1 Department of Physical Therapy, Movement and Cognition Laboratory, University of Haifa, 3498838 Haifa, Israel
2 Department of Neuroscience, Carleton University, Ottawa, ON K1S 5B6, Canada
3 Research Institute, Children’s Hospital of Eastern Ontario, Ottawa, ON K1H 8L1, Canada
Abstract
The early years of life show remarkable brain development and cognitive growth. During this time, the foundations for learning and memory are established, driven by the intricate interplay of various brain structures. Understanding the neuroanatomy of infant learning and retention is crucial in elucidating how these processes evolve and contribute to lifelong cognitive capabilities. Herein, we review the complex processes of brain development, learning and memory in the fetus, and during the first two years of life postpartum. Neural connections and key brain structures start to form during the fetal stage and continue after birth. We discuss how fetuses, infants, and toddlers absorb stimuli from their environment and develop learning and memory capabilities. We also provide an updated review of recent research findings in the field, presenting the latest insights into the development of learning and memory in the fetus and infants. In addition, we compare changes in learning and memory with electroencephalography findings from early childhood.
Keywords
- fetus brain development
- child development learning
- memory
- movement
- cognition
The journey of learning and memory begins before birth. During the fetal stage at around 24 weeks of gestational age (GA), the brain undergoes critical developmental processes that set the stage for learning after birth [1, 2, 3]. Brain structures such as the hippocampus and neocortex start to form during the early weeks of gestation, at around 18 weeks of GA, and play pivotal roles in learning and memory. The cortex, particularly the prefrontal region, is essential for higher cognitive functions such as attention, planning, and problem-solving. Its development commences prenatally and continues into infancy, early childhood and beyond, with the developmental trajectory reported by Leisman and colleagues [2]. Integration of the hippocampus and frontal lobes is vital for cognition, as this enables the encoding, consolidation, and retrieval of memories. It starts developing during the end of the first trimester of pregnancy and continues to mature postnatally [4]. In addition, the neocortex in general and the prefrontal cortex in particular are essential for performing the executive functions of attentional focus, motor planning, and problem-solving. The prefrontal cortex, as part of the broader neocortex, undergoes significant development during infancy. It plays a crucial role in executive functions, including attention regulation, working memory, planning, and decision-making. These functions allow infants to focus on relevant stimuli, ignore distractions, and anticipate outcomes based on experiences [4]. Importantly, the prefrontal cortex also contributes to encoding by enhancing selective attention. Sleep during infancy plays a crucial role in memory consolidation, as it facilitates neural reorganization and strengthens memory traces [5]. Additionally, the association cortices, especially in the temporal and parietal lobes, are involved in storing detailed experiential information, supporting long-term learning processes.
Fetal exposure to stimuli such as sound, movement, and light begins influencing neural connectivity at around 24 weeks GA, suggesting the emergence of early learning and memory capabilities [2]. For instance, fetuses can recognize and respond to familiar sounds, such as their parents’ voices, demonstrating nascent memory functions [4, 5, 6].
Birth represents a major transition in brain development. Transition from the intrauterine to the external environment triggers significant physiological and psychological changes that stimulate learning and memory. From the moment of birth, infants encounter a wide range of sensory inputs, such as sound, light, touch, and smell, which play critical roles in shaping the neural circuits involved in cognition [2]. Emotional responses at birth, such as crying, may serve as mechanisms for environmental engagement and early learning [7]. During the first months of life, the brain undergoes rapid growth, particularly in the cerebral cortex and hippocampus, supporting the ability of the infant to process, retain, and learn from experiences. These processes rely on core mechanisms of encoding, consolidation, and storage [8].
Encoding is the means by which sensory input is transformed into a form that can be stored in the brain. It involves attention, perception, and the initial registration of information. The prefrontal cortex plays a critical role in encoding, as it helps neonates focus on relevant stimuli and ignore distractions.
Consolidation is the stabilization and strengthening of encoded information, making it resistant to forgetting. This process occurs primarily in the hippocampus, where memories are gradually integrated into long-term storage. During infancy, consolidation is influenced by sleep, as this is when a critical part of neural reorganization and memory reinforcement occurs [9].
Storage refers to long-term-memory maintenance in the brain. The association cortices, particularly in the temporal and parietal lobes, are involved in storing detailed information about experiences, including the what, where, when, and how of events.
Research indicates that infants may experience age-related differences in memory retention, with older infants generally demonstrating better recall and learning compared to younger ones. This variance is attributed to the maturation of neural structures, and to the efficiency of encoding and consolidation processes [8, 10].
The prefrontal cortex undergoes significant development during infancy and early childhood, and has multifaceted functions in learning and memory. It supports various executive functions that are crucial for effective learning, including attention regulation, where the prefrontal cortex helps infants to focus on pertinent stimuli and filter out irrelevant information. Working memory facilitates the transient retention and manipulation of information, allowing infants to hold and use information over short periods. As the prefrontal cortex matures, planning and decision-making become possible, and infants become better at anticipating outcomes and making simple decisions based on their experiences. Although the prefrontal cortex is not fully developed in infancy, its ongoing maturation supports increasingly sophisticated learning and memory capabilities [11].
Neuroplasticity is a defining feature of early childhood and denotes the brain’s ability to restructure itself by forming new neural connections. This characteristic allows infants to adapt to their environments and learn from experiences. Synaptogenesis, which is the formation of new synapses, is most prolific during infancy. As infants grow, the brain prunes excess synapses, enhancing the efficiency of neural networks and supporting learning and memory [12]. During the first two years of life, structures such as the cortex, hippocampus, and prefrontal cortex undergo significant development, laying the foundations for learning and memory and providing important insights into information acquisition and cognitive development throughout life [13].
Learning and memory are interconnected, and learning assessments are fundamentally tests of memory. Learning is defined as a generally enduring alteration in behavior that arises from experience, omitting transient changes like fatigue or medication [14]. Memory involves the encoding, storage, and retrieval of experiences. Effective learning requires linking representations of different experiences, which occurs in short-term memory. When the memory is active, it is susceptible to change, but once it transitions to long-term memory, it becomes relatively stable. Previous memories can be accessed and modified, remaining consistent throughout development, although their aspects and substance may vary [15].
The interface between the brain and experience during the first two years of life is critical for developmental and cognitive milestones. During the first two years postpartum, the brain is particularly plastic and responsive to environmental stimuli. The initial interactions a child has with their surroundings play a pivotal role in shaping neural pathways and cognitive functions. Sensory inputs from the environment—such as visual, auditory, and tactile experiences—are integral to the brain’s developmental process. These inputs help to form synaptic connections that are foundational for learning and memory [16].
As infants interact with their caregivers and explore their environment, they begin to develop essential skills such as language acquisition, motor coordination, and social bonding. Studies have shown that the quality and variety of these early experiences can significantly influence cognitive development and brain structure. For example, enriched environments with diverse stimuli are associated with enhanced synaptic growth and neural complexity. The nature of this process is described in more detail below [16, 17].
Furthermore, the use of electroencephalography (EEG) has provided valuable insights into how experience influences brain activity. EEG studies have shown that neural oscillatory patterns change in response to different stimuli, indicating the brain is actively processing and adapting to new information. These changes in brain activity are linked to improvements in cognitive functions such as attention, problem-solving, and memory [18]. Crucially, the dynamic interplay between genetic and environmental factors underscores the importance of early experiences in determining developmental outcomes [19].
Neuroplasticity also refers to the capacity of the central nervous system (CNS) to restructure itself as a result of genetically predetermined limitations and experiential influences [19]. In adulthood, the brain has significant potential to reorganize its neuronal structure in response to specific demands and incoming stimuli. From a developmental standpoint, this is significant because accumulating evidence indicates the cortex can alter its anatomical and functional architecture in response to experience at both the macroscopic and microscopic scale [13, 20].
On a macroscopic scale, neural reshaping at an early age consists of dramatic pathway redeployments that rely on changes in cortical connectivity [21, 22, 23]. On a microscopic scale, the plasticity of neural networks involves structural changes that rely on synaptic efficacy, synapse formation, spine density, and dendritic formation. This growth occurs as a result of experience-driven synaptic remodeling activity following the activation of new gene transcription [24, 25, 26]. The two levels of study offer insight into the interaction between our individual everyday experiences and our brain, resulting in a physiologically and intellectually unique organism. Thus, the generation of human behavior is highly intricate and fluctuates between genetic predispositions and experiential influences.
