1 Paris-Saclay University, CEA, Joliot Institute, NeuroSpin, UNIACT, F-91191 Gif-sur-Yvette, France
2 Paris City University, Inserm, U1141 NeuroDiderot, InDev Team, F-75019 Paris, France
3 Sport Sciences Department, Univ Rouen Normandie, CETAPS, F-76000 Rouen, France
4 Paris Brain Institute, Centre d’Investigation Clinique; Assistance Publique Hôpitaux de Paris, Département des Maladies du Système Nerveux, Hôpital Pitié-Salpêtrière, F-75013 Paris, France
5 Public Assistance Hospitals of Paris, Department of Neurology, Institute of Memory and Alzheimer’s Disease, Pitié-Salpêtrière Hospital, F-75013 Paris, France
6 CEA, NeuroSpin, CNRS, Université Paris-Saclay, F-91191 Gif-sur-Yvette, France
7 BAOBAB, NeuroSpin, CNRS, CEA-SACLAY, F-91191 Gif-sur-Yvette, France
Abstract
Accidental hypoxia has detrimental effects on the brain, particularly on the hippocampal subfields (HS), which are highly sensitive to oxygen deprivation and play a crucial role in episodic memory. This raises the question: could freediving training induce anatomical changes in the HS and lead to significant memory deficits? This study aimed to investigate the impact of a season of freediving training on HS anatomy and episodic memory performance, as freedivers represent a unique natural model for studying the effects of repeated voluntary hypoxic exposure on brain function in healthy individuals. Extending previous research, this study assessed these effects over a prolonged training period.
Seventeen male freedivers were evaluated before and after 7 months of training and compared with a control group of 20 non-freediver athletes. HS anatomical volumes were measured using MRI segmentation and episodic memory performance was evaluated using a pattern separation (PS) task. This task specifically targeted HS-related memory processes by distinguishing between three types of items: (i) identical, (ii) similar, and (iii) new.
No significant differences were observed between freedivers and controls in HS volumes or memory performance, either before or after the 7 month training period. A two-way repeated measures ANOVA revealed that freedivers exhibited the same memory pattern as the control group in the PS task. Specifically, both groups performed better with (i) identical items compared with (ii) similar items (p < 0.001) and were less accurate with (ii) similar items compared with (iii) new items (p < 0.001). This aligns with expectations, as distinguishing similar items from previously presented ones is more cognitively demanding than recognizing new items.
These findings suggest that repeated voluntary hypoxic exposure during freediving training does not impair episodic memory function. Freedivers’ memory performance remained comparable with that of the control group, with no detectable adverse effects on hippocampal anatomy.
Keywords
- episodic memory
- hippocampus
- hypoxia
- diving
- cognition
The hippocampal subfields (HS) within the medial temporal lobe are essential for episodic memory, a form of long-term memory that enables the recall of events in specific spatial and temporal contexts. These subfields, which include Cornu Ammonis (CA) 1, CA2, CA3, and CA4, the dentate gyrus (DG), and the subiculum [1], facilitate episodic memory formation [2] through processes such as pattern separation (PS), which reduces interference between similar events [3]. The DG plays a crucial role in PS, as it supports perceptual discrimination and encoding [4, 5, 6]. Within the DG network, cortical input spike trains are transformed, leading to less similar output spike trains in granule cells; a process further amplified by the CA3 network at the level of single pyramidal cells [7]. Together, these mechanisms allow the HS to balance the retrieval of past experiences while encoding new, yet similar, events.
Another essential function of the HS is its role in neo-neurogenesis, particularly within the DG [8]. This process involves the generation of new neurons, alongside some interneurons and glial cells (e.g., oligodendrocytes or astrocytes). While most newly generated neurons undergo selective pruning, only a small fraction matures and integrates existing networks, thereby maintaining the DG volume. This neurogenic process is essential for encoding new information and supports hippocampal plasticity. Newborn granule cells contribute to PS [9], forming distinct memory traces for overlapping inputs and reducing interference during learning similar items over time, ultimately enhancing memory encoding and retrieval [3, 10, 11, 12].
Involuntary hypoxia, regardless of its cause, significantly affects hippocampal
anatomy and episodic memory. For instance, chronic hypoxia from prolonged
residence at high altitudes induces structural and cellular changes within the
HS, impairing learning and memory functions, and highlighting its vulnerability
to prolonged hypoxia [13]. Similarly, chronic pulmonary diseases such as chronic
obstructive pulmonary disease (COPD), which lead to progressive lung impairment
and chronic hypoxia, are associated with substantial memory deficits. Individuals
with COPD experience significant cognitive impairments, including episodic memory
deficits, which correlate with hippocampal atrophy and reduced hippocampal volume
[14, 15]. Furthermore, obstructive sleep apnea syndrome (OSA), characterized by
frequent breathing interruptions during sleep, leads to intermittent but
prolonged hypoxia. OSA is specifically associated with episodic memory deficits
[16] and is accompanied by cellular and neurophysiological changes in the
hippocampus, contributing to broader cognitive and behavioral impairments [17].
Exposure to extreme altitudes (
These examples illustrate the detrimental effects of involuntary hypoxia on hippocampal function and memory performance, regardless of its cause. In contrast, the impact of voluntary hypoxia in sports remains unclear, with previous studies reporting conflicting results. Allinger et al. (2024) [20] found no adverse effects of hypoxia on memory in freedivers, even following a freediving session. This study assessed episodic memory using a PS task and found no evidence of deficits in individuals with approximately five years of freediving experience. Similarly, an earlier study reported no long-term memory impairments in elite breath-hold divers, using a series of standardized neuropsychological tests evaluating visual and verbal attention and recall tasks abilities [21].
