1 Neurological Institute of Jiangxi Province and Department of Neurology, Jiangxi Provincial People’s Hospital, The First Affiliated Hospital of Nanchang Medical College, and Xiangya Hospital of Central South University at Jiangxi, 330038 Nanchang, Jiangxi, China
2 Queen Mary College, Nanchang University, 330031 Nanchang, Jiangxi, China
3 Department of Psychology, Jiangxi Mental Hospital, Nanchang University, 330029 Nanchang, Jiangxi, China
†These authors contributed equally.
Abstract
Sleep disturbance and autonomic dysfunction are often found in Parkinson’s disease (PD) patients, but little is known about changes in cyclic alternating patterns (CAPs) of electroencephalographic (EEG) activities and heart rate variability (HRV) during deep sleep in PD patients.
To investigate changes in EEG activities and HRV during CAPs and non-CAPs (NCAPs) of N3 sleep in PD patients.
Polysomnographic (PSG) examinations were carried out on 18 PD patients and 18 healthy controls, and power spectral analysis of EEG activities and HRV during CAPs and NCAPs (the segment of sleep without CAPs for more than 60 seconds) of N3 sleep were carried out.
The percentages of N3 sleep with CAPs and CAP A1, as well as the CAP A1 index in the PD patients, were significantly smaller compared with the healthy controls. In addition, the power of α waves in NCAPs was significantly higher, while the powers of δ waves in Phase A and B of CAP A1 and A3, and NCAPs were significantly smaller. Furthermore, the durations of total δ waves and δ waves with an amplitude ≥75 μV were significantly shorter, and the low frequency (LF) power of HRV during CAPs and the LF/high frequency (HF) HRV ratio during both CAPs and NCAPs were significantly smaller.
The changes documented in EEG activities and HRV in PD patients during CAPs and NCAPs of N3 sleep compared with healthy controls suggest that N3 sleep quality and sympathetic function are compromised in PD patients.
Keywords
- Parkinson’s disease
- cyclic alternating pattern
- heart rate variability
- α wave intrusion
- non-rapid eye movement sleep
- autonomic nervous system
Parkinson’s disease (PD), one of the most common movement disorders caused by neurodegeneration, affects more than 1% of the population over the age of 60 [1]. As a typical neurodegenerative disease, PD patients often present with motor and also non-motor symptoms. It has been reported that nocturnal sleep disturbances including insomnia, reductions in rapid eye movement (REM) sleep and slow wave sleep (SWS) sleep, REM sleep behavior disorder (RBD), and excessive daytime sleepiness in most PD patients [2, 3]. Additionally, autonomic dysfunction in PD patients often manifests as typical PD symptoms of constipation, cardiac arrhythmias, and orthostatic hypotension, which significantly impact patients’ life quality [4, 5].
A staggering 80% of PD patients might experience cognitive dysfunction over the disease course, which is attributed to the impaired quantity and quality of Non-rapid eye movement sleep stage 3 (N3) sleep [6]. In addition, alterations in N3 sleep are also correlated with a progression of PD motor symptoms [7, 8]. Cyclic alternating patterns (CAPs) are periodic oscillations in non-rapid eye movement (NREM) sleep, comprising high-amplitude slow waves with a mixture of low-amplitude fast waves in Phase A and the background rhythm in Phase B. CAP A1 indicates a stable sleep while subtype A2 and A3 suggests sleep stability might be disrupted, which leads to arousals. The segment of sleep without CAPs longer than 60 seconds is regarded as non-CAP (NCAP), which is characterized by stable electroencephalographic (EEG) rhythm and fewer events associated with arousals [9]. In addition, changes in N3 sleep including a reduction in N3 sleep duration were often reported in PD patients [2, 10] and there were preliminary findings of changes in CAPs during sleep in previous studies [11, 12, 13].
The autonomic system (ANS) plays crucial roles in regulating various activities such as blood pressure, digestion, stress reactions and metabolisms [14], and many non-motor symptoms of PD, such as constipation, orthostatic hypotension, and cardiac arrhythmias in PD patients, are closely associated with autonomic impairments [4, 5, 15]. Heart rate variability (HRV) serves as a non-invasive tool to evaluate the autonomic function and has increasingly gained attentions these days [16]. In patients with stroke, epilepsy, brain infarctions, and PD, HRV was diminished [17, 18, 19]. Further studies showed shorter mean R-R intervals and reduction of sympathetic regulation during sleep in PD patients [20, 21]. However, it is still not elucidated whether the changes in HRV in PD patients were related to CAPs.
Given that EEG activities and HRV might be changed in PD patients during N3 sleep, the aims of this study were to investigate changes in EEG activities and HRV during CAPs and NCAPs of N3 sleep in PD patients.
A total of 46 PD patients with main extrapyramidal symptoms and 54 healthy controls were recruited and clinically examined at the Department of Neurology of the Jiangxi Provincial People’s Hospital between January 2019 to December 2022. The patients were diagnosed based on the UK Parkinson’s Disease Society Brain Bank clinical criteria and graded by Hoehn and Yahr staging (1–3) [22].
Inclusion criteria included: (1) 50–70 years old; (2) apnea-hypopnea index
(AHI)
The healthy controls and patients were excluded if they had the following
conditions: (1) obstructive sleep apnea with AHI
A computer recording system (Pro Fusion 4 system, Compumedics Limited, Abbotsford, Australia) was used to record EEG (O1-M2, O2-M1, C3-M2, C4-M1, F3-M2, F4-M1, sampling rate: 512 Hz), electrocardiographic (ECG, sampling rate: 1024 Hz), and electrooculographic (EOG; sampling rate: 512 Hz) activities. At the same time, electromyographic (EMG; sampling rate: 512 Hz) activities from jaw muscles, bilateral upper (flexors and extensor carpi radialis) and lower limb muscles (tibialis anterior muscle and gastrocnemius), peripheral oxygen saturation (SpO2) in index finger via a finger pulse oximeter (sampling rate: 16 Hz, Nonin Medical Inc, Plymouth, MN, USA), body posture via a position sensor (sampling rate: 16 Hz), snore via a snore sensor (sampling rate: 256 Hz) as well as respiration via nasal thermistor (sampling rate: 32 Hz), pressure transducer (sampling rate: 256 Hz), and thoracoabdominal plethysmography (sampling rate: 32 Hz) were also recorded during sleep overnight as previous studies [23, 24, 25]. Two infrared video recorders, which were focusing on the head and entire body, respectively, were used to monitor and record movements in the mouth, limbs, and other parts of the body during sleep to aid data analysis.
