1 Department of Neurosurgery, Western Theater Command General Hospital, 610083 Chengdu, Sichuan, China
2 Department of Hyperbaric Oxygen, Western Theater Command General Hospital, 610083 Chengdu, Sichuan, China
Abstract
The pathological increase in brain catabolites after traumatic brain injury strongly correlates with a higher risk of neurodegenerative disease. This review examines the pathogenic role of glymphatic clearance dysfunction in that process. The glymphatic network enables cerebrospinal and interstitial fluid exchange and paracellular flow. These processes are mediated by astrocytic aquaporin-4. Glymphatic function is regulated by arterial pulsatility, sleep-wake cycles, and intramural periarterial drainage, with meningeal lymphatic vessels acting as the final drainage site. Mechanical trauma causes aquaporin-4 depolarization and mislocalization; it also triggers neuroinflammatory activation and blood-brain barrier disruption. These processes ultimately impair glymphatic function and neurotoxic proteins become more localized and overproduced. Previous studies have linked clearance defects to secondary neuron injury. Current evidence in humans has come mostly from pilot studies. Recent advances in neuroimaging provide new assessment tools. Dynamic contrast-enhanced magnetic resonance imaging (MRI) reveals delayed tracer clearance. Diffusion tensor imaging along perivascular spaces shows abnormalities in key parameters. These imaging findings preliminarily associate with fluctuations in cerebrospinal fluid catabolites. Therapeutic research suggests several reparative strategies. Physical exercise improves aquaporin-4 polarization integrity. Cannabidiol administration in experimental models increases meningeal lymphatic drainage and reduces tau pathology. Angiotensin II type 1 receptor antagonists may indirectly improve clearance by stabilizing the blood-brain barrier. Lymphatic pathways have been used as therapeutic targets for cannabidiol. Biological evidence also supports their role in traumatic brain injury progression. Further investigation is needed to validate whether these represent independent contributing processes. Multimodal imaging, novel biomarker assays, and chronobiological modulation strategies are improving visualization. Microfluidic modeling could clarify the glymphatic-biomarker relationship; it may also advance precision medicine approaches for traumatic brain injury.
Keywords
- traumatic brain injury
- glymphatic system
- aquaporin-4
- intramural periarterial drainage
Traumatic Brain Injury (TBI) affects over 50 million individuals annually and represents a significant worldwide health burden [1, 2, 3, 4]. Long-term neuropathological sequelae of TBI, such as chronic traumatic encephalopathy and Alzheimer’s disease (AD), impose substantial socioeconomic costs [2, 3, 5]. As evidenced by clinical investigations, changes in cerebrospinal fluid (CSF) biomarker concentrations are directly associated with cognition deficits in months to years following moderate–severe TBI [4, 6, 7, 8, 9, 10, 11]. The exact mechanisms governing fluctuation in CSF biomarker levels remains incompletely understood. The majority of existing theories propose this fluctuation is caused by irregular production resulting from neuronal injuries, emerging evidence further reveal that deficits of neuronal clearance pathways may equally contribute to change in CSF biomarker levels [12, 13, 14].
As a main pathway for cerebral waste clearance, the glymphatic system maintains
homeostasis through a tightly orchestrated, hierarchical mechanism. The
glymphatic circulation initiates with arterial pulsations driving CSF influx
along periarterial spaces into the brain parenchyma. Polarized aquaporin-4 (AQP4)
channels distributed densely in the astrocyte endfeet mediate rapid
CSF-interstitial fluid (ISF) exchange [8, 13, 14, 15, 16, 17, 18]. Following this exchange,
metabolic waste products, such as
This review introduces a novel glymphatic-biomarker axis model that unites the
molecular, imaging, and clinical aspects to reveal the phenomenon of post-TBI
clearance dysregulation, and reveals the molecular pathogenesis including
different connected pathways and mechanisms. Mechanical shear forces in TBI
disrupt astrocytic endfoot architecture, inducing AQP4 depolarization and
mislocalization. Coexisting blood-brain barrier (BBB) damage will also promote
extravasation of plasma proteins, forming unsoluble A
The glymphatic system, a recently characterized intracranial clearance pathway,
derives its nomenclature from the functional integration of glial cells and
lymphatic-like drainage mechanisms [8, 12, 13, 20, 24, 25, 26]. This system drives
uni-directional flow of CSF into the brain parenchyma via periarterial spaces,
where astrocytic endfoot AQP4 channels mediate exchange with ISF [8, 13, 20, 24, 25, 26, 27]. Substantial AQP4-mediated paracellular flow within the interstitial
fluid (ISF) contributes to the active clearance of metabolic waste products
toward the venous drainage system [28, 29]. These wastes are transported along
perivenous pathways into meningeal lymphatics and deeper cervical nodes through
meningeal lymphatics [12, 16, 17, 18, 19, 30, 31]. The polarized distribution of AQP4 on
astrocytic vascular endfeet serves as the structural foundation of glymphatic
dynamics [9, 10, 32, 33, 34, 35, 36, 37]. This spatial arrangement, which is anchored on the
vascular interface, engages in tripartite interactions with neurons and cerebral
vasculature. It creates an exclusive brain-fluid microenvironment [12, 38].
