1 Hainan Province Key Laboratory of Brain Science and Health Translational Medicine Research Center in Tropical Environment, Hainan Medical University (Hainan Academy of Medical Science), 571199 Haikou, Hainan, China
2 Department of Orthopedic, Affiliated Haikou Hospital of Xiangya Medical College, Central South University, 571199 Haikou, Hainan, China
3 Department of Anatomy, School of Medicine, Southern Illinois University, Carbondale, IL 62901, USA
4 Hainan Provincial Key Laboratory of Carcinogenesis and Intervention, Hainan Medical University, 571199 Haikou, Hainan, China
†These authors contributed equally.
Abstract
Recent studies have indicated that stem cells could provide therapeutic benefits in several neurological conditions, including Alzheimer’s disease (AD). Adipose-derived stem cells (ADSCs) offer many advantages in that they are readily available from individual hosts, are robust, and secrete many factors that promote neuronal growth and homeostasis.
We transfected ADSCs with a viral construct for brain-derived neurotrophic factor (BDNF) and examined the effects of transplanting these cells into the hippocampus of 7-mo-old APPswe/PS1dE9 mice. After 6 mo, the hippocampus was examined for stem-cell survival, effects on BDNF and neprilysin-2 (NEP-2) levels, dendritic morphology using microtubule associated protein 2 (MAP2) immunohistochemistry, and amyloid plaque load.
We found that transplanted BDNF-ADSCs had survived after 6 mo. BDNF and NEP-2 levels were higher than sham controls, and dendritic architecture was improved. In addition, amyloid plaque numbers were reduced.
BDNF-ADSCs appear to confer benefits by simultaneously enhancing amyloid clearance and promoting neuronal structural repair. This multifaceted approach highlights the potential of engineering stem cells to target multiple pathophysiological hallmarks of AD, positioning BDNF-ADSCs as a powerful and synergistic cell-gene therapy strategy for this devastating disorder.
Keywords
- adipose tissue
- Alzheimer’s disease
- brain-derived neurotrophic factor
- hippocampus
- mesenchymal stem cells
Alzheimer’s disease (AD) is the predominant neurodegenerative disorder, with a
profound impact on millions of elderly people across the globe [1]. AD is
characterized by extracellular
Propelled by recent progress, stem-cell technology holds significant promise for generating innovative therapeutic interventions for neurodegenerative disorders [4, 5, 6]. Under appropriate inductive conditions, adipose-derived stem cells (ADSCs) undergo differentiation into cells that express established neuronal and glial protein markers, in addition to their inherent high proliferative potential in vitro [7]. Further, it has been shown that intracerebral administration of ADSCs reduced pathology and memory impairments in mouse models of AD [8, 9, 10, 11]. The clinical translation of ADSCs for neurological disorders is constrained by critical limitations, including uncertain efficacy, poor long-term survival after transplantation, and inefficient homing to target brain areas. These challenges hinder the reliability and therapeutic potential of ADSC-based approaches. Brain-derived neurotrophic factor (BDNF), the predominant neurotrophin in the adult brain, plays a fundamental role in regulating hippocampal synaptic plasticity and memory consolidation [12, 13]. Notably, BDNF levels are altered in both the brain and periphery of AD patients, suggesting its dysfunction in the pathogenesis of the disease [14, 15]. Consequently, restoring physiological BDNF levels represents a promising therapeutic strategy to mitigate the synaptic and neuronal loss underlying cognitive decline in AD.
ADSCs do not normally secrete BDNF [16, 17], although under certain conditions
they may be induced to do so [12, 18, 19, 20]. We therefore reasoned that genetically
engineering ADSCs to become a localized, long-term source of BDNF could
constitute a more effective strategy to counteract both synaptic failure and
pathology progression simultaneously. To test this premise, we transfected ADSCs
with a BDNF-expressing lentivirus to explore the possibility that this
manipulation would enhance their therapeutic potential. The modified ADSCs were
transplanted into the dorsal hippocampus of APPswe/PS1dE9 (APP/PS1) mice. Six
months after transplantation, we assessed the survival and spatial distribution
of BDNF-ADSCs within the hippocampus, their effects on the expression of BDNF and
neprilysin-2 (NEP-2; a neprilysin homolog and a key
Male APP/PS1 mice and C57/BL6J wild-type mice were purchased from Guangdong Medical Laboratory Animal Center (Foshan, Guangdong, China). Mice were maintained under ad libitum feeding conditions with free access to a standard laboratory diet and water. Neonatal C57/BL6J mice, raised at the Hainan Research Institute of Medicine, were used to obtain ADSCs. The experimental mice were divided into three groups (12/gp): (a) wild-type controls (C57/BL6J) and (b) APP/PS1 mice that received either BDNF-ADSCs or (c) sham surgery. All experimental procedures were initiated when mice reached 7 mo, and were conducted in compliance with the ARRIVE guidelines 2.0.