Sleep data were scored in 30 s epochs, and sleep efficiency, total sleep time, durations of NREM sleep stage 1–3 (N1-N3) and REM sleep were scored by one expert clinician and confirmed by another according to the standards established by the American Academy of Sleep Medicine (AASM) [26]. Besides, the sleep bruxism index (SBI; the number of SB events/Hr of sleep), and arousal index, periodic leg movements index (PLMI), as well as mean and minimal SpO2, were also determined as described in previous studies [23, 24]. EEG or ECG recordings were separately exacted at the preprocessing stage and analyzed using different software (see below).
CAP scoring was carried out in accordance with the standards described by Parrino et al. [27, 28]. CAPs were typically observed during NREM sleep, although they could be observed during REM sleep in severe obstructive sleep apnea patients [9, 23]. Each CAP sequence lasted for 2–60 seconds and consisted of phase A (phasic EEG activities) and phase B (background EEG activities). Before the onset of CAP, there must be greater than 60 seconds of continuous NREM sleep without any CAP, except (1) before the first CAP, (2) after the transition from arousal to sleep, and (3) after the transition from REM sleep to NREM sleep. The absence of CAPs in EEG tracings for more than 60 seconds was considered to reflect NCAP [9, 23].
CAPs can be divided into three subtypes, namely CAP A1, A2 and A3 based on the
criteria described by Parrino et al. [27]. Phase A in CAP A1 (Phase A1)
consisted of EEG synchrony (slow waves with high amplitudes, such as
| Parameters | Definitions | |
| CAP index | # of CAPs/Hr of N3 sleep | |
| CAP A1 index | # of CAP A1/Hr of N3 sleep | |
| CAP A2 index | # of CAP A2/Hr of N3 sleep | |
| CAP A3 index | # of CAP A3/Hr of N3 sleep | |
| CAP A1/CAP (%) | CAP A1 as the proportion of total # of CAPs | |
| CAP (A2+A3)/CAP (%) | CAP A2 and A3 as the proportion of total # of CAPs | |
| Total CAP Duration | A sum of all CAPs in duration in N3 sleep | |
| Total NCAP Duration | A sum of all NCAPs in duration in N3 sleep | |
| NCAP/N3 (%) | Total duration of NCAPs as the percentage of N3 sleep time | |
| CAP/N3 (%) | Total duration of CAPs as the percentage of N3 sleep time | |
| Phase A/N3 (%) | Total duration of phase A of CAPs over N3 sleep time in percentage | |
| Phase B/N3 (%) | Total duration of phase B of CAPs over N3 sleep time in percentage | |
| CAP A1/N3 (%) | Total duration of CAP A1 over N3 sleep time in percentage | |
| CAP (A2+A3)/N3 (%) | Total duration of CAPs A2 and A3 over N3 sleep time in percentage | |
| A1/A (%) | Phase A1 duration over total phase A duration in percentage | |
| (A2+A3)/A (%) | Phase A2 and A3 duration of over the total phase A duration in percentage | |
| B1/B (%) | Phase B1 duration over total phase B duration in percentage | |
| (B2+B3)/B (%) | Phase B2 and B3 duration of over total phase B duration in percentage | |
| Phase A1/B1 | Ratio of duration of phase A1 to phase B1 | |
| Phase A2/B2 | Ratio of duration of phase A2 to phase B2 | |
| Phase A3/B3 | Ratio of duration of phase A3 to phase B3 | |
| K complex Index (events/h) | ||
| Index in CAP A1 | # of K complexes in CAP A1/Hr of N3 sleep | |
| Index in CAP A2 | # of K complexes in CAP A2/Hr of N3 sleep | |
| Index in CAP A3 | # of K complexes in CAP A3/Hr of N3 sleep | |
| Index in NCAP | # of K complexes in NCAP/Hr of N3 sleep | |
| Total index | # of K complexes during N3 sleep/Hr of N3 sleep | |
CAP, Cyclic alternating pattern; Hr, Hour; NCAP, Non-cyclic alternating pattern; N3, Non-rapid eye movement sleep stage 3; #, Number.
The EEG data were filtered with a high-pass filter and a low-pass filter (Pro Fusion 4 system, Compumedics Limited, Abbotsford, Australia) set at 0.01 Hz and 35 Hz, respectively, and EEG segments obscured by movements and EMG artifacts were excluded [23]. The power spectral analysis of CAP sequences and NCAPs was based on Fast Fourier transform with 0.25 Hz resolution and cosine window smoothing, and was carried out with the Brainstorm program version 2.0 (GNU GPLv2, McGill University, Montréal, Québec, Canada).
Frontal EEG activities were reported to be more sensitive for evaluation of
changes in EEG activities caused by aging [29], CPAP treatment [29, 30], and
neurodegenerative diseases [31]. In addition, previous study did not find any
significant lateralization of EEG activity [32]. Therefore, EEG activities
recorded from left frontal cortex (F3-A2) were selected for further analysis as
previously described [32, 33, 34]. The power spectral density of frontal EEG
activities associated with N3 sleep was analyzed and the absolute and relative
power of
The R-R intervals were determined by using the software HRV analysis version 1.1 (SNA-EPIS laboratory, Jean Monnet University, Saint-Etienne, France). HRV was analyzed in the frequency domain by using the Fast Fourier transform and power spectral analysis of HRV was carried out using the Welch periodogram algorithm with a Hamming window of 256 points, an overlap of 50%, and an accuracy of 256 points/Hz as described by Pichot et al. [37]. The spectral HRV components were divided into two frequency bands: (1) high frequency (HF) band (0.15 to 0.40 Hz), (2) low frequency (LF) band (0.04 to 0.15 Hz). The normalized spectral indices were defined as LFnu = LF/(LF + HF) and HFnu = HF/(LF + HF) [38]. The LF of HRV mainly reflects sympathetic regulation and the HF of HRV reflected parasympathetic regulation, plus the LF/HF ratio reflects sympathovagal balance [39, 40].