Beyond the classical glymphatic route, the IPAD pathway contributes significantly
to interstitial solute clearance [16, 32, 36, 39, 40]. This alternative mechanism
relies on the structural integrity of the vascular basement membrane,
extracellular matrix composition, and the homeostasis of scaffolding proteins
such as
The kinetics of CSF-ISF exchange within the brain are driven mainly by physiologic forces. Mechanical factors including arterial pulsations from cardiac cycles, respiratory movements, and vasomotion collectively push CSF influx along periarterial spaces into the parenchyma, while perivenous spaces serve as efflux pathways for metabolic waste products [12, 18, 30, 31, 35, 43]. Water transport occurs selectively through the interstitial space via highly specific AQP4 water channels, which are densely expressed in astrocytic end-feet enveloping the central nervous system vasculature [28]. The efficiency of this system exhibits marked state-dependent modulation. During sleep, the cerebral interstitial space undergoes transient expansion exceeding 30%, resulting in enhanced CSF influx and accelerated clearance of neurotoxic metabolites. Wakefulness is correlated with lower activity of the systemic circuit and reduced removal of the waste product [12]. Circadian rhythms show significant regulatory influence on both AQP4 polarization patterns and glymphatic flux [33, 44, 45]. Core clock genes and circadian signaling pathways modulate the subcellular localization and expression of AQP4 in astrocytic endfeet through complex molecular mechanisms, resulting in temporally stratified clearance efficiency that correlates with diurnal variations in cognitive performance observed in aged murine models [38, 46, 47, 48, 49]. The cerebral surfactant protein SP-G demonstrates abundant expression in CSF outflow-associated structures. Its marked elevation in CSF from normal pressure hydrocephalus and central nervous system (CNS) infection cases suggests potential involvement in both cerebrospinal fluid rheology and immunoprotection, with positive correlation to total CSF protein levels reflecting dynamic alterations in CSF drainage under pathological conditions [9]. This leads to multifactorial processes that include not only bidirectional transport across the CSF-ISF exchange but also BBB function. Multiple related systems together function to preserve overall brain homeostasis through such multipoint flux dynamics [39].
Aquaporins (AQPs) constitute a family of integral membrane proteins initially characterized as passive channels dedicated to facilitating water movement across biological membranes, thereby playing an essential role in maintaining osmotic balance [50, 51]. Over the last ten years, growing evidence has broadened the understanding of their functional diversity beyond mere water permeability [52, 53, 54]. In addition to water, certain AQPs, known as aquaglyceroporins, enable transmembrane passage of small neutral solutes including glycerol and various metabolites, linking them to metabolic and inflammatory pathways as well as diverse physiological processes such as renal water reabsorption, cerebral fluid dynamics, adipocyte-liver triglyceride flux, and ocular lens stability [53, 55]. AQPs are also implicated in cellular volume regulation. Recent findings suggest their involvement extends beyond passive pore function to include potential roles in signal transduction, though the underlying mechanisms remain poorly elucidated. The concurrent expression of multiple AQP isoforms in many cell types raises the possibility of physical or functional interactions among different family members [50, 51].
Despite being promising drug targets for conditions like cerebral edema, metabolic dysregulation, and impaired glymphatic clearance, no pharmacological agent targeting AQPs has achieved clinical approval to date [56, 57, 58, 59]. The development of such therapies is hindered by the challenge of simultaneously addressing comorbid processes like edema and dysfunctional waste clearance with a single agent. As a result, the cooperative mechanisms between AQPs and other therapeutic targets remain an underinvestigated area deserving further exploration [54].
