1 Department of Neurobiology, University of Texas Medical Branch, Galveston, TX 77555, USA
Piezo2, a pressure-activated channel that mediates touch, proprioception, and blood-pressure sensitivity in the peripheral nervous system [1], is also expressed in select brain neurons [2] where it can generate local electric field potentials in response to heartbeat-driven arterial pressure pulsations [3]. However, defining a specific function for Piezo2 in the brain has remained elusive, largely because of the difficulty of selectively stimulating Piezo2-expressing neurons in behaving animals. Liu and colleagues [4] have now met this challenge by using a precision magnetic stimulation system (pMSS) to activate Piezo2-expressing oxytocinergic neurons, labelled with paramagnetic nanobeads within the paraventricular nuclei (PVN) of mice, and by testing how different pulsatility-frequency protocols affect behavior.
Liu et al. [4] demonstrated that applying pMSS at 10 Hz for 10 minutes per day over 7 days upregulated Piezo2 expression, enhanced Ca2+ influx, and increased oxytocin release. The same 10 Hz treatment also activated phosphatidylinositol 3-kinase and protein kinase B (PI3K/Akt) signaling and promoted neurite sprouting, leading to improved social behavior in wild-type (WT) mice [4]. Prior genetic knockdown of Piezo2 abolished these effects. Moreover, using a valproic acid (VPA) mouse model of autism spectrum disorder (ASD), it was shown that the 10 Hz pMSS protocol activated Piezo2 to increase Ca2+ influx and oxytocin secretion, improved neuronal morphology, and rescued social deficits. Piezo2 knockdown blocked this rescue and further worsened social deficits, anxiety, and repetitive stereotyped behaviors.
A particularly intriguing finding of Liu et al. [4] was the bidirectional effects of pMSS frequency. In contrast to the beneficial, prosocial effects of 10 Hz stimulation, 1 Hz pMSS decreased Piezo2/oxytocin signaling and reduced sociality, phenotypically mimicking ASD-like behavior [4]. These opposite effects underscore that pulse frequency is not a trivial parameter. At 1 Hz, the slower magnet rotation generates longer-duration individual pulses, producing a prolonged mechanical ‘drag’ on the neuron-nanoparticle complex, akin to a tug. In contrast, the rapid rotation at 10 Hz yields much briefer pulses, approximately ten times shorter, similar to a tap. Interestingly, 5 Hz stimulation produced no measurable effects, possibly reflecting coherence with the mouse resting heart rate (~5 Hz), a regime in which endogenous arterial pulses may already optimally engage Piezo2 (see below).
The opposite frequency-dependent outcomes indicate that while 10 Hz stimulation activates mechanosensory signaling, slow 1 Hz trains of longer-duration pulses may inactivate the same pathway, effectively producing a reversible, non-genetic ‘Piezo2-deficient’ state. This interpretation parallels earlier pressure-clamp experiments by Maroto et al. [5], who demonstrated that repetitive, long-duration (~1 s) pressure pulses induced progressive desensitization of Piezo-like channel responses, whereas shorter (~100 ms) pulses preserved channel responsiveness (see Fig. 1, Ref. [5]). Directly measuring Piezo2 channel kinetics in cultured hypothalamic neurons [4] under different pulse protocols will clarify whether frequency-dependent Piezo2 desensitization underlies the bidirectionally pMSS effects. Finally, because Piezo-like mechanosensitive channels are also expressed in certain human tumor cells [5], analogous frequency-dependent pMSS protocols may hold translational relevance for modulating tumor growth and metastatic behavior.
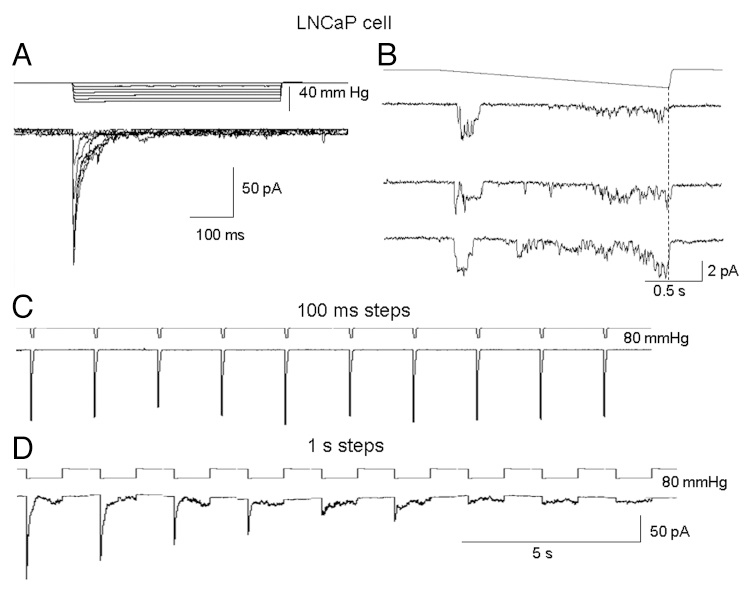 Fig. 1.