According to a previously described method [23], ADSCs were obtained from the
epididymal fat of 2–3-week-old male C57/BL6J mice. Briefly, the adipose tissue
was digested with 0.1% collagenase type I (BS-163, Biosharp, Shanghai, China) at
37 °C for 1 h, followed by mechanical dissociation and centrifugation at 800
Third passage (P3) ADSCs were harvested, fixed in 75% ethanol on ice, and
washed. After filtering, 1
A BDNF-encoding lentivirus was produced by co-transfecting 293T cells with plasmids pSPAX2 and pMD2G using LipoFiter™ (KS-TRLF-200/1000, LipoFiter, Shanghai, China). The viral supernatant was collected, filtered, and concentrated by ultracentrifugation. ADSCs were transduced with the lentivirus (Genomeditech, Shanghai, China) at a multiplicity of infection (MOI) of 40 in the presence of 5 µg/mL polybrene (40804ES76, Yeasen, Shanghai, China), and stable clones were selected with puromycin (ST551, Beyotime, Shanghai, China) over 3 weeks. BDNF expression was confirmed by western blot and immunocytochemistry. The 293T cell line used in this study was obtained from Wanwu Biotechnology (Delf-10618, Hefei, Anhui, China). All cell lines were validated by routine PCR assay and tested negative for mycoplasma.
APP/PS1 mice were anesthetized using isoflurane (PHR2874, Merck KGaA, Darmstadt,
Germany) (3–4% induction, 1.5–2% maintenance) and placed in a stereotaxic
frame (RWD Life Science, Shanghai, China). The skull surface was exposed and
either 2 µL of the DMEM/F12 vehicle or 1
After a survival interval of 180 days, mice were deeply anesthetized with 5% isoflurane and euthanized by cervical dislocation. For immunohistochemical studies, 6 mice from each of the three groups (C57/BL6J control, APP/PS1 sham, APP/PS1 ADSC) were perfused intracardially with phosphate buffered saline (PBS) (pH 7.4), followed by perfusion with 4% paraformaldehyde (12352100, SigmaAldrich, St. Louis, MO, USA) in PBS at 4 °C. After overnight post-fixation at 4 °C, the harvested brains were cryoprotected by immersion in a 30% sucrose-PBS solution. After equilibration, serial 40-µm coronal sections were obtained using a Leica CM1900 freezing microtome (Leica Biosystems, Nussloch, Germany) and stored in a cryoprotectant solution (C0171A, Beyotime) [24] at 4 °C for subsequent processing.
For western blot analysis, the remaining 6 mice/gp were transcardially perfused with ice-cold PBS. Following rapid extraction, the brains were placed on a pre-chilled plate for immediate hippocampal dissection. The isolated hippocampal tissue was snap-frozen and maintained at –80 °C until subsequent protein analysis.
Tissue sections were treated with 1% H2O2/PBS for 30 min, followed by
1 h blocking with 5% normal goat serum containing 0.3% triton X-100 (ST1723,
Beyotime). Sections were then co-incubated overnight at 4 °C with primary
antibodies against BDNF (1:1000, ab108319, Abcam), Flag-Tag (1:1000, ab205606,
Abcam), MAP2 (1:50,000, ab5392, Abcam), and
Protein extracts from ADSCs and hippocampal tissues were prepared using
radioimmunoprecipitation assay (RIPA) lysis buffer (G2002, Servicebio)
supplemented with protease and phosphatase inhibitors (Roche; 04906845001, Roche Diagnostics, Rotkreuz, Switzerland). Hippocampal
samples from 6 mice/gp were pooled before homogenization. Lysates were
centrifuged at 14,000 rpm for 15 min at 4 °C, and supernatants were collected for
protein concentration determination with a bicinchoninic acid (BCA) (P0010,
Beyotime) assay. Proteins (50 µg per lane) were separated on 10% sodium
dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) gels (P0012AC,
Beyotime) and transferred to poly vinylidene fluoride (PVDF) membranes
(IPVH00010, Merck Millipore, Darmstadt, Germany). After blocking, membranes were
incubated overnight at 4 °C with the following primary antibodies in tris-buffered
saline with tween-20 (TBST) (G0004, Servicebio): BDNF (1:1000, ab108319, Abcam),
NEP-2 (1:400, BS-11102R, Bioss Antibodies, Beijing, China).
All data are presented as mean
Flow cytometric analysis of P3 ADSCs confirmed a characteristic mesenchymal surface phenotype. Surface-marker profiling revealed that the cells exhibited strong positivity for the characteristic mesenchymal markers (CD73, CD90, CD105) and were negative for hematopoietic markers (CD34, CD45, HLA-DR) (Fig. 1A). This marker profile, consistent with published data [14], verified the successful isolation of a homogeneous population of mesenchymal stem cells, with minimal contamination from endothelial cells or other non-mesenchymal stromal cells (MSC) types. To assess their multipotency, ADSCs were subjected to specific induction protocols to promote their commitment to osteogenic, adipogenic, and chondrogenic lineages. Successful differentiation was confirmed by lineage-specific staining: Alizarin Red S for calcium deposits in osteocytes, Oil Red O for lipid vacuoles in adipocytes, and Alcian Blue for sulfated proteoglycans in chondrocytes (Fig. 1B–D).
