1 Institute of Acupuncture and Moxibustion, China Academy of Chinese Medical Sciences, 100700 Beijing, China
2 Department of Scientific Research, The Affiliated Hospital of Jiangxi University of Chinese Medicine, 330006 Nanchang, Jiangxi, China
Abstract
The spinal dorsal horn (SDH) plays a crucial role in nociceptive processing. However, the temporal dynamics of neuronal excitability across different laminae during inflammatory pain remain incompletely understood.
Complete Freund’s adjuvant (CFA) was injected into the left hindpaw to induce inflammatory pain. Spontaneous pain behaviors were evaluated using CatWalk gait analysis and weight-bearing tests, while mechanical hypersensitivity was assessed using von Frey filaments. Neuronal activation patterns were mapped using c-Fos (a protein product of the c-Fos immediate-early gene) immunolabeling across superficial and deeper laminae of the SDH. Spontaneous and mechanically-evoked neuronal discharges were recorded in vivo using multi-electrode arrays.
Spontaneous pain behaviors were most pronounced during the first 3 days post CFA injection, with mechanical hypersensitivity persisting through day 7. A marked increase in c-Fos-positive neurons was observed specifically in superficial laminae on day 1, with no significant changes detected in the deeper laminae. Spontaneous and mechanically-evoked firing rates of SDH neurons increased significantly during days 1–5 post CFA injection. Importantly, wide dynamic range (WDR) neurons exhibited the greatest increase in evoked discharge frequency, while low-threshold mechanoreceptor (LTM) neurons showed the greatest proportional increase amongst neuronal subtypes. Furthermore, both WDR and LTM neurons shifted towards a more superficial distribution.
Peripheral inflammatory pain induced distinct alterations in SDH neurons, characterized by an early increase in neuronal activities, followed by changes in the spatial distribution and proportion of WDR and LTM neurons.
Keywords
- spinal cord dorsal horn
- nociceptive pain
- electrophysiological phenomena
- multi-channel electrode
The spinal dorsal horn (SDH) serves as the first relay station for somatosensory information in the central nervous system. It is organized into distinct laminae based on cellular architecture and function. The complex, interconnected network within this laminar organization plays a crucial role in modulating sensory inputs before they reach the supraspinal region [1]. Understanding alterations in neuronal excitability at a population level is critical for elucidating the spinal processing of nociception that underlies pathological pain conditions.
Neuronal populations in the SDH form synaptic connections with the central
terminals of diverse primary sensory neurons in a spatially organized pattern
[2]. Neurons in the superficial and intermedial SDH laminae predominantly receive
nociceptive and innocuous information, respectively. Neurons in the deep laminae
process both noxious and innocuous signals and are described functionally as
having wide dynamic range (WDR). These multi-receptive neurons are predominantly
distributed in the deep laminae (V–VI) [3], with sparse distribution in
superficial laminae [4, 5]. Nociceptive-specific (NS) neurons primarily synapse
with A
Electrophysiological techniques offer superior temporal resolution for monitoring neuronal activity while maintaining organism integrity. Previous single-unit in vivo recordings were often limited in their ability to achieve simultaneous population-level analysis. Although calcium imaging allows real-time, simultaneous assessment of activity across a vast majority of neurons in behaving animals, limitations persist in terms of recording depth and acquisition rate [9]. Multi-electrode arrays (MEAs) with integration of multiple microelectrodes enable simultaneous recording from multiple sites across different laminae of the dorsal horn. The application of this methodology in the rodent dorsal horn may lead to a better understanding of the laminar differences within the SDH network [10]. MEA has been successfully employed in rat models to examine integrated signaling patterns among SDH neuronal populations [11, 12]. Moreover, recent advances in MEA miniaturization have extended these recordings to both superficial and deep laminae in murine models.
In the present study, we systematically investigated functional changes in SDH neuronal excitabilities using complete Freund’s adjuvant (CFA)-induced inflammatory pain mice. Nociceptive sensitization was evaluated through behavioral assessments of spontaneous pain and mechanical hyperalgesia, while SDH activation in laminae I–II was confirmed using c-Fos immunohistochemistry in combination with laminar-specific marker analysis. Finally, 32-channel MEA recording was employed to characterize population-level neuronal dynamics. This work revealed significant alterations in the firing rates, spatial distribution, and functional organization of distinct neuronal subpopulations within the SDH of mice with CFA-induced inflammatory pain.
A total of 95 healthy male C57BL/6J mice (age 6–8 weeks) without physical
abnormalities and weighing 20–22 g [Beijing Vital River Laboratory Animal Technology Co., Beijing, China. license number:
SCXK (Jing) 2021-006] were used in this study. The mice were allocated as
follows: 49 for pain behavioral assays, 15 for immunofluorescence, and 31 for
electrophysiological recordings. Cohorts did not overlap across assays. The
animals were maintained under controlled environmental conditions with a 12-h
light/dark cycle and a temperature of 24
For the CFA-treated group, mice received an intraplantar injection of 20 µL of CFA (F5881, Sigma-Aldrich, St. Louis, MO, USA) into the left hindpaw during isoflurane (R510-22-10, RWD Life Science Co.) inhalation anesthesia (1%–4%). The CFA solution was administered slowly and allowed to remain in situ for 2 minutes before withdrawal to ensure complete absorption, with no observable leakage detected. Mice that failed to develop CFA-induced paw inflammation (swelling/redness) were excluded (Supplementary Fig. 1). For the control group, naïve mice were used without sham handling or injection. For all experiments, mice were allocated to experimental groups using a computer-generated random number sequence upon completion of acclimatization. For behavioral cohorts, this randomization was performed after baseline behavior testing to ensure equivalent starting scores across groups.
Gait analysis was performed using the CatWalk XT system (version 10.6, Noldus
Information Technology, Wageningen, The Netherlands) to observe spontaneous pain
behaviors as previously described [13]. Briefly, the system consists of an
enclosed walkway with a glass plate, illuminated by a fluorescent lamp that emits
light inside the glass plate. Each mouse was placed individually in the CatWalk
system and allowed to walk freely and traverse from one side of the walkway glass
plate to the other. Where the mouse paws made contact with the glass plate, light
was reflected down and the illuminated contact areas were recorded with a
high-speed color video camera positioned underneath the glass plate. The
recordings were carried out when the room was completely dark, except for light
from the computer screen. The software automatically labeled all areas containing
pixels above the set threshold and assigned them to the respective paws. Mice
were trained to traverse the walkway for 2 days before baseline measurements.
