1 Scientific Research Institute of Neuroscience and Medicine, 630117 Novosibirsk, Russia
2 Novosibirsk State University, 630090 Novosibirsk, Russia
3 School of Pharmacy, Southwest University, 400715 Chongqing, China
4 Almazov Medical Research Center, Ministry of Healthcare of Russian Federation, 197341 St. Petersburg, Russia
5 Institute of Translational Biomedicine, St. Petersburg State University, 199034 St. Petersburg, Russia
6 West Caspian University, AZ1001 Baku, Azerbaijan
7 Bioscience Institute, University of Passo Fundo (UPF), 99042 Passo Fundo, RS, Brazil
8 The International Zebrafish Neuroscience Research Consortium (ZNRC), New Orleans, LA 70458, USA
9 The International Stress and Behavior Society (ISBS), New Orleans, LA 70458, USA
10 Graduate Program in Health Sciences, Federal University of Health Sciences of Porto Alegre, 90050 Porto Alegre, Brazil
11 Department of Pharmacology, Oxford University, OX1 3QT Oxford, UK
12 Laboratory of Psychiatric Neurobiology, Institute of Molecular Medicine, Sechenov First Moscow State Medical University, 119991 Moscow, Russia
13 Institute of General Pathology and Pathophysiology, 125315 Moscow, Russia
14 Department of Pharmacology & Toxicology, University of Toronto, Toronto, ON M5S 1A1, Canada
15 Department of Physiology and Biophysics, SUNY, University of Buffalo, Buffalo, NY 14260, USA
16 Federal Research Center Institute of Cytology and Genetics SB RAS, 630090 Novosibirsk, Russia
17 Department of Biosciences and Bioinformatics, School of Science, Xi’an Jiaotong-Liverpool University, 215000 Suzhou, Jiangsu, China
18 Suzhou Municipal Key Laboratory of Neurobiology and Cell Signaling, School of Science, Xi’an Jiaotong-Liverpool University, 215000 Suzhou, Jiangsu, China
Abstract
Encoding a key ‘hub’ scaffolding protein, the ‘Disrupted-In-Schizophrenia-1’ (DISC1) gene has been strongly implicated in brain development and functions. Genetic variance in this gene is associated with major neuropsychiatric disorders, including schizophrenia, bipolar disorder, and major depression. DISC1 is abundantly expressed in the brain of humans and various model organisms. Here, we discuss currently available animal models of DISC1-related brain deficits and their clinical relevance. We focus on evolutionarily conserved (shared) mechanisms and species-specific phenotypes, especially in newly developed zebrafish (Danio rerio) models, to better understand the uniquely complex role of DISC1 in the molecular pathogenesis of neurobehavioral abnormalities relevant to human neuropsychiatric disorders.
Keywords
- DISC1
- zebrafish
- neurogenesis
- animal models
- behavior
- neuropsychiatric disorders
‘Disrupted-In-Schizophrenia-1’ (DISC1) is a critical, evolutionarily conserved central nervous system (CNS) gene [1] encoding a key cellular protein that regulates neurodevelopment, including axonal guidance [2, 3, 4], synaptic plasticity [5, 6, 7], as well as neural precursor proliferation, migration, and maturation [8, 9]. This gene also modulates the functioning of dopamine receptors [10] and the glutamatergic system [11] via direct interactions of DISC1 with the dopamine D2 receptor (D2R) or N-methyl-D-aspartate receptor subunit 1 (NR1) (Table 1, Ref. [2, 3, 4, 5, 8, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59]). The DISC1 protein is crucial for cytoskeletal processes, as mutations of its gene induce aberrant synaptic pruning and dendritic atrophy [5, 60]. Since its discovery, DISC1 has been extensively investigated, especially in the last two decades, yet has recently experienced a notable decline in interest (Fig. 1, Ref. [61, 62]).
| Binding protein/complex | Function of interaction | References |
|---|---|---|
| Glycogen synthase kinase 3 (GSK3) | GSK3 inhibition through the Wnt/ |
[3, 4, 8, 12, 13, 14] |
| Dopamine D2 receptor (D2R) | Regulation of GSK3 via the protein kinase B (AKT)-pathway, internalization of dopamine D2 receptors | [4, 15, 16, 17, 18] |
| Girdin | Prevention of AKT-pathway activation | [19, 20] |
| TRAF2 and NCK interacting kinase (TNIK) | Dynamic regulation of postsynaptic glutamate receptors | [21] |
| Lissencephaly 1 (LIS1) | Dendritic spine density and morphology, filopodia/axon elongation | [22] |
| Nuclear distribution element-like 1 (NDEL1) | Regulation of mitotic length, cell-cycle progression | [23] |
| 14-3-3 | Axonal elongation | [24] |
| NDEL1–LIS1 | Axonal elongation and neurite outgrowth | [5, 25] |
| NDEL1/the nuclear distribution element 1 (NDE1)–LIS1 | Dynein activity regulation | [5] |
| NDEL1/NudE-like (NUDEL) | Neurite outgrowth in differentiating PC12 cells | [26] |
| Kinesin-1–NUDEL–LIS1–14-3-3 |
Axonal elongation | [24] |
| Kinesin-1–Growth factor receptor bound protein 2 (GRB2) | Axonal elongation | [27] |
| Effect on gamma aminobutyric acid (GABA)-ergic transmission by regulating cortical microtubule-based GABA-A receptor trafficking | [28, 29] | |
| Fasciculation and elongation protein zeta 1 (FEZ1) | Neurite outgrow | [5] |
| Pericentriolar Material 1 (PCM1)-Bardet-Biedl Syndrome 4 (BBS4) | Cargo proteins to the centrosome. Neuronal migration | [30] |
| FEZ1–PCM1–BBS4 | Axonal morphology | [5, 30] |
| Ras association domain family member 7 (RASSF7) | Astrogenesis | [31] |
