1 School of Physical Education and Sport Science, Fujian Normal University, 350117 Fuzhou, Fujian, China
2 The Center of Neuroscience and Sports, Capital University of Physical Education and Sports, 100191 Beijing, China
3 School of Kinesiology and Health, Capital University of Physical Education and Sports, 100191 Beijing, China
Abstract
Physical activity (PA) is a widely accepted non-pharmacological therapy for patients with Alzheimer’s disease (AD). Existing studies have demonstrated that PA can improve cognitive function in AD patients. However, few of the meta-analyses conducted to date have included participants with a confirmed AD diagnosis that meets standardized diagnostic criteria, nor have they systematically evaluated the interactions between different intervention parameters. The aim of this study was therefore to investigate the effects of PA on cognitive function improvement in AD patients, and how different intervention parameters may influence the effect sizes.
Two investigators independently conducted systematic searches in four international databases (PubMed, Web of Science, Embase, and Cochrane Library) and two Chinese databases (China National Knowledge Infrastructure [CNKI] and VIP Database [VIP]) while adhering to PRISMA guidelines. The search was limited to randomized clinical trials (RCTs) and covered each database from its inception to March 31, 2025. The methodological quality of included studies was assessed using criteria from the Cochrane Handbook 5.1.0. All analyses were performed using Stata 15.0.
The meta-analysis included 13 RCTs with a total of 813 AD patients. PA significantly improved Mini-Mental State Examination (MMSE) scores in AD patients (Weighted Mean Difference [WMD] = 1.79, 95% CI: 1.03 to 2.55, p < 0.001). Subgroup analyses showed that interventions with moderate intensity (WMD = 2.12), a single session duration of 30 min (WMD = 2.15), a frequency of >3 times per week (WMD = 3.03), a total weekly intervention time of >120 min (WMD = 2.10), and a total intervention duration of >12 weeks (WMD = 1.95) significantly improved MMSE scores. Meta-regression analysis revealed that intervention frequency (p < 0.001) and total intervention duration (p = 0.002) were significantly correlated with improved cognitive function, while the intervention intensity (p < 0.001) and single session duration (p = 0.002) showed negative correlations.
Our findings suggest that PA interventions can improve MMSE scores and enhance cognitive function in AD patients. We recommend that PA interventions for AD patients consist of moderate-intensity, a single session duration of 30 min, a frequency of >3 times per week, a total weekly intervention time of >120 min, and a total intervention duration of >12 weeks.
CRD420250631766. https://www.crd.york.ac.uk/PROSPERO/view/CRD420250631766.
Keywords
- physical activity
- exercise
- Alzheimer’s disease
- cognitive
- meta-analysis
Alzheimer’s disease (AD) is a progressive neurodegenerative disorder primarily affecting older adults. Its hallmark symptoms involve the gradual deterioration of multiple cognitive functions, emotional regulation, daily living activities, and social behaviors [1]. The progression of AD is irreversible, with its onset typically being insidious and difficult to detect [2]. As the disease advances, patients progressively lose cognitive faculties, often experiencing disorientation, memory decline, and the inability to perform routine activities, eventually developing severe dementia [3]. Structural brain damage and neuronal degeneration intensify during late-stage AD, leaving patients with profound cognitive and motor impairments that may become life-threatening in severe cases [4]. A study published in 2024 found that approximately 6.9 million older adults were living with AD in the United States, with over 119,000 deaths attributed to the disease in 2021 alone [5]. This trend is projected to accelerate, with estimates suggesting that more than 152 million people globally will be affected by AD by 2050 [6]. AD is a devastating condition that not only imposes substantial healthcare costs, but also lacks effective diagnostic and treatment options, placing an overwhelming economic burden on patients’ families and society at large [2, 7].
Although pharmacological intervention has always been the primary treatment approach for AD, the types of drugs available are limited and their therapeutic effects are unsatisfactory. These drugs can only prevent or slow down the onset and progression of AD [8]. Consequently, non-pharmacological therapies have attracted widespread attention as safe and relatively inexpensive interventions for maintaining cognitive function in AD patients [9, 10]. Structured and planned physical activity (PA) not only helps to prevent and reduce the risk of secondary diseases caused by declining physical function, but also slows the further deterioration of physical and cognitive abilities [11]. PA has demonstrated significant effects in improving physical health, cognitive ability, and communication skills in older adults, as well as being positively correlated with cognitive performance [12, 13]. Furthermore, regular PA may lower the risk of developing AD, serving as both a preventive measure and as a means to slow disease progression [14]. An intervention study by Yu et al. [15] involving exercise and behavior management programs for AD patients found that PA slowed the rate of cognitive decline, enhanced motor function, and improved overall cognitive performance, while simultaneously promoting better physical health. Additional research findings suggest that PA enhances brain plasticity [16] and improves brain volume [17], with these structural changes being positively associated with brain health [18, 19]. Moderate PA contributes to symptom alleviation, facilitates functional recovery, and supports cognitive improvement, thus making it a crucial strategy for maintaining and enhancing brain function [20]. PA is practically feasible and remarkably effective at reducing the loss of functional independence in patients and delaying the common complications of AD [21].
While numerous studies have suggested that PA may enhance cognitive performance in AD patients, there is significant heterogeneity and uncertainty in the research findings due to variations in study design, sample size, and differences in PA intervention protocols [22, 23, 24]. Previous meta-analyses also have significant limitations. First, some of the included participants did not strictly adhere to the standardized diagnostic criteria for AD. Second, the heterogeneity issue was only addressed through subgroup analyses, and the underlying moderating factors were not explored in depth. Third, although some studies evaluated the effects of intervention parameters, potential interactions between these parameters were not examined. Therefore, the aim of this study was to rigorously evaluate the effect of PA interventions on cognitive function in AD patients by investigating the optimal values for individual intervention parameters (e.g., intensity, frequency), and interaction effects between parameters to clarify their influence on rehabilitation outcomes. Based on current evidence, we hypothesize that: (1) PA can significantly improve the Mini-Mental State Examination (MMSE) scores and enhance cognitive function in AD patients; and (2) significant interaction effects exist among various PA intervention parameters.
