using the VikingSelect experimental system (Nicolet EDX: Viking QUEST, Viking QUEST v20.1.11, CA, USA), and the amplitude and duration were recorded to obtain average values.
All data were analyzed, and graphs were generated using GraphPad software
(GraphPad, v8.0.2, San Diego, CA, USA) or MATLAB. The normality of the continuous
variables was assessed using the Shapiro–Wilk test. For data conforming to a
normal distribution, Student’s t-test (two-tailed) was used for
comparisons between two groups, while one-way analysis of variance (ANOVA) was
used for comparisons among multiple groups, followed by Tukey’s post hoc test.
For data that did not follow a normal distribution, the Mann–Whitney U test and
the Kruskal–Wallis H test were applied for comparisons between two groups and
among multiple groups, respectively. A two-way ANOVA was performed to analyze BBB
scores across groups. When an interaction between group (intervention) and time
was observed, simple effects analysis was conducted and adjusted using Tukey’s
post-hoc test. When no interaction between group (intervention) and time was
observed, main effects analysis was performed followed by Tukey’s post-hoc test.
All results are expressed as mean
We performed WB to detect CS, which is essential for the composition and function of the PNN, and its corresponding sulfated forms (C4S and C6S) (Fig. 1B,C). The results revealed that the expression levels of C4S and C6S in the spinal cord after SCI decreased on SCI-7d and increased on SCI-14d compared to the control group (Sham group) (Fig. 1B,C). The protein expression level of GAD can indirectly reflect the expression level of PV IN to a certain extent [20]. We examined the protein levels of both GAD isoforms after SCI (Fig. 1D). Both protein expressions were significantly reduced on SCI-7d, and GAD67 expression levels increased slightly on SCI-14d as the rat spinal cord repaired itself (Fig. 1E,F). Simultaneously, we analyzed the C4S/C6S ratio, and the results are presented in Fig. 1A. After SCI, this ratio began to decrease gradually, reaching its lowest point on SCI-7d. Subsequently, it began to increase slowly over time, becoming higher than that on SCI-1d by SCI-14d, although it remained lower than that in the control group.
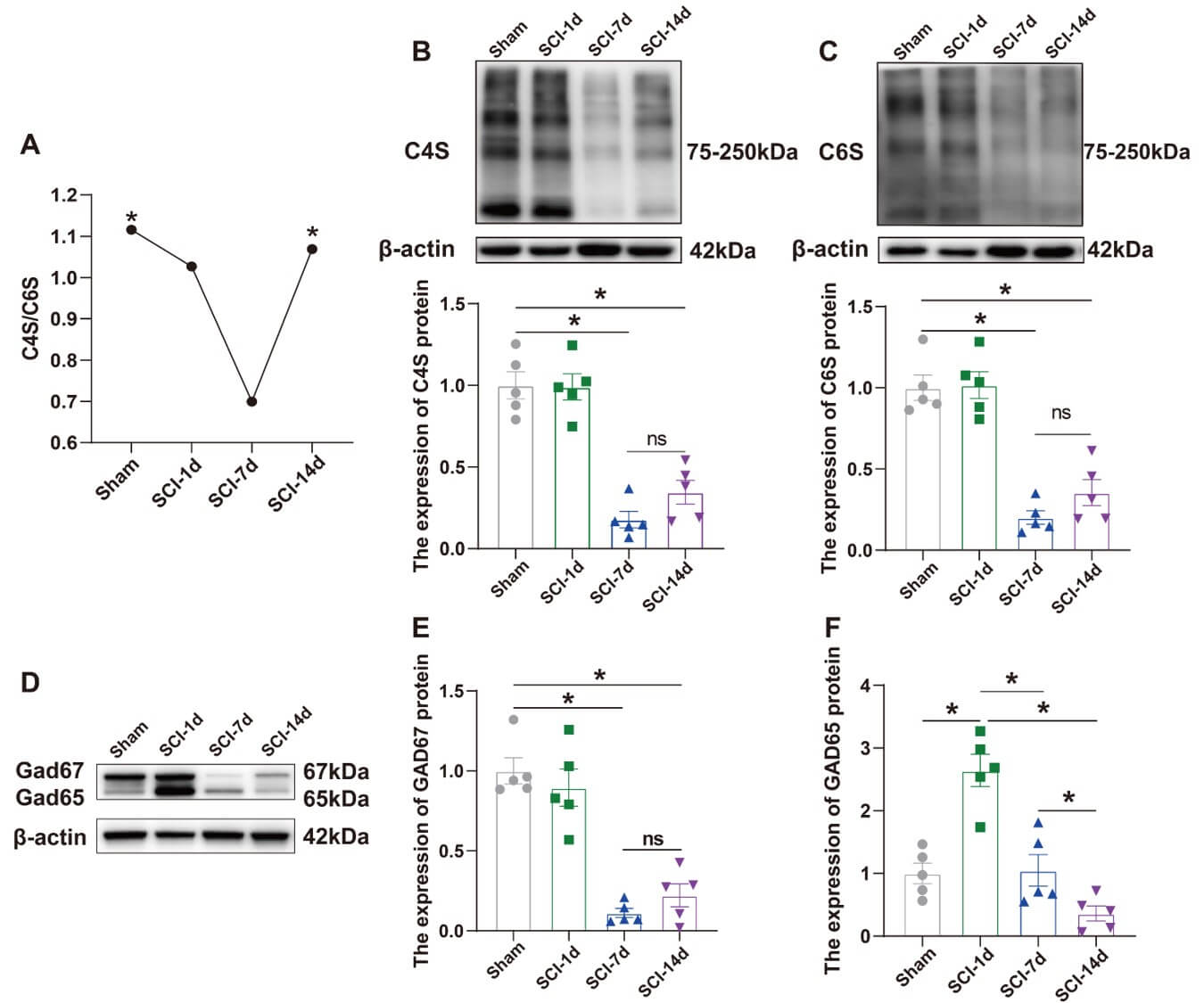 Fig. 1.