Fig. 1.
Pulse-duration–dependent desensitization of pressure-activated Piezo-like currents. (A) Step increases in membrane pressure evoke transient currents in a human prostate tumor cell (Lymph Node Carcinoma of the Prostate, LNCaP) that rapidly inactivate to baseline within ~200 ms. (B) A continuous pressure ramp activates a transient inward current at ramp onset that decays back to baseline despite progressively increasing pressure; later in the ramp, a noisy current is observed, which terminates upon release of pressure. (C) A train of brief (100 ms) pressure pulses evokes reproducible transient currents with no change in peak amplitude or inactivation kinetics across pulses, indicating little or no response desensitization. (D) In the same membrane patch, a train of prolonged (1 s) pressure pulses produces progressive desensitization (“fading”) of the transient peak current along with an increase in the sustained current component; by the ninth pulse, the peak current is reduced by ~90%, from an initial ~150 pA transient to a small, sustained current of ~15 pA. Reproduced from Ref. [5]: Maroto R, Kurosky A, Hamill OP. Mechanosensitive Ca2+-permeant cation channels in human prostate tumor cells. Channels (Austin). 2012; 6: 290–301.
The Liu et al. [4] study thus introduces both a mechanosensory model of ASD and a powerful tool for dissecting and potentially treating abnormal mechanical rhythms that suppress oxytocinergic and affiliative circuits. Related work revealed that pMSS targeted to the prefrontal cortex alleviates depressive-like behavior in mice [6]. Furthermore, transcriptomic data from the genotype-tissue expression (GTEx) project ([7], see also Supplementary Fig. 2D in [2]) indicate that Piezo2 is expressed in other deep brain nuclei, including the nucleus accumbens, amygdala, and substantia nigra — that orchestrate motivation, emotion, motor control, and reward. These regions may also be amenable to therapeutic pMSS modulation. If so, the classic divide between ‘mechanical’ and ‘electrical’ brain stimulation may dissolve into a unified paradigm in which rhythmic energy transduction, including mechanical, electromagnetic, and hormonal, collectively contributes to the therapeutic modulation of dysfunctional brain circuits.
A key question now emerging is: what physiological mechanical rhythms
normally regulate Piezo2/oxytocin affiliative circuits? A compelling candidate
involves heartbeat-driven intracranial pressure (ICP) pulsations (2, 3, 8), which
operate within the frequency domain of heart-rate variability (HRV) and
cardiorespiratory resonance promoted by slow-paced breathing [8]. Supporting this
idea, Buron et al. [9] recently showed that oxytocin release from the
PVN amplifies respiratory-related HRV (i.e., respiratory sinus arrhythmia) via a
hypothalamus–brainstem–cardiac parasympathetic pathway. Specifically,
oxytocinergic projections to the pre-Bötzinger complex enhance glycinergic
inhibition onto cardiac-innervating parasympathetic neurons during inspiration,
thereby increasing HRV amplitude during calm behavioral states ([9], see also
[10]). Thus, oxytocin release may both respond to mechanical pulsatility
associated with slow breathing [8, 11] and feed back to strengthen vagal-cardiac
coupling. This reciprocal dynamic suggests a self-reinforcing HRV
Together, these converging lines of evidence from magnetic stimulation, Piezo2-oxytocin signaling, cardiorespiratory physiology, and social neuroendocrinology support an emerging mechanosensitive-brain hypothesis in which Piezo2 acts as a molecular transducer, linking oxytocin to the mechanical energy of cardiovascular and respiratory pulsations, thereby shaping neural and emotional rhythms. The study by Liu et al. [4] therefore represents a milestone showing that frequency-specific mechanical entrainment can modulate neurohormone-mediated prosocial signaling and opens a new frontier in mechanotransducive neurotherapeutics.
OPH participated in the conception, design, data collection and processing, analysis and interpretation, literature review, writing, and critical review of the manuscript. The author read and approved the final manuscript. The author has participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
The author declares no conflict of interest. Owen P. Hamill is serving as one of the Editorial Board members and a Guest editor of this journal. We declare that Owen P. Hamill had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Bettina Platt.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

