1 Department of Pharmacology and Laboratory of Aging and Nervous Diseases, Jiangsu Key Laboratory of Drug Discovery and Translational Research for Brain Diseases, College of Pharmaceutical Sciences, Soochow University, 215123 Suzhou, Jiangsu, China
2 Department of Anesthesiology, The Second Affiliated Hospital of Soochow University, 215004 Suzhou, Jiangsu, China
Abstract
Exosomes are extracellular vesicles that carry a variety of biomolecules, including nucleic acids, proteins, and lipids, and they play a vital role in intercellular communication. These endogenous carriers offer several advantages over conventional nanocarriers, such as liposomes. These advantages include high biocompatibility, low immunogenicity, and the ability to cross biological barriers such as the blood–brain barrier, making them a promising platform for targeted drug delivery. In this review, we systematically summarize the biological characteristics of exosomes, methods for their isolation and purification, strategies for drug loading (including endogenous and exogenous approaches), and surface engineering techniques (such as genetic engineering and chemical modification) to enhance targeting and therapeutic efficacy, based on a comprehensive PubMed literature search. We particularly focus on the modification of engineered exosomes as drug delivery systems in various clinical contexts, covering multiple diseases including cancer, diabetes, neurological diseases, cardiovascular diseases, and tissue repair. Administration routes include oral, subcutaneous, intranasal, and intravenous delivery. While exosomes have shown promise in preclinical studies, challenges remain in terms of large-scale production, standardized isolation, drug loading efficiency, and safety evaluation. Herein, we aim to provide a theoretical foundation and suggest future directions for developing exosomes as a next-generation drug delivery platform.
Graphical Abstract
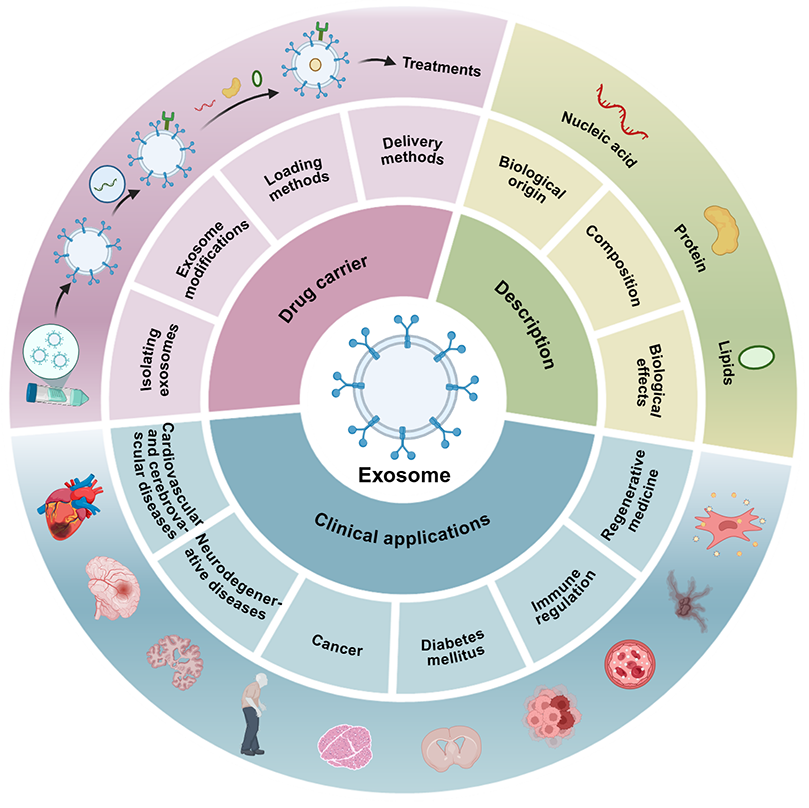
Keywords
- exosomes
- drug delivery systems
- nanomedicine
- cardiovascular diseases
- blood-brain barrier
- diabetes mellitus
- neoplasms
- translational medical research
The term ‘exosome’ was coined by Dr Rose Johnstone in the 1980s during her research into the maturation of reticulocytes [1]. While initially considered cellular debris involved in removing unnecessary proteins and molecular components from cells, they are now recognised as key mediators of intercellular communication. They play essential roles in signal transduction, immune homeostasis, and physiological regulation. There is accumulating evidence that supports the significant impact of exosomes on human health and disease pathogenesis through diverse mechanisms, including developmental regulation, immune modulation, cancer progression, and involvement in neurodegenerative disorders. Furthermore, engineered exosomes with targeted modifications have shown promise in the treatment of diabetic neuropathy and wound healing [2]. Exosomes also show promise in the diagnosis and treatment of various related diseases [3]. Their intrinsic stability in circulation and capacity to deliver functional RNA and small-molecule therapeutics across intact biological barriers, such as the blood–brain barrier (BBB), make them highly effective vehicles for biopharmaceutical delivery. Due to these unique biological properties, there has been increasing interest within the scientific community in using exosomes for the targeted delivery of therapeutic agents or functional nucleic acids to specific pathological tissues [4]. This review summarises recent advances in exosome isolation techniques and current strategies to enhance yield, based on a comprehensive literature search of the PubMed database (https://pubmed.ncbi.nlm.nih.gov). Particular focus is given to engineering approaches for exosome modification and their use as drug delivery systems in various clinical contexts. The cited references encompass all relevant studies on exosomes published up to September 2025, covering a range of diseases.
Extracellular vesicles (EVs) exist in several forms, including apoptotic bodies,
microvesicles, and exosomes. Due to substantial overlap in size, density, and
membrane protein composition between these three subtypes, there are currently no
definitive features or specialised techniques to distinguish between them
reliably. Currently, EVs are classified based on biochemical markers or size
parameters, categorising them as medium/large (
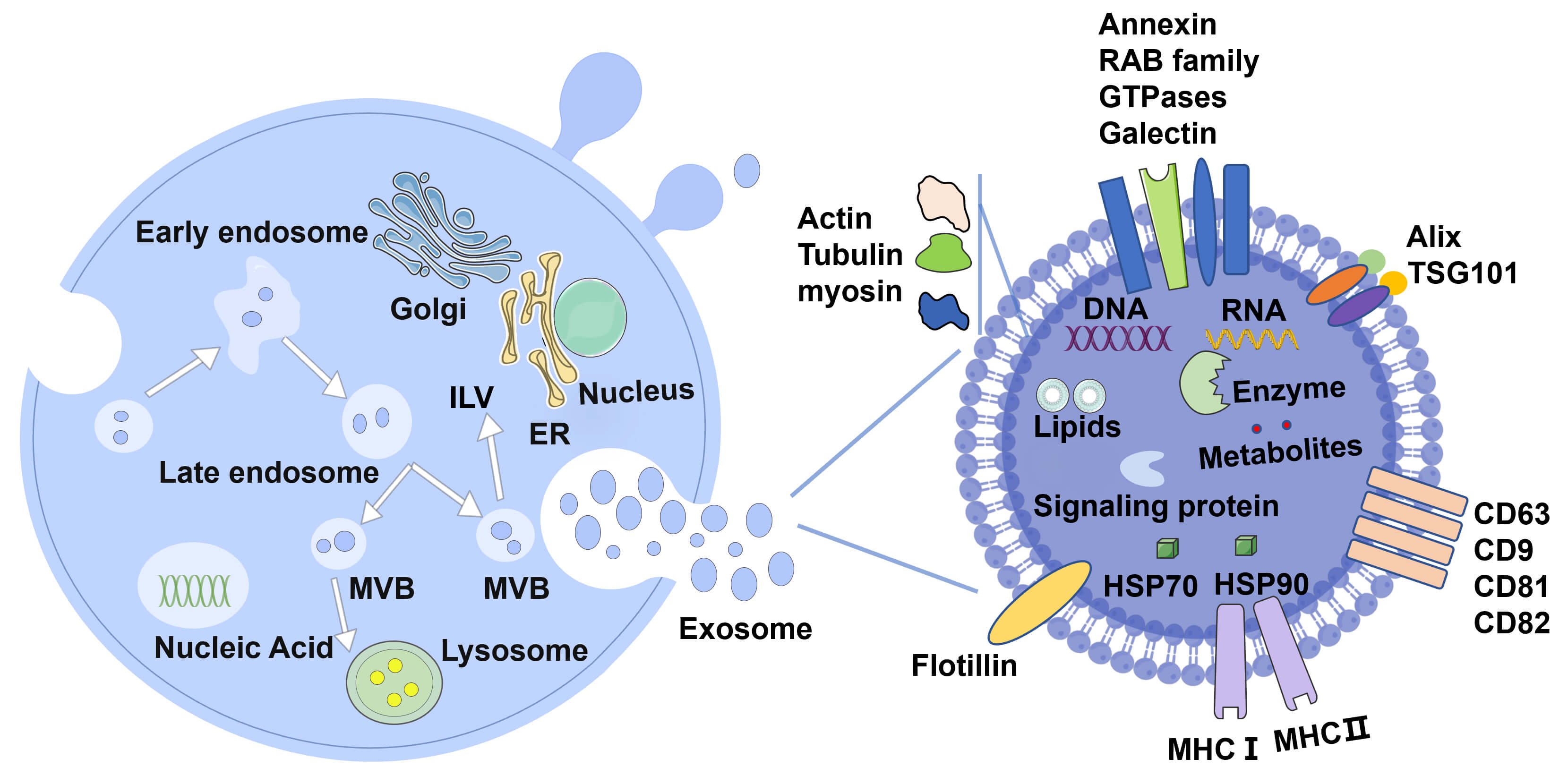 Fig. 1.
Fig. 1.
Exosomes: origin and composition. Exosomes are released by diverse cell types, including immune cells, stem cells (e.g., mesenchymal stem cells), and neurons, among many others. Their biogenesis begins with the endosomal pathway. Early endosomes mature into late endosomes or multivesicular bodies (MVBs). The budding and fusion of the MVB membrane lead to the formation of intraluminal vesicles (ILVs). Some MVBs fuse with lysosomes for degradation, while others fuse with the plasma membrane to release as exosomes into the extracellular environment. Exosomes carry a diverse array of biomolecules in their lumen or on their surface, including nucleic acids (DNA and RNA), proteins, lipids, metabolites, signaling molecules, and receptors. Proteins involved in exosome biogenesis including cytoskeletal proteins (actin, tubulin, myosin), transport/binding proteins (Galectin, GTPases, RAB family, Annexins), ESCRT components, heat shock proteins (HSP70, HSP90), and commonly used exosome markers, such as CD9, CD63, CD81, CD82, TSG101, Alix, Flotillin, MHC-I and MHC-II, are also present on exosomes. ESCRT, endosomal sorting complexes required for transport; GTPases, GTP hydrolases; RAB, ras-associated binding; CD, cluster of differentiation; MHC, major histocompatibility complex; TSG101, tumor susceptibility 101; ER, endoplasmic reticulum.