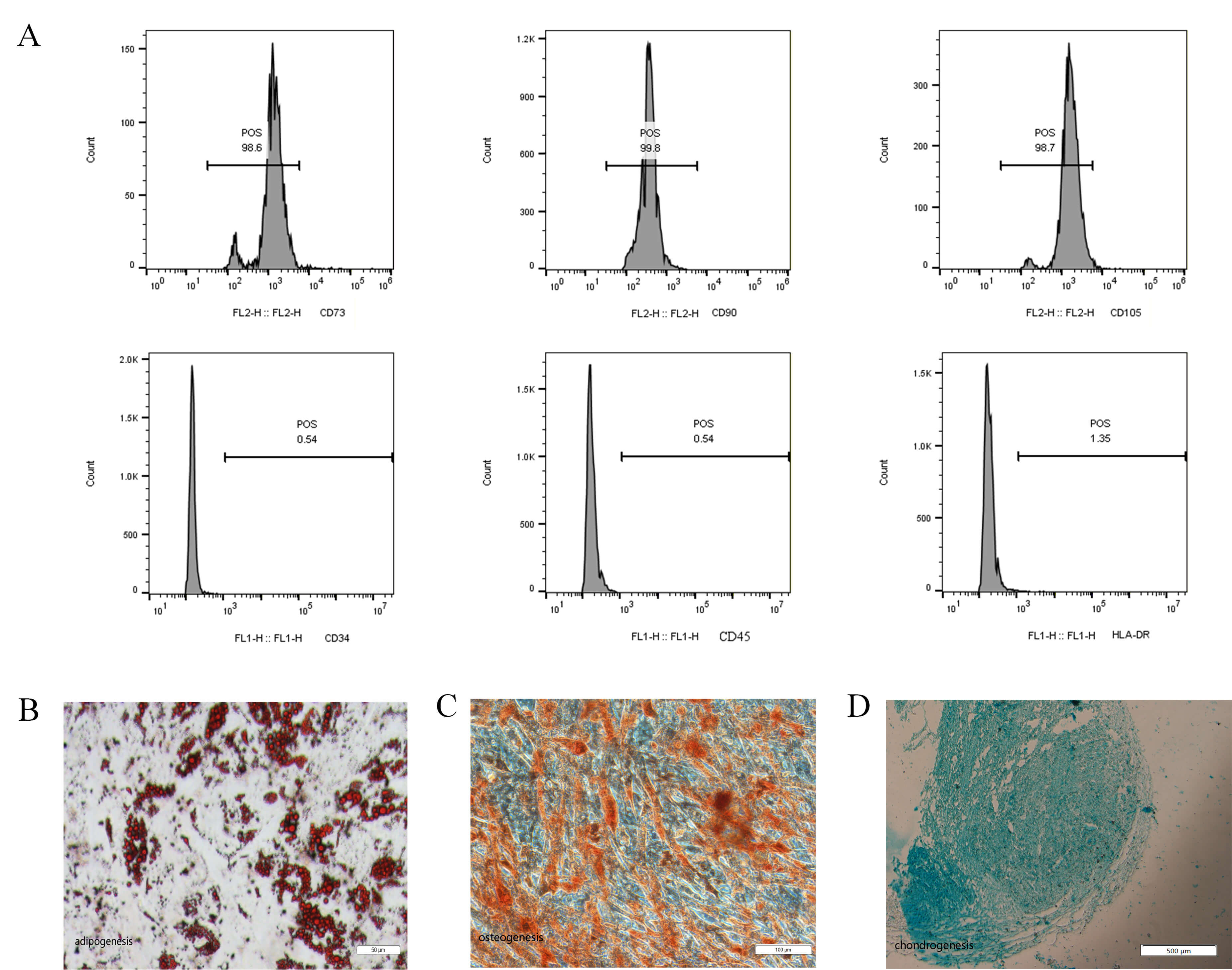 Fig. 1.
Fig. 1.
Cell-surface antigens of ADSCs detected by flow cytometry. (A) Representative results of flow cytometry analysis showed that ADSCs expressed CD73, CD90 and CD105. Bottom: in contrast, almost no expression of the hematopoietic lineage markers CD34, CD45 and HLA-DR was detected. (B) After induction of adipogenic differentiation, the cells showed positive staining for Oil Red O. Scale bar = 50 µm. (C) After induction of osteogenic differentiation, the cells showed positive staining for Alizarin red. Scale bar = 100 µm. (D) After induction of chondrogenic differentiation, the cells showed positive staining for Alcian Blue. Scale bar = 500 µm. (n = 3). ADSCs, adipose-derived stem cells; HLA-DR, human leukocyte antigen-DR; CD, cluster of differentiation; FL1-H, fluorescence channel 1- height; FL2-H, fluorescence channel 2- height; POS, positive.
Successful transduction was confirmed by detecting ZsGreen fluorescence in ADSCs under an inverted microscope at 72 h, indicating expression of the respective exogenous genes (human B‑lymphotropic virus [HBLV]-ZsGreen or HBLV-BDNF-flag-ZsGreen). ZsGreen was highly expressed in both cases (Fig. 2A–C). The expression of BDNF protein was confirmed by western blot analysis using total protein extracts from ADSCs transduced with HBLV-BDNF-flag-ZsGreen, with controls including both untransduced cells and HBLV-ZsGreen-transduced cells. The BDNF-flag-ZsGreen fusion protein was specifically detected at its predicted size via anti-flag western blot in the relevant group, confirming successful expression, whereas control groups showed no signal (Fig. 2D). Immunocytochemistry studies verified that BDNF was expressed in HBLV-BDNF-flag-ZsGreen transfected ADSCs (Fig. 2E).
