1 Department of Radiology, Shenzhen Hospital of Integrated Traditional Chinese and Western Medicine, 518104 Shenzhen, Guangdong, China
2 Department of Radiology, Jieyang People’s Hospital, 522000 Jieyang, Guangdong, China
3 Department of Ultrasound, Shenzhen Hospital of Integrated Traditional Chinese and Western Medicine, 518104 Shenzhen, Guangdong, China
4 Department of Radiology, The Second Affiliated Hospital of Shantou University Medical College, 515000 Shantou, Guangdong, China
†These authors contributed equally.
Abstract
This study aimed to investigate the diagnostic potential of diffusional kurtosis imaging (DKI) parameters in detecting pathological alterations in the normal-appearing white matter (NAWM) associated with cerebral small vessel disease (CSVD).
A total of 56 patients diagnosed with CSVD were enrolled, all exhibiting confirmed lacunar infarction in the corticospinal tract (CST) as verified by conventional magnetic resonance imaging. A control group of 24 healthy individuals who exhibited no discernible abnormalities on conventional magnetic resonance imaging (MRI) scans was also included. The following DKI parameters were recorded, including mean kurtosis (MK), axial kurtosis (Ka), and radial kurtosis (Kr). Regions of interest were placed at representative levels of the CST on the affected side, encompassing the pons, anterior part of the posterior limb of the internal capsule (PLIC), corona radiata, and subcortex.
Variations in MK, Ka, and Kr values in the pons, anterior part of the PLIC, corona radiata, and subcortex of the control group were observed. Notably, the MK and Kr values of the normal-appearing pons in CSVD patients were significantly elevated compared with the control group. The MK, Ka value of the normal-appearing anterior part of the PLIC was significantly higher in the CSVD group than in the control group. The Kr value of the normal-appearing corona radiata exhibited a significant elevation in CSVD patients compared with the control group. Lastly, patients with CSVD displayed lower Ka values and higher Kr values in the normal-appearing subcortex compared with the control group.
DKI is an effective tool for assessing NAWM in patients with CSVD. These findings potentially offer novel insights into the prognosis of CSVD and serve as a foundational platform for future DKI studies on NAWM in other diffuse brain lesions.
Keywords
- cerebral small vessel disease
- corticospinal tract
- normal-appearing white matter
- diffusional kurtosis imaging
- magnetic resonance imaging
Cerebral small vessel disease (CSVD), referring to a disorder of perforating cerebral arterioles, venules, and capillaries, is one of the most common, chronic, and progressive vascular diseases that contribute to 25% of stroke cases and causes 45% of dementia [1, 2]. With the accelerated aging of the world population, the incidence of CSVD has progressively increased over the years, reaching 11.5–35.7% in people aged 50–80 years [3]. The clinical manifestations of CSVD include cognitive impairment, abnormal gait, dementia, sudden stroke symptoms, and psychiatric disorders, depending on the brain regions affected and the cause of the disease [4]. Given the usually mild clinical symptoms associated with CSVD, neuroimaging has become a crucial diagnostic tool for identifying CSVD [5, 6]. The main neuroimaging characteristics of CSVD consist of white matter hyperintensities or leukoaraiosis, lacunes, brain atrophy, small subcortical infarcts, enlarged perivascular spaces, and microbleeds [7, 8]. Functional and structural abnormalities of brain network connections and neural pathways are vital contributors to the pathogenesis of CSVD [9]. Previous studies of CSVD have primarily focused on investigating the relationship between white matter demyelination and cognitive function [10, 11]. More studies investigating brain network disruptions in CSVD patients are warranted.