At the onset of the third millennium, it is widely acknowledged that the cognitive capacities of infants and adults arise from intricate interactions between hereditary factors and the environment. However, this interplay remains poorly understood and is especially nuanced during the initial stages of life when both intrauterine and postnatal brain development contribute to the formation of human behavior. Although considerable effort in developmental cognitive neuroscience research has been directed at this issue, there are still several major methodological and theoretical challenges. Significant knowledge gaps remain concerning the ‘boundaries’ of environmental and genetic factors that influence the entire organism, particularly with regard to the nature of these interactions. There is also significant interest in elucidating the extent to which physiological and genetic factors, as opposed to environmental experiences, influence cognitive development during early stages of life [27].
The interaction between genetic predisposition and environmental conditions plays a central role in shaping cognitive development. Polygenic scores, which summarize the cumulative effect of genetic variants associated with cognitive traits, are increasingly used to explore these interactions [28]. Environmental factors—such as home quality, neighborhood characteristics, and parental involvement—have been estimated to explain approximately 20.6% of the variance in cognitive outcomes [28].
The detection of robust gene-environment interactions remains statistically challenging. Many studies lack sufficient power, making it difficult to identify replicable effects and highlighting the need for larger, more diverse cohorts [28]. Additionally, recent findings suggest the impact of genetics on cognition tends to be consistent across various contexts, and that exposure to cognitive challenges may play a more influential role than mere maturation in driving development [29].
While the interplay of genes and environment is critical for cognitive development, some researchers argue that the focus on interactions may overlook the importance of direct effects, as well as the cumulative impact of environmental factors alone. This perspective highlights the need for a balanced understanding of both genetic and environmental contributions to cognitive outcomes.
A natural example of the interaction between these two aspects is provided by the birth of a preterm infant, which is characterized by abrupt interruption of the biological processes involved in the natural intrauterine maturation of the CNS. Although much evidence has been reported in the literature over the past decade suggesting that cognitive structures are already present at birth, little is known about the neural correlates that underlie them or their development. Work in the field of cognitive developmental neurosciences is now attempting to shed light in this area. This has theoretical interest as well as clinical relevance in terms of cognitive problems associated with preterm birth. The development of tools that allow functioning of the brain to be studied in its spatio-temporal dynamics has led to new insights in this field. Nevertheless, we are still at the dawn of developmental cognitive neurosciences, and much remains to be learned, especially during the early period of life.
Recent studies have begun to identify some of the neural correlates of cognition at birth [29, 30, 31]. These investigations employed advanced neuroimaging techniques to observe brain activity in newborns, providing insights into the functioning of their nascent cognitive structures. Evidence suggests that even at an early age, the brain exhibits activity patterns indicative of rudimentary cognitive processing abilities. This neural activity is believed to underpin the initial stages of learning and adaptation to the environment, laying the groundwork for the development of more complex cognitive functions later in life.
Researchers have focused on specific brain regions in order to understand their involvement in early cognitive tasks. For example, activity in the prefrontal cortex has been linked to early forms of attention and memory, while the auditory cortex shows responsiveness to sound stimuli, reflecting the newborn’s capability to process auditory information. These findings are crucial not only from a theoretical standpoint, but also for clinical applications, particularly in assessing and managing cognitive issues in preterm infants [32].
Learning and memory networks are considered particularly well-suited for investigating brain–environment interactions in infancy for several key reasons. During this period of rapid brain development, these networks are central to acquiring new skills, forming memories, and adapting to external stimuli. Their high degree of plasticity in infancy allows for dynamic reorganization in response to experience, making them an ideal model to study how early interactions shape the brain. These circuits also integrate input from multiple sensory and motor systems, providing a comprehensive view of how infants perceive and engage with their surroundings. Studying the networks reveals not only the mechanisms of learning and memory, but also the role of embodied experiences in cognitive development. Furthermore, early experiences within these networks have lasting effects on behavior and cognition, underlining the importance of this developmental window. Additionally, learning and memory networks are closely linked to observable behaviors in infants, such as imitation, exploration, and problem solving. Studying the networks can therefore provide valuable information about the relationship between brain activity and behavior.
Infants have a complex cognitive and behavioral repertoire indicative of a facility for conscious reflection. Demonstrating cognitive function in infancy is challenging, and even more so within the fetus. Infants cannot report their thoughts and behavioral measurements in the fetal environment, making study difficult. Effective cognitive function requires effective connectivity that can optimize network operations. The physical basis of network function in the brain is the thalamocortical pathway. These sophisticated networks commence development between weeks 24–28 of gestation [33]. Approximately two months later, EEG rhythms between both cortical hemispheres become synchronized, signifying the commencement of generalized integration of nervous system function [34, 35]. As a result, the basis of awareness is developed by the start of the third trimester of pregnancy, as the requisite pathways are already in place, developed, and functioning. Because it is easier to observe and study the functioning of a preterm neonate as compared to a fetus of the same GA in utero, the fetus is often considered, incorrectly, to function in ways similar to preterm neonates. However, this disregards the unique uterine environment of the fetus in a warm cavern, where it is coupled to the placenta which provides blood, nutrients, and hormones to its brain and developing body.
At conception, a zygote commences its development as a cell with a diameter of approximately 100 microns. By the end of a normal pregnancy, the neonate brain has reached approximately 30% of adult brain weight, and by the age of two years, approximately 85%. Frontal lobe development continues from adolescence into the third decade of life, albeit with individual differences [36].
Anatomic investigation shows progressive brain growth in the uterine environment, which continues during the first two years postpartum. By the time a child is around six years of age, its brain weight has achieved almost 90% of adult cranial capacity [37, 38] (see Table 1, Ref. [39]). Although maturation of the nervous system occurs relatively quickly (Fig. 1), specific brain regions require a longer time to mature (Fig. 2). This is especially the case for the prefrontal and frontal neocortical regions, allowing the neonate, infant and child to be more susceptible to extrinsic manipulation and experience. The extended time-course for frontal lobe development commences during fetal development and continues into young adulthood. This development can be affected by numerous experiences, both positive and negative, that may comprise short-term memory effects [40], strategy and planning development [41], associative learning [42], response inhibition or facilitation [43], and effects on emotional function and social behavior. Any event or function that affects the development of the frontal lobes can also affect changes in these functions. Much of what is commonly referred to as temperament, which may relate to personality, has an influence on social behavior, which in turn has a basis in cerebral lateralization and asymmetry.
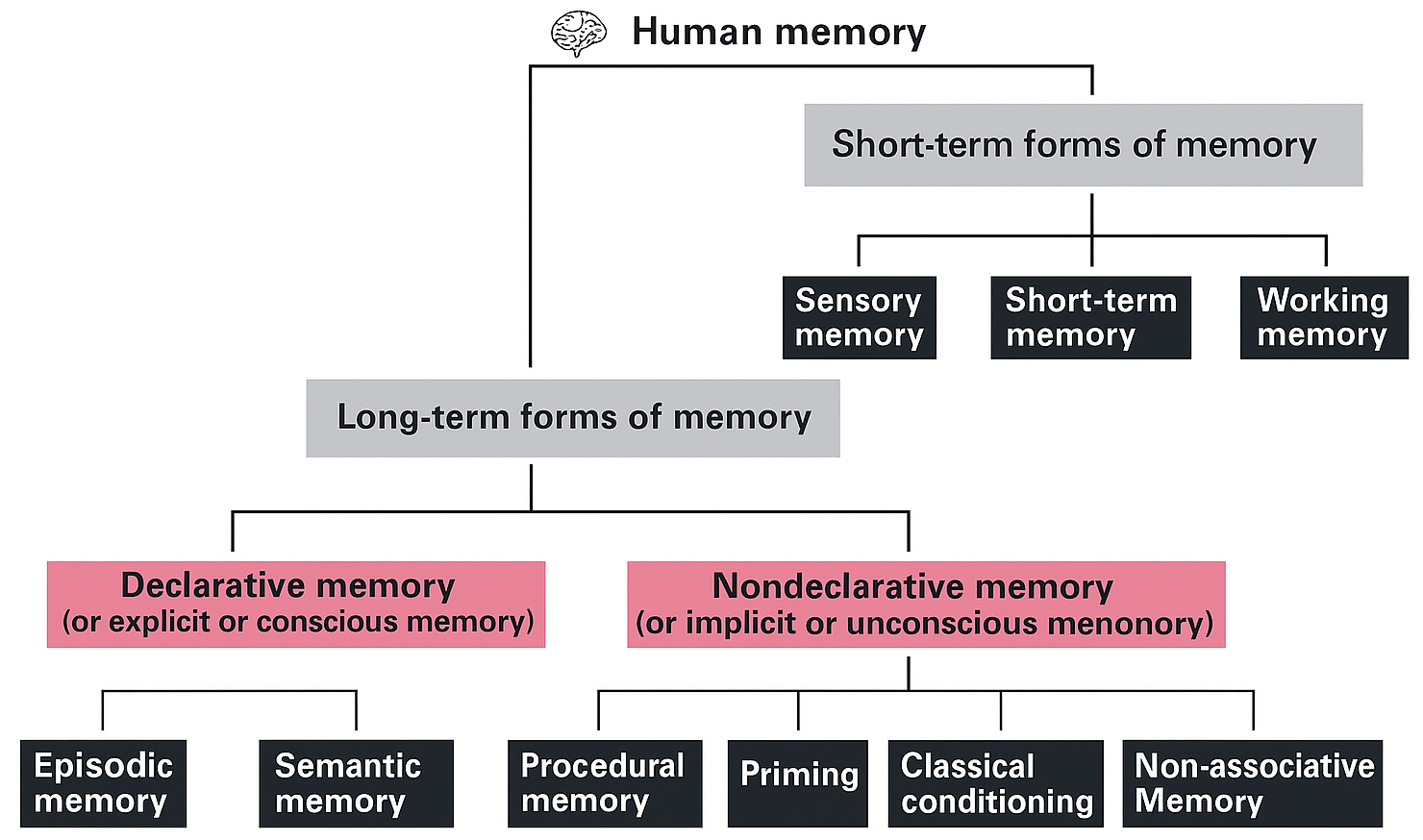 Fig. 1.