Animals were excluded if they failed to run voluntarily. Each mouse was tested at
least three times. Five parameters were analyzed: (1) stand phase: duration in
seconds of paw contact with the glass plate in a step cycle; (2) swing phase:
duration in seconds of no paw contact with the glass plate in a step cycle; (3)
duty cycle: stand duration as a percentage of the step cycle duration [stand /
(stand + swing)
To distinguish between inflammatory pain-induced gait alterations and potential motor deficits, motor coordination and balance were evaluated using the accelerating Rotarod test. Prior to testing, the mice underwent a 2-day habituation protocol on the Rotarod apparatus (Mouse-47650, Ugo Basile, Gemonio, Italy). The assessment was performed 24 h post CFA injection, with the Rotarod accelerating linearly from 0 to 40 rpm over a 300-s period. Three trials were conducted for each mouse, and both the latency to fall and terminal velocity were recorded.
In addition to gait analysis, spontaneous nociceptive behaviors were evaluated using a weight-bearing test system (model BIO-SWB-TOUCH-M, Bioseb, Vitrolles, France). Each mouse was placed in an acrylic glass chamber where independent force plates measured the weight distribution between hindpaws. After a 2-day habituation period, weight-bearing measurements were obtained by averaging the applied force (g) over a 10-s sampling period. The weight-bearing differential was calculated by subtracting the contralateral (right hindpaw) from the ipsilateral (CFA-injected left hindpaw) weight-bearing capacity. The final values represent the mean of six consecutive measurements. Animals showing imbalanced bilateral weight-bearing capacity before CFA injection were excluded (Supplementary Fig. 1).
Mechanical hypersensitivity was assessed using calibrated von Frey filaments
(37450-275, Ugo Basile). Mice were individually housed in transparent glass
chambers positioned on an elevated mesh platform. Prior to testing, animals
underwent a habituation protocol consisting of 1-h sessions for 2 consecutive
days, followed by a 30-min acclimation period before each test session. The
mechanical withdrawal threshold was determined by applying von Frey filaments
perpendicularly to the midplantar surface of the ipsilateral hindpaw with
sufficient force to cause filament bending for 3–5 s. A response was considered
positive if the animal exhibited any nocifensive behaviors, including brisk paw
withdrawal, licking, or shaking of the paw, either during application of the
stimulus or immediately after the filament was removed. Paw withdrawal thresholds
were quantified using the modified Dixon up-down method [14, 15]. The 50%
threshold was calculated according to the formula: 50% threshold (g) = 10 (X + kd)/104, where X represents the log unit value of the final filament, k
is the tabular value for the pattern of positive/negative responses, and d
denotes the mean difference in log units between filaments. Animals exhibiting
abnormal mechanical sensitivity (withdrawal threshold
Baseline measurements for gait parameters, weight-bearing distribution, and mechanical sensitivity were obtained 24 h prior to CFA (F5881, Sigma-Aldrich) injection, with subsequent assessments conducted on days 1, 3, 5 and 7 post injection. Motor coordination was evaluated using the Rotarod test. This was conducted only at the 24 h post-injection timepoint. All behavioral tests were performed at approximately 09:00 AM.
Mice were deeply anesthetized with urethane (1.5 g/kg, U2500, Sigma-Aldrich)
followed by transcardial perfusion with saline. It should be noted that no
additional acute stimulation was conducted before perfusion. The lumbar spinal
cord enlargements were harvested and fixed in 4% paraformaldehyde (PFA, P1110,
Solarbio, Beijing, China), then dehydrated in 30% sucrose until the tissue sank
to the bottom. After 24 h, the tissue was embedded in an optimal cutting
temperature compound (4583, Sakura Finetek, Torrance, CA, USA), frozen, and
sectioned at 30-µm thickness using a cryostat (CM1860, Leica, Wetzlar,
Germany). The sections were collected in PBS-filled wells. Following PBS washing,
the sections were blocked with 3% donkey serum (BMS0140, Abbkine, Wuhan, Hubei,
China) and 0.5% Triton X-100 (T9284, Sigma-Aldrich) in PBS for 45 min at room
temperature (RT). The sections were then incubated overnight at 4 °C in
PBS containing 1% donkey serum and 0.5% Triton X-100, and the following primary
antibodies: anti- isolectin B4 (IB4)-488 (1:500, I21411, Invitrogen, Carlsbad,
CA, USA), mouse anti-protein kinase C-gamma (PKC
In vivo extracellular recordings were performed from SDH neurons in
L4–L6 segments. Mice were anesthetized with intraperitoneal urethane (1.5 g/kg, U2500, Sigma-Aldrich).
A laminectomy was performed at T13 and L1 vertebrae to expose the lumbar
enlargement (L4–L6). Following careful removal of the dura mater, the spine was
immobilized using a stereotaxic frame (SR-6R-HT, Narishige, Tokyo, Japan) to
minimize movement artifacts. The exposed spinal cord was continuously perfused
with warm physiological saline. Core body temperature was maintained using a
heating pad. Neural activity was recorded using a 32-channel silicon probe
(A1
The receptive fields (RFs) of recorded neurons were identified through mechanical probing of the ipsilateral hindpaw. Mechanical stimulation of the RFs consisted of brush stimulation using a camel-hair brush. Calibrated force was applied using an electronic rodent pincher (BIO-RPM, Bioseb) consisting of a pair of blunt forceps. One arm of the forceps was flat, while the opposing arm was tipped with a spherical cap 3.5 mm in diameter and 1 mm in height with smooth edges. For brush stimuli, the center of the hindpaw RF was stroked in the rostral to caudal direction about once per second for 15 s. For touch and pinch stimuli, the spherical cap was placed at the center of the hindpaw RFs. The applied force was then quickly increased by hand until it reached 40 g for touch stimulus and 150 g for pinch stimulus. This was held for 15 s using a timer. Stimuli were given in a predefined order (brush, 40 g touch, followed by 150 g pinch) in order to avoid potential diffuse noxious inhibitory controls (DNIC) elicited by the 150 g pinch. Each type of stimulus was given 2–3 times to improve reliability. The inter-trial interval was approximately 1–2 min to avoid neuronal fatigue in response to repeated stimuli. A 10-s stable response period (Supplementary Fig. 4) from each 15-s mechanical stimulus was analyzed to minimize artifacts from potential hand movements. The spontaneous firing rates were defined as the mean frequency of a 30-s baseline period before any stimulation. The stimulus-evoked firing rates were defined as the mean frequency of the 10-s stable response window minus the mean frequency of the 10-s pre-stimulus baseline. Recordings containing fewer than three neurons per animal were excluded to ensure an adequate sample size for population-level analysis (Supplementary Fig. 1).
