1 Department of Critical Care Medicine, Shenzhen People’s Hospital, The Second Clinical Medical College of Jinan University, 518020 Shenzhen, Guangdong, China
2 Day Surgery Center, The Affiliated Hospital, Southwest Medical University, 646000 Luzhou, Sichuan, China
3 Department of Neurosurgery, The Affiliated Hospital, Southwest Medical University, 646000 Luzhou, Sichuan, China
†These authors contributed equally.
Abstract
Polybutylcyanoacrylate (PBCA) nanoparticles (NPs) were prepared by emulsion polymerization and loaded with an enhanced green fluorescent protein plasmid (pEGFP) encoding human brain-derived neurotrophic factor (BDNF). This study investigated the potential effects of PBCA-pEGFP-BDNF NPs for the treatment of experimental cerebral hemorrhage mouse model animals.
Eight-week-old male mice (30 ± 5 g) were randomly divided into four groups (sham, intracerebral hemorrhage (ICH), ICH+PBCA NPs, and ICH+ PBCA-pEGFP-BDNF NPs; n = 14). An ICH model was constructed by right striatum injection of bacterial collagenase VII. Neurological function was evaluated by modified Garcia score after treatment of ICH mice with PBCA-pEGFP-BDNF NPs. The area of cerebral hematoma was measured and the water content of brain tissues was calculated by the wet/dry ratio method. Finally, immunofluorescence staining was used to detect neuron-specific nuclear protein (NeuN) positive cells around hematomas. Enzyme-linked immunosorbent assay (ELISA), real-time quantitative polymerase chain reaction (qPCR), and western blot were used to detect inflammatory BDNF, nuclear factor kappa-B (NF-κB), interleukin-6 (IL-6), tumor necrosis factor-alpha (TNF-α), and either interleukin-1 beta (IL-1β) mRNA or protein levels.
Treatment with PBCA-pEGFP-BDNF NPs significantly improved neurological function and reduced acute brain edema and neuroinflammation in the mouse model of ICH. qPCR, ELISA, and western blot results showed that PBCA-pEGFP-BDNF NPs increased BDNF expression, inhibited NF-κB signaling pathway activity, and decreased the levels of inflammatory factors (IL-6, TNF-α, IL-1β) when compared with the recombinant plasmid pEGFP-BDNF.
PBCA-pEGFP-BDNF NPs improves neurological function in experimental ICH mice at least in part related to increased BDNF expression and decreased p65 NF-κB signaling axis activation, suggesting that PBCA NPs might be a suitable pEGFP-BDNF-carrying delivery system for ICH treatment.
Keywords
- intracerebral hemorrhage
- polybutylcyanoacrylate nanoparticles
- BDNF, blood-brain barrier
- NF-κB signaling
Intracerebral hemorrhage (ICH) is a common cerebrovascular disease that refers to bleeding caused by the rupture of blood vessels in the parenchyma of the brain in a non-traumatic manner and is characterized by a high mortality and disability rate [1, 2, 3]. The disability rate of patients with ICH is more than 90%, and the mortality rate is more than 30%, which is a heavy social burden [4, 5]. Early surgical treatment to clear hematomas along with neurotrophic drugs is the best treatment method, as it reduces intracranial pressure and controls intracranial infections and both are routine treatment methods for patients with ICH [6]. However, the prognosis for ICH patients remains poor even with aggressive treatment [5, 6]. Additionally, ICH in humans can also be a presenting manifestation of a hematological disease [7]. It is necessary to distinguish ICH caused by hematological diseases from other etiologies of hemorrhagic stroke as they both have different treatment approaches and outcomes.
The inflammatory cascade response secondary to ICH, which leads to neuronal cell dysfunction and even death, is the most critical pathologic factor in secondary brain injury [8, 9]. Recently, neuroinflammation has been regarded an important therapeutic target for improved prognosis of ICH patients, but the presence of the blood-brain barrier (BBB) makes most therapeutic drugs ineffective. While the BBB provides neurons with needed nutrients and oxygen, it protects brain tissue from toxins and pathogens, tightly controlling the microenvironment of the central nervous system (CNS) [10]. However, almost all large molecule drugs and 98% of small molecule drugs as well as nucleotide fragments cannot enter the brain due to the BBB [11, 12]. Therefore, the development of efficient and safe drug delivery systems is significant for the treatment of ICH.
Brain-derived neurotrophic factor (BDNF) is essential for mammalian neuronal
survival, differentiation of synaptic plasticity and regulation of
neuroinflammation [13, 14]. Studies have shown that imbalances in BDNF signaling
and neuroinflammation induced by signals such as NF-
Butylcyanoacrylate (BCA) monomer was purchased from Guangzhou Baiyun Medical Adhensive Co., Ltd. (Guangzhou, China; 6606-65-1). Dextran-70 (D0736000), Tween-80 (P1754) and hexadecyltrimethylammonium bromide (CTAB) (H6269) were obtained from Sigma-Aldrich Company (Saint Louis, MO, USA). Prime STAR Max DNA Polymerase (639241), Hind III (1060A) and Sal I double digestion (1080A), high-purity gel extraction kit (9760) and DNA markers (DL2000) were obtained from Takara Biotechnology Co., Ltd. (Shiga, Japan).