Fig. 1.
Development of the human brain, from conception through to 9 months GA. Development of the frontal lobes does not occur fully until an individual reaches their early 20s. GA, gestational age.
 Fig. 2.
Fig. 2.
The rate of brain maturity is variable, with some regions maturing more slowly than others. The more slowly developing areas are then more susceptible to change as a result of experience.
| Age | Brain weight: male (g) | Brain weight: female (g) |
| Newborn | 380 | 360 |
| 1 year | 970 | 940 |
| 2 years | 1120 | 1040 |
| 3 years | 1270 | 1090 |
| 10–12 years | 1440 | 1260 |
| 19–21 years | 1450 | 1310 |
| 56–60 years | 1370 | 1250 |
| 81–85 years | 1310 | 1170 |
Supporting the development of these functions, the basic structures of the cerebral hemispheres and diencephalic region are developed after the 8 weeks GA, while the brainstem has developed by 7 weeks GA [44]. Cell proliferation during neurogenesis is known to occur at the rate of approximately 250,000 cells/min by 7 weeks GA [45]. Early in the development of the fetus, the proliferating cells migrate and segregate throughout the neocortical layers of the fetus, peaking between approximately 12–20 weeks GA. Under normal circumstances, this process concludes between 26–29 weeks GA [46]. The six-layer laminar distribution of thalamocortical axons is developed by 32 weeks GA [47]. At approximately 20 weeks GA, synaptogenesis commences and continues postnatally and through adolescence [48, 49]. In addition, myelination supports higher-order cognition [50]. The corpus callosum is also important for cognition and is already functioning well by approximately 20 weeks GA [51]. Sensory, cognitive, motor, and emotional functions are combined through the corpus callosum. However, it is during prenatal fetal development that stimulation, both internal and external to the fetus and including both sensory and motor experiences, can modify cortical structure and function.
Jerison recently theorized on how higher mental functions arose in the excess neural tissue, referred to as the proper mass principle [50]. According to this principle, the amount of neural tissue devoted to a specific role should be related to the degree of information processing required by that function. As it grows, the brain organizes itself according to this principle, and organisms develop greater volume representations within the brain depending upon the use of an appendage. For example, a raccoon would have a greater volume of cells dedicated to the forelimb, as it has significant and unique forelimb manipulative abilities in comparison to canines that do not possess these skills. The proper mass principle is also relevant to neural network organization, supporting cognitive function. As the flexibility of response to social information requires significantly increased neural processing, increased brain size has evolved to address that need [49].
In both monkeys and humans, the phase of fetal brain growth (neurogenesis) begins about 40 days after conception and lasts for about 100 days in monkeys and 125 days in humans [51]. Neurogenesis occurs deep within the brain, and the neurons assume specific positions in the neocortex by migrating to locations that are specified by genes. Through this migration, the neurons build the six layers that make up the neocortex, starting with the innermost layer and ending with the outermost layer. The human neocortex is identifiable about two months after conception, and cell migration finishes by the end of the 5th month [39].
Another aspect of maturation is myelination, a process in which fatty sheaths enclose neurons, thereby insulating them and improving their ability to conduct electrical signals. To some degree, the connections that neurons make with one another are genetically programmed. However, the genetic controls are imperfect, and feedback from the body and its sensations influences both the production and elimination of specific connections [39]. Cells that form synaptic connections between neurons receive more nutrition and stimulation than those that do not, and those whose synapses fire off the most frequent messages are particularly well supplied. This process is called synaptic stabilization. Much of this natural selection at the cellular level occurs prenatally, but the process continues well into the postnatal period [52].
Predictably, the maturation of brain tissue parallels the maturation of brain functions. Development of the body and brain has both feedforward and feedback mechanisms. The maturation of a specific brain sector activates comparable bodily functions or related brain regions. The activated function subsequently develops more swiftly through utilization, which in turn promotes growth of the corresponding brain region that governs it. This relationship also molds the developing brain, favoring the fixation of beneficial neural maps and allowing the pruning of useless neural connections. Consequently, although genes specify some traits of the developing brain, neuronal maps in particular are created through environmental interaction, especially during advanced stages of development.
Primates have a lower rate of fetal brain growth than humans. In humans, growth continues throughout the first year of life postpartum [53]. A one-year-old human infant has a large head encasing a brain that is more than double the size of that in an adult chimpanzee [53].
Evolution has allowed consistent development of neuroplasticity for the individual in the short-term, and for the species in the long-term. The homo sapien brain shows unique evolution as a consequence of specific stimulation that serves the behavioral needs of the species. This uniqueness is manifested as bipedalism, as well as a larger cortex than all other species, including hominids. Neural circuitry has been refined in behaviors that control social, emotional, sensory, and cognitive functions [54, 55]. While postnatal development and maturation follow an orderly path with the achievement of developmental milestones, successful neurological maturation of the infant and child is highly contingent on fetal developmental processes that allow for reception, translation, and the ability to act on external stimulation and information [56].
During fetal development, a “blueprint” can serve as a description of a rough framework from which more defined structures and functions will evolve. Alternatively, the operating system develops during normal fetal development, and experience can modify that system in the postnatal world.
Vertebrate species develop their neural architecture early in fetal development, occurring at approximately two months GA in the human fetus [57]. Gene networks drive organizing codes in the development of fetal neocortical structures, with the foundations for different functional areas arising during the first six months of GA [58], as represented in Fig. 3 (Ref. [59]).
 Fig. 3.
Fig. 3.
Development of the human brain from conception through 9 months GA. Full development of the frontal lobes has occurred by the time an individual reaches their early 20s [Konkel [59], 2018, with permission].
The wiring plan controlling axon guidance connectivities during fetal development is not unconditional. Rakic [60] noted that the fetal neocortex mostly develops during gestation, reflecting evolutionary development of the human brain. Neuronal production supporting brain development occurs in an “inside-out” fashion.
Functional cellular connections that form functional control regions and assemblies are supported by the “inside-out” pattern of cell growth [61]. The basis of functional connectivities in the developing fetal nervous system allows the development of cell assemblies through neuron production and migration. This aspect of fetal neuronal development forms the basis for the development of cell assemblies through modifications resulting from learning and life experience.
Modifying the expression of a single genetic transcription factor in genetically manipulated mice can alter the functional connectivities of regions of the cortex [62]. For example, Grove and Fukuchi-Shimogori [63] reported the emx2 transcription factor can influence the expression of Fgf8 close to the anterior cerebrum. Emx2 alone was sufficient to determine which cortical areas received connections, particularly in the somatosensory and frontal regions. This subject will not be pursued further here, other than to state that genetic influences on the development of the fetal cortex provide chemical clues for axonal growth that further promote synaptic growth and configuration [62, 63].
While genetic control may be considered unalterable, genetic processes can clearly be influenced by genetic mutation, as well as environmental or induced effects on regulatory genes, such as inflammation, stressors, toxicities, chemical, alcohol, etc. [64]. In mammals, anatomical structuring of the brain is succeeded by long periods of synaptogenesis, growth, and pruning that commences during fetal development and do not finish until early neurological adulthood [65].
