1 Department of Ophthalmology, Vision Research Center, School of Medicine, University of Missouri-Kansas City, Kansas City, MO 64108, USA
2 Department of Biomedical Sciences, School of Medicine, University of Missouri-Kansas City, Kansas City, MO 64108, USA
†These authors contributed equally.
Abstract
Initial manifestations of neurodegenerative ocular conditions, including age-related macular degeneration (AMD) and glaucoma, often remain undetected in the early stages and can begin after the age of 50 years with the likelihood gradually increasing each year thereafter. This study aimed to explore variances in visual and retinal function and anatomy among C57BL/6J mice, aiming to pinpoint differences between biological age and sex factors that potentially lead to the onset of vision impairment.
A longitudinal study evaluated visual acuity (VA) and contrast sensitivity (CS) using optomotor reflex (OMR), and retinal function, encompassing scotopic and photopic measurements, was recorded by electroretinogram (ERG) at 12 months of age. Tissue was subsequently harvested for histological analysis, complementing the in vivo findings. Disparities in visual function were observed between individual male and female mice, necessitating categorization of visual impairment levels to investigate further sex-specific differences in the study’s aging population. Comparisons between sex and the degree of visual impairment were conducted using ANOVA followed by Tukey’s or Bonferroni’s post-hoc corrections and unpaired t-tests. Pearson correlation analysis determined the association between biological factors.
Sex-related disparities were found in the visual function of male (n = 13) and female (n = 18) mice aged 5–12 months. Eyes were categorized by vision impairment: normal vision, or low, moderate, or severe vision loss at the end of the study. Male and female mice differed in mean contrast sensitivity, indicating less sensitivity to fine detail and moving stimuli in female mice (11–12 months old, p < 0.001). Spectral-domain optical coherence tomography (SD-OCT) revealed a thinner retinal outer nuclear layer in male mice (p < 0.0001), although this did not vary across different levels of vision impairment. ERG indicated slower retinal responses in male mice (p < 0.05), while histology showed a significant reduction in the inner plexiform layer thickness in male mice with severe vision loss (p < 0.0001). Conversely, female mice exhibited greater thinning in the photoreceptor layer when vision was unimpaired (p < 0.01).
The study shows that sex and extent of vision impairment influence visual and retinal health, with individual retinal layers differentially changing in thickness over time.
Keywords
- C57BL/6J
- aging
- retina
- vision
- optomotor reflex
- spectral-domain optical coherence tomography
- electroretinogram
The inbred C57BL/6J wild-type mouse is a staple in research, used in various research areas, including neurobiology, diabetes, obesity, cardiovascular, developmental, immunology, and genetics [1, 2, 3, 4, 5, 6, 7]. Comparative studies encompassing behavior, aging, and or sex differences are necessary to understand the cause or severity of common diseases and treatment outcomes [8, 9, 10, 11]. Mice are advantageous models for aging studies because of their relatively short lifespans compared to humans. Despite the widespread use of the C57BL/6J mouse model in preclinical research and the recognition of age and sex as critical biological variables in humans, researchers have yet to determine the impact of age and sex on many physiological and pathophysiological processes studied with this model.
Age represents a biological variable that affects a similarly wide range of
physiological and pathophysiological processes and encompasses both the
development and aging of the structure and function of organs. Aging is a
significant contributor to the accumulation of oxidative stress that results in
degeneration of the structure and function of tissues, including the retina [12].
Oxidative stress-induced cellular senescence and reduced density of retinal
ganglion cells, bipolar cells, photoreceptors, and pigment epithelial cells can
induce vision loss [13, 14]. Structurally, retinal layer thickness in normal eyes
changes with age, specifically significant decreases in the thickness of the
peripapillary retinal nerve fiber layer (RNFL), pericentral ganglion cell layer
(GCL), peripheral inner plexiform layer (IPL), and foveal outer segment layer
(OSL) thickness with age, and a significant increase in the thickness of the
foveal retinal pigment epithelial (RPE) with age [15]. Similarly, Won and
colleagues determined age-related changes in the thickness and volume of retinal
layers, specifically thinner peripheral RNFL, GCL, and pericentral and peripheral
IPL and thicker foveal inner nuclear layer (INL) and inner retina (IR) in the
older group (
Sexual dimorphism appears in many biological processes, including visual function, where factors such as disease susceptibility, color perception, visual acuity (VA), and photoreceptor cell distribution and density vary depending on sex [18]. Male humans possess a thicker macula [19], a more significant relative number of long wavelength sensitive (L-cones) and middle wavelength sensitive (M-cones) cone photoreceptors [20], increased response to blue light stimulation [21], and significantly heightened retinal sensitivity for fine detail and fast-moving stimuli [22, 23]. Increased iron levels are found in the female retina and RPE [24, 25]. The most well-known dimorphism is color perception. X-linked red-green colorblindness found in males [26] is the most common, while females are more prone to age-related macular degeneration (AMD) [27, 28] and glaucoma [29]. Metabolic profiles from different ocular tissues display sexual dimorphisms [18]. Sex-related structure and function differences in the retina are likely due to hormone profiles [30]. Study has shown that female mice exhibit larger amplitudes in electroretinogram (ERG) recordings than male mice [31]. Spectral-domain optical coherence tomography (SD-OCT) has shown differences in the mean thickness of the outer nuclear layer (ONL), the outer plexiform layer (OPL), and the INL in the macular region, while the RNFL is thicker in females [30]. The estrus cycle may exert a role in these structural and functional differences due to the presence of estrogen receptors in ocular tissues [32]. The menstrual cycle and accompanying hormonal fluctuations modulate ocular structures, including the retina [33]. However, the exact mechanism and implications of these changes are still a topic of ongoing research.
In addition to the significant impact of both age and sex as biological variables, one of the challenges to the development of new therapeutics for age-related neurodegenerative diseases of the retina, such as dry AMD and glaucoma, is the lack of animal models that recapitulate underlying mechanisms of, and phenotypes present in retinal disease. In many studies, animals are genetically modified or exposed to risk factors such as advanced aging or specific diets to establish the pathobiological processes involved with the expectation of treating or delaying its progression [34, 35, 36, 37, 38]. Rigorous studies of wild-type mice are needed to understand better time-related changes that potentially predispose to or contribute to neurodegenerative pathology. Visual processing of moving stimuli evaluated by initial and late phase optokinetic responses deteriorates in aged (21–24-month-old) mice compared to young adult mice [39]. A study utilizing C57BL/6J mice aged between 2 and 32 months reported that natural age-related retinal function and morphology changes occur gradually rather than abruptly [40]. Additionally, other studies described morphological changes, including inappropriate localization of photoreceptor synapses [39], topographic differences in RPE morphology [41], neuroinflammation, and increased glial reactivity in C57BL/6J mice [42]. As new models of retinal degeneration are developed, recent advancements in retinal organoid cultures have emerged as a particularly exciting area of research. These three-dimensional tissue constructs hold significant potential for applications in personalized medicine, drug screening, gene therapy, and cell transplantation. Retinal organoids provide a more precise model for studying human retinal diseases, serving as a valuable complement to traditional animal studies, particularly when animal models exhibit only partial traits of specific disease phenotypes [43, 44].
Our study evaluated visual performance longitudinally in wild-type, middle-aged C57BL/6J mice aged 5–12 months old, retina structure and function using SD-OCT imaging and ERG recordings at the completion of the study, with post-mortem histological analysis. This work sought to clarify early age-related changes in visual and retinal function and structure in the retina of wild-type mice, routinely used in retinal neurodegenerative research and as a potential model for assessing age and sex as biological variables. Empirical stratification of visual impairment, as an additional variable, provides a framework for understanding visual impairment in the early stages of the neurodegenerative process by evaluating sex-dependent differences in visual impairment results.
Five-week-old C57BL/6J male (n = 13) and female (n = 18) mice were purchased from Jackson Laboratories (Bar Harbor, ME, USA). All animals were socially housed in plastic shoebox cages at 25 °C, with ad libitum access to Teklad Global 18% Protein Rodent Diet (Envigo, Somerset, NJ, USA; Cat. #2918.15) and water and maintained on a 12-hour light/dark cycle. Weekly body weights of all animals were recorded, and the average weights were plotted in graphs at two-month intervals. The mice underwent visual performance screenings at 5 months old until they reached 12 months old. Each animal’s right and left eyes were tested separately, depending on the direction of the stimulus pattern, with each eye representing n = 1. Each eye was considered individually for statistical analysis and to capture individual differences in the results. Eyes were removed from the study if found to have ocular injuries, opacities, or abnormal ocular anatomy to ensure accurate and reliable results [45, 46, 47]. A total of 48 eyes were included after excluding ineligible samples.
Behavioral VA assessments using the optomotor response aided in categorizing
individual mouse eyes into 4 groups: normal vision (
Visual acuity or spatial frequency and contrast sensitivity (CS) assessment were determined using the OptoMotry© optomotor testing system with OptoMotry software, version 1.7.7 (CerebralMechanics Inc, Lethbridge, Alberta, Canada) described earlier [49, 50, 51, 52]. Briefly, testing was conducted monthly between 5 and 12 months of age. Each measurement was averaged and plotted in graphs at two-month intervals. Visual acuity is the maximum spatial frequency in which an optomotor response and head tracking has occurred. Visual acuity was measured at 100% contrast, using a drift speed of 12.0 d/s and a cutoff maximum of 0.500 c/d. Contrast sensitivity refers to the minimum contrast level at which head tracking is detected. A lower contrast percentage indicates better contrast vision. The measurement of contrast sensitivity began at a baseline spatial frequency of 0.042 c/d (cycles/degree) and a beginning contrast level of 100%. Animals were placed on a 55 mm or larger platform (dependent on weight) where stimuli of varying spatial frequencies or contrast levels elicited a response [48, 53].