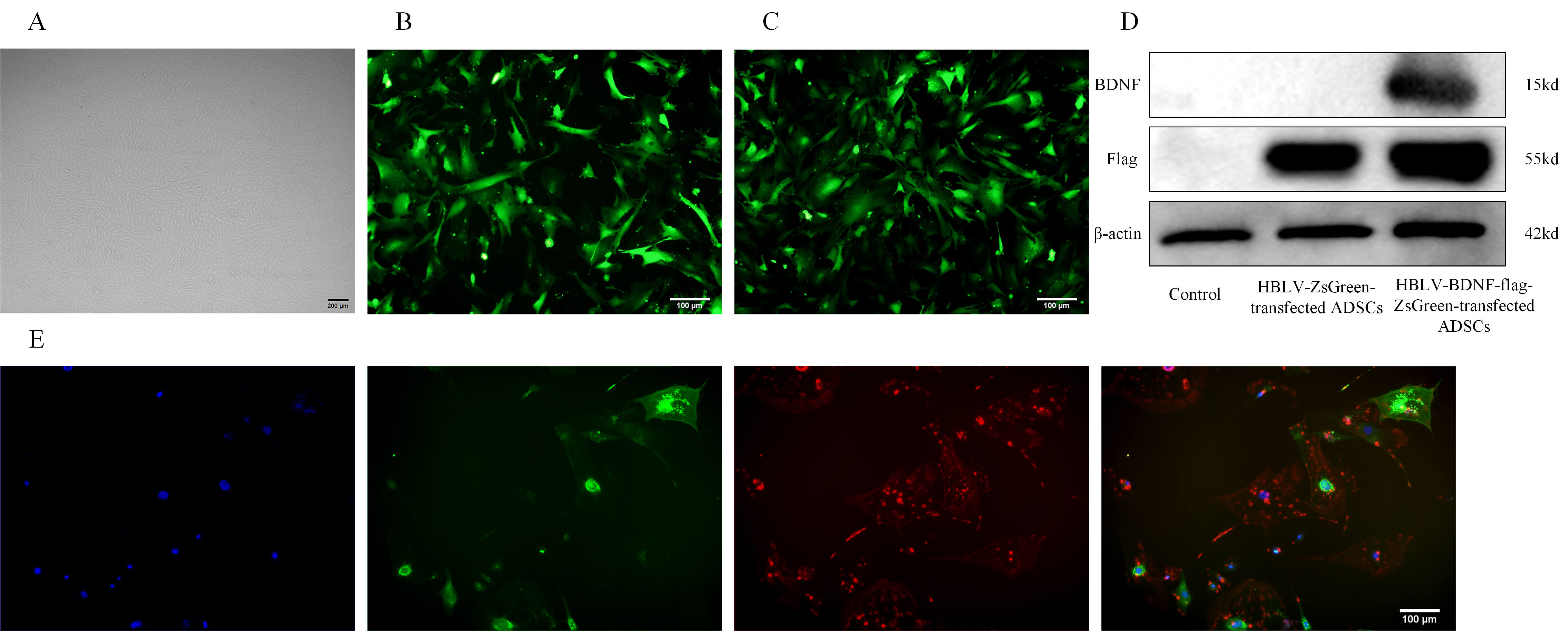 Fig. 2.
Fig. 2.
Expression of exogenous DNA in ADSCs after lentiviral
transduction. (A) Representative images of ADSCs. Scale bar = 200 µm. (B) HBLV-ZsGreen-transfected ADSCs were observed under fluorescence microscope.
Transfection success was estimated at
BDNF-ADSCs were transplanted bilaterally into the anterior hippocampus of 7-mo-old APP/PS1 mice, and analysis was performed 6 mo after transplantation. To investigate the survival and distribution of transplanted BDNF-ADSCs the mice were euthanized and coronal sections containing the anterior–posterior axis of the hippocampus were examined using a confocal microscope. As expected, no BDNF-ADSCs were detected in the AD sham group and the wild type (WT) control group (data not shown). Engrafted BDNF-ADSCs were found within and surrounding the retrosplenial cortex, but most cells were seen in stratum oriens and filed CA1 hippcampus (CA1) stratum pyramidale in the hippocampus. Notably, BDNF-ADSCs were seen well caudal to the injection site, indicating that they had migrated during the survival period (Fig. 3).