The diffusional kurtosis imaging (DKI) technique in magnetic resonance imaging (MRI) has significantly improved the sensitivity and accuracy in evaluating microstructural changes in neural fibers compared to traditional diffusion tensor imaging [12, 13, 14, 15]. A recent study used DKI to explore microstructural alternations in the cingulum of patients with CSVD and found that DKI-derived parameters could be used to characterize mild cognitive impairment in this population [16]. Another study used DKI to characterize white matter in CSVD, focusing on advanced diffusion MRI models to investigate the relationship between brain microstructural alterations and declines in processing speed in CSVD patients [17]. Microstructural changes in the early stage of neuronal degeneration are usually not evident in routine MRI scans, known as normal-appearing white matter regions (NAWM) [18, 19]. In this study, NAWM refers to regions where cerebral infarct lesions have led to early or mild Wallerian degeneration of their downstream nerves without manifesting any abnormalities in conventional MRI signals [20]. We hypothesized that DKI may detect the presence of NAWM in patients with CSVD, thereby providing a more accurate assessment of prognosis and treatment efficacy. Two key factors affecting the accuracy of lesion diagnosis in imaging are the homogeneity of the background and the contrast between adjacent tissues, including the lesion background and adjacent normal tissues. We believe that before studying of NAWM, it is necessary to gain a thorough understanding of the “homogeneity of the background” referring to variations in value uniformity across healthy brain regions in parametric maps, and the contrast of adjacent tissues for each DKI parameter in representative areas of normal brain tissues. A previous study compared the same parameter for different brain structures to identify its variability [21]. Brain mean kurtosis (MK), axial kurtosis (Ka), and radial kurtosis (Kr) provide distinct information in healthy white matter [22]. However, to our knowledge, no studies have specifically focused on these parameters in different brain regions.
In this study, DKI scans were performed on the corticospinal tract (CST) of patients with lacunar infarction. The CST is the largest and most anatomically distinct region of the brain and a key marker of CSVD. Using the CST as the research focus allows for more accurate localization and qualitative analysis of NAWM [23]. The homogeneity of the background and the contrast between adjacent tissues for each parameter of DKI in representative brain regions were explored, and the DKI parameters in different NAWM of the CST were analyzed. We aimed to explore the diagnostic value of DKI parameters in detecting pathological changes in the NAWM associated with CSVD.
In this prospective study, patients diagnosed with CSVD in the Second Affiliated Hospital of Shantou University Medical School between September 2016 and September 2023, with confirmed lacunar infarction in the CST by conventional MRI, were eligible for screening. The inclusion criteria were as follows: (1) the diagnosis of CSVD by two chief neurologists; (2) demonstration of mild neurological dysfunction [24]; (3) confirmation of non-acute lacunar cerebral infarction by conventional MRI [5]; and (4) white matter lesions observed in conventional MRI were consistent with Fazekas Grade 1 [5, 25]. Patients presenting with acute cerebral infarction, cerebral hemorrhage, or infarct lesions exceeding 15 mm in diameter were excluded from the study. The raw data during and after each MRI scan were monitored and inspected. Patients with artifacts were re-scanned. Initially, 60 patients met the eligibility criteria. After the exclusion of three cases due to motion artifacts and one voluntary withdrawal, 56 patients with CSVD were finally enrolled in the CSVD group. Furthermore, we recruited 24 healthy individuals without neurologic symptoms or diseases who displayed no detectable abnormalities on conventional MRI to comprise the Control group. All participants underwent both conventional MRI and DKI scans. This study followed the Declaration of Helsinki and approved by the Research Ethics Committee of the Second Affiliated Hospital of Shantou University Medical College, and all participants or their legal guardians provided written informed consent.
All patients underwent cranial DKI and conventional MRI scans, encompassing T1-weighted images, T2-weighted images, fluid-attenuated inversion recovery, diffusion-weighted imaging, and apparent diffusion coefficient maps. These scans were conducted using a Signa EXCITE HDxT 3.0T whole-body scanner fitted in an 8-channel head coil (GE Medical System, Milwaukee, WI, USA).
The acquisition of DKI data involved a diffusion sequence employing echo planar
imaging with oblique axial slices (thickness/gap: 3.0 mm/1.0 mm). All patients
underwent a whole brain scan within 5 min. The acquisition parameters were
configured as follows: repetition time/echo time = 500/98.6 ms; field of view =
240
Two radiologists at the attending level or above conducted subsequent imaging analyses, following a single-blind protocol and utilizing the DKI software of Functool 9.0 in the GE ADW4.6 workstation (GE Medical System, Rueda Minière, France). The regions of interest (ROIs) were placed at representative levels of the CST on the affected side, including pons, the anterior part of the posterior limb of the internal capsule (PLIC), corona radiata, and subcortex [5, 26]. The ROI area ranges from 5 to 10 mm2, with each ROI measured three times to determine its mean value. In case of disagreement between two radiologists regarding whether the lesion met the inclusion requirements, whether the location of the lesion corresponded to the representative level of the corticospinal tract, or which DKI parameter map most effectively displayed the lesion, a third radiologist at the deputy director level or above would conduct the analysis. A consensus would be reached through discussion.