Electroretinography responses were recorded with an HMsERG electroretinography system with accompanying software, version 4.180 (Ocuscience, Henderson, NV, USA), as described previously [51, 52]. The retinal function of rods (scotopic) and photoreceptors (photopic) was determined between 12–15 months of age. Animals were dark-adapted overnight, with only red light used for setup and performing the test. Anesthesia was initially administered at 4.0% isoflurane (cat. #029405, Covetrus, Dublin, OH, USA) with an oxygen flow rate of 1 L/min and maintained at 2.0% with the same oxygen flow rate for the test duration. Anesthetic drops of 0.5% Proparacaine Hydrochloride Ophthalmologic solution (Henry Schein, Inc., Cat. #2963726, Port Washington, NY, USA), dilation drops of 1% Tropicamide solution (Henry Schein, Inc., Cat. #70069012101, Port Washington, NY, USA), and 0.3% hypromellose (GenTeal® Tears Ophthalmic Gel, Henry Schein, Inc., Cat. #0065806401, Port Washington, NY, USA) eye drops were added to each eye for corneal lubrication. A warming pad maintained the body temperature at 37 °C while a temperature probe monitored the animal’s temperature. The ground electrode (Ocuscience, Henderson, NV, USA) was placed above the base of the tail, and reference electrodes (Ocuscience, Henderson, NV, USA) were placed behind the ear with the tip just behind each eye. Thread electrodes (Ocuscience, Henderson, NV, USA) and a 2.0 mm mini contact lens made of Aclar material were coated with GenTeal tears and placed on each eye to prevent moisture loss throughout the procedure.
ERG scotopic and photopic responses were acquired and analyzed usingERGView 4.380V software (Ocuscience, Henderson, NV, USA) accompanied by the
HMsERG system using a 150 Hz low pass filter and 60 Hz noise-eliminating filter.
The ERGView software determined the b/a wave ratio, which serves as an indicator
of inner-to-outer retinal function. Scotopic threshold responses (STRs) and
photopic negative responses (PhNRs) were calculated from flashes generated from
the scotopic flash intensity series (–4.5 to –3.5 log cd
In vivo imaging determined retinal thickness in wild-type C57BL/6J mice at 15 months. Optical coherence tomography images were obtained by an iVivo® SD-OCT with OctEngine software, version 1.8.41.1 (Ocuscience, Henderson, NV, USA) with 5µm transverse resolution, submicron depth resolution, and a live fundus image-guided alignment. Pupils were anesthetized with 0.5% Proparacaine Hydrochloride Ophthalmologic solution (Henry Schein, Inc., Cat. #2963726, Port Washington, NY, USA) and dilated with 1% Tropicamide solution (Henry Schein, Inc., Cat. #70069012101, Port Washington, NY, USA). GenTeal® Tears Ophthalmic Gel (Henry Schein, Inc., Cat. #0065806401, Port Washington, NY, USA) lubricated the cornea throughout the scans. A platform allowed researchers to manipulate the mice into the correct position for imaging. A SomnoFlow (Kent Scientific, Torrington, CT, USA) anesthesia system administered isoflurane (cat. #029405, Covetrus, Dublin, OH, USA) anesthesia and maintained it at 2.0% for the time needed to acquire images. The platform allowed anesthesia to flow through a nose cone for the imaging duration. Images were exported as JPEG and TIFF files to quantify the thickness of the retinal layers at 500 µm from the optic nerve. An average of 16 B-scan images were post-processed using VQ Enhance software, version 1, provided by Lumedica, Durham, NC, USA. Open-source Fiji Image J, 64-bit software (https://imagej.net/software/fiji/, NIH, Bethesda, MD, USA) was used to measure retinal thickness [54]. Total retinal thickness is defined as the distance from the RNFL (including the RNFL) to the RPE layer (including the RPE layer) in a vertical retina section. Transformation of grey-scale images to pseudo-color was accomplished by applying a royal lookup table (LUT) provided by VQ Enhance software.
After euthanasia and assertion of death, eyes were enucleated and
immersion-fixed in a 4% formaldehyde solution (16% w/v paraformaldehyde (PFA) aqueous solution,
Electron Microscopy Sciences, Cat. #15710, Hatfield, PA, USA) in phosphate
buffer (0.1 M PB, pH 7.4) for 30 min at 4 °C. After 30 min, a cut was
made along the ora serrata, and the eyes were returned to the 4% formaldehyde
solution for O/N fixation. The next day, the tissue was removed from the fixative
and cryoprotected with increasing concentrations of 10% and 20% sucrose
solutions in 1
A Leica DM IL LED inverted microscope (Leica Microsystems, Mannheim, Germany)
and a Q imaging 12-bit camera (Q Imaging, Surrey, BC, Canada) acquired the
light microscopy images at 4
Open-source Fiji ImageJ Windows 64-bit software version (https://imagej.net/software/fiji/downloads) was used for image analysis, histological quantification of layer thickness, and photoreceptor nuclei counts. A representative image of a histological section from a 15-month-old female C57BL/6J mouse with severe vision loss (Supplementary Fig. 6A) demonstrates the quantification of retinal layers and the counting of photoreceptor nuclei rows (Supplementary Fig. 6B). Each image was quantified in triplicate.
Differences between sexes and visual impairment from age-related changes were
determined by a two-tailed, unpaired t-test or one- or two-way ANOVA
with Tukey’s or Bonferroni’s post-test, where indicated using Graph Pad
Prism® 10.2.3 software (GraphPad, San Diego, CA, USA). Results
were considered significant with a p-value of p
We began our study by investigating whether the observed weight differences
between male and female mice were associated with corresponding disparities in
visual acuity impairment. In this study, animals were weighed before each VA
assessment. As shown in Fig. 1, male C57BL/6J mice displayed an overall
statistically significant increase in body weight compared to age-matched females
throughout this study (Fig. 1A) [55]. At 5 months, male mice initially weighed 34
g
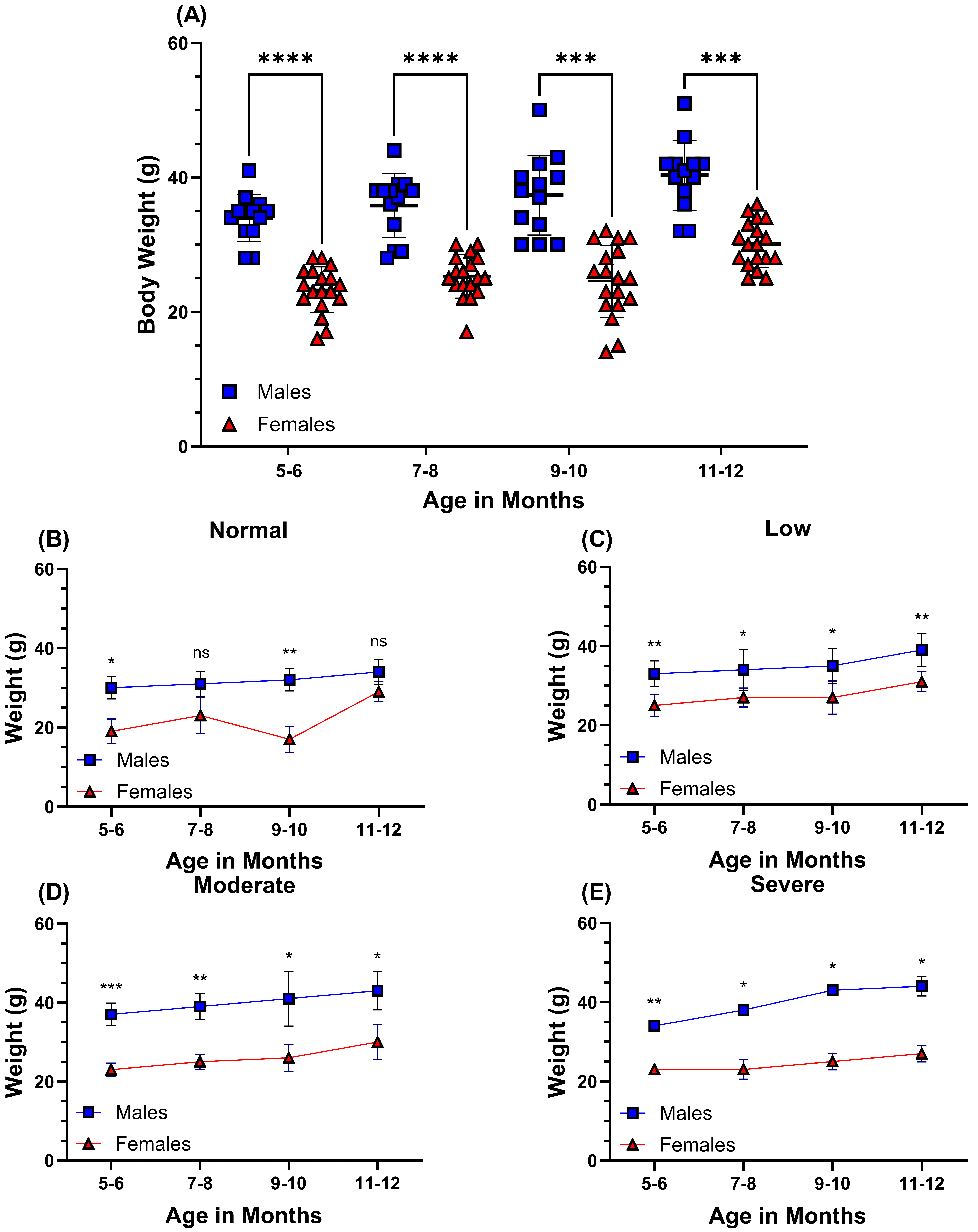 Fig. 1.