Exosomes are primarily composed of cytoplasmic and cell surface metabolites, including lipids, proteins, and nucleic acids (such as RNA and DNA) [12]. They are characterised by enriched lipid bilayers that maintain their integrity and protect biological activity [14, 15]. Various lipids, such as phosphatidylinositol, phosphatidic acid, cholesterol, sphingomyelin, sphingolipids, phosphatidylserine, and ceramides, have been identified within exosomes. Evidence suggests that modulating cholesterol levels in the exosomal membrane enhances cargo transfer via membrane fusion-mediated mechanisms [16]. Exosomes contain numerous nucleic acid molecules, including RNA (such as messenger RNA [mRNA], microRNA [miRNA], and other types of non-coding RNA) and DNA. Notably, miRNAs in exosomes are more abundant than in parental cells and body fluids [17, 18]. miRNAs are short non-coding RNA segments of around 20 nucleotides. Target mRNAs can have their translation inhibited by miRNA sequences that selectively bind to their 3’ untranslated region. Thus, miRNAs are essential for controlling several biological functions, including differentiation, cell division, and programmed cell death [19]. Exosomes are structurally abundant in transmembrane proteins, receptors, and functional molecules, including adhesion molecules such as tetraspanins (cluster of differentiation (CD)37, CD63, CD9, CD53, CD82, CD81 and integrins), cytoskeletal proteins (actin, tubulin, cofilin and moesin), transport/binding proteins (galectin, GTP hydrolases (GTPases), ras-associated binding (RAB) family, and Annexins), ESCRTproteins (Alix and tumor susceptibility 101 (TSG101)), and cofactors such as vacuolar protein sorting 4 (VPS4) [20, 21], heat shock proteins (HSP70 and HSP90) and lipid raft components (Flotillin) [22]. Flotillin, TSG101, CD81, and CD63 are the most frequently utilised marker proteins for exosomes [23]. Exosomes also contain integrin-related membrane signalling proteins, including the mast/stem cell growth factor receptor (c-Kit, also known as stem cell factor receptor (SCFR) or CD117) and the epidermal growth factor receptor (EGFR) [24], as well as RNA-editing enzymes, lipases, proteases, glycosyltransferases, glycosidases, and metabolic enzymes. These contribute to the functional versatility of exosomes in intercellular communication [24] (Table 1).
| Category | Representative components | Biological functions |
| Lipids | Cholesterol, Sphingomyelin, Phosphatidylserine, Ceramide | Form bilayer structure |
| Mediate membrane fusion | ||
| Enhance cargo transfer | ||
| Proteins | Tetraspanins (CD9, CD63, CD81), Integrins | Cellular adhesion & uptake |
| Serve as identification markers | ||
| Proteins | ESCRT proteins (Alix, TSG101), Heat shock proteins (HSP70, HSP90) | Facilitate exosome biogenesis |
| Maintain protein stability | ||
| Proteins | Signaling Receptors (e.g., c-Kit, EGFR), Metabolic Enzymes | Activate signaling pathways in recipient cells |
| Remodel the recipient cell metabolism | ||
| Nucleic acids | miRNAs, mRNAs, Noncoding RNAs | Regulate gene expression |
| Inhibit mRNA translation | ||
| Other components | Metabolic intermediates, Cytoskeletal proteins | Maintain structural integrity |
| Reflect the cellular metabolic state |
EGFR, epidermal growth factor receptor; c-Kit, stem cell factor receptor.
The composition of exosomal contents largely depends on their cellular origin. For example, adipose tissue-derived (AD-) versus bone marrow-derived (BM-) mesenchymal stem cells (MSCs) comprise different miRNA and transfer RNA (tRNA) sets [25]. Furthermore, exosomes originating from mast cells can promote the proliferation of B and T lymphocytes in both experimental settings and living organisms. These exosomes also display markers such as major histocompatibility complex (MHC)-II, CD86, LFA-1 (lymphocyte function-associated antigen 1), and ICAM-1 (intercellular adhesion molecule 1) [26].
Exosomes are typically taken up by recipient cells via three primary mechanisms: endocytosis, membrane fusion, and ligand-receptor interaction (Fig. 2). Endocytosis is the main mechanism by which recipient cells absorb exosomes and includes phagocytosis, clathrin-mediated endocytosis, macropinocytosis, and lipid raft-mediated endocytosis. During membrane fusion uptake [27], exosomal transmembrane proteins interact directly with receptors on the recipient cell membrane [28], leading to the integration of the exosomal membrane with the plasma membrane and the direct release of contents into the cytoplasm of the recipient cell [29]. The exosomal membrane also integrates into the recipient cell’s membrane [30]. Additionally, exosomes can also facilitate intercellular communication through ligand-receptor interaction, similar to mediating antigen presentation.
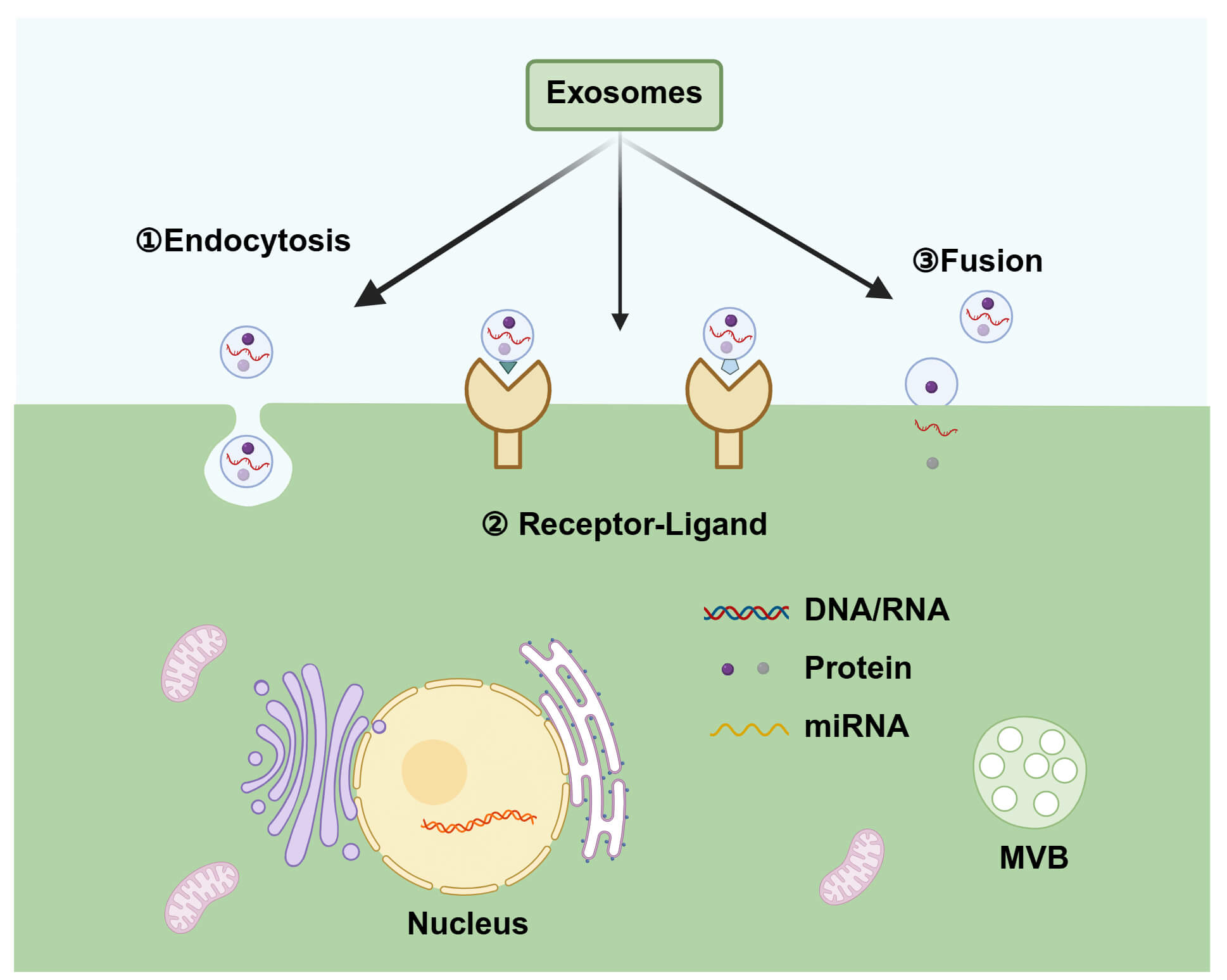 Fig. 2.
Fig. 2.
Mechanisms of exosome uptake and cargo delivery by recipient cells. After secretion, exosomes deliver their cargo through several mechanisms, including endocytosis, receptor-ligand interactions, and membrane fusion. Note that upon membrane fusion, the exosomal membrane integrates with the plasma membrane, resulting in the direct release of its contents into the cytoplasm. miRNA, microRNA. Created in BioRender (https://BioRender.com/).
The protein composition of the exosomal membrane directly facilitates cellular uptake. Specific surface proteins, such as tetraspanins (e.g., CD9 and CD81) and integrins, promote adhesion and internalisation via endocytosis. Furthermore, specific viral or glycoproteins can induce membrane fusion, enabling the direct delivery of exosomal cargo into the cytoplasm [31].
To date, exosomes have been found to participate in nearly all investigated physiological and pathological activities, and they can deliver a wide variety of biomolecules to specific target cells via regulated secretion mechanisms [32]. They are capable of utilising endocrine, paracrine, or autocrine pathways to execute their functions [33]. Following uptake by recipient cells through the mechanisms above, they elicit a variety of biological effects. Firstly, they contribute to cellular homeostasis and survival by eliminating harmful substances from parent cells [34]. Secondly, they can deliver functional cargo, such as proteins and various RNA species (miRNA, mRNA, and non-coding RNA), as well as DNA fragments, to neighbouring or distant cells. During this process, the cargo can be delivered to the cytoplasm or nucleus of recipient cells, and membrane components can be transferred to the recipient cell’s membrane [35, 36]. The introduction of these exogenous molecules can initiate cascading reactions that modulate signaling pathways, gene expression, and overall functionality in recipient cells.