| DIX domain containing 1 (DIXDC1) | Neural progenitor proliferation | [2, 4, 32] |
| DIXDC1–NDEL1 | Neuronal migration | [4] |
| Amyloid precursor protein (APP) | Neuronal migration, translocation of DISC1 to the centrosome | [33, 34] |
| Phosphodiesterase 4B (PDE4B) | Synaptic plasticity through cyclic adenosine monophosphate (cAMP) response element (CRE) binding protein (CREB), synaptogenesis and/or maturation of synapses | [5, 35, 36] |
| Activating Transcription Factors 4 (ATF4) | DNA-binding transcription activator activity | [37] |
| Nuclear receptor corepressor (N-CoR) | Modulation of CRE-mediated gene transcription | [38] |
| ATF4/ATF5/N-CoR | CRE-mediated gene transcription, epigenetic regulation | [4] |
| N-methyl-D-aspartate (NMDA) receptor | Neuronal migration | [39] |
| Kalirin-7 (Kal‐7) | NMDA‐mediated activation of Rac1, spine morphology | [5, 21, 40] |
| Erb-b2 receptor tyrosine kinase 4 (ErbB4) | Development of cortical inhibitory interneuron dendrite and synaptic growth | [41] |
| Exocyst complex component 1 (EXOC1) | Endoplasmic reticulum Ca2+ signaling | [42] |
| Microtubule actin crosslinking factor 1 (MACF1) | Synapse structure, intracellular transport and cytoskeletal stability | [43] |
| Bardet–Biedl syndrome 1 (BBS1) | Neuronal migration and corticogenesis | [44] |
| Serine/arginine-rich protein (SR) | D-serine generation and NMDA neurotransmission | [45, 46] |
| Coiled-coil protein associated with myosin II and DISC1 (CAMDI) | Radial migration of developing neurons | [47, 48] |
| Dynein | Movement of intracellular transport and cell division | [49, 50] |
| Coiled-coil helix crista morphology 1/Coiled-coil-helix-coiled-coil-helix domain containing 6 (CHCM1/CHCHD6) | Regulation of mitochondrial cristae morphology | [51] |
| Mitofilin | Mitochondrial function | [52] |
| Dynein Intermediate Chain (DIC) | Microtubule‐based cargo transport | [5, 24, 53] |
| Dynactin | Direction of dynein activity | [5, 53] |
| Growth factor receptor-bound protein 2 (Grb2) | Neurotrophin-induced axon elongation | [27] |
| Kinesin family member 5A (KIF5A) | Intracellular transport | [24, 54] |
| Kinesin light chain 1 (KLC1) | Microtubule‐based cargo transport | [5, 24, 55] |
| Basic helix-loop-helix ARNT like 1textbf (BMAL1) | Regulation of glucocorticoid synthesis | [56] |
| Fragile X messenger ribonucleoprotein 1 (FMR1) | Regulation of glutamatergic synaptogenesis | [57] |
| Brain-derived neurotrophic factor (BDNF) | Sensorimotor gating | [58] |
| Growth arrest specific gene 7 (Gas7) | Maturation and morphological differentiation of cerebellar neurons | [59] |
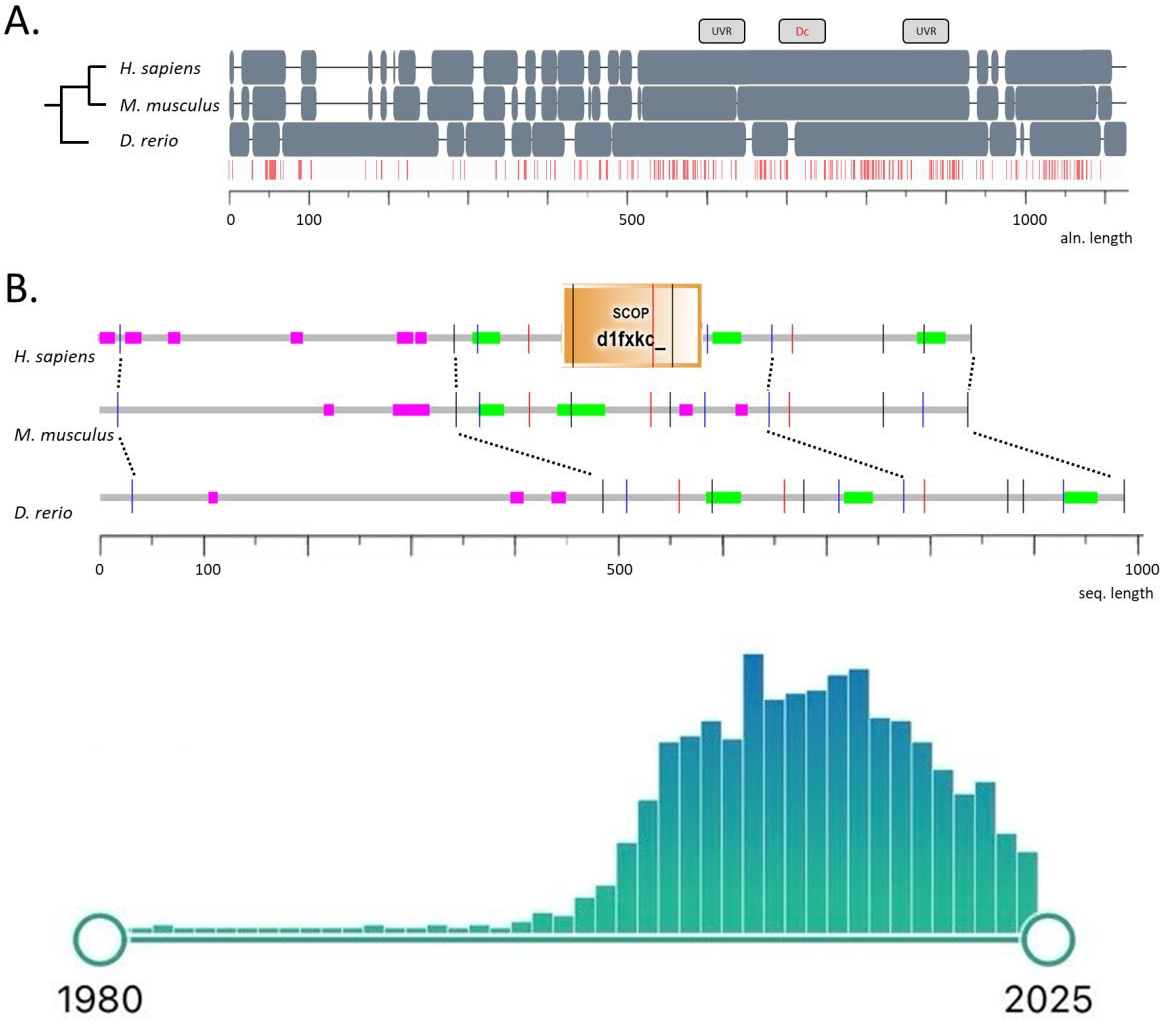 Fig. 1.
Fig. 1.
Protein sequences of DISC1 orthologs in humans, mice, and zebrafish. (A) shows multiple sequence alignment (positions identical in three species are marked in red). The location of DISC1 UVR-like repeats and the DISC1 conserved region (DC box) according to [61] are shown above the sequences. (B) compares annotations of protein regions using the SMART tool [62], where coiled-coil motifs and low complexity regions are marked in color, and positions of introns are given by vertical lines. Inset: Timeline of DISC1-focused publication output based on a PubMed search for “DISC1”, with a total of 1079 results as of February 2025 (note a measurable recent decline in interest in this gene). DISC1, Disrupted-In-Schizophrenia-1; UVR, the family of ultraviolet radiation (UV) resistance globular domains; SCOP, Structural Classification of Proteins.
The human DISC1 gene encodes an 854 amino acid-long protein (Fig. 1) [63], and is expressed ubiquitously as a 7.5 kB mRNA transcript [63, 64]. Human brain imaging studies do not link common single nucleotide polymorphisms (SNPs) in DISC1 to schizophrenia, yet associate its most-studied SNPs, Ser704Cys (rs821616) and Leu607Phe (rs6675281), with neurodevelopmental deficits [65, 66, 67, 68]. As schizophrenia is often linked to dopaminergic hyperactivity [18, 69], positron emission tomography (PET) detects higher striatal dopamine synthesis in Ser704Cys homozygote probands vs. cysteine homo- or heterozygotes [70]. Lifetime suicide attempts in opioid abusers are also linked to the DISC1 rs2738888 SNP, hence implicating DISC1 in opioid dependence and, likely, drug abuse in general [71]. Finally, ablation of DISC1 increases canonical Wnt signaling in neuronal progenitor cells, suggesting the DISC1-dependent suppression of basal Wnt signaling as an important determinant of cell types in cortical neurogenesis [72]. Clinical association studies also link haplotypes and SNPs of DISC1 to reduced gray matter density in the prefrontal cortex, smaller hippocampi, and impaired cognitive performance [73]. Although genetic variance in DISC1 has been reported in probands with schizophrenia, major depression, generalized anxiety, and alcoholism [74], recent genome-wide association studies (GWAS) failed to link its locus to psychiatric disorders [75, 76], necessitating further reconsideration of its role in CNS pathobiology. As will be discussed further, Disc1 mutant animal models display various behavioral and anatomical deficits [77, 78, 79], implicating this gene in behavioral disorders and psychopathology.