Both MRI parameters and DKI parameters, including the MK, Ka, and Kr, were recorded. The coefficient of
variation (CV) for MK, Ka, and Kr values in each brain region was also calculated
using the following formula: CV = Standard deviation/mean
Data analysis was conducted utilizing SPSS 26.0 software (IBM SPSS statistics
26.0, Chicago, IL, USA). MK, Ka, and Kr values within distinct brain regions were
categorized as measurement data, and their adherence to a normal distribution was
evaluated through the Shapiro-Wilk test. When these data demonstrated a normal
distribution, they were presented as mean
The CSVD group, comprised of 33 men and 23 women, exhibited a mean age of 61
| Item | CSVD group (n = 56) | Control group (n = 24) | |
| Age (years; mean |
61 |
60 | |
| Male/Female (n) | 33/23 | 9/15 | |
| NIHSS (n) | |||
| Minor/Moderate/Severe | 56/0/0 | 0/0/0 | |
| Risk factors for cerebral infarction | |||
| Hypertension | 35/56 (62.50%) | 4/24 (16.67%) | |
| Type 2 diabetes | 15/56 (26.79%) | 6/24 (25%) | |
| Dyslipidemia | 29/56 (51.79%) | 10/24 (41.67%) | |
| Coronary heart disease | 4/56 (7.14%) | 1/24 (4.17%) | |
CSVD, cerebral small vessel disease; NIHSS, National Institute of Health Stroke Scale; SD, standard deviation.
Note: NIHSS score: minor (1–4); moderate (1–15); severe (15–42).
The MK, Ka, and Kr values of the pons, anterior part of the PLIC, corona
radiata, and subcortex in healthy brains of the Control group were significantly
different (all p
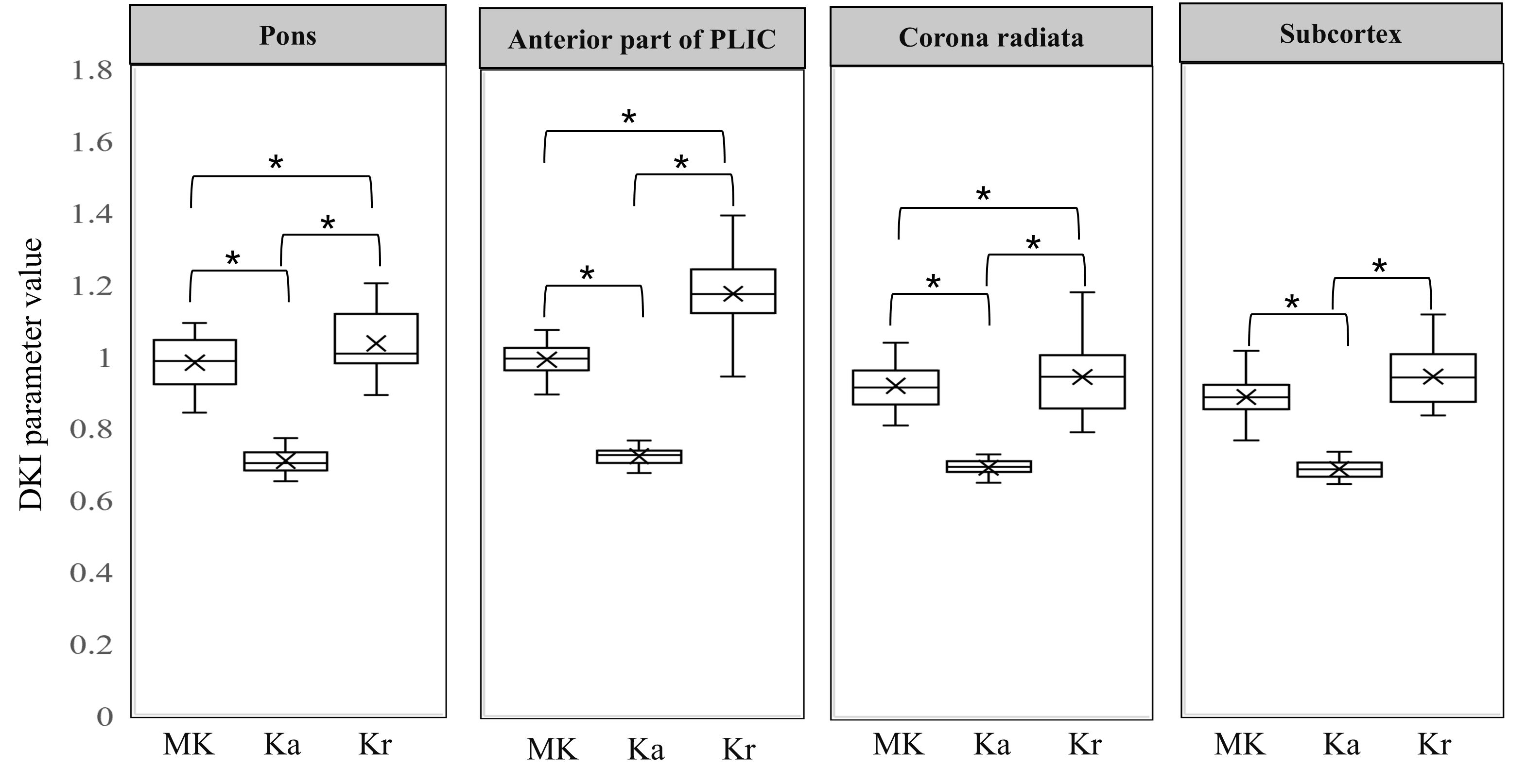 Fig. 1.