Following PRISMA guidelines [25], two investigators (Shengyu Dai [SD] and Shunling Yuan [SY]) independently conducted systematic searches in four international databases (PubMed, Web of Science, Embase, and Cochrane Library) and two Chinese databases (China National Knowledge Infrastructure [CNKI, https://www.cnki.net/] and VIP Database [VIP, https://qikan.cqvip.com/]). The search was limited to randomized controlled trials (RCTs), and each database was searched from inception to March 31, 2025. The systematic literature search employed Medical Subject Headings (MeSH) to identify core search terms. MeSH terms including “Exercise”, “Alzheimer’s Disease”, and “Cognitive Function” were selected and a comprehensive search strategy was implemented that combined both MeSH subject terms and free words for term retrieval (see Supplementary Table 1). To ensure comprehensive coverage of the literature, we also supplemented this search strategy by manually screening reference lists of identified meta-analyses and published reports to identify additional studies relevant to the topic. The review protocol was registered with the International Prospective Register of Systematic Reviews (PROSPERO), registration number CRD420250631766.
Inclusion criteria: (1) Population: patients with a confirmed diagnosis of AD meeting standardized diagnostic criteria; (2) Intervention: the intervention group received PA interventions (e.g., aerobic exercise, cycling, or walking) without restriction on the type of activity; (3) Control: the control group received conventional rehabilitation, or no intervention; (4) Outcomes: cognitive impairment was assessed using MMSE scores; and (5) Study design: only RCTs were included.
Exclusion criteria: (1) Patients without AD; (2) The intervention group received multimodal therapeutic interventions (e.g., cognitive-behavioral interventions); (3) Non-RCTs; (4) Review articles or meta-analyses; (5) Low-quality studies or duplicate publications; and (6) Studies with unavailable full-text or unextractable data.
Two researchers independently screened article titles and abstracts, with data extraction conducted using standardized forms. In the event of a discrepancy, the two researchers discussed or consulted with a third investigator to facilitate resolution. The following data were extracted: title, first author, publication date, country (region) of the study, age of participants, number of participants, diagnostic criteria, duration of disease, specific intervention measures, and outcome measures.
The risk of bias assessment method recommended in the Cochrane Handbook 5.1.0 [26] was strictly adhered to by both independent researchers when evaluating each included study. Assessment items included random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, handling of incomplete outcome data, selective reporting, and other potential biases. Each item was classified as low-risk, high-risk, or unclear-risk. In case of disagreement between the two researchers, a third investigator was consulted to arbitrate and reach a consensus. This independent dual-review with third-party arbitration ensured the objectivity of assessment.
All analyses were performed using Stata software (Version 15.0, StataCorp LLC,
College Station, TX, USA; Official website: https://www.stata.com/). Since the
scoring scales used in the combined trials (such as MMSE) were consistent, we
employed weighted mean difference (WMD) and 95% confidence intervals (CI) to
analyze continuous variable data. A fixed-effects model was applied when an
I2 value of
We systematically searched six databases, retrieving 5691 studies. After initial screening and removal of duplicates, 3727 articles remained. Of these, 3583 were excluded following assessment of their relevance based on the title and abstract. Next, the screening process strictly followed the predefined inclusion and exclusion criteria, with 131 studies undergoing full-text review and evaluation. This resulted in 118 articles being excluded for the following reasons: inconsistency in experimental design (n = 36), no available data (n = 39), non-RCT (n = 25), and systematic review (n = 18). Eventually, 13 RCTs were included in the final analysis (Fig. 1).
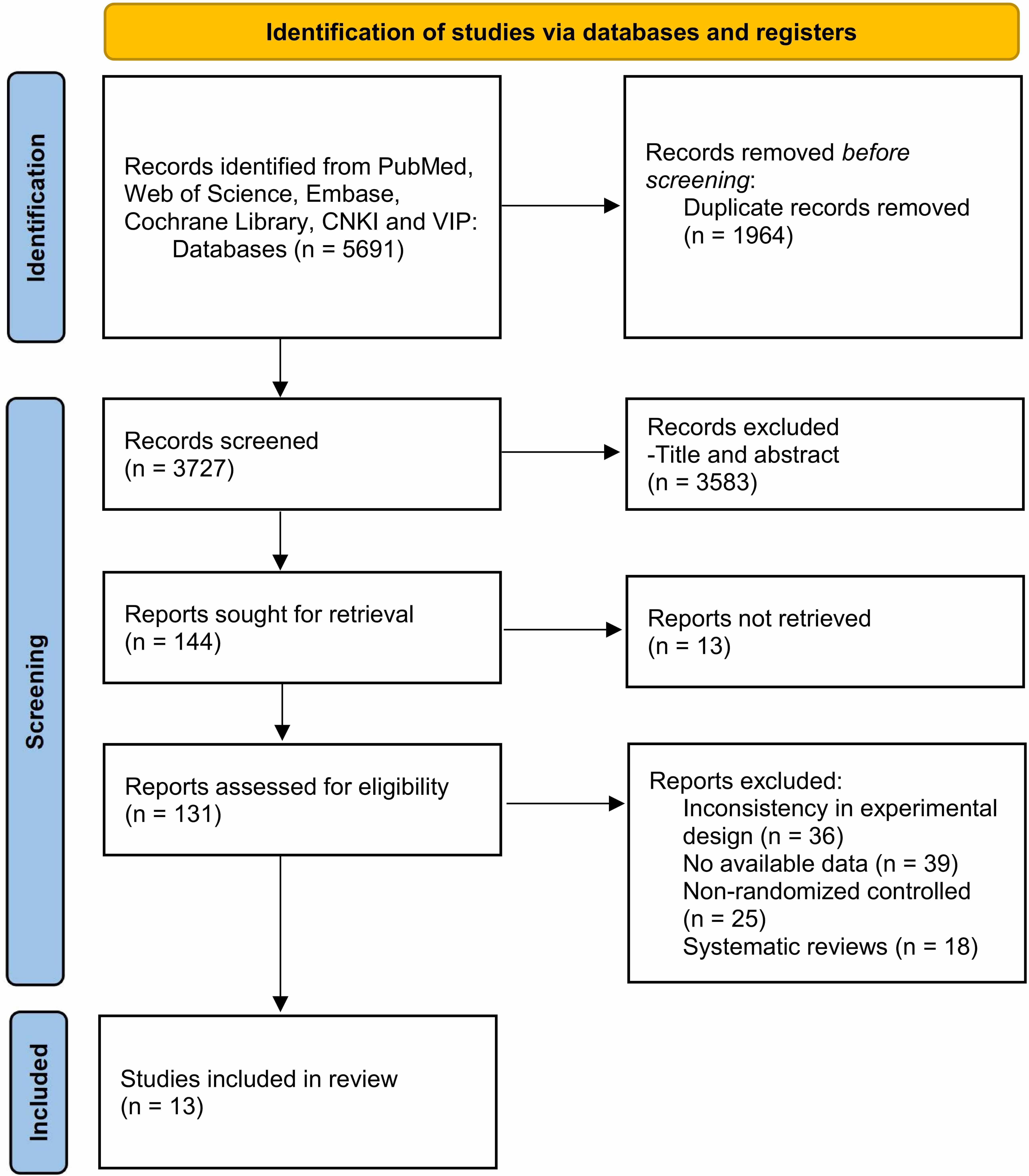 Fig. 1.