While previous animal research has been largely focused on rodents (see further), zebrafish (Danio rerio) have recently emerged as a viable model to investigate DISC1 mechanisms across taxa. With considerable genetic and physiological homology to mammals, zebrafish offer several practical advantages over traditional rodent models, such as their small size, ease of maintenance, and high fecundity—allowing for robust experimental designs and large sample sizes [80, 81]. Their neuroanatomical and neurotransmitter systems are highly conserved with mammals, increasing translational relevance of behavioral findings and fostering the development of novel pharmacological interventions for CNS-related pathologies [82, 83, 84]. Since our complete understanding of the role of this gene in CNS processes necessitates focus on evolutionarily conserved molecular mechanisms and traits, here we discuss currently available animal models of Disc1-related CNS deficits, particularly in regard to the growing potential of zebrafish CNS models. Critically evaluating evolutionarily conserved (shared) and species-specific phenotypes, we outline the translational value of cross-species analysis to further dissect the complex role of DISC1 in CNS pathogenesis.
The mouse and rat Disc1 proteins consist of 852 and 824 amino acids [25, 85, 86], respectively, with highly conserved amino acid sequences (82–91%). Although there is less conservation (50–60% in the globular region and 75% in the helical region) between human DISC1 and rodent Disc1, their intron/exon structure is identical [25]. The largest exon of both human and mouse genes is exon 2 [25, 87]. To date, 16 exons are known in different isoforms in mouse Disc1, and 14 exons in rat Disc1 (see Fig. 1 for details).
Several Disc1 rodent models have been developed [8] whose overt behavioral and anatomical phenotypes can be relevant to major psychiatric disorders. In C57BL/6J mice, N-ethyl-N-nitrosourea (ENU) mutagenesis produced two mutations, L100P and Q31L, with schizophrenia- and depression-like phenotypes, respectively, as well as aberrant neurotransmission and an impaired ability to reorganize cortical plasticity [4, 88]. 129S mutants express truncated Disc1 protein in synapses, impairing the scaffolding of excitatory postsynaptic receptors and reducing the firing rate in pyramidal cells and interneurons [89]. Disc1 knockdown mice display developmental entorhinal-hippocampal hypo-communication [90]. DISC1-N mutants display reduced firing of basolateral amygdala (BLA) neurons marked by Wolfram Syndrome 1 (WFS1) expression (BLAWFS1) and impaired communication with astrocytes [91], whereas DISC1-NTM mice display reduced firing of nucleus accumbens (NAc) parvalbumin (NAcPV) interneurons [92]. Deletion of exon 12 in the Disc1 gene evokes altered phosphodiesterase 4 (PDE4) signaling and synaptic abnormalities in mouse brain [93]. Conversely, overexpression of Disc1 causes misassembly of DISC1 and, consequently, disrupted interactions and downstream signaling [94]. Several mouse Disc1 models also have enlarged lateral ventricles [95, 96, 97], paralleling some clinical signs of schizophrenia.
The most common behavioral deficit in mouse Disc1 models is spontaneous hyperactivity [13, 36, 95, 97, 98] (seen only in males), whereas females display impaired spatial memory [99]. Biallelic functional deletion of the Disc1 gene produces a sexually dimorphic modulation of anxiety and neophobia [79]. Disc1-Q31L mice exhibit aberrant fear memory extinction and emotional behavior, which is also sex-dependent [100]. 129S mutants demonstrate impaired cognition, with decreased exploratory behavior [89]. Disc1 knock-down evokes memory and executive deficits as a result of impaired prefrontal-hippocampal communication throughout development [101]. Neuronal and network activity in the olfactory bulb, as well as the drive from the bulb to the prefrontal-hippocampal network, is also reduced in Disc1+/– mice [102]. Disc1 gene overexpression in tgDISC1 lines leads to a decrease in both cognitive flexibility and social interaction [94]. Disc1 ablation also leads to greater immobility in the forced swim test [95, 98], aberrant social behavior [13, 36, 95, 97, 103, 104], disrupted risk avoidance [91, 92], and impaired pre-pulse inhibition (PPI) [36, 95, 97, 98, 103, 104, 105] (Table 2, Ref. [13, 17, 36, 42, 77, 96, 97, 98, 99, 100, 103, 104, 106, 107, 108, 109, 110, 111, 112, 113, 114]). Overall, neurodevelopmental perturbations induced by the Disc1 knockout result in overt behavioral (i.e., emotional, social, and sensorimotor) effects consistent with a wide range of CNS pathologies seen in humans.
| Models | Details | Physiological, morphological, and behavioral abnormalities | References |
| Mice | |||
| Spontaneous mutations | |||
| 129DISC1Del mutation (functional knockout) | Deletion in exon 6 leading to stop codon in exon 7 as the result of truncated DISC1 | Decreased prefrontal cortex (PC) volume, altered dentate gyrus organization, pre-pulse inhibition (PPI) deficits, increased immobility in the forced swim test, altered locomotion (hyperactivity in males and hypoactivity in females) | [98, 99, 106, 107] |
| Point mutations | |||
| Q31L | Missense point mutations in exon 2 | Reduced brain volume and lower DISC1 binding to GSK3 |
[17, 100] |
| L100P | Reduced brain volume and DISC1-binding, lower accumbal monoamine levels, DS density, hyperlocomotion, impaired PPI, latent inhibition and working memory deficits | [36, 108] | |
| D453G | Missense mutation of the C-terminus | Decreased DISC1-GSK3 |
[13] |
| Constitutive transgenic models | |||
| CaMK-DN (Dominant negative)-DISC1 tg | Expression of truncated DISC1 under control of an |
Enlarged lateral ventricles, decreased immunoreactivity in the medial prefrontal cortex, impaired olfaction, increased activity, deficits in PPI and working memory, increased despair—like immobility in the forced swim test, decreased social interaction | [109] |
| BAC (Bacterial artificial chromosome)-DISC1 tg | Expression of two copies of truncated DISC1 protein (first 8 exons) | Smaller cortex (reduced neuronal proliferation), enlarged lateral ventricles, callosal agenesis, impaired latent inhibition and fear memory, despair-like behavior in the forced swim test | [96] |
| Inducible transgenic models | |||
| Parv2A-tTA2; mutant DISC1 | Selective inducible expression of truncated DN-DISC1 in the cerebellum | Fewer large Purkinje cells and more small-somata cells, increased spontaneous activation of the Purkinje cells, poorer spatial and social recognition memory | [103] |
| CaMK-DISC1-cc tg (inducible at postnatal day 7) | Encoding residues 671–852 in the C-terminal portion of DISC1 | Reduced hippocampal dendritic organization, impaired spatial and working memory and social interaction; high immobility in forced swim test | [104] |
| DN-DISC1 tg (mhDISC1) | Tet-off system under control of |
Mild enlarged lateral ventricles, decreased neurite outgrowth in primary neurons, hyperlocomotion and aggression, decreased social interaction, delayed methamphetamine sensitization (males), impaired spatial memory and conditioned place preference (females) | [97] |
| HRM/DISC1 double mutant | Heterozygous reelin (RELN) haploinsufficiency mice (HRM) crossed with C-terminally truncated DN DISC1 | Impaired pre-pulse inhibition, altered cognition, and decreased activity, increased |
[77] |
| Deletion of exons | |||
| DISC1Δ2-3 | Deletion of exons 2–3 | Shifted induction threshold of long-term potentiation (LTP) in the dentate gyrus, compulsive-like behavior (males), impaired PPI and increased methamphetamine induced locomotor activity (females), reduced anxiety (both sexes) | [42, 110] |
| DISC1Δ1-3 | Deletion of a large locus generating exons 1–3 | Excursive APP trafficking in cortical neurons leads to amyloid-beta peptide generation | [111] |
| Rats | |||
| Inducible transgenic models | |||
| Overexpression of full-length human DISC1 tg (transgenic) | CosSHa.tet vector with full-length, non-mutant human DISC1 with the F607 and C704 polymorphisms | Increased hippocampal and decreased callosal volume, fewer tyrosine hydroxylase (TH)-positive fibers and parvalbumin (PV) interneurons in the striatum, lower dopamine and more dopamine transporter levels in the striatum, impaired long-term-memory and attention-related behavior, increased anxiety, hyperlocomotion after amphetamine administration | [112, 113] |
| Knockout models | |||
| DISC1 svΔ2 | Nonsense mutation in exon 2 (CRISPR-Cas9) with nonsense, frame-shifted, and truncated DISC1 | Decreased neurite density in the hippocampus, basal ganglia, and neocortex, altered latency to startle (males) | [114] |
CaMK, Ca2+/calmodulin-dependent protein kinase; CRISPR, clustered regularly interspaced short palindromic repeats; Cas9, CRISPR-associated protein 9.