Data were analyzed with the Statistical Package for the Social Sciences (SPSS)
(Version 26, IBM, Armonk, NY, USA). Data with normal distributions in the PD patients
and healthy controls were expressed as mean
A total of 100 subjects were recruited and 64 subjects (28 PD patients and 36
healthy controls) including 18 subjects with insomnia, 43 subjects with
obstructive sleep apnea, and 3 subjects with arrhythmia were excluded. Thus, 18
early-stage PD patients (8 males and 10 females) aged 58–69 (median: 62) with
H-Y staging of 1–3, and 18 healthy controls (9 males and 9 females) aged 55–67
(median: 60) were included in the study. PD patients were under regular
dopaminergic medications (L-dopa/carbidopa, or plus either ropinirole,
pramipexole or entacapone). Daily L-dopa equivalent dose was calculated according
to the previous study [41]. The PD patients had a disease duration of 2.5
Table 2 summarizes demographic data and general sleep parameters of the PD patients and healthy controls. No significant differences in BMI and AHI were found between the two groups. The total sleep time, sleep efficiency, durations of N3 and REM sleep, and the ratio of REM sleep duration to total sleep time in percentage in the PD patients were significantly shorter or smaller than those in the healthy controls while the wakefulness after sleep onset (WASO) and the ratio of N2 sleep duration to total sleep time in the PD patients were significantly longer or higher than those in the healthy controls.
| Variables | PD patients | Healthy controls | p | |
| Age (years) | 62 (58–70) | 60 (55–67) | 0.10 | |
| Male | 8 | 9 | ||
| Female | 10 | 9 | ||
| BMI, Kg/m2 | 22.14 |
23.67 |
0.21 | |
| H-Y stage | 2 (1–3) | |||
| Duration of disease from being diagnosed (years) | 2.5 |
|||
| L-Dopa equivalent dose (mg/day) | 454.7 |
|||
| AHI (events/h) | 2.0 (0.3–8.3) | 2.2 (0.2–7.4) | 0.61 | |
| Mean heart rate during sleep | 62 (53–75) | 65 (57–75) | 0.46 | |
| Total sleep time (min) | 360.7 |
421.3 |
||
| N1 (min) | 27.0 |
36.6 |
0.11 | |
| N2 (min) | 189.4 |
192.0 |
0.87 | |
| N3 (min) | 87.5 |
107.5 |
||
| REM (min) | 56.8 |
85.3 |
||
| N1 (%) | 7.5 |
8.6 |
0.44 | |
| N2 (%) | 52.2 |
45.0 |
||
| N3 (%) | 24.5 |
25.9 |
0.55 | |
| REM (%) | 15.8 |
20.5 |
||
| WASO, min | 67.6 |
38.8 |
||
| Sleep efficiency (%) | 80.3 |
88.5 |
||
| Arousal index | 20.1 |
15.2 |
||
| SBI (events/h) | 2.7 |
3.2 |
0.39 | |
| PLMI (events/h) | 14.9 |
10.7 |
0.33 | |
| Mean SpO2 (%) | 96 (93–98) | 97 (95–98) | 0.25 | |
| Minimal SpO2 (%) | 90 (87–93) | 91 (89–93) | 0.17 | |
Data are expressed as mean
AHI, Apnea-hypopnea index; BMI, Body Mass Index; N1–N3, Non-rapid eye movement sleep stage 1-3; PD, Parkinson’s disease; PLMI, Periodic leg movements index; REM, Rapid eye movement sleep; SBI, Sleep bruxism index; WASO, Wakefulness after sleep onset; SpO2, oxygen saturation of peripheral capillary blood.
As shown in Table 3, CAP index and CAP subtype A1 index in the PD patients were significantly lower, while subtype A2 and A3 indices in the PD patients were significantly higher than those in the healthy controls. In addition, the total CAP duration and CAP/N3 during N3 sleep in the PD patients were significantly shorter or smaller. In contrast, NCAP duration and NCAP/N3 were significantly longer and higher in the PD patients, respectively, than those in healthy controls.