Critical primary environmental interactions need to occur for cognitive and behavioral function to progress normally, and so that development of the fetal nervous system and neural network organization fundamental to these behaviors can progress on a normal trajectory, supported by neuroplasticity [66]. Certain neurological processes are responsive to environmental inputs only for a specified window of time. If the stimulation does not transpire or is aberrant, the “sensitive period” passes, and growth of the nervous system will not follow its normal trajectory [67].
Greenough and associates termed this aspect of neurodevelopment “experience-expectant” [68]. In this process, synaptic connectivities form after some nominal experience that occurs commonly in most species. This optimizes the genome’s function and allows it to avoid organizing and controlling all facets of development in the members of a species.
The frontal lobes and hemisphere asymmetries directly address the processes of change in early infant development. Markham and Greenough [68] have proposed that the foundational structure of expectation consists of a transient overproduction of synapses distributed across an extensive area during early development. This was followed by subsequent pruning of synapses that either failed to establish connections, or formed aberrant connections. Fig. 4 (Ref. [69]) below illustrates the nature of neural connectivities.
 Fig. 4.
Fig. 4.
Illustration of dendritic connectivities at (A) 5 days, (B) 6 years of age (corresponding to an era of exuberant connectivities), and (C) adulthood. The complexity of dendritic structures of cortical neurons is consistent with the expansion of synaptic connectivities in middle childhood, and is pared down in adulthood [69].
The anticipated experience generates patterns of neural activity that focus on the synapses designated for preservation. Synaptic connectivities are presumed to be initially temporary, and need some form of validation for their continued existence. If confirmation is not received, synaptic connections will be retracted according to a developmental timeline, or due to competition from established synaptic connections [70].
Experience-expectant neurogenesis differs markedly from other forms of plasticity, which Greenough designates as experience-dependent. This process enhances an individual’s adaptation to distinctive characteristics of the environment, such as learning [69]. Consequently, diverse forms of information will be obtained and retained for future utilization, facilitating the development of individual variances across multiple cognitive domains, and encompassing emotionality and temperament [71]. The fundamental difference between experience-expectant and experience-dependent development is that the former is universally present and manifests similarly across nearly all individuals of the species, whereas the latter pertains to specific individuals and is evident in typical emotional development during fetal stages and infancy.
There is compelling evidence that proper neuronal pruning is regulated by synaptic activity. Cellular function that could regulate synaptic remodeling had been studied by Paolicelli et al. [71], who observed upregulation of the signaling molecule fractalkine during synaptic development. The microglia receive a signal from fractalkine to aggressively prune synapses. In healthy mice, left brain circuits do not develop normally and remain immature into adulthood when fractalkine communication between neurons and microglia is disrupted. This suggests that microglia-mediated synaptic pruning is an essential and crucial process in the development of circuitry [71, 72]. Stevens et al. [73] discovered that immune system-related “complement proteins”, which are essential for retinal axon pruning, have a similar role.
Complement proteins from the immune system are thought to inform microglia regarding what to engulf and extract. Stevens et al. [73] hypothesized this might be linked to the emergent function of microglia in pruning and synaptic remodeling. In support of this, they found that microglia in the visual system of newborn mice can phagocytize synapses in the lateral geniculate nucleus (LGN) through mechanisms involving both complement proteins and neuronal activity. Analogous to the findings by Gross on fractalkine, inhibition of complement signals disrupted the formation of visual circuits [73], leading them to propose that complement proteins may label low-activity synapses for elimination by microglia.
Dance [74] used time-lapse imaging to study the process of trogocytosis, which is used by immune cells to kill pathogens. They observed microglia “nibbling” on presynaptic assemblies of live neurons in culture, even though there is no convincing evidence that microglia can prune or eliminate full synapses.
During human fetal brain development, little synaptic growth occurs before the start of the third trimester of pregnancy. Synaptic development then accelerates to approximately 40,000 synapses per minute, which continues until around the second year of life [75]. This dramatic increase in synaptic formation is followed by an asymptote during which no significant change occurs in the number of synapses. However, significant restructuring of the synaptic architecture takes place, including synaptic type and location, as well as the relationship between excitatory and inhibitory synapses. Synaptic pruning of extra connections begins during early childhood and continues through adolescence, with the process completed by early adulthood [76, 77].
In the same way that the genetic contribution of a fetus, infant, and child continuously interacts with experience to regulate the course of development [78], diverse experiences can interrelate with the individual’s genetics to refashion the path of development. In theory, the timing, perturbation, type of cell population affected, and mechanism of molecular signaling could all influence and change developmental trajectories.
The human brain is complex and built to perform cognitive tasks including attention, perception, action, movement planning, learning, and memory. All of the nervous system is engaged during prenatal development, and we describe here the progression of fetal cognitive development. The fetus begins to process sensory information, starting cortically at around 25 weeks GA. At 34 weeks GA, the fetus can perceive complicated acoustic stimuli and differentiate between various auditory inputs. Fetal movement and action planning are established by 22 weeks GA, and research utilizing four-dimensional ultrasound has shown that the complexity of fetal motor behaviors and action accelerates as pregnancy advances [42]. The fetus possesses an extraordinary aptitude for learning and memory.
Neonates already possess well-developed subcortical structures, with high activity in primary cortical regions and low activity in neocortical association areas. More data on fetal cognitive function is needed to support better neurodevelopmental outcomes for high-risk pregnancies and prematurely born neonates [42].
After conception, brain cells do not emerge in the human embryo until approximately three weeks GA [52, 75]. Studies indicate the cognitive and sensory processing brain regions do not become effective until 28 weeks GA, even though the initial development of these processes starts earlier. Fetal synaptic connectivity develops progressively throughout gestation, laying the foundation for postnatal neural function and cognitive processing [79]. All of the nervous system is functional during prenatal development. From approximately 25 weeks GA, the cortical level of the fetus processes sensory stimuli, including pain [80]. At 34 weeks GA, the fetus can perceive externally generated complex acoustical signals and sounds, and can also discriminate between them [2, 79, 80]. Action planning by the fetus occurs at approximately 22 weeks GA [81]. The increase in complicated fetal motor action and behavior with the advancement of pregnancy can be observed using four-dimensional ultrasound [82]. The fetus has significant capacity for learning and memory. The subcortical configurations of the brain in the neonate are already well developed at birth, with significant activity in primary cortical regions, but less activity in association areas [43, 83]. Brain development of the fetus includes cognitive function, which does not start instantly at birth but rather progresses gradually through pregnancy and continues postpartum in the neonate and infant.
O’Rahilly and Müller [84] studied the structure and function of the brainstem in the fetus at 8 weeks GA. At this age the 3.75 mm embryo already has a face, hands, and feet, resembling a neonate. Brainstem growth is likely responsible for the majority of movement by the embryo, known to start at approximately 6 weeks GA, and demonstrating bodily pulsations that may be considered a startle reflex [84, 85]. The embryo, however, does not possess a neocortex supporting intentional movement. At this point, an embryo that demonstrates a startle-like movement response in the absence of a developed neocortex cannot be considered human, since it is the neocortical region of the embryo and ultimately the fetus and neonate that enables awareness and cognitive function. The embryo cannot, at this point in development, be aware and selectively respond to environmental stimulation.
Neocortical cells first arise at approximately 4 weeks GA, but as shown in Fig. 1, most of the neocortex does not form until the fetus is almost 6 months GA [46, 86]. Interconnecting neuronal fibers are required for the neocortex to function effectively. Effective cognition is determined by the optimized functional connectivities between synapses [87]. Isolated connections between neocortical neurons and the neuromuscular system can be detected in a fetus as young as 15 weeks, and part of the cortex controlling fetal limb movement has functionally matured by 22 weeks GA [88]. The fetus is unlikely to be capable of pre-planned movement until at least 28 weeks GA, since the cognitive apparatus for this has not yet developed. Tactile stimulation of a fetal limb will result in a motor response that is a reflexive action, without a cognitive component controlling that action.
Fetuses develop physical adaptations for postnatal functioning and begin to sense their environment early in gestation, laying the groundwork for early attachment to their mothers and the adjustment to life after birth [2, 89]. At approximately 28 weeks GA, cell connectivities in the neocortex are facilitated by interneurons, leading to complex functions that support cognition. This period is considered even more critical than birth. Fetuses at 28 weeks GA can see similarly to neonates [89]. Prematurity does not accelerate synaptogenesis in the visual neocortex, suggesting that birth is not essential for brain development [90].
EEG measurements show that the neocortex becomes active around 28 weeks GA, with fetal EEG patterns resembling those of neonates by 29 weeks [91]. However, the exact timing of when a fetus becomes aware and capable of intentional responses remains unclear. After recording EEG data from 30 fetuses, Eswaran et al. [92] found that 60% exhibited at least one recording with discontinuity, and various patterns were observed before and after 35 weeks GA.