The method for synthesizing PBCA NPs is described in [22]. Briefly, 1% (w/v) Dextran-70 and 0.6% (w/v) Tween-80 were dissolved in deionized water, and the pH was adjusted to 2.5 by adding 1 N HCl (North Weiye Measurement Co., Ltd., Henan, China; BWZ7351). A BCA monomer (100 µL) was pipetted slowly into this solution until the final concentration was 1.0% at 1000 rpm and at room temperature for two hours, adjusted the pH to 6.5 with 0.1 N NaOH (FeiMoBio, Beijing, China; FB30092-500) and stirred for one hour. The NPs were separated by ultracentrifugation at 40,000 rpm for two hours at 4 °C and rinsed with distilled water three times. Thereafter, the NPs were dissolved in 0.25% CTAB at 1000 rpm and room temperature for two hours, at 4 °C, 20,000 rpm ultracentrifugation for one hour, then washed with distilled water three times. Finally, the large aggregates and other impurities were removed by 0.22 µm microfiltration bilayer membrane filtration. The colloid was freeze-dried and stored at –20 °C before use. The morphology of NPs was observed by transmission electron microscopy (Kyky Technology Co., Ltd., Beijing, China) and the surface charge (zeta potential) and PNs particle size determined by the Malvern Zetasizer (ZEN3600, Malvern Panalytical, Malvern, UK) laser diffraction light scattering method.
A synthesized eukaryotic expression vector was created as previously described [23]. Briefly, based on mouse genomic DNA, the DNA fragment was PCR amplified (CFX Opus 96, Bio-Rad Laboratories, Hercules, CA, USA). The amplified DNA fragments were purified, the pEGFP vector and BDNF cDNA fragments were digested with Hind III and Sal I, respectively, and T4 DNA ligase was then used to construct the pEGFP-BDNF eukaryotic expression vector. The pEGFP-BDNF DNA was transformed into competent cells derived from Escherichia coli JM109; the white single colonies were collected, placed into liquid luria-bertani (LB) culture medium, and incubated on a shaking table overnight. The recombinant extraction plasmid, pEGFP-BDNF, was then digested by Hind III and Sal I to verify its accuracy and then detected by DNA automatic sequencing analysis (MGI Tech Co. Ltd., Guangdong, China; MGISEQ-2000). Finally, PBCA NPs and pEGFP-BDNF were mixed at a mass ratio of 9:1 at pH 7.0, then stirred at 20,000 rpm for 30 minutes at 25 °C. The NPs were separated by ultracentrifugation at 20,000 rpm for 20 minutes at 4 °C then rinsed with distilled water three times. The colloid was freeze-dried and stored at 4 °C before use.
Fifty-six healthy male C57BL/6J wild-type mice were obtained from the Laboratory
Animal Center of Southwest Medical University (Sichuan, China). A
collagenase-induced ICH animal model was adopted in this study [24]. The mice
(eight weeks, 30
The neurological function of mice was blind assessed by two
investigators with no knowledge of the experimental group using the modified
Garcia score after ICH modeling and at 24, 48 and 72 hours after treatment [24].
The Garcia score ranged from 3 (severely impaired neurological function) to 18
(normal neurological function). Following anesthesia with
Avertin (100 mg/kg), the mice were euthanized via decapitation. The brains were
then carefully removed and serially sectioned into one mm thick slices using a
vibrating microtome. Consecutive brain slices were photographed, and the ICH
volumes were calculated by multiplying the area of the blood clot in each slice
by the distance between the slices, using Image J software (1.52s version,
National Institutes of Health, Bethesda, MD, USA) for analysis. As described
previously, the wet/dry method was used to assess cerebral edema: (wet weight –
dry weight)/(wet weight)
First, total RNA isolation was performed using TRIzol reagent (Vazyme, Nanjing, China; R401-01). Oligomer (dT) primer and Moloney murine leukemia virus (M-MLV) reverse transcriptase (Thermo Fisher Scientific, Grand Island, NY, USA; 28025013) were then used for transcription. qPCR was performed with Archimed-X6 real-time PCR (Rocgene, Beijing, China) equipment after preparing the reaction solution according to the SYBR Green PCR kit (Accurate Biology, Hunan, China; AG11701). Each 10 µL reaction contained 5 µL SYBR Green mix, 0.5 µM each primer, 1 µL cDNA template, and 3 µL double distilled water. Cycling conditions were: 95 °C for 30 s (initial denaturation), 40 cycles of 95 °C for 5 s and 60 °C for 30 s. GAPDH was used as the reference gene throughout this procedure. Based on mouse genomic DNA, the following primers (Table 1) were then used.
| Gene | Forward | Reverse |
| BDNF | 5′-AGGTCTGACGACGACATCACT-3′ | 5′- CTTCGTTGGGCCGAACCTT-3′ |
| TNF- |
5′-ATGTCTCAGCCTCTTCTCATTC-3′ | 5′-GCTTGTCACTCGAATTTTGAGA-3′ |
| IL-6 | 5′-CTCCCAACAGACCTGTCTATAC-3′ | 5′-CCATTGCACAACTCTTTTCTCA-3′ |
| IL-1 |
5′-TTCAGGCAGGCAGTATCACTCATTG-3′ | 5′-ACACCAGCAGGTTATCATCATCATCC-3′ |
| GAPDH | 5′-GCCAAGGCTGTGGGCAAGGT-3′ | 5′-TCTCCAGGCGGCACGTCAGA-3′ |
BDNF, brain-derived neurotrophic factor.
