1 Department of Clinical Pharmacy, Meizhou People’s Hospital (Huangtang Hospital), 514031 Meizhou, Guangdong, China
2 Department of Pharmacy, The Second Affiliated Hospital of Shantou University Medical College, 515000 Shantou, Guangdong, China
†These authors contributed equally.
Abstract
The main symptoms of Parkinson’s disease (PD) include olfactory impairment and tremor. Current treatment methods for PD generally have limitations such as short duration and severe side effects. The novel phosphodiesterase 4 (PDE4) inhibitor Roflupram (Roflu) mitigates inflammatory responses and enhances cognitive functions in individuals with neurological conditions. However, it remains unknown whether Roflu provides neuroprotection in a PD model induced by 6-hydroxydopamine (6-OHDA).
Cell viability was assessed using a 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay and flow cytometry. The expression level of tyrosine hydroxylase (TH) was evaluated by immunoblotting or immunofluorescence. Lactate dehydrogenase (LDH) release was measured to assess cytotoxicity. Intracellular reactive oxygen species (ROS) levels and mitochondrial membrane potential (MMP) were determined using fluorescent probes.
Roflu significantly increased cell viability in 6-OHDA-treated cells, as demonstrated by both MTT assay (17.18%, p < 0.001) and flow cytometry (12.20%, p < 0.001). It also upregulated the expression level of TH by 28.53% (p < 0.05). Furthermore, Roflu reduced LDH release by 23.54% (p < 0.001), indicating decreased cellular damage. Roflu markedly suppressed 6-OHDA-induced ROS accumulation by 57.82% (p < 0.001) and enhanced mitochondrial membrane potential (MMP) by 21.07% (p < 0.01). In addition, Roflu downregulated PDE4B expression in 6-OHDA-treated cells by 88.40% (p < 0.001). Knockdown of PDE4B mimicked the protective effects of Roflu, increasing cell survival by 18.43% (p < 0.001) and reducing LDH release by 21.54% (p < 0.001). Conversely, overexpression of PDE4B completely abolished the protective effects of Roflu, reversing both the increase in cell survival and the reduction in LDH release induced by Roflu in 6-OHDA-treated cells.
Roflu has demonstrated a clear protective effect against cell damage caused by 6-OHDA, which is closely related to the inhibition of PDE4B. These findings indicate that Roflu has substantial preclinical potential as a therapeutic candidate for PD and other neurodegenerative disorders involving oxidative damage.
Keywords
- Parkinson’s disease
- cyclic nucleotide phosphodiesterases
- neuroprotection
- oxidopamine
- oxidative damage
The classic feature of Parkinson’s disease (PD) is the massive production and
aggregation of the pathological form of
Phosphodiesterase 4 (PDE4) regulates the cyclic adenosine monophosphate (cAMP) signaling pathway, and abnormal cAMP signaling activation can accelerate neuronal damage by promoting neuroinflammatory responses and oxidative stress; thus, PDE4 has been implicated in the pathogenic mechanisms of various neurodegenerative conditions [7, 8]. PDE4 inhibitors can improve neuroinflammation and oxidative stress by enhancing the cAMP signaling pathway and exhibit protective effects in various neurodegenerative disease models [9, 10]. PDE4 inhibitors are considered candidates for the treatment of PD because of their anti-inflammatory, antioxidant, and neuroplasticity-promoting properties [9]. Previous studies demonstrated that PDE4 inhibitors exert therapeutic benefits in PD models by safeguarding dopaminergic neurons and improving impaired spatial working memory; however, their exact molecular mechanisms are unclear [11, 12]. Currently used PDE4 inhibitors generally cause dose-limiting adverse reactions (especially gastrointestinal toxicity, such as nausea and vomiting), e.g., roipram, which was discontinued in a clinical trial for depression because of intolerable gastrointestinal side effects [7].
Roflupram (Roflu; also termed FFPM) is a potent PDE4 inhibitor (Fig. 1A). Roflu demonstrated superior selectivity for the PDE4A4, PDE4B2, and PDE4D4 subtypes compared with rolipram, along with notable anti-inflammatory and neuroprotective activities [13, 14]. Further, Roflu shows a low incidence of adverse effects such as vomiting, and it can cross the blood-brain barrier [13]. Roflu enhances autophagy and inhibits the activation of the inflammatory body of microglia, thereby reducing inflammatory responses and neural damage. Its advantages have been confirmed in neurodegenerative disease models, i.e., it can significantly improve cognitive dysfunction, and exert neuroprotective effects through the cAMP/protein kinase A (PKA)/cAMP-response element binding protein (CREB) signal transduction axis. Roflu also alleviates neuronal damage in brain ischemic injury models [9, 13, 15], suggesting its potential as a safe therapeutic intervention for stroke. However, the therapeutic potential of Roflu in PD requires further research.