Fig. 1.
Flowchart of the literature screening process. CNKI, China National Knowledge Infrastructure; VIP, VIP Database.
Following the rigorous screening process, 13 RCTs [28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40] published between 2010 and 2024 and including a total of 813 AD patients were identified (Table 1). Among these, seven were conducted in China, three in Brazil, and one each in Germany, Denmark, and France. All studies reported baseline characteristics including age, sample size, and diagnostic criteria. However, six studies failed to document disease duration. The intervention duration ranged from 4 to 48 weeks across studies. Primary outcomes were assessed using the MMSE.
| First Author | Year | Country | Age (IG/CG) | Cases (IG/CG) | Diagnostic criteria | Duration of disease (year) | Dosage of intervention | Intervention details (IG/CG) | |
| Hernandez [28] | 2010 | Brazil | 77.70 |
9/7 | DSM-IV | 2.50 |
24 weeks | (1) Intensity: 60–80% of HRmax | Routine medical treatment |
| 84.00 |
3.50 |
3 days/week | (2) Motor development components: Stretching activities, weight training, circuits, pre-sport games, dance sequences, recreational activities and relaxation | ||||||
| 60 min | |||||||||
| Arcoverde [29] | 2014 | Brazil | 78.5 (64–81.2)/ | 10/10 | NINCDS-ADRDA/ | 4.3 |
16 weeks | Treadmill exercise: | Routine medical treatment |
| 79 (74.70–82.20) | NINDS-AIREN | 4.10 |
2 days/week | (1) Weeks 1–4: Adaptation period | |||||
| 30 min | (2) Weeks 5–16: 10-min warm-up, 20-min exercise at 60% VO2max, and 5-min stretching exercises | ||||||||
| Wang [30] | 2014 | China | 71.19 |
26/28 | DSM-IV/ | NA | 12 weeks | Cycle ergometer training: | Usual daily activities |
| 70.04 |
NINCDS-ADRDA | 3 days/week | (1) Intensity: Moderate (70% of HRmax) | ||||||
| 40 min | (2) 5-min warm-up, 30-min target-intensity exercise, and 5-min cool-down | ||||||||
| Hu [31] | 2014 | China | 63.26 |
50/50 | NINCDS-ADRDA | 3.96 |
48 weeks | Table tennis exercise: | Routine medical treatment |
| 60.45 |
4.67 |
3–4 days/week | (20-min play, and 10-min rest) | ||||||
| 60 min | |||||||||
| Holthoff [32] | 2015 | Germany | 72.40 |
15/15 | NINCDS-ADRDA | 4.13 |
12 weeks | Lower-body training: | Usual treatment |
| 70.67 |
2.80 |
3 days/week | Alternating modes (passive, motor-assisted, or active resistive) and directions (forward and reverse) every 5 min | ||||||
| 30 min | |||||||||
| Yan [33] | 2015 | China | 71.50 |
18/18 | DSM-IV/ | 2.80 |
24 weeks | Cycle ergometer training (two sessions): | No intervention |
| 70.60 |
NINCDS-ADRDA | 3.10 |
2 days/week | (1) Intensity: Moderate (70% of HRmax) | |||||
| 30 min | (2) 5-min warm-up, 20-min target-intensity exercise, and 5-min cool-down | ||||||||
| Qi [34] | 2015 | China | 71.80 |
8/7 | DSM-IV | NA | 12 weeks | Cycle ergometer training: | Routine medical treatment |
| 76.40 |
3 days/week | (1) Intensity: Moderate (70% of HRmax) | |||||||
| 40 min | (2) 5-min warm-up, 30-min target-intensity exercise, and 5-min cool-down | ||||||||
| Hoffmann [35] | 2016 | Denmark | 69.80 |
107/93 | NINCDS-ADRDA | NA | 16 weeks | (1) Weeks 1–4: Adaptation period (lower extremity exercise and aerobic exercise) | Usual treatment |
| 71.30 |
3 days/week | ||||||||
| 60 min | (2) Weeks 5–16: Intensity: 70–80% of HRmax; Aerobic exercise: 3 × 10-min sessions (ergometer bike, cross trainer, and treadmill) | ||||||||
| Mu [36] | 2016 | China | 72.9 |
39/39 | NINCDS-ADRDA | 3.21 |
16 weeks | Aerobic exercise: | No intervention |
| 73.69 |
3.28 |
Brisk walking | |||||||
| 60 min | |||||||||
| de Souto Barreto [37] | 2017 | France | 88.3 |
44/47 | DSM-IV | NA | 24 weeks | (1) Intensity: Moderate | Social Activity: |
| 86.9 |
2 days/week | (2) 10-min warm-up, 10-min coordination and balance exercises, 10–15-min muscle strengthening, 20-min aerobic exercise, 5–10-min cool-down | Therapeutic music mediation or arts and crafts | ||||||
| 60 min | |||||||||
| Liu [38] | 2017 | China | 70.9 |
24/24 | DSM-IV | 3.2 |
12 weeks | Aerobic calisthenics: | Usual daily activities |
| 70.3 |
3.1 |
3 days/week | (1) Intensity: Moderate (70% of HRmax) | ||||||
| 40 min | (2) 5-min warm-up, 30-min target-intensity exercise, and 5-min cool-down | ||||||||
| de Oliveira Silva [39] | 2019 | Brazil | 81.22 |
13/14 | DSM-IV | NA | 12 weeks | (1) Intensity: 70% of VO₂max or 80% of HRmax | No intervention |
| 77.54 |
2 days/week | (2) 5-min balance training, 30-min aerobic exercise (5-min warm-up, 20-min at target intensity, and 5-min cool-down), 20-min strength training, and 5-min stretching | |||||||
| 60 min | |||||||||
| Wei [40] | 2024 | China | 73.09 |
48/50 | ICD-10 | NA | 4 weeks | Aerobic Baduanjin | Usual treatment |
| 72.39 |
7 days/week | ||||||||
| 30 min | |||||||||
IG, intervention group; CG, control group; NA, not available; DSM-IV, Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition; NINCDS-ADRDA, National Institute of Neurological and Communicative Disorders and Stroke-Alzheimer’s Disease and Related Disorders Association; NINDS-AIREN, National Institute of Neurological Disorders and Stroke-Association Internationale pour la Recherche et l’Enseignement en Neurosciences; ICD-10, International Classification of Diseases, Tenth Revision; HRmax, Maximum Heart Rate; VO₂max, Maximum Oxygen Consumption/Uptake.