Fig. 1.
The protein expression of CS and GAD was reduced
following SCI. (A) Changes in the C4S/C6S ratio in normal rats at SCI-1d,
SCI-7d, and SCI-14d (n = 4, F = 5.225, p = 0.0174). (B) Confirmation of
two-dimensional gel electrophoresis results by WB. Analysis of C4S protein levels
in normal rats at SCI-1d, SCI-7d, and SCI-14d, using
To investigate the changes in PNN during SCI repair, we administered ChABC to
the T10 region for 14 days. The results of the BBB scores revealed a significant
interaction between group (intervention) and time (F(9,84) = 192.6,
p
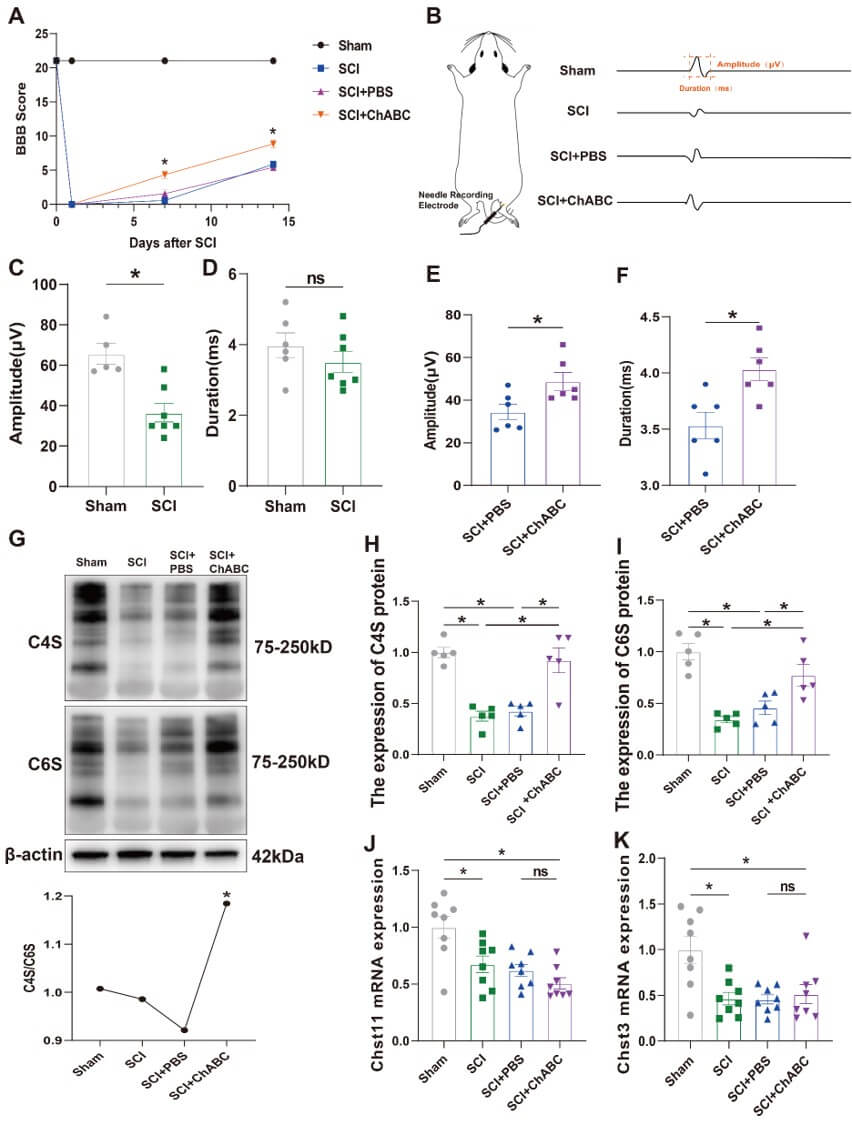 Fig. 2.
Fig. 2.
ChABC increases CS protein expression after SCI and
promotes motor function recovery. (A) Changes in BBB scores at 14 days
post-establishment of the acute spinal cord injury model in the Sham group, SCI
group, SCI + PBS group, and SCI + ChABC group (n = 6–7). There was an
interaction between group (intervention) and time in the BBB scores. A two-way
ANOVA was performed, followed by Tukey’s post hoc test. (B) Schematic
representation of EMG waveforms recorded from different groups. (C) Statistical
analysis of EMG amplitude in the gastrocnemius muscle of the Sham and SCI group
(n = 5–6, t = 4.136, p = 0.0020). (D) Statistical graph of EMG
duration in the gastrocnemius muscle of the hind limb in the Sham and SCI group
(n = 6, t = 1.025, p = 0.3272). (E) Statistical graph of EMG
amplitude in the gastrocnemius muscle of the hind limb in the SCI + PBS and SCI +
ChABC group (n = 6, t = 2.596, p = 0.0267). (F) Statistical
graph of EMG duration in the gastrocnemius muscle of the hind limb in the SCI +
PBS and SCI + ChABC group (n = 6, t = 3.213, p = 0.0093). (G)
Confirmation of two-dimensional gel electrophoresis results by WB. Changes in
GAD67 and GAD65 protein expression and the C4S/C6S ratio in Sham, SCI, SCI + PBS,
and SCI + ChABC groups (n = 5, F = 5.321, p = 0.0146). (H) Analysis of
C4S protein levels in Sham, SCI, SCI + PBS, and SCI + ChABC groups, using
Changes in CS are closely related to the progression of SCI repair, and the ratio of C4S to C6S affects PNN structure stability after SCI. Changes in CS affect the activity and expression of PV IN. We injected AAV indicative of changes in calcium signaling and labeled PV IN in the spinal cord of rats at the junction of T9 and T10. We detected and recorded calcium signals on day 14 after preparing the SCI model (Fig. 3A,B). Our previous study confirmed that neurons labeled by the jGCamp7f virus are PV IN, and the virus co-infects with PV [21]. We also validated the virus in rats and demonstrated that jGCaMP7f binds to PV IN with a significant effect (Supplementary Fig. 1). After ensuring that the surroundings were quiet and suitable and that the rats were emotionally stable, we recorded calcium signals from 2 s before stimulation to 10 s after stimulation. The results (Fig. 3C,D) revealed that the amplitude of calcium signaling in PV IN was significantly reduced in the SCI group than in the control group (Fig. 3E), and the peak calcium signal during neuronal activity decreased (Fig. 3F). The amplitude of calcium signaling in PV IN at the injury site was significantly increased in SCI + ChABC than in SCI + PBS (Fig. 3E), with no significant increase in the peak of calcium signal (Fig. 3F). WB results (Fig. 3G) revealed that the GAD67 expression level was significantly increased in the SCI + ChABC group than in the SCI + PBS control group, with no significant difference in GAD65 expression among the groups (Fig. 3H,I).