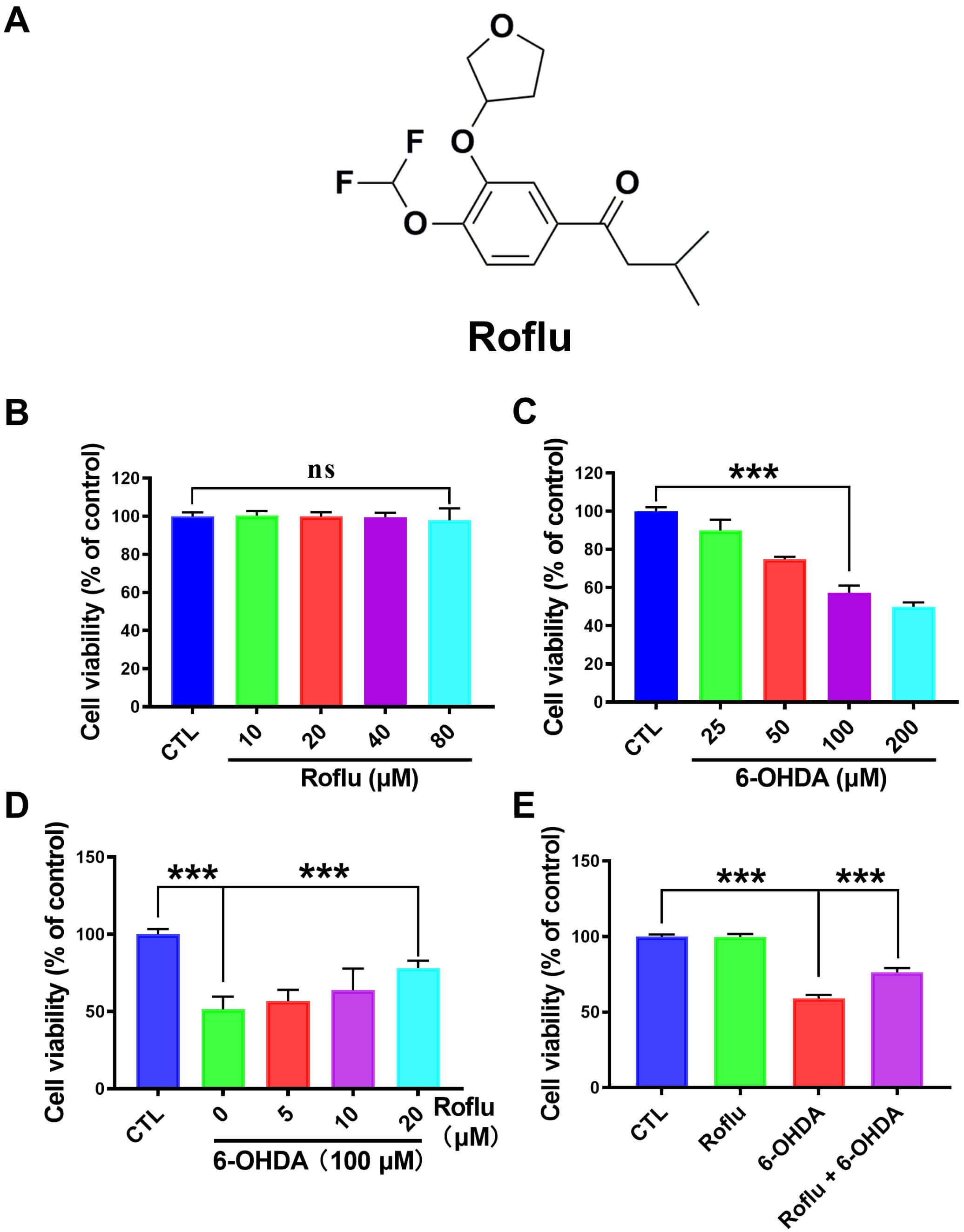 Fig. 1.
Fig. 1.
Roflu increases the cell viability in the context of 6-OHDA-induced
injury. (A) Structural diagram of Roflu. (B) The viability of cells treated with
a gradient of Roflu concentrations (10–80 µM) for 48 h was determined by
MTT assay. (C) The cytotoxicity of 6-OHDA (25–200 µM) following a 48 h
exposure was assessed in SH-SY5Y cells using the MTT method. (D) A 1 h
pre-incubation with Roflu (5–20 µM) was applied to cell cultures, and then
exposed to 6-OHDA (100 µM) for 48 h. (E) SH-SY5Y cell viability was
assessed by MTT after pretreatment with 20 µM Roflu for 1 h prior to
exposure to 100 µM 6-OHDA for 48 h. Data are presented in the form of mean
Cellular oxidative damage and stress play pivotal roles in the pathogenesis of
PD. Oxidative stress is a state of stress caused by the overproduction of
reactive oxygen species (ROS) [16, 17]. The high metabolic rate and the
propensity for dopamine auto-oxidation render dopaminergic neurons particularly
vulnerable in PD. Oxidative stress leads to a vicious cycle of mitochondrial
dysfunction. The deficiency in mitochondrial complex I results in excessive ROS
production, which is further exacerbated by a significantly compromised
antioxidant defense system in the brains of PD patients, preventing cells from
clearing excessive ROS. Abnormal aggregation of
This study evaluated the neuroprotective effects of Roflu, a PDE4 inhibitor, in
a PD model. Previous study showed that PDE4 inhibitors exerted potential
neuroprotective effects in PD, including 1-methyl-4-phenylpyridinium
(MPP+)/1-methyl-4-phenyl-1,2,3, 6-tetrahydropyridine (MPTP), rotenone, and
The manufacturers and item numbers of the reagents were as follows: MPP+ (#D048, Sigma-Aldrich, St. Louis, MO, USA); LDH Cytotoxicity Assay Kit (#C0017, Beyotime Institute of Biotechnology, Shanghai, China); bicinchoninic acid (BCA) protein assay kit (#23225, Thermo Fisher Scientific, Waltham, MA, USA); siPDE4B was purchased from GenePharma (Shanghai, China); Roflu (#SML2106, Sigma-Aldrich); dimethyl sulfoxide (DMSO; #D2650, Sigma-Aldrich); 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT; #M2128, Sigma-Aldrich); tetramethylrhodamine, ethyl ester, perchlorate (TMRE; #T669, Thermo Fisher Scientific); annexin V-FITC/PI double staining cell apoptosis detection kit (#KGA108, KeyGen Biotech, Nanjing, Jiangsu, China); phosphate buffered saline (PBS; #10010023, Gibco, Waltham, MA, USA); penicillin/streptomycin (#15140122, Gibco); anti-GAPDH (#2118, Cell Signaling Technology, Danvers, MA, USA); and anti-TH (#ab152, Merck-Calbiochem, La Jolla, CA, USA); anti-PDE4B (ab124772, Abcam, Cambridge, UK).