Two researchers independently evaluated the methodological quality of the included studies using the Cochrane Handbook 5.1.0 criteria [26], with any discrepancies resolved through third-party arbitration. The majority of the 13 RCTs were of moderate methodological quality (Fig. 2). However, the proportion of “unclear” ratings for allocation concealment and participant/researcher blinding was relatively high. These ratings stemmed from inadequate reporting in the original studies. Attempts were made to contact the authors of studies with missing key information, but no responses were received. To avoid speculative judgments, these items remained categorized as “unclear”.
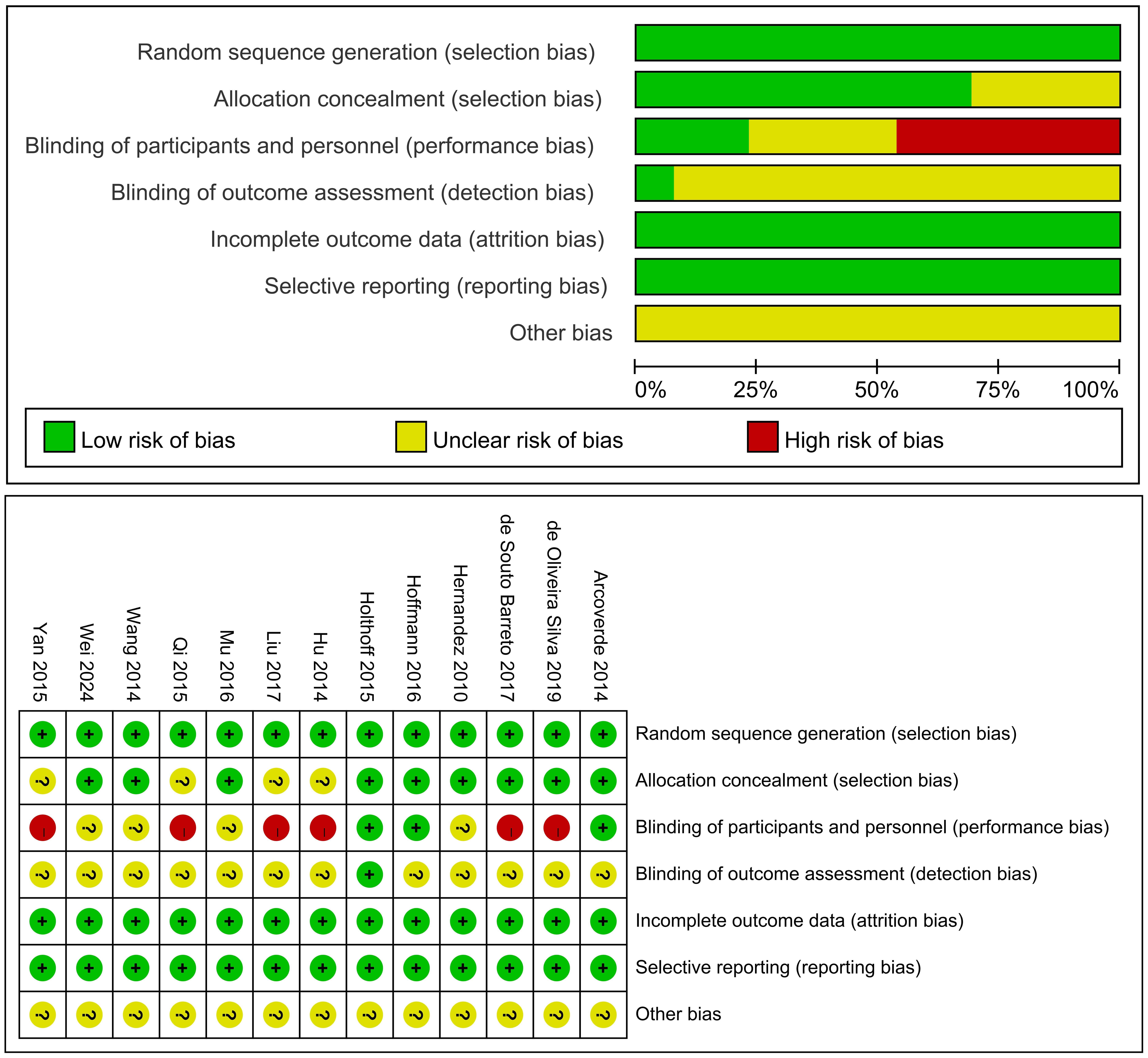 Fig. 2.
Fig. 2.
Schematic diagram of risk bias. “?”, unclear-risk; “+”, low-risk; “-”, high-risk.