Following an anatomical description of nervous system operations and discussion of the processes of sensation, perception, and learning, we proceed to discuss the role of cognitive components that are essential for effective sensation/perception.
Fetal sensation, perception, and cognition are crucial areas of study that provide insight into how fetuses interact with their environment and prepare for life after birth. During gestation, fetuses develop various sensory and cognitive abilities that enable them to process and respond to external and internal stimuli.
From around 20 weeks GA, fetuses respond to light, sound, taste, and touch, demonstrating their ability to detect and react to stimuli in a coordinated manner. Body awareness begins to develop after 25 weeks GA [2].
The neural pain system develops from nociceptors to sensory areas in the cerebral cortex, supporting pain perception. Somatosensory-evoked potentials recorded after 29 weeks GA indicate pain processing in the somatosensory cortex, and facial expressions of pain in preterm infants suggest they are conscious of pain. Fetuses also develop taste and smell sensations, reacting differently to pleasant and unpleasant tastes in amniotic fluid, and may also acquire food preferences during fetal life. These sensory experiences contribute to early attachment to the mother and learning about the postnatal world [93, 94, 95].
Understanding fetal sensation, perception, and learning may require more than just neural reactivity, as behavioral observations can enhance our comprehension of fetal life. Fig. 5 (Ref. [92]) illustrates the fetal developmental process, setting the stage for an in-depth discussion on the development of sensation, perception, and learning in utero [93].
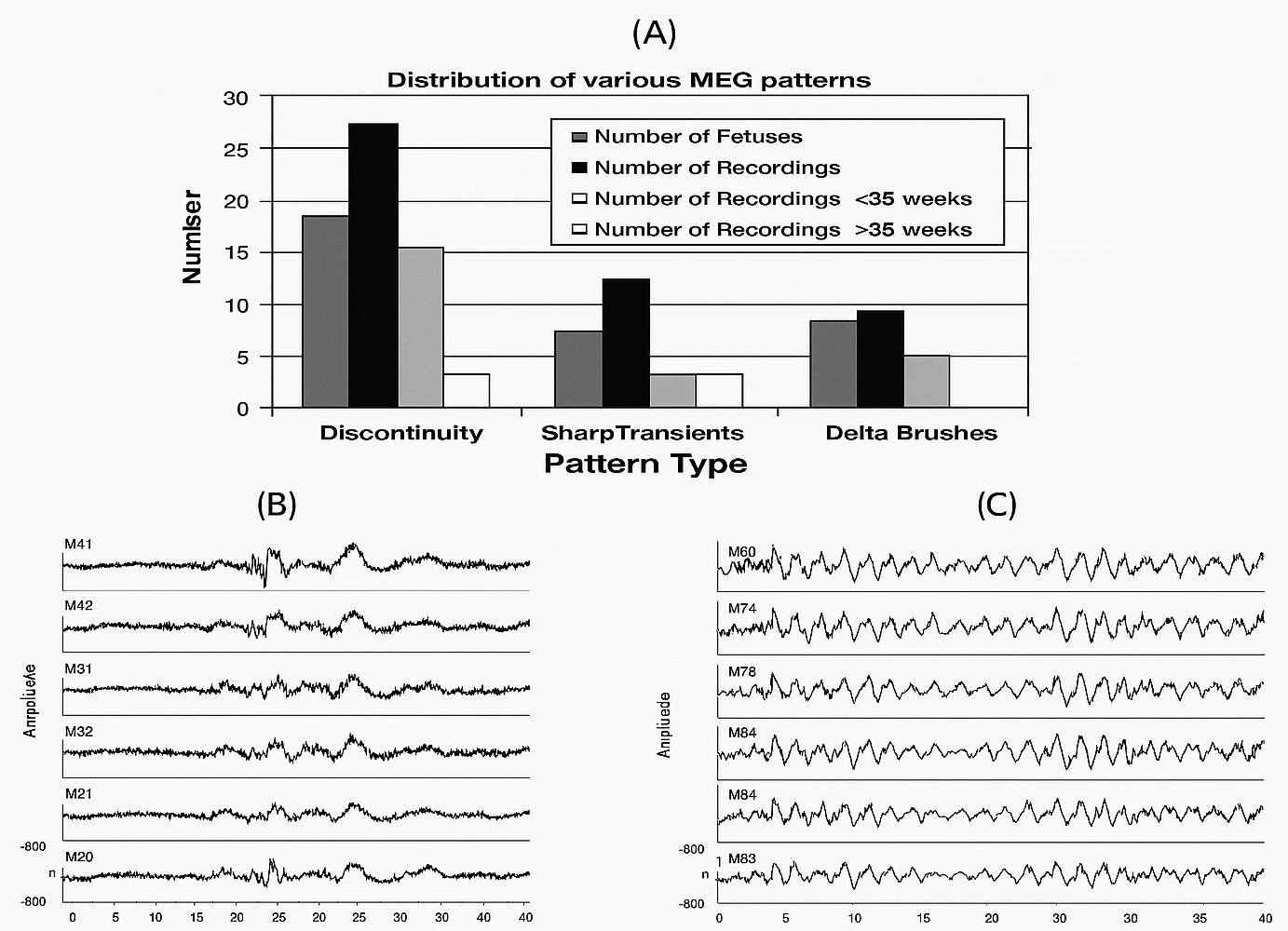 Fig. 5.
Fig. 5.
Developmental maturation of fetal cortical activity measured by magnetoencephalography (MEG). (A) Aggregated recording data of the distribution of three MEG patterns before and after 35 weeks of gestational age. (B) MEG recordings from a fetus at 29 weeks post-conception. The observed pattern is a representative discontinuity, analogous to “trace discontinue” in the EEG of preterm newborns. (C) MEG recordings of a 36-week GA fetus exhibit a characteristic discontinuous pattern morphologically akin to a trace alternant observed in the EEG of premature babies [Eswaran et al. [92], 2007, with permission.]. EEG, electroencephalography.
Fetuses also develop taste and smell sensations, reacting differently to pleasant and unpleasant tastes in amniotic fluid, and may acquire food preferences during fetal life. These sensory experiences contribute to early attachment to the mother and learning about the postnatal world. Understanding fetal sensation, perception, and learning may require more than just neural reactivity; behavioral observations can enhance our comprehension of fetal life [2, 96, 97]. Fig. 5 illustrates the fetal developmental process, setting the stage for a deeper discussion on the development of sensation, perception, and learning in utero. Table 2 (Ref. [2, 98, 99, 100]) provides a summary of the relationship between the physiological development of the brain, and sensory and cognitive function.