The SH-SY5Y (American Type Culture Collection, Manassas, VA, USA, ATCC, #CRL-2266, RRID: CVCL-0019) was cultivated in a humidified incubator (#51036153, Thermo Fisher Scientific) (37 °C; 5% CO2). Cells were cultured in DMEM/F12 (#C11330500BT, Thermo Fisher Scientific) growth medium containing 10% fetal bovine serum (FBS) (#10099141, Thermo Fisher Scientific) and 1% penicillin–streptomycin. At 80%–90% confluence, as monitored daily via morphology, the cells were passaged every two cycles using a 0.25% trypsin-EDTA solution (#25200056, Thermo Fisher Scientific). All cell lines were authenticated via autosomal short tandem repeat (STR) profiling and confirmed negative for mycoplasma contamination.
Following overnight culture in 96-well plates, cells were subjected to a 60 min pretreatment with serial dilutions of Roflu (10–80 µM) after the growth medium was aspirated. In the additional toxicity test, cells underwent a 48 h treatment with varying concentrations of 6-OHDA (25–200 µM). Moreover, cells were pretreated with Roflu (5–20 µM) for 1 h prior to a 48 h exposure to 100 µM 6-OHDA. The control (CTL) group was untreated. The Roflu group was treated with 20 µM Roflu, the 6-OHDA group was treated with 100 µM 6-OHDA, and the Roflu + 6-OHDA group was treated with 20 µM Roflu for 1 h and then 6-OHDA for 48 h. Cells in the above experiment were treated respectively, cell viability was assessed by the MTT assay. This involved a 4 h incubation using MTT reagent (0.5 mg/mL). The absorbance was measured at 570 nm after 10 min of orbital shaking.
Before experimentation, the cells had reached approximately 70% confluence.
The experiment comprised four treatment groups: an untreated CTL group, a group
treated with 20 µM Roflu, a group treated with 6-OHDA (100 µM), and a
group (Roflu + 6-OHDA) receiving a 1 h pre-treatment with 100 µM Roflu
prior to a subsequent 48 h challenge with 6-OHDA. Following 1 h pre-incubation
with Roflu (20 µM), cultures were exposed to 100 µM 6-OHDA for 48 h.
Plates were centrifuged (400
We divided the cells into four groups (CTL, Roflu, 6-OHDA, Roflu+6-OHDA), and the treatments for each group were carried out as described above. After discarding the culture medium, cells were treated with Roflu (20 µM) for 1 h and then challenged with 100 µM 6-OHDA for 24 h. Cells were then harvested by trypsinization. After centrifugation, following resuspension in 500 µL of binding buffer, the cells were stained with 5 µL Annexin V-FITC and 5 µL PI. Measurement of the apoptosis was performed using flow cytometry.
After being seeded in 24-well plates and cultured overnight. The experiment comprised four treatment groups: an untreated CTL group, a group treated with 20 µM Roflu, a group treated with 6-OHDA (100 µM), and a group (Roflu + 6-OHDA) receiving a 1 h pre-treatment with 100 µM Roflu prior to a subsequent 48 h challenge with 6-OHDA. After the cells are processed, they were washed three times. Subsequently, CellROX Deep Red Reagent (#C10422, Thermo Fisher Scientific) (10 µM) was applied to cells. Following three washes with PBS, ROS levels were assessed.
The CTL group was the untreated control group. The Roflu group was treated with a concentration of 20 µM Roflu, the 6-OHDA group was treated with a concentration of 100 µM 6-OHDA, and the Roflu + 6-OHDA group was treated with 100 µM Roflu for 1 h, with subsequent exposure to 6-OHDA for 24 h. Washing of the cells was gently carried out three times using PBS. We then stained the cells with 50 nM TMRE for 20 minutes. Following three additional PBS washes (5 min each) to remove unbound dye, the MMP was assessed by imaging TMRE fluorescence using an inverted confocal microscope (ECLIPSE Ti2-A, Nikon, Tokyo, Japan).
For PDE4B knockdown, we utilized a siRNA duplex (designated siPDE4B) with the following strands: sense: 5′-CCUGCAAGAAGAAUCAUAUTT-3′; antisense: 5′-AUAUGAUUCUUCUUGCAGGTT-3′. The negative control (NC) consisted of a non-targeting scrambled siRNA. Transfection complexes were prepared by incubating Lipofectamine 3000 (#L3000001, Thermo Fisher Scientific) with Opti-MEM (#31985070, Thermo Fisher Scientific), and 50 nM siRNA for 20 min. Following a 6 h exposure to the complexes, the medium was replenished with fresh culture medium. Cell harvesting for analysis was subsequently performed 24 h post-transfection.
The transfection mixture was prepared by separately diluting the following in Opti-MEM: (1) the PDE4B expression plasmid complexed with P3000™ reagent, and (2) Lipofectamine 3000. To enable complex formation, the separately equilibrated solutions were pooled, mixed gently, and subjected to a 20 min incubation. To assess functional changes, the cells were analyzed 24 h after being treated with the transfection complexes.
To extract cellular proteins, the samples were lysed using radioimmunoprecipitation assay buffer supplemented with the protease inhibitor PMSF (#P7626, Merck-Calbiochem) at a 1:100 volume ratio. Centrifugation was used to separate the soluble proteins into the supernatant fraction, which was retained. Loading consistency across samples was ensured by quantifying protein levels using a BCA assay. Size-based fractionation of proteins was achieved by electrophoresis on SDS-PAGE gels. Following separation, the proteins were blotted onto a polyvinylidene fluoride (PVDF) (#IPVH00010, Merck-Calbiochem) membrane. Following overnight incubation with primary antibodies at 4 °C, the membranes underwent extensive tris buffered saline with tween 20 (TBST) washes. Subsequently, they were probed with enzyme-conjugated secondary antibodies for 2 h to detect the target complexes. Finally, the target protein bands were visualized by chemiluminescence following the reaction with the necessary detection substrate, allowing visualization of their position and relative quantity.
