1 Henan International Joint Laboratory of Immunology and Model Animals, Henan Collaborative Innovation Center of Molecular Diagnosis and Laboratory Medicine, School of Medical Technology, Xinxiang Medical University, 453003 Xinxiang, Henan, China
2 Collage of Pharmacy, Xinxiang Medical University, 453002 Xinxiang, Henan, China
†These authors contributed equally.
Abstract
Quercetin is a naturally occurring flavonoid widely distributed in plants that exhibits various biological activities, including anti-inflammatory, antioxidant, and neuroprotective effects. It exhibits a potential role in sleep regulation and homeostasis; however, its specific effects on sleep-wake cycles and underlying mechanisms remain unelucidated.
To systematically investigate the regulatory role of quercetin in sleep architecture and homeostatic recovery, polysomnography (PSG) was used to monitor sleep parameters in mice under normal circadian rhythms and acute sleep deprivation (ASD). Immunofluorescence staining was performed to assess the expression of cellular proto-oncogene protein Fos (c-Fos) and microglial activation in sleep-related brain regions, including the medial prefrontal cortex (mPFC), nucleus accumbens (NAc), bed nucleus of the stria terminalis (BNST), paraventricular thalamic nucleus (PVT), hippocampal dentate gyrus (DG), basolateral amygdala (BLA), and periaqueductal gray (PAG).
Under normal circadian conditions, high-dose quercetin promoted non-rapid eye movement (NREM) sleep in mice. In ASD models, quercetin enhanced NREM sleep rebound during the early recovery phase. It sustained higher levels of wakefulness during the subsequent light phase, exhibiting its dual role in accelerating homeostatic recovery while balancing circadian arousal. Immunofluorescence analyses showed that quercetin markedly suppressed c-Fos expression in the mPFC, BLA, and PVT under sleep-deprived conditions. Additionally, it inhibited microglial activation in the mPFC and NAc.
These results mechanistically associate the sleep-regulatory effects of quercetin with its dual inhibition of neuronal hyperactivity in sleep-associated brain regions and neuroinflammatory responses. Altogether, this study identifies quercetin as a novel natural modulator of sleep homeostasis, underscoring its therapeutic potential for sleep disorders via anti-excitatory and anti-inflammatory mechanisms.
Keywords
- quercetin
- sleep homeostasis
- polysomnography
- sleep deprivation
- c-Fos
- microglia
Sleep is a fundamental physiological process essential for maintaining nervous system function and immune homeostasis, with sleep homeostasis regulation involving a bidirectional interplay with the neuroimmune network [1, 2]. Clinical and animal studies suggest that disrupting sleep homeostasis can activate the immune system, exacerbate the increase in peripheral proinflammatory cytokines, and stimulate microglial activation within the central nervous system (CNS), thereby increasing the risk of infectious, autoimmune, and neurodegenerative diseases [3, 4]. Microglia, the primary immune cells in the brain, regulate wake-sleep cycles by modulating neuronal activity in specific brain regions [5, 6]. For instance, the activation of inhibitory G-protein signaling within microglia has been shown to promote sleep. Conversely, sleep states can regulate the morphology of microglia. Notably, animal studies have associated enhanced neuronal activity during wakefulness with elevated microglial branch extension and dynamic synaptic interactions, whereas microglia exhibit a more quiescent morphology during sleep. These findings closely associate microglial morphology with synaptic plasticity during the sleep-wake cycle [7, 8, 9]. Microglia undergo a morphological shift toward a proinflammatory phenotype, characterized by reduced branching and increased phagocytic activity, during sleep deprivation (SD). This shift, along with inflammatory infiltration in the meninges and brain parenchyma, reveals that sleep states influence immune responses by regulating microglial morphology [10]. Therefore, exploring approaches to maintain sleep homeostasis through immune system modulation provides a theoretical foundation for developing “anti-inflammatory–sleep-promoting” intervention strategies.
Quercetin, a natural flavonoid abundantly present in Chinese herbal medicines,
is well known as its anti-inflammatory, antioxidant, and neuroprotective
properties [11, 12]. Recent study has shown that quercetin nanoparticles can
modulate the gut-brain axis, attenuate the translocation of proinflammatory
cytokines into the brain, and improve sleep quality [13]. Additionally, quercetin
has been reported to alleviate SD-induced behavioral alterations by inhibiting
the inflammatory pathway in the hippocampus and restoring brain-derived
neurotrophic factor-mediated neuroplasticity in SD models [14]. The
anti-neuroinflammatory effects of quercetin mainly include suppression of the
nuclear factor-
This study aimed to employ polysomnography and SD paradigms to systematically investigate quercetin-mediated reshaping of sleep architecture in mice under both basal and homeostatic imbalance conditions. Additionally, the effects of quercetin on brain region-specific neuronal activity and microglial dynamics were investigated to elucidate the “natural compound–neuroimmune–sleep homeostasis” regulatory axis and provide mechanistic insights for interventions in sleep disorders.
Adult male C57BL/6J mice (age: 8–10 weeks) were obtained from Beijing Vital River Laboratory Animal Technology Co., Ltd. and housed in a specific pathogen-free (SPF)-grade animal facility under a 12-h light-dark cycle (light on: 7:00–19:00) and controlled room temperature (24–26 °C), with ad libitum access to food and water. Mice were acclimated to the housing environment for 1 week before experimental manipulation to minimize stress. During acclimation and experimental periods, animals were monitored daily for general health status in accordance with the Guidelines for the Care and Use of Laboratory Animals (8th edition, National Institutes of Health, Bethesda, Maryland, USA). Animals were euthanized via intraperitoneal injection of an overdose of sodium pentobarbital (100 mg/kg body weight, Sigma-Aldrich, St. Louis, MO, USA; Cat. No.: P3761). All procedures involving animals were approved by the Henan Provincial Animal Care and Use Committee and conducted in accordance with the experimental guidelines of the Animal Experimentation Ethics Committee of Xinxiang Medical University (No. XYLL-20240390), China.
The experimental mice were anesthetized via intraperitoneal injection (i.p.) of ketamine (100 mg/kg; Cat. No. K-002; Supelco, Merck KGaA, St. Louis, MO, USA) and diazepam (5 mg/kg; Cat. No. D-907; Supelco, Merck KGaA), which was dissolved in 0.9% saline. The injection was administered at a volume of 0.1 mL/10 g of body weight. Subsequently, the implantation of electroencephalogram (EEG) and electromyography (EMG) electrodes was performed with a stereotaxic apparatus (Model No. 68803; RWD Life Science Co., Ltd., Shenzhen, China). The EEG recording electrode was placed in the medial prefrontal cortex (mPFC) at the coordinates: Bregma anteroposterior (AP) + 1.75 mm, mediolateral (ML) + 0.45 mm, with the EEG reference electrode implanted on the surface of the cerebellar skull. Two EMG electrodes were inserted into the bilateral neck muscles. The electrodes were secured with dental cement (Super-Bond C&B, Cat No.: FG2971, N Corporation, Moriyama, Shiga Prefecture, Japan). After surgery, the mice were returned to their home cages for recovery.
After EEG-EMG electrode implantation, the mice were allowed to recover for 1 week and then placed in a sound-attenuated chamber to adapt the resistance of the electrical commutator for 3 days. The polysomnographic recorder (Model No. Neurokey, Nanjing Greathink Medical Technology Co., Ltd., Nanjing, China) synchronously recorded EEG and EMG signals at a sampling frequency of 1000 Hz. The manual scoring of sleep states was performed following downsampling of the signals to 250 Hz. The sleep-wake states were classified into wakefulness, non-rapid eye movement (NREM) sleep, and rapid eye movement (REM) sleep based on the EEG and EMG signals. Wakefulness was recognized by high-frequency, low-amplitude EEG activity and high-amplitude EMG activity. NREM sleep was recognized by low-frequency (0.5–4 Hz), high-amplitude EEG dominated by delta waves and low-level EMG activity. REM sleep was recognized as low-amplitude EEG with theta wave dominance (4–9 Hz) and near-absent EMG activity.
Following manual scoring to identify NREM sleep states, EEG signals were subjected to fast Fourier transform (FFT) with a time window (4-second window length) to calculate the power spectral density (PSD, unit: µV2/Hz). The delta wave band was defined as 0.5–4 Hz, and the gamma wave band as 30–80 Hz.
Sleep latency was defined as the duration from the initiation of EEG-EMG signal
acquisition to the mouse’s first transition into NREM sleep. If NREM sleep was
not detected in the first hour, the latency was recorded as 3600 s. To minimize
artifact interference, the first episode of NREM sleep was only included in the
analysis if it was sustained continuously for
The mice were subjected to acute sleep deprivation (ASD) or chronic sleep deprivation (CSD) using the Rodent Sleep Deprivation System (Model XR-XS108; Shanghai XINRUAN Information Technology Co., Ltd., Shanghai, China). The mice were allowed to move freely with ad libitum access to food and water. Sleep deprivation was initiated at 7:00 during the light phase. The sleep deprivation rods rotated unidirectionally at 10 revolutions per minute (rpm). A 5-min interval was maintained between each full rotation of the rods. Each sleep deprivation period lasted 6 h.