Neurons were classified into three distinct categories based on their response properties to mechanical stimulation as described previously [16]: (1) LTM neurons that responded exclusively to innocuous stimuli (dynamic brush and/or static touch); (2) WDR neurons that exhibited graded responses to both innocuous and noxious stimuli (pinch), with greater responses to the latter; and (3) NS neurons that responded selectively to noxious pinch.
The depth of each probe was calculated based on the distance from the electrode tip to the spinal cord surface and the spacing between probes. The depth bin for analysis was set as 25 µm. It should be noted that tissue edema caused by surgery and exposure of the spinal cord beyond control may alter the estimated depths. Other potential limitations such as the tilt of the electrode tip and zeroing deviation were controlled as described in the insertion procedure above.
All data are presented as the mean
CatWalk gait analysis was performed to objectively evaluate pain-induced locomotor alterations. This method has been used previously to assess mechanical allodynia in animal models [17]. As shown by the timing view and footprint intensities (Fig. 1A), CFA-treated mice exhibited significant alterations in their bilateral hindlimb stepping pattern. Compared with baseline measurements, CFA-treated mice showed notable reductions in stand phase duration (Fig. 1B), duty cycle (Fig. 1D), single stance duration (Fig. 1E), and max contact max intensity (Fig. 1F) of the affected hindpaw, together with an increase in the swing phase duration (Fig. 1C). These abnormalities were most prominent during the first 3 days post CFA injection, suggesting peak spontaneous pain during this period. However, gait parameters showed a gradual recovery beginning on day 5. To exclude potential motor dysfunction induced by CFA injection, we also performed Rotarod testing. Both the latency and maximal rotational velocity at fall remained unchanged in CFA-treated mice compared with controls (Fig. 1G), indicating the observed gait abnormalities were primarily pain-driven rather than because of motor impairment. We also examined complementary weight-bearing balance and reflexive pain-related behaviors. Both these measurements revealed that CFA-treated mice exhibited sustained spontaneous pain and evoked hypersensitivity, especially in von Frey filament-evoked mechanical allodynia (Fig. 1H,I). While gait analysis indicated partial recovery by at least day 5, weight-bearing asymmetry and decreased mechanical withdrawal thresholds persisted through day 7. Collectively, these behavioral analyses demonstrate that intraplantar CFA injection induces both dynamic temporal changes and persistent static pain.
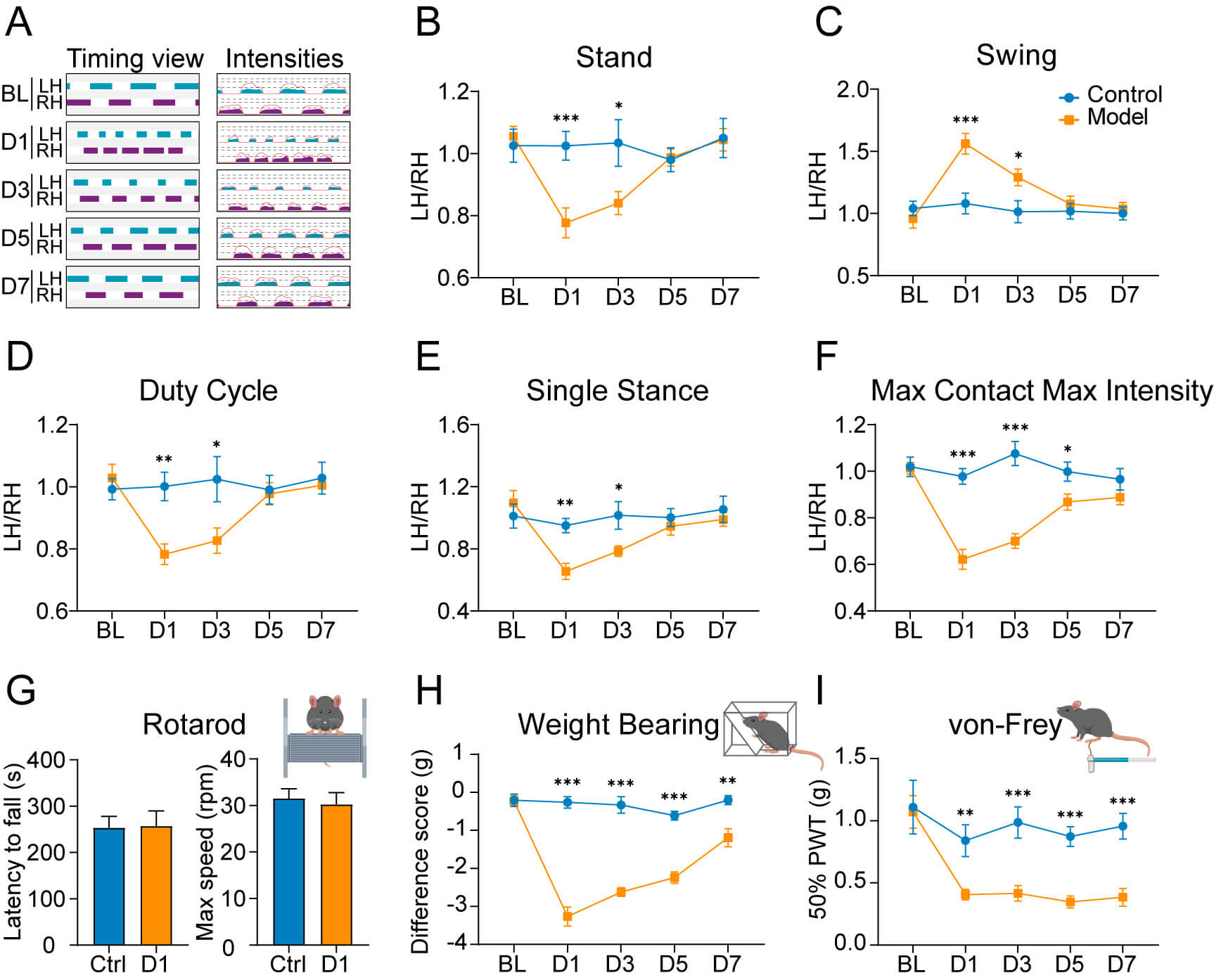 Fig. 1.
Fig. 1.