For example, the secretion of exosomes by fibroblasts in both the peripheral and central nervous systems, macrophages, and Schwann cells can promote regenerative and developmental processes in the nervous system. Exosomes secreted by adipose-derived stem cells (ADSCs), MSCs, neurons, and glial cells are essential for mediating angiogenesis and remodelling of the neurovascular unit. These exosomes act as key signalling molecules that promote the repair and regeneration of the neural and vascular systems. Meanwhile, it has been found that microglia, oligodendrocytes, and MSCs mediate cell signal transduction, intercellular communication, nervous system growth and repair, vascular remodelling, and the preservation of homeostasis in the nervous system by secreting exosomes [37]. Furthermore, exosomes are involved in immune regulation. Exosomes from antigen-presenting cells play a crucial role in the immune response, facilitating intercellular communication within the immune system by activating specific cell surface receptors, for example. Antigen-presenting cells, especially dendritic cells (DCs), release exosomes containing MHC molecules. These exosomes can trigger adaptive immune responses by directly binding to T cell receptors and activating T cells [38]. Exosomes actively participate in antigen presentation by expressing MHC class I and class II molecules on their surface, thereby inducing diverse immune responses [39]. Notably, MSC-derived exosomes resemble their parent cells, which have limited MHC-I expression and no MHC-II, and therefore usually have low immunogenicity [40].
As various types of recipient cells possess the ability to uptake exosomes, exosomes play a crucial role in intercellular communication. Thus, exosomes enhance the efficacy and effectiveness of intercellular communication by facilitating the transfer of nucleic acids, proteins, and lipids between cells. Notably, exosomes can also encapsulate various drugs for intercellular transport to specific injury or disease sites by overcoming biological barriers. This establishes a foundation for the clinical utilization of exosomes in medical diagnostics and treatment.
Due to their unique characteristics, exosomes are considered promising candidates for drug delivery systems. Compared with synthetic nanovesicles such as liposomes, the advantages of exosomes as biologically derived sources of endogenous functional biomolecules include biocompatibility, low toxicity [41], enhanced stability and bioavailability [42], and an extended in vivo half-life [43]. Importantly, as endogenous delivery systems, they can cross the BBB autonomously [44] and infiltrate various tissues via specific surface ligands and receptors [45]. These properties give exosomes an advantage in clinical applications, offering more possibilities for targeted drug delivery systems. To deliver drugs, genes, and other macromolecules precisely to target cells or tissues in various therapeutic settings, further investigation of exosome-based delivery systems is necessary.
Exosomes can serve as potential diagnostic indicators for various illnesses and can be isolated from bodily fluids or cell cultures. However, extracting exosomes from bodily fluids is not only a practical challenge, but the process is also complicated by the heterogeneity resulting from the contributions of different cell types to the exosome population. This interferes with the analysis results [46]. Consequently, cell-derived exosomes are currently favoured for drug delivery.
Almost all types of cells secrete exosomes, although immune cells, cancer cells,
and MSCs are the primary sources of exosomes that carry drugs. Exosomes produced
from immune cells have shown considerable promise in immunomodulation and disease
treatment due to their ability to deliver antigens [47]. Exosomes derived from
immune cells possess the inherent potential to modulate various immunological
reactions. For example, treg cell-derived exosomes can effectively lower systemic
inflammation by delivering miRNAs that inhibit
interferon-
MSCs are pluripotent stem cells that have the capacity to differentiate into multiple cell types in vivo. They are present in several human tissues, including adipose tissue, amniotic fluid, dental tissue, the placenta, the umbilical cord, and bone marrow tissue [50, 51, 52]. The most commonly studied MSCs are bone mesenchymal stem cells(BMSCs), gingival MSCs, ADSCs, and umbilical cord-derived MSCs (UC-MSCs) [53]. MSCs have the capacity to home to tissue, differentiate into multiple lineages, and self-renew. They have demonstrated therapeutic potential by protecting against fibrosis, oxidative damage, and apoptosis, promoting angiogenesis and tissue repair, exhibiting anti-inflammatory properties, and playing an immunomodulatory and neuroprotective role [54]. MSCs also show promise in producing more exosomes. Furthermore, mesenchymal stem cell-derived extracellular vesicles (MSC-EVs) typically exhibit excellent immunogenicity and inherit several key characteristics of their parent cells. Therefore, MSCs are an ideal source for the clinical extraction of exosomes [55]. However, although MSCs can serve as drug carriers, issues such as embolism and infection may arise.
To realise the therapeutic potential of exosomes in practical applications, an efficient and reliable separation method is crucial. Several techniques currently exist for exosome extraction, including ultracentrifugation, precipitation, immunoaffinity, size-exclusion chromatography, ultrafiltration, differential supercentrifugation, density gradient centrifugation, tangential flow filtration, and affinity capture [56, 57]. Each method has its own advantages and disadvantages (Table 2, Ref. [58, 59, 60, 61, 62, 63, 64, 65, 66, 67, 68, 69]). The extraction technique chosen impacts the production and purity of MSC-EVs [50].
| Isolation method | Principle | Yield | Purity | Advantages | Disadvantages | Contaminants | Ref. |
| Ultracentrifugation | Differential sedimentation based on size and density | Low-Moderate | Low-Moderate | Low cost; Handled large quantities | Time-consuming; requires specialized equipment | Protein aggregates, apoptotic bodies | [58, 59] |
| Density gradient centrifugation | Buoyant density equilibration | Low | High | High purity separation from contaminants | Time-consuming with multiple steps; low yield | - | [60] |
| Membrane filtration | Size-based sieving through pores of defined molecular weight | High | Low | Easy-operated; Efficient; High yield | Low purity; Exosome deformation | Protein aggregates, other vesicles of similar size | [61, 62] |
| Precipitation | Polymer-based precipitation reduction of solubility | High | Low | Easy-operated; High yield; no special equipment | Co-precipitation of non-vesicular material | Polymers, proteins, nucleic acids | [63, 64] |
| Size-Exclusion chromatography | Size-based elution | High | High | Decreased aggregation of exosomes; Preserves vesicle integrity and function | Special equipment; Low specificity; Time-consuming | Co-eluting lipoproteins (e.g., very low density lipoprotein (VLDL)) | [65, 66, 67] |
| Immunoaffinity capture | Antibody-antigen binding to specific surface markers | Low | High | High specificity for exosome subpopulations | High cost; limited to exosomes with known markers | Non-specifically bound proteins | [68, 69] |
Ultracentrifugation is currently the most commonly used technique for isolating exosomes. This technique reduces costs and facilitates the processing of large volumes of samples through high-speed centrifugation [50]. Immunoaffinity uses immobilised antibodies to capture exosomes containing specific ligands. However, for samples with low exosome content, concentration is required before immunoaffinity, which makes it unsuitable for large sample volumes. Immunoaffinity is also commonly used as a purification step. The yield of differential centrifugation is relatively high [70], as it enables efficient separation. However, this method cannot distinguish between particles with overlapping size ranges, such as exosomes and microvesicles [46]. To overcome the issue of co-purification, a sucrose gradient can be used to achieve a higher purity by utilising different densities [71]. Additionally, the affinity capture technique can be used to differentiate various high-purity exosomes based on markers found on the surface of extracellular vesicles. This method enables molecules to adhere to multiple carriers independently of the size or density of the extracellular vesicles. However, this approach has the disadvantage of a low yield [72]. Although these extraction and purification techniques can improve exosome purity, achieving total isolation from other biological products and complete purification remains a challenge. A variety of approaches should be employed to achieve the best results, despite the absence of a universally accepted ‘gold standard’ method for isolating exosomes. This integrated approach can ensure reliable and repeatable findings, while also aiding in increasing separation efficiency and purity [41, 73]. Further research is needed to investigate the isolation and purification of exosomes.
Exosomes can achieve targeted therapeutic effects by delivering specific cargo to recipient cells. However, the limited secretion of exosomes by parent cells hinders the application of large-scale therapy. One way to overcome this challenge is to genetically manipulate cells and modify their environmental conditions to enhance exosome production while preserving their functionality [50, 74].
Studies have been conducted to enhance exosome production by genetically modifying cells. For example, researchers utilised the Myc gene to transfect MSCs, resulting in the generation of immortalised MSCs and subsequently increasing exosome production [75]. The results demonstrated that exosomes derived from immortalised MSCs exhibit a similar biological function and set of properties to those of non-transfected MSCs, thus establishing a viable approach to exosome generation.
In addition to the genetic manipulation of parent cells, optimising the external environmental conditions for cell culture is also a key strategy for increasing the yield of exosomes. Studies on exosome production emphasise the importance of the culture medium. Investigations on human and mouse neuroblastoma cell lines have demonstrated that the biophysical and size characteristics of exosomes remain unchanged when transitioning from a medium containing fetal bovine serum (FBS) to a serum-free medium [76]. However, exosome production increases substantially when a serum-free medium is used. Furthermore, the limited surface area of the culture vessel used for monolayer cells restricts exosome production. To overcome these limitations, hollow fibre bioreactors with semipermeable capillary fibres and cylinders were employed. Importantly, such closed-system bioreactors enhance yield and facilitate good manufacturing practice (GMP)-compliant manufacturing by ensuring sterility, reducing batch variability, and enabling the establishment of rigorous release criteria, including endotoxin testing [77]. In Watson et al.’s study [78], a hollow fibre bioreactor was used to prepare exosomes from human embryonic kidney cells (HEK293). The results showed that this method was about 10 times more efficient than conventional methods [78]. Large-scale production often employs stirred tank bioreactors, with the cell density achieved varying according to operating modes and culture types. Therefore, compared to traditional static culture systems (e.g., T-bottles), bioreactor-based culture systems can significantly improve exosome production and are essential for generating clinically relevant quantities of EVs under controlled conditions that meet regulatory requirements [79]. Therefore, the culture medium, production system, or production method used by each cell line should be optimised to enhance the efficiency of exosome production and enable large-scale, clinically translatable manufacturing.
In many applications, exosomes are utilized to transport and deliver various therapeutic agents, including proteins, lipids, miRNAs, and other substances [80, 81], thereby achieving the desired therapeutic effects [82]. Several general techniques for effectively loading specific cargoes (drugs) into exosomes and ensuring their stability have so far been discovered [83]. Drugs can be bound to exosomes or exosome mimics through two main approaches: endogenous and exogenous loading [84].