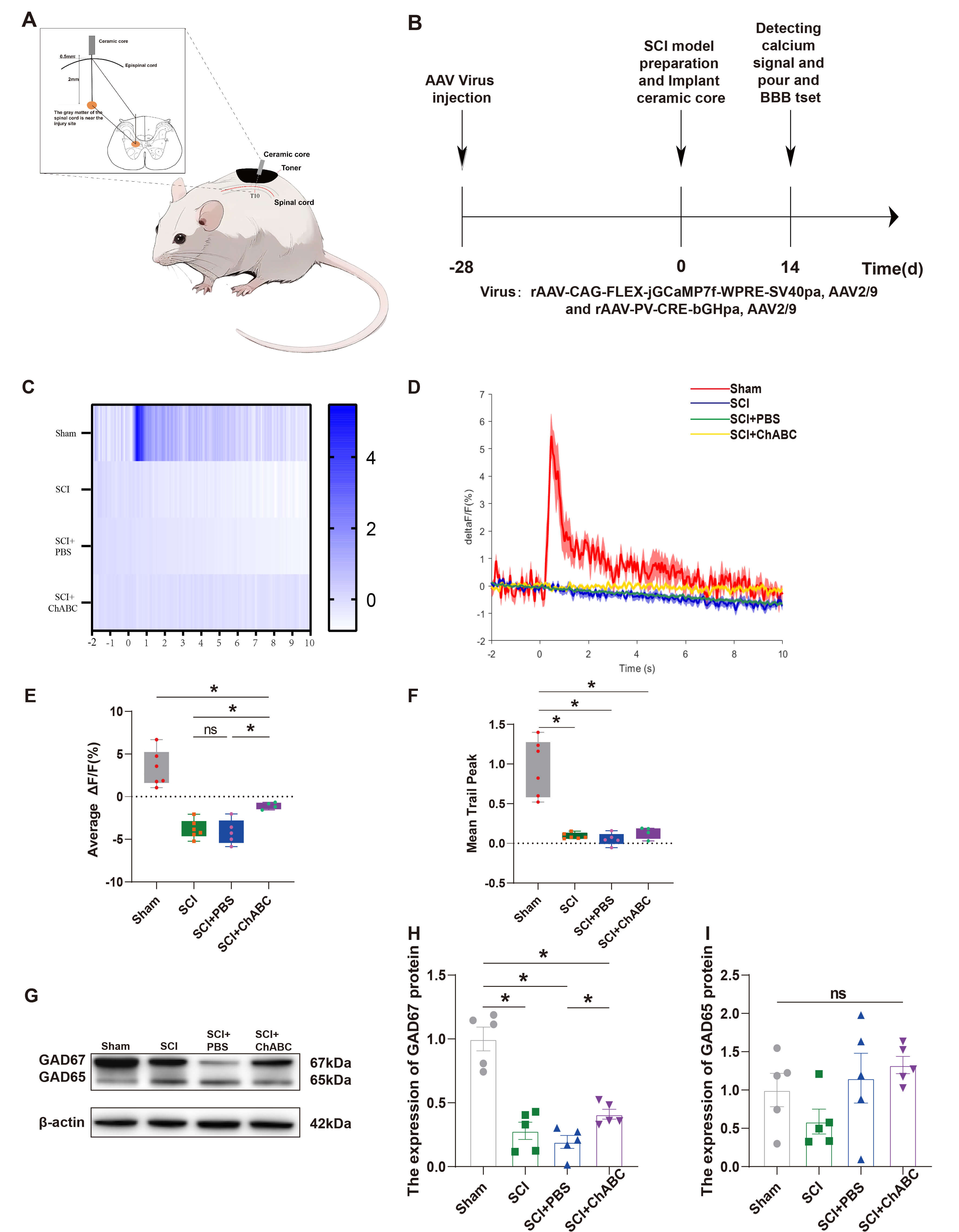 Fig. 3.
Fig. 3.
ChABC enhances PV IN activity and GAD protein
expression after SCI. (A) Schematic diagram of the fiber-optic placement in the
rat spinal cord. (B) Schematic timeline of AAV virus injection and fiber-optic
placement. (C) Heatmaps illustrating calcium signal changes in Sham, SCI, SCI +
PBS, and SCI + ChABC groups recorded after excitation at 470 nm calcium-related
signal wavelength. (D) Schematic diagram of folded calcium signal changes in
Sham, SCI, SCI + PBS, and SCI + ChABC groups, reflecting changes in PV IN
activity. (E) Statistical graph of the mean calcium signal change (n = 4–6, F =
30.53, p
Previous studies have demonstrated that ChABC significantly promotes PNN
stability and enhances PV IN activity. Subsequently, we used EA to intervene in
SCI rats and verified its therapeutic effects to determine whether the repair
effect of EA on SCI was similar to or significantly different from that of ChABC.