CFA-induced inflammatory pain induces time-dependent changes in
spontaneous and evoked pain behaviors. (A) Representative CatWalk gait analysis
showing timing views (left) and pressure intensity maps (right) of hindpaw prints
in control mice and at the indicated time points after CFA injection (days 1, 3,
5 and 7). (B–F) Quantitative analysis of various gait parameters assessed by
CatWalk: (B) stand phase duration, (C) swing phase duration, (D) duty cycle, (E)
single stance duration, and (F) max contact max intensity. Data were collected
from control mice and CFA-treated mice. Values were normalized as the ipsilateral
(affected left hindpaw)/contralateral (unaffected right hindpaw) ratio. Mean
Protein expression of the immediate early gene c-Fos has been widely
used as a marker for neuronal activation [18]. To study dynamic and
laminar-specific sensory processing in the SDH, we examined the temporal and
spatial distribution of c-Fos expression across distinct SDH laminae following
CFA injection. IB4 and PKC
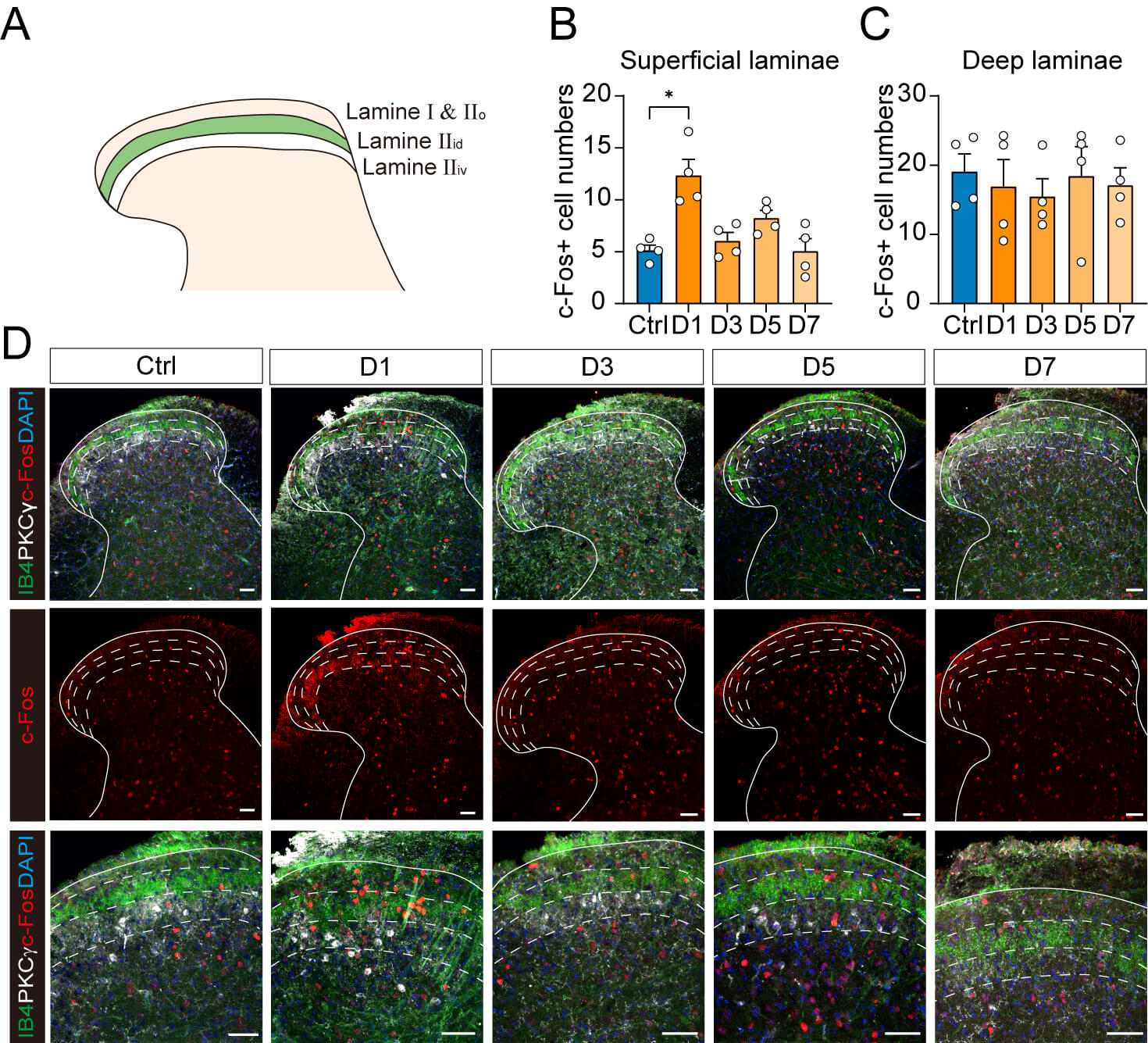 Fig. 2.
Fig. 2.
Time-dependent c-Fos expression across superficial and deep
laminae of the SDH after CFA injection. (A) Schematic representation of mouse
spinal dorsal horn showing lamina IIi delineated by IB4 (green) and
PKC
A comprehensive view of real-time neuronal responsiveness in persistent inflammatory pain was obtained using multi-lamina extracellular recordings to characterize the spatiotemporal dynamics of neuronal excitability throughout the dorsal horn. Conventional c-Fos mapping fails to detect this. Neurons in the SDH were categorized into superficial (0–250 µm) and deep regions (250–850 µm) based on the locations of their recording (Fig. 3A–C, Ref. [20] for C). Each experiment yielded an average of 12 isolated single units per mouse.