DISC1 modulates dopaminergic function by interacting with several proteins involved in dopamine signaling, including fasciculation and elongation protein zeta 1, phosphodiesterases (PDEs) PDE4D9 and PDE4B, serine/threonine protein kinase Akt, and glycogen synthase kinase 3 (GSK3, Table 1). DISC1 transgenic mice with inducible expression of mutant human DISC1 (hDISC1) display lower cortical dopamine, whereas combined pre- and postnatal expression of hDISC1 triggers aggression [115]. Transient knockdown of Disc1 during development in pyramidal neurons of the prefrontal cortex impairs mesocortical dopamine neurons maturation and associated behaviors (e.g., impaired PPI and novel object recognition) [116, 117].
Mutant Disc1 mice (line 1001) also show delayed methamphetamine-induced sensitization and impaired conditioned place preference (CPP) [118], while DISC1-L100P mouse mutants show a greater response to amphetamine and more striatal dopamine receptors, as well as deficits in PPI and latent inhibition (LI) that are reversed by haloperidol [108]. Chronic N-acetylcysteine (NAC) administration normalizes the amphetamine-induced hyperactivity in heterozygous Disc1 mice [78]. In Disc1 knockdown mice clozapine elevates dopamine levels via blocking dopamine D2 auto-receptors as well as normalizes dopamine in the medial prefrontal cortex (mPFC), PPI, and novel object recognition [116]. DISC1 mutant mice (line 1001) show greater responses to an N-methyl-D-aspartate (NMDA) receptor antagonist, dizocilpine (MK-801), in the open field and PPI of the acoustic startle tests, and are significantly more sensitive to the ameliorative effects of D-serine [45]. Deletion of exon 12 in Disc1 produces mice with elevated levels of PDE4 with social and cognitive deficits, reversible by pharmacological inhibition of the PDE4 signaling pathway [93].
Although attenuated hippocampal serotonin neurotransmission has been reported in Disc1 mice [119], and low serotonin levels were detected specifically in the NAc in Disc1-Q31L mutant mice [120], no serotonergic drugs have been tested systematically in these Disc1 mouse mutants. In adult mice expressing a putative dominant-negative (DN) DISC1 mutation, chronic adolescent treatment with delta-9-tetrahydrocannabinol (THC) exacerbates deficits in fear-associated memory [121]. Selective expression of DN-DISC1 in astrocytes of mice exposed to THC decreases recognition memory [122]. In DN-DISC1 mice, exploratory activity, hippocampal short-term synaptic facilitation, and brain-derived neurotrophic factor (BDNF) levels are reduced, and their cannabinoid receptor 1 (CB1R) expression is down-regulated following THC administration [123]. Overall, DISC1 models show different pharmacological modulations and various mutations in the gene may alter CNS drug responses in rodent models. The involvement of DISC1 in the regulation of specific neurotransmitter systems (i.e., dopamine, serotonin, glutamate, and endocannabinoid, as discussed above) is particularly critical, as it may help elucidate mechanisms of DISC1 in disease onset and development, based on targeting these neurotransmitter systems.
Chronic exposure to adverse events, especially in early life, can affect behavior and brain formation of animal models, hence increasing the likelihood of neuropsychiatric disorders [124]. Similarly, juvenile stress increases hippocampal expression of Disc1 in male, but decreases in female, mice [125]. Likewise, chronic mild stress (CMS) increases DISC1 hippocampal expression in male mice [126]. In Disc1 knockdown rats, a 1-h restraint stress impairs working memory, whereas in non-stressful conditions, the loss of Disc1 has no effects [127]. The 3-week isolation stress induces PPI deficits as well as impairs locomotor activity and performance in the forced swim test in adolescent DISC1-DN transgenic mice [124]. Lower dopamine levels in the ventral tegmental area, elevated expression of dopamine D2 receptors, and reduced expression of tyrosine hydroxylase are also seen in the frontal cortex of DISC1-DN transgenic mice [124]. Chronic social defeat stress decreases baseline locomotor activity and exploration in L100P+/-, but not wildtype or Q31L+/- mice [128]. In contrast, decreased sociability and social novelty-seeking are observed in both wildtype and L100P+/- mice [128]. Adolescent isolation impairs adult social memory, neurogenesis, and synaptic functions in the hippocampus of DISC1-L100P point mutant mice [129].
Chronic, lifelong exposure of mDISC1 mice to Pb2+, a potent selective NMDA antagonist, is not associated with gross developmental abnormalities but produces sex-dependent hyperactivity, exaggerated responses to the NMDA receptor antagonist MK-801, mildly impaired PPI, and enlarged lateral ventricles [130]. Other environmental stressors can lead to immune activation which, in turn, evokes pathological phenotypes. For instance, in male tgDISC1 rats, juvenile immune activation disrupts spatial and recognition memory [131]. Introduction of DN-Disc1 mice to PolyI:C daily during embryonic development, impairs short-term memory in the object recognition task and contextual fear memory [132]. Prenatal adverse events, such as excessive maternal immune activation and activation of cytokine signaling, can alter the expression and epigenetic regulation of DISC1 [133]. DNA methylation at the tyrosine hydroxylase gene promoter in dopaminergic projection neurons of the ventral midbrain is altered in Disc1 mutant mice [124].
Significant gene–environment interactions (G
Over the last decades, numerous DISC1 rodent models have been generated and characterized for histological, anatomical, neurochemical, and behavioral phenotypes [137, 138, 139]. Yet there is considerable variability among models given the genetic manipulation and assays used to characterize it (e.g., Disc1 point mutants vs. C’-truncated transgenic lines). For instance, DN human DISC1 transgenic mice under the alpha calmodulin kinase II (CaMK) promoter induces expression of DN-DISC1 in the pyramidal neurons of the forebrain, whereas a prion protein (PrP) promoter induces its expression in most brain regions and cell types, hence contributing to substantial phenotypic heterogeneity [137]. This diversity significantly complicates comparison between models, underscoring the need for simpler, genetically tractable models.