The results of BBB scoring revealed a significant interaction between group
(intervention) and time (F(9,96) = 167.9, p
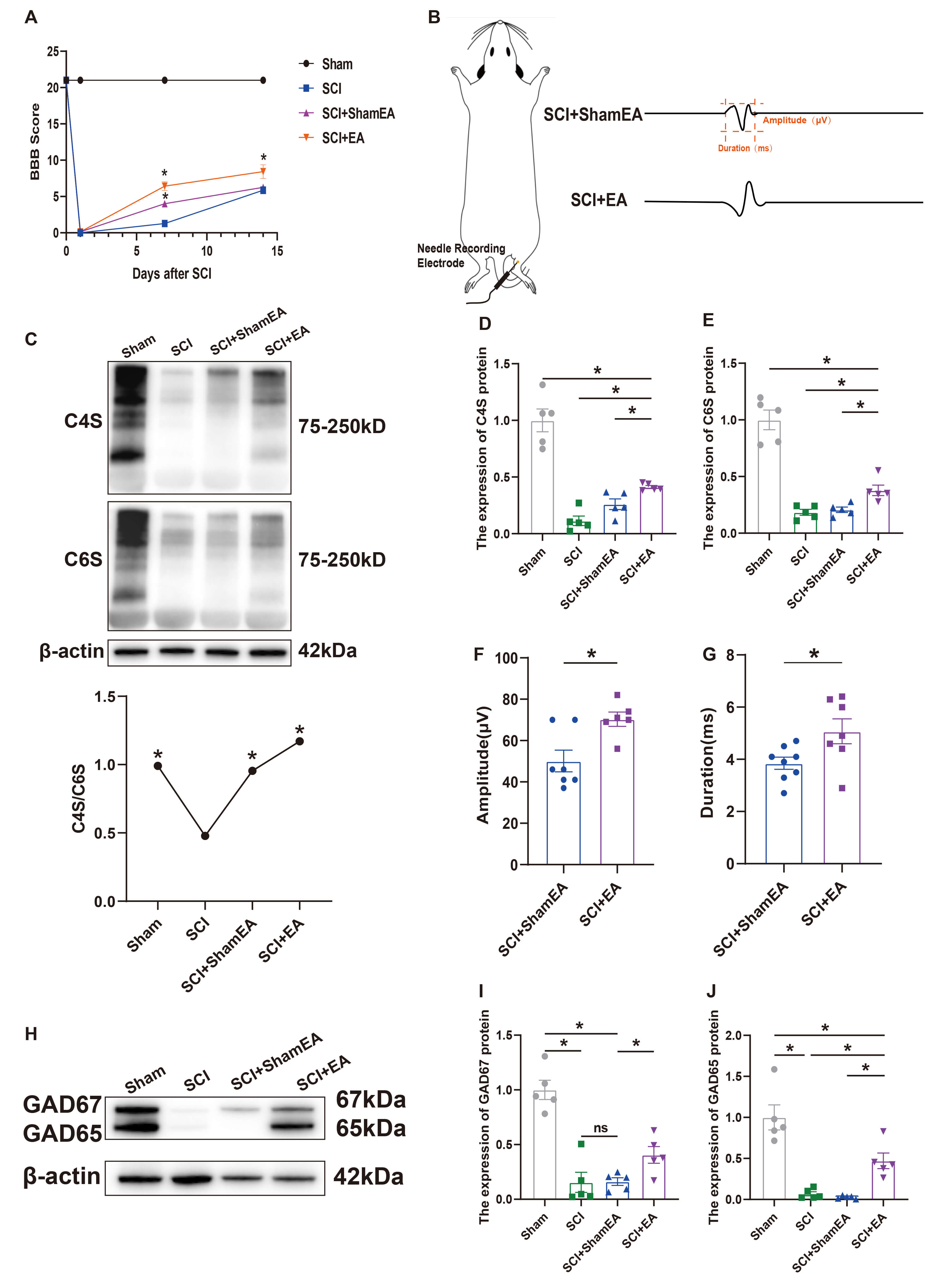 Fig. 4.
Fig. 4.
EA increased the protein expression levels of CS and GAD after
SCI and improved the relative stability of the PNN structure. (A) BBB motor
function scores of Sham, SCI, SCI + ShamEA, and SCI + EA groups at 7 and 14 days
after acute SCI model preparation (n = 7). There was an interaction between group
(intervention) and time in the BBB scores. A two-way ANOVA was performed,
followed by Tukey’s post hoc test. (B) Schematic diagram of the waveforms of EMG
signals measured in SCI + ShamEA and SCI + EA groups. (C) Protein expression of
C4S and C6S and variation of C4S/C6S in Sham, SCI, SCI + ShamEA, and SCI + EA
groups (n = 4, F = 9.189, p = 0.0020). (D,E) Confirmation of
two-dimensional gel electrophoresis results by WB. Analysis of C4S and C6S
protein levels in Sham, SCI, SCI + ShamEA, and SCI + EA groups, using
When normal rats contained more Chst11 than the normal range, their PNN
inhibitory properties were enhanced, and the rate of spinal cord repair was
inhibited following SCI. Based on this characterization, we injected the virus
overexpressing Chst11 into the spinal cord of rats at the junction of T9 and T10
28 days prior, and it was fully expressed in the T10 segment (Fig. 5A,B).
Subsequently, we verified the efficiency of Chst11 overexpression (Fig. 6C,D).