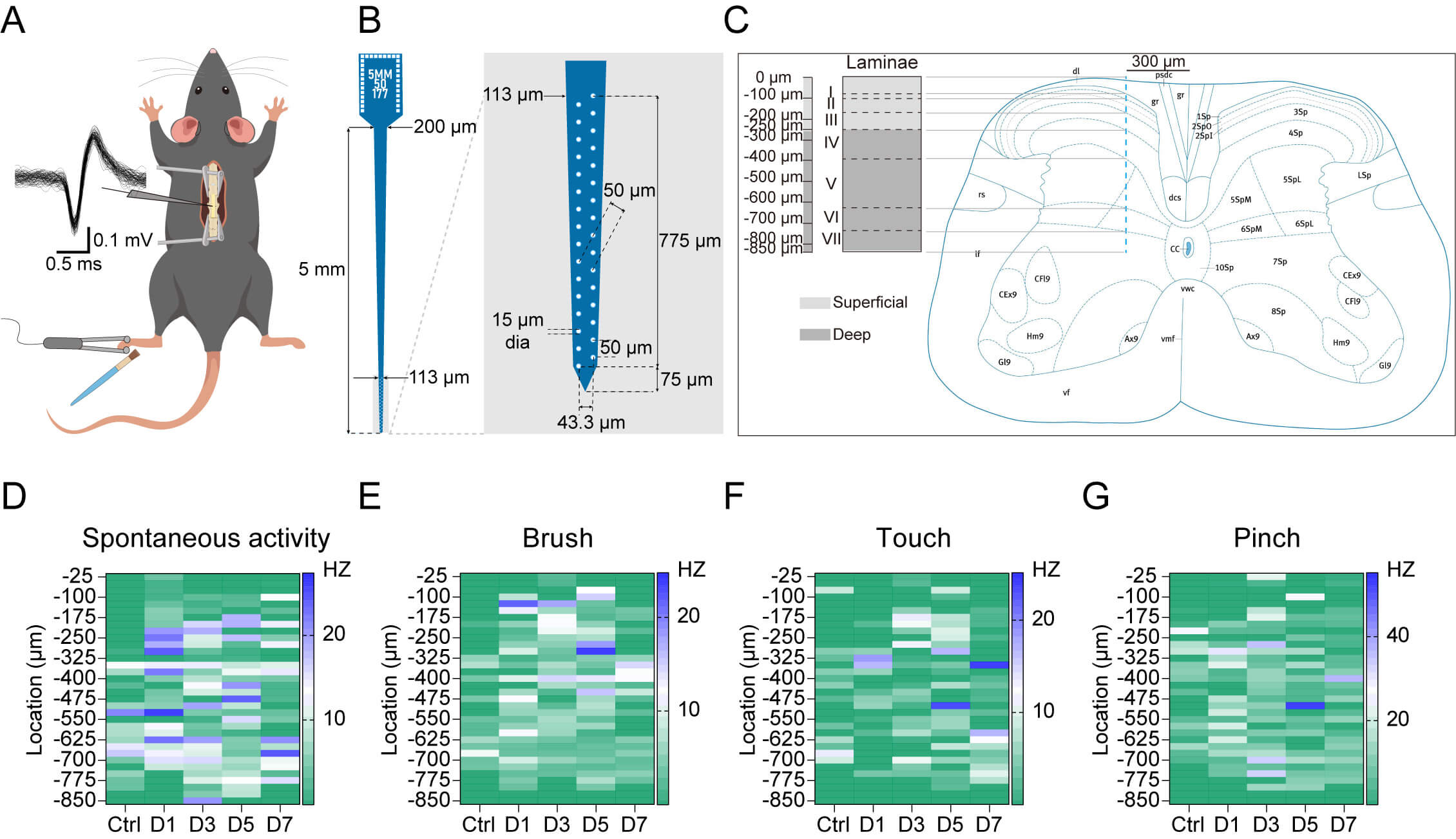 Fig. 3.
Fig. 3.
Temporal changes in spontaneous and evoked neuronal activity across different depths of the SDH after CFA injection. (A) Experimental setup for in vivo SDH neuronal recordings. A 32-channel MEA was used to record action potentials from SDH neurons while applying mechanical stimuli (touch, brush and pinch) to the ipsilateral hindpaw receptive fields. (B) Configuration of the 32-channel microelectrode array showing the recording sites distributed across an 850 µm depth. (C) Predicted extent and borders of spinal laminae across the 850 µm depth for 300 µm lateral to the middle spinal artery. The SDH laminar organization were divided into superficial (0–250 µm) and deep (250–850 µm) regions. The annotated L4 spinal cord section was adapted from the Mouse Spinal Cord Atlas provided by the Allen Institute for Brain Science (https://mouse.brain-map.org/experiment/siv?id=100050402&imageId=101006501&imageType=atlas). (D–G) Depth-dependent neuronal activity represented as heatmaps and showing (D) spontaneous, (E) brush-evoked, (F) touch-evoked, and (G) pinch-evoked firing rate changes (Hz). Depth bin for each row was 25 µm. The color scale represents the firing frequency in Hz. Animal numbers: control: n = 6; day 1: n = 6; day 3: n = 6; day 5: n = 7; day 7: n = 6. Temporal profile of spontaneous and evoked neuronal activity in the superficial and deep dorsal horn following CFA injection. A detailed analysis of spontaneous and evoked neuronal activity across the superficial and deep SDH is presented in Supplementary Fig. 5 and Supplementary Table 3. Adobe Illustrator 2020 (version 24.1.3, Adobe Inc.) was used to create the illustrations in (A–C). MEA, microelectrode array.
Heatmap analysis showed laminar-specific differences in spontaneous and evoked neuronal activity between control and CFA-treated mice. In control animals, neurons in the superficial laminae exhibited minimal activity, whereas deep laminae displayed higher levels of spontaneous firing (Fig. 3D). Following CFA injection, the spontaneous excitabilities of neurons among all laminae increased rapidly on day 1, then gradually declined to normal levels (Fig. 3D). As for evoked activity, neurons across all laminae displayed limited responses to mechanical stimuli under physiological conditions. In mice injected with CFA, brush stimulus elicited the most pronounced neuronal activity in the superficial laminae, whereas touch and pinch stimuli induced comparable neuronal hyperactivity across all laminae (Fig. 3E–G).
Collectively, these electrophysiological recordings suggest that neurons across the dorsal horn in mice with inflammatory pain tend to exhibit an increase in spontaneous and evoked excitability. The temporal profile of these alterations correlates well with both spontaneous and evoked pain behaviors, providing a more comprehensive understanding of the neuronal basis of inflammatory pain processing.
To further elucidate the neuronal population underlying inflammatory pain, the proportion and distribution of SDH neurons were characterized across three functional subtypes: WDR, LTM, and NS neurons. Mechanical response was assessed using dynamic brush and static touch as innocuous stimulation, and a 150-g calibrated forceps pinch was used as noxious stimulation. Neurons responding to both stimuli were classified as WDR, while neurons responding exclusively to innocuous stimuli were LTM, and neurons responding exclusively to noxious stimuli were NS (Fig. 4A). Quantitative analysis revealed dynamic shifts in the composition of mechanically-responsive neuronal populations (Fig. 4B). Under physiological conditions, WDR neurons constituted the predominant population (44.44%) of recorded neurons, whereas LTM and NS neurons were present in comparable proportions (27.78%). On day 1 after CFA injection, the proportion of WDR neurons decreased markedly (36.99%) but returned to prominence in later observations. LTM and NS neurons exhibited divergent dynamics at day 1, with LTM peaking at 41.10%, whereas NS neurons declined to 21.92%. Both neuronal subtypes gradually returned to baseline levels thereafter.