Specimens were fixed in neutral formalin, embedded in paraffin and then sectioned (5 µm). Sections were dewaxed with Xylene (Aladdin, Shanghai, China; 1330-20-7), endogenous peroxidase activity was blocked with 3% hydrogen peroxide (Aladdin; 7722-84-1) and then washed with DPBS (Thermo Fisher Scientific; 21600069). After blocking with 10% goat serum for one hour, sections were washed, then incubated overnight at 4 °C with either primary antibody rabbit anti-BDNF (Abcam, Cambridge, UK; ab108319, 1:500) or rabbit anti- neuron-specific nuclear protein (NeuN) (Abcam; ab177487, 1:500), washed and then incubated with Alexa Fluor™ 647 donkey anti-rabbit IgG (Abcam; ab150075, 1:500) for one hour. Immunofluorescence images were captured by confocal laser-scanning microscopy (Leica TCS SP5 II, Wetzlar, Germany). Image J software was used to calculate the number of NeuN-positive cells per unit area around the hematoma. Immunohistochemically stained sections were photographed after incubation with 3, 30-diaminobenzidine substrate kit (Beyotime, Shanghai, China; P0202). Image J software was used to calculate the percentage of BDNF positive cells per unit area.
The cerebral tissue of the same hemisphere of ICH were
homogenized in a volume of 10
The cerebral tissue of the same hemisphere of ICH were homogenated and centrifuged at 12,000 g for 30 minutes at 4 °C. A bicinchoninic acid assay protein assay kit (Beyotime; P0010) was used to determine protein concentration. Equal amounts of protein were extracted from each group, separated by 15% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE), and transferred to a polyvinylidene difluoride membrane. The sealed membrane of milk was incubated with primary antibody mouse anti-p65 (Cell Signaling Technology, Danvers, MA, USA; CST6956T, 1:500) and mouse anti-GAPDH (Beyotime; AF0006, 1:2000) at 4 °C overnight, after washing with PBS at pH 8 to eliminate non-specific binding, membranes were incubated with a biotinylated goat anti-mouse IgG (Sangon Biotech, Shanghai, China; D110087, 1:1000) at 37 °C for one hour. Finally, protein bands were displayed by chemiluminescence. To verify the level of phospho-p65 (p-p65) on the same polyvinylidene fluoride membrane, the membrane was stripped after the initial immunoblotting to remove primary and secondary antibodies, and re-incubated with primary antibody rabbit anti-p-p65 (Cell Signaling Technology; CST3033T, 1:500) and a biotinylated goat anti-rabbit IgG (Sangon Biotech; D110058, 1:1000). Finally, the phosphorylation level of p65 was quantified by p-p65/p65.
Numeric variables are given as mean
The plasmid PEGFP-BDNF was cut by Hind III and Sal I double digestion. PBCA NPs
and PBCA–pEGFP-BDNF NPs exhibited monodispersed spheres with uniform particle
size (Fig. 1A). The mean diameters and zeta potentials of the PBCA NPs and
PBCA-pEGFP-BDNF NPs complexes are given in Table 2. Mean diameters and absolute
value of zeta potentials of the PBCA NPs increased significantly after complex
formation with pEGFP-BDNF (p
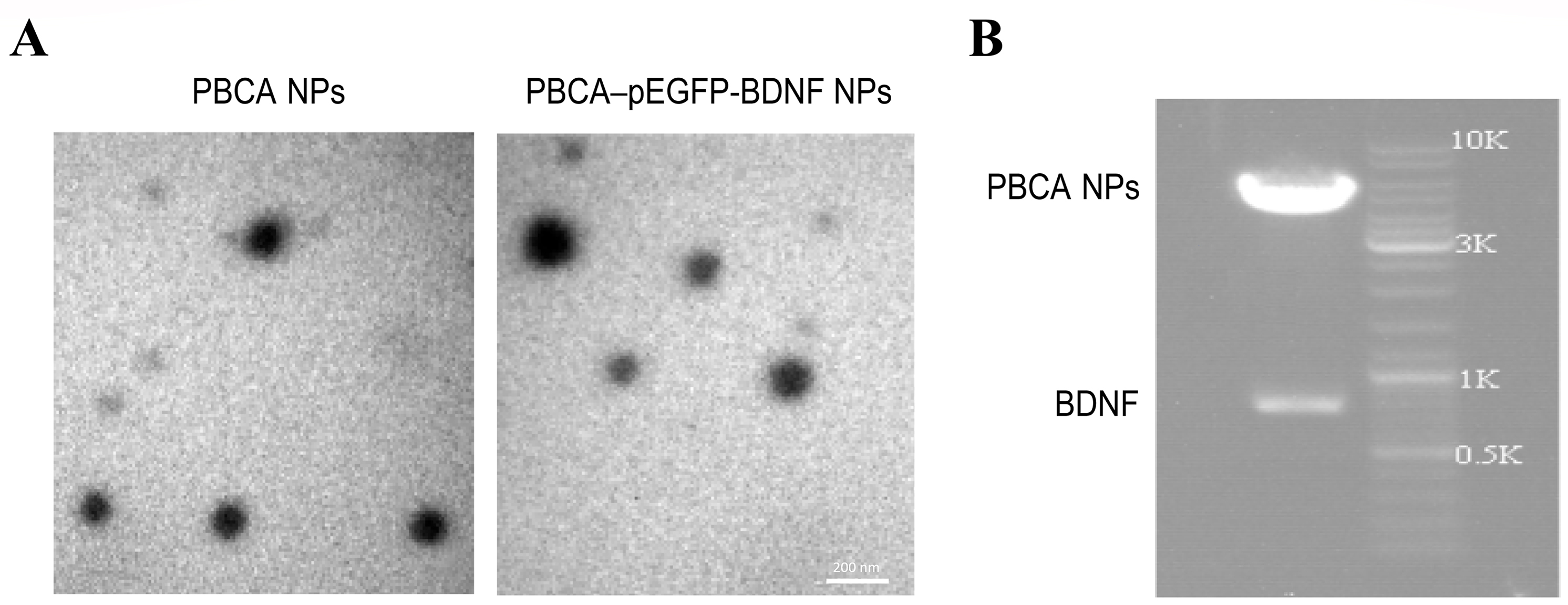 Fig. 1.