To investigate the potential of EA in promoting SCI repair by inhibiting Chst11
and modulating CS and GAD expression, we administered EA with a virus containing
Chst11 to rats for 14 days, followed by detection and analysis of T10 spinal cord
segments. The results of BBB scores revealed a significant interaction between
group (intervention) and time (F(6,60) = 6.563, p
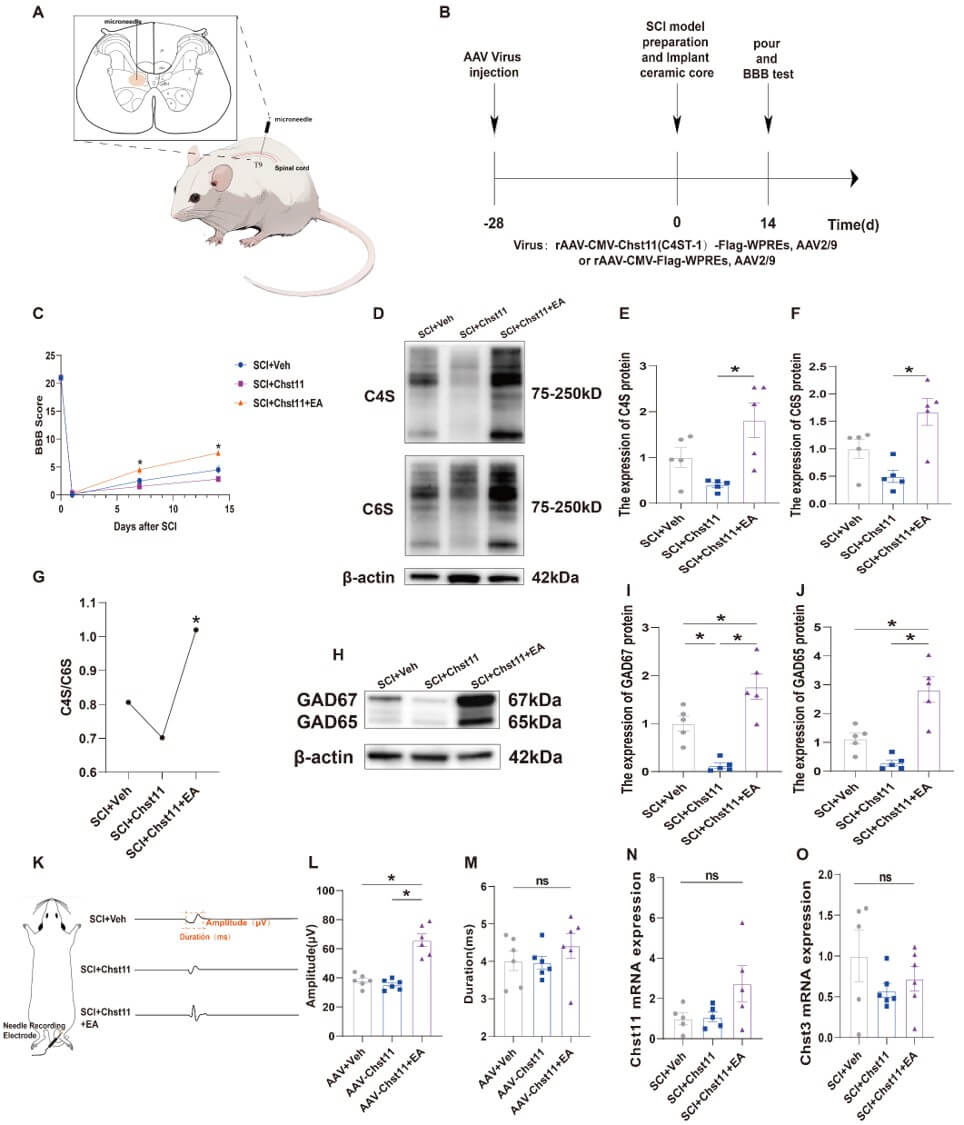 Fig. 5.
Fig. 5.
EA reversed the inhibitory effect of Chst11 and
increased the protein expression levels of CS and GAD. (A) Schematic diagram
illustrating AAV injection. (B) Timeline of AAV virus injection and SCI model
preparation. (C) Statistical graphs of BBB scores of SCI + Veh, SCI + Chst11, and
SCI + Chst11 + EA groups at 7 and 14 days after SCI model preparation (n = 6).
There was an interaction between group (intervention) and time in the BBB scores.