| CAP parameters | PD patients | Healthy controls | p | |
| CAP index (events/h) | 60.84 |
75.39 |
||
| CAP A1 index (events/hr) | 48.37 |
74.34 |
||
| CAP A2 index (events/hr) | 7.25 (1.05–18.00) | 0 (0–1.74) | ||
| CAP A3 index (events/hr) | 2.61 (0.64–11.68) | 0 (0–1.60) | ||
| CAP A1/CAP (%) | 88.56 |
99.08 |
||
| CAP (A2+A3)/CAP (%) | 11.44 |
0.92 |
||
| Total CAP Duration (min) | 52.75 |
81.04 |
||
| Total NCAP Duration (min) | 28.45 |
16.66 |
||
| NCAP/N3 (%) | 32.51 |
15.59 |
||
| CAP/N3 (%) | 59.72 |
74.28 |
||
| Phase A/N3 (%) | 24.34 |
35.46 |
||
| Phase B/N3 (%) | 35.38 |
38.82 |
0.31 | |
| CAP A1/N3 (%) | 47.98 |
73.63 |
||
| CAP (A2+A3)/N3 (%) | 11.74 |
0.64 |
||
| Total Duration (min) | ||||
| Phase A | 21.01 |
38.30 |
||
| Phase A1 | 17.11 |
38.03 |
||
| Phase A2 | 2.64 |
0.12 |
||
| Phase A3 | 1.27 |
0.15 |
||
| Phase B | 31.74 |
42.74 |
||
| Phase B1 | 25.48 |
42.25 |
||
| Phase B2 | 4.73 |
0.26 |
||
| Phase B3 | 1.52 |
0.24 |
||
| Average Duration (s) | ||||
| Phase A1 | 12.66 |
11.87 |
0.49 | |
| Phase A2 | 10.70 |
6.70 |
||
| Phase A3 | 10.35 |
6.04 |
||
| Phase B1 | 22.31 |
18.07 |
0.13 | |
| Phase B2 | 17.40 |
11.93 |
0.24 | |
| Phase B3 | 15.68 |
11.68 |
0.30 | |
| A1/A (%) | 81.16 |
99.29 |
||
| (A2+A3)/A (%) | 18.84 |
0.71 |
||
| B1/B (%) | 78.17 |
98.81 |
||
| (B2+B3)/B (%) | 21.83 |
1.19 |
||
| Phase A/B (%) | 72.13 |
96.54 |
||
| Phase A1/B1 (%) | 77.16 |
97.17 |
||
| Phase A2/B2 (%) | 74.13 |
50.24 |
0.12 | |
| Phase A3/B3 (%) | 81.23 |
66.16 |
0.25 | |
| K complex Index (events/hr) | ||||
| Index in CAP A1 | 16.21 (2.85–41.00) | 20.94 (4.44–33.96) | 0.24 | |
| Index in CAP A2 | 0.75 (0–6.34) | 0.61 (0–2.79) | 0.68 | |
| Index in CAP A3 | 1.64 (0–5.92) | 0.51 (0–3.58) | ||
| Index in NCAP | 5.50 (1.24–19.65) | 2.93 (0–10.80) | 0.10 | |
| Total index | 25.90 (4.59–50.76) | 25.29 (7.27–39.01) | 0.85 | |
Student’s t-test was used to compare data with a normal distribution
(shown as mean
In the PD patients, phase A/N3, phase B/N3 and CAP A1/N3 were significantly smaller while CAP (A2+A3)/N3 was significantly greater than those in the healthy controls. Similarly, in the PD patients, CAP A1/CAP, A1/A and B1/B were significantly smaller and CAP (A2+A3)/CAP, A2+A3)/A and (B2+B3)/B were significantly greater than those in the healthy controls. In addition, phase A2/B2 and phase A3/B3 in the PD patients were significantly greater than those in the healthy controls. Furthermore, the durations of phase A2 and phase A3, but not phase A1, were significantly longer in the PD patients than those in the healthy controls while the durations of phase B1, B2 and B3 were not significantly different between the two groups (Table 3).
The K complex index in CAP A3 was significantly higher in the PD patients, but there were no significant differences in K complex index, K complex index in CAP A1 and A2 as well as K complex index in NCAP of the PD patients compared to the healthy controls (Table 3).
As shown in Fig. 1, in phases A1, B1 and A3, the absolute power of
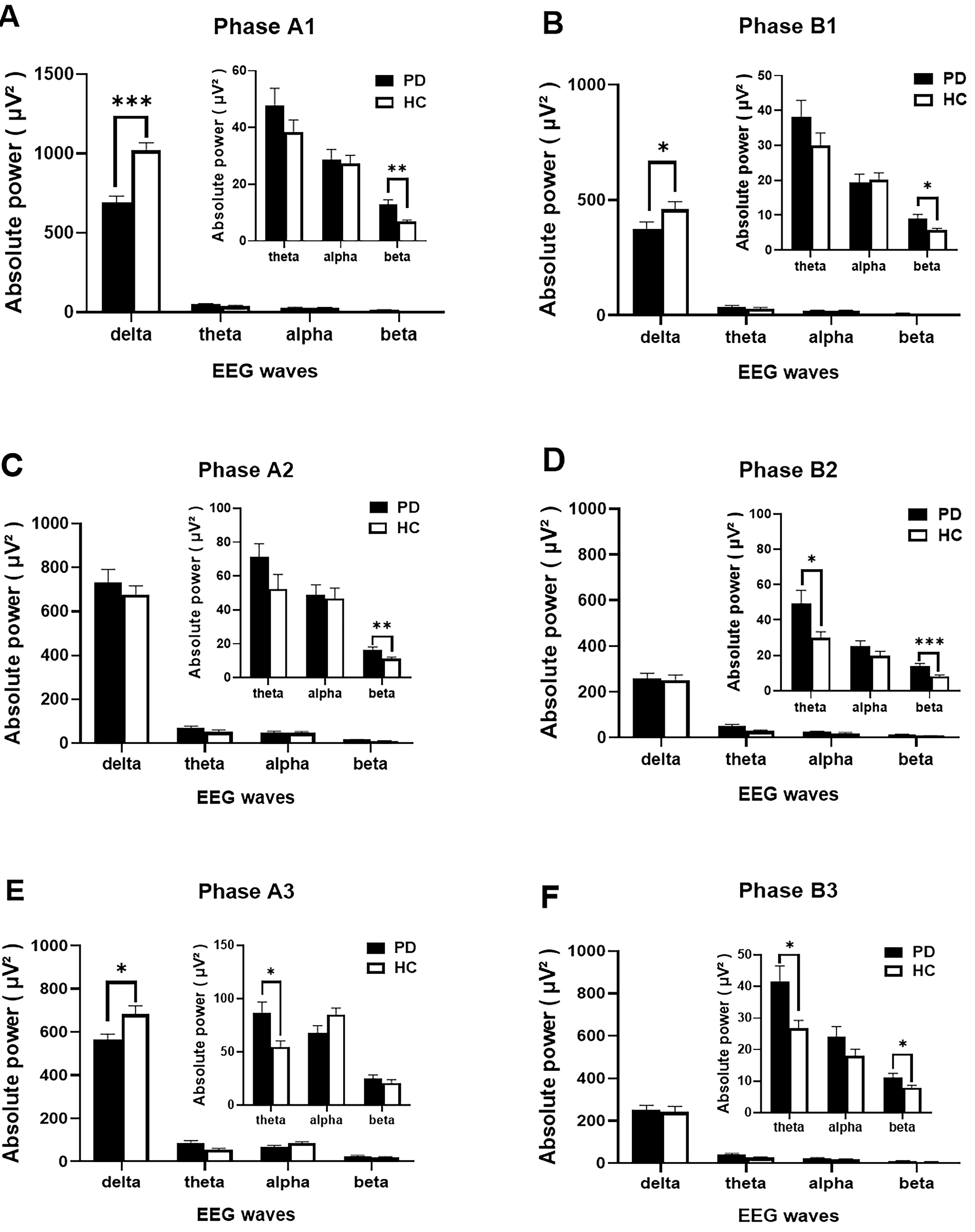 Fig. 1.