Before the implementation of isolation techniques, endogenous loading involved modifying parent cells and then introducing the cargo into the exosome. This was achieved by using gene editing techniques on the parent cells [85], such as overexpressing therapeutic agents via cell transfection. Another approach involves incubating the drug directly with the parent cells to generate drug-containing exosomes. Endogenous loading ensures the integrity of exosome membranes, offering a relatively direct and stable method for producing exosomes. However, it is time-consuming and inefficient, typically resulting in limited cargo capacity.
To overcome the limitations of endogenous loading, researchers have developed various exogenous loading strategies. Exogenous loading involves encapsulating drugs in exosomes using passive or active loading methods after the isolation of intact exosomes [86]. Passive loading techniques, such as exosome or parent cell incubation, are primarily suitable for hydrophobic drugs. Mixing exosomes with therapeutic agents enables the lipid bilayer of the vesicle membrane to engage with hydrophobic medications, allowing them to permeate the exosomes in response to the concentration gradient. The effectiveness of the encapsulation process is affected by the hydrophobic characteristics of the drug molecules. This relatively simple approach does not require the inclusion of any active components in the system [87, 88] and has been used to load miRNA, small hairpin RNA (shRNA), mRNA, and proteins into exosomes [89]. For instance, brain-derived neurotrophic factor (BDNF) can be passively loaded into macrophage-derived exosomes via co-incubation. When intravenously administered, these exosomes efficiently crossed the blood-brain barrier and delivered BDNF to the brain, with enhanced accumulation under inflammatory conditions, demonstrating the utility of this simple loading method for targeted protein delivery [90]. Passive loading relies on concentration gradients of molecules and the hydrophobicity of cargo, typically resulting in a lower drug loading capacity [7].
Several active loading techniques have been developed for hydrophilic medications to facilitate drug diffusion into exosomes by temporarily penetrating the hydrophobic lipid membrane through physical or chemical approaches. These active loading methods include electroporation, sonication, chemical processes, click chemistry, extrusion, freeze-thaw cycles, and surface engineering technologies (Table 3, Ref. [39, 57, 91, 92, 93, 94, 95, 96, 97, 98, 99, 100, 101, 102, 103, 104, 105, 106, 107]) [81, 108]. The method chosen depends on the type of drug: electroporation is commonly used for nucleic acids, with a typical loading efficiency of 10–25%. Sonication and extrusion are suitable for proteins and hydrophilic small molecules, with an efficiency of 15–30% typically achieved. Hydrophobic small molecules can be effectively loaded via incubation or freeze-thaw cycles. Electroporation is the most popular technique, particularly for RNA encapsulation. However, electroporation can cause RNA aggregation and exosome instability, which may reduce the loading capacity [109]. Another active loading technique is sonication. A study has shown that sonication dramatically reduces the microviscosity of exosome membranes. After an hour of incubation at 37 °C, the membrane integrity of the exosomes can be restored [87]. Chemical methods, such as the use of transfection reagents or osmolytes like saponins, facilitate the entry of cargo into exosomes without disrupting their lipid bilayer structure. This enables functional molecules to be loaded into the exosomal cavity or onto its surface for therapeutic applications [110]. Similarly, click chemistry can directly bind molecules to the exosome surface via covalent bonds. For example, the azide-Fluor 545 compound can be conjugated to cross-linked alkyne-modified exosomes via carbodiimide-mediated chemistry [111]. The efficiency and stability of drug loading into exosome vesicles vary between these methods, each of which has its limitations. Therefore, it is imperative to continuously explore advanced exosomal drug loading techniques to enhance efficiency and optimise drug stability.
| Engineering strategies | Mechanisms | Advantages | Disadvantages | Optimal drugs | Ref. |
| Drug loading | |||||
| Co-incubation | Passive diffusion of drugs into exosomes during incubation | Simple operation, no extra reagents required | Low loading efficiency, potential drug cytotoxicity | Hydrophobic small molecule drugs | [91, 92, 93] |
| Electroporation | Electrical pulses create reversible pores in the exosome membrane | High efficiency, low equipment requirements | May compromise exosome integrity; cargo aggregation | Nucleic acids, proteins, peptides, and nanomaterials | [57, 94] |
| Sonication | Physical disruption of the exosome membrane using sonication | High efficiency, capable of loading large molecules. | May alter exosome integrity and size | Hydrophilic drugs, proteins | [95, 96] |
| Transfection | Introduction of genetic material into exosomes via chemical/biological agents | Maintaining exosome integrity and function | Complex, uncertainty of effectiveness | Nucleic acids, proteins, peptides, hydrophobic molecules, and high-molecular-weight drugs | [39, 97] |
| Extrusion | Mechanical forcing of exosome-drug mixture through a nanofiltration membrane | High loading efficiency | Requires special equipment; low yield | Small hydrophobic drugs, nucleic acids | [98, 99] |
| Freeze–thaw cycles | Membrane permeabilization through repeated freezing and thawing | Simple operation | May cause exosome aggregation | Water-soluble drugs, low-molecular-weight compounds, proteins, peptides, nucleic acids | [100, 101] |
| Surfactant-mediated membrane penetration | Surfactants create pores in the exosome membrane | High efficiency, Simple operation | Potential toxicity; may disrupt exosome integrity | Hydrophobic drugs, hydrophilic biomacromolecules | [102] |
| Surface engineering technologies | |||||
| Genetic engineering (transfection) | Modification of parent cells to produce engineered exosomes | Maintaining exosome integrity and function | Complex, high cost, low yield | —— | [103, 104] |
| Chemical modification (click chemistry) | Covalent conjugation via specific chemical reactions (e.g., azide-alkyne) | Efficient and precise | May alter membrane proteins, time-consuming | —— | [105, 106] |
| Hybrid membrane engineering (membrane fusion) | Fusion of exosomes with synthetic liposomes or other plasma membranes | Optimizing characteristics of the exosome surface | May weaken exosome function | —— | [107] |
Exosomes are native vesicles with a wide range of potential applications; however, their therapeutic efficacy may be limited by the capacity and stability of the drugs they carry. Furthermore, the targeting efficacy of native exosomes is often insufficiently precise, posing significant challenges in ensuring the accurate delivery of therapeutic agents to the lesion site. Following intravenous administration, for example, MSC-EVs are sequestered in the spleen, liver, and lungs, resulting in a low concentration of EVs in the target tissue. Furthermore, unmodified exosomes are rapidly cleared by the mononuclear phagocyte system, resulting in short circulation half-lives and potential off-target accumulation in organs such as the liver and spleen. To overcome this issue, MSC-EVs must be modified to selectively target the target tissue [112, 113]. Recently, scientists have attempted to create exosomes that can deliver medications and beneficial chemicals to wounds, neurons, and the circulatory system to generate specific therapeutic effects [114].
Modifying the surface characteristics of exosomes, particularly their protein composition, can significantly alter their biodistribution and targeting. To enhance local exosome concentration in the target region while minimising systemic toxicity, surface engineering technologies can be employed. Currently, mainstream surface engineering technologies can be classified into three main categories: genetic engineering, chemical modification, and hybrid membrane engineering (Table 3). Genetic engineering is an effective method for transfecting parental cells with targeted ligand fusion protein and selected exosome transmembrane protein plasmids, thereby displaying targeted ligands on the exosome membrane surface. Lysosome-associated membrane protein 2 (Lamp2b), for instance, is one of the most commonly used scaffolds in this approach. Genetically fusing a variety of targeting ligands to Lamp2b greatly increases the selectivity of exosomes for recipient cells [115]. Chemical modification techniques enable molecules to bind to the exosome membrane via covalent and non-covalent interactions without compromising the membrane’s structural integrity [116]. Chemical modification of exosomes encompasses two main approaches: exogenous and endogenous modifications. In the exogenous approach, exosomes are isolated and modified in vitro before clinical application. In the endogenous method, parent cells are genetically modified to produce engineered exosomes reliably. A common exogenous strategy is polyethylene glycol conjugation (PEGylation) of the exosome surface, which reduces immunogenicity and macrophage-mediated clearance [117]. Both of these methods are widely utilised [6]. Hybrid membrane engineering involves combining exosomes with liposomes or other plasma membranes to deliver drugs. This can include solid lipid nanoparticles (SLNs), lipid-polymer nanoparticles, or lipid dendrimer nanocarriers. For example, direct membrane fusion between exosomes and liposomes is achieved using a hybrid system known as EXOPLEXs (an exosome-liposome fusion complex), which efficiently delivers macromolecules while preserving the integrity of the exosome membrane structure [118]. Many studies have confirmed that these surface modification technologies can significantly enhance the targeting ability and therapeutic efficacy of exosomes.
One of the most effective ways to enhance the ability of exosomes to target the
brain following intravenous administration is through surface modification.
Exosomes with specific surface modifications have been shown to dramatically
increase miRNA loading efficiency, raising the prospect of using them to treat
neurodegenerative diseases [119]. For instance, MSC-derived exosomes (MSC-Exos)
conjugated with the rabies viral glycoprotein (RVG) peptide, which is specific to
the central nervous system (CNS), can significantly enhance their brain-targeting
capabilities. Exosomes treated with RVG exhibit increased accumulation in the
hippocampus and cerebral cortex following intravenous administration.
RVG-modified exosomes considerably decreased A
However, there is also evidence suggesting that, although surface modification of MSC-EVs could enhance their efficacy in targeted therapy [75], these methods of modification, especially genetic engineering techniques for the EV membranes and contents, could alter the morphology and function of EVs, as well as have detrimental and unnecessary influences on recipient cells/tissues. To guarantee the safety, specificity, and effectiveness of modification techniques, it is crucial to carefully evaluate any possible adverse effects when investigating exosome surface alterations.
Building upon engineering strategies that enhance the targeting specificity of exosomes, selecting an appropriate administration route is the next critical step to ensure these precisely engineered vesicles reach target tissue efficiently in vivo. Exosome-based medications can be administered intravenously, subcutaneously, intranasally, intraperitoneally, or orally, demonstrating great adaptability and compatibility [39]. MSC-EVs injected intravenously have been shown to alleviate spinal cord injury and promote functional recovery in rat models [121]. This involves the release of nutrients or miRNAs via exosomes by MSCs in the injured area. Additionally, the intraperitoneal administration of exosomes has been shown to significantly increase the expression of tyrosine hydroxylase-positive cells in the substantia nigra of animals used to model Parkinson’s disease (PD), thereby promoting recovery [122]. These findings support the ability of intravenous and intraperitoneal exosome injections to deliver therapeutic agents across the BBB. However, the systemic administration of drugs via the intraperitoneal or intravenous routes still limits the amount that enters the brain to some extent [123, 124]. To address this, intranasal administration (IN) offers a more direct route for treating central nervous system diseases as a non-invasive alternative. Compared with intravenous administration, intranasally administered exosomes can traverse the BBB, exhibit enhanced accumulation in the brain, and have been found in multiple brain regions [125]. Importantly, exosomes may also actively migrate to lesion sites [126, 127].