Quercetin (Cat. No. S2391; Selleckchem, Houston, TX, USA) was dissolved in freshly opened dimethyl sulfoxide (DMSO; Cat. No. D8371; Solarbio Science & Technology Co., Ltd., Beijing, China). Quercetin working solutions were then formulated at concentrations of 20 mg/kg and 200 mg/kg. The diluent for working solutions was prepared by sequentially mixing 5% DMSO, 40% polyethylene glycol 300 (PEG300; Solarbio Science & Technology Co., Ltd., Beijing, China; Cat. No.: IP9020), 5% Tween 80 (Solarbio Science & Technology Co., Ltd.; Cat. No.: IT9000), and 50% ddH2O (DNase/RNase-Free Water; Solarbio Science & Technology Co., Ltd.; Cat. No.: R1600).
For ASD, quercetin (20 mg/kg or 200 mg/kg body weight) was administered via i.p. 30 min before the end of the deprivation period. For CSD, quercetin was administered intraperitoneally 30 min before the end of the SD on day 7. Immediately after the SD, a 24-h sleep recording was conducted.
At 30 min before the start of the night phase (active period), the experimental mice were injected via i.p. with quercetin. The control mice were administered an i.p. injection of the vehicle (the diluent for working solutions). The EEG-EMG signal was recorded continuously for 24 h, starting 30 min after quercetin injection.
To observe the effects of quercetin on the cellular proto-oncogene protein Fos (c-Fos) expression and microglial activation in the brain, quercetin was administered 30 min before the end of SD. After 60 min of quercetin administration, the mice were subjected to transcardiac perfusion. Subsequently, the brain tissues were harvested and subjected to post-fixation with 4% paraformaldehyde (PFA) for 8–12 h. Then, 40-µm-thick coronal brain sections were prepared by using an automated vibratome (Model: VT1200S; Leica Biosystems, Nussloch, Baden-Württemberg, Germany). The brain sections were collected into 24-well plates, and 4–6 slices containing the target brain regions were selected for immunofluorescence staining.
The brain sections were washed three times with 0.01 M phosphate-buffered saline (PBS), with each wash lasting 10 min. Then, the sections were blocked with 0.01 M PBS containing 0.3% Triton X-100 and 10% goat serum for 90 min at room temperature (RT). Following the removal of the blocking solution, the sections were incubated overnight at 4 °C with primary antibodies. The primary antibodies included: ionized calcium-binding adapter molecule 1 (Iba1) (Wako Pure Chemical Industries, Ltd., Osaka, Japan; Catalog No.: 019-19741; Dilution: 1:1500) and c-Fos antibody (Synaptic Systems GmbH, Göttingen, Germany; Catalog No.: 226008; Dilution: 1:5000).
The brain sections were subsequently rinsed thrice with 0.01 M PBS, with each
rinse lasting 10 min. Fluorescent secondary antibodies (Goat anti-rabbit Alexa
Fluor 594; catalog number: A-11012, Thermo Fisher Scientific, Waltham, MA, USA; Dilution:
1:800)
diluted in 0.01 M PBS were added, and the brain sections were incubated for 2 h
at RT. After three additional PBS washes (10 min each time), the brain sections
were incubated with 4′,6-diamidino-2-phenylindole (DAPI; Roche, Basel,
Switzerland; Cat. No.: 10236276001, 5 µg/mL) for 15 min at RT. Then,
the stained sections were mounted in 50% glycerol. The immunofluorescence images
were acquired using NIS-Elements Advanced Research software (NIS-Elements AR 5.42.06; Nikon Instruments Inc., Tokyo, Japan), a dedicated software for the Nikon Ti2-E
confocal microscope (Model: Ti2-E; Nikon Instruments Inc., Tokyo, Japan) with
10
All statistical analyses were performed using GraphPad Prism 8.0 software
(GraphPad Software, San Diego, CA, USA). The experimental data were presented as
the mean
To investigate the regulatory effects of quercetin on sleep under physiological conditions, mice received a single i.p. injection of either low-dose (20 mg/kg) or high-dose (200 mg/kg) quercetin, and the control mice were administered a vehicle diluent. Following treatment, continuous 24-h EEG-EMG sleep signals were recorded (Fig. 1A). Notably, high-dose quercetin significantly increased the percentage of NREM sleep and decreased that of wakefulness, with no significant differences observed in REM sleep (Fig. 1B,C). These effects were consistent across both active and inactive phases (Fig. 1D,E).
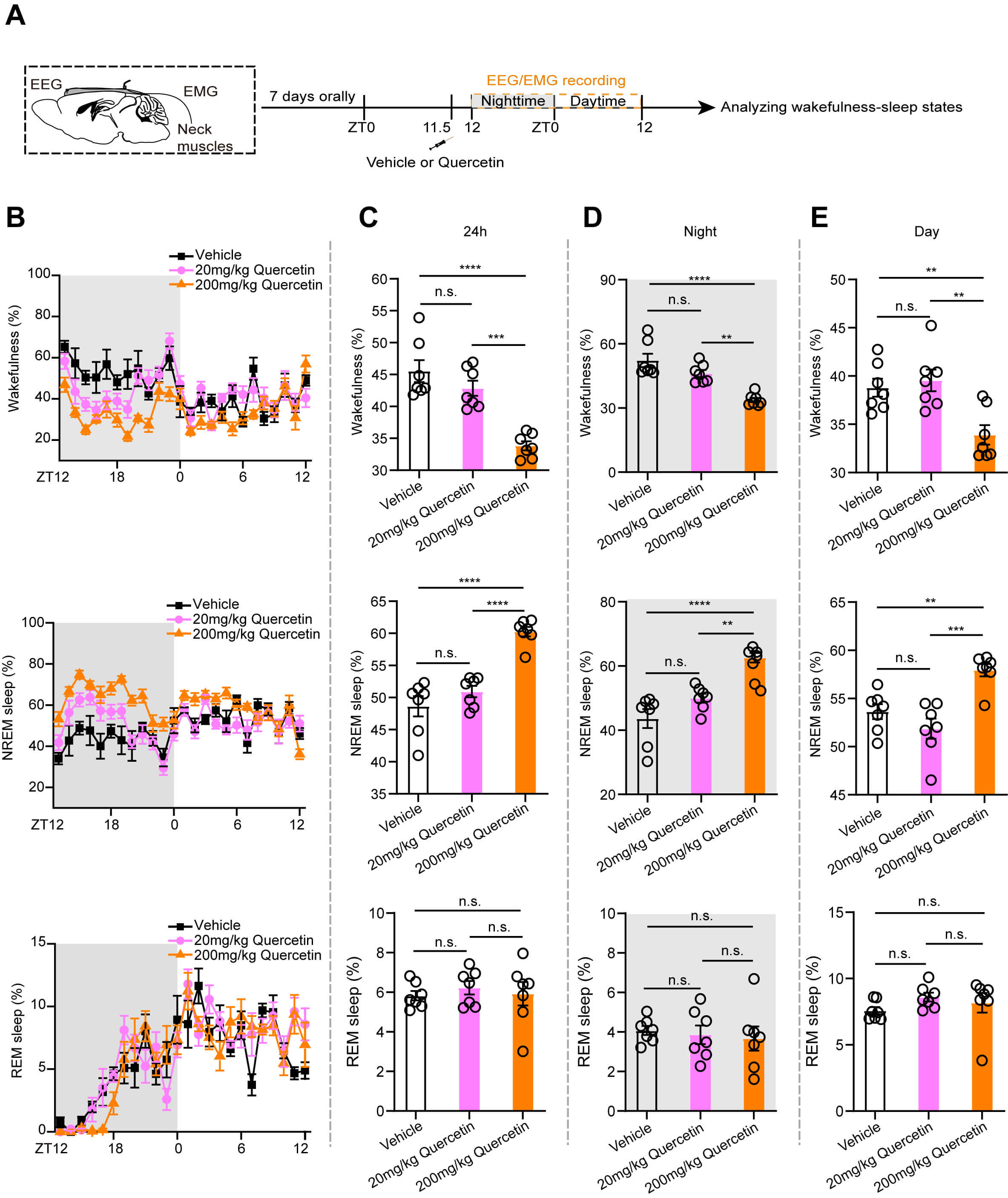 Fig. 1.
Fig. 1.
Quercetin significantly increases the NREM sleep under basal
conditions in mice. (A) Flow chart of electroencephalogram-electromyogram
(EEG-EMG) recording after quercetin injection. (B) The line chart shows the
percentages of wakefulness (upper), NREM sleep (middle), and REM sleep (lower) in
mice after quercetin administration. (C–E) The statistical charts show the
percentage of wakefulness (upper), NREM sleep (middle), and REM sleep (lower)
over three time periods: 24 h (C), nighttime active phase (D), and daytime
inactive phase (E). n = 7 per group. Each circle represents one mouse. The data
are expressed as mean
To determine if quercetin facilitates the wakefulness-to-sleep transition in mice, post-treatment sleep latency during the 1st hour was analyzed (Fig. 2A–C). Sleep latency decreased dose-dependently, with high-dose quercetin significantly reducing sleep latency compared with the control group (Fig. 2D).