Fig. 1.
DKI parameters value of different brain regions in healthy
brains of the control group. DKI, diffusional kurtosis imaging; PLIC, posterior
limb of the internal capsule. * indicates statistical significance among
different DKI parameters (p
According to the conventional MRI, substantial changes in the Ka maps were observed when the non-acute stage lacunar infarction foci were situated in the anterior part of the PLIC (positive rate: 62.5%). Substantial alterations in the Kr maps were found when the lesion was in the subcortex (positive rate: 64.29%). Lesions in the pons and corona radiata were most prominently displayed in the MK maps (positive rate: 48.21% and 39.29%, respectively) (Fig. 2).
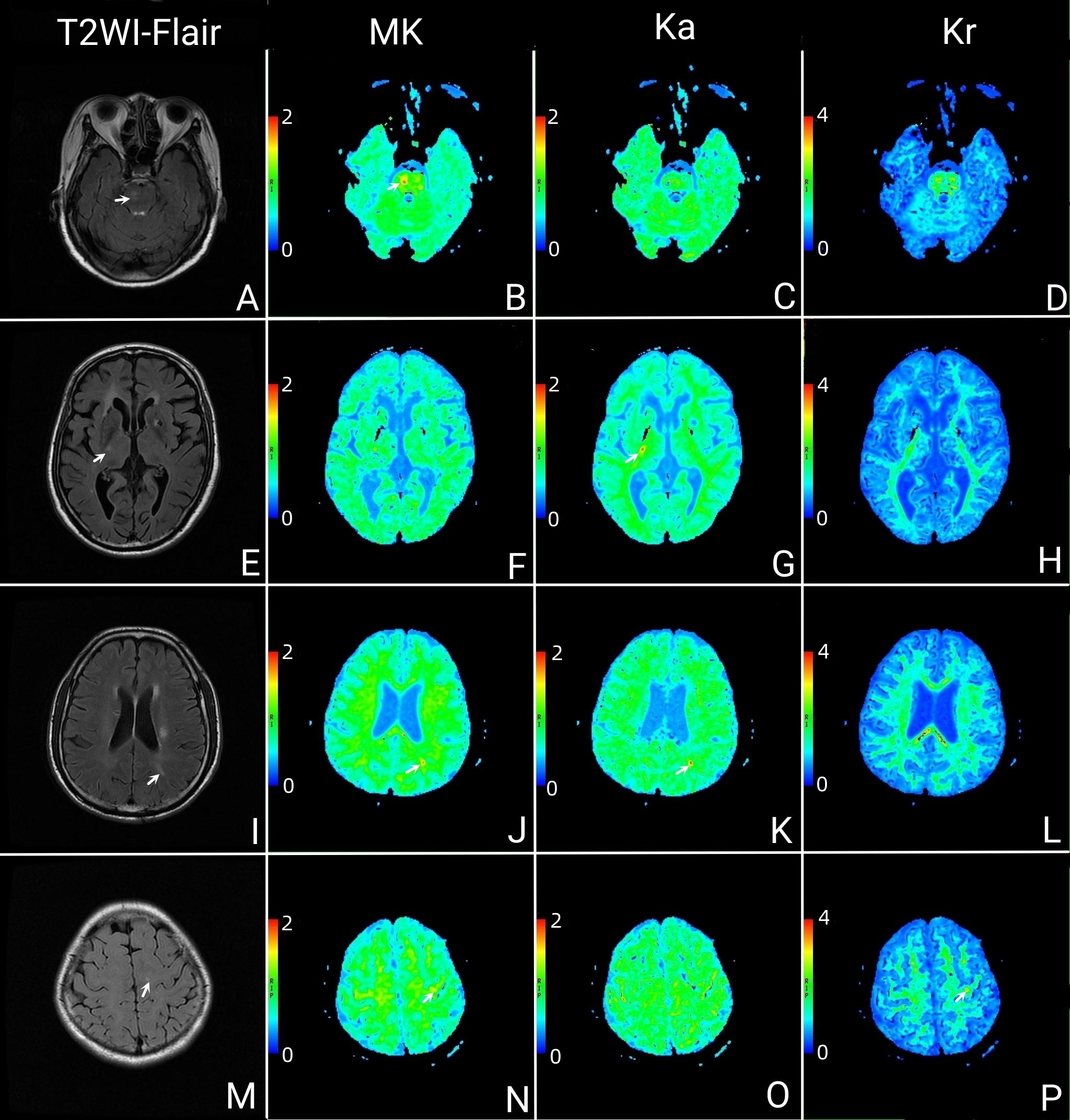 Fig. 2.
Fig. 2.
Characteristics of lacunar infarct foci in different brain regions of the CST on DKI. The lesion within the pons showed high signal on Flair (A, white arrow), and it was most prominently depicted in the MK maps (B, white arrow). Lesions within the pons were barely visible in the Ka maps (C) and Kr maps (D). The lesion within the PLIC showed high signal on Flair (E, white arrow), and it was most prominently depicted in the Ka maps (G, white arrow). Lesions within the PLIC were barely visible in the MK maps (F) and Kr maps (H). The lesion within the corona radiata showed high signal on Flair (I, white arrow). Lesion within the corona radiata was most prominently depicted in the MK maps (J, white arrow), but it was unclear displayed in the Ka maps (K, white arrow) and invisible in the Kr maps (L). The lesion within the subcortex showed high signal on Flair (M, white arrow). Lesion within the subcortex was most prominently depicted in the Kr maps (P, white arrow), but it was unclear displayed in the MK maps (N, white arrow) and invisible in the Ka maps (O). T2WI, T2-weighted images; Flair, fluid-attenuated inversion recovery; CST, corticospinal tract; DKI, diffusional kurtosis imaging. Note: (1) Diagnostic efficacy: The lesions were first found with the naked eye following a single-blind protocol, and the differences between the lesions and the mirrored normal brain areas were measured at the workstation. The DKI parameter map with the greatest difference between the lesion and the mirror normal brain area is defined as the one with the highest diagnostic efficacy (white arrow). (2) The diagnostic efficacy of MK, Ka, and Kr maps in identifying lacunar infarcts in different brain regions displayed variability. (3) The different rows of panels related to different patients.