All 13 articles used the MMSE to evaluate the cognitive function of research
subjects (Fig. 3). Compared with the control group, PA had a positive effect on
the MMSE scores of AD patients (WMD = 1.79, 95% CI: 1.03 to 2.55, p
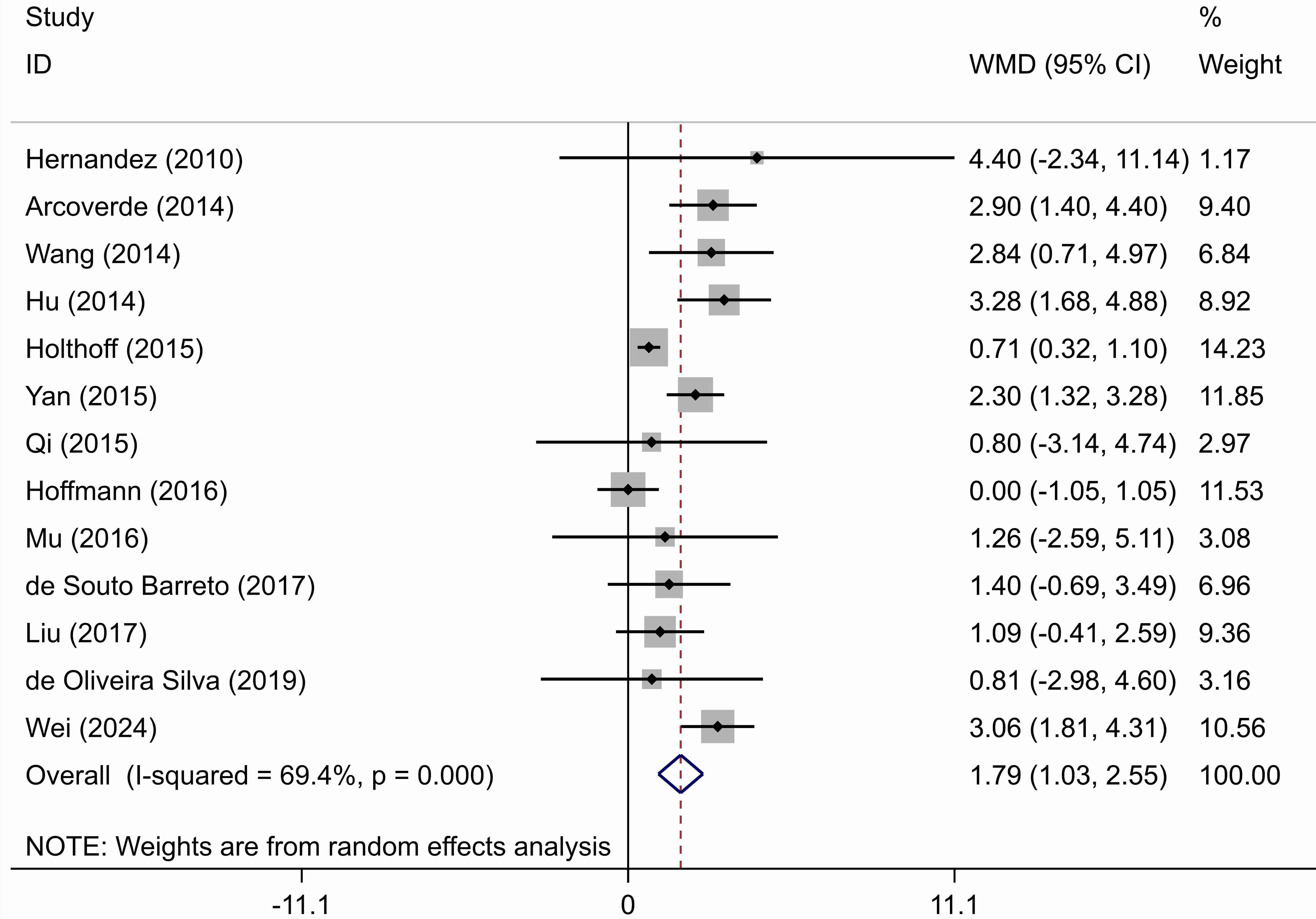 Fig. 3.
Fig. 3.
Forest plot showing the effect of physical activity on cognitive function (MMSE scores) in AD patients. WMD, Weighted Mean Difference; MMSE, Mini-Mental State Examination; AD, Alzheimer’s disease; CI, confidence interval.
Due to the high heterogeneity observed among the included studies, we conducted subgroup analyses stratified by: (1) country; (2) intervention intensity; (3) single session duration; (4) intervention frequency; (5) total weekly intervention time; and (6) total intervention duration. The results for this subgroup analysis are shown in Table 2.
| Subgroup | Number of Studies | Heterogeneity Test | Results of Meta-Analysis | |||
| I2(%) | p-value | WMD (95% CI) | Z-value | p-value | ||
| Country | ||||||
| China | 7 | 5.1 | 0.388 | 2.40 (1.78, 3.02) | 7.60 | |
| Other than China | 6 | 56.2 | 0.044 | 1.10 (0.18, 2.03) | 2.35 | 0.019 |
| Intervention intensity | ||||||
| Moderate | 6 | 0.0 | 0.507 | 2.12 (1.48, 2.76) | 6.48 | |
| Other than moderate | 7 | 75.8 | 1.62 (0.45, 2.78) | 2.71 | 0.007 | |
| Single session duration | ||||||
| 30 min | 4 | 87.7 | 2.15 (0.81, 3.49) | 3.14 | 0.002 | |
| 40 min | 3 | 0.0 | 0.386 | 1.60 (0.42, 2.77) | 2.67 | 0.008 |
| 60 min | 6 | 59.6 | 0.030 | 1.51 (–0.01, 3.03) | 1.94 | 0.052 |
| Intervention frequency | ||||||
| 2 times weekly | 4 | 0.0 | 0.590 | 2.28 (1.53, 3.03) | 5.96 | |
| 3 times weekly | 6 | 28.5 | 0.221 | 0.81 (0.15, 1.46) | 2.42 | 0.015 |
| 3 | 0.0 | 0.636 | 3.03 (2.07, 3.98) | 6.22 | ||
| Total weekly intervention time | ||||||
| 3 | 86.7 | 0.001 | 1.85 (0.42, 3.28) | 2.54 | 0.011 | |
| 120 min | 5 | 0.0 | 0.724 | 1.50 (0.51, 2.49) | 2.97 | 0.003 |
| 5 | 79.0 | 0.001 | 2.10 (0.23, 3.96) | 2.20 | 0.028 | |
| Total intervention duration | ||||||
| 6 | 67.7 | 0.008 | 1.64 (0.51, 2.76) | 2.84 | 0.004 | |
| 7 | 67.5 | 0.005 | 1.95 (0.80, 3.11) | 3.32 | 0.001 | |
First, by country:
Studies conducted in China showed significantly greater improvement in MMSE scores (WMD = 2.40, 95% CI: 1.78 to 3.02; I2 = 5.1%, p = 0.388) than those from outside China (WMD = 1.10, 95% CI: 0.18 to 2.03; I2 = 56.2%, p = 0.044) (Supplementary Fig. 1). Treatment response therefore appears to vary by region. Although the small sample size makes it difficult to draw definitive conclusions, this trend is noteworthy.
Second, by intervention intensity:
Moderate intensity intervention produced superior cognitive improvement (WMD =
2.12, 95% CI: 1.48 to 2.76; I2 = 0%, p = 0.507) compared to
non-moderate intensity (WMD = 1.62, 95% CI: 0.45 to 2.78; I2 = 75.8%,
p
Third, by single session duration:
Interventions conducted for 30 min per session (WMD = 2.15, 95% CI: 0.81 to
3.49; I2 = 87.7%, p
Fourth, by intervention frequency:
The significant reduction in heterogeneity within each subgroup after
stratification suggests that intervention frequency is a source of heterogeneity.