Fig. 1.
Body weight differences of aged-matched male and female C57BL/6J
mice. The data presents monthly measurements of mean body weights (in grams)
recorded for all mice throughout the study (A). Smaller representative graphs
indicate mean body weights for male and female mice eyes stratified according to
the degree of vision loss: normal vision (n = 7), low (n = 18), moderate (n =
15), and severe vision loss (n = 8), determined by behavior assessment of
optomotor reflex (B–E). The visual acuity scale is shown in Fig. 2B. Male mice
(n = 13) are symbolized by blue squares, and female mice (n = 18) are symbolized
by red triangles. Results are mean
Visual function (VA and CS) was quantified by behavioral assessment of optomotor
reflex (OMR) beginning at 5 months of age (Fig. 2A,B). No statistically
significant differences were found in visual function between male and female
mice aged 5–12 months. Visual acuity started to decline at 9–10 months of age
and declined even further by the study’s conclusion. Male mice’s mean initial VA
at 5 months of age was 0.490 (c/d)
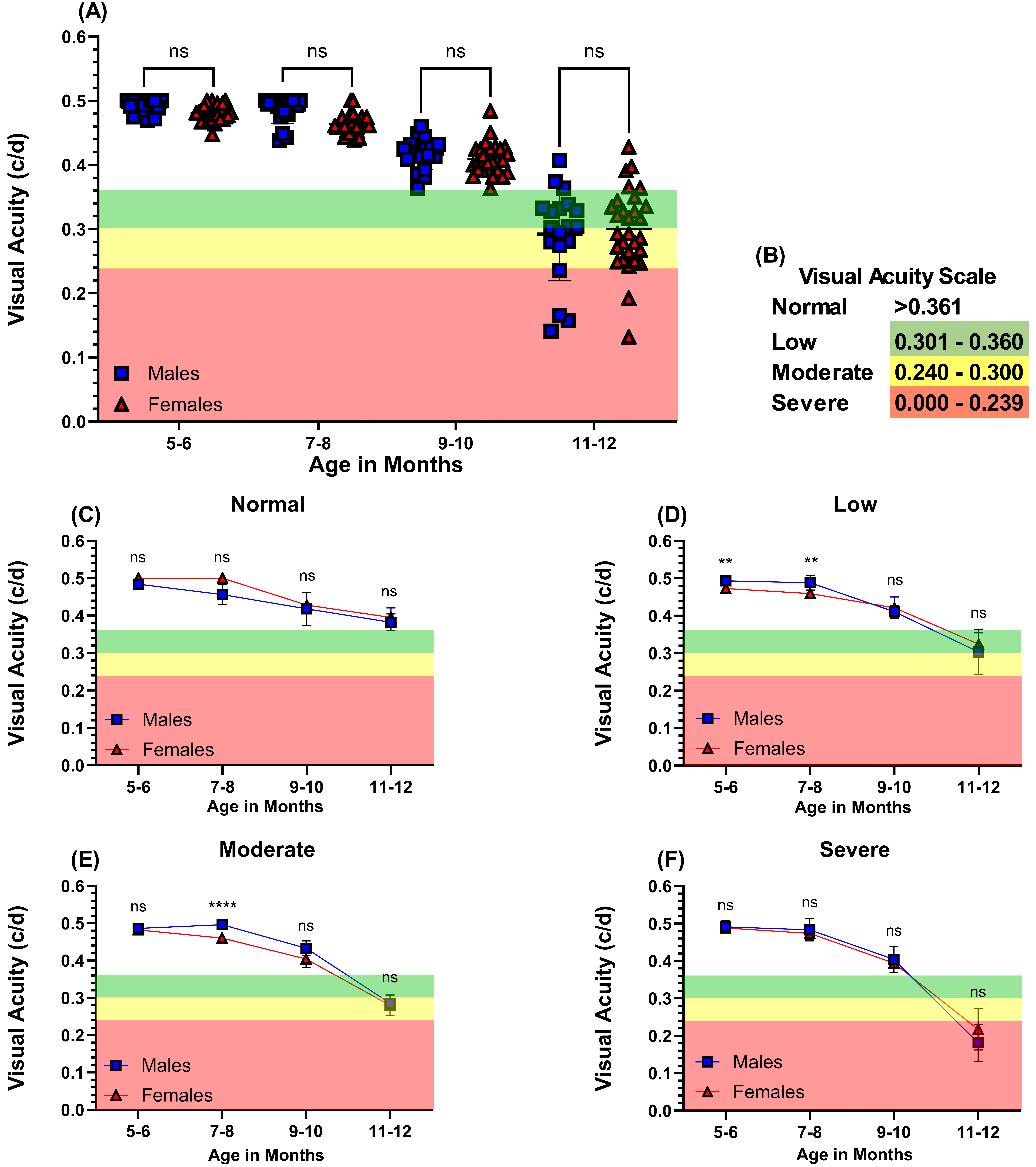 Fig. 2.
Fig. 2.
Visual acuity in middle-aged C57BL/6J mice. Visual acuity (A)
was assessed in mice monthly by behavior assessment of optomotor reflex expressed
as spatial frequency units, cycles/degree (c/d). Blue squares symbolize
individual male mice eyes (n = 19), and red triangles symbolize individual female
mice eyes (n = 29). Scale for the severity of visual loss expressed by spatial
frequency (c/d) is normal (n = 7; white)
Assessment of CS also identified sex as a biological variable with statistically
significant differences between male and female mice (Fig. 3A). Contrast
sensitivity started to decline at 9–10 months of age and continued to decline
further by the end of the study. The mean initial CS for male mice at 5 months of
age was 14.7
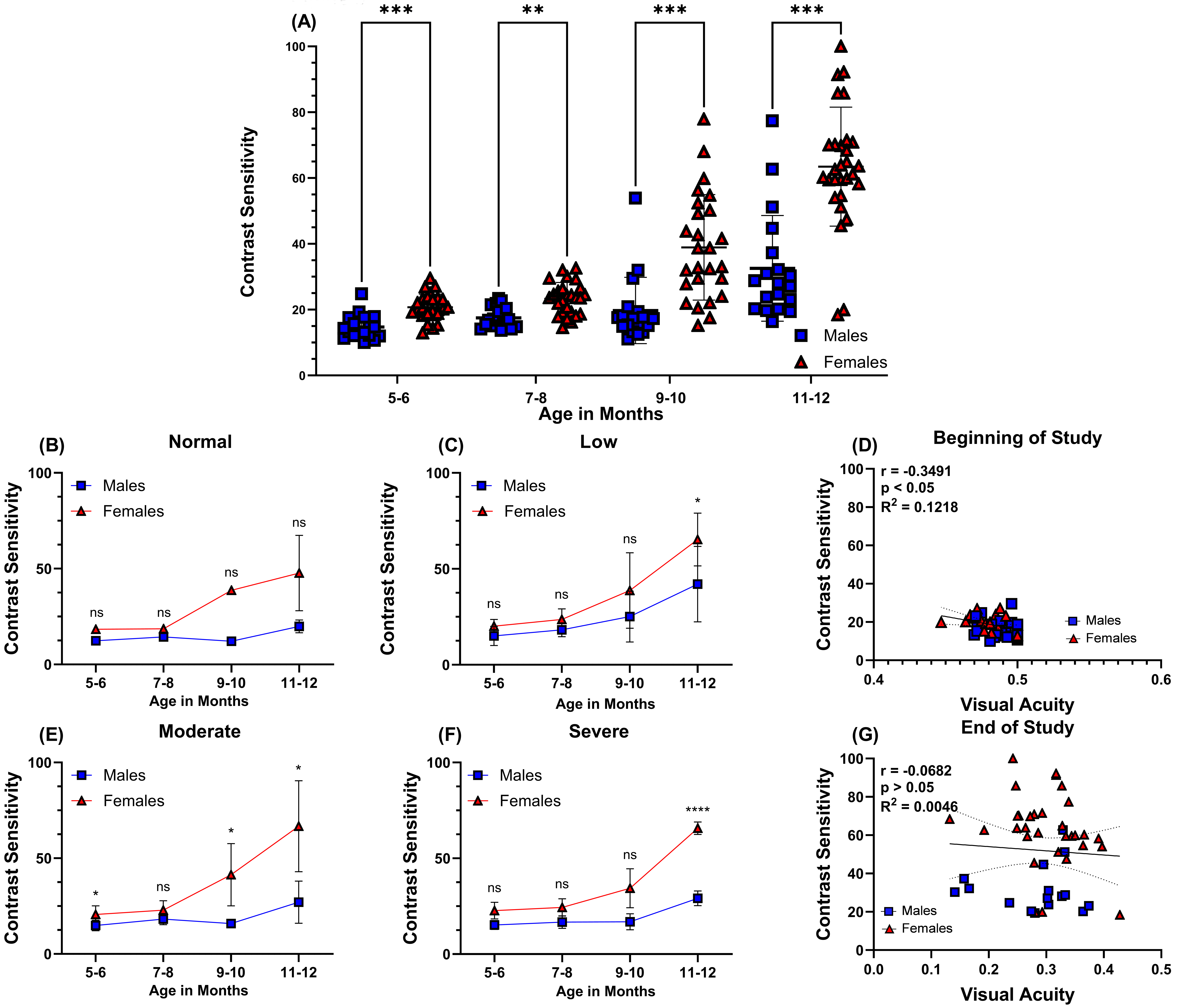 Fig. 3.