In addition to delivering to the central nervous system, exosomes have also demonstrated significant advantages in treating localised diseases. For instance, subcutaneous injection of exosomes is an effective method of targeting skin-related diseases, particularly skin malignancies. The transformation of M2 (anti-inflammatory) macrophage polarisation via exosomes was successfully promoted by injecting M2 macrophage-derived exosomes subcutaneously at the edge of the wound. This significantly increased collagen deposition, re-epithelialization, and angiogenesis, thereby accelerating the wound healing process [128].
Furthermore, for chronic diseases requiring long-term or non-invasive drug administration, the oral route offers the advantages of convenience and high patient compliance. Therefore, oral therapy represents a simple and non-invasive method for exosome delivery. This approach has been used to administer exosomes carrying chemotherapeutic agents, such as curcumin, achieving curcumin levels in multiple organs that are three to five times higher than with free compound delivery [129]. Furthermore, Arntz et al. [130] were the first to reveal that, in a mouse arthritis model, the onset of rheumatoid arthritis (RA) was considerably delayed when milk exosomes were taken orally.
Therefore, multiple routes are available for exosome administration, and selecting an appropriate one can effectively enhance therapeutic efficacy.
Exosomes possess attributes that make them a versatile foundation for various biomedicine applications, including tissue engineering, genetic therapy, targeted drug delivery, oncological diagnostics and treatment, and vaccine development [114, 131]. They have been shown to interact with a wide range of recipient cells, thereby influencing various biological processes [132]. Through intervention and modification, they can be engineered for three main applications: vaccines, disease biomarkers for diagnosis, and delivery systems for treatment. Exosomes have been widely utilised in clinical research worldwide to identify or treat various diseases, including cardiovascular diseases, cancer, neurodegenerative diseases (such as PD and Alzheimer’s disease (AD)), autoimmune syndromes, infectious diseases [133], bone and cartilage lesions [134], and for drug and vaccine delivery [6]. The subsequent sections outline the use of exosomes in the treatment of cardiovascular and cerebrovascular diseases, neurological disorders, cancer, diabetes mellitus, immunomodulation, and regenerative medicine (Fig. 3, Table 4, Ref. [135, 136, 137, 138, 139, 140, 141, 142, 143, 144, 145, 146, 147, 148, 149, 150, 151, 152, 153, 154, 155, 156]). It is important to note that the term ‘exosomes’ is used only when the original studies provided evidence for specific isolation and characterisation. However, many therapeutic studies utilise heterogeneous preparations obtained by methods such as ultracentrifugation. In cases where the particular subtype of vesicle was not rigorously defined, the more general term ‘EVs’ is used, or the terminology reported by the original literature (e.g., MSC-EVs) is used to reflect the nature of the biological material used accurately.
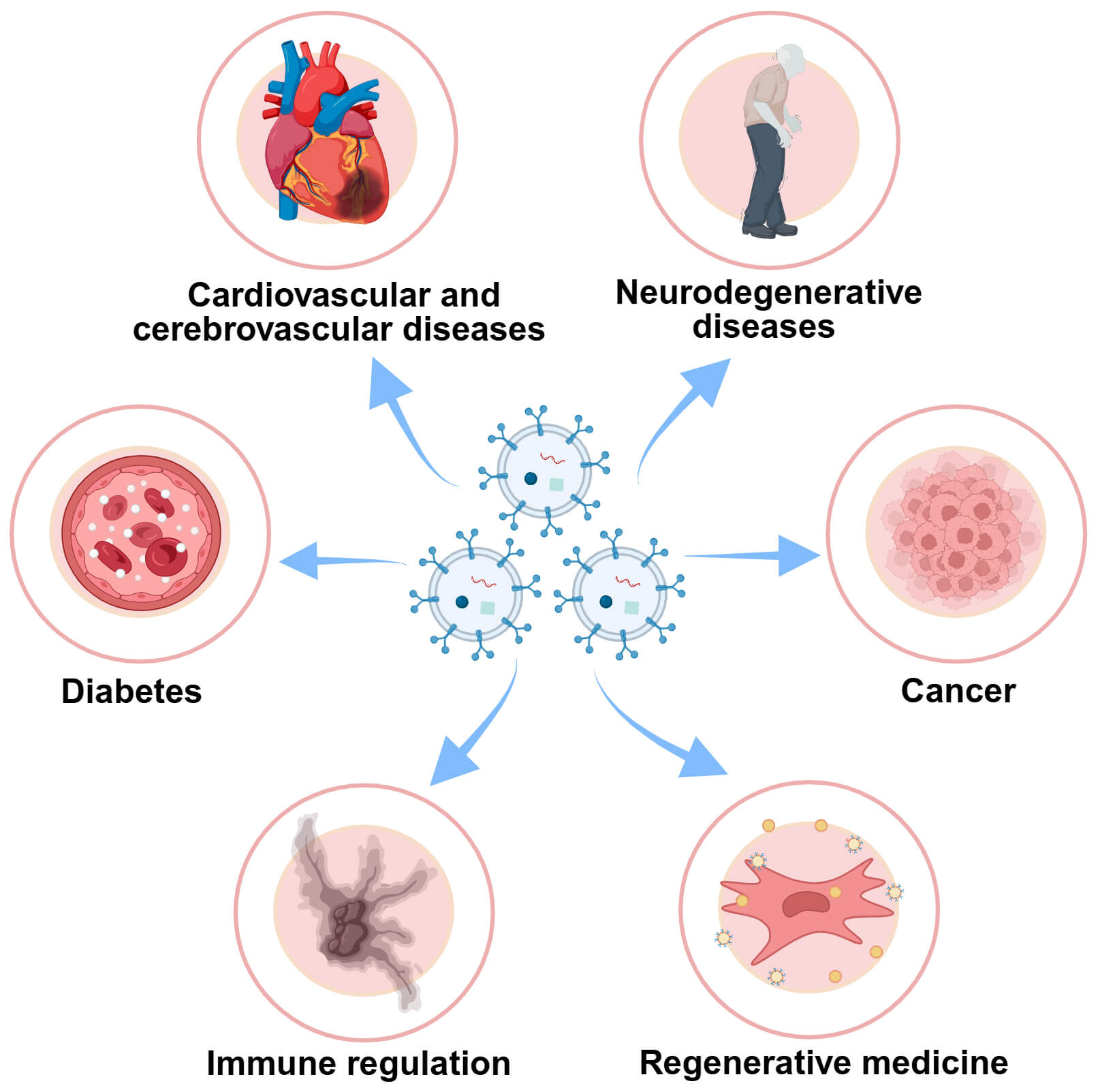 Fig. 3.
Fig. 3.
Clinical application of exosomes in multiple diseases. As efficient drug carriers, proteins, lipids, and miRNA can be delivered and transferred more easily via exosomes for therapeutic uses. These applications encompass an extensive number of clinical conditions, including cardiovascular and cerebrovascular diseases (CVD) like myocardial infarction (MI) and cerebral ischemia-reperfusion (CI/R) injury, neurodegenerative diseases including Alzheimer’s disease (AD), Huntington’s disease (HD), amyotrophic lateral sclerosis (ALS) and Parkinson’s disease (PD), cancer, diabetes mellitus (DM), and immune regulation. Created in BioRender (https://BioRender.com/).