The convention of reporting the mean
As an initial evaluation of its neuroprotective properties, the cytotoxicity of
Roflu was tested in SH-SY5Y cells via the MTT assay. The cell survival rate after
treatment with Roflu (10–80 µM) did not show significant changes, and cell
viability did not differ significantly (p
The previous experimental results indicated that Roflu enhanced the viability
following 6-OHDA exposure. To further confirm the neuroprotective effects of
Roflu, we used flow cytometry to determine the cell survival rate. The
experimental results were consistent with the aforementioned results. Roflu
conferred a significant protective effect, enhancing the survival of
6-OHDA-challenged SH-SY5Y cells (p
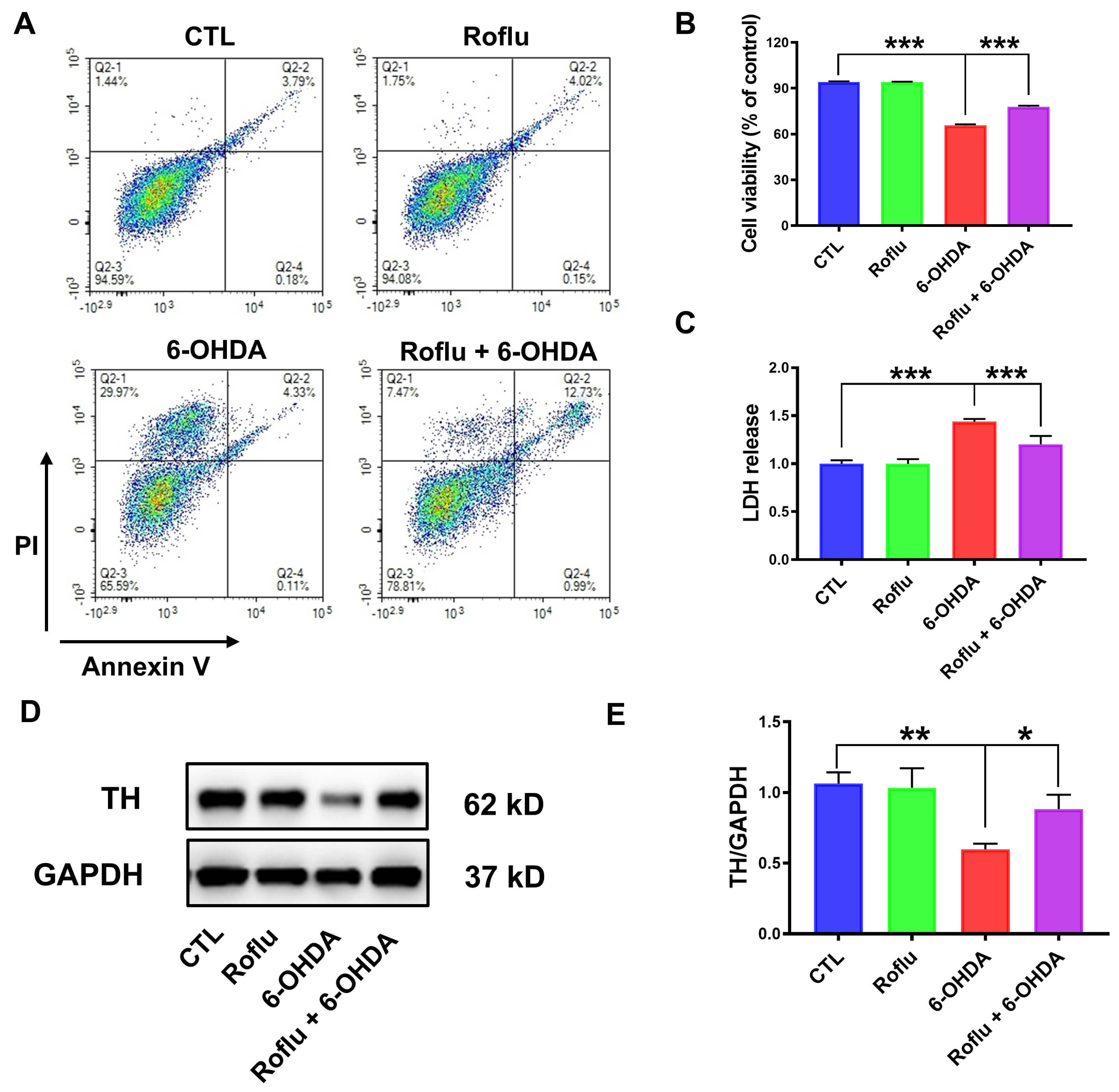 Fig. 2.
Fig. 2.
Roflu protects SH-SY5Y cells from cytotoxic effects caused by
6-OHDA. (A,B) After a 1 h pretreatment with 20 µM Roflu, cells were
subjected to 100 µM 6-OHDA for 24 h, with viability assessed using flow
cytometry. Data are presented as mean
Intracellular oxidative damage and mitochondrial dysfunction play crucial roles
in the neurotoxicity following 6-OHDA insult [28]. Previous findings demonstrate
that Roflu attenuates the cytotoxic effects of 6-OHDA, thereby improving cell
viability. However, its effect on intracellular oxidative damage is unknown.