AQP4 is the primary aquaporin water channel expressed in the CNS, where it is
highly enriched in astrocytic end-feet and serves as a key regulator of water
homeostasis and glymphatic clearance. Its polarized localization on astrocytic
endfoot membranes facilitates rapid water exchange between CSF and ISF, with its
spatial distribution critically determining the directionality and efficiency of
fluid transport [11, 38, 47, 48]. MR imaging utilizing H217O as a
tracer revealed a substantial bulk flow of ISF, facilitating significantly
accelerated clearance of waste molecules from the parenchyma compared to purely
diffusion-driven transport [28]. Experimental evidence demonstrates that
AQP4-knockout mice exhibit markedly reduced CSF-ISF exchange efficiency,
accompanied by impaired clearance of A
The IPAD pathway represents an alternative clearance mechanism that depends on
the structural integrity of the vascular basement membrane and extracellular
matrix [16, 63, 64]. Genetic ablation of
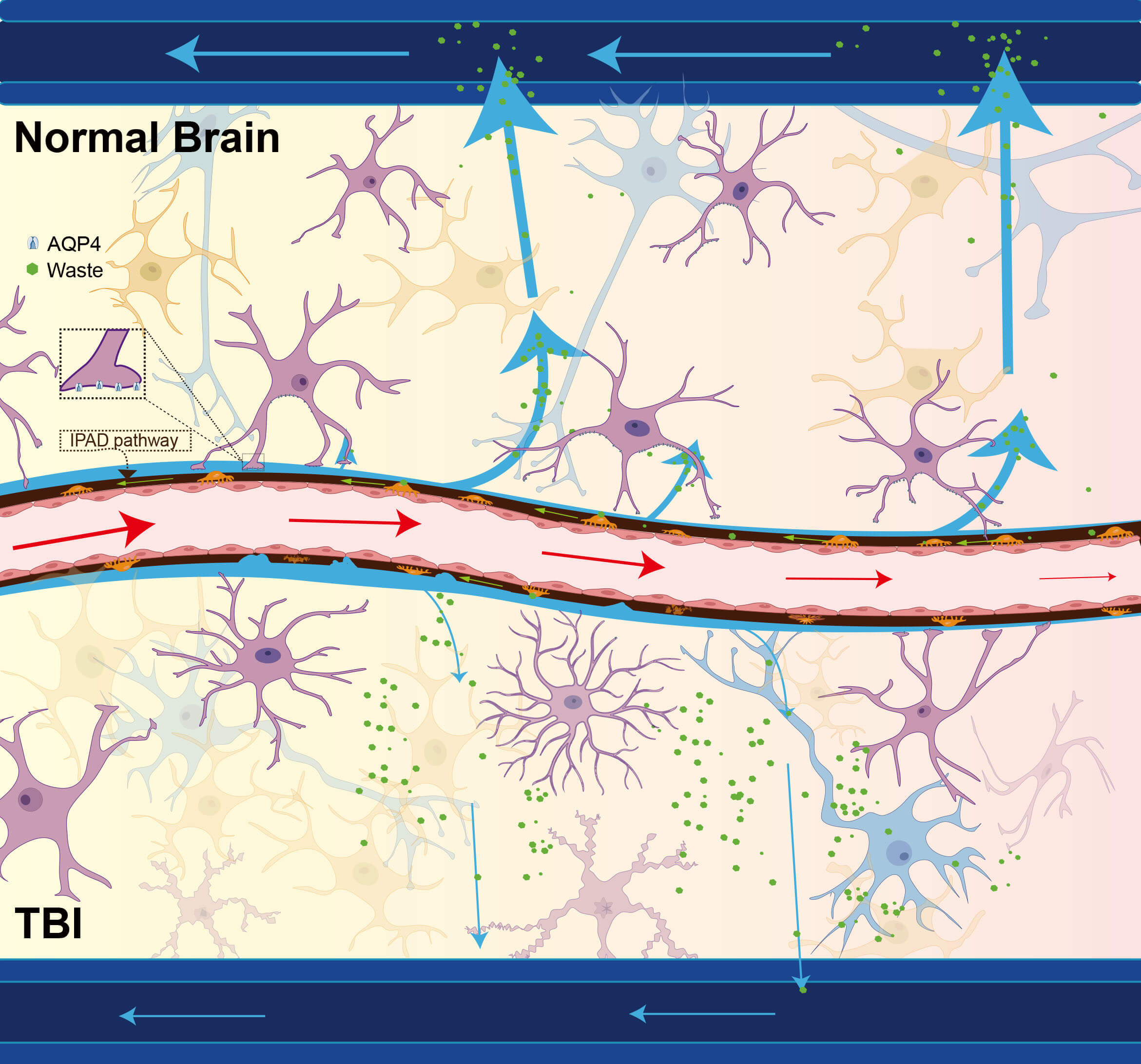 Fig. 1.