| Disease category | Specific condition | Exosome type | Model system | Administration route | Key therapeutic effects | Ref. |
| Cerebrovascular disease | Ischemic Stroke | MSC-Exos (BMSC-Exos) | Rat MCAO model | Intravenous | Reduced infarct volume, promoted neurogenesis/angiogenesis, improved functional recovery | [135, 136] |
| Ischemic Stroke | Engineered plasma exosomes (Edaravone-loaded) | Rat MCAO model | Intravenous | Enhanced brain targeting, stronger neuroprotection, reduced acute injury | [137] | |
| Cardiovascular disease | Myocardial Infarction | MSC-Exos (miR-25-3p enriched) | Mouse I/R model | Intravenous | Promoted M2 macrophage polarization, mitigated inflammation | [138, 139] |
| Myocardial Infarction | MSC-Exos (GATA-4 overexpressing) | Rat MI model | Intramyocardial | Delivered anti-apoptotic miRNAs, robust cardioprotection | [140] | |
| Neurodegenerative diseases | Alzheimer’s Disease | BMSC-Exos (miR-29b overexpressing) | Rat AD model | Intracerebroventricular | Inhibited A |
[141] |
| Alzheimer’s Disease | Engineered exosomes (CoQ10-loaded) | Rat AD model | Intracerebroventricular | Improved spatial memory, increased neuronal density, and neurogenesis | [142] | |
| Parkinson’s Disease | hUCMSC-Exos | Rodent PD model | Intravenous | Crossed the BBB, ameliorated behavioral deficits, and provided neuroprotection | [143] | |
| Parkinson’s Disease | Engineered blood exosomes (Dopamine-loaded) | Mouse PD model | Intravenous | 15-fold higher brain dopamine delivery, reduced systemic toxicity | [144] | |
| Amyotrophic Lateral Sclerosis | AT-MSC-Exos | SOD1G93A ALS mouse model | Intravenous Intranasal | Suppressed glial activation, mitigated neuroinflammation, improved motor function | [145, 146] | |
| Huntington’s Disease | Astrocyte-derived exosomes | HD 140Q KI mouse model | Intrastriatal | Reduced mHTT aggregate load | [147] | |
| Cancer | Hepatocellular Carcinoma | ADMSC-Exos (miR-122 loaded) | Mouse HCC model | Intratumoral | Enhanced sorafenib effect, suppressed tumor growth | [148] |
| Breast Cancer | hBMSC-EVs (miR-379 loaded) | Preclinical BC model | Intravenous | Inhibited tumor progression via the COX-2 pathway | [149] | |
| Various Cancers | Native exosomes (Paclitaxel-loaded) | Murine models (Lung, Breast, Ovarian) | In vitro culture | Suppressed tumor growth and metastasis | [150] | |
| Diabetes | Type 1 Diabetes | human UCB-MSC-Exos | Mouse T1DM model | Intravenous | Promoted |
[151] |
| Type 2 Diabetes | hBMSC-Exos | Rat HFD/STZ model | Intravenous | Alleviated hyperglycemia, enhanced insulin sensitivity, protected |
[152] | |
| Diabetic Neuropathy | Schwann cell-derived exosomes | Mouse T2DM model | Sciatic nerve administration | Alleviated neuropathy symptoms, promoted nerve regeneration | [153] | |
| Immune regulation | Asthma | BMSC-EVs | Human BMSCs in vitro | In vitro culture | Enhanced Treg function, upregulated TGF- |
[154] |
| Regenerative medicine | Sciatic Nerve Injury | GMSC-derived EVs | Rat nerve defect model | Local administration | Accelerated axonal regeneration, functional recovery | [155, 156] |
MACO, middle cerebral artery occlusion; BBB, blood-brain barrier; HFD/STZ, high-fat diet and streptozotocin; GATA-4, GATA-binding protein 4; HCC, hepatocellular carcinoma; mHTT, mutant huntingtin protein; MSC-Exos, mesenchymal stem cell-derived exosomes; BMSC-Exos, bone marrow-derived mesenchymal stem cell exosomes; hUCMSC-Exos, human umbilical cord mesenchymal stem cell-derived exosomes; AT-MSC-Exos, adipose tissue-derived mesenchymal stem cells -derived exosomes; ADMSC-Exos, adipose-derived mesenchymal stem cells-derived exosomes; hBMSC-Exos, human bone marrow-derived mesenchymal stem cell-derived exosomes; GMSC, gingival mesenchymal stem cell; EVs,extracellular vesicles; MI, myocardial
infarction; AD, Alzheimer’s disease; PD, Parkinson’s disease; SOD1G93A, mutant superoxide dismutase 1 (G93A); ALS, amyotrophic lateral sclerosis; KI, knock-in; BC, breast cancer; T1DM, Type 1 diabetes mellitus; TGF-
The brain is susceptible to hypoxia and ischemia. Ischemic cerebrovascular disease is the second leading cause of global mortality [157], accounting for 11.6% of all deaths and 62.4% of stroke cases worldwide in 2019 [158]. Current standard therapies, including mechanical thrombectomy and the intravenous administration of fibrinolytic agents, aim to rapidly restore cerebral perfusion and minimise neurological damage following ischaemic events. Nevertheless, secondary brain injury after reperfusion remains a significant clinical challenge, driven by complex pathophysiological mechanisms. Exosomes have been linked to the recovery of cerebral function following a stroke, as well as to the regulation of peripheral immunological responses. Both of these have been linked to long-term neuroprotective effects [159]. In adult rat models of middle cerebral artery occlusion (MCAO), mesenchymal stem cell-derived exosomes (MSC-Exos) have been shown to significantly enhance cell proliferation in the subventricular zone, as well as promote angiogenesis and neurogenesis in the ischemic penumbra. Furthermore, these exosomes facilitate functional recovery and neurite remodelling after stroke [135]. In rat stroke models, bone marrow-derived mesenchymal stem cell exosomes (BMSC-Exos) administered within 24 hours after ischemia reduced cerebral infarct volume and brain water content in a dose-dependent manner. BMSC-Exos also promoted beneficial microglial polarization and suppressed NOD-, LRR- and pyrin domain-containing protein 3 (NLRP3) inflammasome-mediated inflammation and pyroptosis. Notably, sustained improvements in neurological function were observed over five weeks following BMSC-Exos administration, suggesting both acute and prolonged neuroprotective effects in the MCAO model [136].
In addition to modulating inflammation and neuronal function, exosomes have the potential to facilitate structural repair of the nervous system. Compared with the PBS control treatment, the intravenous administration of MSC-Exos significantly increased the proportion of BrdU-positive/Doublecortin-positive cells in the ischaemic penumbra of MCAO rats, indicating enhanced neurogenesis after stroke [160]. Another research report suggests that the intravenous injection of MSC-EVs can promote white matter repair in a rat model of ischaemic stroke [161]. To further improve therapeutic outcomes, ongoing research focuses on engineering exosomes as targeted drug delivery vehicles to achieve greater specificity and reduced systemic exposure. For example, in a rat model of MCAO, edaravone (EDV) was loaded onto exosomes via co-incubation. The results showed that EDV-loaded rat plasma exosomes were more effective at alleviating acute ischaemic brain injury than free EDV, suggesting they have stronger brain-targeting ability, higher bioavailability, and are safer. This targeted effect was mediated by the transferrin receptor, with a significant reduction in infarct volume and a marked improvement in neurological function observed within seven days of cerebral ischemia [137]. In the MCAO model, quercetin (Que) was loaded onto surface-engineered rat plasma exosomes via co-incubation. These exosomes were then conjugated with monoclonal antibodies against growth-associated protein 43 (GAP43), enabling targeted delivery to damaged neurons and enhancing their survival rate. This dual-functional system increased quercetin accumulation at the injury site and effectively reduced reactive oxygen species (ROS) generation by modulating the nuclear factor erythroid 2 -related factor 2 (Nrf2) /heme oxygenase-1 (HO-1) signalling pathway [162].
Collectively, owing to their intrinsic biological properties and high degree of engineering feasibility, exosomes represent a novel and versatile approach for treating cerebral ischemia-reperfusion injury. MSC-EVs, in particular, hold promise as a therapeutic candidate.
Myocardial infarction is characterised pathologically by the death of cardiomyocytes and inflammatory activation [163]. There is extensive preclinical evidence demonstrating that MSC transplantation can lead to moderate yet consistent improvements in post-infarct left ventricular remodelling and heart failure [164]. Notably, MSC-EVs confer cardioprotective effects comparable to those of whole-cell transplantation in the treatment of myocardial infarction, while offering distinct advantages, including excellent immunocompatibility and minimal tumourigenic potential.
MSC-EVs are critical mediators of MSC-mediated cardioprotection, transporting diverse bioactive molecules and demonstrating regenerative potential for post-infarction cardiac repair in both in vitro and in vivosettings [165, 166]. For instance, studies using murine models of myocardial ischaemia-reperfusion and in vitro systems have shown that MSC-Exos exert cardioprotective effects by delivering miR-25-3p. This miRNA promotes the polarization of macrophages from the pro-inflammatory M1 phenotype to the anti-inflammatory M2 phenotype. This mitigates inflammation and protects the myocardium against reperfusion injury [138, 139]. Furthermore, MSC-Exos contain multiple miRNAs, including miR-210, miR-199a-3p, miR-21, and miR-22, which collectively form a synergistic regulatory network. These molecules help reduce cardiomyocyte apoptosis, attenuate fibrosis, and improve cardiac function in mouse models of myocardial infarction [167].
In addition to miRNA-mediated mechanisms, the transcription factor GATA-binding protein 4 (GATA-4) plays a pivotal role in modulating the cardioprotective properties of exosomes. As a member of the GATA family of transcription factors, GATA-4 binds specifically to GATA motifs in the promoters of target genes and is constitutively expressed in cardiomyocytes, where it regulates cardiac development and homeostasis from embryogenesis through to adulthood. MSC-Exos engineered to overexpress GATA-4 are enriched with anti-apoptotic miRNAs, including miR-22, miR-451, miR-221, and miR-19a. When injected intramyocardially into a rat model of myocardial infarction, these exosomes exhibit significant cardioprotective effects [140]. Long non-coding RNAs (lncRNAs) also contribute to exosome-mediated cardiac protection. MSC-EVs pretreated with macrophage migration inhibitory factor (MIF) exert protective effects via long non-coding RNA nuclear-enriched abundant transcript 1 (lncRNA NEAT1), a transcript located on chromosome 11 that has been shown to protect cardiomyocytes and can be effectively delivered via MSC-EVs [53]. Building upon these promising preclinical findings, exosome-based therapies are progressing towards clinical translation. Recent efforts have focused on establishing GMP-compliant protocols for the large-scale production of clinical-grade exosomes derived from cardiac progenitor cells [168]. Several biotechnology start-ups are now leveraging similar exosome platforms as cell-free therapeutics for the treatment of acute coronary syndrome and myocardial infarction.
AD is a prevalent neurodegenerative disorder characterised by the pathological
hallmarks of amyloid-
Beyond their diagnostic potential, exosomes possess unique therapeutic
capabilities. Accumulating evidence indicates that they can effectively cross the
BBB via mechanisms such as transport through the choroid plexus to reach the
brain parenchyma and deliver their cargo. This intrinsic ability, combined with
their capacity to encapsulate bioactive molecules, target injured regions, and
modulate neurodegenerative processes, establishes them as a promising delivery
platform offering advantages over synthetic carriers in terms of biocompatibility
and CNS penetration [171, 172]. In murine models of AD, BMSC-Exos have been shown
to upregulate the expression of miR-146a in the hippocampus, thereby attenuating
astrocyte-mediated neuroinflammation and promoting synaptic regeneration and
cognitive recovery [178]. Another study reported that, in a rat model of AD,
MSC-Exos that overexpress miR-29b and are stereotactically injected into the cornu
ammonis 1 (CA1) region of the dorsal hippocampus effectively inhibit
A
Furthermore, exosomes can be engineered to carry additional neuroprotective agents, thereby enhancing their therapeutic efficacy. For example, in rat models of AD, exosomes loaded with coenzyme Q10 have been shown to improve spatial memory more effectively when administered via intracerebroventricular injection. They have also been found to increase hippocampal neuronal density and upregulate the expression of BDNF and SRY-related high-mobility group box 2 (SOX2), which highlights their roles in neuroprotection and neurogenesis [142]. Additionally, exosomes loaded with quercetin (Exos-Que) enhanced the neuroprotective effect of Que by increasing its bioavailability and achieving brain targeting. As an effective tau protein aggregation inhibitor, Exos-Que is more effective than Que alone at improving cognitive function in AD mice, and is regarded as a potential treatment strategy for AD [179].