Complementing rodent and clinical evidence, the zebrafish is widely used to
study CNS disorders [84], due to the ability to perform rapid loss- and
gain-of-function assays with a high-throughput capability [140, 141]. The
zebrafish DISC1 (zdisc1) gene is annotated on chromosome 13
[142] and, like in rodents, shows synteny with the human chromosome 1 locus, with
the neighboring genes TSNAX (encoding translin-associated factor X) and
EGLN1 (encoding hypoxia-inducible factor prolyl hydroxylase 2) located
upstream of DISC1 [142]. Despite the whole-genome duplication in the
evolution of teleost fishes, there is only one disc1 ortholog in the
zebrafish genome [143], and its second copy has probably been lost [144]. The
zdisc1 gene is expressed maternally and zygotically, with strong
expression in the developing nervous system [145], especially in the midline of
the hindbrain near the optic tectum, at the midbrain-hindbrain boundary, and the
ventral diencephalon [142]. Zebrafish DISC1 and human DISC1
functions are generally conserved, since expression of the human gene in
zebrafish DISC1-morphants rescues the phenotype, also necessitating the
GSK3
As shown in Fig. 1 (Ref. [61, 62]), genomic sequences and transcription maps in humans, mice, and zebrafish show that the amino acid sequence of DISC1 is rapidly diverging, although the nuclear localization signal, coiled-coil regions, and the alternatively spliced variants of DISC1 protein are also conserved [146]. Comparing protein sequences across the three species (Fig. 1) shows the N-terminal part of the multiple alignment contains deletions, insertions, and a relatively low-density of conservative positions, while the C-terminus is aligned between mammals and zebrafish practically without inserts, containing multiple conserved positions [61]. The conserved region (amino acids 530–850) contains binding sites to other proteins (Fig. 2, Ref. [147]), whereas the N-terminal small group of sites (GSK3, kalirin 7/Kal7, and PDE4B) reveals multiple conservative positions in all three species, hence supporting the evolutionarily conserved molecular functions and structure of DISC1. Analysis of disc1 in embryonic zebrafish (from 24–55 hours post fertilization/hpf to 2–3 days post fertilization/dpf) reveals prominent expression in the basal part of the brain, especially the hypothalamus [148]. The 55-hpf hypothalamic disc1 is restricted to cells around the lateral recesses and posterior tuberal 3rd ventricle—the neurogenic zones that harbor proliferating progenitors [149].
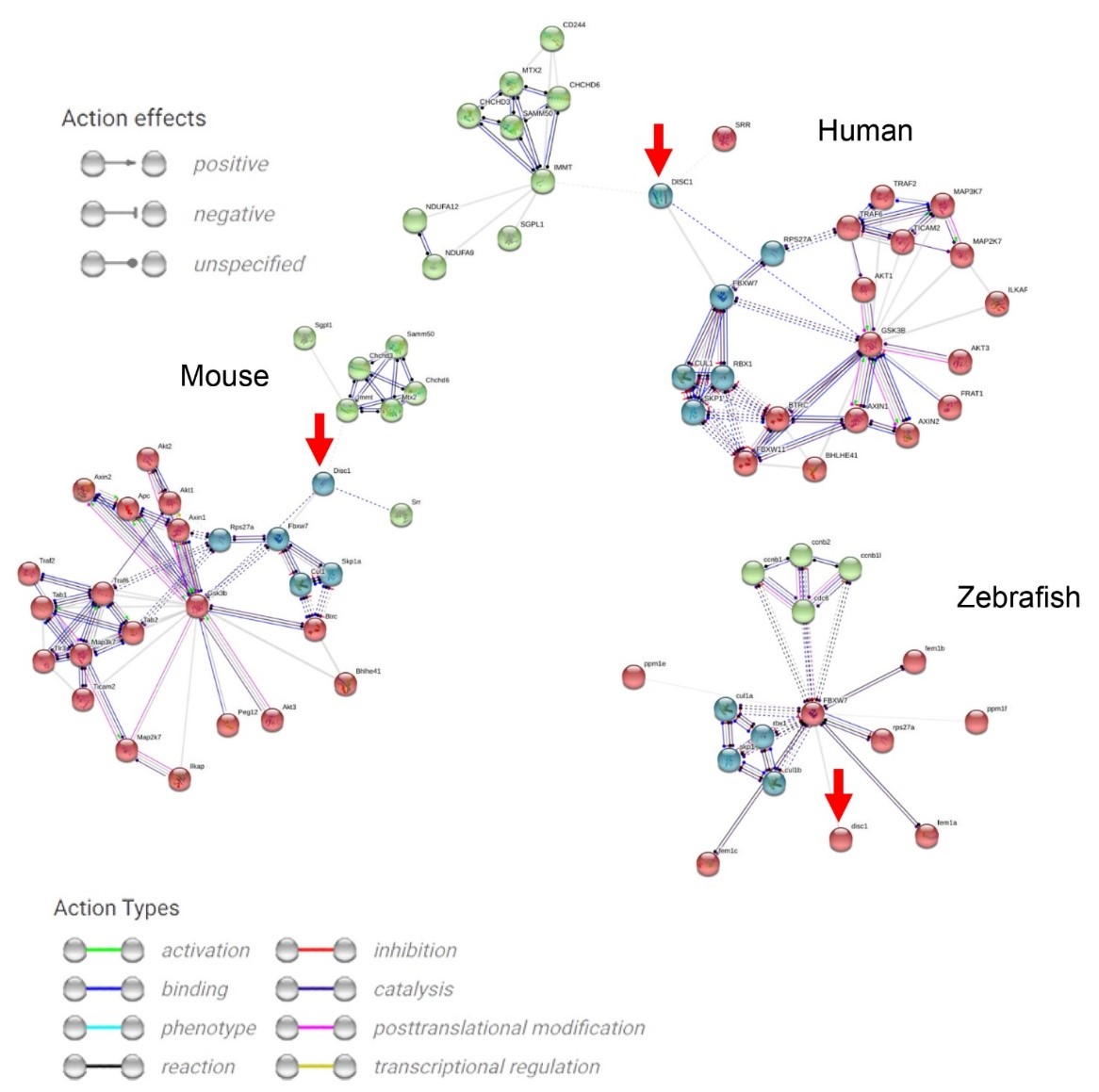 Fig. 2.
Fig. 2.
Molecular networks generated for known protein interactions for
human, mouse, and zebrafish DISC1 (denoted by red circles and arrows) using the
STRING database [147] (https://string-db.org/, accessed February 2025). Edges
represent various types of known molecular interactions using only experimental
data (excluding text mining, databases data, co-expression, neighborhood, gene
fusion, and co-occurrence) with the minimum required interaction score set to
0.4, and the number of interactors restricted to 30, and 2nd shell interactors
calculated as 30 minus the suggested number of the 1st shell interactors. The
kmeans clustering was used to cluster the nodes, with each cluster represented by
a different color. Note, for example, the green cluster containing mostly
mitochondrial function proteins (e.g., inner mitochondrial membrane protein
encoded by the IMMT gene, the sorting and assembly machinery component 50 homolog
SAMM50, mitochondrial contact site and cristae organizing system MICOS), the red
cluster formed by proteins involved in the Wnt- and the mitogen-activated protein
kinase (MAPK)-signaling cascades (e.g., glycogen synthase kinase 3
Analyses of human, mouse, and zebrafish data shows that aberration in the
DISC1 gene similarly affects neuronal migration (e.g., amyloid precursor protein (APP),
dixin/Dixdc1, lissencephaly 1 (LIS1), the nuclear distribution E neurodevelopment protein 1/NDE1
and NDE1–like protein NDEL1), neural progenitor proliferation (GSK3
While the differences in zebrafish and mammalian DISC1 signaling pathways,
binding sites, and molecular interactors have not been systematically assessed,
individual signaling pathways involved in the DISC1 interactome seem to be
conserved across species. We also constructed the interactome (Fig. 2) and
molecular networks for known protein interactions for human, mouse, and zebrafish
DISC1 (denoted by red circles and arrows), showing that DISC1 is involved in both
canonical (Wnt/
Another important protein in the DISC1-interactome is caveolin-1, regulating neuronal signaling, membrane/lipid raft formation, and dendritic development [151]. It also modulates DISC1 expression in both humans and rodents, as well as regulates dorsoventral patterning in embryonic zebrafish [152, 153]. Interestingly, caveolin-1 knockout zebrafish display developmental abnormalities and embryonic lethality, unlike their mouse counterparts [153]. Zebrafish models also support a critical role of the DISC1 gene in oligodendrocyte development and olig2-positive cerebellar neurons [142], as well as dorsolateral cell migration during gastrulation, causing abnormally small brain ventricles and axonal growth defects [145]. Disc1 is also implicated in transcriptional repression of foxd3 and sox10, promoting aberrant zebrafish cranial neural crest migration and differentiation [143].