Interestingly, Loprinzi et al. (2019) [22] reported unexpected advantages of mild acute hypoxia on episodic memory, primarily through the upregulation of brain-derived neurotrophic factor (BDNF). BDNF, a crucial factor in memory function, is regulated by hypoxia-inducible factor-1 during acute hypoxic exposure, enhancing synaptic plasticity and, consequently, memory function. This hypoxia-induced BDNF production not only facilitates episodic memory by strengthening synaptic connections but also improves PS by reducing memory interference. Acute hypoxia exposure may thus mitigate retroactive interference, where new information disrupts previously learned content. These cognitive benefits have been linked to mechanisms such as enhanced synaptic plasticity, neurotransmitter modulation, and improved cerebral blood flow, all contributing to optimizing memory and learning processes.
Additionally, the beneficial effects of aerobic physical exercise on memory (e.g., endurance), particularly PS, are well established [23]. Physical exercise enhances the survival rate of newly generated neurons in the DG [24], which are crucial for PS. Notably, endurance sports primarily rely on aerobic capacity, except during intense and prolonged efforts that engage anaerobic energy systems, whereas apnea training focuses on enhancing the body’s adaptability to anaerobic conditions. The combined effects of exercise and hypoxia on memory and cognition reveal a complex interaction between the two factors. Ando et al. (2020) [25] found that cognitive performance seems to be significantly influenced by the interplay between exercise’s beneficial effects and hypoxia’s detrimental effects. The severity of hypoxia emerges as a critical determinant: moderate hypoxia during moderate exercise may enhance cognitive performance, whereas severe hypoxia often impairs tasks requiring memory and attention. These findings highlight the importance of balancing exercise intensity, hypoxia severity, and exposure duration to optimize cognitive outcomes.
Recently, Damgaard et al. (2023) [26] reported potential neuroprotective benefits of moderate hypoxia in brain disorders, with most human studies indicating cognitive improvements. Low-dose hypoxia combined with exercise was associated with enhanced cognitive performance and neuroplasticity, emphasizing the therapeutic potential of this combination. However, higher doses of hypoxia were linked to adverse effects, underscoring the need to carefully calibrate hypoxic exposure to maximize benefits while minimizing risks. Despite the detrimental effects of accidental hypoxia, previous studies on memory performance in freedivers, who voluntarily subject themselves to intermittent hypoxia during prolonged breath-hold activities, have reported mixed findings, ranging from preserved function to deficits. This is why a precise exploration of episodic memory with HS anatomy is necessary to clarify the impact of hypoxic training in this sport. While previous research has primarily focused on cognitive outcomes at a single time point after a freediving performance (maximal performance) in elite freedivers (considering several years of practice), a notable gap remains regarding the effects of a season of freediving training (several months) in recreational athletes. To date, no study has examined cognitive and anatomical changes before and after a structured training period.
The present study aimed to examine the effects of a freediving training season on HS volume and its relationship to performance in the PS task. To this end, 17 male freedivers were recruited during their training season and assessed at the beginning and end of a seven-month training period, which included three training sessions per week. Their results were compared to those of an age-matched control group of non-freediving male athletes. Data were collected at the beginning (Ft0) and at the end (Ft1) of the freediving training season. We employed Hippocampal Segmentation Factory (HSF© 4.0.0 CEA Saclay, Gif-sur-Yvette, France), an automatic MRI segmentation tool of the HS [27], and a PS task, HippoPS [28], to assess episodic memory. Given the role of the DG in neo-neurogenesis and its neuroprotective function for episodic memory, we hypothesized that freedivers would exhibit changes in the volume of DG and comparable PS scores to those of the control group between the beginning and the end of the training season, as no memory deficits have been previously reported in this population.
Data acquisition was performed following the Good Clinical Practice guidelines and approved by an appropriate Ethical Committee (2022-A01030-43). Written informed consent was obtained from each participant. Forty-seven male volunteers participated in this study, including 17 freedivers (FD) and 20 controls (NC) aged 27–55 years and recruited between 2022 and 2024 in the pools of Ile de France region (Paris, Yvelines, Hauts de Seine, France). A schematic of our protocol is shown in Fig. 1.
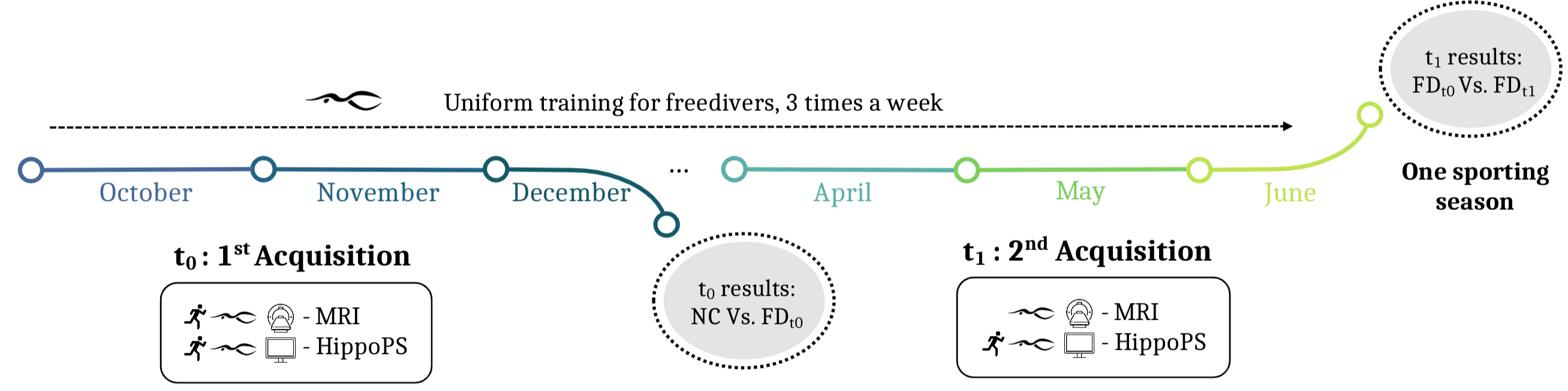 Fig. 1.