Fig. 1.
Comparative schematic of the glymphatic system. The glymphatic
system facilitates CSF circulation and interstitial waste clearance through a
coordinated paravascular network. CSF originating from the subarachnoid space
flows along periarterial channels into the brain parenchyma, driving bulk ISF
movement toward perivenous drainage routes. Solutes and fluids subsequently exit
through either SAS reabsorption or meningeal lymphatic vessel connected to
cervical lymph nodes. A critical component of this pathway involves AQP4 water
channels, densely clustered in astrocytic endfeet, which mediate efficient
CSF-ISF exchange across paravascular spaces. This system primarily supports the
clearance of metabolic byproducts, including lactate and soluble A
Recent advances in microphysiological systems have enabled the development of
“glymphatics-on-a-chip” platforms [47, 65]. These in vitro models have
validated the mechanistic link between neuroinflammation (e.g., lipopolysaccharide or A
A hallmark neuropathological feature of AD is cerebellar A
Pharmacological studies using the selective AQP4 inhibitor TGN-020 have established the central importance of AQP4-mediated glymphatic flow in tau clearance [31, 60, 67]. TGN-020 has been proposed as a potential AQP4 inhibitor candidate. Huber et al. [68] demonstrated, through osmotic swelling assays in Xenopus laevis oocytes, that this compound inhibits AQP4-mediated water transport in a dose-dependent manner. Experiments conducted in primary human and rat astrocytes have indicated a lack of AQP4 inhibitory activity, suggesting that its functional efficacy requires further investigation [69, 70]. Experimental inhibition not only obstructs CSF-ISF exchange but also significantly reduces tau clearance rates, identifying this pathway as a potential therapeutic target [60]. The analysis of experiments that tracked the fluorescent label of tau injected into mouse brain in vivo provided a clear sign of glymphatic outflow path, and through the quantification of the data it was indicated that the outflow was reliant upon the function of AQP4, showing the exact steps needed to allow for the movement of tau and its removal [71].
Neuroinflammatory mediators such as Chitinase-3-like protein 1 (CHI3L1, commonly referred to as YKL-40), elevated in AD and other
neuroinflammatory disorders, are secreted by activated astrocytes and may
indirectly impair glymphatic waste clearance through Chemoattractant Receptor-Homologous Molecule expressed on T Helper 2 Cells (CRTH2) receptor-mediated
suppression of neural stem cell
Evidences implicate glymphatic dysfunction plays an important role in multiple
protein aggregation disorders like subarachnoid hemorrhage (SAH) and Parkinson’s
disease (PD). In contrast to the initial view focusing only on alterations in
A
Cerebrovascular pathologies, including ischemia-reperfusion injury, post-stroke
neuroinflammation, subdural hematoma, and intracerebral hemorrhage with
consequent CSF pathway obstruction, consistently demonstrate glymphatic
impairment accompanied by pathological accumulation of metabolic byproducts such
as A
These observations indicate that the coexistent abnormality of glymphatic and BBB transport systems is the essential mechanism responsible for the buildup of pathological proteins in cerebrovascular disorders, while both neurological imaging and biochemistry play an essential role in diagnosis and prognosis.