Fig. 3.
Contrast sensitivity in middle-aged C57BL/6J mice. Contrast
sensitivity (A) was assessed in mice monthly by behavior assessment of optomotor
reflex. Blue squares symbolize individual male eyes (n = 19), and red triangles
symbolize individual female mice eyes (n = 29). Smaller representative graphs
(corresponding animals chosen from representative VA graphs from Fig. 2C–F
indicate differences between age-matched males in blue and females in red
according to the degree of vision loss: normal vision (n = 7), low (n = 18),
moderate (n = 15), and severe vision loss (n = 8), determined by behavior
assessment of optomotor reflex (B–E). Correlations between VA and CS were
determined at the beginning (F) and end of the study (G) for all eyes. Dashed
lines around the regression line indicate 95% CI. Specific Pearson correlation
coefficient r, respective p-values, and coefficient of determination
R2 are listed directly in panels. Data are presented as mean
Horizontal cross-sections of retinas from male (Fig. 4A) and age-matched female (Fig. 4B) C57BL/6J mice were imaged by SD-OCT. Images are shown in black and white and pseudo-colored to visualize the reflective signal’s intensity better, allowing for greater contrast between individual retinal layers. Layers colored red to white indicate tissues with high reflectivity as opposed to layers colored green to yellow. Representative images are grouped according to the degree of visual impairment using spatial frequency OMR stratification described above (normal vision, low, moderate, and severe vision loss). Images in Supplementary Fig. 1 identify and indicate the labeling of layers for quantification purposes using a 5-month-old C57BL/6J female mouse retina (Supplementary Fig. 1A) compared to a 15-month-old C57BL/6J female mouse retina (Supplementary Fig. 1C). No differences in thickness were noted between the nasal and temporal retina to the optic nerve (data not shown). Representative OCT images were arranged to visualize differences between male and female retinas belonging to 5-month-old animals (Supplementary Fig. 1B) and 15-month-old male (Supplementary Fig. 1D) and female (Supplementary Fig. 1E) mouse retinas stratified according to the degree of visual impairment. The ONL contains the cell bodies of rod and cone photoreceptors. The structure of the ONL serves as an essential biomarker of retina neurodegeneration [56, 57]. ONL thinning can be seen in the representative images for low, moderate, and severe vision loss compared to normal vision, though more pronounced in male mice (Supplementary Fig. 1D).
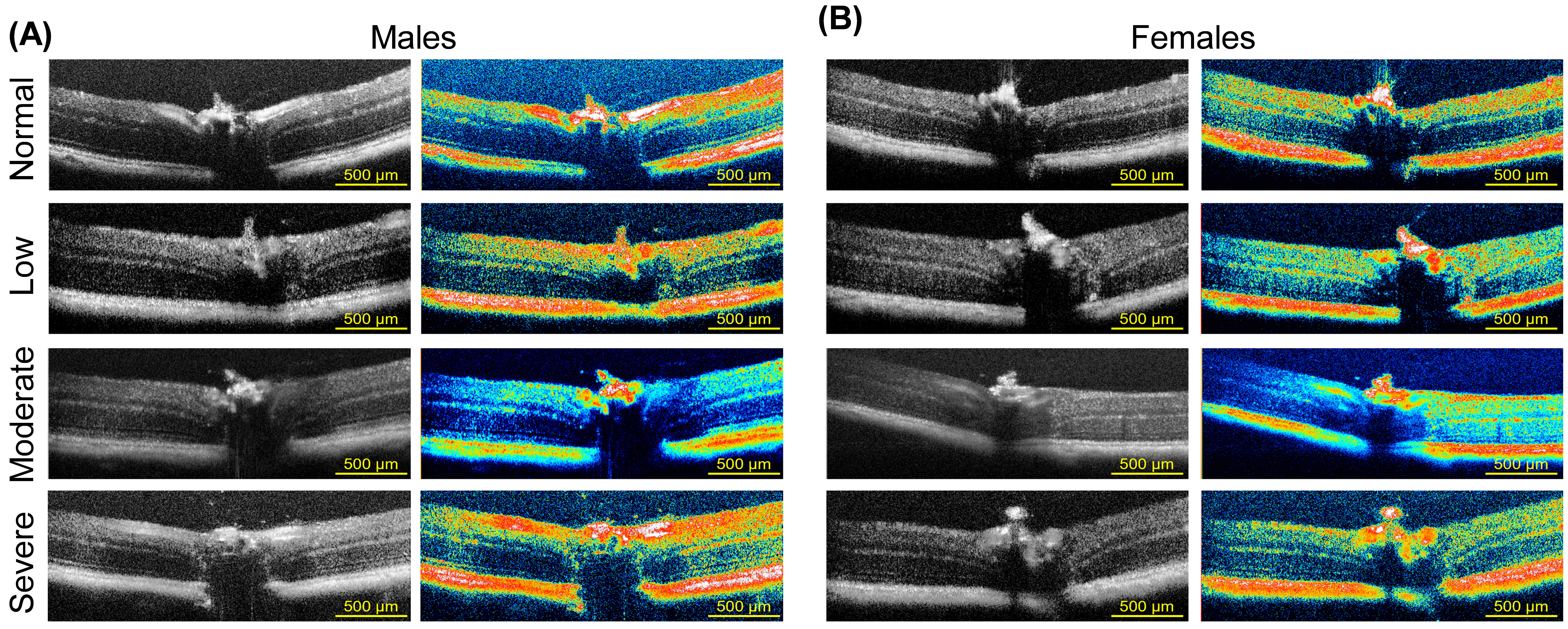 Fig. 4.
Fig. 4.
In vivo SD-OCT imaging in middle-aged C57BL/6J mice. Representative OCT B-scan images demonstrating retinal structure (black and white images) and corresponding royal pseudo color to enhance the contrast between retinal layers in male (A) and age-matched female mice (B). Images were also stratified according to the degree of vision loss: normal vision, low, moderate, and severe vision loss determined by behavior assessment of optomotor reflex. Scale bar = 500 µm. OCT, optical coherence tomography; SD-OCT, Spectral-domain-OCT.
Individual retinal layer thicknesses were quantified from SD-OCT scans as
detailed above, with the ONL in male mice showing a statistically significant
decrease in thickness compared to female mice (54.83 µm
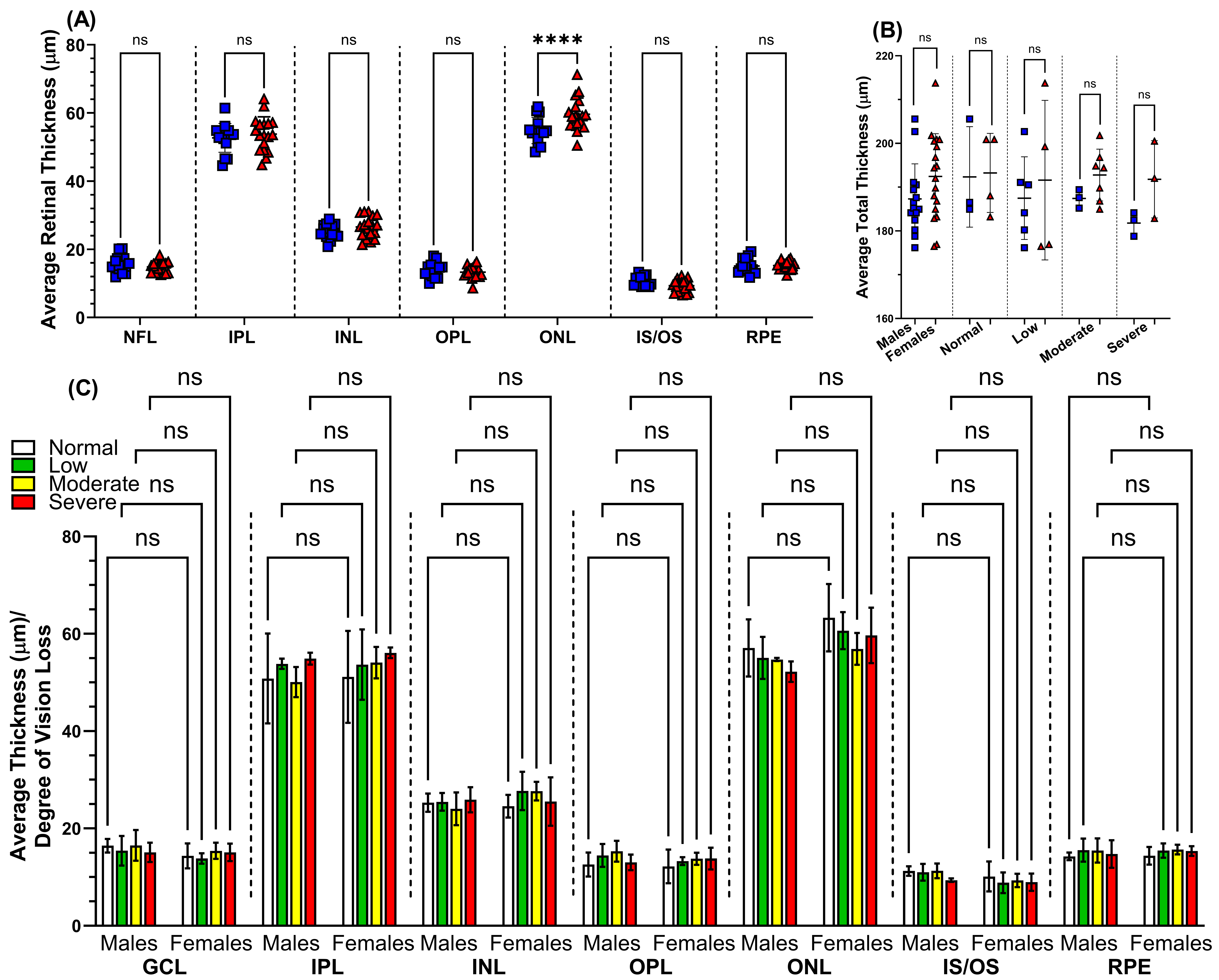 Fig. 5.