Fig. 1.
Preparation and characterization of PBCA-pEGFP-BDNF NPs. (A) Electron micrographs of PBCA NPs and PBCA-pEGFP-BDNF NPs exhibiting monodispersed spheres with uniform particle size. Scale bar: 200 nm. (B) Agarose gel electrophoresis of the product of pEGFP-BDNF complexes after restriction and digestion. PBCA, polybutylcyanoacrylate; pEGFP, enhanced green fluorescent protein plasmid; NPs, nanoparticles.
| Nanocarrier | Mean diameter (nm) | Zeta potential (mV) |
|---|---|---|
| PBCA NPs | 101.1 |
–2.1 |
| PBCA-pEGFP-BDNF NPs | 124.6 |
–16.5 |
Inverted fluorescence microscopy assay confirmed that PBCA-pEGFP-BDNF NPs
treated mice had green fluorescent protein (GFP) expression in their brain
tissues (Fig. 2A). To further illustrate the effect of PBCA-pEGFP-BDNF NPs on
BDNF expression in mouse brain tissues, the mRNA of BDNF was assessed
using qPCR analysis. The mRNA level of BDNF was significantly increased
in brain tissues of mice treated with PBCA-pEGFP-BDNF NPs when compared with PBCA
NPs (p
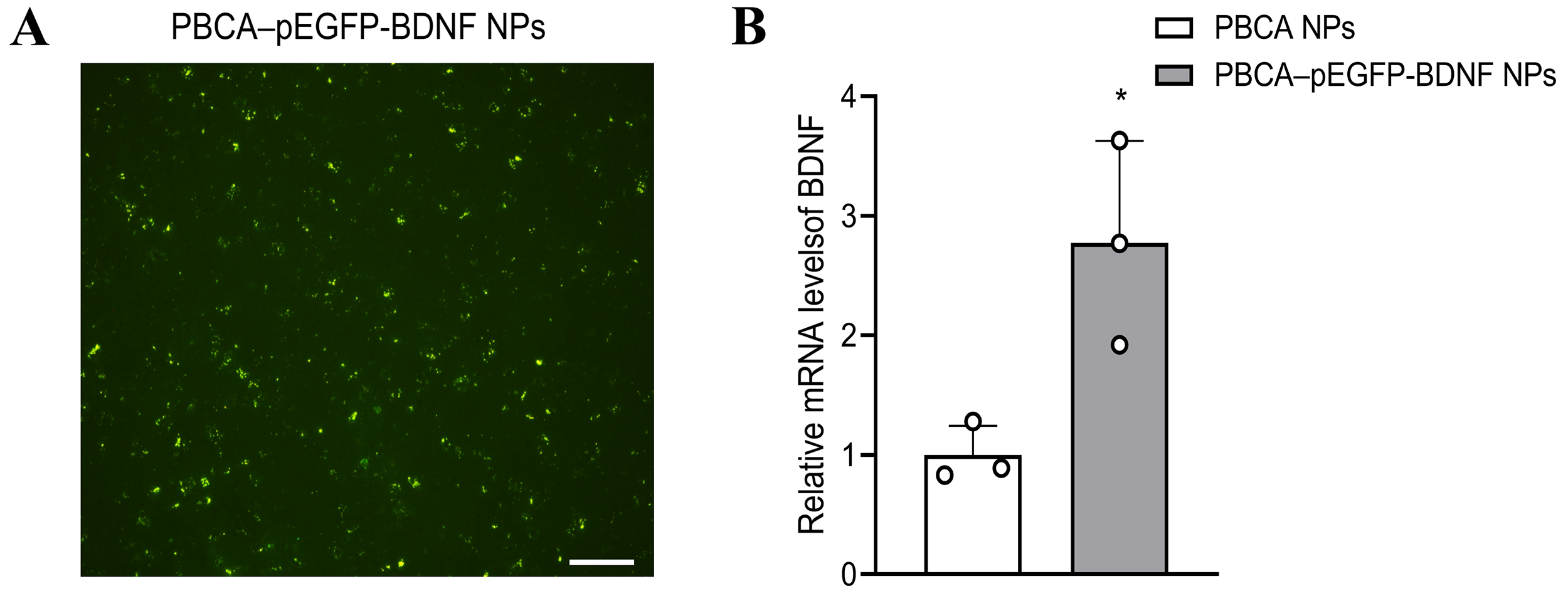 Fig. 2.