The MK, Ka, and Kr values of the normal-appearing pons of the
CSVD group were elevated compared with the Control group, with statistical
significance observed in the MK and Kr values (FMK = 23.90, PMK
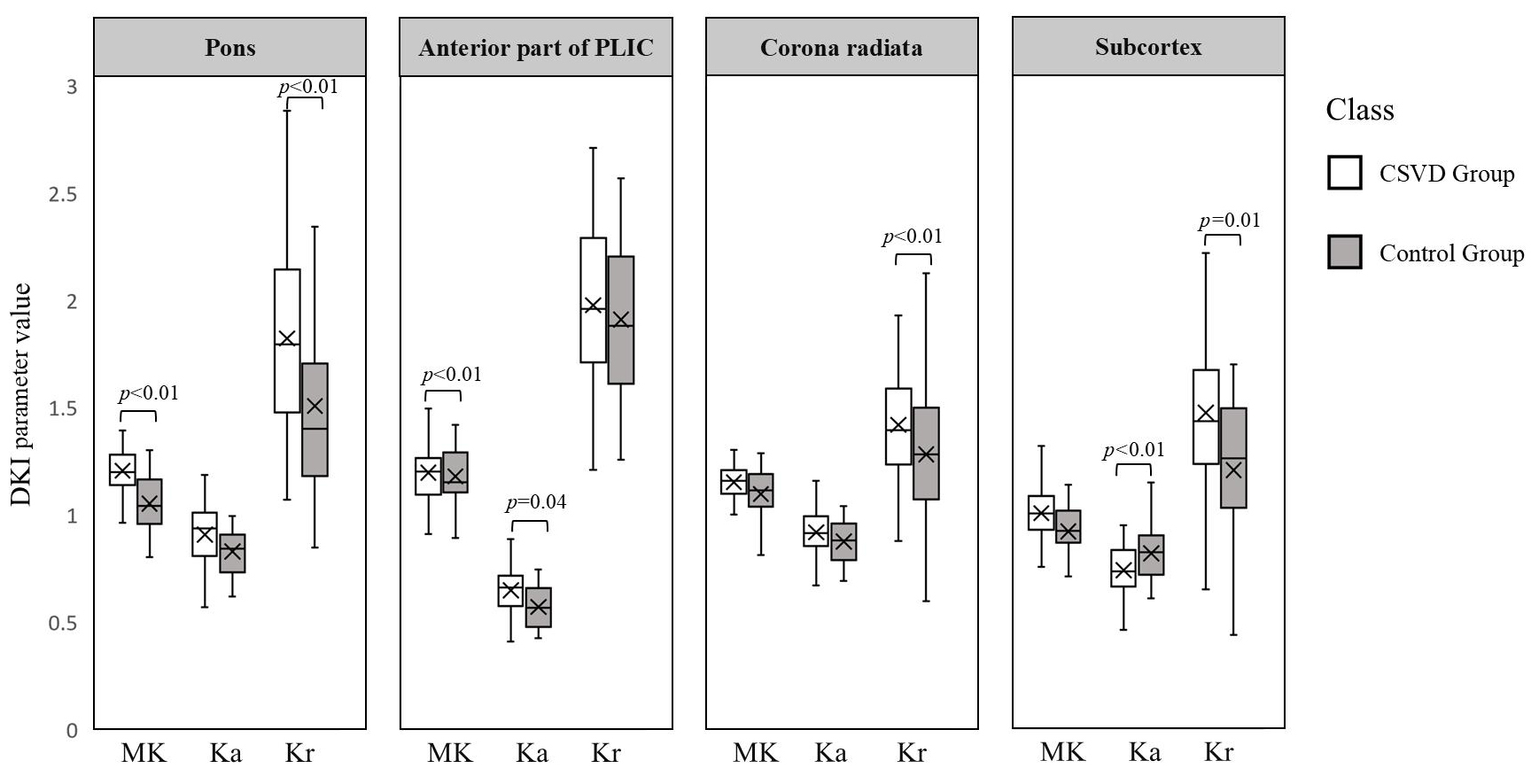 Fig. 3.
Fig. 3.
The MK, Ka and Kr values of different brain regions of the CSVD and Control groups. PLIC, posterior limb of the internal capsule; CSVD, cerebral small vessel disease. Note: The differences (anisotropy) of Brownian motion of water molecules in brain tissues in various directions are closely related to the direction of nerve fiber routing.
Nodular lesions within the brain, which lead to a cascade of neuronal dysfunction or damage, contribute significantly to the pathogenesis of CSVD [27]. Previous investigations have demonstrated that conventional mean diffusivity maps show increased values in white matter lesions but not in NAWM [12]. Brain abnormalities induced by cerebral infarction may be undetected in routine MRI sequences, such as T1 and T2 weighted images, fluid-attenuated inversion recovery, and conventional diffusion-weighted MRI sequences, including diffusion-weighted imaging, apparent diffusion coefficient maps, and diffusion tensor imaging [28]. However, these abnormalities can be effectively identified using DKI [29, 30, 31]. In this study, we demonstrated the presence of NAWM in cases of lacunar infarction within the CST and proved that DKI is a valuable tool for detecting microstructural alterations within these regions.