Fig. 1.
Acquisitions calendar over one sporting season. 3T anatomical MRI and cognitive tests (HippoPS task) before and after homogeneous freediving training 3 times a week, including 1 hour 30 minutes of training for each session, and for non-freediver sport controls. FD, freedivers; NC, controls; MRI, magnetic resonance imaging; PS, pattern separation.
The FD selected for this study had prior experience with freediving as a hobby but were not professional athletes. After inclusion, FD participants underwent seven months of intense, regular, and uniform hypoxia training [29], including swimming pool sessions three times per week. Training sessions were made available through an application, where participants logged their performance immediately after completion or noted any exceptional absence. Swimming pool sessions lasted 1 hour and 30 minutes and consisted of repeated apnea series of varying durations interspersed with recovery periods of similar lengths. Typically, with bifins, 50-meter apnea series, used for hypercapnia training, lasted around 45–50 seconds with short recovery periods, while longer-distance apnea series, such as 100 meters (approximately 90–100 seconds), included a few minutes without breathing, followed by equivalent recovery periods. Throughout the season, training progressed from general physical preparation to more specific hypoxia exposure. By the end of the training period, all freedivers were able to complete dynamic apnea swims of 100 meters using fins, ensuring their physiological adaptation to hypoxia. All participants were members of a freediving club, ensuring consistent training throughout the year. Data collection was conducted at two time points: the first at the beginning of the training season (Ft0) and the second at the end of the training season (Ft1).
Age- and sex-matched controls with similar socio-cultural backgrounds were assessed under similar conditions (Table 1). These controls did not participate in freediving but maintained a similar weekly training load (~5 hours per week) through physical activities such as swimming, running, or cycling (aerobic sports). Control participants underwent a single anatomical MRI assessment at t0 and completed the memory task at t0 and t1.
| Characteristics | Freedivers (FD) | Controls (NC) |
| n | 17 | 20 |
| Sex | M | M |
| Age* | 47.9 |
44.8 |
| Years of study* | 16.9 |
16.5 |
| Training load |
* Data are means
Cerebral MRI was performed at 3T (VE11, 3T PRIMA Fit Siemens Healthineers, Erlangen, Germany)
including the acquisition of T1 and high-resolution T2-weighted images of the HS.
T1 parameters were Repetition Time (TR) = 2300 ms, Echo Time (TE) = 3.05 ms, 0.9
The HS were automatically segmented using the Hippocampal Segmentation Factory (HSF©) software, a segmentation tool using deep learning. Starting with a preprocessing step where MRI scans are prepared for segmentation, the HSF© pipeline entails a standalone tool, ROILoc, to extract the left and right hippocampi from the raw T2 MRI. The hippocampi are cropped with a safety margin and normalized (Z-normalization). This preprocessing step also registers MRI data to a standardized template, enabling consistent localization of hippocampal regions. Additionally, the system incorporates test-time augmentation, generating multiple versions of each HS through flips, elastic deformations, and affine transformations, which are aggregated for final segmentation.
HSF© produces voxel-wise segmentation and uncertainty maps, assigning confidence levels to each voxel. It segments the major HS, including the DG, essential for PS processes; the CA1, CA2, CA3, and the subiculum, crucial regions in episodic memory.
This approach addresses challenges in segmenting every type of HS by leveraging high-quality datasets and an efficient training process. HSF© achieves results closer to manual segmentations than existing tools, ensuring accuracy and reproducibility while significantly reducing processing time. More details about this tool can be found at https://hsf.readthedocs.io/en/latest/ [27].
We assessed fluid intelligence using the Raven’s Progressive Matrices task [30], which evaluates logical reasoning and problem-solving skills. The task consists of five series (A, B, C, D, E) of twelve numbered items, with increasing difficulty. Participants were required to identify the missing piece to complete the logical sequence. A total score was computed to approximate the participant’s intellectual capacity and subsequently compared to age- and socio-culturally adjusted normative data [31].
To assess memory span, we used the digit span task, comprising two components: the forward span, measuring short-term memory, and the backward span, which requires more manipulation and visualization of numbers and measures working memory. Participants listened to a sequence of numbers and repeated it correctly. Sequence length increased progressively following each correct response. Each level included two trials with different sequences, and the task concluded when the participant failed both trials at a given level. The participant’s span was defined as the level at which they made two consecutive errors. Performance was compared against normative data stratified by age [32].
The global intelligence scores indicated no cognitive deficits in either cohort (NC and FD), and the two populations had similar scores (Table 2) [31, 32].
| Tasks | Freedivers (FD) | Controls (NC) | |
| Raven task (percentiles)* | 88.9 |
87.8 | |
| Memory span (score)* | |||
| Forwards | 7.1 |
7.4 | |
| Backwards | 6.4 |
5.9 | |
*Data are means
Episodic memory was assessed using the HippoPS task [28] (Fig. 2), a validated ecological tool specifically designed to study PS. This task is particularly suited for evaluating episodic memory, as it provides a more precise assessment of its defining characteristics compared to methods commonly described in literature.