Fig. 1.
Comparisons of the absolute power of
The absolute power of
In phases A1, B1–B2 and A3, relative power of
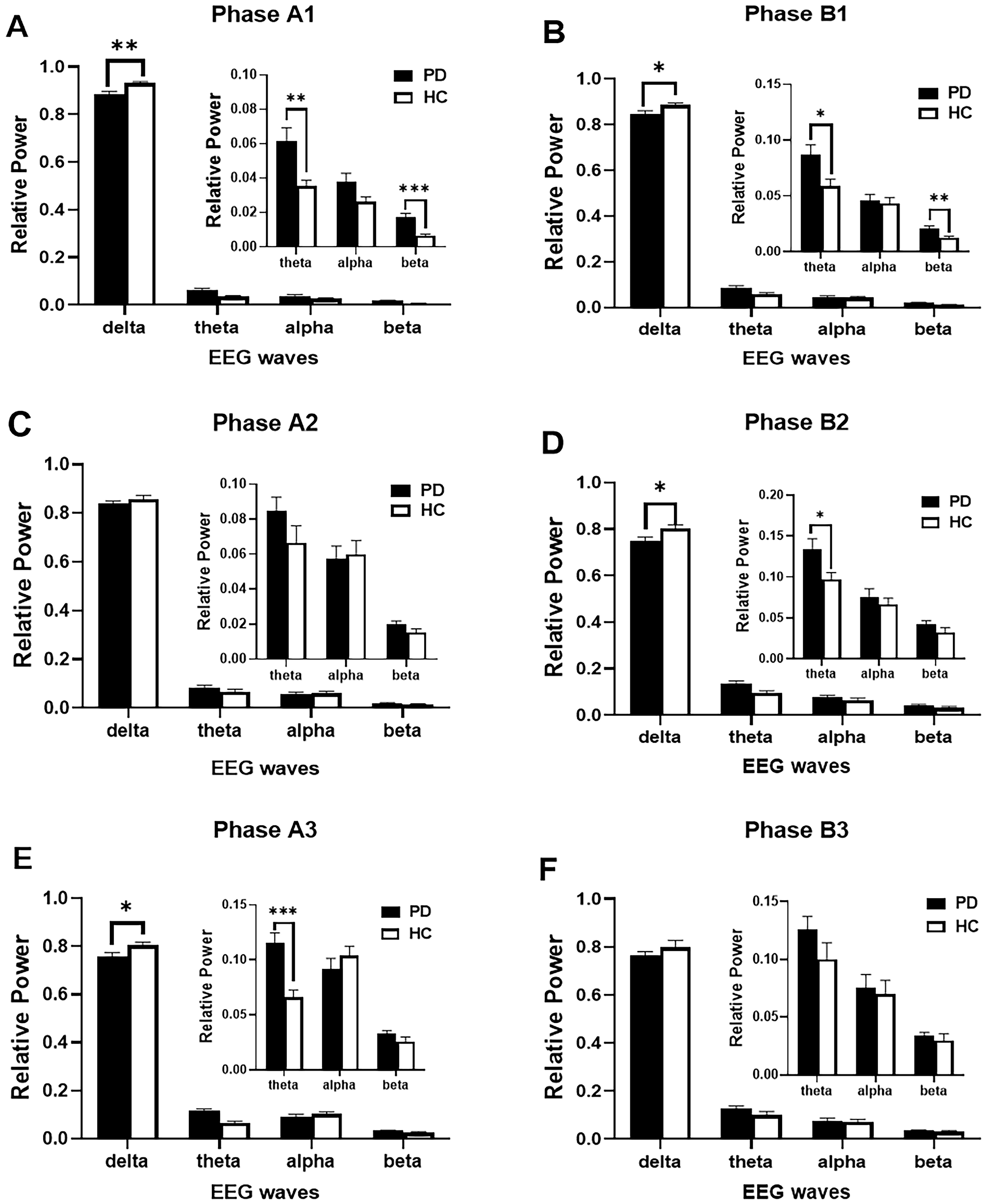 Fig. 2.
Fig. 2.
Comparisons of the relative power of
In the PD patients, the absolute power of
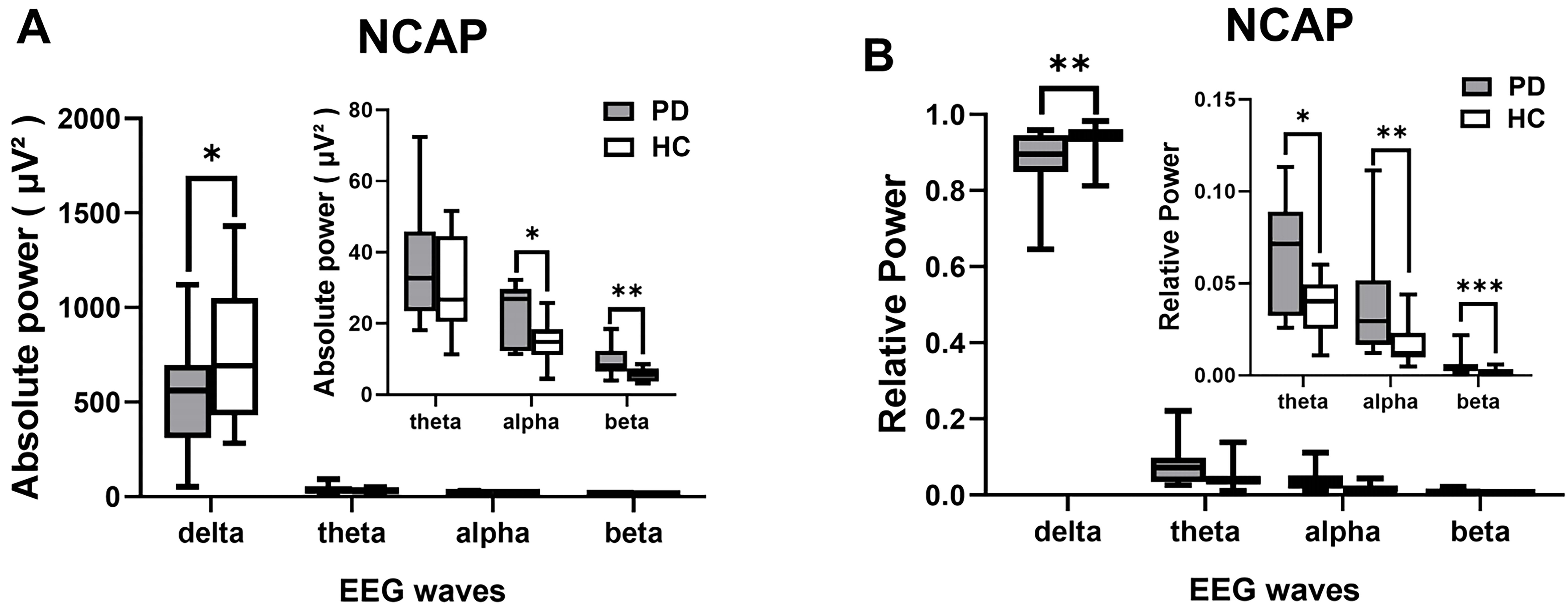 Fig. 3.