Fig. 2.
PBCA-pEGFP-BDNF NPs enter the mouse brain and increase BDNF
transcription. (A) The expression of enhanced green fluorescent protein (EGFP)
was detected by fluorescence microscopy (scale bar: 100 µm). (B) qPCR
analysis of BDNF expression. mRNA of BDNF was increased in
mouse brain tissue after treatment with PBCA-pEGFP-BDNF NPs (n = 3,
*p
Experimental ICH mice exhibited significant neurological
deficits when compared to the sham-operated group (p
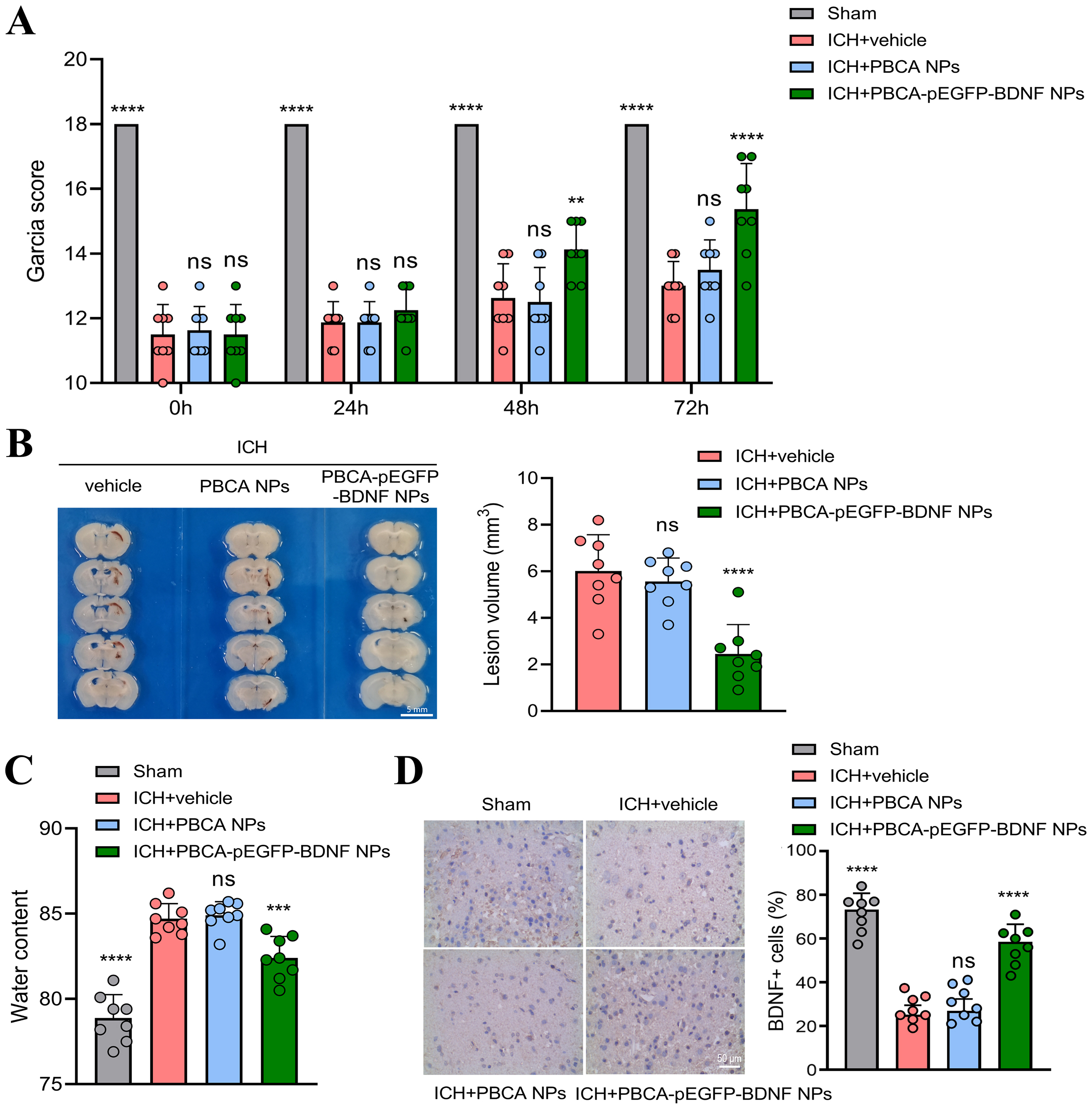 Fig. 3.
Fig. 3.
Effect of PBCA-pEGFP-BDNF NPs treatment on neurologic function
in experimental ICH mice. (A) Garcia scores of mice in each group. (B)
Representative cerebral hemorrhage brain slices and statistical results (scale
bar: 5 mm). (C) Results of quantitative analysis of brain water content. (D) The
level of BDNF was detected by immunohistochemistry and analyzed statistically
(scale bar: 50 µm). (n = 8, “ns” indicates no statistical difference,
**p
Neuronal loss in the brain tissue surrounding hematomas was analyzed.