| Gestational age (weeks) | Brain development | Reference – fetal sensation, perception and cognition | ||
| Sensation | Perception | Cognition | ||
| 20–24 | • Significant development of the cerebral cortex, forming primary brain waves and distinct brain regions. | • Development of the visual system, including retinal cells and visual cortex formation. | • Physical responses to stimuli like movement and light, showing basic perceptual ability. | • Early signs of learning through conditioned responses to repeated sounds or music. |
 |
• Beginning of synaptogenesis, creating essential neural connections for cognition. | • Beginning light sensitivity, though vision is still immature. | • Early recognition of rhythm or repetition in sounds, indicating sensory processing. | • Evidence of short term memory for familiar auditory stimuli. |
| • Neuronal migration to form various brain structures. | • Progress in auditory system; fetus begins hearing environmental and maternal sounds. | • Beginning to differentiate between types of stimuli (e.g., sound, touch). | • Initial development of simple emotional responses to familiar sounds, without complex emotional understanding. | |
| • Development of the central nervous system, linking brain and spinal cord. | • Initial development of taste and smell, experiencing flavors via amniotic fluid. | • Physical reactions showing distinction between familiar and novel stimuli. | ||
| • Formation of key brain structures such as the amygdala and hippocampus, involved in emotion and memory. | • Formation of nerve endings enabling touch sensation and reflexes. | • Initial processing of emotional stimuli, such as response to mother’s voice. | ||
| 25–28 | • More complex brain waves form, reflecting advanced brain activity. | • Development of blinking reflex in response to light. | • Distinguishes between different sounds, including mother’s voice, with physical responses like movements or rhythm changes. | • Developing ability to learn from repeated stimuli, such as recognizing familiar sounds or music. |
 |
• Accelerated synaptogenesis increases neural connections. | • Beginning of pain sensation with physical reactions. | • Begins responding to visual stimuli, such as light, with movements or rhythm changes. | • Emerging memory for recurring auditory experiences, indicating early retention. |
| • Development of structures like the corpus callosum, improving communication between hemispheres. | • Increased sensitivity to complex sounds, including speech and music. | • Experiences physical sensations, developing basic body awareness. | • Initial formation of basic associations between stimuli and responses, though still very limited. | |
| • Growth of glial cells, enhancing support and protection for neurons. | • More developed taste and smell experiences. | • Shows facial expressions, indicating early emotional responses to stimuli. | ||
| • Early development of basic sensory responses to sound and movement. | ||||
| 29–32 | • Continued maturation of the cerebral cortex with new nerve cell layers, boosting cognitive function. | • Improved hearing with recognition of familiar voices. | • Somatosensory-evoked potentials recorded from about 29 weeks, indicating pain perception in the somatosensory cortex. | • Fetus shows controlled responses to stimuli, such as head and eye movements in reaction to light, stopping when the stimulus is removed. |
 |
• More complex brain waves reflect advanced cognitive and sensory processing. | • Ability to distinguish between high and low sound frequencies. | • Increased responsiveness to tactile stimuli and stronger reactions to familiar sounds. | • Improved ability to retain and recognize recurring sensory input. |
| • Ongoing synaptogenesis strengthens neural communication. | • Response to changes in light and dark. | • Improved visual attention to light changes and patterns. | • Early signs of stimulus-response regulation, showing growing cognitive control. | |
| • Further development of brain structures like the thalamus and hippocampus, linked to memory and emotion. | • Development of taste and smell preferences influenced by mother’s diet. | • Beginning of more coordinated multisensory perception (e.g., combining touch and sound). | • Continued development of associative learning from repeated experiences. | |
| • Central nervous system stabilizes with more complex brain-spinal cord connections, improving motor and sensory functions. | • Growing coordination between different senses. | • Enhanced recognition of rhythmic and repetitive stimuli. | ||
| 33–36 | • Continued maturation of the cerebral cortex with development of gyri and sulci, increasing surface area and connectivity. | • Cortical activation in response to sound detected from 33 weeks. | • Formation of facial expressions with about 19 facial action units (AUs) observed by 34–35 weeks. | • Detects changes in acoustic frequency and anticipates auditory patterns (evidenced by MEG). |
 |
• Intensified electrical brain activity showing advanced cognitive and sensory functions. | • Increased sensitivity to speech and complex sounds. | • Increasing complexity of facial movements, with simultaneous AUs forming facial gestalts (e.g., cry expression). | • Begins to show habituation, reduced response to repeated sounds, indicating learning. |
| • Strengthening interhemispheric connections via the corpus callosum. | • Enhanced coordination between touch, hearing, and vision. | • Enhanced sensory integration, coordinated responses to combined stimuli (e.g., light and movement). | • Early sensory integration supports more complex cognitive processing. | |
| • Proliferation of glial cells, improving neuron support and protection. | • Heightened sensitivity to smells in the amniotic fluid, influenced by maternal diet. | • Signs of habituation, reduced response to repeated stimuli, indicating early perceptual learning. | ||
| • Preparation for postnatal cognitive processing. | ||||
| 37–40 | • Increased myelination, speeding up neural signal transmission. | • Sensory development focuses on refining and deepening existing senses rather than creating new ones. | • Fetus responds to mother’s voice with head-turning, increased heart rate, and reflexive movements. | • Learns sequences of auditory beeps, showing advanced auditory learning. |
 |
• Maturation of brain areas involved in higher cognitive functions. | • The fetus develops more precise and complex responses to sounds, light, and tastes. | • Demonstrates selective attention to music and rhythmic patterns. | • Brainstem reflexes respond to and differentiate specific auditory frequencies. |
| • Enhanced and more organized brain activity with synchronized brain waves. | • These changes reflect maturation of the senses and preparation for life outside the womb. | • Shows preference for familiar auditory stimuli, indicating memory based perception. | • Improved processing and memory of complex sounds. | |
| • Preparation for motor control and reflexive movements. | • Able to distinguish between different voices, especially maternal vs. unfamiliar. | • Early attention regulation, focusing on meaningful stimuli like mother’s voice. | ||
| • Improved communication between brain regions through wave synchronization. | • May exhibit anticipatory responses to repeated stimuli, suggesting early predictive processing. | • Begins rudimentary predictive processing. | ||
| • Critical stage where sensory, cognitive, and motor systems integrate, preparing for life outside the womb. | ||||
We previously reviewed converging evidence showing that the transition from basic aspects of cognition (implied in perceptual activity) to primary consciousness occurs between the second and third trimesters of gestation [2]. This all-important initial transition is followed by successive transitions to higher-order forms of awareness after birth, including self and extended awareness in older children and adults. Environmental interactions are initially cognition, and are then experienced as embodied neurobiological and neurophysiological processes. The emergent transitions starting in utero carry over and complete a developmental cycle ending at approximately two years postpartum. These pass through the newborn period, which will be reviewed next.
Despite the growing interest in fetal cognition, significant methodological challenges remain. Measuring EEG in utero is technically challenging due to the need for noninvasive methods. The fetal environment is difficult to access, and maternal tissues often interfere with signal reception. Noise and artifacts also make it difficult to accurately interpret data and draw reliable conclusions, often affecting the quality of fetal EEG signals. Furthermore, fetal brain development varies considerably between different stages of pregnancy, affecting the consistency of EEG measurements and complicating interpretation. Claims regarding fetal “awareness” or intention are particularly difficult to substantiate, as the presence of brain activity does not necessarily indicate conscious awareness or intentional behavior.
These methodological limitations highlight the need for caution when interpreting findings related to fetal cognition. While advances in technology and research methods continue to improve our understanding, it is essential to acknowledge current limitations and avoid overgeneralizing claims regarding fetal awareness or intent.
We will now describe some of the learning and memory processes available over the first two years postpartum. Many of the most exciting findings over the past decades have involved the discovery of a range of learning and memory systems in infancy. Here, we review studies that provide evidence for statistical learning, spatial learning, threat-based learning, reward and reinforcement learning, and early memory retrieval processes. We examine evidence for the neurological basis and developmental trajectories of these processes, and address their interplay with environmental factors and their influence on the development of the infant’s nervous system.
The subject of neonatal cognitive abilities has in the past led to controversial debate with extreme positions, such as the notion of “tabula rasa”. Despite this, the study of early human cognition has advanced, and many cognitive abilities have been reported to exist at birth and before. According to the contemporary view in developmental cognitive neuroscience, many human cognitive capacities are now thought to be innate [101, 102].
Apart from the simple and automatic sensory-motor prerequisites that allow early interaction with the environment (e.g., following a bright stimulus with eyes, or turning the head toward an acoustic stimulus), newborns also show complex cognitive abilities. Numerous studies over the last few decades have reported that newborns possess perceptual and attentional abilities that allow them to organize a complex sensorial world [102, 103]. For instance, newborns can coordinate their oculomotor and attentional systems to orient toward a peripheral stimulus [104, 105]. They can also select and encode visual information [106], beginning postnatal life with the ability to perceive and organize visual information and recognize real face images [107, 108], highly schematic face-like patterns [107, 109], and biological movement [110].
Newborns are also able to elaborate auditory complex inputs, such as pitch [111, 112] and speech stimuli [113], and recognize and prefer their mother’s voice in comparison to an unknown voice [114]. Therefore, differences between the cognitive abilities of adults and infants, which have long been assumed to be due to deficiencies in the human system at birth, are now regarded as being overcome by developmental processes. The widespread use of the term “immature” to describe the behavioral performance of infants supports the view that functions and structures should be understood in terms of what they will become, rather than what they are at a given point during development.
Consequently, our understanding of the neurocognitive function of the newborn has transformed from a bio-behavioral organism that passively receives stimulation, to one that is active in processing sensation and perceives, discriminates, remembers, cognizes, attends, and repeats learned responses to carry out evolutionarily important behaviors [115, 116].
Human infancy from birth to the age of two years represents a period of remarkable physical and cognitive transformation. During this critical phase, not only do infants grow rapidly, they also develop foundational cognitive abilities that will shape their future learning and memory processes. The study of infant learning and memory has evolved significantly, revealing that even newborns possess sophisticated cognitive abilities. This contradicts the “tabula rasa” argument, supported by behavioral psychology, which suggests that infants are born without any innate knowledge or cognitive abilities. However, contemporary developmental cognitive neuroscience has shown that many cognitive abilities are innate.
The predictive processing framework suggests the brain is constantly generating and updating a model of the environment based on incoming sensory information. Essentially, the brain makes predictions about what it expects to perceive, and then updates these predictions based on the actual sensory input it receives. This ongoing process helps the brain in reducing the discrepancy between its expectations and the actual sensory information, referred to as prediction errors. In infant learning, the predictive processing framework views infants as active learners who predict and interpret their environment, rather than passively receiving sensory input. This supports rapid adaptation and unifies perception, action, and learning into one process.