Exposure to 6-OHDA significantly elevated intracellular ROS levels (p
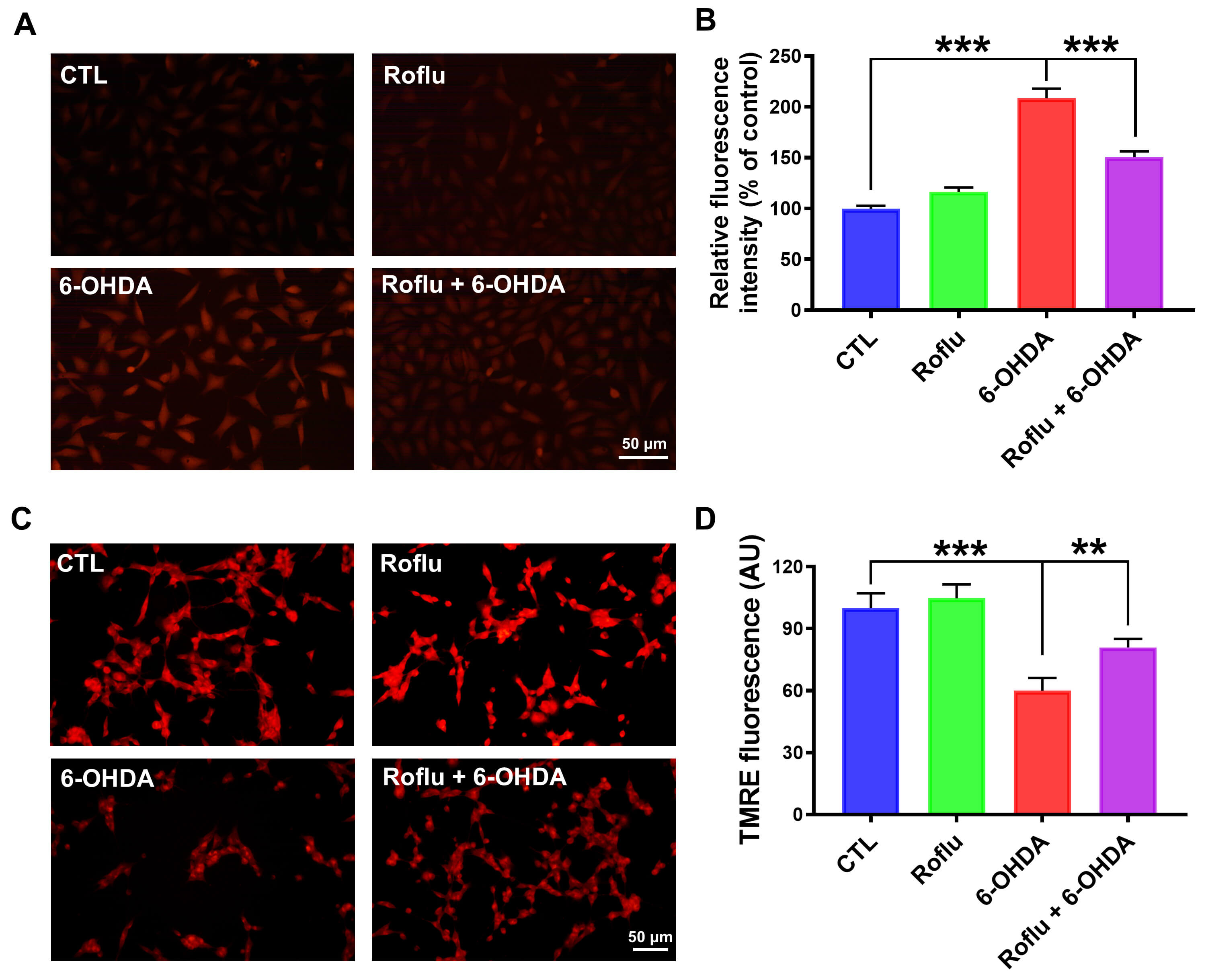 Fig. 3.
Fig. 3.
Roflu protects SH-SY5Y cells from the oxidative damage caused by
6-OHDA. (A,B) SH-SY5Y cells were first treated with Roflu (20 µM) for 1
h, then exposed to 6-OHDA (100 µM) for 24 h. After the treatment, the ROS
level was detected using CellROX Deep Red Reagent (10 µM). Scale bar = 50
µm. (C,D) SH-SY5Y cells were pre-incubated with Roflu (20 µM) for 1
h, followed by a 24 h treatment with 6-OHDA (100 µM), MMP was detected
using TMRE (50 nM). Scale bar = 50 µm. Data are presented as means
PDE4 shows prominent expression in multiple brain areas, notably the striatum,
hippocampus, and cortex, and cerebellum. Notably, among all PDEs, PDE4B
mediates key aspects of striatal neuronal activity, suggesting a potential link