Fig. 5.
Quantification of retinal layer thickness from optical coherence
tomography imaging. OCT B-scan images were manually reviewed and segmented, and
each layer was measured for thickness by comparing male and female mice using
Fiji – ImageJ processing software (A). The mean total retinal thickness for male
and female mice was stratified according to the degree of vision loss: normal
vision (n = 7), low (n = 18), moderate (n = 15), and severe (n = 8) vision loss
determined by behavior assessment of optomotor reflex (B). Blue squares symbolize
male mice (n = 13), and red triangles symbolize female mice (n = 18). The mean
thickness of individual retinal layers for male and female mice was also
categorized according to the degree of vision loss determined by behavior
assessment of optomotor reflex (C). Data are presented as mean
Pearson correlations were performed for visual function variables, VA and CS, to
total retinal thickness (Supplementary Fig. 2A,B), INL thickness
(Supplementary Fig. 2C,D), and ONL thickness (Supplementary
Fig. 2E,F) quantified from OCT images of each eye at the end of the study, to
test for consistency in the data. No appreciable correlation was found
for VA to total retinal thickness, INL thickness, and ONL thickness in male and
female mice. However, CS for C57BL/6J male mice to total retinal thickness
(Supplementary Fig. 2A) showed a moderate, negative correlation that was
statistically significant (r = –0.5318, p
Full flash ERG measurements of scotopic (Fig. 6A–E,K) and photopic (Fig. 6F–J,L) functions were recorded from aged-matched male and female mice. Delays
in mean ERG responses for scotopic b-wave implicit time from male mice were
statistically significant compared to female mice (123.2 ms
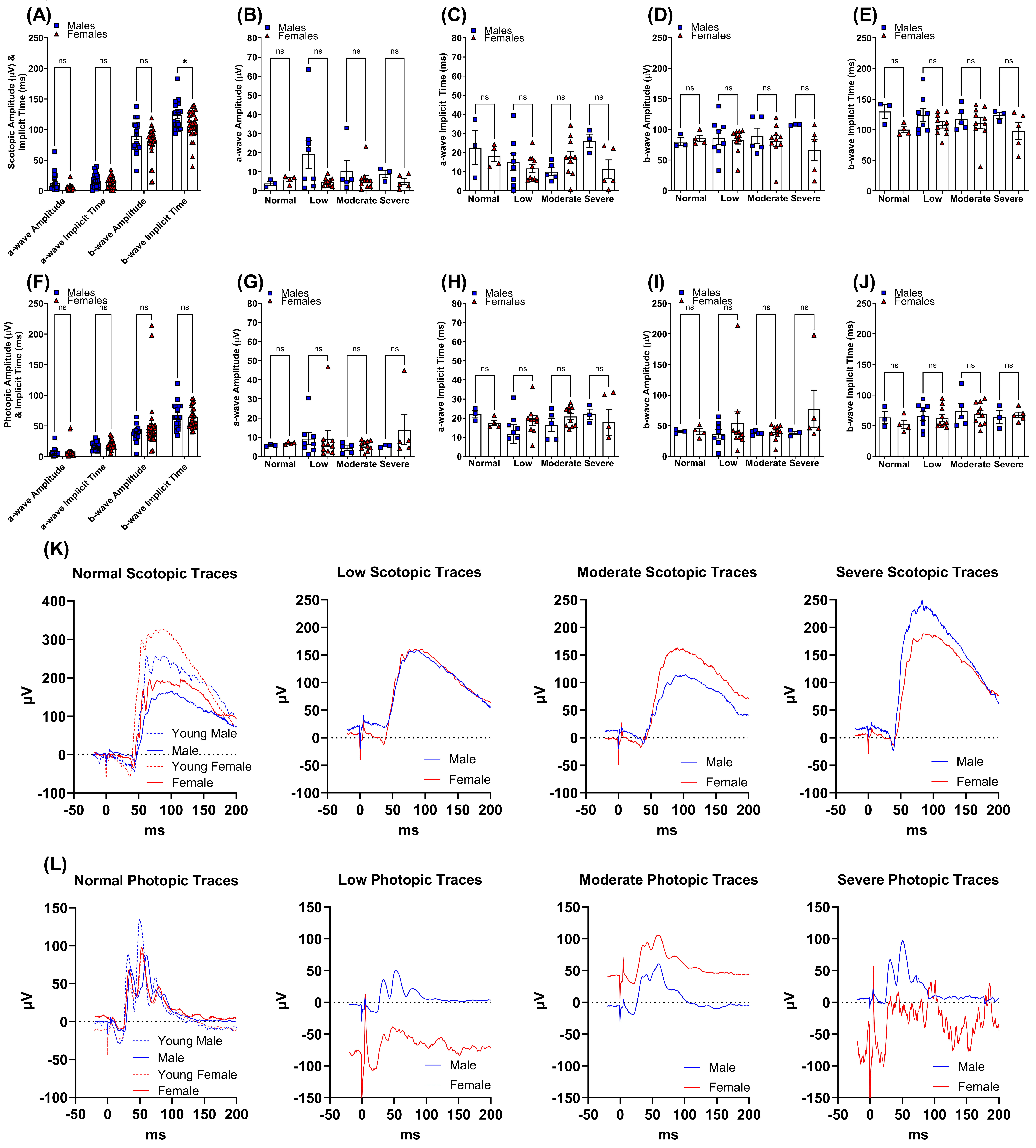 Fig. 6.
Fig. 6.
Retinal function differences determined by electroretinogram of
middle-aged C57BL/6J mice. Full-field flash ERG was used to determine scotopic
and photopic amplitude (A,B,D and F,G,I, respectively) and implicit time
(A,C,E and F,H,J, respectively) in 15-month-old age-matched male mice
eyes (n = 19) represented by blue squares and female mice eyes (n = 29) represented
by red triangles. Scotopic and photopic ERG responses are categorized into normal
vision (n = 7), low (n = 18), moderate (n = 15), and severe (n = 8) vision loss
(B–E,K, and G–J,L, respectively) determined by behavior assessment of
optomotor reflex. Mean representative line graphs indicate ERG differences
between males in blue and females in red according to the degree of vision loss.
Individual male eyes (n = 10) and individual female eyes (n = 8) of 5-month-old
C57BL/6J mice were used as comparisons (open blue squares with dashed lines and
open red triangles with dashed lines). Data are presented as mean
Single flash recordings for scotopic (Supplementary Fig. 3A–D) and
photopic (Supplementary Fig. 3E–H) amplitude and implicit time show
differences between male and female mice. Individual flashes for scotopic b-wave
implicit time (Supplementary Fig. 3D) at –5.5 log cd
The b/a wave ERG ratio is a predictive measure for assessing the functional relationship between the inner and outer layers of the retina. Pearson correlation analyses were conducted to evaluate the association between VA and CS with the scotopic b/a wave ratios (refer to Fig. 7A,B) and the photopic b/a wave ratios (see Fig. 7C,D) at the conclusion of the study. No correlations were found between VA and CS with b/a wave ERG ratios. Neither scotopic b/a wave ratios (Fig. 7E) nor photopic b/a wave ratios (Fig. 7F) exhibited appreciable statistical significance between age-matched male and female mice when eyes were stratified by degree of visual impairment.