Interventions conducted
Fifth, by total weekly intervention time:
Interventions conducted for
Sixth, by total intervention duration:
Interventions conducted for an overall duration of
Meta-regression analysis was conducted on each variable (country, intervention intensity, single session duration, intervention frequency, total weekly intervention time, and total intervention duration) in order to assess its influence on effect size (Table 3).
| Variable | Coef. | Std. Err. | z | p |
95% CI |
| Country | 1.621 | 0.349 | 4.64 | (0.936, 2.306) | |
| Intervention intensity | –0.337 | 0.760 | –0.44 | 0.657 | (–1.828, 1.153) |
| Single session duration | –0.021 | 0.028 | –0.75 | 0.456 | (–0.076, 0.034) |
| Intervention frequency | 0.271 | 0.532 | 0.51 | 0.609 | (–0.770, 1.314) |
| Total weekly intervention time | 0.009 | 0.046 | 0.19 | 0.846 | (–0.081, 0.099) |
| Total intervention duration | 0.304 | 0.777 | 0.39 | 0.695 | (–1.219, 1.828) |
Coef, coefficient; Std. Err., standard error.
The results showed that the country of study, as a covariate, had a
statistically significant effect on MMSE scores (p
Univariate analysis showed no significant correlation for intervention frequency
(coefficient = 0.271, p = 0.609). However, multivariate meta-regression
analysis revealed significant positive associations for intervention frequency
(coefficient = 2.06, p
Egger’s test found no significant publication bias (t = 1.94, p = 0.078; Fig. 4, Supplementary Table 4). Similarly, Begg’s test also revealed no significant bias (Z = 0.18, p = 0.855; Fig. 5), indicating the reliability of the data.
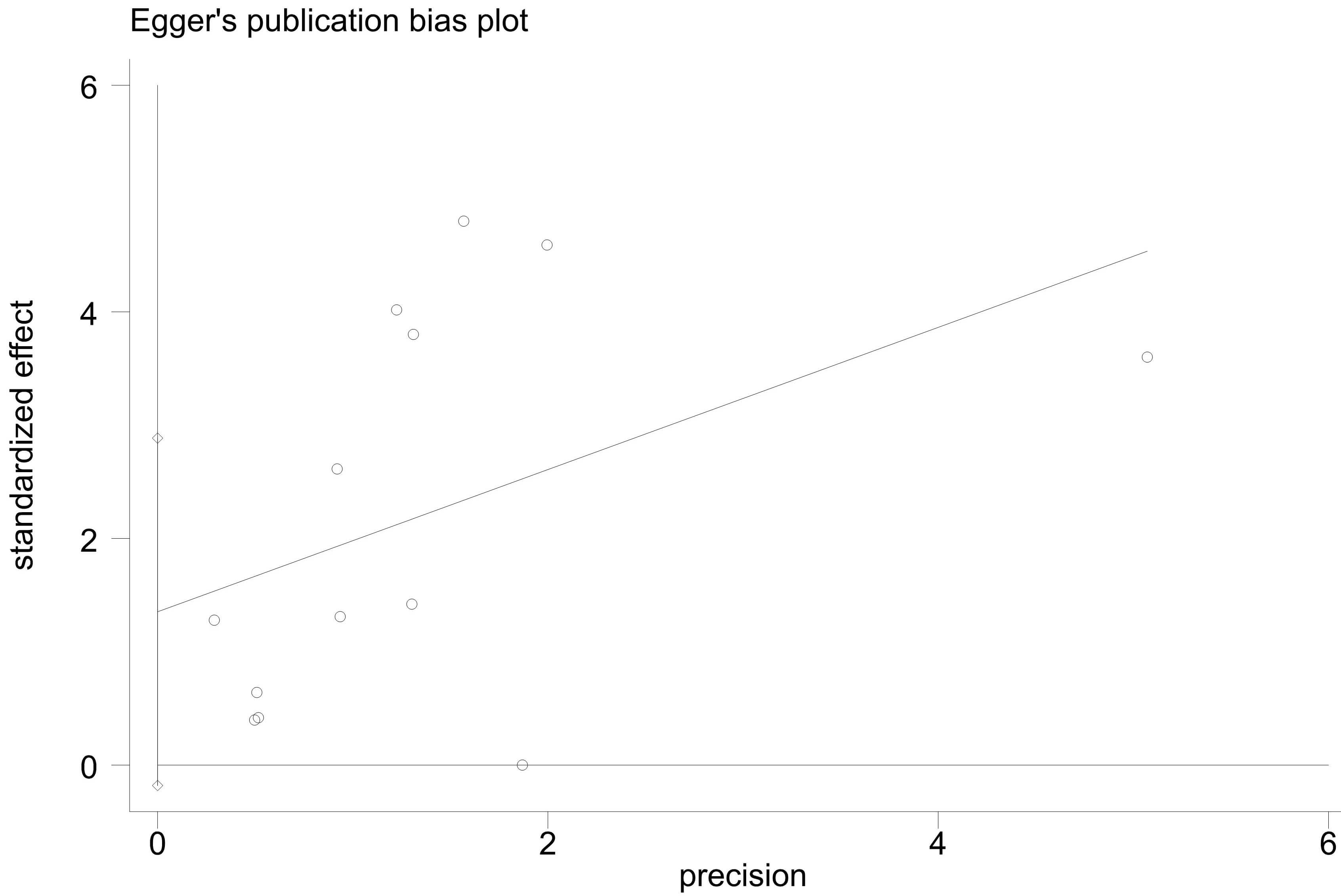 Fig. 4.
Fig. 4.
Egger’s publication bias plot of physical activity on the cognitive function of AD patients.