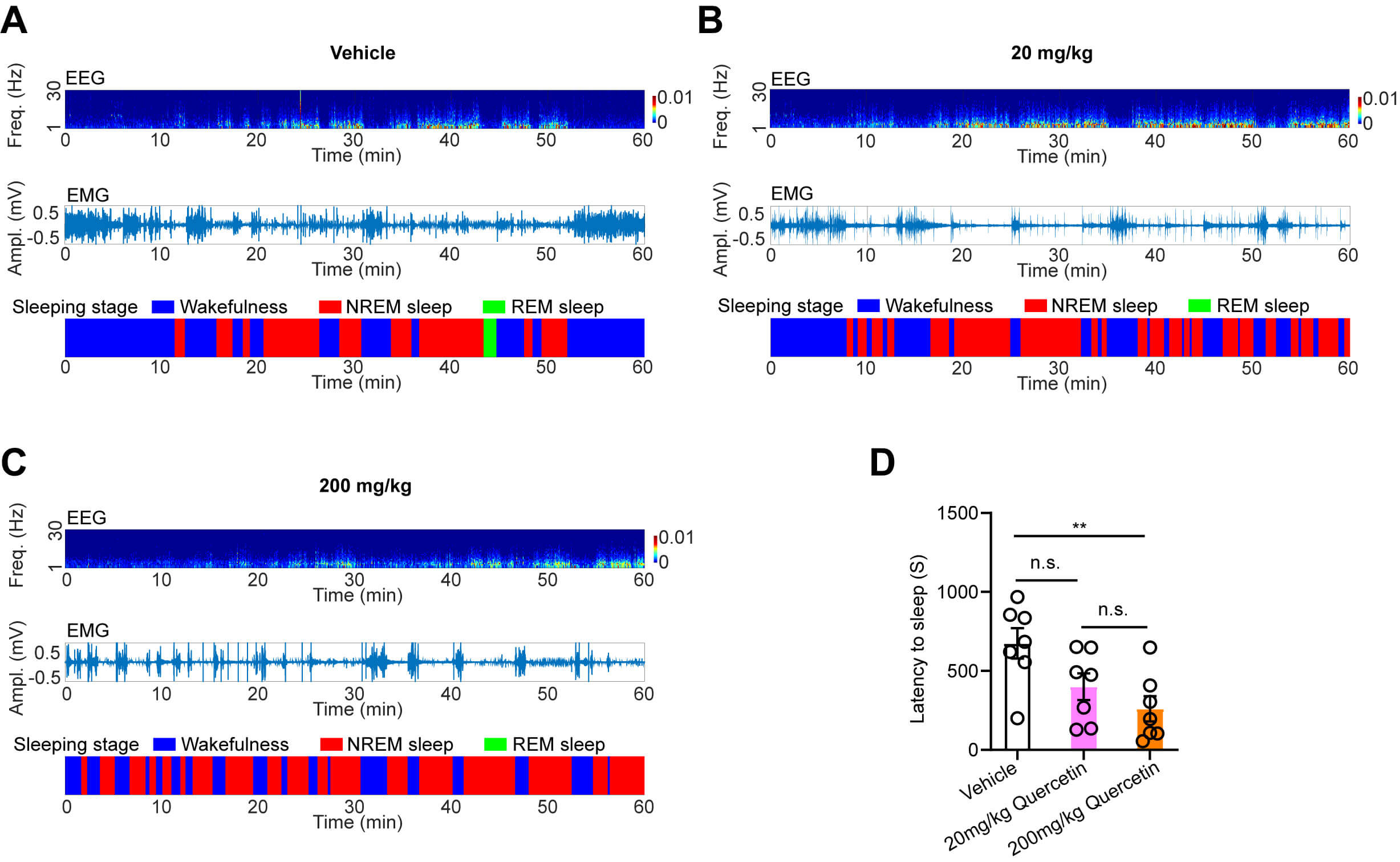 Fig. 2.
Fig. 2.
Quercetin effectively reduces the sleep latency in mice. (A–C)
Representative traces of EEG, EMG wave, and the sleeping stages in the vehicle
control group (A), 20 mg/kg quercetin-treated group (B), and 200 mg/kg
quercetin-treated group (C) during the first hour post-administration. (D) The
statistical chart illustrates the sleep latency in vehicle-treated mice, 20 mg/kg
quercetin-treated mice, and 200 mg/kg quercetin-treated mice. n = 7 per group.
Each circle represents one mouse. The data are expressed as mean
To further assess quercetin-mediated regulation of sleep pressure, EEG power spectra were evaluated during both day and night. Notably, the EEG power of delta (0.5–4 Hz) and gamma (30–80 Hz) waves did not differ significantly between quercetin-treated and control mice during both periods (Fig. 3A–F). Overall, these findings suggest that quercetin exerts an NREM sleep-promoting effect without elevating sleep pressure.
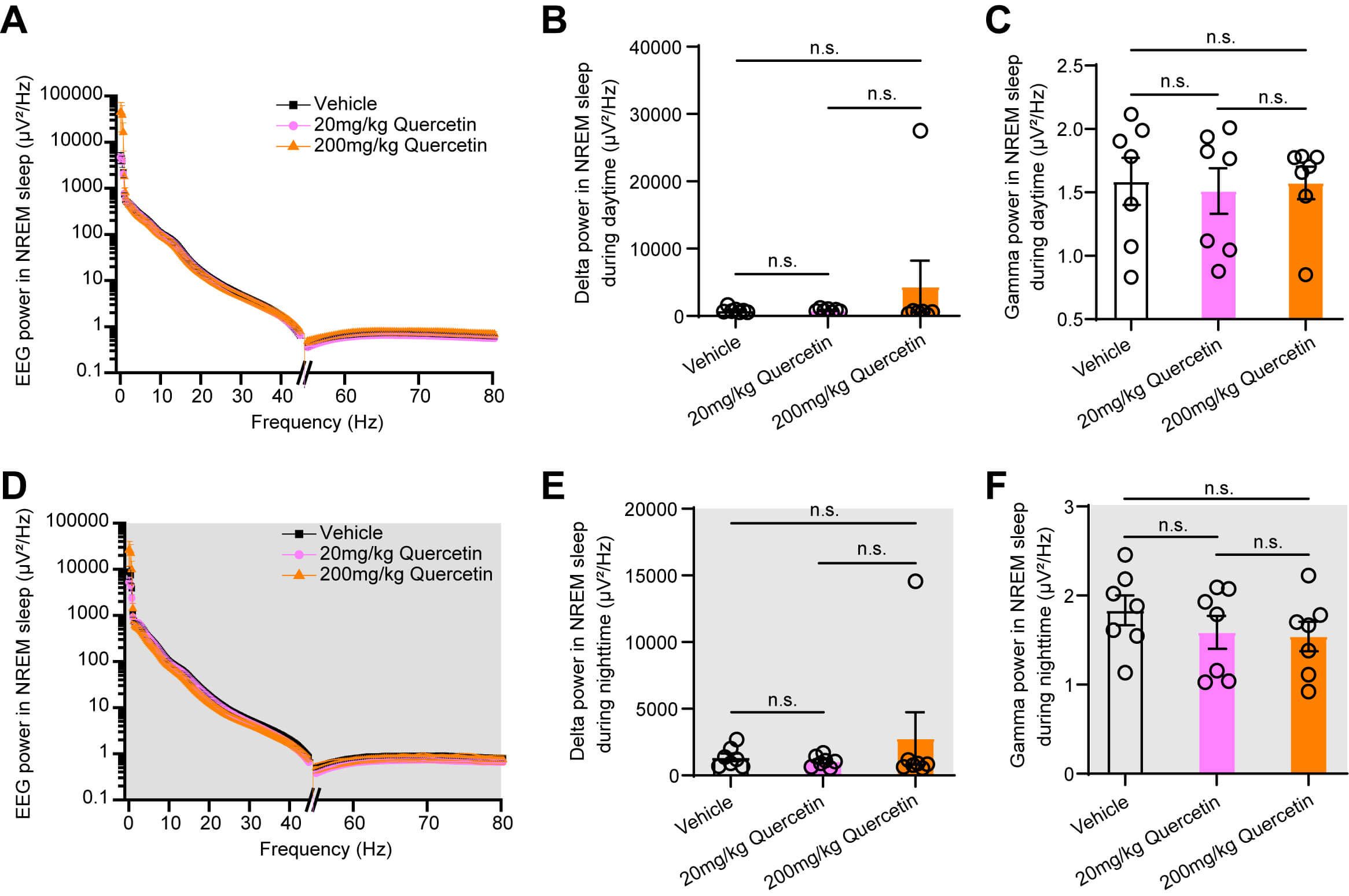 Fig. 3.
Fig. 3.
Quercetin has no effect on the EEG power spectrum in mice under
the basal state. (A,D) Statistical graph showing EEG power analysis of NREM
sleep after quercetin injection during daytime (A) and nighttime (D). (B,C)
Statistical charts show that quercetin had no significant effect on delta power
(B) and gamma power (C) during daytime. (E,F) Statistical charts show that
quercetin did not affect delta power (E) and gamma power (F) during nighttime. n
= 7 per group. Each circle represents one mouse. Data are presented as mean
Sleep homeostasis is essential for overall physiological health, as disruption can impair immune function and increase susceptibility to infections and mood disorders. To determine whether quercetin regulates the maintenance of sleep homeostasis, we modeled a high-arousal state induced by sleep disruption using ASD in mice (Fig. 4A). Animals were subjected to a single-cycle of 6-h SD beginning at zeitgeber time 6, followed by a 24-h EEG-EMG recording to evaluate sleep latency and sleep-wake states across the following four recovery phases: 1–6 h, 7–12 h, 13–18 h, and 19–24 h post-SD (Fig. 4B,C). Notably, quercetin did not affect sleep latency after ASD (Fig. 4B). During the first 1–6 h of recovery, the ASD group exhibited significantly increased NREM sleep and REM sleep compared with the vehicle group, concomitantly reducing wake time. In SD + quercetin mice, the percentage of NREM sleep was increased and REM sleep was decreased, while reducing wake time and increasing NREM sleep compared with that in the control group, with no difference in REM sleep (Fig. 4D).