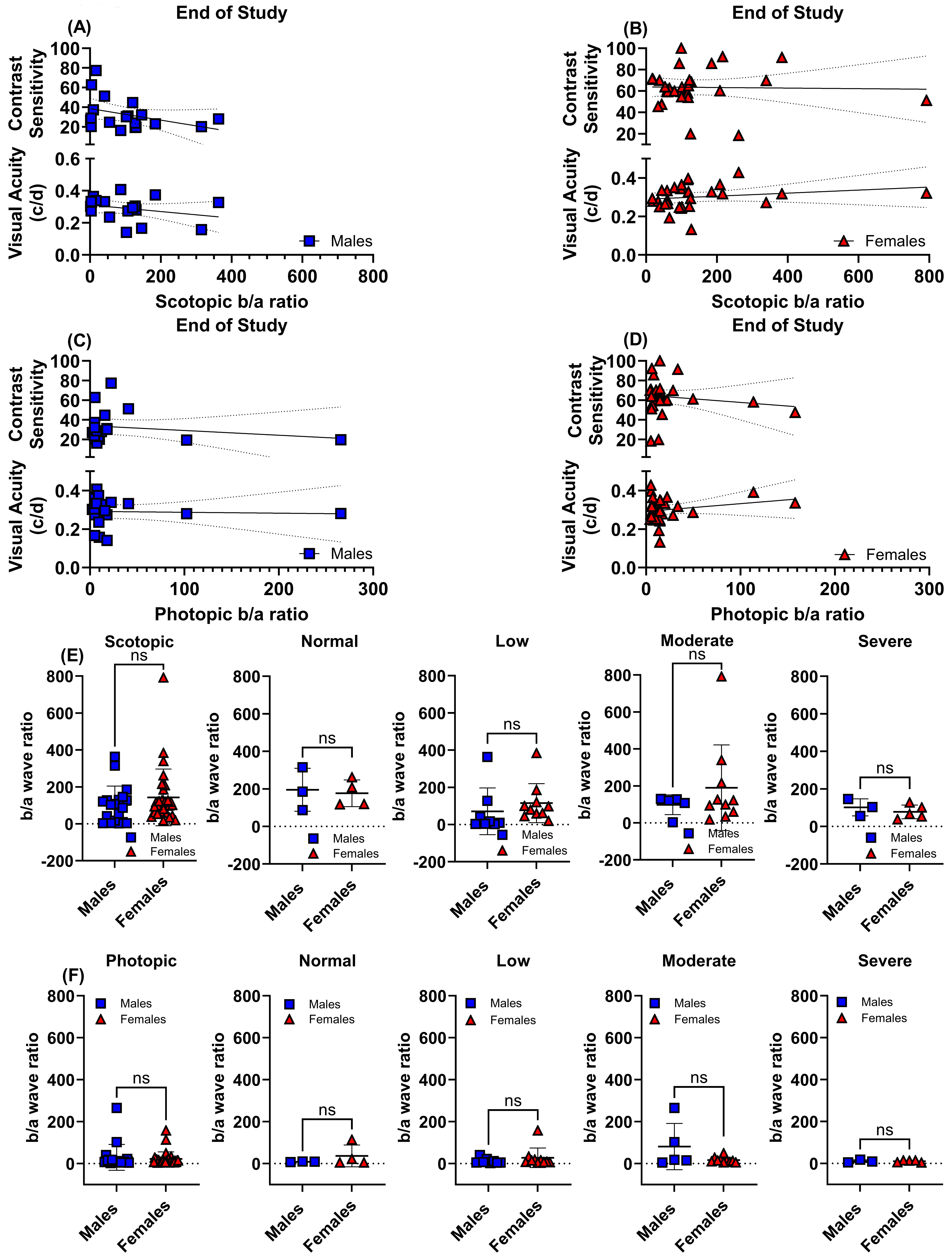 Fig. 7.
Fig. 7.
Relationship between visual function and retinal function
represented by ERG b/a ratio. Correlations were determined for male and female
C57BL/6J mice eyes between behavior assessment of visual function (VA and CS) and
scotopic (A,B, respectively) and photopic (C,D, respectively) b/a amplitude
ratios calculated at the end of the study. Dashed lines around the
regression line indicate 95% CI. Pearson correlation coefficient r,
p-values, and goodness of fit R2 are listed directly in panels
where significance was found. The mean amplitudes corresponding to the ERG b/a
wave ratio between age-matched male and female mice were calculated for scotopic
(E) and photopic (F) responses and stratified according to visual impairment:
normal vision (n = 7), low (n = 18), moderate (n = 15), and severe (n = 8) vision
loss. Blue squares symbolize male mice’s eyes (n = 19), and red triangles
symbolize female mice’s eyes (n = 29). Data are presented as mean
Components of scotopic threshold responses (STRs) (Supplementary Fig. 4A–D) and photopic threshold
responses (PhNRs) (Supplementary Fig. 4E,F) showed statistically significant differences in
middle-aged animals compared to young animals for pSTR, STR amplitude, and PhNR
latency and young female mice compared to young male mice for STR amplitude
(Supplementary Table 1). Oscillatory potential (OP 1-4) amplitudes (Supplementary Fig. 5B,D,F,H) and latency (Supplementary Fig. 5A,C,E,G), thought to be generated by inner
retinal neurons [58], showed statistically significant differences with
15-month-old female mice compared to age-matched male mice and for middle-aged
animals compared to young animals for flash intensities –2.0 and –1.5
cd
Histological studies on retinal tissue sections stained with H&E for male (n =
11) and age-matched female mice (n = 11), stratified according to the degree of
vision loss: normal (n = 2), low (n = 3), moderate (n = 2), and severe (n = 4)
for each sex, are shown by representative images in Fig. 8A. A detailed overview
of retinal layers representing visual impairment shows overall gross changes in
morphology compared to retinal layers representing normal vision. Histological
analysis demonstrated that the IPL for male mice was significantly thinner than
for female mice (37.63 µm
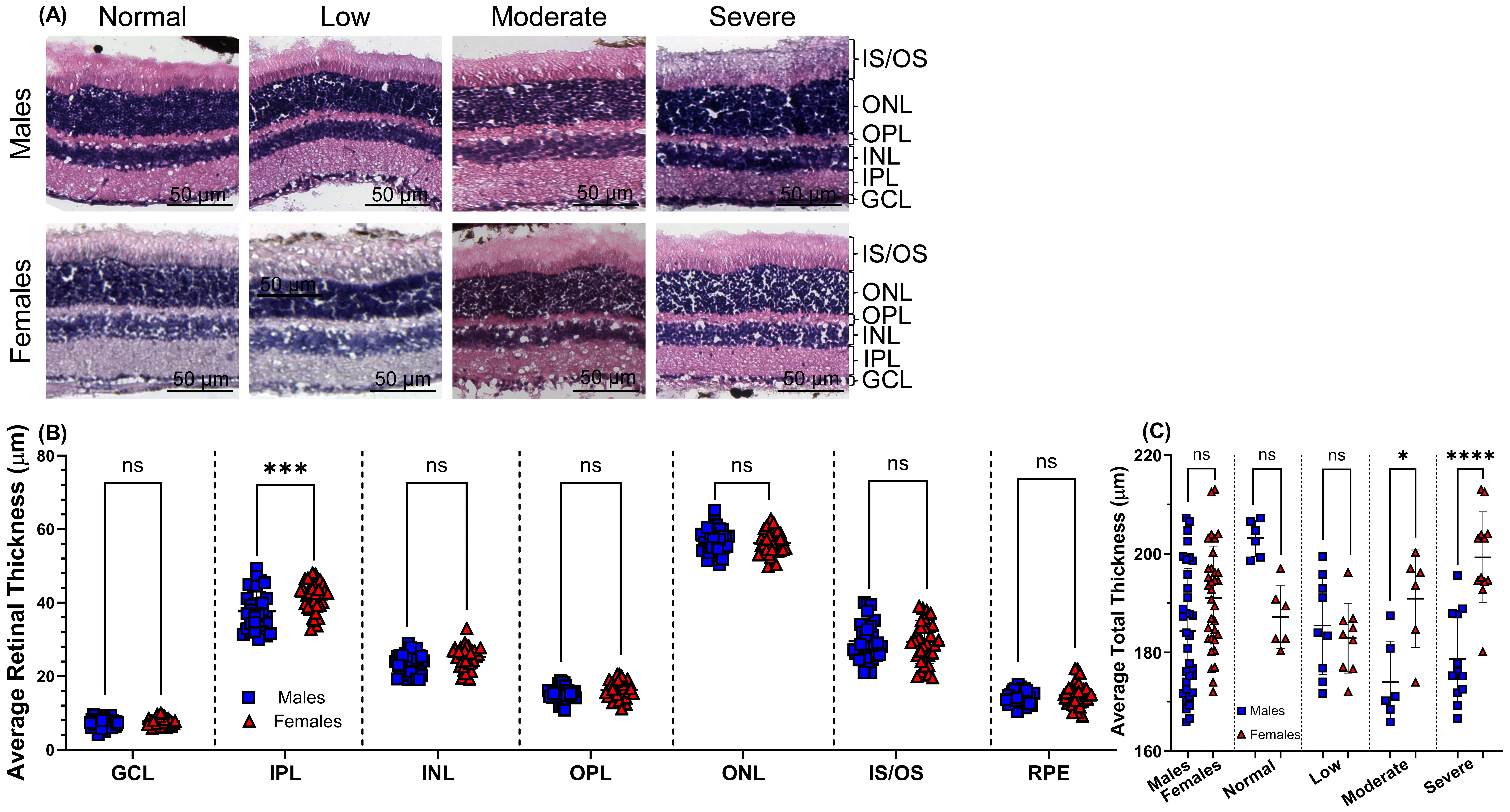 Fig. 8.