Fig. 3.
Comparisons of the absolute (A) and relative power (B) of
As described in the previous study [42],
The total duration of the selected
| Alpha waves | PD patients | Healthy controls | p |
| Total duration, min | 55.3 (20.9–104.7) | 8.5 (2.8–29.3) | |
| N3 sleep with |
62.37 |
14.61 |
|
| Mean frequency, Hz | 10.98 (10.38–11.55) | 10.40 (9.29–10.70) |
Student’s t-test was used for comparisons of data with a normal
distribution (shown as mean
As shown in Fig. 4, the percentage of N3 sleep containing
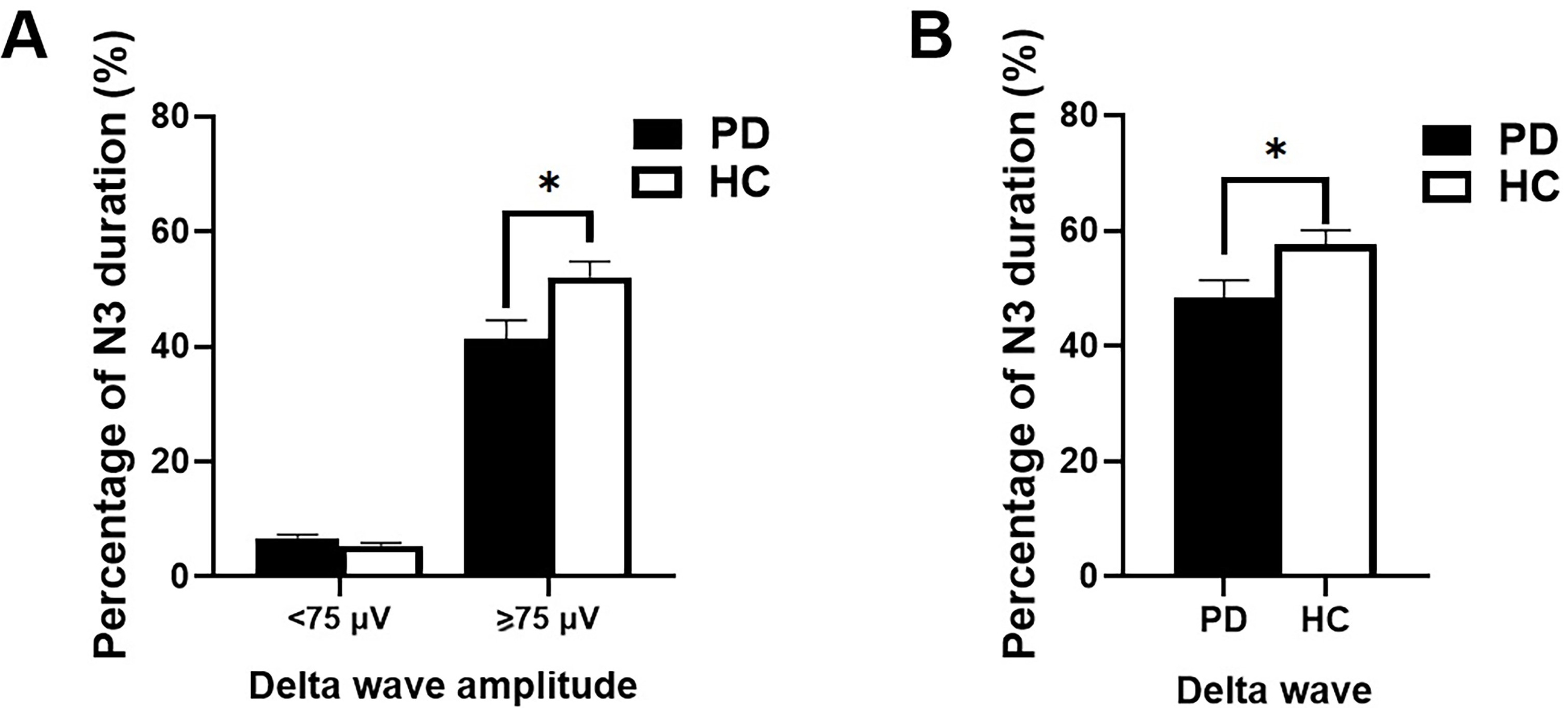 Fig. 4.
Fig. 4.