The predictive processing framework offers a powerful explanation for various infant learning processes, such as object recognition, categorization, causal understanding, and social cognition. By continually improving predictions based on sensory input, infants build increasingly accurate models of their environment, supporting cognitive and behavioral growth. This model also emphasizes the essential role of experience and environmental input in shaping development through dynamic brain-environment interactions. However, the framework has been criticized for its limited empirical basis in infancy research, insufficient evidence for similarity to adult neural mechanisms, and possible overinterpretation of infant behavior. Some researchers suggest that developmental systems approaches may be better at explaining aspects of early cognition.
While the predictive processing framework has had an impact on adult neuroscience, its application to infancy research is still evolving. Köster et al. [117] have argued that despite its theoretical appeal as a unifying principle for understanding infant learning, including statistical learning, motor development, and environmental understanding, only a few direct empirical investigations have so far applied this model to infants. This gap makes it difficult to assess the explanatory power of the framework in early development. For instance, while infants show brain activity patterns (e.g., theta synchronization in the anterior cingulate cortex) related to prediction error processing, it is still not known how early these mechanisms develop, and whether they operate identically across ages [118].
Neuroconstructivism is a theoretical framework that explains cognitive development as an evolving, experience-dependent process shaped by dynamic interactions at genetic, neural, cognitive, and environmental levels [27, 119, 120]. Rather than viewing abilities as fixed or predetermined, it emphasizes that mental representations and brain structures are constructed gradually over the course of development [119, 120]. This perspective allows for a deeper understanding of both typical and atypical trajectories, and emphasizes the plasticity and context sensitivity of cognition.
Although modeling such multilevel interactions poses methodological challenges, particularly given the lack of longitudinal data, neuroconstructivism remains a powerful framework for investigating developmental change. At the same time, the predictive processing framework, which views learning as driven by the brain’s attempts to minimize prediction errors, offers complementary insights into early cognition. Together, these models emphasize the central role of experience and the interplay between brain and environment in shaping the developing mind.
The first two years of life are crucial for the growth and expansion of learning and memory systems. Various learning processes, including spatial and statistical learning, threat, reward and reinforcement learning, are all observed in infants. These processes are supported by neural substrates that undergo significant maturation during this period.
Despite the historical belief that infants are incapable of long-term memory, current research indicates otherwise. An infants’ experiences, even those from the earliest days of life, can have enduring impacts on their cognitive development. This challenges the notion of infantile amnesia, where adults cannot recall events from the first few years of life [121].
The ability to detect patterns and regularities in the environment is termed statistical learning. Infants are remarkably adept at this type of learning, allowing them to make sense of the world around them. Spatial learning involves understanding the physical layout of the environment. Infants develop spatial awareness through interactions with their surroundings, such as crawling and exploring. This type of learning is essential for navigating and understanding spatial relationships.
Reward and reinforcement learning are mechanisms by which behaviors are shaped by their consequences. Infants learn to associate certain actions with positive or negative outcomes, thereby reinforcing behaviors that lead to rewards, and reducing those that result in adverse effects [122, 123].
Threat-based learning involves recognizing and responding to potential dangers. Infants can learn to associate specific cues with threatening situations, helping them to safely navigate their environment [124].
Memory retrieval processes in infancy are critical for the long-term retention of information. Infants can remember and recall experiences, demonstrating that memory systems are functional even at a young age. This is evident by their ability to recognize familiar faces, objects, and sounds [125, 126].
Interaction between the infants’ cognitive processes and their environment is vital for cognitive development. The environment provides stimuli that drive learning and memory processes, while the infants’ cognitive abilities shape their responses to these stimuli. This dynamic interaction influences the development of neural substrates and cognitive functions. The schematized pathways involved in infant learning are represented in Fig. 6.
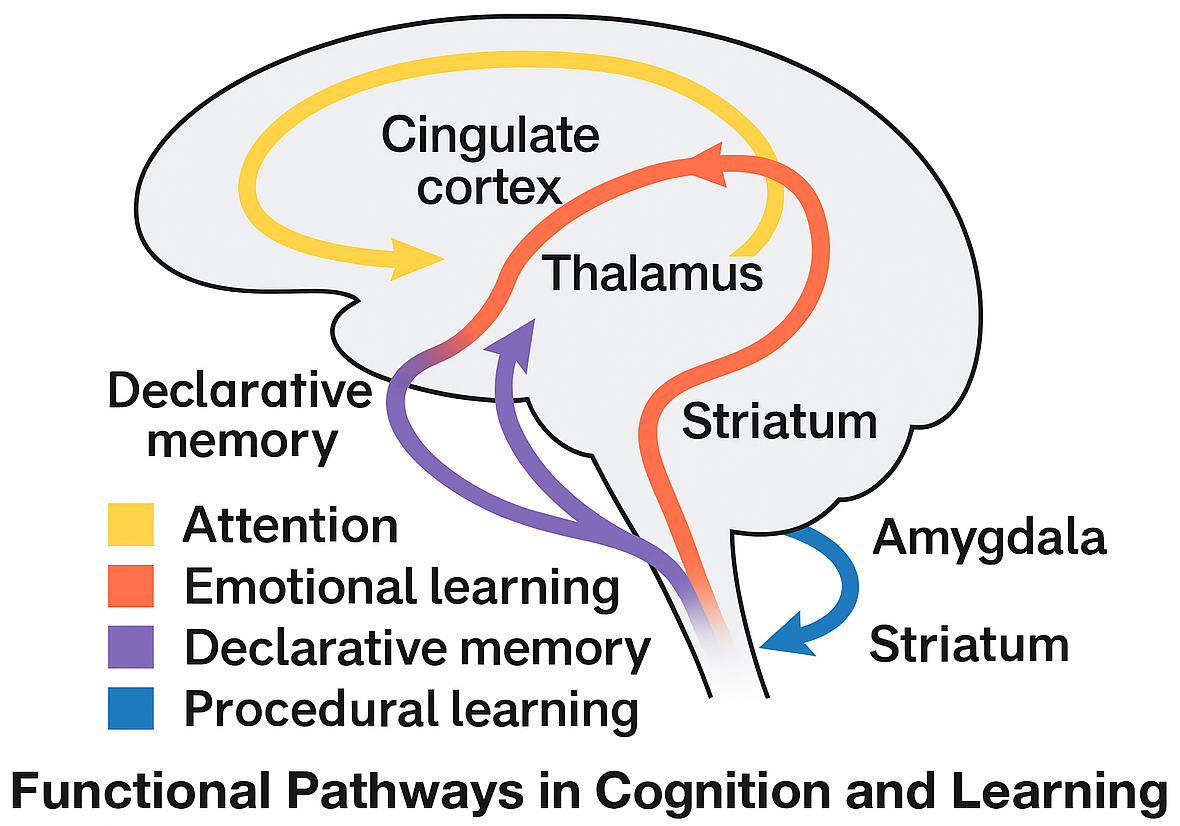 Fig. 6.
Fig. 6.
Illustration of the functional pathways in cognition and learning. The arrows depict the following connections: attention from thalamus to cingulate cortex; emotional learning from amygdala to cingulate cortex; declarative memory from hippocampus through thalamus to cingulate cortex; and procedural learning from striatum via thalamus to cortex. The diagram is overlaid on a neutral gray brain outline to provide anatomical context.
The environment offers a rich array of stimuli that contribute to learning and memory development. Social interactions, sensory experiences, and physical exploration all play a role in shaping cognitive abilities. Infants learn from their surroundings, forming associations and building knowledge through continuous engagement.
Human infancy spans from birth to two years old, during which time infants experience significant physical and behavioral changes. By the end of this period, few traces of their newborn state remain. Freud was the first to suggest that adult behavior could be linked to early childhood experiences, sparking considerable theoretical interest in the long-term effects of these early experiences. Many developmental scientists believe the experiences of infants build upon each other and are crucial for later cognitive development, implying that infants have some capacity for long-term memory. If behavior and cognition are involved, infants must have a way to retain a lasting record of events. However, most researchers also argue that preverbal infants cannot retain long-term memories of their experiences. The phenomenon of infantile amnesia, where adults cannot recall events from before the age of three to four, supports this belief. This paradox continues to shape contemporary studies and theories about newborn learning and memory [126].