Fig. 8.
Differences in retinal morphology of middle-aged C57BL/6J mice.
Representative retinal sections comparing each category of vision loss for
15-month-old males to age-matched female mice eyes and stratified according to
the degree of visual function loss (A). Quantification for individual mean
retinal layer thickness for male (n = 11) and age-matched female (n = 11) mice
eyes (B). The mean total retinal thickness for male and female mice eyes was also
determined and stratified depending on visual impairment: normal vision (n =
6/group), low (n = 9/group), moderate (n = 6/group), and severe vision loss (n =
4/group) (C). Blue squares symbolize male mice eyes, and red triangles symbolize
female mice eyes. Data are presented as mean
Rows of nuclei that make up the INL and ONL were counted, and the resulting data
was stratified based on the severity of vision loss using VA testing as described
above (normal vision, low, moderate, and severe vision loss; Fig. 9A). No
statistically significant differences were noted in the ONL and INL of male and
female retinas. Overall, the mean number of nuclei rows counted in both layers
from middle-aged male mice decreased as vision became more impaired. No
discernible pattern of nuclei row loss was observed in female mice. Counts of
nuclei rows of INL and ONL that were not stratified according to visual
impairment showed no significant differences between male and female retinas
(4.45
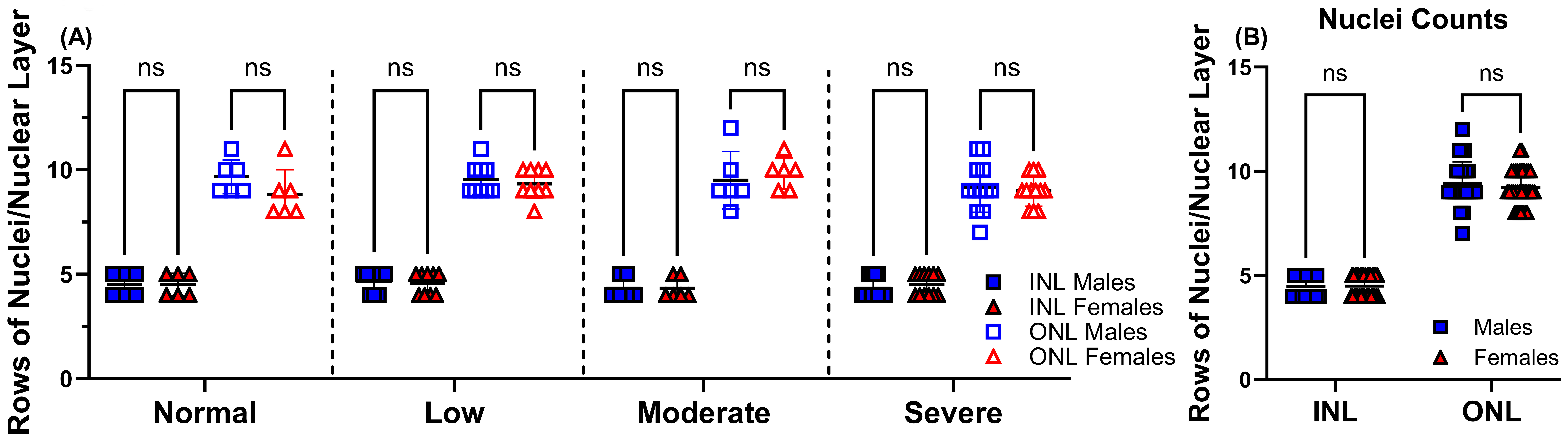 Fig. 9.
Fig. 9.
The number of nuclei rows in the INL and ONL does not vary
depending on visual impairment. Rows of nuclei in the INL (filled symbols) and
ONL (open symbols) were counted for 15-month-old male (n = 11) and age-matched
female (n = 11) mice eyes and stratified according to the degree of vision loss:
normal vision (n = 6/group), low (n = 9/group), moderate (n = 6/group), and
severe vision loss (n = 4/group), determined by behavior assessment of optomotor
reflex for 15-month-old male (blue squares) and female (red triangles) C57BL/6J
mice (A). Rows of nuclei in the INL and ONL were counted for 15-month-old males
(n = 11), represented as blue squares, and females (n = 11), represented as red
triangles (B). Data are presented as mean
Statistically significant sex-specific differences were found in the mean
thickness of the IPL in mice as visual impairment worsened, with male mice
displaying the most significant decrease in IPL thickness with severe vision
impairment (33.65 µm
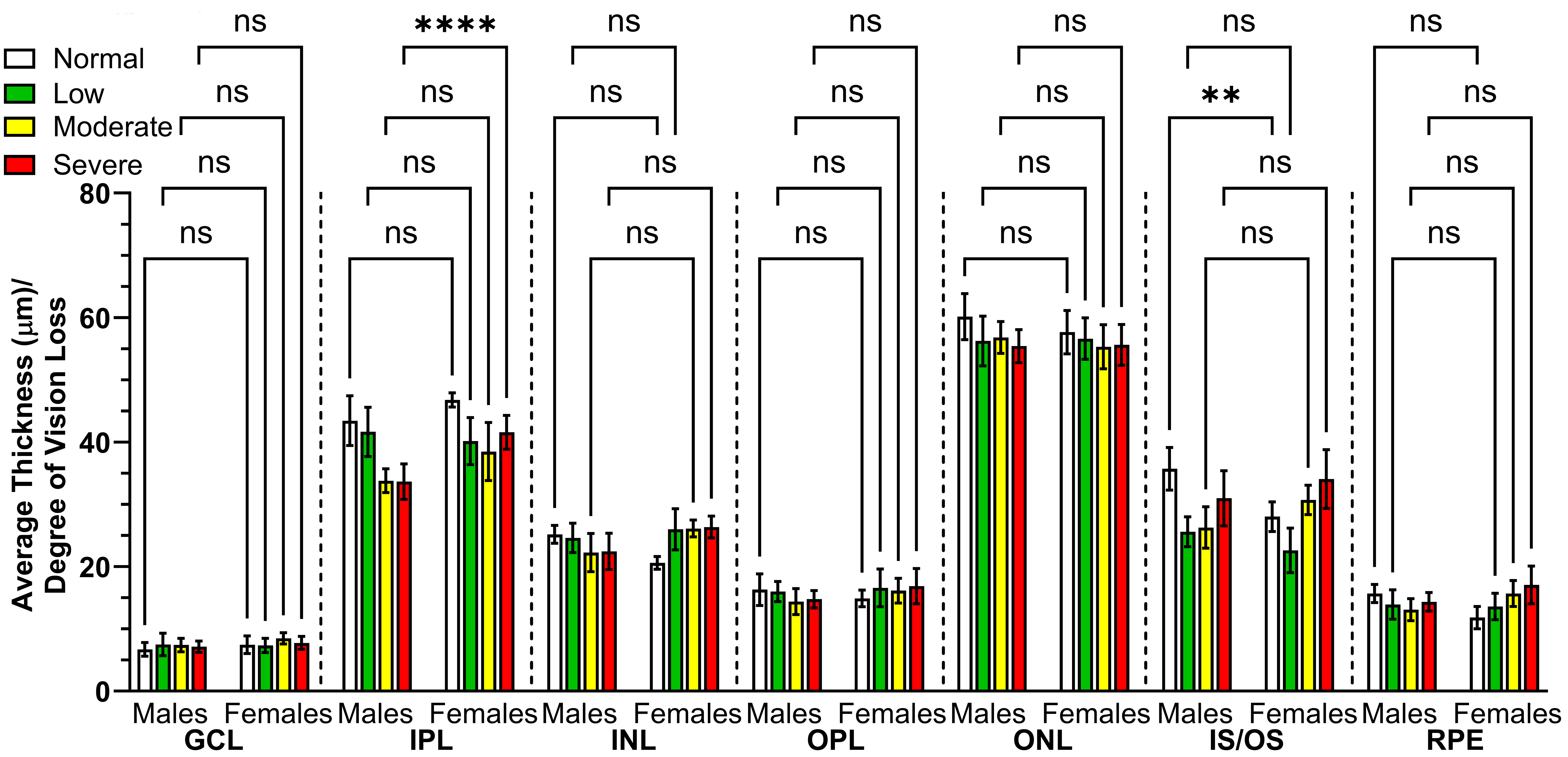 Fig. 10.
Fig. 10.
Disparities observed in the mean thicknesses of the IPL and
IS/OS retinal layers after stratification. The mean thickness of individual
retinal layers for 15-month-old male (n = 11) and age-matched female (n = 11)
mice eyes were stratified according to the degree of vision loss: normal vision
(n = 6/group; white), low (n = 9/group; green), moderate (n = 6/group; yellow),
and severe vision loss (n = 4/group; red), determined by behavior assessment of
optomotor reflex. Data are presented as mean
We determined age- and sex-specific differences in visual function and retinal structure in C57BL/6J mice as potential underlying differential aging effects leading to visual loss. To our knowledge, this is the first study to empirically stratify visual impairment based on behavioral measurements of visual performance, identifying age and sex as biological variables and reproducible parameters in the absence of disease.