Despite offering a novel approach to treating neurodegenerative diseases [180], research into exosomes and their translation into clinical practice is still in its early stages. To ensure safety, reliability, and clinical benefit, large-scale, multicentre, randomised controlled trials are essential in order to rigorously evaluate the efficacy and tolerability of using exosomes to carry miRNAs or drugs before human application. Notably, some studies have indicated that certain exosomal miRNAs may exert cytotoxic effects under specific conditions [17]. Furthermore, the majority of preclinical evidence supporting exosome-based therapies originates from studies using young animal models (typically under one year old), which do not fully replicate the physiological environment of aged patients with AD. Therefore, further investigation is required to assess the therapeutic outcomes of exosomal miRNAs or drugs in aged animal models. Future research should focus on systematically validating the functional efficacy and translational potential of exosomes as dual-purpose carriers for therapeutics and diagnostic biomarkers in AD.
PD is the second most common neurodegenerative disorder worldwide. Its incidence and prevalence increase with age, and it affects men more than women, with a male-to-female ratio of approximately 2:1. Reported incidence rates range from 47 to 77 and from 108 to 212 per 100,000 people among individuals aged 45 and over and 65 and over, respectively [181]. PD pathogenesis is characterised by depletion of dopaminergic neurotransmitters, accumulation of Lewy bodies in specific brain regions, and progressive loss of neurons in the substantia nigra pars compacta [182]. Clinically, PD is characterised by the progressive degeneration of dopaminergic neurons and the subsequent development of severe motor impairments. Current standard therapies for providing symptomatic relief primarily rely on dopamine precursor agents, such as levodopa [183]. While these treatments can improve patients’ quality of life to some extent, they do not halt disease progression or restore dopaminergic neuronal function. Furthermore, many therapeutic agents have a limited capacity to cross the BBB, which poses a significant challenge to the effective delivery of drugs to the central nervous system.
Notably, when administered via intravenous injection, human umbilical cord mesenchymal stem cell-derived exosomes (hucMSC-Exos) have been shown to efficiently traverse the BBB and target the substantia nigra (SN), leading to the amelioration of behavioural deficits in rodent models of PD [143]. This capability underscores their potential as a novel therapeutic platform for PD [184]. Cell-based therapies utilising MSCs and MSC-EVs have demonstrated the ability to enhance dopaminergic neuron activity, promote angiogenesis, stimulate neurogenesis, mitigate neuroinflammation, and support functional recovery. This positions them as a promising intervention for PD treatment. Beyond their therapeutic applications, disease-specific alterations in exosomal miRNA expression profiles also provide valuable biomarkers for early diagnosis and monitoring. For example, Bhattacharyya et al. [185] found that the level of miRNA-128 was significantly lower in plasma-derived exosomes from patients with PD than in healthy controls.
Furthermore, exosomes derived from mouse blood have demonstrated potential as
targeted drug delivery vehicles for PD. In a murine PD model, dopamine was loaded
onto these exosomes via co-incubation. The results showed that, when the
dopamine-loaded exosomes were injected intravenously into mice, the distribution
of dopamine in the brain was almost 15 times higher than with free dopamine
administration, and systemic toxicity was significantly reduced. This delivery
strategy substantially improves upon conventional systemic levodopa therapy,
which is typically limited by poor BBB penetration [144]. Exosomes from diverse
cellular origins, loaded with different bioactive molecules, demonstrate distinct
therapeutic advantages. In PD mouse models, for example, exosomes derived from
mesenchymal stem cells and enriched with miR-100a-5p have been shown to protect dopaminergic
neurons and ameliorate motor deficits by reducing oxidative stress via the Nox4/ROS/Nrf2 signaling pathway [186]. Additionally,
exosomes have the potential to target important pathogenic proteins. For example,
by loading an antisense oligonucleotide sequence targeting human
Despite these advances, the clinical application of MSC-EVs for PD remains in its early stages. Most existing clinical trials have small sample sizes, which limits the generalisability of the findings to broader patient populations. Furthermore, the higher cost and technical complexity of isolating and expanding MSCs impedes the scalable production and widespread clinical adoption of MSC-EVs [183]. In summary, although exosomes show promise as a tool for achieving sustained, minimally invasive drug delivery in PD, their true clinical value must be substantiated through more extensive clinical research.
Amyotrophic lateral sclerosis (ALS) is a progressive neurodegenerative disorder involving the degeneration of motor neurons in the brainstem and spinal cord. This leads to muscular denervation and the progressive loss of motor function. According to a meta-analysis, the global standardised incidence rate of ALS is 1.68 per 100,000 people per year, with significant variations between regions [188]. Key pathological hallmarks include the mislocalisation and aggregation of cytoplasmic superoxide dismutase 1 (SOD1) and TAR DNA-binding protein 43 (TDP-43) [189, 190]. Hypoxic and other cellular stress conditions have been shown to modulate the expression of various miRNAs, thereby influencing key proteins involved in ALS mechanisms. In patients with sporadic ALS (sALS), dysregulation of circulating miRNAs has been closely associated with disease progression. This includes elevated serum levels of miR-1825 and miR-1234-3p, as well as altered plasma concentrations of miR-151b, miR-221-3p, and miR-130a-3p [191]. Given the critical regulatory roles of miRNAs in the central nervous system, profiling the content of exosomal miRNAs in biofluids such as blood and urine offers a promising, non-invasive strategy for elucidating the molecular basis of ALS onset and progression.
Furthermore, exosomes secreted by adipose tissue-derived mesenchymal stem cells (AT-MSCs) have demonstrated neuroprotective properties in in vitro ALS models [192]. In the SOD1G93A transgenic mouse model of ALS, repeated intravenous or intranasal administration of AT-MSC-derived exosomes (AT-MSC-Exos) has been shown to suppress glial activation, mitigate neuroinflammation, and improve motor function, highlighting their potential as an intervention therapy for ALS [145, 146].
Huntington’s disease (HD) is an autosomal dominant neurodegenerative disorder caused by a CAG trinucleotide repeat expansion in the huntingtin (HTT) gene. This results in the mutant protein (mHTT) gaining a toxic gain-of-function. HD is clinically characterised by progressive motor dysfunction and cognitive decline, and currently lacks disease-modifying therapies [193]. The global prevalence of HD is 2.7 cases per 100,000 people [194]. Studies indicate that soluble, misfolded forms of the mutant protein appear in the striatum during the early stages of the disease, before overt neurodegeneration. In 2019, Giampà et al. [195] reported that conditioned medium from amniotic MSCs exerted neuroprotective effects in vitro and improved motor performance in the R6/2 HD mouse model. Similarly, exosomes derived from astrocytes and ADSCs exhibit robust neuroprotective capabilities. Notably, the intrastriatal delivery of astrocyte-derived exosomes in Huntington’s disease 140Q knock-in (KI) mice significantly reduced the load of mHTT aggregates [147]. Importantly, exosomes isolated from the primary astrocytes of 140Q KI mice were found not to contain mHTT, which suggests that they are safe and suitable as a potential therapeutic vehicle for HD.
Several studies have reported significant downregulation of miRNA-124—a CNS-abundant miRNA critical for neurogenesis—in the R6/2 mouse model, STHdhQ111/HdhQ111 cells, and postmortem brains of patients with HD [196]. As miR-124 exists in both free and exosomal forms in body fluids, its role in HD pathogenesis warrants further investigation. Lee et al. [197] demonstrated that exosomes engineered to carry miR-124 effectively reduced the expression of REST (repressor element 1-silencing transcription factor), a key mediator of HD-related transcriptional dysregulation. While a single administration did not result in significant functional improvement, these findings imply that optimising dosing regimens or combining exosomal miR-124 with other neuroprotective miRNAs, such as miR-9 and miR-125b, could enhance therapeutic efficacy [198].
Cancer remains a leading cause of death worldwide. It is projected that the United States will see around 2,041,910 new cases of invasive cancer in 2025, equating to an average of 5600 new diagnoses each day [199]. While multiple therapeutic modalities, including immunotherapy, targeted therapy, radiotherapy, chemotherapy, and surgical intervention, are currently available, the development of drug resistance significantly limits their clinical efficacy [200, 201]. Consequently, there is an urgent need to develop more effective cancer management strategies. In recent years, exosomes have emerged as promising biomarkers with diagnostic potential across various malignancies. Their utility has been demonstrated in prostate cancer and glioblastoma [202], as well as in breast, liver, lung, and bladder cancers [203]. To evaluate the diagnostic value of exosomes in non-small cell lung cancer (NSCLC), serum exosomes were isolated from healthy donors (n = 46) and NSCLC patients (n = 125) by ultracentrifugation [204]. Proteomic analysis (via mass spectrometry) revealed multiple differentially expressed proteins between the groups. Specifically, the exosomal expression levels of alpha-2-HS-glycoprotein (AHSG) and extracellular matrix protein 1 (ECM1) were significantly increased in NSCLC patients. This study provides evidence that serum exosomes contain specific proteins with potential diagnostic and prognostic value for NSCLC, thus supporting their potential application as novel non-invasive tools. Further investigation is required to validate these exosomal protein biomarkers and explore their clinical utility.
In cancer therapy, engineered MSC-EVs, characterised by favourable tumour tropism, nanoscale dimensions, high biocompatibility, and low immunogenicity, are regarded as candidates for targeted drug delivery [12]. For instance, in a hepatocellular carcinoma model, the intratumoral administration of ADMSC-derived exosomes (ADMSC-Exos) loaded with miR-122 significantly enhanced the anti-tumour effects of sorafenib and suppressed tumour growth [148]. Similarly, both in vitro and in vivo studies have demonstrated that ADMSC-EVs overexpressing miR-199 can sensitise tumour cells to doxorubicin (DOX) by inhibiting the mTOR signalling pathway [205]. miR-379 exerts potent tumour-suppressive effects in breast cancer, and a preclinical study has shown that intravenously injected human bone marrow-derived mesenchymal stem cell (hBMSC)-derived extracellular vesicles (hBMSC-EVs) can inhibit breast cancer progression by targeting the COX-2 signalling pathway with miR-379 [149]. Notably, exosomes carrying conventional chemotherapeutic agents can serve as therapeutic vehicles. Melzer et al. [150] successfully isolated paclitaxel-encapsulating exosomes, which efficiently delivered the drug to tumour cells in murine models, thereby suppressing tumour growth and metastasis in lung, breast, and ovarian cancers. Various types of drug-loaded exosomes exhibit distinct therapeutic advantages in multiple tumour models. For example, BMSC-Exos carrying DOX exhibited enhanced cytotoxicity against osteosarcoma cells (with an IC50 of 6.48 µg/mL for Exo-DOX versus 10.13 µg/mL for free DOX). Interestingly, the IC50 value of Exo-DOX in myocardial cells (29 µg/mL) was approximately four times higher than that of free DOX (7.312 µg/mL), confirming superior safety characteristics for non-target cells [206]. In a murine osteosarcoma model, MSC-derived exosomes loaded with DOX functioned as efficient targeted delivery vehicles, significantly enhancing anti-tumor efficacy while reducing cardiotoxicity compared to free drug [207].