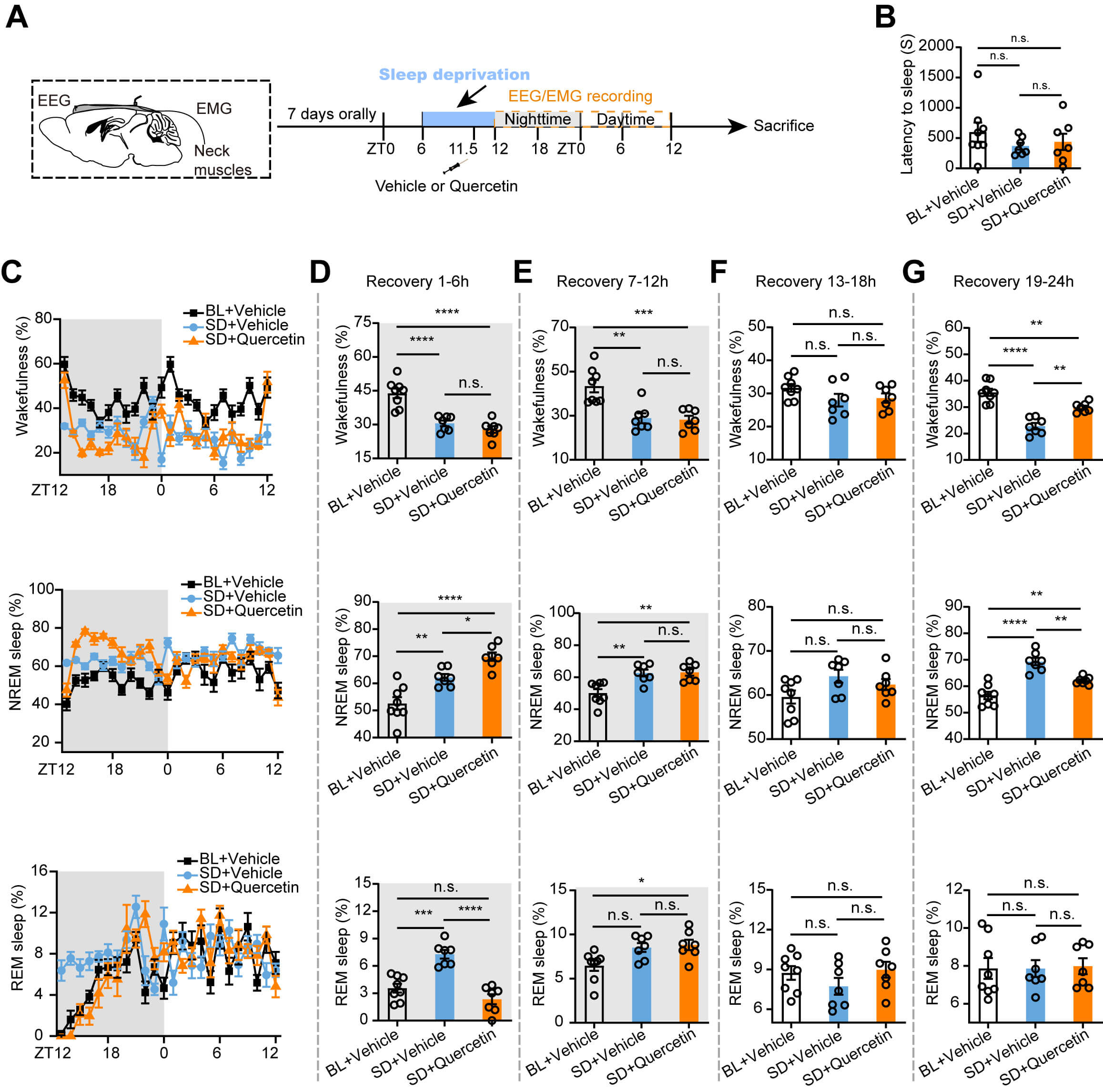 Fig. 4.
Fig. 4.
Quercetin significantly improves sleep homeostasis in ASD mice.
(A) Flow chart showing sleep recording after ASD. (B) The statistical chart shows
that i.p. injection of quercetin has no significant difference in sleep latency
after ASD. (C) The line chart shows the percentage of time spent in wakefulness
(upper), NREM sleep (middle), and REM sleep (lower) during a 24-hour sleep
recovery period in mice. (D–G) Statistical graphs show the percentage of
wakefulness (upper), NREM sleep (middle), and REM sleep (lower) during the 1–6 h
(D), 7–12 h (E), 13–18 h (F), and 19–24 h (G) recovery periods, respectively.
BL + Vehicle: n = 8, SD + Vehicle: n = 7, SD + Quercetin: n = 7. Each circle
represents a mouse. Data are expressed as mean
During 7–12 h of recovery, the SD group exhibited increased NREM sleep and decreased wake time compared with the vehicle group. In quercetin + SD mice, no differences were observed in wakefulness, NREM, and REM sleep compared to the SD group. However, quercetin + SD mice spent less time in wakefulness and exhibited increased NREM and REM sleep compared with that in the control group (Fig. 4E). No statistical differences were observed among vehicle, SD, and quercetin groups during the 13–18 h recovery phase (Fig. 4F). Notably, at 19–24 h post-SD, the SD mice exhibited reduced wakefulness and increased NREM sleep compared to the control group, whereas quercetin + SD mice exhibited increased wakefulness and decreased NREM sleep compared with that in the SD mice, indicating a trend toward normalization (Fig. 4G).
Herein, EEG power spectra were analyzed during NREM sleep in 6-h intervals across the 24-h recovery period to assess the effects of quercetin on sleep pressure during recovery. Notably, delta (0.5–4 Hz) and gamma (30–80 Hz) wave powers among the control, SD, and quercetin + SD groups showed no significant differences across all recovery phases (Fig. 5A–L). These findings suggest that quercetin may have the potential to promote the recovery of sleep homeostasis following ASD without altering sleep pressure.
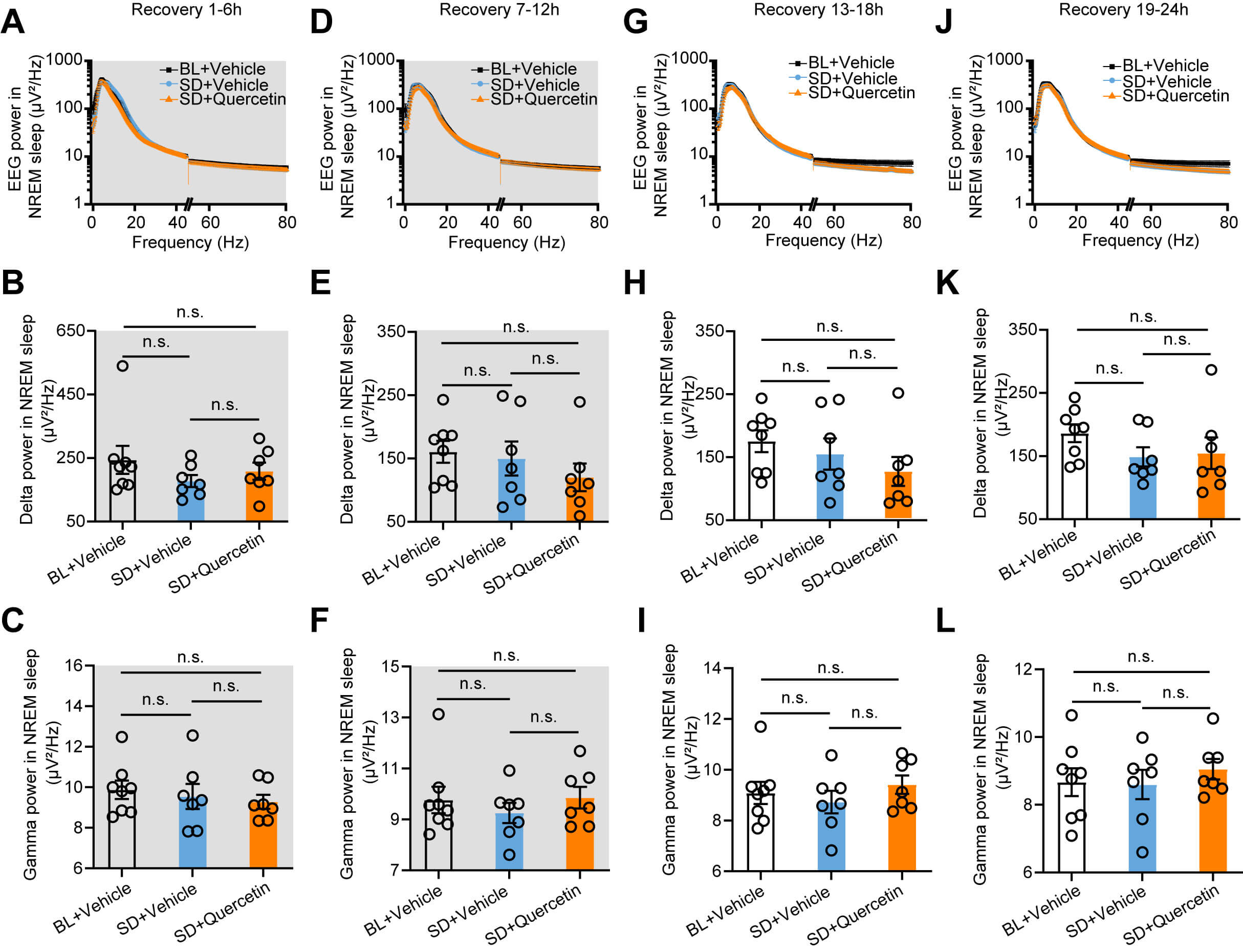 Fig. 5.