to PD pathogenesis under pathological conditions [29]. 6-OHDA treatment induced a
significant upregulation of PDE4B expression (p
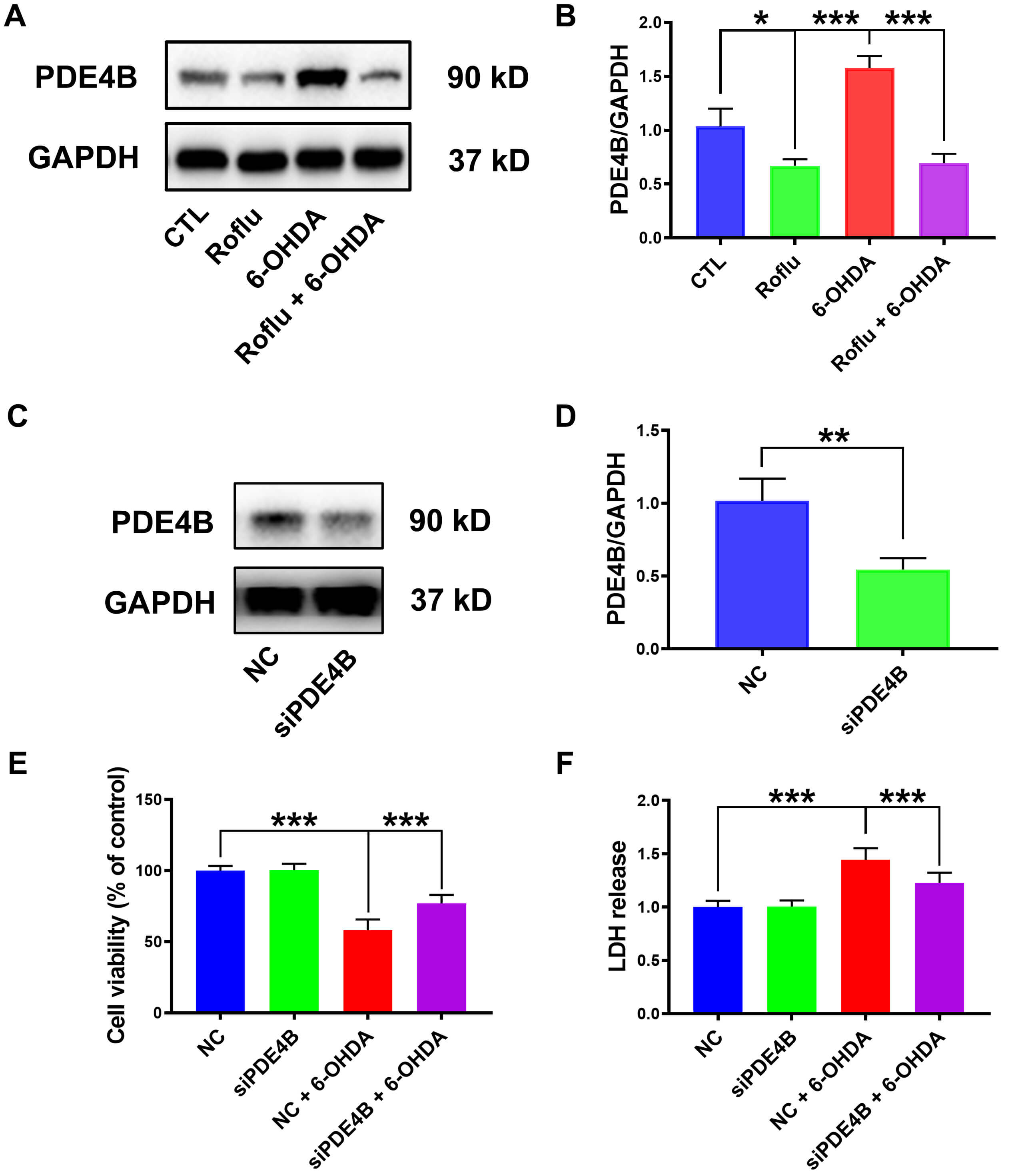 Fig. 4.
Fig. 4.
Roflu reduces cytotoxic effects by decreasing PDE4B in
cells induced by 6-OHDA. (A,B) Western blot analysis was performed to assess
PDE4B expression in cells following pretreatment with 20 µM Roflu
for 1 h and subsequent co-culture with 100 µM 6-OHDA for 24 h. The data are
presented in the form of mean
To establish a causal link between the PDE4B pathway and Roflu’s
neuroprotective effects against 6-OHDA in cells, PDE4B was
overexpressed. We generated an SH-SY5Y cell line overexpressing pDE4B. A
substantial increase in PDE4B expression was observed in these cells
(p
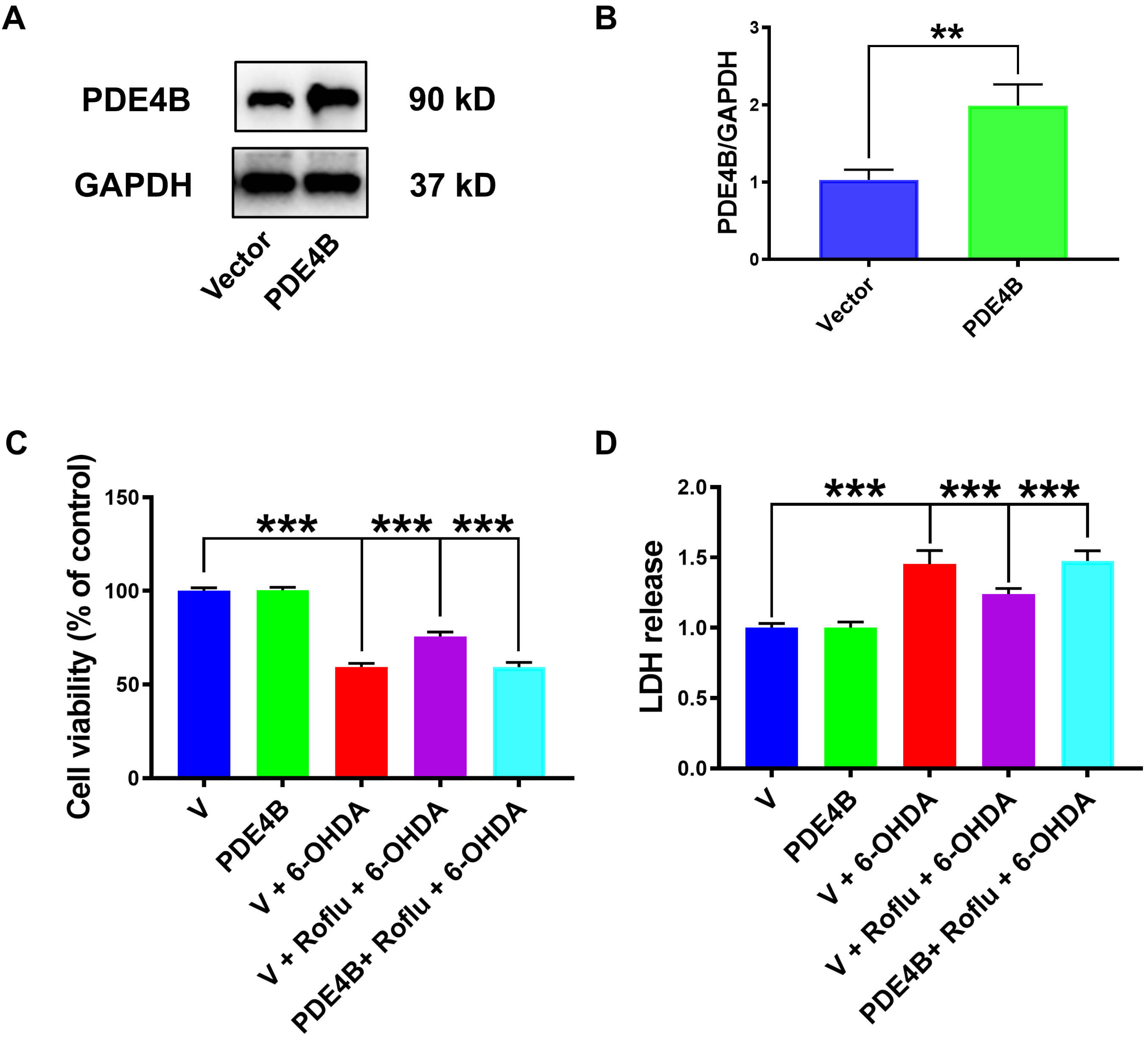 Fig. 5.