In healthy mammals, aging is associated with changes in body weight. Body weight and percent body fat generally increase with age due to the accumulation of body fat and free fat mass [60]. These alterations in body composition can be sex or strain-specific. Results from a recent study by Rathod and colleagues suggest that satiation and satiety in the control of energy intake are regulated in a sex and age-dependent manner, with fat mass accumulation more significant and variable in males than females [61]. The association of sex and age-related weight gain of the mice in this study confirms this finding. Generally, physiological and behavioral factors influence rodent health, particularly responses to visual acuity challenges, which may, in turn, affect their weight [62, 63, 64]. It is also essential to consider genetic makeup, environmental conditions, and the specific type of visual impairment involved. Further research into the weight disparities observed in male and female mice due to visual impairments could provide deeper insights into the physiological and behavioral nuances that may alter rodents’ responses to deficits in visual function.
Visual acuity refers to the highest spatial frequency of a visual stimulus that can trigger a response. On the other hand, CS starts with a stimulus at 100% contrast and is gradually decreased until a threshold is determined. This threshold is identified by examining the tracking behavior exhibited during the OMR [48]. The lower the contrast percentage, the better an animal can distinguish a stimulus in low-contrast environments, such as dusk, dawn, or dense foliage. This test provides a simple and rapid screening of visual function through behavioral observation of the animal and has been used as a first-line screening of vision in rodents [65, 66, 67, 68]. OMR plays a crucial role in maintaining visual stability in mice, and its characteristics are influenced by various factors, including ocular abnormalities and genetic variations in different mouse strains [45, 46, 47, 69]. Our decision to stratify visual impairment was based on VA instead of CS data. This choice was influenced by the superior resolution for fine detail and the increased reproducibility in VA testing. These findings align with reports from other researchers suggesting that the duration of the test could potentially skew CS measurements and affect animal performance.
Furthermore, it is still unclear if the CS impairments associated with specific eye disorders develop quickly or how these impairments vary based on the nature of the pathology [69, 70]. It has been documented in patients that CS deficits may develop at early stages of a specific ocular disease even when VA is relatively normal. Our study has identified several indicators that connect early degenerative changes with CS. Through deep learning, Shamsi and colleagues discovered a significant correlation between the thickness of the human ganglion cell layer, the inner plexiform layer, and CS [71]. In vivo imaging by spectral-domain optical coherence tomography displayed a significantly thinner ONL in male mice but no significant layer differences among the vision impairment groups. OCT analysis also identified a significant correlation between total retinal thickness and CS in male mice. Histological examination found the IPL of male mice was significantly thinner than female mice with severe visual impairment. Thinning of the photoreceptor IS/OS layers was noticeably more pronounced in female mice compared to males with normal vision. These findings suggest a causal relationship with behavioral assessed CS. Contrast sensitivity is a crucial aspect of visual perception and a more comprehensive assessment of visual function than acuity [72] that enables us to discern and perceive variations in brightness levels. However, as we undergo changes in our retina related to age and sex, including a decrease in the density of specific retinal layers, our ability to detect spatial patterns with low contrast is affected.
Impairment of retinal function measured by ERG responses was affected by age to the same extent in male and female mice. ERG amplitude is defined as the maximum displacement of the wave evoked by the stimulus intensity, while implicit time is the time between the flash and peak response [73]. We demonstrated that retinal function decreases with age, evidenced by a reduction in a- and b-waves and delays in implicit times. Statistically significant differences in scotopic b-wave implicit time delays were evident in female retinas compared to male retinas but not when visual function is separated by stratification of vision loss. Delays in scotopic and photopic implicit time indicate outer retinal photoreceptor degenerative changes and reflect the rate at which an electrical signal is at maximum amplitude [74]. No significant differences were found in a- and b-wave amplitudes when the biological sex variable was considered. Our results suggest that sex is a considerable confounding variable concerning implicit time, but not for amplitude, and confirm earlier reports of a decline in retinal function with age [75, 76]. While we identified age-related impairment of both rod and cone photoreceptor cell-mediated vision, the higher susceptibility of rod photoreceptors to age-mediated decline in function when compared to cones is typically a focus of preclinical research [76].
Structural changes in neurons and CNS tissue are common signs of neurodegeneration [77, 78, 79]. SD-OCT enables the non-invasive, cross-sectional visualization of the retina in vivo and the identification of morphological differences at high resolution. Histochemistry of vertical retinal tissue sections is a complementary approach for comparative analysis alongside SD-OCT. The average thickness of the retina, as observed through OCT, aligns well with the thickness determined from histological sections, according to our findings. The structure of the ONL, where photoreceptor cell somata and nuclei are located, serves as an essential biomarker of retina neurodegeneration [56, 57]. The IPL, in contrast, is the area of the retina where synaptic connections between bipolar cells and dendrites of retinal ganglion cells (RGCs) are found [80]. As visual impairment increased with age, the IPL, ONL, and IS/OS layers displayed the most significant differences between male and female mice, but also among other groups of differently reduced VA. As visual impairment increased with age, the concomitant decrease in IPL thickness also resulted in more pronounced differences between males and females as well as among other groups of differentially reduced VA. Visual function measured by behavior assessment of OMR is partly driven by synaptic signaling in the IPL. Impaired motion perception and tracking in human multiple sclerosis (MS) patients are found to be associated with combined RGC/IPL thinning [81] and are associated with deficits in CS, as mentioned earlier in the discussion. A decrease in synaptic density equates to a reduction in IPL thickness.
Direct comparisons between OCT imaging can influence our results from this study due to factors like acquisition region, layer aggregation, or animal age in addition to the process of histological preparation. Differences between OCT and histology quantification can stem from various factors such as imaging techniques, resolution and detail, quantification methods, sample preparation, and the context of use. These differences underscore the distinct nature of OCT and histology, with each technique providing unique advantages based on the specific context of the results. The differences between female and male mice found here exhibit a consistent trend, even though these differences may not be statistically significant. Our findings lend additional support to the research outcomes of other groups, such as results from a study by Batista et al. [82] revealed that total retinal thickness decreases with age, while the thickness of individual retinal layers exhibits varying patterns of change in wild-type C57BL6/129S mice. While the literature consistently reports average total retinal thickness values of approximately 200 µm for wild-type mice, the thicknesses of individual retinal layers or layer aggregates exhibit more significant variability with age. Comparatively, few studies have systematically examined these retinal layers and layer aggregate thicknesses, nor have studies examined how the biological factor of sex affects retinal layer thickness in aging [40, 82, 83, 84]. Fundamentally, a comparison of retinal tissue thickness by OCT and histological analysis revealed that as mice age, the stratification of visual impairment helps disclose the contribution of sex as a biological variable in our results and is most reliably reflected in thickness changes of the IPL, ONL, and IS/OS layers. Retinal layer thickness in normal eyes generally decreases with age and visual impairment in both sexes. Aging studies in mice and rats have observed significant thinning in the IPL, OPL, INL, and ONL [40, 85, 86]. In light of this study, stratifying visual function results to understand the contribution of sex further as a biological variable should be considered in experimental planning and evaluating results. The results presented herein of aging mice can serve as a reference for future studies as we advance our understanding of age-related neurodegenerative disease progression.
In conclusion, the present study provides an essential foundation to further our understanding of age-related pathology’s molecular mechanisms, advance therapy development, and integrate visual performance into both fields. Future work aims to delve deeper into the mechanisms that regulate outcomes associated with slowing the progression of age-related retinal degenerative changes and controlling the therapeutic response. By utilizing the C57BL/6J mouse model and incorporating the additional variable of visual function impairment into the results, we can observe a more accurate estimate of the nature of retinal aging changes alongside differences present from sexual dimorphisms.
The article/supplementary material encompasses the original contributions from the study. For additional queries, please reach out to the corresponding author.
Conceptualization and design, PK. Investigation and data curation, GE, SR, CB, MM, CE, RPM, AR, and AE. Analyzation of data, all authors. Writing, GE and PK. Supervision, PK. GE, SR, CB, MM, CE, RPM, AR, and AE performed experiments, analyzed data, edited and reviewed the manuscript, and approved the submitted version. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The Institutional Animal Care and Use Committee (IACUC) at the University of Missouri-Kansas City (protocol 1902-02) approved all experimental animal procedures, which were performed in accordance with institutional and federal guidelines and the Association for Research in Vision and Ophthalmology (ARVO) Statement for the Use of Animals in Ophthalmic and Vision Research.
We would like to thank Megan Florance, Heather Johnson, and Jacob Player for their technical assistance.
Research reported in this publication was supported in part by a grant from the National Eye Institute (EY030747) of the National Institutes of Health (PK). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. Additional support by the Felix and Carmen Sabates Missouri Endowed Chair in Vision Research, a Challenge Grant from Research to Prevent Blindness, and the Vision Research Foundation of Kansas City is gratefully acknowledged.
The authors declare no conflict of interest. Peter Koulen is serving as one of the Editorial Board members of this journal. We declare that Peter Koulen had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Gernot Riedel.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/JIN25805.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.