The zdisc1 mutant embryos also show aberrant proliferation of rx3+ hypothalamic progenitor cells and neuronal differentiation, both critical for anxiety-related behaviors and corticotrophin-releasing hormone (CRH) signaling [149] (see Table 3, Ref. [1, 142, 143, 145, 149, 154, 155] and Fig. 3, Ref. [13, 36, 110, 149] for details). Homozygous disc1 mutants have altered expression of CRH in the tuberal hypothalamus and preoptic hypothalamus [149]. The effects of disc1 mutation are dynamic across the early life period—with increased CRH expression in the neurosecretory preoptic area (NPO) region at embryonic stages, but decreased expression in the larval hypothalamus [156]. The reduction in CRH-expressing neurons at larval stages correlates with a blunted behavioral and endocrine response to acute stress exposure in mutant larvae, consistent with the attenuated hypothalamus-pituitary-inter-renal (HPI) axis reactivity [157]. Given the essential role of CRH in stress regulation and the altered stress response in disc1 mutant zebrafish [149], it is likely that this gene regulates the HPI axis via CRH, clearly meriting further scrutiny [156].
| Models | Physiological and morphological abnormality | Behavioral abnormality | References |
| Mutations | |||
| Disc1L115—a premature stop codon in the N-terminal head domain of DISC1 (Adult) | Lower rx3 expression in the lateral recesses, absent expression in the 3rd ventricle | Insensitivity to alarm pheromone | [149] |
| Disc1Y472—a premature stop codon in the N-terminal head domain of DISC1 (Adult) | Lower rx3 expression in the lateral recesses, absent expression in the 3rd ventricle | Increased freezing and fast swimming, no natural preference for light arenas, failure to modulate shoaling, insensitivity to alarm pheromone | [149] |
| Disc1Y472 (Adult) | Not assessed | Lower swimming activity, no differences in startle habituation | [154] |
| Disc1Y472 (Larvae) | Not assessed | Hypolocomotion | [154] |
| Disc1 L115 (Adult) | Not assessed | Increased bottom dwelling, fewer entries to the top of the novel tank, impaired reversal learning | [154] |
| CRISPR-Cas9 knockout (Adult) | Not assessed | Aberrant shoaling (staying close but not moving as a coherent group) | [155] |
| Knockdown | |||
| Morpholino antisense oligonucleotide (MO, Larvae) | Near total loss of olig2-positive cerebellar neurons, embryonic failure to inflate the swim bladder, misshapen eyes and head, curved trunk and tail | Not assessed | [142] |
| MO targeting donor site between exon 1 and intron ½ (Larvae) | Defects in forebrain formation, reduced brain ventricles, failure of axon outgrowth throughout the brain, absent tails | Not assessed | [1] |
| MO targeting between exons 8–9 (Larvae) | Reduced hindbrain and midbrain ventricles, short and bent tail, abnormal axon growth | Not assessed | [1] |
| MO altering the splicing of the disc1 pre-mRNA, deleting exon 3 (Larvae) | Aberrant neural crest cell migration, enhanced peripheral glial population | Not assessed | [143] |
| MO targeting the intron 1/exon 2 boundary (Larvae) | Abnormal movement of dorsolateral cells during gastrulation, truncated tail, abnormal brain morphology, reduced ventricles | Not assessed | [145] |
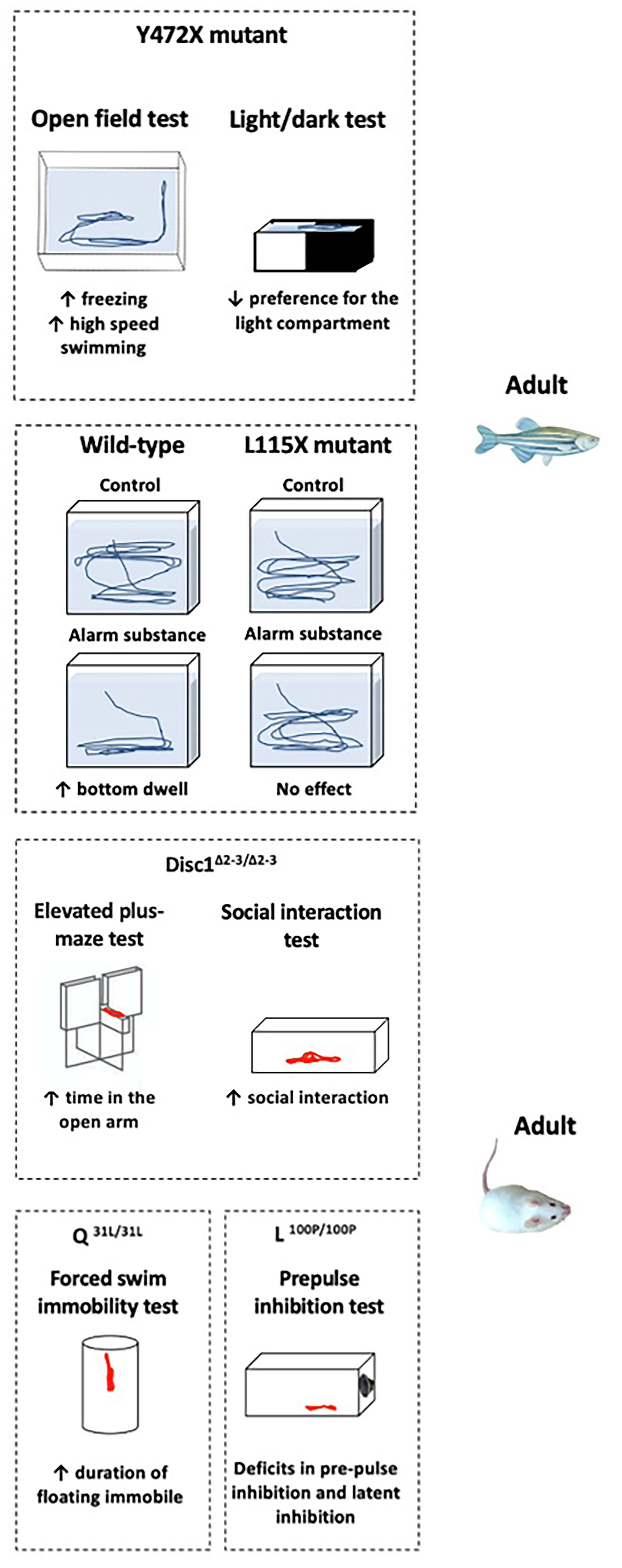 Fig. 3.
Fig. 3.
Selected behavioral phenotypes of zebrafish and rodent DISC1 gene mutants. The left panel shows larval Y472X and L115X exposure to alarm substance that unalters shoaling behavior [149]. The next panel shows adult zebrafish Y472X mutants with increased freezing and high-speed swimming in the open field test, and reduced preference for the light compartment in the light/dark test [149]. Alarm substance increases bottom dwelling in the wild-type fish, but has no effect on L115X mutants [149]. The third panel shows young adult female DISC1D453G mice displaying hyperlocomotion in the open field test, an anxiogenic-like behavior (e.g., less open arm exploration) in the elevated plus-maze, and reduced social interaction [13]. Adult DiscΔ2-3/Δ2-3 mutant mice display lower anxiety in the elevated plus-maze test and increased social interaction [110]. The last two panels show that adult mice with the Q31L mutation show depressive-like behavior in the forced swim test, and L100P mutant mice exhibit poorer pre-pulse and latent inhibition [36]. However, given the challenge of comparing complex behaviors across such evolutionarily distant species, caution must be used when drawing direct parallels. Arrows directed upwards denote the increase, and directed downwards - the decrease, of the respective behaviors.