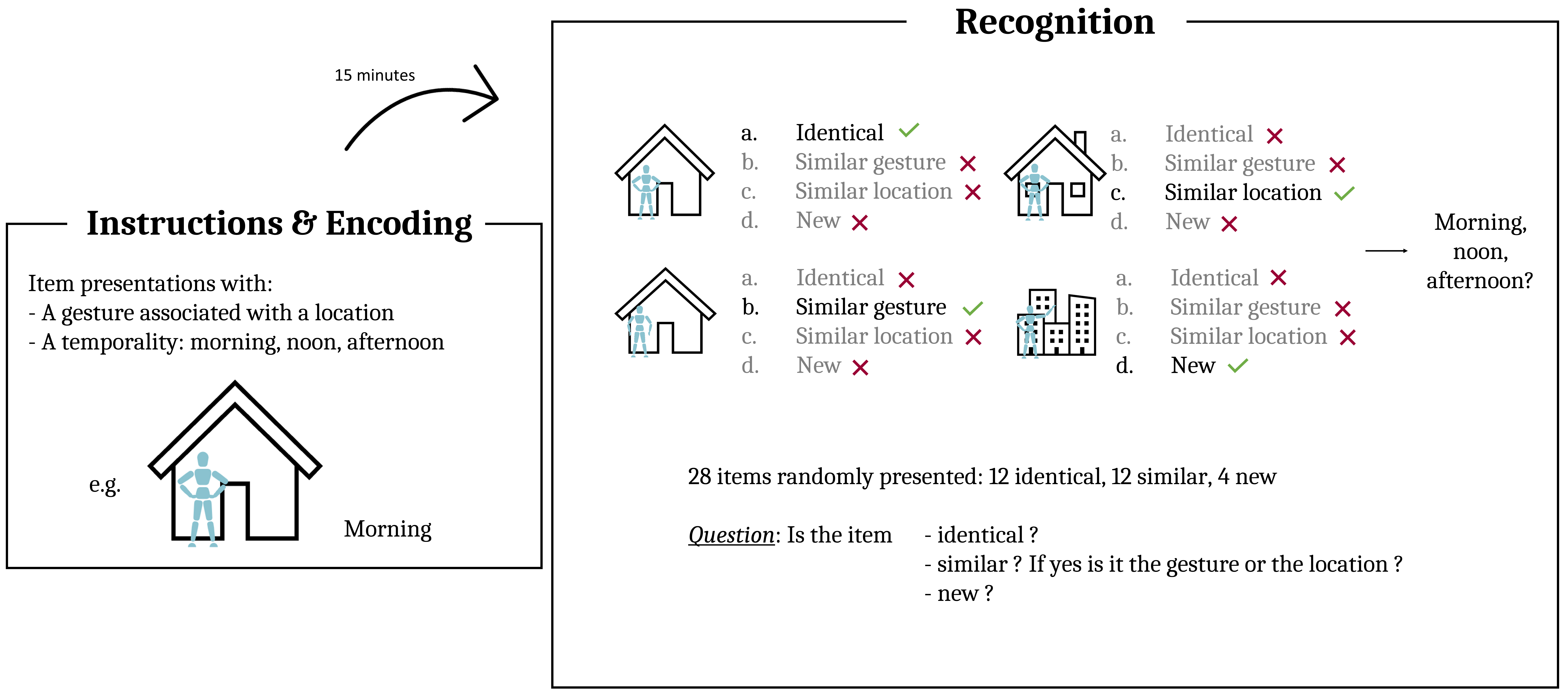 Fig. 2.
Fig. 2.
Encoding and recollection phase of the HippoPS task. The encoding phase involves presenting images of a location paired with a gesture and a temporal context (morning, noon, or afternoon). The recognition phase takes place 15 minutes later and consists of 28 questions: is the item (i) identical? (ii) similar? (iii) new?
The experiment consists of two parts: the encoding phase and, after 15 minutes delay, the recognition phase. The experiment begins with an instruction: “Lisa is a foreign tourist coming to Paris for the first time. She wants to show you around and associate a different gesture for each place she has visited. Watch the entire video carefully so that you can discuss it with her afterward. Do you recognize these places? Have you seen these gestures before?”.
In the encoding phase, participants watch a series of items presented on video, each comprising an image of a location associated with a gesture and a temporal context (morning, noon, afternoon). After 15 minutes, the recognition phase begins, with an instruction as a transition, asking the participant to distinguish photos, with the question: (i) is it an event with the same gesture and the same location (‘identical’), or (ii) is it an event with a different location but the same gesture or an event with the same location but a different gesture (‘similar location’ or ‘similar gesture’), or (iii) is it an event with a different gesture and a different location (‘new’)?
Four examples are provided to ensure that the instructions are clearly understood. 28 items were randomly presented, 12 of which were ‘identical’ events, 12 ‘similar’ events (i.e., 6 ‘similar location’ and 6 ‘similar gesture’), and 4 ‘new’ events. The results are calculated in percentage for (i) identical, (ii) similar, and (iii) new items.