Percentages of N3 sleep containing
As shown in Table 5, there were no significant differences in RR intervals and HF power of HRV in all CAP subtypes between the PD patients and healthy controls. In contrast, LF power of HRV during CAP A1-A3 in the PD patients was significantly lower than that in the healthy controls. In addition, LFnu and HFnu of HRV during CAP A1 and A2 was significantly lower and higher than those in the healthy controls, respectively. Nevertheless, no significant differences in LFnu and HFnu during CAP A3 were found between the two groups. Furthermore, the LF/HF ratio during CAP A1-A3 in the PD patients was significantly lower than that in the healthy controls.
| Variable | PD patients | Healthy controls | p | ||
| CAP | |||||
| A1 | RR, ms | 971.5 (719.4–1107.9) | 964.0 (831.4–1243.6) | 0.59 | |
| A2 | 973.2 (726.0–1145.9) | 967.3 (775.2–1300.0) | 0.88 | ||
| A3 | 962.3 (777.3–1139.1) | 974.5 (842.4–1147.0) | 0.78 | ||
| A1 | HF, ms2 | 833.8 (265.3–1527.6) | 955.3 (241.7–1761.5) | 0.14 | |
| A2 | 646.6 (310.6–1694.1) | 790.3 (215.3–1574.1) | 0.47 | ||
| A3 | 850.6 (266.9–1555.3) | 837.5 (231.0–1789.6) | 0.58 | ||
| A1 | LF, ms2 | 360.1 (58.0–760.4) | 848.0 (233.3–1058.2) | ||
| A2 | 160.1 (34.5–282.2) | 430.4 (47.0–582.9) | |||
| A3 | 224.0 (65.6–513.1) | 296.3 (109.5–714.7) | |||
| A1 | HFnu, % | 86.6 (60.6– 96.7) | 64.3 (31.5–83.7) | ||
| A2 | 81.7 (57.4– 93.7) | 60.0 (6.2–85.5) | |||
| A3 | 73.9 (51.4– 94.2) | 72.9 (41.1–85.7) | 0.32 | ||
| A1 | LFnu, % | 13.4 (3.3–39.4) | 35.7 (16.3–68.5) | ||
| A2 | 18.3 (6.3–42.7) | 40.0 (14.5–93.8) | |||
| A3 | 26.1 (5.8–48.5) | 27.1 (14.3–85.7) | 0.32 | ||
| A1 | LF/HF | 0.22 (0.04–0.63) | 0.41 (0.15–1.46) | ||
| A2 | 0.23 (0.05–0.45) | 0.59 (0.18–1.63) | |||
| A3 | 0.25 (0.10–0.80) | 0.36 (0.21–1.02) | |||
| NCAP | |||||
| RR, ms | 930.4 (779.5–1055.2) | 933.7 (797.1–1114.5) | 0.67 | ||
| HF, ms2 | 680.7 (280.0–1381.1) | 572.5 (287.3–1254.0) | 0.63 | ||
| LF, ms2 | 120.8 (31.4–295.5) | 217.2 (60.7–318.8) | 0.25 | ||
| HFnu, % | 85.2 (77.2–94.8) | 80.3 (42.2–82.1) | |||
| LFnu, % | 14.8 (5.2–22.8) | 19.7 (17.9–57.8) | |||
| LF/HF | 0.18 (0.17–0.31) | 0.32 (0.23– 0.92) | |||
| N3 Sleep | |||||
| RR, ms | 1003.8 (819.1–1109.7) | 932.4 (827.1–1261.2) | 0.53 | ||
| HF, ms2 | 783.8 (200.8–1584.0) | 743.3 (510.2–1439.8) | 0.77 | ||
| LF, ms2 | 226.3 (133.8–526.0) | 298.1 (77.7–772.5) | 0.13 | ||
| HFnu, % | 80.7 (65.0–91.0) | 72.2 (56.7–84.7) | |||
| LFnu, % | 19.3 (9.0–35.0) | 27.8 (15.3–43.3) | |||
| LF/HF | 0.30 (0.11–0.6) | 0.52 (0.16–0.98) | |||
Data were shown as median (min–max) and examined with the Mann-Whitney U test. RR, RR interval; HRV, heart rate variability; HF, high frequency; LF, low frequency; LFnu, low frequency power of heart rate variability expressed in normal units; HFnu, high frequency power of heart rate variability expressed in normal units.
During NCAP and N3 sleep, RR intervals, and HF and LF power of HRV in the PD patients were not significantly different from those in the healthy controls. In contrast, LFnu was significantly lower and HFnu was significantly higher in the PD patients than those in the healthy controls. Furthermore, the LF/HF ratio was significantly lower in the PD patients than that in the healthy controls.
Sleep disturbance is prevalent in PD patients, and greatly decreases their life
quality [1]. In this study, the authors systematically examined N3
sleep especially CAPs and NCAPs and their associated EEG activities and HRV in
the PD patients compared to the healthy controls and found in the PD patients,
the N3 sleep duration, proportion of N3 sleep occupied by CAPs and CAP A1, as
well as CAP A1 index were significantly smaller while the proportion of N3 sleep
occupied by CAP A2 and A3, and NCAP, as well as CAP A2 and A3 indices were
significantly greater. In addition,
The current study has documented sleep disturbance in the PD patients such as shorter total sleep time and N3 and REM sleep, longer WASO and N2 sleep, and lower sleep efficiency (Table 2). These findings are in line with previous studies [3, 13, 43] and indicate that sleep disturbance, as a non-motor symptom in the PD patients, is very common. In addition to shorter N3 sleep, the proportion of N3 sleep containing CAPs and CAP A1, as well as CAP A1 index in the PD patients was significantly smaller in the current study (Table 3). CAP is a form of periodic EEG activity that may signify sleep disturbance or sleep instability and contribute to arousals in both physiological and pathological conditions [9, 27]. CAP A1 is thought to be associated with deeper and more efficient SWS, which promotes phase-amplitude coupling and synaptic plasticity processes, as well as the brain’s efforts to maintain stable sleep [23, 27]. It has been reported that a positive correlation occurs between CAP A1 and frontal cognitive functions such as language learning and memory processes [5, 7]. Decreased CAP A1 indicate that N3 sleep in the patients is disturbed and becomes less stable. Previous studies have shown alternation in sleep microstructure such as CAPs and CAP A indices even in patients with an early stage of PD [11, 12, 13]. For example, it was found that CAP rate (Duration of CAPs/Duration of NREM sleep) as well as CAP index in the PD patients were significantly decreased, and CAP metrics in the PD patients were significantly correlated with reduced norepinephrine transporter density in arousal prompting nuclei (i.e., locus coeruleus, raphe nuclei) in the brainstem as well as arousal propagating brain structures such as temporal cortex and thalamus. These findings suggest a more severely altered microstructure than macrostructure of sleep in PD patients, which might be associated with widespread dysfunction of the noradrenergic arousal system [12]. Furthermore, the proportion of N3 sleep containing CAP A2 and A3, and NCAP, as well as CAP A2 and A3 indices were found to be significantly greater in the PD patients (Table 3). CAP A2 and A3 (especially CAP A3) are positively correlated with AHI in obstructive sleep apnea (OSA) patients, SB index in SB patients, and periodic limbic movement index in restless leg patients [44, 45], and are always linked with activities that promote arousals and sleep segmentations [46, 47]. These findings further indicate sleep instability in PD patients.