Immunofluorescence staining revealed a significant decrease in NeuN-positive
cells in ICH mice (192
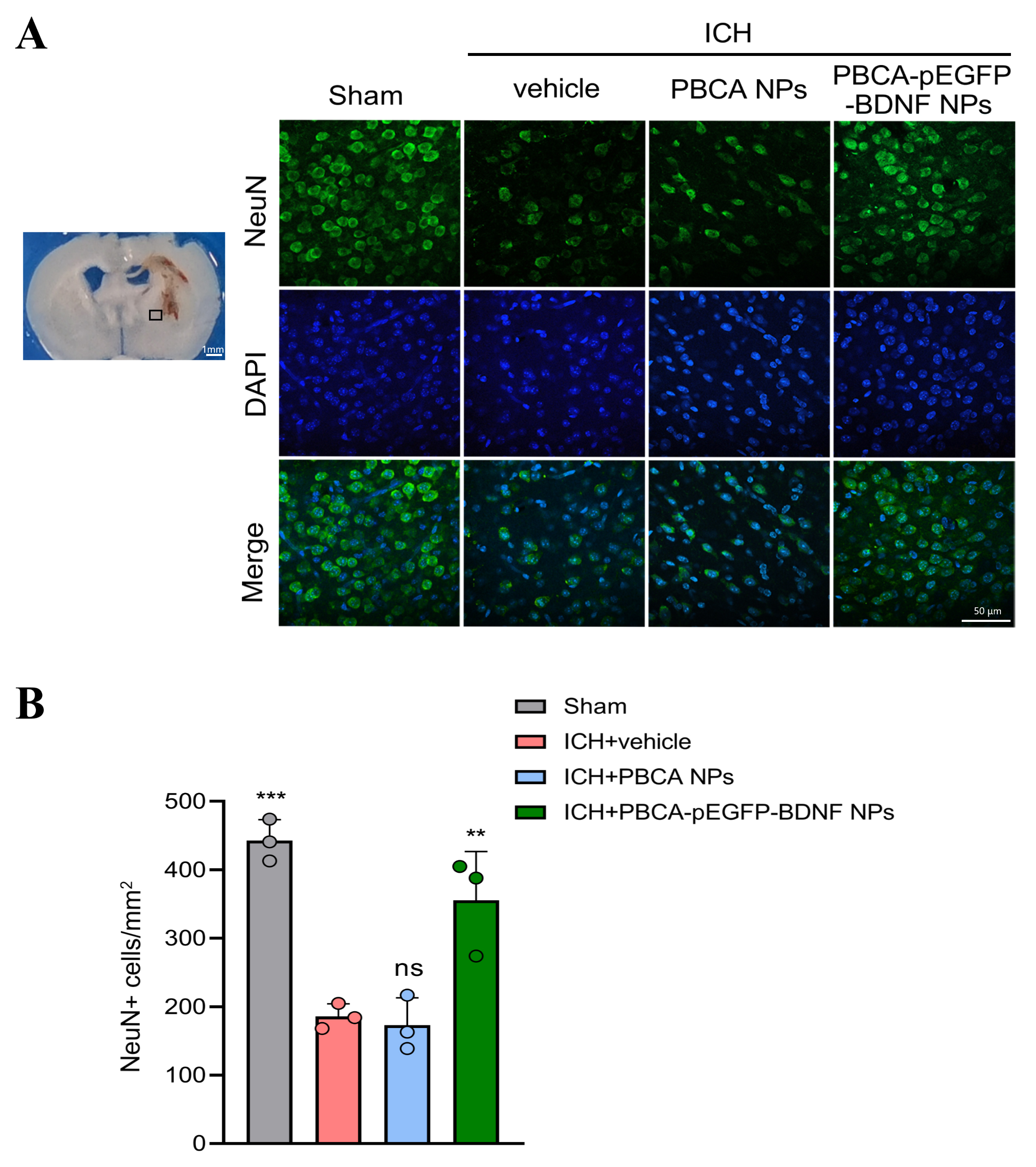 Fig. 4.
Fig. 4.