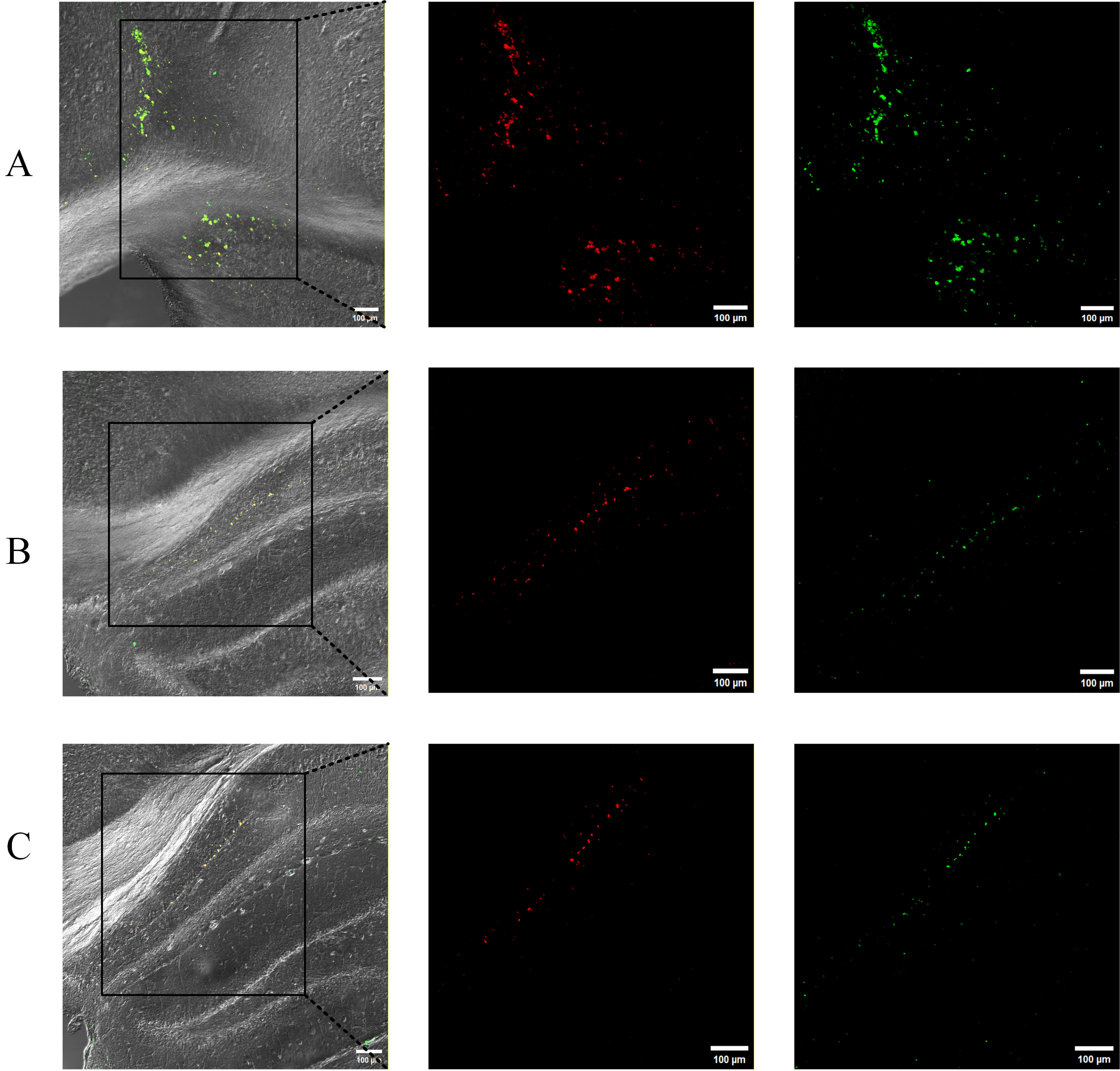 Fig. 3.
Fig. 3.
Representative distribution of transplanted BDNF-ADSCs in the hippocampus of mice. ZsGreen (Green) was used to label the BDNF-ADSCs, Cy3 (Red) was used to label the Flag expression. Each panel displays, from left to right: low-magnification image with higher power area shown in subsequent panels. On the far right are illustrations of the coronal section at which the image was taken. In (A), labeled cells are present in the retrosplenial cortex as well as in the hippocampus. In (B,C), labeled cells are seen in the medial CA1 pyramidal cell layer. Coronal sections shown are, from bregma: –0.94 mm (A); –1.46 mm (B); –1.70 (C); scale bar = 100 µm; n = 6.
Previous study has shown that the neurotrophin BDNF plays a key role in synaptic
plasticity [18]. NEP-2 is an important A
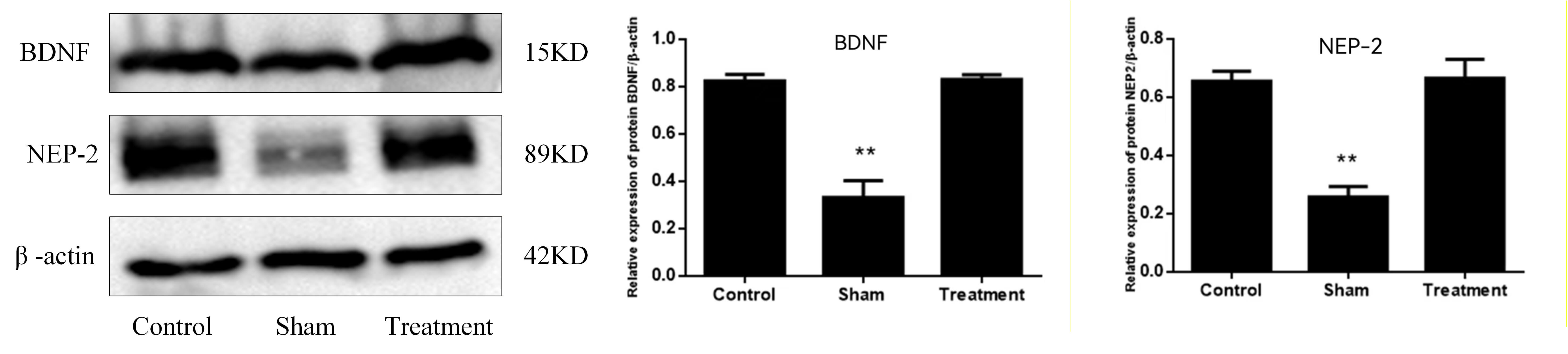 Fig. 4.