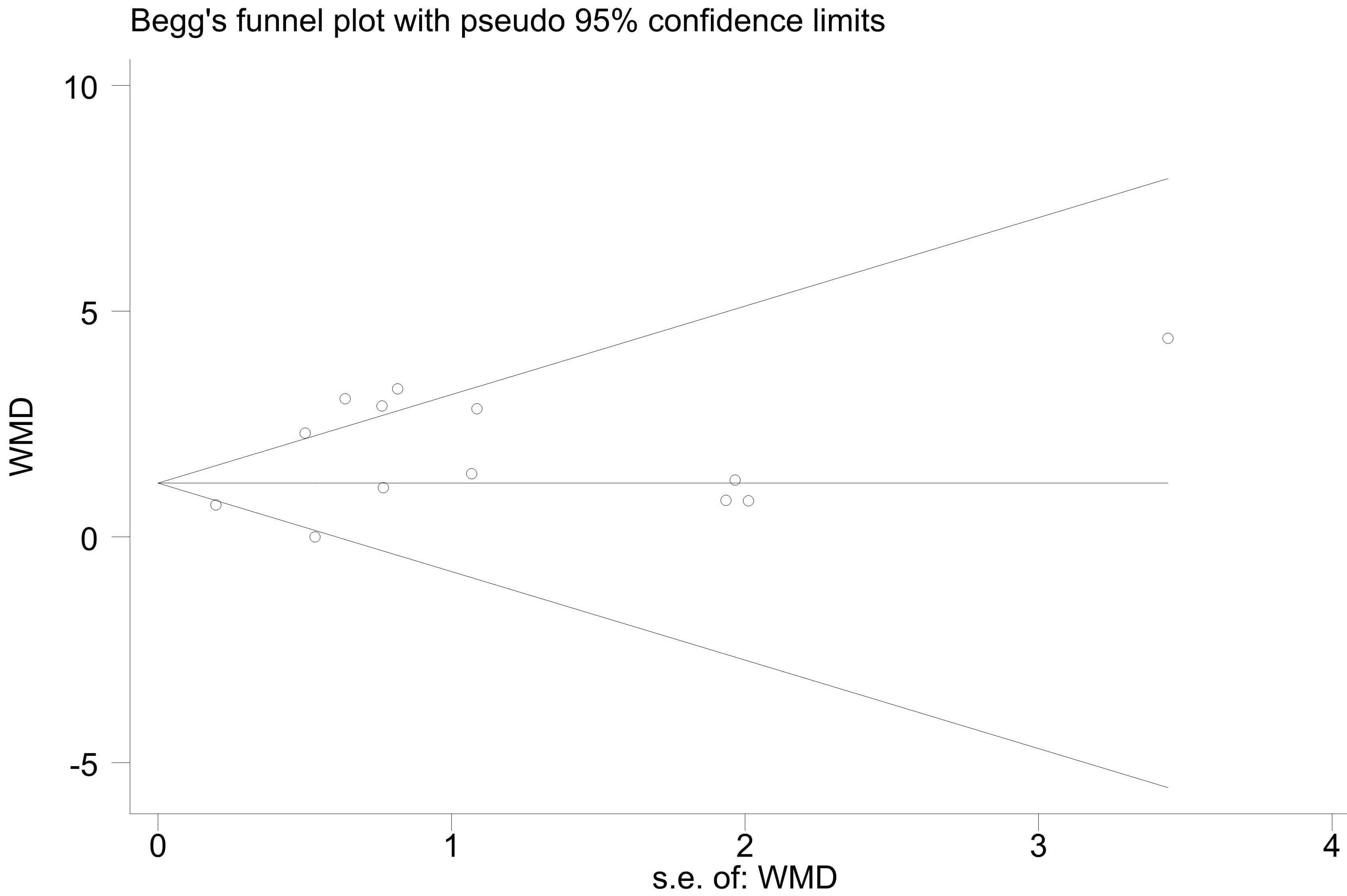 Fig. 5.
Fig. 5.
Begg’s publication bias plot of physical activity on the cognitive function of AD patients.
Through systematic screening of the literature, we identified 13 RCTs that
assessed cognitive function using the MMSE. Analysis of the results from these
studies revealed a significant improvement in MMSE scores among the PA
intervention group, indicating that it can effectively enhance cognitive function
in AD patients. Subgroup and meta-regression analyses revealed that sources of
heterogeneity were the country in which the study was performed, and the
intervention frequency. Specifically, the subgroup analysis showed that MMSE
scores were significantly improved by interventions with moderate intensity,
interventions with a single session duration of 30 min, an intervention frequency
of
This study specifically examined the effects of PA interventions on cognitive function in patients with a clinical diagnosis of AD. Our findings showed that PA significantly improves cognitive function in AD patients, in line with the results of previous meta-analyses. Du et al. [41] conducted a meta-analysis of cognitive function in 869 patients diagnosed with AD. Their results also showed a statistically significant improvement in cognitive function after PA interventions. However, five of the included studies in their meta-analysis did not show beneficial effects. Jia et al. [27] conducted a meta-analysis of 13 RCTs involving 673 subjects diagnosed with AD. As in the present study, these authors used subgroup analysis to explore a possible dose-response relationship between PA interventions and improvement in cognitive function. Based on the MMSE scores, they found a statistically significant cognitive enhancement in the intervention group, with different doses of PA producing different effects. A systematic meta-analysis by Yang et al. [42] demonstrated that aerobic exercise significantly improved cognitive function in AD patients. Subgroup analyses revealed that patients showed significant improvements in both MMSE and Alzheimer’s Disease Assessment Scale-Cognitive Subscale (ADAS-cog) scores when the total intervention duration exceeded 16 weeks.
These studies suggest that PA has a positive effect on cognitive function. However, most previous studies did not apply internationally recognized diagnostic criteria (e.g., National Institute of Neurological and Communicative Disorders and Stroke-Alzheimer’s Disease and Related Disorders Association (NINCDS-ADRDA), Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV), or International Classification of Diseases, Tenth Revision (ICD-10)) when enrolling AD patients. This inconsistency in diagnostic standard can increase the heterogeneity of study outcomes, potentially compromising accurate assessment of the intervention effect. Moreover, previous meta-analyses typically relied only on subgroup analysis to explore potential influencing factors when heterogeneity was detected, without further investigating the specific effect of these factors on effect sizes. In contrast, the current study only enrolled patients who met established clinical diagnostic criteria for AD, thereby ensuring homogeneity of the study population. In addition to subgroup analysis, we also employed meta-regression to systematically evaluate the influence of intervention parameters on effect sizes. As a result, our study effectively controlled for bias arising from inconsistent diagnostic criteria, providing more targeted evidence for PA interventions in AD patients.
Using subgroup analysis, we systematically evaluated the effects of the following factors on cognitive function in AD patients: country of the study, intervention intensity, single session duration, intervention frequency, total weekly intervention time, and total intervention duration. This analysis revealed subtle differences in treatment effects between studies conducted in China and those in other countries, in line with previous findings by Jia et al. [27] and Yang et al. [42]. However, unlike their findings, the results of our meta-regression analysis revealed that differences in the country of study had a significant impact on intervention effects. In the five sets of bivariate meta-regression analyses, the country of study variable consistently maintained significant correlations, indicating that national differences are an important factor influencing intervention outcomes. In particular, differences between countries were most significant after adjusting for the intensity of intervention, suggesting this may be the most prominent regulatory factor responsible for the observed differences in treatment effects. Although we adjusted for several key variables in the intervention protocol, most studies did not explicitly report differences in environmental factors (e.g., level of medical infrastructure, cultural acceptance) and demographic characteristics (e.g., dietary habits, ethnicity/race), and consequently our findings may still be affected by residual confounding factors. Future research should aim to systematically collect relevant background data to further explore the association between potential environmental/demographic factors and the intervention effects.