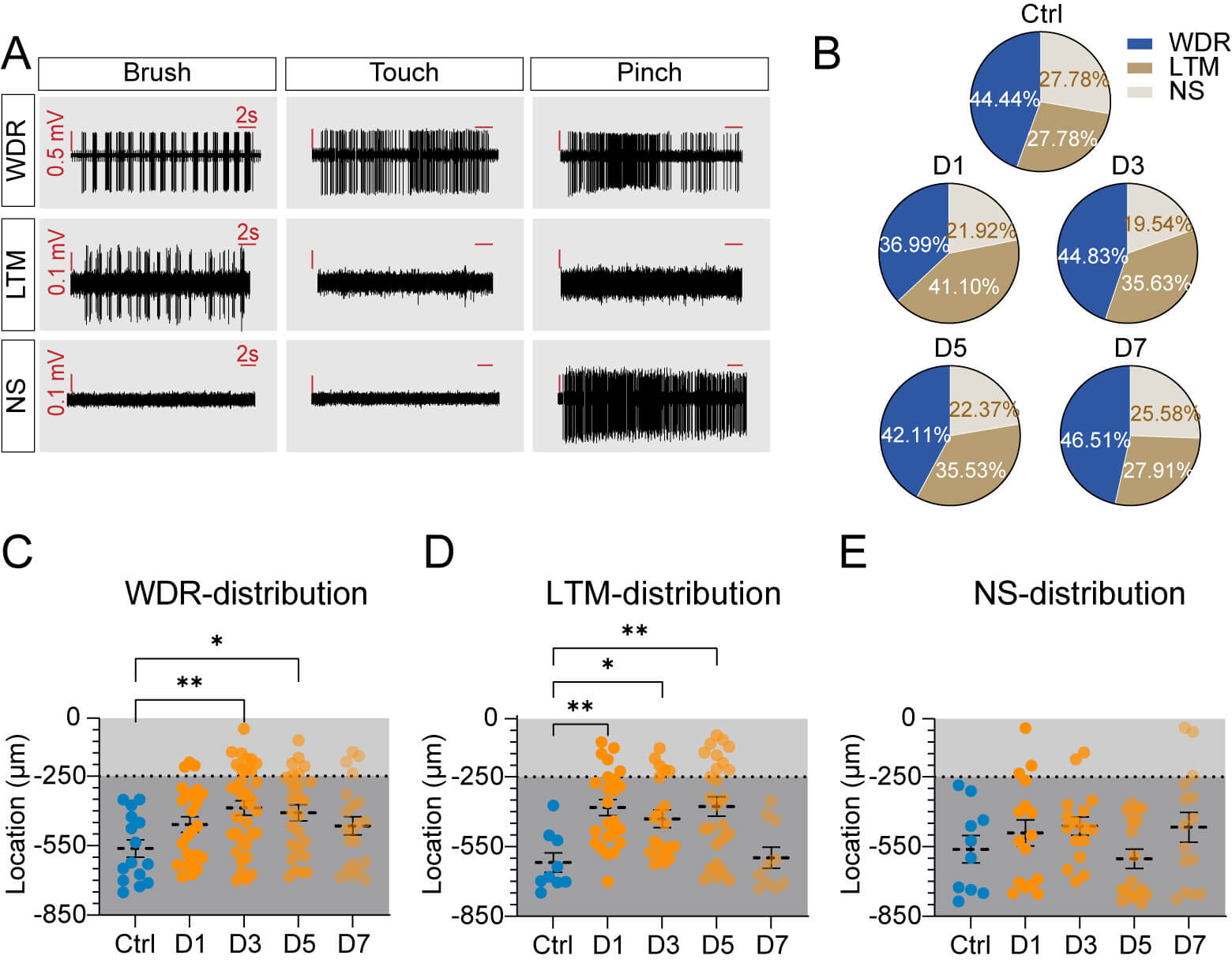 Fig. 4.
Fig. 4.
Temporal changes in neuronal subtypes and their spatial
distribution in SDH following CFA-induced inflammatory pain. (A) Representative
response patterns defining neuronal subtypes. WDR neurons show graded responses
to brush, touch, and pinch stimuli. LTM neurons respond only to brush
stimulation, while NS neurons respond exclusively to pinch stimulation. (B)
Proportion of neuronal subtypes (WDR, LTM, and NS) in control mice and
inflammatory pain mice at indicated time points. Values are shown as the
percentages of total recorded neurons. (C–E) Spatial distribution analysis of
recorded neurons across dorsal horn depth: (C) WDR neurons (control, N = 16; day
1, N = 27; day 3, N = 40; day 5, N = 32; day 7, N = 27). (D) LTM neurons
(control, N = 10; day 1, N = 30; day 3, N = 28; day 5, N = 27; day 7, N = 12).
(E) NS neurons (control, N = 10; day 1, N = 16; day 3, N = 17; day 5, N = 17; day
7, N = 16). N represents neuron number (animal n values match Fig. 3). Scatter
plots display the depth distribution, where light, and dark gray regions
correspond to superficial and deep laminae, respectively. Values are presented as
the mean
Spatial distribution analysis revealed a shift in depth of the neuronal populations following inflammatory pain. WDR neurons, which normally predominate in deeper laminae, exhibited a significant shift toward more superficial locations on days 3 and 5 (Fig. 4C, Supplementary Table 4). LTM neurons were typically concentrated in deep laminae under normal conditions, but showed an enhanced superficial distribution on days 1, 3, and 5 post-CFA compared with controls (Fig. 4D, Supplementary Table 4). The distribution of NS neurons remained unchanged throughout the observation period (Fig. 4E, Supplementary Table 4). The concurrent superficial shift of both LTM and WDR neurons suggests enhanced nociceptive processing through increased integration of both noxious and innocuous inputs in pain-processing circuits. Overall, alterations in both the proportion and distribution of SDH neurons may represent a form of maladaptive plasticity that contributes to the heightened pain sensitivity observed in our behavioral studies.
After characterizing the changes in proportion and distribution of SDH neurons, we next assessed their mechanical excitabilities by quantifying evoked discharges in response to various stimuli (brush, touch, and pinch) following CFA injection. The discharge frequency of WDR neurons evoked by brush and pinch stimuli was significantly higher on days 1 and 3 post CFA injection compared with controls. Additionally, these neurons showed increased mean firing frequency to touch specifically on day 1 (Fig. 5A–C). Interestingly, despite their altered proportion and distribution patterns, LTM neurons in mice with inflammatory pain showed a slight increase in their brush-evoked excitabilities (Fig. 5D). NS neurons maintained relatively stable activities to pinch stimuli throughout the observation period (Fig. 5E). The differential evoked activities of WDR, LTM, and NS neurons suggests that WDR neurons serve as key cellular mediators for the manifestation of both mechanical allodynia and hyperalgesia in inflammation.