A two-way ANOVA was performed, followed by Tukey’s post hoc test. (D) C4S and C6S
protein expression in SCI + Veh, SCI + Chst11, and SCI + Chst11 + EA groups at
day 14 after SCI (n = 5). (E,F) Statistical graphs of C4S and C6S protein
expression (n = 5. C4S: F = 7.805, p = 0.0067. C6S: F = 10.23,
p = 0.0026). (G) Changes in the C4S/C6S ratio in SCI + Veh, SCI +
Chst11, and SCI + Chst11 + EA groups (n = 4, F = 4.880, p = 0.0367). (H)
GAD67 and GAD65 protein expression in SCI + Veh, SCI + Chst11, and SCI + Chst11 +
EA groups on day 14 after SCI (n = 5). (I,J) Statistical graphs of GAD67 and
GAD65 protein expression (n = 5. GAD67: F = 21.16, p = 0.0001. GAD65: F =
20.70, p = 0.0001). (K) Schematic diagrams of waveforms obtained by EMG
in SCI + Veh, SCI + Chst11, and SCI + Chst11 + EA groups. (L,M) Statistical
graphs of EMG signals of the gastrocnemius muscle in the hind limbs of rats in
SCI + Veh, SCI + Chst11, and SCI + Chst11 + EA groups (n = 6. Amplitude: F =
36.06, ns: not significant, p
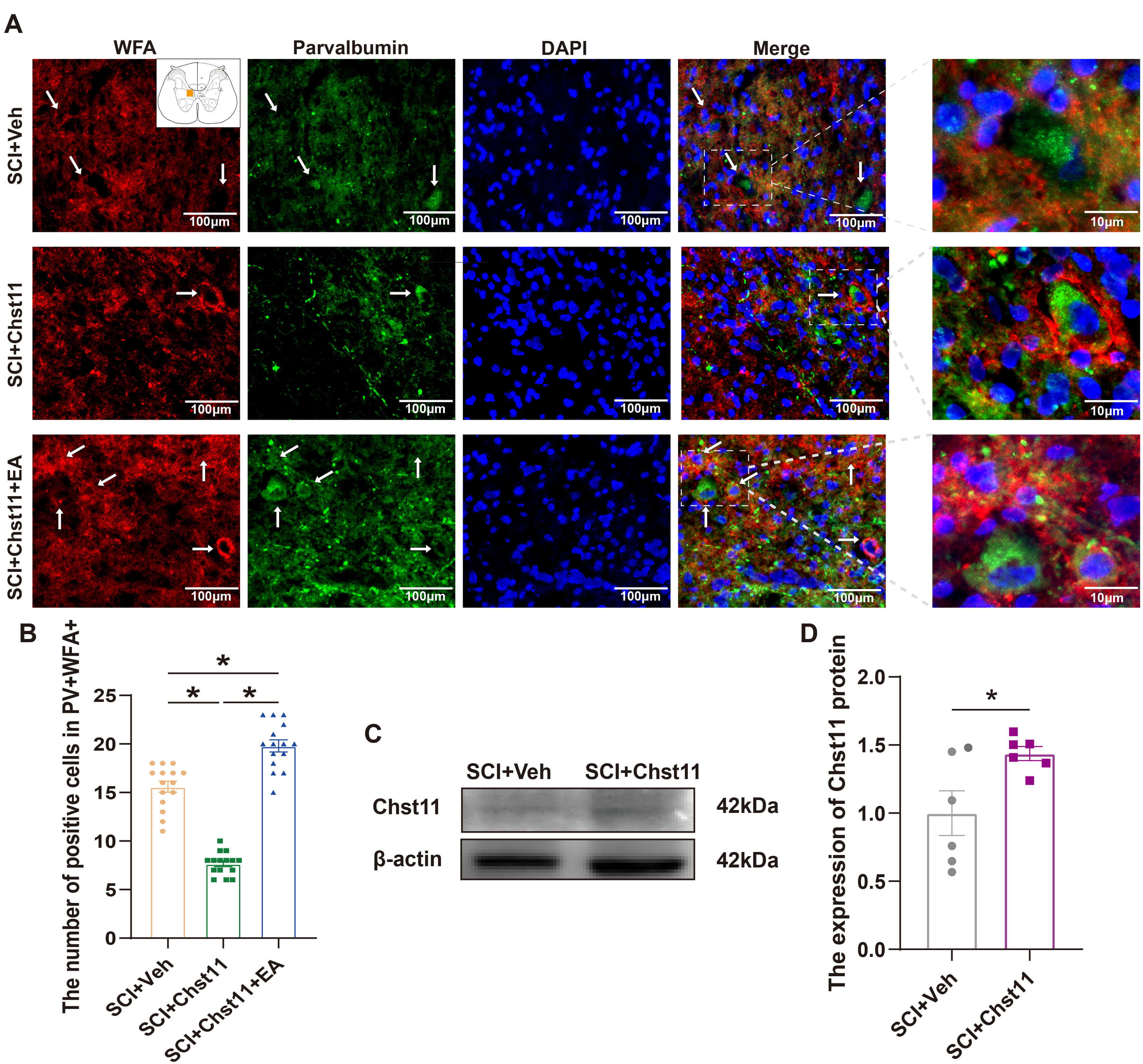 Fig. 6.
Fig. 6.
EA reversed the Chst11-induced reduction in the number
of PNN and PV IN positive cells. (A) Representative fluorescence plots of WFA +
PV+ in SCI + Veh, SCI + Chst11, and SCI + Chst11 + EA groups at day 14. (Scale bar = 100 µm and 10 µm). White arrows indicate relevant positive staining points. The white box marks the rightmost magnified area. (B)
Statistical graph of the number of WFA + PV + positive cells in SCI + Veh, SCI +
Chst11, and SCI + Chst11 + EA groups illustrated by IF. Each rat contains 5
sections. Statistical analysis of the data results was performed using one-way
ANOVA, followed by Tukey’s post hoc test (n = 3, F = 141.3, p
The results of qRT-PCR similarly indicated that EA did not alter the endogenous expression of CS-related synthase genes in rats while promoting SCI recovery, suggesting that its impact on the organism was minimal (Fig. 5N,O). IF results were consistent with those of WB, and EA effectively increased the number of PNN and PV IN after SCI, facilitating SCI repair (Fig. 6A,B). The original western blot data for this study are provided in the Supplementary Materials-WB.
After SCI, the volume and number of individual PNN around motor neurons at the site of injury decrease, and structural and functional remodeling occur [22], which is accompanied by atrophy of inhibitory gamma-aminobutyric acid ergic (GABAergic) interneurons and decreased expression of PV IN [23]. We observed reduced calcium activity of PV IN, as well as CS, and GAD expression levels, in response to PV IN activity and expression in an SCI model [24, 25]. In contrast, EA intervention reversed this effect and effectively facilitated SCI repair.
PNN, a densely aggregated form of ECM with a lattice-like network structure and
a negative charge, is widely present around many neurons in the spinal cord [26].