Fig. 4.
BDNF-ADSC transplantation normalized BDNF and NEP-2 protein
levels in the hippocampus of 13-month-old APP/PS1 mice. Levels of the two
proteins were significantly lower in Sham mice than in comparably aged C57/BL6J
controls, but treated mice had protein levels that were not different from
controls. Data are presented as mean
Degradation of dendritic cytoarchitecture is a hallmark of AD, and is reflected in reductions in MAP2 protein levels and patterning in mouse models of the disease [25, 26]. Compared to the control group (Fig. 5A), MAP2 staining in hippocampal area CA1 dendrites in 13-mo-old APP/PS1 mice revealed a shortened dystrophic pattern, indicating that microtubule structure was disrupted (Fig. 5B). In contrast, in mice that had received BDNF-ADSC injections, MAP2-stained dendrites were elongated and more densely distributed, similar to what was seen in control C57/BL6J mice (Fig. 5C).
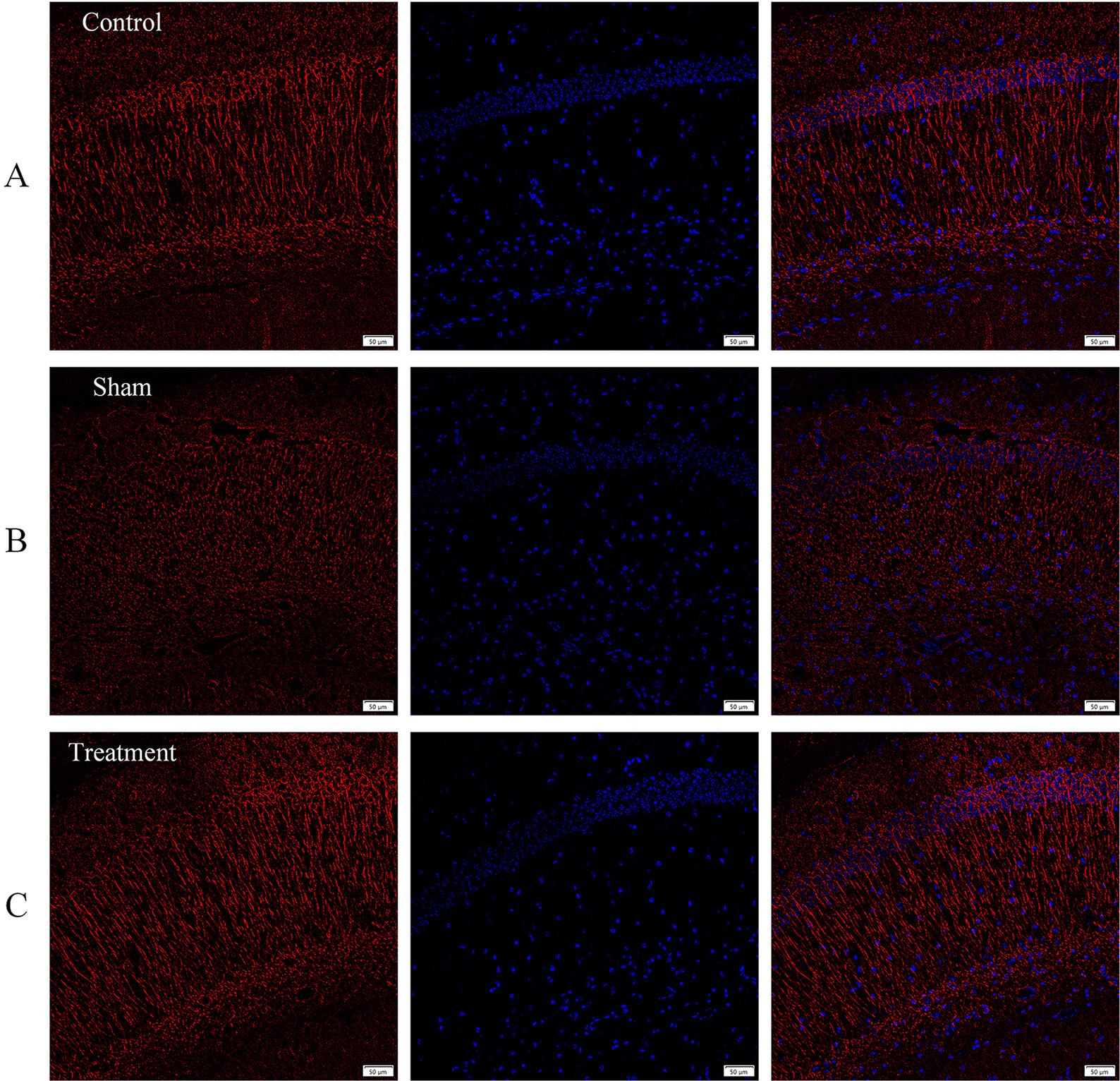 Fig. 5.