With regard to the intervention intensity and based on the American College of Sports Medicine (ACSM) guidelines, moderate intensity was defined as 64–76% of Maximum Heart Rate (HRmax) [43]. The current results showed the intervention effect for the moderate intensity group was better and more consistent (i.e., without heterogeneity) compared to the other intensities. In contrast to previous meta-analyses by Yang et al. [42] and Zhang et al. [44], our study is the first to incorporate classification by intervention intensity. Although the results were statistically significant, certain differences were apparent in the definition of intervention intensity among the different studies. Some studies used 60–80% of HRmax as the standard [28, 35, 39], while others did not clearly report the intensity [31, 32, 36, 40]. This inconsistent methodology may be the main reason for the high heterogeneity observed in the non-moderate intensity group in our analysis. Differences in the classification of moderate intensity were also noted. Most studies used percentage of HRmax as the evaluation criterion [30, 33, 34, 38], while others employed VO2max [29], and some described the intensity as “moderate” without precise quantification [37]. Therefore, a standardized intensity assessment system that utilizes objective indicators is necessary to determine the optimal intensity threshold for improving cognitive function in AD patients.
Our study found that interventions with a single session duration of 30 min and
a frequency of
Our subgroup analysis revealed that a total weekly intervention time of
Our study also found that a total intervention duration of
Of note, most covariates in our study were found to be non-significant in univariate models, but significant in the multivariate model. This discrepancy is likely to stem from the interconnectedness of intervention parameters. The multivariate model revealed that intervention frequency and total intervention duration were independent predictors of improved cognitive function, while intensity and single session duration exert independent negative effects. These associations were masked in univariate analyses, probably due to their interaction with other parameters, highlighting that PA interventions in AD patients require comprehensive consideration of multiple interacting parameters.
A dose-response relationship exists between PA and health benefits. An appropriate exercise load can not only mitigate the degradation of physiological function caused by aging, but also reduce the risk of various chronic diseases [53]. Another study found an inverted U-shaped relationship between PA and cognitive function [54]. PA is a crucial stabilizing factor that can slow the decline of cognitive functions [55]. A large number of studies have shown that PA can enhance cognitive function in elderly patients with AD [28, 29, 56]. A study by Machado et al. [55] confirmed that regular PA can enhance neural function in the brain. Tyndall et al. [57] found that long-term PA significantly reduces the risk of developing AD and exerts positive regulatory effects on related blood biomarkers. A systematic analysis concluded that PA had moderate positive effects on neurocognitive functions, such as improving attention, accelerating the speed of information processing, enhancing executive function, and improving memory [58]. Finally, a systematic review by Li et al. [59] also supports the efficacy of PA.
A study in transgenic AD mice demonstrated that PA can reduce extracellular
A
Several mechanisms might explain the positive effect of PA on cognitive function
in patients with AD: (1) PA can stimulate the release of calcium-metabolizing
hormones, thereby increasing the calcium ion level in the blood. This
subsequently promotes dopamine synthesis in the brain, while increasing the
density of acetylcholine receptor. Ultimately, elevated dopamine levels can
regulate the function of the cholinergic system, thus contributing to the
improvement of cognitive function [63, 64]. (2) Cardiovascular-related risk
factors are closely associated with cognitive decline. PA can improve
cardiovascular function and alleviate inflammatory response and oxidative stress,
thus exerting a protective effect on neurons. Concurrently, PA can enhance and
redistribute cerebral blood flow, accelerate the degradation of
This research has several strengths. First, it only included RCTs, thereby improving the quality of the study and the reliability of the results. Second, the study population was limited to AD patients who met standardized diagnostic criteria, ensuring consistency in patient disease characteristics. Third, a multi-subgroup design was employed to explore the reasons for the high heterogeneity and for the effects between PA and cognitive function in different AD patient subgroups. Finally, our study used multivariate regression analysis to quantify significant interactions between intervention parameters (frequency, intensity, session duration, and total intervention duration). However, this study also has several limitations. Firstly, it was limited to the English and Chinese language literature. Second, the included studies did not use blinding of participants during the intervention process, and hence there may have been some bias in the study design. Third, the lack of consistency in the intervention protocols and the use of different criteria across studies may have influenced the final results and increased the heterogeneity of studies. Fourth, the possible effects of potential confounding factors could not be controlled. For example, some studies did not report both disease severity and disease duration. Fifth, included studies used varying diagnostic criteria (DSM-IV, NINCDS-ADRDA, ICD-10). Because only one study used ICD-10 and the others used overlapping criteria, it was not feasible to quantify this effect using robust subgroup or sensitivity analyses. Future research should adopt a unified, standardized criterion to reduce such heterogeneity. Sixth, some subgroup analyses included only three studies. This small sample size reduces the statistical power, thereby affecting the reliability of effect size estimates. Finally, we have noted that meta-regression is an ecological analysis, and individual-level confounding cannot be excluded. Additionally, adherence to interventions and differences in implementation across studies may introduce heterogeneity that is difficult to control for in meta-analysis.
Our findings suggest that PA interventions can improve MMSE scores and enhance
cognitive function in AD patients. We recommend that interventions for AD
patients consist of moderate-intensity PA, a single session duration of 30 min, a
frequency of
PA, physical activity; AD, Alzheimer’s disease; RCT, randomized controlled trials; MMSE, Mini-Mental State Examination; WMD, weighted mean difference; 95% CI, 95% Confidence Intervals; IG, intervention group; CG, control group; NA, Not available; DSM-IV, Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition; NINCDS-ADRDA, National Institute of Neurological and Communicative Disorders and Stroke–Alzheimer’s Disease and Related Disorders Association; NINDS-AIREN, National Institute of Neurological Disorders and Stroke–Association Internationale pour la Recherche et l’Enseignement en Neurosciences; ICD-10, International Classification of Diseases, Tenth Revision; HRmax, Maximum Heart Rate; VO2max, Maximum Oxygen Consumption/Uptake; Coef, coefficient; Std. Err., standard error; ACSM, American College of Sports Medicine; BDNF, brain-derived neurotrophic factor levels.
All data are available in the main text or as a supplementary file. Additional requests can be made to the corresponding author upon reasonable request.
SD, SY, and XBZ conceptualized and designed the study. CJ provided administrative support. SY and XBZ were responsible for document retrieval. SD, SY, and XKZ were in charge of the collection and assembly of data. SD, XKZ, and CJ analyzed and interpreted the data. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This study receives the support from the National Natural Science Foundation of China [grant number: 32371132].
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/JIN42702.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