Mechanical shear forces and pressure waves induced by TBI trigger immediate
astrocyte activation and loss of AQP4 polarization. Astrocytes play a complex and
dual role in the progression of TBI, with their functions differing significantly
between acute and chronic phases. During the acute phase, reactive astrogliosis
primarily exerts protective effects. Activated astrocytes form a glial scar,
referred to in the literature as the perilesional glial barrier, which sequesters
the core injury area and confines the spread of damage and infiltration of
inflammatory cells, thereby preserving surrounding healthy tissue. Astrocytes
also contribute to the formation and regulation of cerebral edema through
specific ion channels like AQP4 [79]. Although this activity may initially
exacerbate cytotoxic edema, astrocyte activation and barrier formation are
crucial for controlling subsequent secondary injury. During the chronic phase,
the function of astrocytes is more contradictory: they maintain the activation
and stabilization of the glial scar through long-term activity, thereby forming
an obstacle to axonal regeneration and the recovery of neural circuits; yet, at
the same time, it also has an impact on recovery in some active ways, including
secretion of various neurotrophic factors. To promote neurogenesis and
synaptogenesis, sustain angiogenesis, and aid in axonal remodelling, GDNF and
VEGF stimulate additional factors, such as astrocytes. Through their effects on
inflammation, cellular homeostasis, and the establishment of an appropriate
environment for brain plasticity and repair, astrocytes ensure sufficient levels
of GDNF and VEGF that facilitate these processes [79, 81, 82]. Experimental
studies utilizing murine TBI models demonstrate significant reductions in
parenchymal CSF-ISF exchange efficiency post-injury, accompanied by decreased
polarized distribution of AQP4 at astrocytic endfeet membranes [44, 83]. These
pathological changes facilitate rapid cerebral accumulation of metabolic
byproducts (including A
It is clear that the outcomes can reveal possible impacts of post-TBI on various causes responsible for the glymphatic function disorders in general as well as present a basis for a targeted intervention to improve neurological outcomes after TBI. Besides, it has been demonstrated that the TBI affected clearance may involve neurovascular regulation and inflammatory pathways. Both seem essential elements of a treatment.
The BBB and the GL arranged for their separate but coordinated functions to
accomplish the maintenance of neural homeostasis; however, a TBI would
concurrently impair the BBB and GL despite their inherently divergent physiology.
The TBI damages not only the tight junctional protein of the vascular endothelium
(claudin-5 and occludin) in the BBB to increase its permeability, but also alters
cerebral hemodynamics to decrease the glymphatic inflow driven by cardiac
pulsation [39]. Extravasated blood components including albumin and hemoglobin
infiltrate the interstitial compartment, triggering activation of
neuroinflammatory cascades in astrocytes and microglia, and formation of
persistent A
The relationship between the BBB, iron deposition, and cognitive function is increasingly recognized as a critical axis in both neurodevelopmental and neurodegenerative processes. The BBB serves as a dynamic interface that regulates the exchange of substances between the blood and the brain, including iron, an essential element for neuronal metabolism, myelination, and neurotransmitter synthesis. Dysfunction of the BBB, through increased permeability or impaired water exchange, can lead to aberrant iron accumulation in susceptible brain regions such as the globus pallidus, substantia nigra, and thalamus [87, 88].
Quantitative susceptibility mapping (QSM) studies reveal that iron deposition follows a region-specific trajectory, with rapid accumulation during early development and a plateau in later life. These iron dynamics are closely coupled to BBB function, as measured by techniques such as diffusion-prepared pseudo-continuous arterial spin labeling (DP-pCASL), which quantifies the water exchange rate across the BBB. This rate serves as a sensitive indicator of BBB efficiency, with lower values associated with higher iron accumulation and potentially impaired clearance mechanisms [89].
In pathological contexts such as AD and cerebral small vessel disease, BBB
breakdown facilitates the extravasation of neurotoxic substances promoting
oxidative stress, neuroinflammation, and neuronal damage. The APOE
These pathophysiological insights demonstrate that optimal post-TBI metabolic clearance requires coordinated therapeutic strategies targeting both BBB integrity restoration and glymphatic pathway reconstruction, emphasizing the need for dual-system intervention approaches in clinical management.