Fig. 5.
Engrafted BDNF-ADSCs changed dendritic morphology in the hippocampus of APP/PS1 mice. (A) Immunohistochemical analysis of MAP2 staining in control group. (B) In the sham group, MAP2 stained CA1 dendrites were shortened and dystrophic. (C) In the BDNF-ADSC treatment group, MAP2-stained dendrites appeared elongated and parallel to each other, similar to those seen in the control group. Red: MAP2; blue: 4,6-diamidino-2-phenylindole (DAPI). Scale bar = 50 µm (n = 6). MAP2, microtubule associated protein 2.
Immunofluorescence staining was performed on brain sections using the 6E10
anti-A
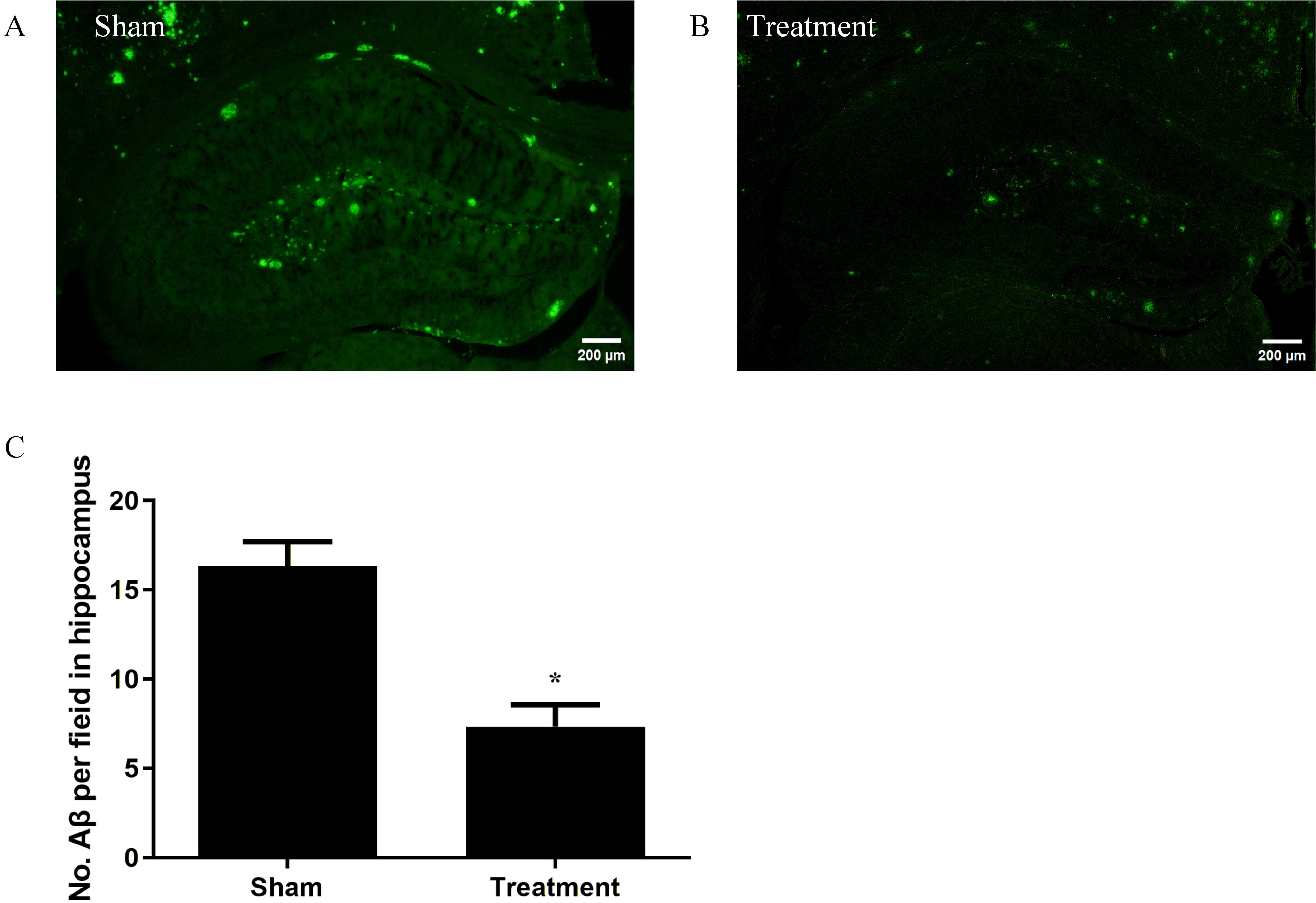 Fig. 6.
Fig. 6.