Fig. 5.
Quercetin did not affect the EEG power spectrum during the sleep
recovery period after ASD. (A,D,G,J) Statistical graphs show EEG power spectrum
analysis of NREM sleep during the 1–6 h (A), 7–12 h (D), 13–18 h (G), and
19–24 h (J) of sleep recovery after acute sleep deprivation. (B,E,H,K)
Statistical charts illustrate the effects of quercetin on delta power during the
recovery periods of 1–6 h (B), 7–12 h (E), 13–18 h (H), and 19–24 h (K) after
ASD. (C,F,I,L) Statistical charts show the effects of quercetin on gamma power
during the recovery periods of 1–6 h (C), 7–12 h (F), 13–18 h (I), and 19–24
h (L) after ASD. BL + Vehicle: n = 8, SD + Vehicle: n = 7, SD + Quercetin: n = 7.
Each circle represents a mouse. Data are presented as mean
The role of quercetin in restoring sleep homeostasis under CSD was examined in mice undergoing CSD for seven consecutive days, followed by 24-h sleep recording (Fig. 6A). No statistical differences were present in sleep latency among the control, CSD, and CSD + quercetin groups (Fig. 6B). We then analyzed the 24-h recovery period to examine the proportion of wakefulness, NREM sleep, and REM sleep (Fig. 6C).
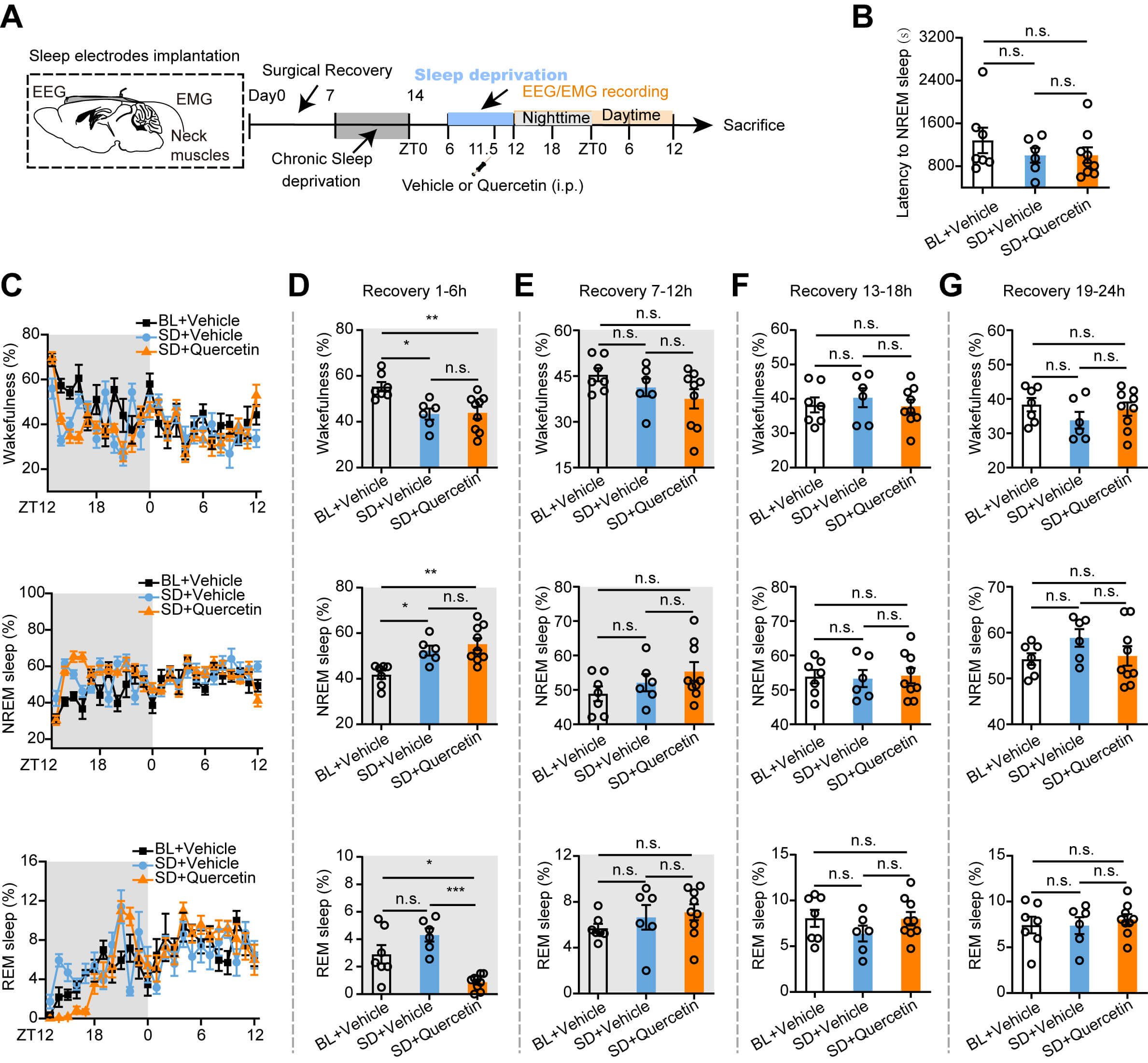 Fig. 6.
Fig. 6.
Quercetin has few significant effects on sleep homeostasis in
CSD mice. (A) Flow chart of sleep recording after CSD. (B) Statistical chart
showing the sleep latency in CSD mice after quercetin treatment. (C) Percentages
of wakefulness (upper), NREM sleep (middle), and REM sleep (lower) in different
groups. (D–G) Statistical charts show the percentage of wakefulness (upper),
NREM sleep (middle), and REM sleep (lower) during the 1–6 h (D), 7–12 h (E),
13–18 h (F), and 19–24 h (G) recovery periods, respectively. BL + Vehicle: n =
7, SD + Vehicle: n = 6, SD + Quercetin: n = 9. Each circle represents a mouse.
Data are presented as mean
During the first 1–6 h of sleep recovery, the CSD group exhibited decreased wakefulness and increased NREM sleep compared to control mice. Unlike the ASD group, the CSD + quercetin group exhibited decreased REM sleep compared to the CSD group, with no significant differences in wakefulness and NREM sleep. Compared with the control mice, the CSD + quercetin mice experienced increased NREM sleep, following decrease in time spend in wakefulness and REM sleep (Fig. 6D). No significant differences were observed in wakefulness, NREM, and REM sleep among the control, CSD, and CSD + quercetin groups during the subsequent recovery phases (7–12, 13–18, and 19–24 h, Fig. 6E–G).
Additionally, post-CSD EEG power analysis revealed no significant differences in delta or gamma wave bands during NREM sleep (Fig. 7A–L). Altogether, these results suggest that a single administration of quercetin exerts minimal influence on restoring sleep homeostasis following CSD.