The pathological accumulation of metabolic byproducts following TBI represents a
critical determinant of long-term neurodegenerative consequences [91]. Impaired
fluid dynamics coupled with elevated A
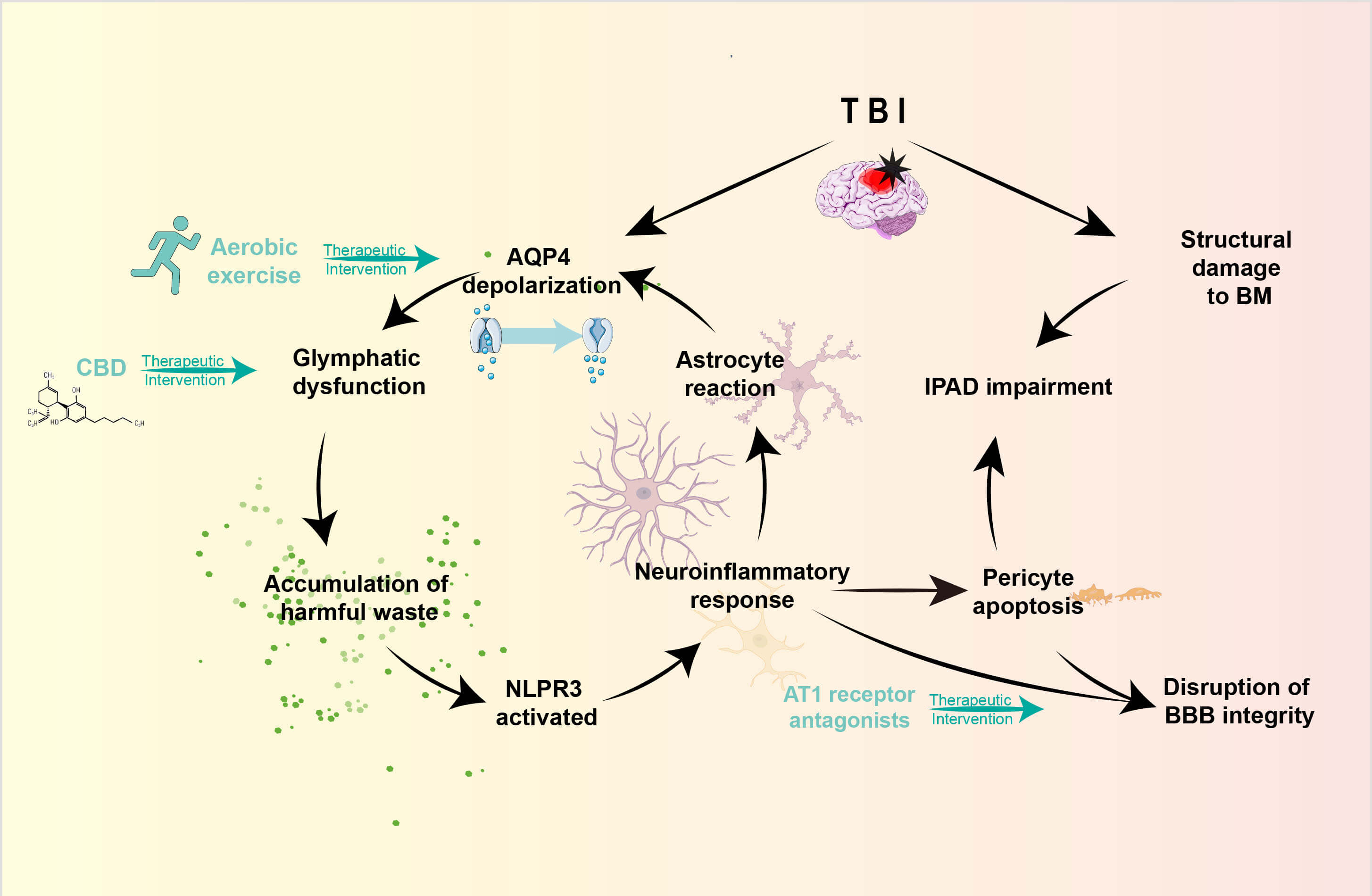 Fig. 2.
Fig. 2.
The alterations in glymphatic function following TBI and the therapeutic interventions. TBI induces multifaceted pathological alterations that compromise both the glymphatic system and the IPAD pathway. The disruption of AQP4 polarization critically diminishes CSF-ISF exchange, resulting in the accumulation of neurotoxic metabolites and subsequent NLRP3 inflammasome activation. This neuroinflammatory cascade triggers reactive astrogliosis, which further exacerbates AQP4 depolarization. Concurrently, structural degradation of BM, pericyte loss, and reactive astrogliosis synergistically impede interstitial clearance. These pathological modifications establish a self-perpetuating cycle of dysfunctional waste clearance and sustained neuroinflammation, ultimately amplifying secondary injury progression post-TBI. NLRP3, NACHT, LRR and PYD domains-containing protein 3; BBB, blood-brain barrier. The figure was created with Adobe Illustrator (version 29.0).