It predominantly wraps around the rapidly spiking
CSPGs are the primary components of PNN [38], and all types of CSPGs are composed of core proteins and a variable number of CS-GAG chains [39, 40], whose high negative charge is an essential determinant of PNN functions [41, 42]. This is because the high-intensity negative charge carried by CS-GAG can buffer cations generated after oxidative stress or toxic metal ions, thereby conferring neuroprotection to PNN [43, 44, 45]. Previous studies have suggested that C4S inhibits axonal growth and suppresses neuronal plasticity, whereas C6S promotes axonal growth and enhances plasticity [10, 46, 47]. CS-GAG is essential for PNN stability. Excessive degradation of CS-GAG causes excessive plasticity and increases the susceptibility of nerves to neurotoxic stimuli [5]. This study revealed that after SCI, the expression of CS, which represents an essential component of PNN function, decreased, accompanied by downregulation of GAD expression. This suggests that PNN structure was damaged differently after SCI due to the decrease in CS expression caused by the injury, leading to a sudden reduction in the number of PNN that WFA can recognize in the injured area and a similar decrease in the number of PV IN. Simultaneously, the homeostatic balance of neurons, initially maintained by PNN, was disrupted, and the plasticity of neurons was enhanced due to the reduction in PNN. When the damaged area was repaired and CS expression was significantly upregulated, the number of PNN also increased, as did the number and activity of PV IN, indicating that CS has an essential influence on the production and functional expression of PNN and the activity of PV IN. However, in this study, we found that GAD67 protein expression remained unchanged on day 1 post-injury, whereas GAD65 expression significantly increased during this period. As GAD65 and GAD67 are primarily expressed in GABAergic interneurons, the difference in their expression levels stems from the distinct regulatory mechanisms governing different interneuron subtypes. We hypothesized that GAD67 expression may decrease post-injury due to hypoxia and glutamate toxicity compared to GAD65 at the same time point. Conversely, GAD65 remains inactive under normal conditions but rapidly transitions to an active state in response to neurotrauma-induced GABAergic synaptic activity within a short timeframe, thereby increasing its expression levels [48]. The precise mechanisms underlying these differences remain to be explored, and we plan to investigate this phenomenon in subsequent studies.
Following SCI, mechanical trauma induces glial cells, which maintain central nervous system stability, to release toxins and multiple cytokines, endowing them with pathological and reparative properties. SCI activates perivascular cells and astrocytes, causing peripheral fibroblasts and Schwann cells to aggregate at the injury site, thereby forming glial scars and fibrotic scar tissue [49]. Astrocytes are the primary components of glial scar formation. Following SCI, astrocytes become activated, exhibiting hypertrophic changes, synapse growth, and increased expression of glial fibrillary acidic protein, transforming into reactive astrocytes (RA) [50]. Continuous aggregation and overlapping of RAs transform them into scar-forming astrocytes, forming extensive glial scars at the injury site. This inhibits axonal regeneration and limits SCI recovery [51]. Following SCI, disruption of PNN structure and stability leads to extensive deposition of ECM molecules at the injury site, thereby forming fibrotic scar tissue [52]. Concurrently, astrocyte formation interferes with ECM deposition, inhibiting endogenous repair of SCI. The resulting glial and fibrotic scars may impede the transport of CS-GAG to the damaged PNN, thereby hindering PNN repair. Since CS-GAG is a crucial component of the neuroprotective function of PNN, injury sites encapsulated by the glial scar lack the protective effects of a structurally intact PNN, further obstructing SCI repair.
The C4S/C6S ratio in the spinal cord affects PNN function and production. An increase in the C4S/C6S ratio promotes PNN formation during the developmental stage of neurons, suggesting a significant increase in PV IN during the neuronal developmental stage. However, a decrease in the C4S/C6S ratio at the stage of neuronal injury opens up the plasticity of PNN, suggesting that the structure of PNN is disrupted to some extent at the injury site [10]. The lower the C4S/C6S ratio, the more susceptible the mouse brain is to external stimuli that induce epileptic symptoms [53]. The results of this study on rats under different interventions after SCI revealed that a decrease in the C4S/C6S ratio represented the disruption of PNN stability and neuronal plasticity. When SCI was intervened and repaired, the upregulation of the ratio represented the reorganization of PNN structure, and the stabilizing function for maintaining the homeostatic environment of neurons was restored. Since PV IN was encapsulated by PNN, the restoration of PNN caused by the increase in the C4S/C6S ratio in the localized spinal cord after the intervention promoted the formation and expression of PV IN to some extent.
C4ST-1/Chst11 is an enzyme involved in CS sulfation that catalyzes the transfer of the sulfate group from the donor to the carbon-4 position of N-acetylgalactosamine to complete C4S sulfation [4]. Excessive C4S expression increases the inhibitory properties of PNN [54], increases structural stability, and decreases the ability to repair after injury. Transferase enzymes, such as Chst11, have been suggested to be key factors in fine sulfation homeostasis and indirectly influence neural maturation and stabilization of the CNS [8, 55]. The damaged spinal cord of zebrafish was repaired more rapidly after Chst11 knockdown, while the presence of Chst11 inhibited the progress of spinal cord repair [9]. In this study, we found that after injecting a virus overexpressing Chst11 into normal rats, the inhibitory properties of the rats were enhanced by localized genetic changes. After preparing the SCI model, the results revealed that rats injected with a virus containing Chst11 did not express sufficient CS-GAG. Combined with the results of previous studies, we suggest that this is due to the inhibitory effect of Chst11, causing a significant reduction in the repair capacity at the site of injury. This phenomenon was alleviated after subsequent EA intervention. This suggests that excessive Chst11 causes deterioration of the neuronal environment and increased inhibition at the injury site. However, EA can alleviate this situation, improve the neuronal external environment at the injury site, and promote injury repair.
In the CNS, especially in the spinal cord, PNN is predominantly wrapped around
the fast-spiking PV IN [27], a type of inhibitory
The effect of CS-GAG on PV IN function is also important, as CS deletion reduces the spiking response of PV IN [11]. Maturation of PV IN promotes PNN development and the sulfation pattern of CS-GAG. Thus, the sulfation mode pattern of CS-GAG can promote the maturation of PV IN and maintain cellular function [11]. The stable PNN structure, in which CS-GAG participates, also provides neuroprotection to PV IN.