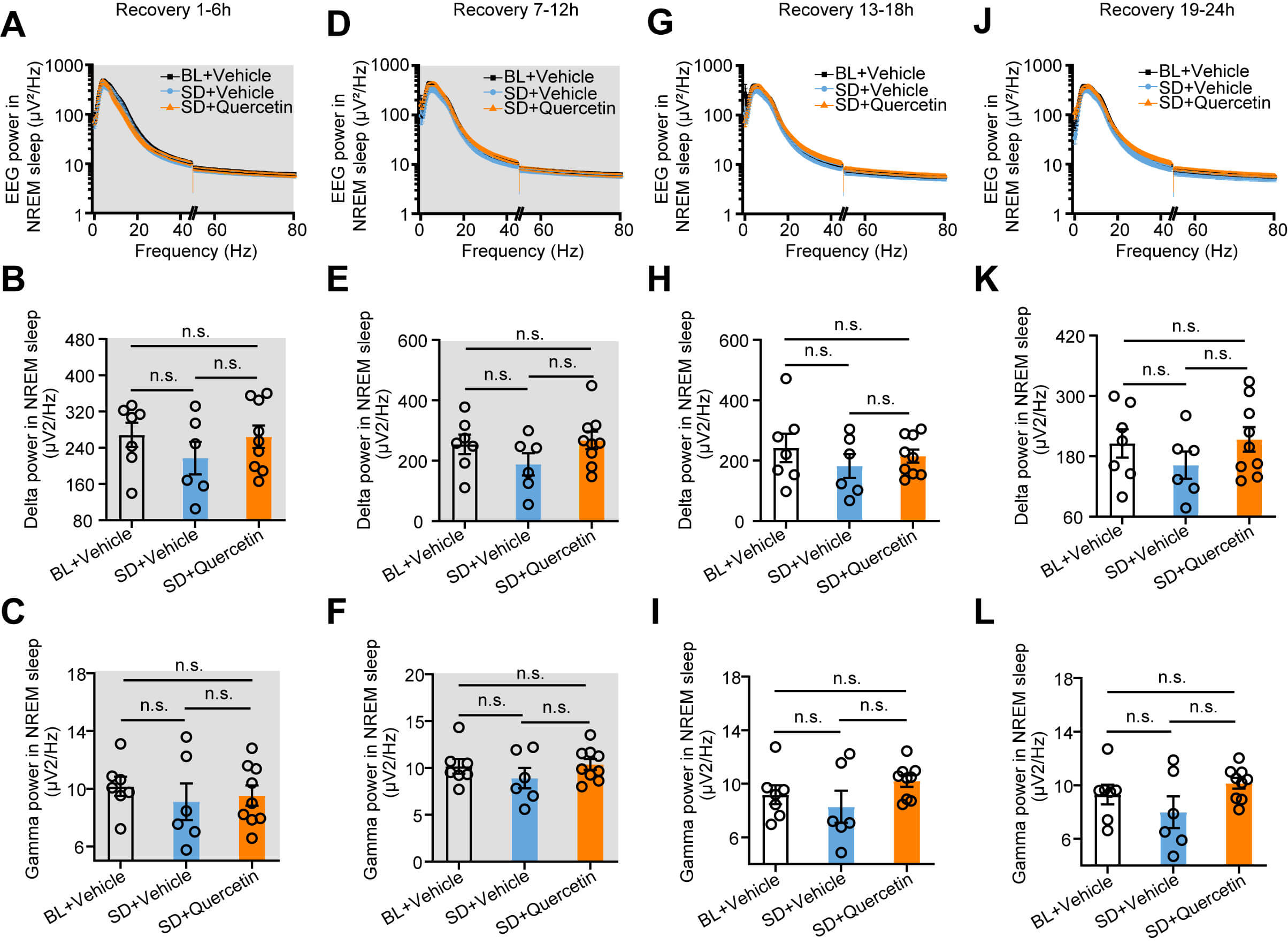 Fig. 7.
Fig. 7.
Quercetin did not affect the EEG power spectrum in chronically
sleep-deprived mice. (A,D,G,J) Statistical graphs showing EEG power spectrum
analysis in NREM sleep during the 1–6 h (A), 7–12 h (D), 13–18 h (G), and
19–24 h (J) recovery periods after CSD. (B,E,H,K) Statistical charts show the
effects of quercetin on delta power during the 1–6 h (B), 7–12 h (E), 13–18 h
(H), and 19–24 h (K) sleep recovery periods after CSD. (C,F,I,L) Statistical
charts show the effects of quercetin on gamma power during the sleep recovery
periods of 1–6 h (C), 7–12 h (F), 13–18 h (I), and 19–24 h (L) after CSD. BL
+ Vehicle: n = 7; SD + Vehicle: n = 6; SD + Quercetin: n = 9. Each circle
represents a mouse. Data are presented as mean
Based on the sleep homeostasis-restoring effect of quercetin after ASD, its impacts on neuronal activity and microglia activation in specific brain regions were explored. Mice received a single i.p. injection of quercetin (200 mg/kg) immediately after ASD, followed by immunofluorescence staining for c-Fos (Fig. 8A) and Iba1 (Fig. 9A). Examination included various key sleep-related brain regions, including the mPFC, nucleus accumbens (NAc), bed nucleus of the stria terminalis (BNST), paraventricular thalamic nucleus (PVT), hippocampal dentate gyrus, basolateral amygdala (BLA), and periaqueductal gray (PAG).
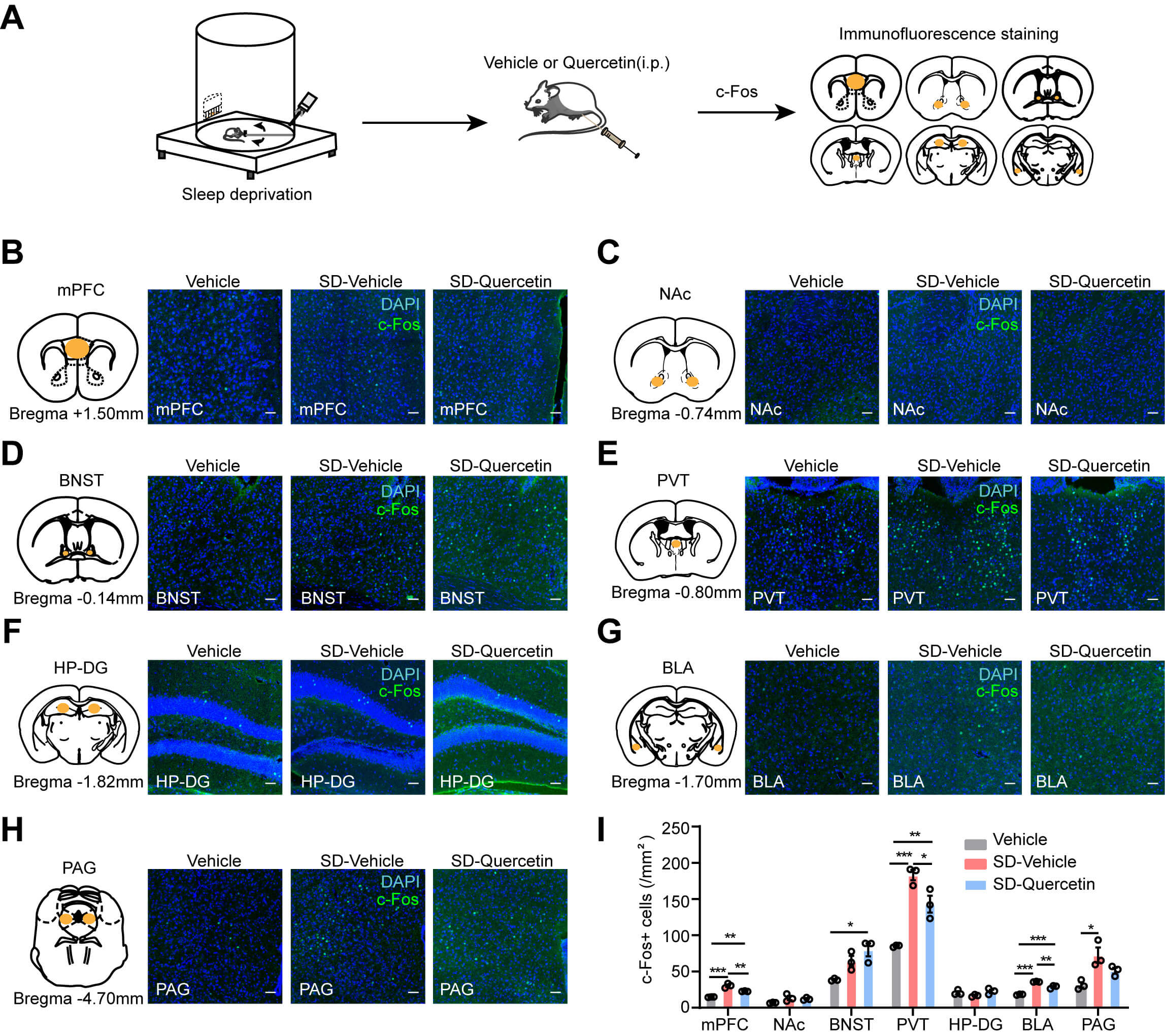 Fig. 8.
Fig. 8.