Preclinical investigations with murine TBI models indicated that CBD
administration could enhance motor and cognitive recovery, accelerate cerebral
tracer clearance, restore AQP4 polarization, and reduce p-tau/A
Translational clinical studies employing DTI-ALPS have pioneered noninvasive
glymphatic assessment in TBI patients. Initial correlational analyses reveal an
inverse relationship between diminished ALPS indices and elevated CSF
A
Even though existing clinical reports of accumulating evidence are currently few in number, such proof-of-concept studies lay an important groundwork for future multicenter clinical trials. Combining our preclinical mechanistic insights with the growing body of clinical biomarkers will continue to form the basis upon which we may provide disease-targeted interventions.
The A
Limbrick et al. [6] reported in their clinical investigation elevated
concentrations of CSF sAPP
All these findings present a deeper understanding on how the glymphatic system
is controlling the levels of certain proteins after TBI by regulating post-TBI
changes in A
The water channel protein AQP4 serves as the principal molecular effector of
glymphatic function, with its quantitative expression and polarized distribution
directly governing CSF-ISF exchange efficiency. Aerobic exercise training has
been shown to upregulate AQP4 expression while reorganizing its polarized
orientation, thereby enhancing A
The research into both lysosome-associated membrane proteins LAMP-1 and LAMP-2 in the field of neurodegenerative diseases and lysosomal storage disorders is very extensive. Little attention has been given to the assessment of glymphatic biomarkers following TBI [42, 74]. Given the critical dependence of astrocytes and neurons on endosomal-lysosomal pathways for clearing both exogenous and endogenous protein aggregates, quantitative changes in LAMP-1/2 expression may serve as indicators of compromised intracellular degradative capacity. Such impairment could significantly influence the glymphatic system’s ability to intercept and transport metabolic waste products to clearance pathways [42].
Future investigations should employ integrated methodologies combining ELISA-based quantification of CSF LAMP-1/2 levels with advanced neuroimaging techniques to elucidate their biological significance in post-TBI clearance dysfunction. This multimodal approach may reveal novel mechanistic insights into the dysregulation of protein homeostasis following neural trauma [17]. SP-G is postulated to reflect alterations in the extracellular matrix, while LAMP-1/2 is indicative of lysosomal function/autophagy activity. These emerging molecular biomarkers have the potential to complement and enrich the AQP4-centric framework from orthogonal perspectives. They may provide critical insights into distinct pathological processes, such as neuroinflammation and impaired protein degradation. Looking forward, it may be feasible to construct a multimodal biomarker panel that integrates AQP4 (water transport), neuroimaging (system-level function), and these molecular indicators (microscopic pathology), thereby enabling more precise disease subtyping and therapeutic target selection. The key molecules of glymphatic system and their functional consequences following TBI are summarized in Table 1.
| Key molecules | Consequences following TBI | Influence on clearance efficiency | Interventions |
| AQP4 | Depolarization and reduced expression | Impaired CSF-ISF exchange leads to pathological accumulation of metabolic waste products. | Aerobic exercise, CBD |
| LAMP-1/2 | Lysosomal storage disorders | Compromised endosomal-lysosomal system leads to defective exogenous and endogenous protein aggregates clearance. | A potential candidate |
| SP-G | Elevated in CSF | Alterations in fluid rheological properties result in drainage resistance. | Uncertain |
| CHI3L1 | Excessively released by activated astrocytes | Indirectly impair glymphatic waste clearance through CRTH2 receptor-mediated suppression of neural stem cell |
CRTH2/ |
CBD, cannabidiol; LAMP, lysosome-associated membrane protein; SP-G, surfactant protein-G; CHI3L1, Chitinase-3-like protein 1; CRTH2, Chemoattractant Receptor-Homologous Molecule expressed on T Helper 2 Cells.
Dynamic contrast-enhanced MRI enables real-time visualization and quantification of glymphatic fluid dynamics through sequential monitoring of contrast agent (Gd-DTPA) influx, retention, and clearance patterns in cerebral tissue [96]. As reviewed by Verheggen et al. [39], this technique provides critical metrics including CSF-ISF exchange rates and contrast retention duration, reflecting both arterial pulsation-driven flow and AQP4 functional status [97].