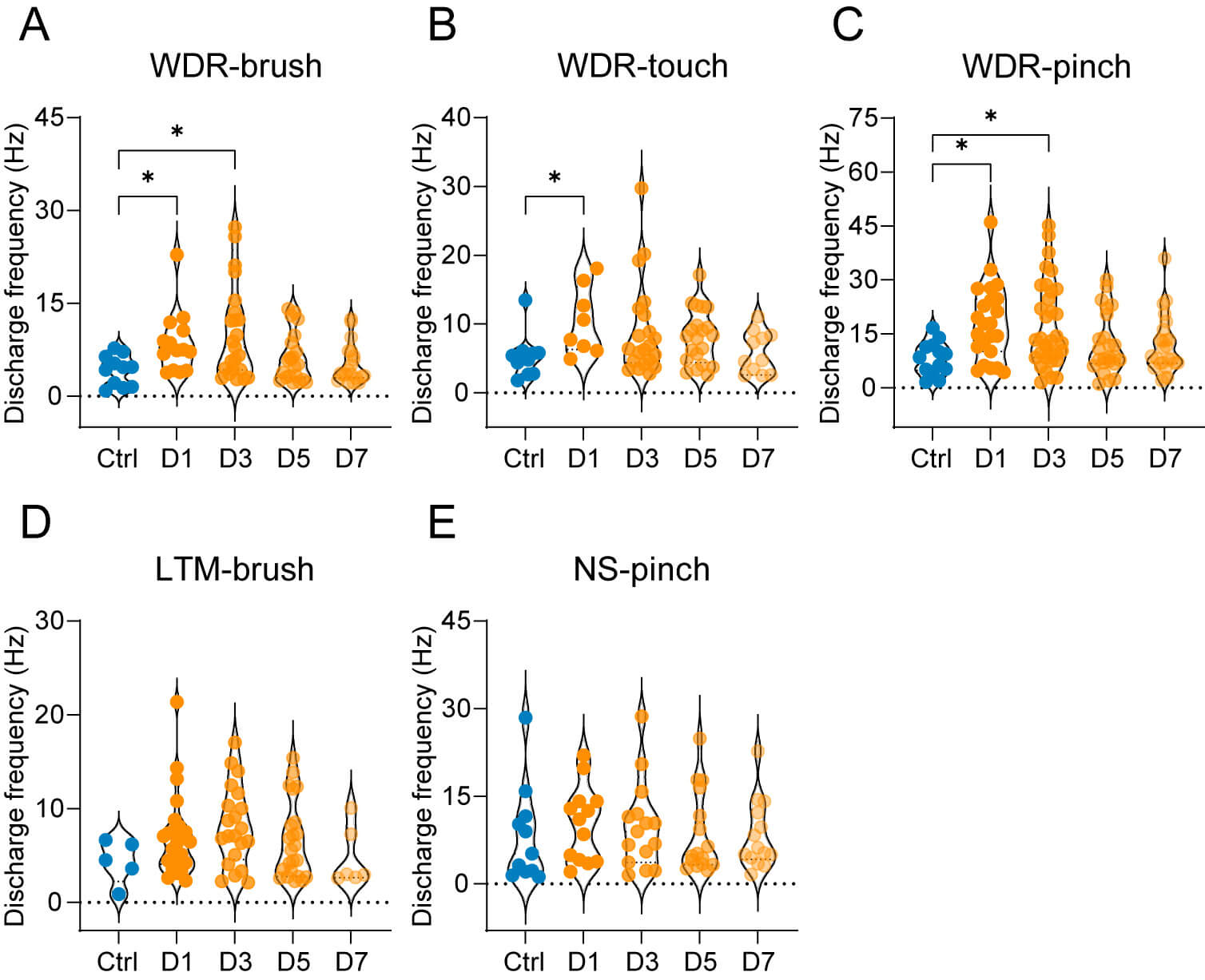 Fig. 5.
Fig. 5.
Temporal changes in stimulus-evoked responses of distinct
neuronal subtypes after CFA injection. (A–C) Analysis of WDR neuron firing
rates in response to (A) brush, (B) touch, and (C) pinch stimulation. (D)
Brush-evoked firing rates of LTM neurons. (E) Pinch-evoked firing rates of NS
neurons. Values are presented as individual data points on violin plots (N values
for neurons as shown in Fig. 4). Statistical significance was determined using
Kruskal-Wallis test followed by Dunn’s multiple comparisons. * p
In the present study we utilized immunofluorescence and extracellular recordings to systematically investigate changes in SDH neuronal excitability that underlie nociceptive behaviors in inflammatory pain. Our results demonstrate that spontaneous nociceptive behaviors in mice with CFA-induced inflammatory pain exhibit parallel temporal dynamics with SDH neuronal hyperactivity. Furthermore, pathological pain states induced significant shifts in the depth, proportion and stimulus-responsive profiles of SDH neuronal subpopulations. Moreover, WDR hypersensitivity and an increased proportion of LTM neurons were the most robust electrophysiological correlates of inflammatory pain development.
Objective assessment of nociceptive somatosensory responses in animal models requires reliable behavioral assays [21]. Von Frey testing is an established, standardized metric for evoked nociception in pain research, based on the consistently decreased withdrawal thresholds observed in rodents. Pain-affected animals frequently exhibit spontaneous behaviors including tactile hypersensitivity in the injured region and compensatory postural adjustments. The present study incorporated gait analysis and weight-bearing tests to evaluate spontaneous pain, alongside von Frey testing for mechanical sensitivity assessment. Consistent with a previous study [22], our results demonstrated transient gait alterations accompanied by progressive restoration of bilateral weight-bearing balance in mice with inflammatory pain, together with persistent hyper-responsiveness to mechanical stimuli. Unlike murine models of neuropathic and osteoarthritic pain which exhibit persistent gait abnormalities for several weeks [23, 24], our inflammatory pain model showed complete resolution within 5 days post CFA injection. This divergence may originate from the self-limiting nature of inflammatory processes, which differs fundamentally from the chronic and maladaptive neuroplasticity observed in neuropathic and osteoarthritic conditions. Therefore, timepoints and a diversity of behavior assessments are of great importance for the study of pain conditions in animals.
The SDH functions as the primary central sensory integration site, with discrete
laminae exhibiting specialized functional segregation [25]. Nociceptive
A
It should be noted that the subsequent reduction in superficial c-Fos+neurons does not preclude the persistence of nociception. Our electrophysiological recordings showed lasting hyperexcitability in both spontaneous and evoked neuronal responses across SDH laminae, indicating a spatial expansion of sensitization during chronic pain maintenance. Interestingly, we observed that dynamic brush stimulus induced a higher neuronal discharge frequency than static touch. This parallels the well-established behavioral dissociation between dynamic allodynia and static hyperalgesia in both clinical and preclinical pain models [38, 39, 40]. Genetic manipulation studies further demonstrate that distinct neural circuits mediate these differential sensory phenotypes [41, 42]. Our findings extend this dichotomy to the level of spinal neuronal response, suggesting that laminar-specific plasticity underlies modality-selective pain hypersensitivity.