All statistical analyses were conducted using R Statistical Software 4.4.2 (https://cran.r-project.org/src/base/R-4/) and JASP Team (2024 0.19.1, https://jasp-stats.org/previous-versions/), with significance set at a 0.05 threshold. The volumes of the whole hippocampus and each HS were analyzed separately. Due to deviations from normality, a Mann-Whitney test was employed to NC and FD at Ft0 (pre-training), and NC and FD at Ft1 (post-training). Within the FD group, comparisons between Ft0 and Ft1 were performed using a Wilcoxon signed-rank test. Equality of variances was assessed using Levene’s test, and an False Discovery Rate (FDR) correction was applied to account for multiple volume comparisons. Episodic memory (EM) scores were analyzed using a two-way repeated measures ANOVA. The factors included Group effect (FD vs. NC), Item effect (3 scores: (i) identical, (ii) similar, and (iii) new items), and Training effect for FD (FDt0 vs. FDt1). Between-group contrasts (FD vs. NC) and within-group contrasts (FDt0 vs. FDt1) were assessed. Levene’s test confirmed the equality of variances. However, Mauchly’s test revealed a violation of sphericity; therefore, degrees of freedom were corrected using Greenhouse-Geisser estimates of sphericity where necessary.
No significant differences were observed between the left and right hippocampal
volumes; therefore, the two hippocampi were pooled for each subject (FDt0 p = 0.455; FDt1 p = 0.567; NC p = 0.250). At the
beginning of training (t₀), the FD and NC groups showed comparable hippocampal
anatomy, regardless of the HS volume considered (NC (M = 3.69, SD = 0.37)
At the end of the training season (t1), no significant differences were
observed either within the freediver group or between freedivers and controls,
whether in specific HS regions or the whole hippocampus (FDt0 (M = 3.67, SD
= 0.31)
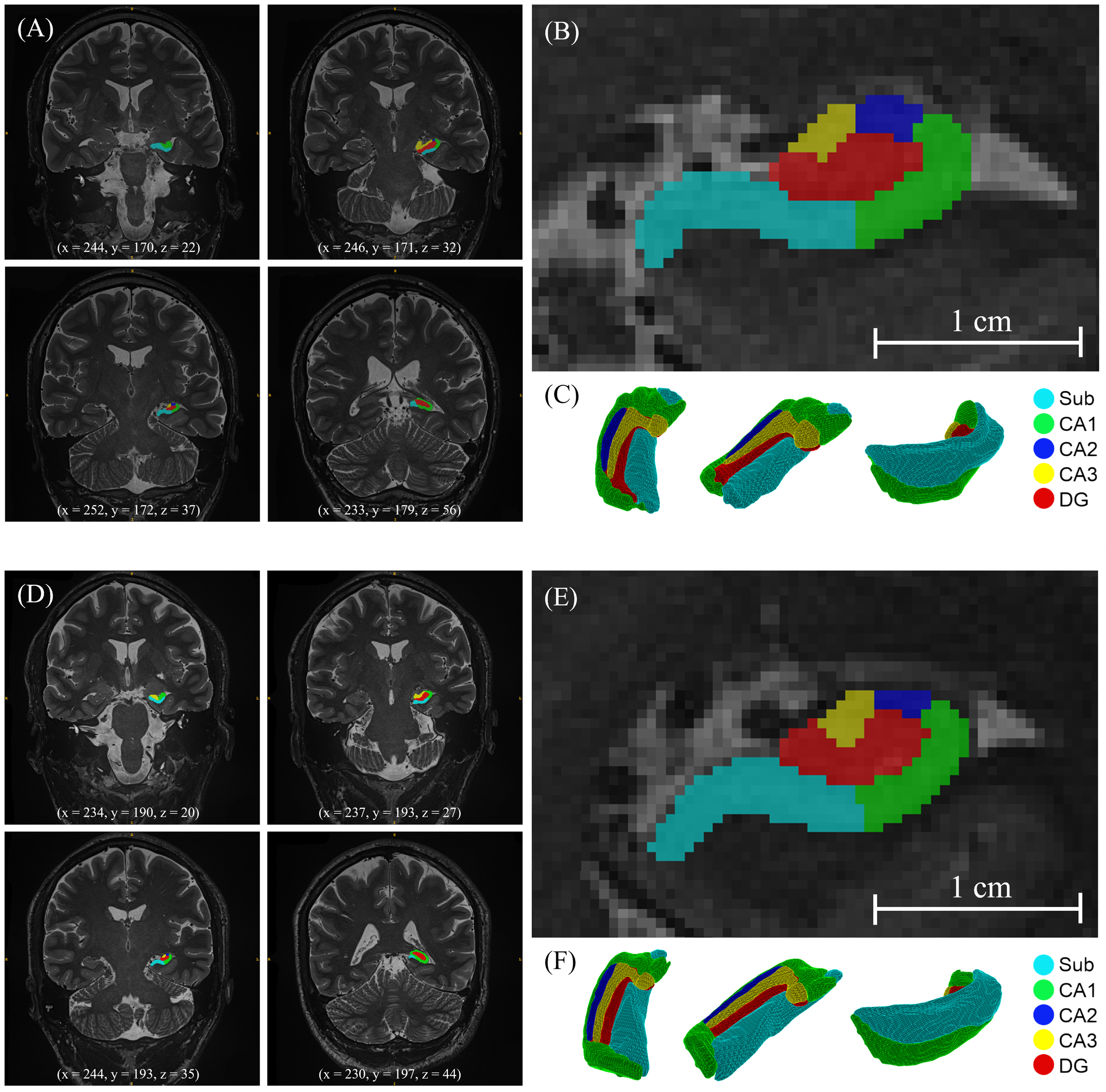 Fig. 3.
Fig. 3.