CAP A1 is generally considered to be related to the establishment and
maintenance of stable sleep that facilitates phase-amplitude coupling and
synaptic plasticity processes, and phase A1 predominantly comprises slow EEG
waves in the frontal cortex, which is associated with a strengthened network and
enhanced cognitive function. Phase A1 was found to be positively correlated with
cognitive functions such as memory and language learning, and
CAP A2 is a form of CAP occurring between unstable sleep related to CAP A3 and
stable sleep related to CAP A1. In the current study, it was found in the PD
patients significantly greater CAP A2 index (Table 3) and
CAP A3 is often related to incidents during sleep that increase sleep fragments
and arousals such as movements and OSA [23, 27]. In the current study,
significantly more CAP A3 and greater absolute and relative power of
In the current study,
In the current study, increased
At the same time, a significantly greater power of
HRV is a noninvasive tool to evaluate the autonomic functions [16].
Attenuated HRV has been found during daytime and night in PD patients, strongly
reflecting the blunted autonomic nervous system [21]. Orthostatic
hypotension is a common symptom reflecting impairments in the autonomic system in
PD patients [64]. In addition, exercise-evoked sympathetic activation is
decreased in PD patients irrespective of dopaminergic mediations. Finally, PD
patients show reduced LF power of HRV, which can be considered to reflect
compromised sympathetic modulation of vascular tone [65, 66]. HRV
impairment could also be more overt in PD patients with worse motor symptoms [67], reminding concomitant changes of the autonomic system with motor
system. In the present study, the authors focused on HRV during N3 with
consideration of CAPs and found a significantly lower LFnu and LF/HF ratio but no
significant differences in RR interval, LF and HF power of HRV during N3 sleep
and NCAP in the PD patients compared to the healthy controls. In addition,
smaller LF power of HRV was also found during CAPs (Table 5). The HF HRV is
considered to reflect the parasympathetic activity mediated by vagal nerve, while
the LF power of HRV reflects the baroreflex modulation of the autonomic outflow
mediated by both sympathetic and parasympathetic system [39, 40]. Sympathetic
regulation was found to be blunted while parasympathetic regulation was
relatively intact during N3 sleep in the PD patients in the current study, which
was consistent with previous findings showing blunted sympathetic regulation
during REM and NREM sleep [21]. In PD patients, accumulation of
In summary, in N3 sleep, significantly fewer CAP A1, more CAP A2 and A3, smaller
LF power of HRV and LF/HF ratio during CAPs and NCAPs, smaller power of
While the current study showed changes in sleep especially CAPs and NCAP of N3
sleep and associated changes in HRV in the PD patients compared to the healthy
controls, there are some limitations to be considered. First, it was possible
that dopaminergic drugs might have impacted the results since the PD patients in
the present study were not drug naïve, but were taking dopaminergic
medications such as L-dopa, ropinirole and pramipexole. However, application of
controlled-release Co-beneldopa (200/50 mg) before sleep has been reported to
increase the total SWS time by about 10% without significant influences on other
sleep stages [72]. In addition, dopamine agonist pramipexole does not affect
CAPs and most sleep parameters except for prolonged N2 sleep and increased sleep
efficiency, decreased WASO, which might be considered as a result of relief of
pre-sleep restless leg syndrome (RLS) symptoms and abolition of periodic limb
movements, not action on sleep abnormalities in RLS patients [73]. Moreover,
acute or chronic usage of dopamine agonist pramipexole has been reported not to
change HRV parameters and tonic sympathovagal regulation [74, 75]. Thus, future
studies need to include anti-PD medication naïve PD patients. Secondly, the
number of subjects was also relatively small as the standards for inclusion of
healthy controls and PD patients were quite strict. Nevertheless, the power of
each test with p
In the PD patients, it was found in the current study that N3 sleep duration,
percentage of N3 sleep occupied by CAPs and CAP A1, as well as CAP A1 index
during N3 sleep were significantly shorter or smaller compared to healthy
controls. In addition,
The data used to support the findings of this study are included in the article.
DY conceived and supervised the study, and ZC finalized the data analysis and drafted the article. ZC, QL, XZ, ZZ, QOuyang, CG, FY, YL, YM, and DY contributed to study design and execution, recruiting Parkinson’s disease patients and healthy controls, data acquisition, analysis and interpretation, sort references, preparation of the figures and tables, critically reviewing and editing the manuscript, and read and approved the final manuscript. All the authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
All research protocols and experimental procedures were reviewed and approved by the Jiangxi Provincial People’s Hospital Research Ethics Committee (No. 2019-014) in compliance with the principles of the Declaration of Helsinki. Informed consent was acquired from all the subjects.
The authors are grateful to Professor Barry Sessle of the University of Toronto for his assistance in editing this article and helpful discussions, and also wish to thank all the subjects for participating in the study.
This research was supported by the Jiangxi Provincial People’s Hospital Grant 2019-009, and Jiangxi Province Key Laboratory of Neurology Grant (No. 2024SSY06081).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