Behaviors of zebrafish DISC1 models are also characterized in detail (Table 3). For example, Y472X mutant fish show increased freezing and fast swimming, as well as absent light-dark test preference [149]. L115X mutants display normal top/bottom swimming in the novel tank test following exposure to an anxiogenic alarm substance. Y472X mutants swim more slowly and also fail to modulate shoaling behavior in response to acute stress exposure due to abnormalities in the lateral line development or the visual system [149]. Zebrafish disc1 mutants also exhibit altered sociability, staying close to one another but not moving together as a coherent group [155]. While their underlying mechanism is unknown, it is possible that these observed behaviors are due to impaired vision in the Y472X mutant strain.
Longitudinal neurobehavioral analysis of disc1 mutant zebrafish across key stages of life further implicates disc1 in sensorimotor processes and the genesis of anxiogenic behaviors. During early developmental stages, the loss-of-function disc1 mutants exhibit abrogated behavioral responses to sensory stimuli, including abnormal activation of neurons in the pallium, cerebellum, and tectum—anatomical sites involved in the integration of sensory perception and motor control—when exposed to acoustic stimuli. Adult zebrafish exhibit a sexually dimorphic reduction in anxiety-like behavior in the novel tank test, supporting the role of disc1 in zebrafish HPI axis function [158]. Together, these findings implicate disc1 in sensorimotor processes and anxiogenic-like behaviors, which can be exploited for the development of novel treatments in addition to investigating the biology of sensorimotor function in the context of disc1 deletion.
The role of disc1 in modulating pharmacological effects has also been explored in zebrafish. For example, 15 dpf larvae exposed to water-soluble fraction of crude oil or lead show decreased disc1 gene expression [159]. Exposure of larvae with the loss-of-function mutation in DISC1 to a synthetic cannabinoid 1-pentyl-3-(1-naphthoyl)-indole (JWH-018) leads to an impairment of locomotion not seen in drug-exposed wild-type fish [160]. Later testing of adult fish revealed their higher anxiety-like behavior—opposite to anxiolytic-like effects in exposed wild type fish, further supporting the role of disc1 in the zebrafish HPI axis [149, 160]. Alterations in zebrafish disc1 disrupt the specification of oligodendrocytes and neurons [142] and the migration and differentiation of the neural crest (i.e., cells that form the craniofacial cartilage) [143], hence contributing to aberrant behavior of disc1 mutants.
The main epigenetic mechanisms are also generally evolutionarily conserved and
shared between mammals and zebrafish [161]. Because cortisol-mediated stress
responses to different environmental stimuli are also disrupted in zebrafish
disc1 mutants [156, 158], this makes zebrafish DISC1 models a valuable
tool to study G
As already mentioned, the vast information on DISC1 function in the CNS has been generated by human and rodent evidence. However, despite overt differences with mammals in neurodevelopment and the lack of a neocortex, zebrafish have been extensively used as a vertebrate model organism in genetic studies of CNS pathologies. Large-scale investigation of gene function has been achieved primarily through ENU- [163] and retroviral mutagenesis [164, 165], also referred to as ‘forward’ genetics. With the development and application of engineered endonucleases, including zinc finger nucleases (ZFN) [166, 167], transcription activator-like effector nucleases (TALEN) [167], and the CRISPR/Cas system [168, 169, 170], ‘reverse’ genetics has expanded rapidly [171, 172]. The generation of transgenic lines for direct visualization of neuronal populations in vivo during migration, axon extension, and establishment of connections, particularly facilitates morphogenetic studies using zebrafish [173]. In zebrafish, the loss of disc1 triggers persistent CNS cell medial migration, dorsal to the developing neural epithelium, and hinders their migration away from the region dorsal to the neural rod [143]. Reduced zdisc1 expression also disorganizes the formation of the axon tracts (e.g., the post-optic and anterior commissures and the supraoptic tract) [67]. The synaptic location of zebrafish DISC1 implicates this gene in synaptic plasticity. Due to the availability of stable transgenic lines fused to fluorescent proteins, larval zebrafish models have become most promising (among in vivo vertebrate models) for imaging the spatial and temporal dynamics of synaptogenesis [174].
Measuring neuronal activity in zebrafish using in vivo whole brain calcium imaging can help establish zebrafish models specific for cell type, especially given cell-specific expression of disc1 (e.g., glial cells) [143, 175]. This technology offers several advantages, such as recording activity from thousands of neurons, including sub-cellular structures, as well as tracking of the same cells over time [175, 176, 177, 178]. The precise spatial localization afforded by calcium imaging also enables post hoc characterizations in situ by immunohistology, cell-attached/whole-cell recordings, or electron-microscopy [178]. In particular, the tool may further decipher the function of, for example, CRH neurons. Calcium imaging in disc1 mutant fish with and without stressor exposure can provide insights into whether disc1 regulates the activity of CRH neurons in vivo. Similarly, calcium imaging of CRH neurons in animals reared under different contexts (such as early life stress exposure) can reveal how development shapes the stress response [156]. Single cell RNA sequencing studies in mammals indicate that there are multiple subsets of hypothalamic CRH cells [179, 180] whose sequencing in disc1 mutant fish can be used to tease out specific sub-population differences in CRH neurons resulting from a lack of disc1 function, as well as identifying molecules linking disc1 to the development and function of CRH neurons [156].
Altering the function of an animal orthologous disease-associated gene either by mutation or knock down of the gene product is a common approach to study its role in human pathologies [181]. However, genetic DISC1 models, especially in novel organisms (such as zebrafish) and coupled with various environmental stressors, often present challenges and limitations. For example, on one hand, depletion of DISC1 at developmental stages can turn on putative compensatory mechanisms that attenuate pathological phenotype. On the other hand, multiple expression of DISC1 isoforms may provide a wide spectrum of possible phenotypes of animal models [182]. Different lifetimes of DISC1 isoforms may also affect behavior [182]. Nevertheless, due to the high reproductivity and ease of genetic manipulation of zebrafish models, these obstacles can be overcome by creating stable genetic zebrafish lines.
Given that numerous pharmacological compounds have already been assessed in zebrafish disc1 mutants [149, 159, 160], the application of emerging screening platforms for further high-throughput examination is an attractive prospect. The integration of machine learning and computer vision tracking technologies for automated behavioral phenotyping provide a rapid, yet nuanced screening of pharmacological agents or gene functions relevant to schizophrenia and other neuropsychiatric disorders [183]. For example, algorithms using deep neural networks can track multiple adult zebrafish simultaneously, reconstruct their 3D swim paths, and accurately classify movements into specific behavioral categories [184, 185]. This reduces human error and allows for large-scale, high-throughput analysis of behavior—especially beneficial when investigating subtle phenotypic differences induced by genetic mutations [186, 187].
Despite intense efforts to decipher the molecular genetic mechanisms of, and to develop effective therapeutics for, devastating brain disorders such as schizophrenia and bipolar depression, their underlying pathology remains poorly understood, and their presence severely debilitating [157, 188]. The use of rodent models for the identification of new therapeutic options for patients has proven extremely difficult due to the prohibitively high costs associated with generating and analyzing comprehensive mutants and the limited throughput of drug testing. Thus, genetically tractable zebrafish may offer a powerful investigative tool for the dissection of genetic risk alleles and high-throughput molecular screening of novel therapeutics.