Nevertheless, the role of exosomes in cancer is a double-edged sword. While they hold promise for diagnosis and therapy, there is growing evidence that they may also contribute to tumour progression and metastasis. As key components of the tumour microenvironment, MSCs can influence oncogenic processes via exosome-mediated intercellular communication [208]. For example, in xenograft models of colon and gastric cancers, MSC-EVs have been shown to promote tumour growth and progression. One study reported that co-implantation with MSC-Exos resulted in substantially larger tumours, reaching over 150 mm3 by day 20, compared to less than 100 mm3 in the control group. Even by day 40, there was an approximate 1.5-fold increase in tumour volume [209]. While such pro-tumourigenic effects are less evident under in vitro conditions, these findings emphasise the importance of carefully evaluating exosome-based therapies. In summary, the clinical transformation of exosomes as therapeutic carriers requires a thorough evaluation of efficacy and safety. In particular, BMSC-EVs exhibit functional heterogeneity in modulating chemotherapy resistance, tumourigenesis, and disease progression. Therefore, future research must focus on elucidating their long-term biosafety profile and defining their appropriate therapeutic applications.
Diabetes mellitus (DM) is a chronic metabolic disorder characterised by an
absolute or relative deficiency in insulin secretion, resulting in persistent
hyperglycaemia. Type 1 diabetes mellitus (T1DM) results from the
autoimmune-mediated selective destruction of pancreatic
In recent years, therapeutic approaches utilising MSC-EVs have emerged as a promising strategy for diabetes management. In type 1
diabetes, MSC-EVs exert immunomodulatory effects that contribute to repairing
autoimmune damage to pancreatic islet tissue. Evidence indicates that, following
intravenous administration, MSC-Exos can attenuate diabetes,
facilitate insulin production, and promote
MSCs can secrete exosomes that exhibit immunomodulatory properties and retain
the key biological characteristics of their parent cells, such as
anti-inflammatory and anti-apoptotic effects [217]. Furthermore, MSC-EVs act as
endogenous carriers of bioactive molecules, facilitating the transfer of
proteins, miRNAs, and genetic material to recipient cells. This process
recapitulates critical MSC functions such as immunomodulation and pro-angiogenic
activity. Proteomic analysis has revealed that the secretome of MSCs derived from
adipose tissue, bone marrow, and fetal tissues is enriched with growth factors,
immunomodulatory mediators, and antioxidant components. These collectively
constitute the molecular foundation for the cells’ immunoregulatory capabilities
[218]. There is accumulating evidence that MSC-EVs have significant
immunoregulatory effects in various autoimmune disorders, including multiple
sclerosis (MS), rheumatoid arthritis, uveitis, and type 1 diabetes mellitus [51].
For instance, BMSC-EVs have been shown to enhance the proliferation and
immunosuppressive function of regulatory T cells (Tregs) in asthma patients,
thereby upregulating the expression of inhibitory cytokines such as transforming
growth factor-
In addition to their therapeutic potential, exosomes also have diagnostic value in the context of immune-related diseases. Urine is a valuable, non-invasive source of disease-relevant biomolecules, particularly in the context of renal pathologies. Urinary exosomes, for example, contain a large number of distinct proteins, many of which are associated with specific disease states, making them a promising source of novel biomarkers. For example, hemopexin (HPX) and tetraspanin 1 (TSPAN1), which have been identified in urinary exosomes, are potential early biomarkers for T-cell-mediated rejection in kidney transplant recipients [221].
However, the immunological functions of exosomes are not always beneficial and can have dual roles depending on the pathological context. Emerging evidence suggests that exosomes can contribute to immune evasion mechanisms, potentially promoting tumour progression or allograft acceptance by suppressing immune surveillance in an undesirable way. This paradoxical behaviour warrants further investigation to fully understand the contextual determinants of exosomal activity and ensure their safe and effective clinical application.
MSCs possess multilineage differentiation potential, meaning they can differentiate into various cell lineages, including chondrocytes, adipocytes, and osteoblasts. Furthermore, they exert critical paracrine effects by secreting growth factors, cytokines, chemokines, and extracellular matrix-degrading proteases, thereby fully harnessing their regenerative potential in tissue repair and remodelling [74]. Compared to conventional cell-based therapies, MSC-EVs offer several advantages, including greater compositional stability, reduced risk of tumour formation or immunogenic reactions, and ease of storage and handling. Due to these qualities, they are valuable candidates for cell-free regenerative therapeutics [222]. Consequently, there has been increasing interest in the field of regenerative medicine in exosomes derived from MSCs, which have been extensively investigated for their ability to promote tissue regeneration and accelerate wound healing [223, 224]. Among these, bone marrow-derived MSCs produce bioactive components with reparative properties, demonstrating promise in tissue engineering and regenerative applications [225, 226]. Preclinical evidence suggests that MSC-EVs exhibit many of the biological functions of their parent cells and have therapeutic potential in various disease models, including inflammatory disorders, cutaneous injuries, autoimmune-associated neural regeneration, and oral and maxillofacial pathologies [222], as well as diabetic skin ulcers [227], palatal wound healing [228], periodontitis [229], and subclavian artery defects. For instance, in a rat model of a severe tongue defect, the functionalisation of small intestinal submucosa extracellular matrix (SIS-ECM) with gingival mesenchymal stem cell (GMSC)-derived exosomes was found to significantly enhance the regeneration of myo-mucosal tissue and taste buds [230]. Similarly, in murine models of sciatic nerve crush injury [155] and rat models of sciatic nerve segmental defects [156], local administration of GMSC exosomes markedly accelerated axonal regeneration and functional neurological recovery.
Taken together, MSC-EVs show promise as a potential cellular therapeutic agent for promoting tissue regeneration and functional restoration. However, their clinical translation requires further validation through well-designed, high-quality clinical trials.
Exosomes have demonstrated potential applications in various fields, including regenerative medicine, drug delivery systems, and disease diagnosis and treatment. As key mediators of intercellular communication, they play a crucial role in regulating both physiological and pathological processes, and possess several unique advantages. Compared with artificial carriers such as liposomes, they have excellent biocompatibility and low immunogenicity, as well as a superior ability to penetrate biological barriers. This makes them efficient drug carriers for the precise delivery of therapeutic substances to specific tissues or organs. Furthermore, engineering modifications enable the use of exosomes for targeted therapy, significantly enhancing treatment efficacy in areas such as cancer, cardiovascular diseases, and neurodegenerative disorders. The specific biomarkers they carry are crucial not only for early disease diagnosis but also for applications in tissue regeneration, gene therapy, and vaccine development.
Despite their promising potential, translating exosomes from basic research to clinical application still faces multiple technical challenges. Firstly, the exact roles of exosomes in maintaining homeostasis or regulating abnormal conditions in vivo are unclear. This necessitates the development of more comprehensive models and technical systems to study their biogenesis, transport mechanisms, endocytosis processes, and functional signalling pathways systematically. Secondly, the clinical application of exosomes is hindered by low extraction yields, insufficient purity, limited drug loading efficiency, and high costs. Current separation techniques struggle to meet the simultaneous requirements of high purity, high yield, scalability, and economic feasibility. Standard drug loading methods, such as electroporation, sonication, and incubation, still present challenges, including low encapsulation efficiency, damage to the membrane structure, and drug aggregation. In terms of targeting, although engineering can enhance the targeting ability of native exosomes, their actual targeting accuracy and off-target risks in vivo still need to be systematically evaluated through in-depth studies. Additionally, the optimal administration route, dosage regimen, and treatment cycle remain to be determined. Furthermore, advancing exosomes towards clinical application requires addressing critical regulatory and manufacturing challenges, particularly ensuring consistency between batches, achieving long-term storage stability, and enabling cost-effective large-scale production. Therefore, these challenges must be addressed, with future research focusing on developing standardised, scalable, and clinically viable platforms for exosome production, engineering, and quality control. In this endeavour, international standardisation initiatives such as the Minimal Information for Studies of Extracellular Vesicles (MISEV 2023) guidelines provide an essential framework [231]. Adherence to guidelines on harmonised practices in nomenclature, separation, characterisation, and functional analysis will be pivotal in translating exosome research into reliable clinical applications.
Compared with traditional treatment methods, the development and production costs of exosome therapy are significantly higher. Therefore, it is crucial to outline its key benefits over current therapies to demonstrate its usefulness in real-world applications. Pharmacokinetic studies reveal limited data on the metabolism and tissue accumulation of exosomes in vivo, highlighting the need for more comprehensive preclinical and clinical evaluation. These gaps directly impact critical safety considerations, including potential immunogenicity, tumourigenicity, and dose optimisation. These issues must be systematically addressed in conjunction with efforts to improve production and targeting. Further basic experiments should focus on clarifying the pharmacokinetic and pharmacodynamic characteristics of exosomes as drug carriers, to enhance therapeutic efficacy while minimising adverse reactions. The introduction of additional animal models and experimental conditions that more closely mimic human physiological and pathological states is necessary to facilitate their clinical translation.
In conclusion, while preclinical studies highlight the potential of exosomes as an emerging drug delivery platform, their clinical translation remains in its infancy. Significant scientific and technical challenges must be overcome before broader therapeutic application becomes feasible, including the standardisation of production, targeted delivery, comprehensive safety evaluation, and demonstrating efficacy in multicentre, controlled, large-scale comparative trials. Therefore, pending successful validation in clinical settings, exosomes hold the prospect of becoming a valuable tool in future medicine.
XQL: Writing—original draft & editing, Conceptualization, RS: Writing—review & editing, Visualization, Supervision, Funding acquisition, Conceptualization. Both authors read and approved the final manuscript. Both authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This work was supported by grants from the National Natural Science Foundation of China (No. 82473917, 82173811), Jiangsu Key Laboratory of Neuropsychiatric Diseases (BM2013003) and the Priority Academic Program Development of the Jiangsu Higher Education Institutes (PAPD) and Suzhou International Joint Laboratory for Diagnosis and Treatment of Brain Diseases.
The authors declare no conflict of interest. Rui Sheng is serving as one of the Editorial Board members of this journal. We declare that Rui Sheng had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Bettina Platt.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