We chose to use jia-ji acupoints (EX-B2) because they are primarily located on both sides of the spinal column, approximately 1.5 cm away from the spinal cord. There are 17 pairs of acupoints from T1 to L5, all of which are in proximity to the spinal cord. These acupoints can effectively promote recovery in the adjacent spinal cord. In this study, we selected the jia-ji acupoints near T9–T11, which are located at the upper and lower segments of the T10 injury site, and the EA intervention can effectively promote the recovery of SCI, which has been confirmed in our previous studies and clinical data analysis [13, 15, 21, 60]. Due to the proximity of these points to the chest and spinal cord, they can cause several adverse reactions if not handled appropriately. Through our observations and studies, we did not find any adverse reactions to EA in mice and rats, and no cases of death in mice and rats were attributed to EA. In clinical treatment, acupuncture has minor adverse reactions when targeting different diseases and patients; however, these can be avoided by adjusting the patient’s position and adequate preparation. Individual adverse effects may vary in degree due to personal differences, but overall, EA or acupuncture remains a safe intervention [61].
Electroacupuncture stimulation of EX-B2, followed by a specific parameter of electrical stimulation, is a traditional acupuncture technique that has been modernized in the context of therapy. EA stimulation of the EX-B2 can regulate the local blood supply and stimulate local nerve excitation, thereby promoting nerve repair at the site of injury. EA can improve the microenvironment of neurons at the SCI site, improve local blood supply, alleviate lower body paraplegia caused by SCI, and promote long-term recovery of neurological function in patients [62]. Previous treatment options for SCI, such as the application of biomaterials [63], stem cell therapy [64], delivery of nanomedicines [65] or ultrasound stimulation [66], and other traditional invasive and novel non-invasive therapies, have been associated with high costs, drug side effects, and high technical requirements. In contrast, EA only requires a cheap stainless-steel needle and an electrical stimulation device with an appropriate frequency for SCI treatment, which is effective and safe. From this perspective, EA has several advantages, including high-cost performance, simplicity of operation, safety, and effectiveness, and is expected to become a new technology for SCI treatment.
Additionally, through BBB scoring and hindlimb EMG testing in this study, we found that EA demonstrated favorable effects on restoring hindlimb motor function in rats with spinal cord injury. Based on changes in BBB scores, we can clearly determine the recovery stage of the rats’ hindlimbs. However, since BBB scoring is limited to evaluating hindlimb motor function in rats and cannot be directly applied clinically, existing guidelines or standards should be referenced for a more detailed assessment of patients’ limb motor function in actual clinical practice. Although the BBB scoring system cannot be directly applied in clinical practice, it indirectly reflects that EA treatment for SCI encompasses motor function aspects. This finding may further expand the clinical application scenarios for EA.
In this study, we observed that EA significantly enhanced PNN stability after SCI, increased PV IN activity, and promoted CS and GAD expression. Additionally, EA reversed the inhibitory effect of Chst11 and increased the expression levels of PNN and PV IN, thereby maintaining neuronal stability and promoting the recovery of neuronal functions. In summary, EA enhanced the stability of PNN and PV IN by increasing CS-GAG expression, thereby promoting SCI rehabilitation in rats.
In conclusion, the effectiveness and mechanisms of EA must be validated and elucidated; however, this does not prevent EA from being used as a safe and effective means of treating SCI.
The mechanism and effects of EA on SCI repair were investigated. The results revealed that EA could regulate the recovery of PNN structure and function via CS-GAG and GAD, improve PV IN activity, and reverse the inhibitory effect of Chst11 to promote SCI rehabilitation in rats.
BBB, Basso–Beattie–Bresnahan; Chst11, Carbohydrate sulfotransferase 11; C4ST-1, Chondroitin-4-sulfotransferase-1; CNS, Central nervous system; CSPG, Chondroitin sulfate proteoglycan; CS-GAG, Chondroitin sulfate glycosaminoglycan; C4S, Chondroitin 4-sulfation; C6S, Chondroitin 6-sulfation; CS, Chondroitin sulfate; ChABC, Chondroitinase ABC; EA, Electroacupuncture; ECM, Extracellular matrix; GAD65/67, Glutamic acid decarboxylase 65/67; IF, Immunofluorescence; PV IN, Parvalbumin interneuron; PNN, Perineuronal net; PV, Parvalbumin; qRT-PCR, Quantitative Real-Time Polymerase Chain Reaction; SCI, Spinal cord injury; WB, Western blotting; WFA, Wisteria floribunda lectin; EMG, Electromyography; SD, Sprague-Dawley.
This paper has been posted as a preprint on Research Square with doi: https://doi.org/10.21203/rs.3.rs-3404696/v1, which is available from: https://www.researchsquare.com/article/rs-3404696/v1.
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
RM, BC, and RH designed the study; BC, RH, XW, and JZ conducted the research; MS, YC, YH, and DH contributed to obtaining results and data; XY organized results and data and provided preliminary interpretations; BC, RH, MS, YH, and RM performed data analysis; BC and RH drafted the manuscript. YC and XY refined the manuscript structure and content and provided assistance and suggestions regarding the impact factor section. RM and DH provided major constructive feedback during manuscript revisions and finalized the draft. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
All animal experiments were conducted in accordance with all relevant animal testing and research ethics regulations and in accordance with the animal protocol approved by the Animal Ethics Committee of Zhejiang University of Chinese Medicine (ZSLL, 2017183). This study was conducted in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
We sincerely thank the third clinical college of Zhejiang Chinese Medical University, Hangzhou, Zhejiang, China for offering the experimental areas and instruments.
This study was supported by National Natural Science Foundation of China (No. 82174487 and 82505780). Special project of the Affiliated Hospital of Zhejiang Chinese Medical University (No. 2022FSYYZZ08 and 2022FSYYZY09).
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/JIN46448.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