A practical challenge in cross-species analysis of DISC1-mediated pathological mechanisms is the lack of established neurobehavioral models in zebrafish. As shown in Table 3, data are rather scarce in regard to the spectrum of zdisc1-associated behavioral phenotypes, thereby complicating their interpretation and analyses from an evolutionary standpoint. Likewise, data are limited on region-specific aspects of Disc1-mediated regulation in the brain, including all three species. Moreover, disc1-associated phenotypes often involve volumetric alterations in the brain structures, and such studies in zebrafish are becoming critical to ensure cross-species translation of volumetric phenotypes.
In silico analyses using public databases further support this notion. For example, DISC1 in general has low tissue specificity, and is in multiple brain areas, including the cortex, cerebellum, olfactory region, hippocampus, amygdala, basal ganglia, thalamus, hypothalamus, and corpus collosum, with the lowest expression in the cerebellum, cerebral cortex, and olfactory region and the highest in basal ganglia, amygdala, hippocampus, thalamus, pons, medulla, and midbrain (Human Protein Atlas https://www.proteinatlas.org/ and Allen Human Brain Atlas https://human.brain-map.org/, both accessed February 2025) [189, 190]. Similarly, in the mouse brain, Disc1 is expressed throughout most areas, but has a different expression pattern, most prominently expressed in the cortical subplate, isocortex, olfactory bulbs, hippocampus, and striatum, and least prominently in the pallidum, thalamus, hypothalamus, midbrain, pons, and medulla (Allen Mouse Brain Atlas https://mouse.brain-map.org/, accessed February 2025) [191]. Therefore, DISC1 in human cortical and olfactory structures, pons, and medulla, is under-expressed compared to mice—which possibly relates to the unique aspects of human cortical development and its vulnerability in psychiatric disease. While no histological zdisc1 brain expression data are yet available for the adult zebrafish, complicating direct cross-species comparisons, DISC1 is expressed in 24–55-hpf larvae in the hypothalamus, supporting its role in neuronal cell differentiation in vertebrates [149].
Mounting evidence suggests putative sex differences in the effects of DISC1 on the onset of neuropathological phenotypes [192], including sex-specific effects of common DISC1 variants on the volumes of the basal ganglia, the amygdala, and the cortical surface area [193]. This notion is supported by Disc1 mutant mouse data of sex-dependent behavioral deficits [13], such as novelty-induced hyperlocomotion, an anxiogenic profile in the elevated plus-maze and open field tests, reduced social exploration in DISC1D453G females, and impaired passive avoidance in males [13]. Presently, zebrafish have not been used to study sex differences in DISC1-related behavioral deficits, although mounting evidence indicates important sex differences in zebrafish behavioral and neuropharmacological responses [194], making it a promising tool for future behavioral research of DISC1.
A strong advantage of zebrafish models is their high neurogenerative potential. In contrast to mammals, zebrafish can constitutively produce new neurons along the whole rostrocaudal brain axis, due to the presence of continuously proliferating stem/progenitor cells [195]. DISC1 plays a key role in neurodevelopmental processes, regulating cortical development (progenitor proliferation and neuronal migration) and hippocampal neurogenesis [105]. Overexpressing DISC1 in primary neural stem cells via lentiviral transfection significantly affects their proliferation and differentiation [196]. Moreover, suppression of DISC1 expression reduces neural progenitor proliferation, leading to premature cell cycle exit and differentiation [8]. Given their superior neurogenic ability, zebrafish models more than rodents may help understand the role of DISC1 in complex signaling pathways, as well as its effect on neural stem cell proliferation.
Environmental pollution has increasingly been implicated in the development of certain neuropathologies in humans, including schizophrenia, bipolar disorder, and attention deficit hyperactivity disorder (ADHD) [197, 198, 199]. With the continuous development of precise detection technology, a growing number of contaminants within the environment have been detected [200]. Neurotoxic pollutants have attracted particular attention due to their potential threat to human and animal life. Studies on the toxicity of environmental pollutants to the zebrafish CNS have largely focused on morphology and behavior regulation, gene expression, and neural development [201, 202, 203, 204]. However, research into epigenetic toxicity, blood-brain barrier damage, and regulation of the brain-gut-microbiota axis require further investigation at the molecular and signaling levels to discern the mechanisms of pollutant toxicity in zebrafish [200]. In particular, identification of changes in gene-specific methylation represents a fundamental issue in the emerging field of environmental epigenetics. Several recent studies have suggested that pollutant exposure can induce alteration in DNA methylation patterns by disturbing the expression of certain genes during zebrafish embryogenesis [200, 205, 206, 207]. Further research is needed to determine if zebrafish disc1 expression can be influenced in a similar way. Importantly, epigenetic alterations in certain human neurological disorders, acquired through environmental factors, can not only influence brain functions throughout the entire lifespan, but can be transmitted across generations via epigenetic germline inheritance as well [208]. While this phenomenon has been well documented in rodents [209, 210, 211, 212], it has yet to be explored in zebrafish, as modifications regulating germline epigenetic plasticity are plausible molecular sources of phenotypic heterogeneity and offer a viable target for therapeutic interventions [208].
The complex genetic origins of many human CNS-related disorders suggest that
epistatic interactions between genes (G
In summary, mounting translational evidence discussed here indicates the importance of cross-species analyses that have multiple implications for the future research of DISC1-related CNS pathologies. With the availability of constantly emerging modern genetic tools, zebrafish can make an ideal case for exploring unveiled questions of DISC1 biology (briefly summarized in Table 4), and complement clinical and rodent studies in elucidating its exact fundamental role in pathological circuits. Given the inconsistencies and variability in rodent studies of DISC1, zebrafish may also offer a higher predictive validity in the search for novel therapeutics. Overall, a more comprehensive understanding of complex CNS functions of this key cellular protein and its gene in vivo may result in renewed interest in these biomolecular markers.
| Questions |
| Environment-related |
| Cellular mechanism-related |
| Disease-related |
| Translational |
ADV: Formal analysis, Conceptualization, Writing – review & editing, Writing – original draft, Illustration, Investigation. SVC: Formal analysis, Conceptualization, Writing – review & editing. NDC: Formal analysis, Visualization, Writing – review & editing. KVS: Formal analysis, Writing – review & editing. AVD: Data analysis, Writing – review & editing. DSG: Formal analysis, Conceptualization, Writing – review & editing. MSA: Formal analysis, Conceptualization, Writing – review & editing, Investigation. TS: Formal analysis, Conceptualization, Writing – review & editing. TL: Formal analysis, Writing – review & editing, Formal analysis. MP: Writing – review & editing, Formal analysis. LY: Writing – review & editing, Formal analysis, Conceptualization. LWL: Writing – review & editing, Formal analysis. AMS: Writing – review & editing, Investigation, Formal analysis, Revision. TGA: Writing – review & editing, Investigation, Conceptualization, Supervision, Investigation, Revision. AVK: Writing – original draft, Supervision, Project administration, Investigation, Funding acquisition, Formal analysis, Conceptualization. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work
Not applicable.
Not applicable.
The study was supported by budgetary funding for basic scientific research of the Scientific Research Institute of Neuroscience and Medicine (project AAAA-A16-116021010228-0). The funders had no role in the design, analyses and interpretation of the submitted study, or decision to publish. AVK prior research was supported by St. Petersburg State University funds. His current research, as well as LY and LWL, are supported by the School of Science, Xi’an Jiaotong-Liverpool University, Suzhou, China.
The authors declare no conflict of interest. Allan V. Kalueff is an Editorial Board member of this journal. He had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Bettina Platt.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