Effect of treatment with PBCA-pEGFP-BDNF NPs on neuronal loss
due to ICH. (A) Immunofluorescence staining was used to assess the number of
NeuN-positive cells in the tissues surrounding cerebral hematomas in each group
of mice (scale bar: 50 µm). Brain sample with schematic showing the area around the hematoma used for NeuN-positive cell counts (indicated by black squares). (B) Quantitative analysis of NeuN-positive
cells. (n = 3, “ns” indicates no statistical difference, **p
To assess whether the improvement of neurologic function in ICH mice by
treatment with PBCA-pEGFP-BDNF NPs is associated with neuroinflammation, the mRNA
levels and concentration of IL-6, TNF-
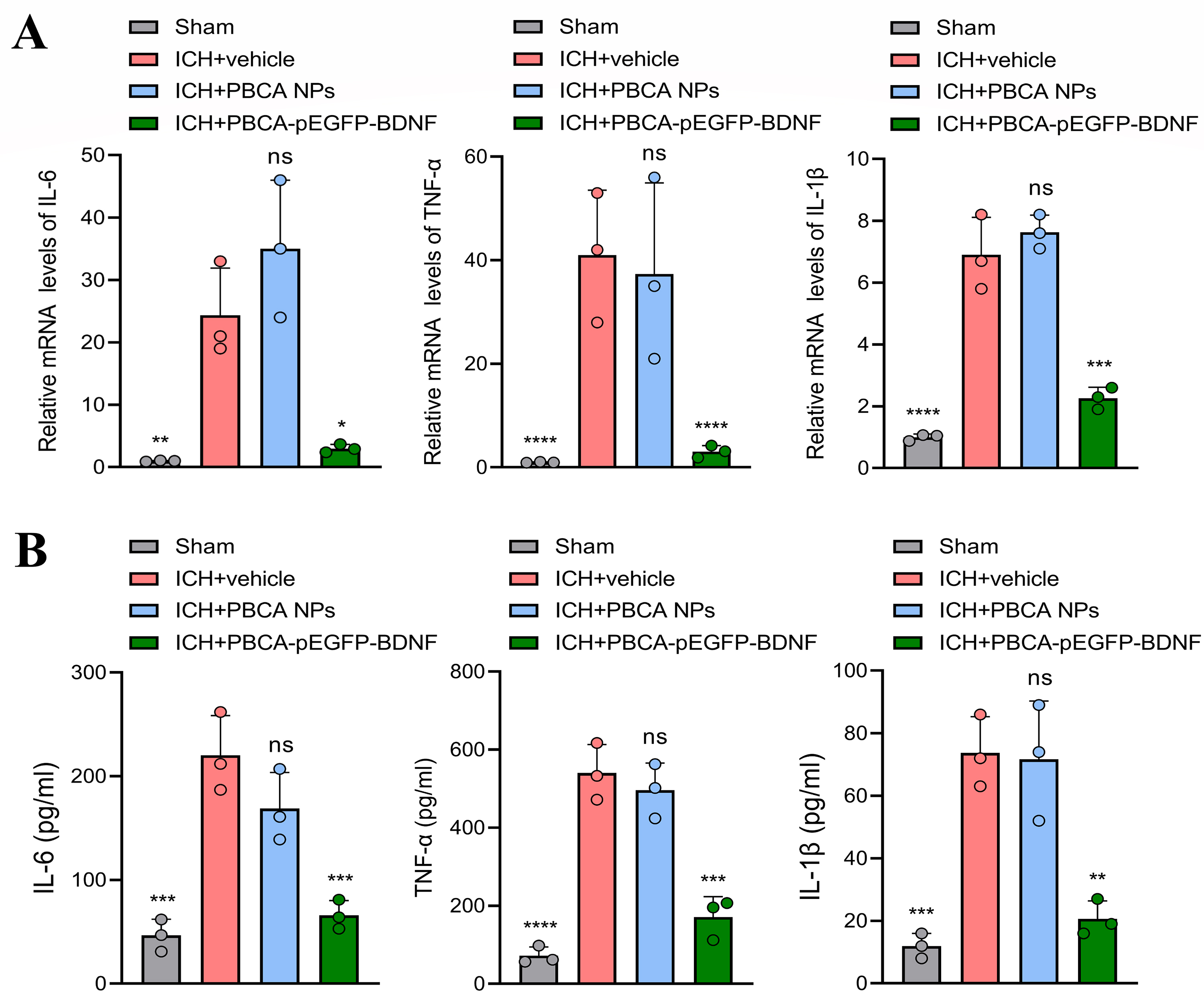 Fig. 5.
Fig. 5.
Effect of treatment with PBCA-pEGFP-BDNF NPs on pro-inflammatory
cytokines. (A) qPCR and (B) ELISA were performed to detect the mRNA and protein
levels of IL-6, TNF-
BDNF is a key factor for neuronal survival and normal function and is
neuroprotective in ICH [25, 26, 27]. High BDNF levels have been reported to inhibit
inflammation levels by modulating p65 NF-
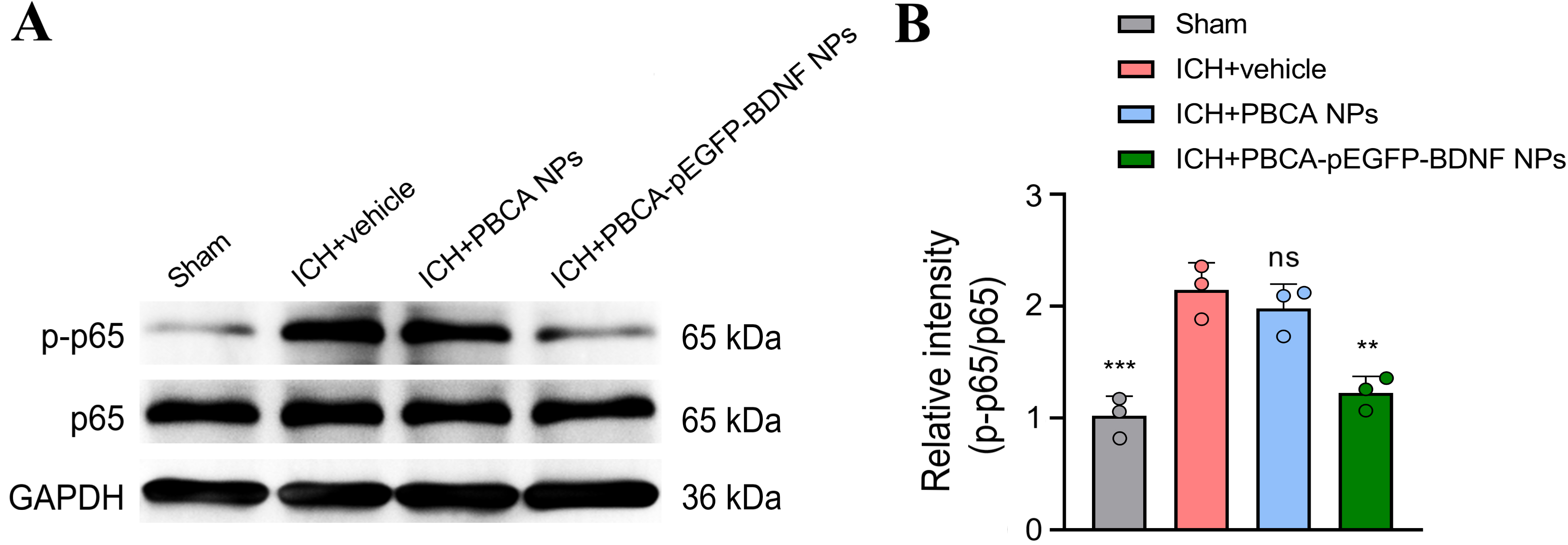 Fig. 6.