Fig. 5.
PDE4B overexpression abolished Roflu’s protective
effect on SH-SY5Y cells from 6-OHDA. (A,B) Following the protocol provided by
the transfection reagent manufacturer, transfection of SH-SY5Y cells with a
PDE4B plasmid led to robust protein expression, as detected by western
blot. Data are presented as mean
Our findings confirmed that the antioxidative protection offered by Roflu
against 6-OHDA in SH-SY5Y cells, which was previously observed but
mechanistically unexplained, was specifically mediated through PDE4B
inhibition, as PDE4B overexpression completely prevented the suppression
of ROS generation with Roflu (p
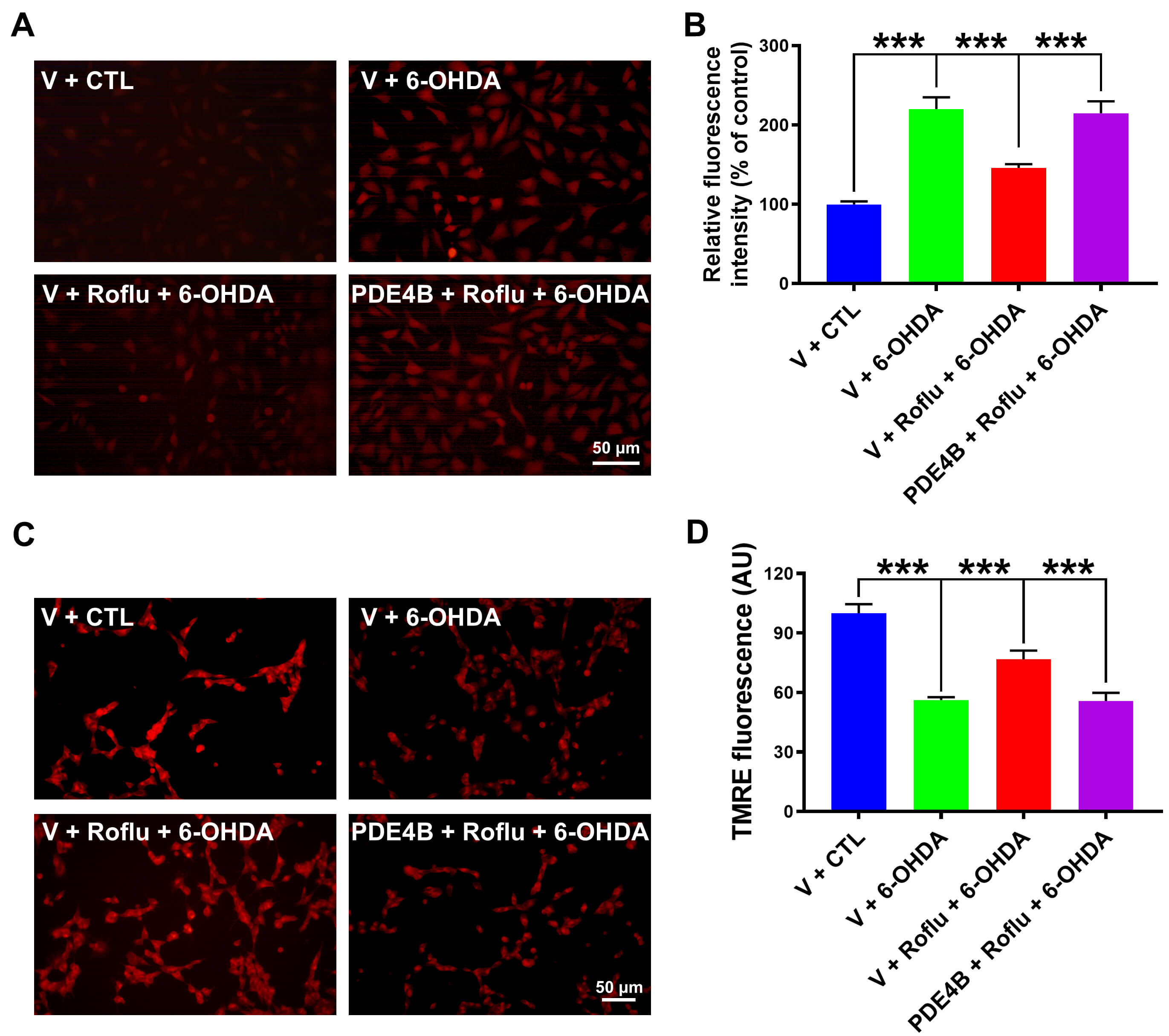 Fig. 6.
Fig. 6.