Automatic HS segmentation of an NC (A–C) and FD (D–F) obtained with HSF©: dentate gyrus, CA1, CA2, CA3 regions and subiculum. (A,D) Coronal MRI slices at MNI coordinates (B,E) Zoom of a coronal MRI slice at MNI coordinates (x = 244, y = 193, z = 35) (C,F) 3D volume rendering. HS, hippocampal subfields; HSF©, Hippocampal Segmentation Factory; DG, dentate gyrus; CA, Cornu Ammonis; Sub, subiculum; MNI, Montreal Neurological Institute.
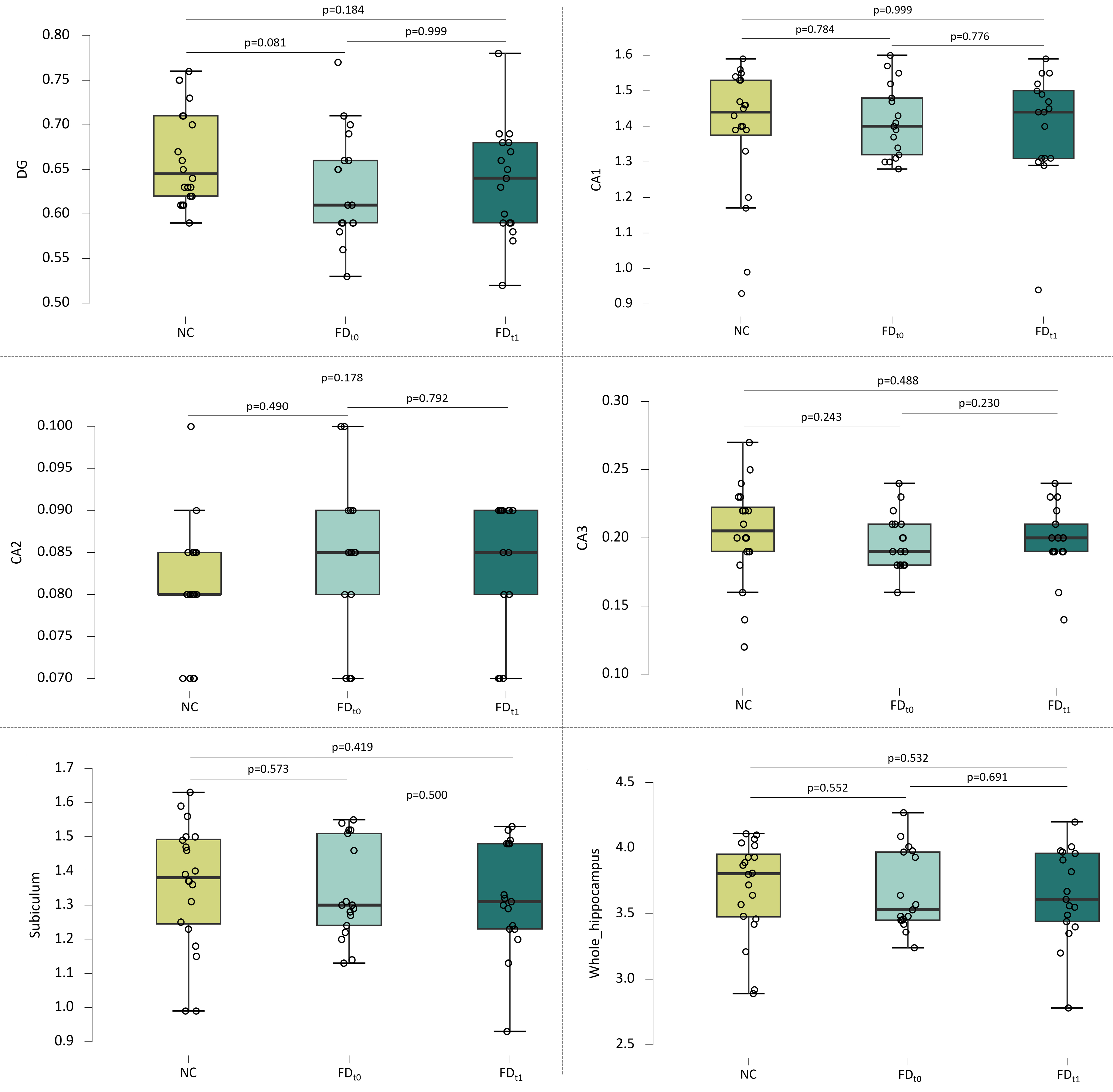 Fig. 4.
Fig. 4.
HS volumes in cm3 in the NC group before training and the FD group before and after training. p-values are the Mann-Whitney test and the Wilcoxon signed-rank test with FDR correction. Mean and standard deviation are indicated. FDR, False Discovery Rate.
In addition, there was no change in the variability whether examining specific HS volumes or the whole hippocampus before (t₀) and after (t1) training (Fig. 4).