Fig. 6.
Effect of treatment with PBCA-pEGFP-BDNF NPs on p65
NF-
ICH is a serious disease in which blood enters the brain parenchyma due to rupture of a cerebral blood vessel, which can be caused by a number of factors (e.g., hypertension, vascular pathology) [29]. Patients with ICH have a high mortality rate, and those who survive often exhibit severe sequelae (mobility problems, emotional abnormalities, cognitive impairment) that affect their quality of life [30, 31]. Recently, the treatment of cerebral hemorrhage has primarily focused on the surgical removal of hematomas. Surgical treatment has somewhat reduced the mortality rate of ICH patients, but does not seem to have had a significant effect on long-term neurological recovery [32]. The pathophysiology of ICH is very complex, and different bleeding sites and etiologies are accompanied by different prognoses. For example, acute spontaneous lobar hemorrhage in humans (the common site of ICH in non-hypertensive mechanisms) has a different clinical profile and a more severe early prognosis than deep subcortical ICH [33]. Neuronal dysfunction and death have been found to be the most critical determinants of patient prognosis, while reduced BDNF and enhanced inflammation are the important causes of secondary injury [34, 35, 36]. In this study, it was found that treatment with PBCA-pEGFP-BDNF NPs significantly improved neurological function, reduced cerebral edema, reversed neuronal loss and suppressed neuroinflammation in experimental ICH mice.
BDNF is essential for maintaining neuronal growth and normal function and has been associated with a variety of CNS disorders including neurodegenerative diseases, cerebral hemorrhage and brain tumors [37, 38, 39]. A wide range of studies have reported that the prognosis of ICH is improved by increased BDNF levels [40, 41]. However, the BBB prevents most drugs from entering the brain to take effect and mitigates the effect of all drugs. The emergence of novel nanocarriers provides a better alternative that not only improves drug penetration through the BBB, but also excels in improving bioavailability, controlling drug release, and reducing side effects [42]. The synthesized PBCA-pEGFP-BDNF NPs by wrapping the DNA fragment of BDNF with PBCA NPs efficiently cross the BBB to reach the intracranial area and is not degraded by nuclease. However, the mechanism by which BDNF reduces blood clot size is unclear and here it is speculated to accelerate hematoma absorption.
Neuroinflammation plays a crucial role during secondary brain injury after ICH
[43]. Products of erythrocyte breakdown generate a strong inflammatory cascade by
activating microglia and astrocytes [44]. Inflammatory cells also release large
amounts of pro-inflammatory cytokines (e.g., IL-6, TNF-
Although NPs such as PBCA have shown promise in enhancing drug delivery across the BBB, their ability to penetrate this barrier may still be limited by factors such as particle size and surface characteristics. Additionally, while PBCA nanoparticles have been approved by the FDA for clinical use due to their biocompatibility and biodegradability, the metabolic breakdown of BDNF after injection poses a further challenge. This degradation reduces the therapeutic efficacy of BDNF before it reaches the intended site of action, necessitating the development of more stable formulations or delivery methods. Furthermore, the potential for neuroinflammation induced by the nanoparticles themselves could counteract the intended neuroprotective effects of BDNF, complicating treatment outcomes.
The current study reveals that PBCA-pEGFP-BDNF NPs promote the expression level
of BDNF in the brain. Treatment with PBCA-pEGFP-BDNF NPs improves neurological
function in experimental ICH mice at least in part related to increased BDNF
expression and decreased p65 NF-
The data during the current study are available from the corresponding author on reasonable request.
XL and YX: Conceptualization, Data curation, Investigation, Formal analysis, Writing—original draft. XG: Data curation, Investigation, Formal analysis, Writing—original draft. CC: Conceptualization, Supervision, Project administration, Writing—review. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study has been approved by the Ethics Review Committee of Southwest Medical University (approval number: 20231013-008). All animal experiments were performed in accordance with relevant guideline for the Care and Use of Laboratory Animals of Southwest Medical University.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/JIN26971.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