The experimental application on rodent models of ischemic stroke verifies that
the DCE-MRI parameters undergo synchronous changes in association with the APP
deposition in secondary thalamic infarcts, confirming the feasibility of
utilizing this method to analyze the abnormal glymphatic function due to
post-traumatic or vascular injuries [19, 97]. Some preliminary studies applied
DCE-MRI on the TBI population and found that compared to un-injured groups, both
the lesioned and peri-lesional areas showed prolonged CSF clearance time which
correlated with higher CSF A
The DTI-ALPS approach is an objective method to quantify the glymphatic system
by depicting the water molecule diffusion pattern inside the peri-vessels and
demonstrate the reduction of ALPS index after TBI presents strong inverse
correlations with cognitive impairment and increased A
In order to cope with the inherent shortcomings of single-modality imaging
techniques, researchers in today’s era increasingly rely on multimodal
neuroimaging. For example, there is a typical scheme that jointly employs kinetic
parameters acquired from DCE-MRI and DTI-ALPS indices to evaluate the rates of
contrast agent clearance and the flow of aqueous diffusion simultaneously [17, 97]. This integrated approach, when correlated with multi-omics data
including CSF/plasma concentrations of A
Evidences show that glymphatic-targeted interventions improve the efficiency of
waste clearance in TBI as well as other neuropathologies. Aerobic exercise
changes both the cardiac-induced arterial pulsatility and AQP4
expression/repolarization to boost the CSF-ISF exchange and diminish the
deposition and inflammation of A
| Interventions | Mechanisms | Preclinical findings | Clinical translation potential |
| Priority 1: Aerobic exercise | Augmenting arterial pulsatility | Cognitive deficits improved and enhanced A |
An effective prevention strategy in high-risk populations |
| Upregulated AQP4 expression | |||
| Improved AQP4 polarization efficiency | |||
| Priority 2: T1 receptor antagonists | Preserve BBB integrity | AT1 receptor knockout mice exhibit preserved expression of BBB tight junction proteins, restoration of AQP4 polarization, and concomitant reductions in both A |
A new indication for an established drug with a favorable safety profile |
| Restore glymphatic flow | |||
| Priority 3: CBD | Augmentation of meningeal lymphatic and glymphatic flow velocities | Enhances motor and cognitive recovery in TBI rat | Phase I clinical trial is currently in progress |
| Restoration of AQP4 polarization | |||
| Reduction of p-tau/A |
The development of microfluidic chip technology (glymphatics-on-a-chip)
represents a significant advancement in elucidating the molecular regulatory
mechanisms governing glymphatic system dynamics. These innovative platforms
employ co-culture systems incorporating human astrocytes and cerebral
microvascular endothelial cells to accurately simulate CSF-ISF flow dynamics
[102]. Experimental validation using this model has demonstrated the dual effects
of LPS, A
Research on the circadian regulation of glymphatic activity suggests that good
sleep quality and circadian rhythms stabilization may be alternative ways to
improve post-TBI metabolic clearance without pharmaceutical intervention. Chronic
sleep-deprivation animal models reveal much higher nocturnal AQP4 polarization
and glymphatic clearance capacity than those during daytime [103]. Sustained
sleep limitation annihilates this circadian difference with correspondingly high
brain A
Advancing investigations should focus on multidimensional data integration (incorporating neuroimaging, CSF/plasma biomarkers, and genomic profiles) to establish international consensus standards for the glymphatic-biomarker axis, facilitating early TBI diagnosis and precision therapeutics. Beyond AQP4, emerging molecular targets including surfactant protein SP-G, CHI3L1, and LAMP-1/2 may expand both diagnostic and therapeutic landscapes [9, 42, 102, 104]. Multifactorial studies incorporating microgravity simulation and environmental stressors will elucidate complex endogenous/exogenous influences on glymphatic function. Ultimately, interdisciplinary collaboration will prove pivotal in transforming fundamental discoveries into innovative clinical management strategies for neural trauma and related disorders.
We present glymphatic dysfunction as an important pathologic feature observed after TBI in post-TBI biomarkers for development of TBI precision therapies, and propose the foundations of chronotherapeutic targeting to restore glymphatic function clearance. Continued investigation regarding multimodal biomarker validation is needed along with investigation of chronotherapeutic interventions aimed at maximally enhancing glymphatic function.
TY and SY conceptualized the study, established the research framework, and coordinated the project. YY conceived the review topic, provided overall supervision and guidance, and critically revised the manuscript for important intellectual content. YM and MY contributed to the conceptualization and scope of the review, and designed the literature search and synthesis strategy. JC and XC conducted the literature review and drafted the initial manuscript. TY played a leading role in manuscript writing and was responsible for figure preparation and visualization. HS contributed to data interpretation from included studies and assisted in manuscript revision and final editing. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