The Group effect was not significant before (t0) or after training
(t1), indicating comparable scores between FD and NC (p = 0.397,
The Training effect was significant (p = 0.002,
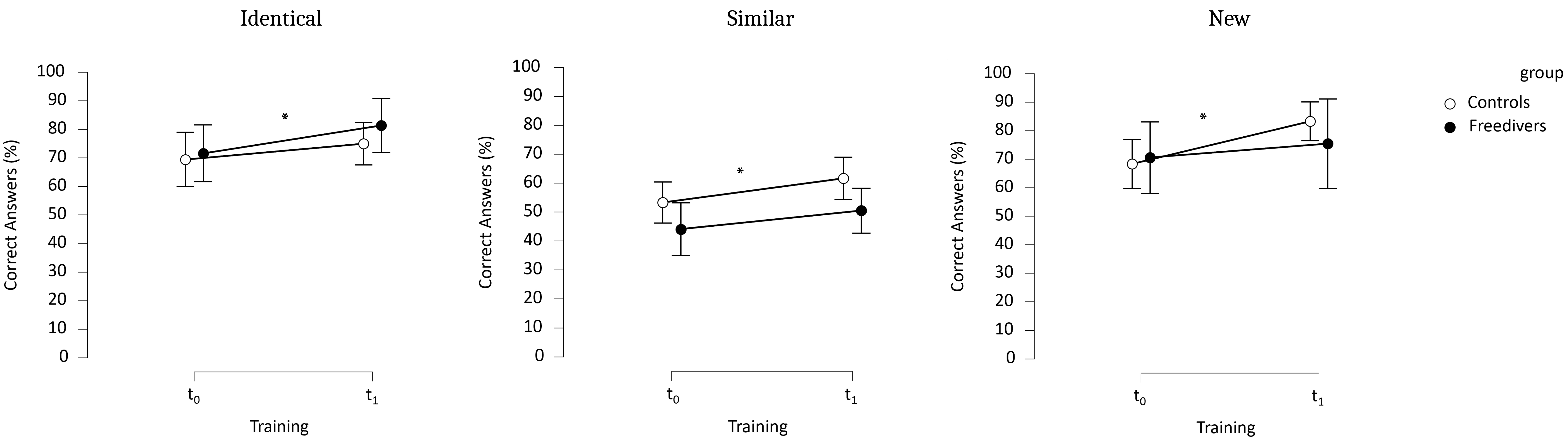 Fig. 5.
Fig. 5.
Correct answers in % in the PS task before and after training, for items (i) identical, (ii) similar, (iii) new in both groups. *, significant differences between t0 and t1.
This study aimed to determine the impact of intermittent voluntary hypoxia through seven months of freediving training on HS volumes using MRI automatic segmentation by HSF© [27], and episodic memory scores using a sensitive ecological PS task [28].
Our results showed (1) no significant differences in HS volumes between freedivers and controls or between freedivers before and after training, regardless of the specific HS examined; (2) no significant differences in episodic memory performance between freedivers and controls, either before or after training. Both groups demonstrated similar performances in the PS task, with better accuracy for identical items compared to similar ones, and for new items compared to similar ones, showing improved performance at the second assessment. These findings suggest that male freedivers maintain hippocampal volume and function essential for episodic memory despite repeated voluntary exposure to hypoxia.
Unlike studies reporting hippocampal atrophy in response to pathological or environmental hypoxia, such as COPD or high-altitude exposure [13, 14, 16, 18, 19], our results suggest that hippocampal anatomy remains stable in healthy individuals following repeated voluntary hypoxia during freediving training. This highlights the resilience of the hippocampus under controlled hypoxic conditions, warranting further confirmation in larger samples.
Previous studies on accidental hypoxia consistently highlight the vulnerability of the hippocampus to hypoxia, often leading to structural atrophy associated with memory impairments [15, 17]. While many studies have focused on total hippocampal volumes or specific HS such as CA1 or the DG [4, 33], few studies have systematically examined all HS using advanced segmentation methods. Recent improvements in segmentation techniques, such as HSF©, allow detailed analysis of HS, yet their application in hypoxia studies remains limited. By employing such methods, we ensured a comprehensive examination of CA1, CA2, CA3, DG, and subiculum. Despite their sensitivity to oxygen deprivation [13, 19], no significant volume changes were detected across the freediving training period. This stability may reflect limitations in current segmentation approaches, which typically analyze the hippocampal head, body, and tail as separate regions. Further studies should consider more detailed analyses along the anterior-posterior axis or employ a gradient-based approach to better capture subtle adaptations. Furthermore, functional or microstructural changes may precede detectable volumetric changes, necessitating advanced imaging techniques for identification.
Our study also enhances the understanding of episodic memory performance under hypoxic conditions, particularly regarding PS, a process critical for distinguishing overlapping memories. The PS task assessed the hippocampus’s ability to encode unique representations for each event and tested memory consolidation by evaluating participants’ ability to distinguish between identical, similar, and new items after a 15-minute delay. Additionally, the task included associative learning components, linking actions to specific locations, and temporal contexts. Designed with ecologically valid stimuli, this task offers greater relevance to real-life memory functions compared to abstract or artificial cognitive assessments [28, 34].
We observed no significant differences in memory performance between freedivers and controls, either before or after training. However, both groups improved their PS performance post-training, consistent with the established cognitive benefits of regular physical exercise [23]. In addition, regardless of training, both groups were less adept at identifying similar items and better at recognizing identical ones. This is understandable, as distinguishing between similar items is a more complex task than recognizing a previously seen item. Thus, freedivers and controls adopt the same approach to the task. These findings contrast with studies reporting hypoxia-induced memory deficits in clinical populations, such as those with OSA [16] or COPD [14], where PS impairments were linked to hippocampal damage. Unlike these pathological conditions, freedivers who are exposed to controlled, intermittent hypoxia, preserved their memory performance. This aligns with prior studies highlighting the neuroprotective effects of physical exercise and moderate hypoxia [22, 26].