NAWM refers to regions where cerebral infarct lesions have led to early or mild
Wallerian degeneration of their downstream nerves without manifesting any
abnormalities in conventional MRI signals [20]. Signal heterogeneity in MK, Ka,
and Kr maps plays a pivotal role in assessing NAWM in cases of CSVD [12, 13]. Our
initial findings revealed significant differences among MK, Ka, and Kr in each
brain region of the Control group (all p
When lacunar infarction foci were localized in the anterior part of the PLIC, noticeable changes were observed in the Ka maps. Similarly, significant alterations were detected in the Kr maps when the lesion was situated in the subcortex. Lesions within the pons and corona radiata were most prominently depicted in the MK maps. These findings are consistent with previous reports [12, 13]. A homogeneous background and the contrast between adjacent tissues (including contrast between lesion-background and contrast between adjacent normal tissue) in the DKI maps are the two pivotal factors for the comprehensive analysis of NAWM in cases of CSVD.
The differences (anisotropy) of Brownian motion of water molecules in brain tissues, which varies in different directions, are closely related to the direction of nerve fiber routing [13]. The values of DKI parameters (i.e., MK, Ka, and Kr) differ in different brain regions. Ka reflects the Brownian motion of water molecules in the direction of the axons. Kr represents the influence of the hydrophobicity of the axonal myelin sheath on the Brownian motion of water molecules. MK indicates the complexity of the local microstructure. These DKI parameters have been extensively discussed in previous DKI studies, and our results are consistent with these findings. The pons represent a region where nerve fibers converge with relatively consistent directional alignment, displaying relatively high anisotropy [13]. In the case of CSVD patients, the MK, Ka, and Kr values of the pons were elevated compared to the Control group, with statistically significant differences observed in the MK and Kr values. In the anterior part of the PLIC, nerve fibers exhibit the highest level of anisotropy, indicating directional consistency [13]. Due to the elevated background signal in this region, the Ka value showed a significant increase in CSVD patients compared to healthy subjects. In contrast, the corona radiata primarily consists of divergent nerve fibers characterized by low directional consistency and anisotropy [13]. In this case, the Kr value of the corona radiata was significantly higher in the CSVD group than in the Control group. Lastly, the subcortex primarily comprises arcuate fibers, exhibiting the lowest directional consistency and anisotropy compared to the pons, the anterior part of the PLIC, and corona radiata. CSVD patients displayed lower Ka and higher Kr values in the subcortex compared to healthy individuals. These discrepancies in the changes of Ka, MK, and Kr values may suggest compensatory alterations in proximal axons due to injury. Previous animal studies have indicated aberrant expression of neuro-related antibodies, such as neurofilament-200 and glial fibrillary acidic protein, at different stages of cerebral infarction [32, 33, 34]. Further studies are required to ascertain whether NAWM in patients with CSVD is linked to compensatory changes in proximal nerve fibers.
The current study is limited by a relatively small sample size. Future investigations are needed to enhance statistical power by enlarging the sample size, allowing for a more comprehensive analysis of subgroups within the case cohort. Additionally, longitudinal follow-up studies on a patient cohort are warranted to provide insights into the dynamic progression of CSVD. In addition, using 15 gradient directions for DKI scanning is insufficient, as it may compromise the accuracy and precision of the estimates. Finally, to enhance the understanding of the microstructural changes in minor vessel diseases, it is necessary to improve the establishment of diffusion models, such as the reproducibility of diffusion metrics.
DKI is an effective method for evaluating NAWM in patients with CSVD. We recommend establishing evaluation criteria based on the anisotropy and background signal of NAWM. Our findings may offer new perspectives for the prognostic assessment of CSVD and serve as a basis for DKI studies of NAWM in other diffuse brain lesions, such as multiple sclerosis and neuromyelitis optica.
The data sets generated and/or analyzed during the current study are not publicly available due to their containing information that could compromise the privacy of research participants, and some of which will also be used for further research.
YG, SC, HR, WZ and LK conceived and designed the experiments. YG, SC, HR, WL, QL, FL and YW performed the experiments. SC, LK, WL, FL and YW analyzed the data. HR, LK, WL, QL, FL and YW prepared the figures and tables. SC wrote the manuscript. YG and WZ revised it critically for important content. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This study was follow the Declaration of Helsinki and approved by the Research Ethics Committee of the Second Affiliated Hospital of Shantou University Medical College, and all participants or their legal guardians furnished written informed consent (approval number: 2016-29).
Not applicable.
This research was funded by the Natural Science Foundation of Shenzhen (JCYJ20190808095403639), Shenzhen Bao’an TCM Development Foundation (2022KJCX-ZJZL-4) and the Sanming Project of Medicine in Shenzhen (AZZYSM 202106006).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