BDNF-ADSCs diminish A
This study investigated whether transplantation of BDNF-ADSCs would attenuate hippocampal neuronal pathology in APP/PS1 mice, an established model of AD. We used a novel combinatorial strategy by genetically engineering ADSCs to overexpress BDNF through lentiviral transduction. We found that BDNF-ADSCs survived in the hippocampus for 6 mo after transplantation, and appeared to migrate a substantial distance from the injection site. BDNF-ADSC transplantation significantly increased BDNF and NEP-2 expression in treated mice. Moreover, the dendrites of hippocampal neurons, as illustrated by MAP2 staining, were longer and more densely distributed than in sham mice.
Postmortem histopathological analyses of AD patients have demonstrated significant neuronal loss in key brain regions, including the cortex and hippocampus [27, 28]. By repopulating lost cells and exerting protective effects, cell-based therapies may mitigate the progression of neurodegenerative diseases [6]. Stem cells have been reported to be short-lived in the brain, in some cases surviving less than one month after transplantation [29]. Therefore, the therapeutic benefits of stem-cell transplantation may be mediated primarily by indirect mechanisms activated by the grafted cells, rather than by the direct actions of the cells themselves. For example, the repair mechanisms observed after cell transplantation are primarily mediated by the secretion of paracrine factors from the grafted cells [14, 29, 30, 31, 32]. The paracrine function of ADSCs involves the release of a broad spectrum of bioactive molecules, including growth factors and exosomes. These secretions enable them to contribute significantly to key biological processes: including the inhibition of apoptosis and inflammation, promotion of angiogenesis, and modulation of immune responses [33, 34, 35]. We confirmed that BDNF-ADSCs survive for at least six months after transplantation in the mouse brain. These cells exhibited distribution within the hippocampus and were associated with the amelioration of key pathological features in the APP/PS1 mice. Consistent with this concept, Yu et al. [36] demonstrated that genetic modification of ADSCs with vascular endothelial growth factor (VEGF) modified messenger RNA (modRNA) transfection enhanced the survival of the transplanted cells. Those engineered ADSCs, when co-transplanted with human fat into a murine model, exhibited elevated proangiogenic capacity, which in turn ensured long-term survival of the fat graft [36]. We do not know whether BDNF transfection itself contributed to the longevity of the transplanted cells because we did not do a direct comparison between transfected and nontransfected ADSCs. This is an important topic for future studies.
BDNF is critically involved in key neuronal processes, including promoting
neuronal survival, facilitating differentiation, regulating synaptic
transmission, and guiding structural plasticity through dendritic remodeling and
axon growth [37, 38]. Consistently reported low levels of BDNF, at both the mRNA
and protein levels, are a hallmark of postmortem AD brains, particularly
affecting the hippocampus and frontal, parietal, and temporal cortices [39, 40].
Extensive research has demonstrated that BDNF administration mitigates multiple
Alzheimer’s-related pathologies in APP transgenic mice, including reducing
A
NEP and NEP-2 are A
Dendrites, which are crucial for neuronal signal processing, are structurally
and functionally impaired by key AD pathogenic agents, including A
The presence of amyloid plaques, a defining neuropathological hallmark of AD, is a requisite feature of any valid animal model for this condition. Reducing plaque formation or eliminating existing plaques has long been a goal of the development of therapies aimed at forestalling or relieving cognitive symptoms. However, the relationship between amyloid plaques and AD is complex, as it appears that plaque accumulation in the brain occurs many years prior to the appearance of cognitive deficits [55]. Nevertheless, brain amyloid reduction remains a major focus, and is a particular target of new antibody agents [56]. Among other potential benefits, we found that transplantation of BDNF-ADSCs elicited a sustained reduction in hippocampal amyloid plaque load in APP/PS1 mice.
Despite the promising findings, this study had several limitations. First, the absence of a control group involving wild-type mice transplanted with ADSCs limits our ability to fully dissect the contribution of the disease environment to the observed effects and to rule out non-specific outcomes of the transplantation procedure itself. Furthermore, the most significant functional limitation is the absence of cognitive behavioral assays, which leaves the functional impact of the structural improvements on learning and memory unconfirmed. In addition, although MAP2 immunohistochemistry revealed enhanced dendritic architecture, direct evidence for functional synaptic plasticity, such as from electrophysiological recordings, is lacking. Finally, the observed increases in BDNF and NEP-2, although correlated with reduced plaque load, did not establish a definitive causal mechanism, and future studies using specific pathway inhibitors are needed to elucidate the precise molecular interplay.
Our study demonstrated that intra-hippocampal transplantation of BDNF-ADSCs
achieved long-term (6 mo) engraftment, elevated expression of BDNF and the
amyloid-degrading enzyme NEP, ameliorated dendritic pathology, and reduced
A
The datasets used and analysed during the current study are available from the corresponding author on reasonable request.
HZ, GMR: Conceptualization, Supervision, Writing. HZ, LM: Acquisition, Analysis. YZL, YHL, HL, CP: Investigation. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
All animal procedures were approved by the Ethics Committee of Hainan Medical University (approval number: HYLL-2021-071#) and were conducted in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals (NIH Publication No. 8023, revised 1978).
We acknowledge Hainan Medical University and Public Research Laboratory for providing the research facilities.
This research was funded by Academic Enhancement Support Program of Hainan Medical University [XSTS2025125]; Hainan Province Science and Technology Special Fund of China [ZDYF2022SHFZ290].
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/JIN46077.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