Our results are consistent with the literature on episodic memory in freedivers, involving similar cohorts in terms of the number of participants, freediving experience, age, and years of education. These studies have reported no memory deficits following hypoxemia exposure in freedivers. For instance, research using a 4-item memory task after at least 12 hours without freediving found that all divers performed within one standard deviation of published norms, suggesting no significant impairment despite years of hypoxemia exposure [21]. More recent work investigating PS performance using the Mnemonic Similarity Task (MST) found no decline in memory performance when participants completed the task 20 minutes before and 10 minutes after a freediving session [20], and testing divers 30 minutes post-exercise also reported no significant differences between freedivers and controls [35]. It is important to note that while these studies focused on acute effects, based on data collected during and immediately after performance, our study is the first to provide longitudinal data on hippocampal volume and episodic memory in the context of repeated voluntary hypoxia, allowing us to isolate the effects of freediving training on hippocampal adaptability and cognitive function.
Hippocampal plasticity is modulated by various factors, suggesting an adaptive mechanism that responds to environmental changes. Physical exercise, an intrinsic component of freediving training, plays a crucial role in hippocampal adaptability and episodic memory, promoting neurogenesis through different mechanisms [36, 37, 38]. The combination of hypoxia and physical exercise, as seen in freediving, may offer unique benefits for episodic memory, that exceed those of either factor alone. Moderate hypoxia during physical exercise has been shown to enhance episodic memory through mechanisms such as increased synaptic plasticity and neurotransmitter modulation [24]. However, the extent of hypoxia is critical; while moderate levels improve memory and neuroplasticity, severe hypoxia may impair cognitive functions [25]. The neuroadaptive benefits of freediving, who experience controlled and intermittent hypoxia, likely result from the interplay of these factors, optimizing hippocampal function while avoiding the detrimental effects associated with pathological or accidental hypoxia [39, 40].
While the frequency and duration of apnea episodes in freedivers may resemble those observed in pathological conditions, some variations exist. Both freediving and involuntary hypoxia (e.g., OSA or COPD) expose the brain to repeated oxygen deprivation, a known risk factor for hippocampal atrophy and memory deficits [13, 15]. However, the main distinction in our study lies in the voluntary nature of hypoxia during freediving, combined with regular physical exercise. This controlled exposure, paired with the physiological benefits of exercise, may explain the absence of detectable hippocampal damage or cognitive impairments in freedivers.
A recent animal study [41] suggests that controlled, moderate exposure to intermittent hypoxia can yield significant neuroprotective effects, including enhanced hippocampal angiogenesis and neurogenesis, as well as reduced brain lesion size in neonatal mice, highlighting its potential to mitigate cognitive impairments and brain injuries. Thus, carefully calibrated intermittent hypoxia could be explored as a therapeutic strategy for conditions like hypoxic-ischemic encephalopathy or other neurological disorders, opening new avenues for translational research in human medicine.
While our findings provide valuable insights, several limitations must be
acknowledged. Although the HSF© software proves highly effective,
outperforming other known automatic segmentation models to date (HSF’s dice
coefficient compared to ASHS (p
Our findings strongly suggest that hippocampal function and memory performance remain intact in recreational freedivers following repeated hypoxic exposure, ensuring that freediving, as an increasingly popular sport, does not negatively impact the hippocampus, a brain region particularly sensitive to oxygen deprivation. This raises the intriguing possibility of exploring voluntary, controlled intermittent hypoxia as a non-invasive therapeutic intervention. Whether combined with physical exercise or implemented through other protocols, moderate intermittent hypoxia holds promise as a potential strategy for enhancing neuroplasticity and cognitive resilience.
HS volumes and memory performance remained comparable between freedivers and controls before and after the seven-month training period. These findings suggest that repeated voluntary hypoxic training, as experienced in freediving, does not induce detectable hippocampal damage or memory deficits in recreational athletes. They highlight the potential neuroprotective effects of regular physical exercise, even under hypoxic conditions, and position freediving as a valuable natural model for investigating the impact of intermittent hypoxia on cognitive function. Moreover, this study provides insights into potential therapeutic applications for neurological conditions involving hippocampal dysfunction, such as stroke, perinatal hypoxia, and transient global amnesia. Future research should explore potential functional and metabolic adaptations to further elucidate the neurobiological mechanisms underlying these findings.
DG, dentate gyrus; FD, freedivers; HS, hippocampal subfields; NC, controls; PS, pattern separation.
The datasets referenced in this article are not publicly available, as participants consented to their data being used solely under the supervision of the principal investigator. Requests to access the datasets should be directed to MN.
MN developed the research questions and the design of experiment (PI). The HSF software was developed by CP and MN. The HippoPS task was developed by JZ and MN. FM, FB and MN designed the MRI sequences. CC made substantial contributions to the acquisition of data as the medical investigator and was responsible for overseeing the project’s ethical compliance. Recruitment and data collection were conducted by JM, AEH, MT and MN. Data analyses and results interpretation were led by JM under the supervision of MN. JM and MN drafted the manuscript, ATH contributed to statistical analysis and edited the English language. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was carried out in accordance with the guidelines of the Declaration of Helsinki and the research protocol is adhering to ethical guidelines for research involving humans and was approved by the Ethics Committee (Committee for the Protection of Persons Ile de France II, Ethic Approval Number: 2022-A01030-43). All participants provided signed informed consent.
We would like to warmly thank all the MRI technologists and the entire nursing and medical team for their assistance throughout all phases of the human experimentation process, with a special thanks to Mrs. Carré for her care of the participants.
Julia Micaux received a PhD fellowship from the CEA. This study was funded by the Agence Nationale de la Recherche (ANR-21-CE37-0022 HippoXia).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