Overexpression of PDE4B blocked the ability of Roflu to
attenuate oxidative damage in 6-OHDA-treated SH-SY5Y cells. (A–D) Following
transfection with the PDE4B plasmid, prior to a 24 h treatment with 100
µM 6-OHDA, SH-SY5Y cells were pretreated with 20 µM Roflu for 1 h.
Subsequently, intracellular levels of ROS and MMP were measured. Scale bar = 50
µm. Data are presented as means
We found that specific inhibition of PDE4B by Roflu underlies its
neuroprotective effects in a PD model established by 6-OHDA. Specifically, Roflu
mitigated oxidative stress during the injury of SH-SY5Y cells, thereby reversing
the degeneration of dopaminergic neurons. These findings highlight PDE4B
inhibition as a potential therapeutic strategy for PD and deepen the
understanding of the regulatory mechanism of the PDE4/cAMP signaling axis in
neurodegenerative diseases, offering new candidate compounds and mechanism-based
evidence for the targeted treatment of PD and related oxidative damage-induced
neurodegenerative diseases. Although current mainstream drugs for PD, such as
levodopa, can temporarily improve motor function, they cannot stop the
pathological process of neuronal degeneration and are prone to cause adverse
reactions, such as the wearing-off and on-off phenomena, when used for a long
time [3]. Therefore, new intervention targets and drugs that can delay neuronal
damage are needed. Conventional PDE4 inhibitors, such as rolipram, were
discontinued in Phase II trials owing to adverse gastrointestinal reactions such
as vomiting [7]. Compared with rolipram, Roflu shows higher selectivity for
PDE4A4, PDE4B2, and PDE4D4 subtypes (IC₅₀ values for PDE4A4/B2/D4 subtypes are 3
to 10 fold lower than those of rolipram), and demonstrates minimal emetic
potential along with favorable blood-brain barrier penetration in animal studies
[13, 14], suggesting that its therapeutic window may be significantly better than
that of rolipram. Roflu demonstrates significant advantages in safety, with no
cytotoxicity observed within the concentration range of 10 to 80 µM in
experiments. Additionally, Roflu has been proven to enhance the autophagic
clearance of
Previous research showed that PDE4 inhibitors have potential neuroprotective
effects in PD, including in MPP+/MPTP, rotenone, and
The core manifestations of oxidative damage in PD include an overproduction of
ROS and a compromised antioxidant system (such as glutathione) [32]. Dopamine
metabolism is a major source of ROS, while mitochondria, the cellular
powerhouses, are also the primary site for its generation. Further amplify this
oxidative burden, their dysfunction (especially the reduced activity of complex
I) directly leads to an ATP synthesis disorder, causing neurons to be in an
energy crisis which initiates a vicious cycle of sustained ROS overproduction
[33]. Excessive ROS can directly attack and oxidatively damage lipids, proteins
(e.g.,
Next, we investigated the connection between the neuroprotective effects of Roflu and PDE4. The PDE4B expression within the substantia nigra is the highest among all phosphodiesterases and is related to the function of the striatum and the pathology of PD [29]. We detected a marked upregulation of PDE4B expression in the 6-OHDA-treated SH-SY5Y cell model, and Roflu could markedly inhibit this trend. Moreover, knockdown of PDE4B simulated the effect of Roflu, whereas overexpression of PDE4B counteracted the improvement effect of Roflu on cell viability and LDH release and even reversed its regulatory effects on ROS and MMP. These findings collectively demonstrate that the neuroprotective mechanism of Roflu involved specifically through the inhibition of the PDE4B subtype and that PDE4B is the key target mediating its neuroprotective effect. This study addresses the lack of attention to PDE4B in previous studies, clarifying the role of PDE4B with regard to the protective effects of Roflu. This study confirmed that Roflu can exert neuroprotection in the PD model by attenuating oxidative stress. However, the specific mechanism of action still requires further research. The nuclear factor-erythroid 2-related factor 2 (Nrf2)/ heme oxygenase-1 (HO-1) pathway is the core regulatory hub for the cellular capacity to combat oxidative stress. Previous study have shown that inhibiting PDE4 can regulate it [10], which provides a direction for further exploration of more downstream molecular pathways.
Our study thus provides experimental evidence that Roflu exerts neuroprotective effects in a well-established 6-OHDA-induced PD model, complementing the previous data from different PD models such as MPP+/MPTP. These findings strengthen the universality of PDE4 inhibitors in various models of PD. Our results clearly identify PDE4B as the core target for Roflu in regulating oxidative damage, resolving the key controversy of “functional heterogeneity of PDE4 subtypes” in previous studies [22, 23, 24]. Given its high selectivity and favorable safety profile, Roflu shows significant promise as a drug candidate targeting of PD and other neurodegenerative diseases that share common pathological features.
All data generated or analysed during this study are included in the research article.
JHZ: Conceptualization, Methodology, Investigation, Funding acquisition, Writing—original draft. XHY: Conceptualization, Methodology, Investigation, Writing—original draft. JLX: Conceptualization, Methodology, Investigation. QMW: Data curation, Formal analysis, Software. ZML: Conceptualization, Funding acquisition, Project administration, Resources, Writing—review & editing. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This work was supported by National Nature Science Foundation of China (Grant No. 82304434), GuangDong Basic and Applied Basic Research Foundation (Grant No. 2023A1515111199), China Postdoctoral Science Foundation (Grant No. 2022M713263), Scientific Research Starting Foundation for High-level Talents of Meizhou People’s Hospital (Grant No. KYQD202501, KYQD202502), PSM Guangdong Pharmaceutical Popular Science Research Foundation (Simcere Foundation) (Grant No. 2025KP178, 2025KP78), Shining Across China-Medicinal Research Fund (Grant No. Z04J2023E095), Social Development Science and Technology Plan Project of Meizhou (Grant No. 2024C0301079).
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/JIN45490.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