In vivo electrophysiological recordings have primarily been performed in rats because of their technical accessibility [43, 44]. However, in a recent study we successfully implemented this methodology in murine SDH using MEA. Building upon a prior innovative framework [6, 45, 46], our high-density 32-channel MEA with 50-µm inter-electrode spacing enabled depth-resolved monitoring of neuronal population dynamics during CFA-induced inflammatory pain progression. Neurons were categorized into LTM, WDR, and NS, based on stimulus-response profiles, as previously described [47, 48]. Interestingly, all three neuronal populations exhibited significant plasticity, as evidenced by both their shifts in proportion and laminar distribution patterns. Peripheral inflammation in mice induced a particularly pronounced increase in the proportion of LTM neurons, along with a ventral-to-dorsal shift of all three neuronal types. This spatial reorganization aligns with previous reports of altered neuronal distribution in the SDH under pathological pain conditions [49]. The observed alteration may partly be attributed to impaired inhibitory transmission. Under physiological conditions, GABAergic and glycinergic neurons inhibit the activation of neurons in superficial laminae by large-diameter, non-nociceptive afferents [50, 51]. In inflammatory states, however, these inhibitory circuits become modified and lead to enhanced responses in superficial laminae to low-threshold peripheral inputs [52, 53, 54]. Such disinhibition represents a key mechanism underlying touch-evoked pain-like behaviors [35, 55, 56], consistent with the persistent mechanical allodynia observed in our von Frey tests.
While WDR neurons are considered predominantly located in the deep dorsal horn, numerous studies have confirmed their presence in laminae Ⅰ and Ⅱ [4, 57, 58]. Our data similarly demonstrated an appearance of WDR neurons in superficial laminae in inflammatory pain mice. Besides, a previous study of cancer pain models has reported a shift from NS to WDR predominance in superficial layers [59]. However, this specific transition was not observed in our inflammatory pain model. This discrepancy may stem from the less severe and distinct pathophysiology of inflammatory pain compared with neuropathic or cancer-related pain states. Furthermore, our electrophysiological recordings captured a greater number of active neurons in mice with inflammatory pain than in those under normal conditions. This likely results from inflammatory mediators sensitizing previously unresponsive sensory afferents and enhancing presynaptic glutamate signaling [60, 61]. The resultant increase in excitatory drive may recruit once-silent dorsal horn neurons, explaining the increased number of recorded units [62]. Therefore, the observed shifts in neuronal subtype proportions not only derive from neuronal functional modification in the dorsal horn, but also likely attribute to functional recruitment of previously silent neurons into the active network.
Spinal WDR neurons encode mechanical stimulus intensity through changes in
firing rate, which not only increase with escalating stimulus intensity but also
exhibit hyperactivity under various pathological pain conditions [43, 63].
Accordingly, the discharge frequency of WDR neurons serves as a valuable
indicator for assessing nociceptive transmission in the SDH. These neurons
integrate inputs from large-diameter primary afferents, A
Additionally, a previous study indicates that WDR neurons exhibit greater sensitivity to peripheral stimulation than LTM and NS neurons [67]. Our data similarly demonstrated that WDR neurons displayed heightened activity that coincided with spontaneous pain behaviors on the 1st and 3rd days after CFA injection, as evidenced by gait abnormalities. Notably, neither LTM nor NS subtypes showed similarly pronounced changes in evoked discharge frequency. This differential response may be attributed to injury-related sensitization specific to WDR neurons, a phenomenon not observed in NS subtypes [68]. However, this correlation between neuronal activity and pain behaviors derives from separate cohorts for terminal electrophysiology and behavioral testing. Future studies employing chronic recording techniques would be valuable to establish definitive causal links between individual neuronal response profiles and the manifestation of specific pain behaviors. Besides, methodological considerations regarding neuronal receptive fields should be noted. To ensure consistent laminar localization, the lateral recording position was fixed across experiments. Meanwhile, unlike single-unit recordings that identify receptive fields based on individual neuronal responses, our approach defined receptive fields as locations eliciting multi-unit activity. Given that WDR neurons possess broad receptive fields that expand further under pathological conditions [47], our recordings likely overrepresented WDR neuronal responses. This sampling bias should be considered when interpreting the observed discharge patterns.
There are several limitations to our study. First, while anesthesia allows stable acute recordings, it unavoidably masks the dynamics of chronic pain. Emerging technologies such as ultra-flexible electrode arrays enable longitudinal spinal monitoring in awake mice [69], although further validation of their signal stability and efficacy is needed. These advanced tools may provide new opportunities to investigate spinal sensory processing under natural pain conditions. Additionally, our classification of SDH neurons relied exclusively on mechano-sensitivity without specific neurochemical markers, reflecting a fundamental constraint of current in vivo electrophysiological approaches. Future research should focus on developing multimodal methodologies that combine morphological characterization with in vivo electrophysiology, thereby enabling more comprehensive functional analysis of neuronal populations.
The spontaneous pain behaviors observed in mice with inflammatory pain exhibited parallel dynamics with SDH neuronal hyperactivity. Following CFA-induced inflammation, SDH neuronal subpopulations underwent significant shifts in depth and proportion, and exhibited altered stimulus-responsive profiles. Among these alterations, the sensitization of WDR neurons and expansion of the LTM population served as dominant mechanisms of inflammatory pain pathogenesis.
The data that support the findings of this study are available from the corresponding author upon reasonable request.
HZ: Investigation, Methodology, Formal analysis, Writing original draft. XS: Investigation, Methodology, Writing review & editing. QY: Investigation, Methodology, Writing review & editing. XW: Methodology, Resources, Validation, Writing review & editing. YS: Methodology, Resources. Validation, Writing review & editing. XJ: Conceptualization, Resources, Validation, Writing review & editing, Supervision. ZZ: Conceptualization, Methodology, Formal analysis, Writing review & editing, Supervision, Funding acquisition. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
All experimental procedures were approved by the Institutional Animal Care and Use Committee of the Institute of Acupuncture and Moxibustion, China Academy of Chinese Medical Sciences (approval no. D2024-01-26-09) and conducted in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
We gratefully acknowledge the technical advice from Yani Li, Liaoyuxian Deng, and Junkang Chen.
This work was supported by National Natural Science Foundation of China [grant number 82204796], and the Fundamental Research Funds for the China Academy of Chinese Medical Sciences [grant number ZZ18-YQ-045].
The authors declare no conflict of interest.
During the preparation of this work the authors used Claude in order to check spell and grammar. After using this tool, the authors reviewed and edited the content as needed and takes full responsibility for the content of the publication.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/JIN46163.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