Effects of quercetin on c-Fos expression in brain regions of
acutely sleep-deprived mice. (A) Flow chart showing the effects of quercetin on
c-Fos expression in brain tissues of acutely sleep-deprived mice. (B–H)
Representative images show the expression of c-Fos in the mPFC (B), NAc (C), BNST
(D), PVT (E), HP-DG (F), BLA (G), and PAG (H) of mice in the Vehicle, SD-Vehicle,
and SD-Quercetin groups. Scale bar, 50 µm. (I) Histograms display
the number of c-Fos-positive cells in the mPFC, NAc, BNST, PVT, HP-DG, BLA, and
PAG regions across the three groups. n = 3 per group. Each circle represents a
mouse. Data are presented as mean
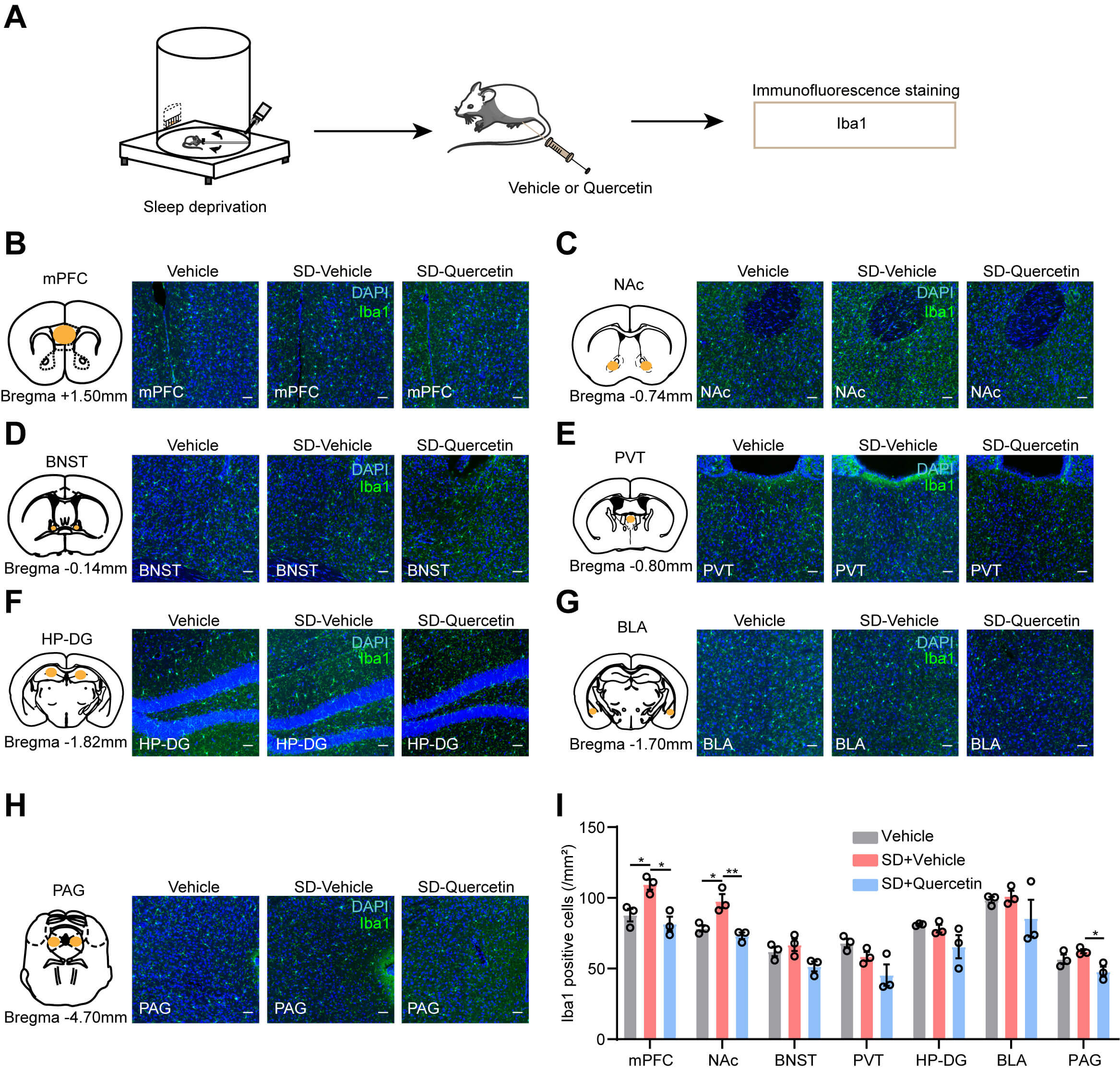 Fig. 9.
Fig. 9.
Quercetin regulates the activation of microglia in brain regions
of acutely sleep-deprived mice. (A) Experimental flow chart for evaluating the
effect of quercetin on microglial activation after ASD. (B–H) Representative
images showing the expression of Iba1 in the mPFC (B), NAc (C), BNST (D), PVT
(E), HP-DG (F), BLA (G), and PAG (H). Scale bar, 50 µm. (I)
Histograms showing the number of Iba1-positive cells in the brain regions
corresponding to (B–H). n = 3 per group. Each circle represents an individual
mouse. Data are presented as mean
Notably, c-Fos staining revealed that quercetin attenuated the SD-induced c-Fos upregulation in the mPFC, PVT, and BLA, whereas it enhanced c-Fos expression in the BNST (Fig. 8B–I). Iba1 immunostaining revealed that quercetin significantly reduced SD-induced microglial activation, as indicated by decreased Iba1 immunoreactivity in the mPFC, NAc, and PAG (Fig. 9B–I). Overall, these results suggest that quercetin suppresses microglial activation and c-Fos expression in critical sleep-regulatory brain regions, potentially underpinning its modulatory effects on sleep homeostasis.
This study aimed to systematically elucidate the regulatory effects of quercetin in sleep homeostasis and its underlying neuroimmunological mechanisms. Notably, quercetin exerts an NREM sleep-promoting effect under basal conditions, and after ASD, it modulated sleep architecture, thereby suppressing excessive neuronal activation in specific brain regions and alleviating microglial inflammatory responses. Altogether, these findings provide novel insights into the mechanisms underlying the natural compound-mediated regulation of sleep and neuroimmunity.
Due to its limited blood–brain barrier (BBB)-penetrating ability, quercetin requires higher doses in studies on CNS mechanisms in mice compared to those in peripheral anti-inflammatory/antioxidant experiments. According to reports, doses of 10–200 mg/kg are considered effective for quercetin-mediated regulation of sleep-wake cycles in rodents [21, 22]. Recent study on regulating CNS-mediated sleep regulation has consistently used a dose of 200 mg/kg, which effectively targets brain neurotransmitter systems to modulate sleep [23]. Regarding safety, a toxicity study on the hydroethanolic extract of Dolichandra unguis-cati L. leaves showed that a single, high-dose oral administration did not cause mortality or abnormal behaviors in male and female rats over 14 d. Furthermore, the rats exhibited normal weight gain, with no pathological abnormalities in core indicators of liver and kidney function [24]. Based on these findings, the 200 mg/kg dose setting (upper limit) was included in this study, which also exhibits basic safety.
A Study investigating quercetin’s role in treating sleep deprivation-related memory impairment has shown that acute REM SD can activate inflammatory signaling pathways in the hippocampus [18]. A study by Wu et al. [13] reported that orally administered quercetin nanoparticles could regulate sleep duration under high-altitude conditions, providing critical evidence for developing drug treatments for altitude-related sleep disorders. The present study induced SD by disrupting sleep via the rotarod method and employed the i.p. method to administer quercetin, thereby expanding its application in modulating normal sleep rhythms and addressing both ASD and CSD. Additionally, this study investigated cellular activity and microglial activation in key brain regions involved in sleep regulation.
Despite accumulating evidence indicating the therapeutic potential of quercetin in sleep research, various challenges persist in its clinical translation. For instance, free quercetin can be enzymatically hydrolyzed in the gastrointestinal tract or be challenging to absorb owing to low solubility [25]. Nevertheless, recent advancements in nanoformulation technology have provided promising solutions to these issues. For example, quercetin-maize peptide nanoliposomes, synthesized using multifrequency pulsed ultrasonic methods, exhibited significantly enhanced quercetin-encapsulation efficiency, gastrointestinal stability, and transmembrane transport efficiency (they could penetrate the tight junctions of small intestinal epithelial cells) [25], thereby enhancing their potential for CNS targeting. Regarding the clinical translation of neuropsychiatric disease treatment, quercetin-loaded chitosan nanoparticles (Qu-Ch NPs) have demonstrated notable advantages, including a low dose and high safety [26]. Low-dose Qu-Ch NPs exert strong neuroprotective effects by modulating antioxidant and anti-inflammatory pathways.
Advances in nose-to-brain delivery technology have further established an effective route for quercetin entry into the CNS. Notably, traditional oral and intravenous methods achieved extremely low quercetin concentrations in the brain due to the restrictive BBB. In contrast, intranasally delivered quercetin nanoparticles exhibited direct entry into the brain via the olfactory nerve pathway [27]. Notably, mitochondria-targeted quercetin nanoformulations have been shown to combat Alzheimer’s disease-related ferroptosis through a triple mechanism of “iron chelation-pathway regulation-lipid protection” [28], offering strong experimental evidence for their potential in treating neurodegenerative diseases. However, regardless of their potential for clinical translation, further research is necessary on quercetin nanoformulations to clarify the correlation between “dose-central concentration-pharmacodynamic effect” and comprehensive long-term safety. Overall, optimizing quercetin nanoformulations and delivery systems can effectively overcome the major limitation of low bioavailability, highlighting their substantial potential for clinical application in treating neuropsychiatric diseases.
The power of delta waves (0.5–4 Hz) during NREM sleep is a classical indicator of sleep pressure dynamics. Additionally, sleep latency directly reflects the intensity of sleep drive and represents an intuitive metric of sleep pressure. Reportedly, the interaction between “homeostatic pressure (Process S)” and “circadian rhythm (Process C)” governs the sleep-wake balance [29]. Notably, Process S, also referred to as the “traditional homeostatic pathway” of sleep, has the following characteristic core feature: the longer the duration of wakefulness, the greater the accumulation of sleep pressure. Accumulated sleep pressure results in a compensatory increase in delta wave power during sleep, ultimately facilitating pressure dissipation [30, 31]. During wakefulness, neuronal activity-associated metabolite accumulation, coupled with elevated proinflammatory cytokine levels, enhances neuronal excitability and synergistically promotes the buildup of sleep pressure [32]. Together, they act on specific brain regions, including the basal forebrain and hypothalamus, to inhibit wake-promoting pathways, activate sleep-promoting pathways, and induce sleep [33]. Following sleep onset, slow-wave sleep (SWS) and delta wave activity direct the traditional homeostatic pathway into a pressure-dissipation phase, as this pathway activation relies on various factors beyond delta wave power alone, including adenosine concentration, sleep latency, and SWS proportion. It is important to emphasize that the traditional sleep homeostatic pathway does not regulate sleep independently.
Reportedly, sleep initiation involves two parallel pathways: the “homeostasis-dependent” and the “homeostasis-independent” pathways [34]. The traditional pathway relies on the accumulation of homeostatic factors, such as adenosine. In contrast, the homeostasis-independent pathway initiates sleep by directly regulating sleep-wake switching neurons, for example, through gamma-aminobutyric acid (GABA), which mediates receptor-driven inhibition of wake-promoting pathways [35]. Quercetin has been shown to act on the CNS primarily by inhibiting neuroinflammation and regulating oxidative stress levels [21, 36].
In the present study, quercetin treatment reduced sleep latency under basal conditions; however, it exhibited no significant effect on post-SD sleep latency. Additionally, quercetin did not significantly affect delta wave activity during sleep under both baseline conditions and post-SD recovery phase. Integrating the aforementioned literature findings with the present results, we hypothesize that quercetin regulates sleep by modulating cerebral neuronal excitability and microglial activity, rather than interfering with sleep pressure accumulation reflected by delta waves. This finding is consistent with a previously reported study [21]. Additionally, this process is likely associated with the cortical-limbic system and brainstem multilevel neural circuits that maintain sleep-wake homeostasis. To clarify this association, the roles of key sleep-regulatory brain regions were delineated in this study.
Notably, sleep-wake homeostasis regulation depends on coordination among multilevel neural circuits spanning the cortex, limbic system, and brainstem. Insomnia is often associated with hyperactivity or disrupted network connectivity in different important brain regions. For instance, the mPFC has been shown to drive wakefulness by enhancing thalamocortical synchronization [37]. Similarly, the NAc regulates sleep-wake cycles primarily via dopaminergic and glutamatergic signaling, with hyperactivity of dopaminergic neurons potentially prolonging wakefulness in chronic stress or insomnia conditions [38]. Furthermore, the BNST and BLA integrate limbic inputs to modulate the sleep-wake balance. The BNST affects this balance by regulating GABAergic signaling. For example, pharmacological or optogenetic inhibition of GABAergic neurons in the BNST reportedly improved sleep continuity and reduced stress-induced wakefulness [39]. The BLA processes stress and emotional memory, structural and functional alterations and chronic insomnia [40, 41]. Notably, acute stress can trigger the PVT, a relay station connecting the cortex, limbic system, and brainstem, to activate glutamatergic projections to the central amygdala, thereby promoting hyperarousal [42]. Additionally, the hippocampus and ventrolateral PAG (vlPAG) further contribute to sleep-wake regulation via homeostatic and neuroprotective mechanisms. For instance, the hippocampus can suppress excessive stress responses via the hypothalamic-pituitary-adrenal (HPA) axis, and its dysfunction has been associated with anxiety and depression comorbid with insomnia [43]. In contrast, neural pathways in the vlPAG are critically implicated in stress-induced recovery sleep. For example, neurotensinergic neurons in the vlPAG have been shown to promote NREM sleep via GABAergic projections to arousal-promoting nuclei, directly linking brainstem sleep circuits and stress [44]. Altogether, these findings suggest that dysfunction across multiple brain regions can induce insomnia. Further studies on dynamic interactions between these neural circuits are required to fully elucidate the mechanisms underlying quercetin-induced precise therapeutic effects on insomnia.
The results of this study showed that quercetin facilitated a compensatory increase in NREM sleep during the early recovery phase (1–6 h) post-ASD, whereas in the late recovery phase (19–24 h), it promoted normalization of the sleep-wake cycle. Notably, the compensatory increase in NREM sleep was primarily concentrated in the early recovery phase. Importantly, NREM sleep contributes to synaptic pruning, metabolic waste clearance, and overall bodily recovery [45]. These results suggest that quercetin-mediated regulation of NREM sleep during the early recovery phase aligns with the physiological demands of the body and provides important support for restoring physiological homeostasis.
SD-induced neuroinflammation in the brain has been characterized by the
upregulation of inflammatory factors, such as IL-6, IL-1
A key aspect warranting detailed discussion is that quercetin exerts sleep-regulating effects by attenuating neuroinflammation within specific brain regions. Recent studies report that quercetin exerts anti-inflammatory effects in neurodegenerative diseases by modulating inflammatory cytokine levels in the brain [47, 48]. Furthermore, quercetin can cross the BBB and is widely distributed across the cerebral cortex and limbic system. A previous study confirmed that quercetin mitigated sodium fluoride-induced structural and functional damage to the rat mPFC by enhancing antioxidant capacity and inhibiting apoptosis, offering a potential strategy for protecting the CNS from neurotoxicity [49]. However, direct evidence for the anti-inflammatory effect of quercetin within the NAc remains limited. Therefore, the observed reduction of microglia in the NAc in the present study may be attributed to quercetin-mediated suppression of inflammation in the mPFC, which then reduced the transmission of pro-inflammatory signals to the NAc, thereby alleviating local neuroinflammation.
c-Fos, a key marker of neuronal activation, plays a critical role in assessing neural activity in sleep-regulatory brain regions. This study showed that quercetin inhibited c-Fos expression in the mPFC, PVT, and BLA, while increasing its expression in the BNST, compared with that in the control group. This pattern needs to be further explored to elucidate underlying mechanisms. As a key component of the extended amygdala, the BNST is closely linked with high-arousal behavioral states. It is a highly heterogeneous region, exhibiting extensive diversity in cell types, with notable variations in spatial organization, molecular expression, synaptic inputs, axonal outputs, and functional regulation [50]. Various subtypes of BNST neurons project to brain regions essential for sleep-wake regulation, including the lateral hypothalamus (LH), ventral tegmental area, and arcuate nucleus [50]. Notably, optogenetic activation of BNST GABAergic neurons in mice has been shown to promote rapid transitions from NREM sleep to wakefulness; these neurons are also highly active during wakefulness and REM sleep [51]. Studies have confirmed bidirectional functional connectivity between the LH and BNST [52, 53]. Additionally, excitatory BNST projections have been reported to activate REM-active neurons in the sublaterodorsal nucleus of the brainstem, contributing to REM sleep regulation [54]. Notably, quercetin treatment further increased c-Fos expression in the BNST in this study, suggesting that its effects may involve the neurochemical and cellular heterogeneity of the BNST. Future studies need to focus on integrating EEG-EMG and fiber photometry recordings, along with two-photon imaging, to monitor quercetin-induced sleep regulation in the cell-type-specific BNST neurons.
In contrast to its pronounced efficacy in ASD models, quercetin exhibited
limited effectiveness in restoring post-CSD sleep homeostasis, showing only a
reduction in REM sleep during the early recovery phase. ASD mainly impairs
attention and working memory, along with inducing transient oxidative stress in
the hippocampus and prefrontal cortex [55]. Mechanistically, ASD transiently
activates the HPA axis and triggers microglial activation, leading to localized
inflammation without systemic spread. In contrast, CSD causes “multi-target,
irreversible” damage. Prolonged overactivation of the HPA axis results in a
“depleted state”, accompanied by dysregulation of multiple neurotransmitter
systems [4]. Reportedly, a study on rodents has shown that CSD disrupts BBB
integrity, exacerbates microglial activation, and elevates proinflammatory
cytokine levels (such as TNF-
The findings of this study reveal the potential of quercetin in promoting NREM sleep under basal conditions and post-ASD sleep homeostasis restoration, without influencing sleep pressure. The underlying mechanisms may involve inhibiting neuroinflammation and excessive neuronal activation in specific sleep-regulatory brain regions. These findings provide novel evidence supporting the role of natural compounds in regulating sleep and neuroimmune interactions, along with offering quercetin as a promising candidate for treating acute sleep disorders. Future research needs to aim to refine quercetin administration protocols, elucidate region-specific molecular mechanisms, and develop formulations with improved bioavailability, thereby laying a solid foundation for the clinical treatment of sleep disorders.
The data during the current study are available from the corresponding author on reasonable request.
RH and BW designed the research study. BW, ZH, and PF performed animal surgeries, sleep recordings, and immunohistochemistry. ZL, BN, and JZ analyzed EEG data and conducted statistical analyses. RH and BW wrote the initial draft. JZ offered experimental guidance. All authors contributed to editorial changes in the manuscript. All authors revised and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
All animal experiments were approved by the Institutional Animal Care and Use Committee of Xinxiang Medical University (IACUC Protocol No. XYLL-20240521), and we comply with all relevant ethical regulations. Every effort was made to minimize the number of animals used and reduce their suffering throughout the study. This study was conducted in accordance with the Guidelines for the Care and Use of Laboratory Animals (8th edition, National Institutes of Health, USA). All protocols were designed to adhere to the 3Rs principles (Replacement, Reduction, Refinement) of animal research and were periodically reviewed by the IACUC to ensure ongoing compliance with ethical standards.
We sincerely thank Professor Hui Wang for providing the valuable experimental platform and offering rigorous guidance on the experiments. We thank Dr. Yunwei Lou (Xinxiang Medical University) and Dr. Zhitao Gao (Xinxiang Medical University) for their technical guidance and constructive comments. Additionally, the authors appreciate the contributions of all laboratory members for their help with animal care and sample preparation during the study.
This work was financially supported by Key Scientific and Technological Projects of Henan Province (Grant No. 242102310372), and Key Scientific Research Projects of Higher Education Institutions in Henan Province (Grant No. No.23B320001), and China National University Student Innovation & Entrepreneurship Development Program of Xinxiang Medical University (Grant No. 202410472007), and the Natural Science Foundation of Henan Province (Grant No. 202300410314), and Higher Education Discipline Innovation Project (111 Project, No. D20036).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.