Today, learning across all domains has been recorded from the moment of birth. Early studies into newborn learning were conducted to record the foundations of adult behavior. Employing methodologies intended for adults, they presented an unsatisfactory perspective on infant learning [127]. Pavlov, for instance, ascribed the unsuccessful classical training efforts with early newborns to cerebral immaturity. Years later, it was noted that increasing the optimal interstimulus interval by a factor of 2–3-fold compared to adults enabled classical eyelid conditioning of eye blink in sleeping neonates and 10-day-old infants [128]. Adults in a state of sleep are unable to form new associations. Using methods designed for infants, researchers discovered that neonates quickly develop conditioned feeding reflexes [121]. At a designated feeding time, a tone was paired with tactile stimulation on the right cheek, prompting a right-head turn that led to milk being dispensed from the right side. In a similar manner, a buzzer was paired with stimulation on the left cheek, prompting a left-head turn that led to milk being dispensed from the left side. Neonates learned to turn right when they heard a tone and left when they heard a buzzer [129]. Infants showed a preference for a chamomile scent associated with breastfeeding within their first 8 days of nursing. At 7 months, infants preferred chamomile-scented teething rings, and by 21 months they favored a chamomile-scented toy for play [130].
It is evident that children acquire knowledge and derive advantages from prior experiences, indicating the presence of some form of memory. Several decades ago, DeCasper and Spence [131] proposed that prenatal experiences can influence subsequent behavioral responses. Just hours after birth, neonates can differentiate between a new story and one that their mothers recited during the final weeks of pregnancy [132]. The study employing these paradigms has found evidence of remarkably strong memory in very young infants [133].
The early stages of neural development in infants lay the crucial groundwork for future learning and memory capabilities. This development is a complex and dynamic process, characterized by innate cognitive capacities that enable infants to interact with their environment and acquire knowledge. Various learning processes, supported by neural substrates, contribute to cognitive development during the first two years of life [3, 134].
During this critical period, the brain undergoes rapid growth and organization, marked by the formation of neural connections and the pruning of unused pathways [77]. Heightened plasticity allows infants to adapt to their environments and acquire new skills. Key processes such as synaptogenesis and myelination play significant roles in enhancing cognitive functions, enabling infants to process sensory information, develop language skills, and form attachments [135, 136].
Interactions between the cognitive abilities of infants and their environment shape their learning experiences and influence long-term cognitive outcomes. Understanding these processes provides valuable insights into the foundations of human cognition and the factors contributing to healthy cognitive development. The maturation of neural substrates is closely tied to the development of learning and memory systems, with various brain regions and neural networks and hubs supporting different types of learning and memory processes [137].
The development of networks and hubs supporting cognitive development in infancy is a complex process characterized by the maturation and integration of functional brain networks. Research indicates that during early childhood, significant changes occur in the connectivity of brain regions, particularly in networks associated with cognitive control and higher-order functions. This is critical for the development of cognitive abilities such as language, motor skills, and visual processing. As they grow, infants shift towards networks linked to higher-order cognitive processes, with increased recruitment of functional networks correlating with skill complexity [138]. The cingulo-opercular and frontoparietal networks exhibit increased connectivity from early infancy to age nine, indicating they play a role in cognitive control during activities like story listening [139].
Functional hubs facilitate communication among brain networks. They begin to stabilize in late childhood, with connections to non-hub regions strengthening throughout development. The architecture of these hubs is essential for efficient information flow, supporting cognitive functions as they mature [140].
While the development of these networks is generally seen as beneficial for cognitive growth, some studies suggest that disruptions in early connectivity could lead to challenges in language acquisition and other cognitive skills. This highlights the importance of early intervention in cases of atypical development.
Newborns exhibit perceptual and attentional abilities that allow them to recognize and respond to social stimuli, such as faces and biological motion. They can also process complex auditory inputs, including pitch and speech stimuli. These foundational abilities are essential for the development of more advanced cognitive processes. The brain’s plasticity enables infants to learn and remember information effectively, supporting the formation of new neural connections and the strengthening of existing ones.
Sensory Preconditioning (SPC) is a form of learning that occurs without observable behavior, whereby a link is established between two neutral environmental stimuli in the absence of reinforcement, before formal training [140]. An additional training program is essential to provide participants with a clear method of expression. The SPC paradigm includes three components: (1) pre-exposure, where subjects experience two paired stimuli (S1, S2); (2) a training phase, during which subjects learn to link S1 with a reinforcement source; and (3) a transfer-test phase, where subjects are evaluated with the untrained stimulus (S2). When respondents act during the test as if S2 had received reinforcement, it can be inferred that an association was established between S1 and S2 during the pre-exposure period. The standard control group undergoes the same treatment, with S1 and S2 being exposed independently (unpaired) during phase one. This group should avoid performing the reinforced response during the transfer test with S2 [141].
Spear and colleagues [141] noted that the increased ability of neonatal rat pups to develop concurrent associations with SPC concludes after two weeks postnatally. Eight- and 12-day-old rat pups could establish simultaneous associations, whereas 21-day-old pups could not [141]. In applying SPC to human infants, six-month-old infants were pre-exposed simultaneously to two distinct and adjacent (cloth panels) for one hour daily over seven days. In contrast, infants in the unpaired group were pre-exposed to the two panels for the same duration, but at different times of the day. One day later, all participating infants were trained to kick a mobile while one of the panels was present (phase 2). This was followed by a 24 h transfer test with the trained mobile in the presence of the alternate panel (phase 3). Six-month-old infants did not recognize the mobile which they were trained to kick in a different context [142]. The paired pre-exposure group showed significant retention in the untrained setting, while the unpaired pre-exposure group did not. The paired group had linked the two panels in phase one, facilitating the transfer of conditioned responses to the untrained environment.
Barr et al. [143] utilized Boller’s pre-exposure protocol to investigate SPC through a deferred imitation task. Their findings showed that six-month-old infants who were pre-exposed to two hand puppets demonstrated a significantly higher mean imitation score of target actions with one puppet than did a baseline control group. In contrast, the unpaired control group, which was exposed to the puppets at different times, did not show this enhanced imitation, suggesting that pre-exposure to paired stimuli facilitates the ability to imitate learned behaviors in infants.
To evaluate the specificity of the connection, a different paired pre-exposure group was tested with a new puppet, C. This group was unable to replicate the activities demonstrated with puppet C, confirming the association was unique to puppets A and B. Similar results were observed when infants were pre-exposed to the paired puppets for only two days. Inquiring about the duration for which the association between puppet A and puppet B could remain dormant before retrieval. Rovee-Collier and Cuevas [144] studied the retention of learned behaviors in infants aged 6-, 9-, and 12-months after pre-exposure to paired puppets. Their results showed that 6- and 9-month-old infants could replicate target actions after a two-week delay, but not after a three-week delay. In contrast, 12-month-old infants did not imitate the actions at all, indicating they did not establish the initial relationship between the puppets. By employing an alternative deferred imitation challenge, Barr et al. [145] also found that 12-month-old infants were unable to establish a contemporaneous association. Cuevas and Davinson [126] reported that 6- and 9-month-old infants established simultaneous associations, but 12- and 15-month-old infants did not. At 18 months, the capacity of infants to establish simultaneous associations reemerged.
Giles and Rovee-Collier [146] investigated how the duration and number of pre-exposure sessions influenced the retention of associations in infants aged 6 and 9 months. They found that while both age groups showed notable imitation of puppet behaviors after specific intervals, the advantage of memory retrieval at the start of an additional session outweighed the benefits of longer exposure times. Additionally, when retrieval occurred on the same day rather than 24 h later, infants retained the association for a week longer, indicating significant forgetting.
Infants aged three months can associate several stimuli presented simultaneously without reinforcement. Bhatt and Rovee-Collier [147] demonstrated that three-month-old infants could form associations between multiple stimuli presented simultaneously without reinforcement. The findings indicated these infants could (1) create numerous associations among the attributes of a multi-element event, (2) encode different information from the same event compared to adults, and (3) acquire more knowledge about the event than adults.
In stimulus-response relationships, potentiation refers to the enhancement of memory for a less salient stimulus when it is presented alongside a more salient stimulus. This occurs because the more noticeable stimulus, which gained its strength through previous associations, helps to strengthen the association of the weaker stimulus when both are presented together. Essentially, the associative strength of the stronger stimulus boosts the weaker one. For example, in studies with infants, potentiation has been observed when a more salient stimulus, like a train, is used as a memory cue to enhance the memory of a less salient stimulus, like a puppet task. This results in the infant remembering both tasks for a longer period. In adults, similar experimental conditions were found to give rise to overshadowing instead of potentiation. Wagner and Rescorla [148] examined the phenomenon of overshadowing in the 1970s, referring to a situation in which a more salient or stronger stimulus reduces the learning or association of a weaker stimulus when both are presented together. Essentially, the presence of the stronger stimulus “overshadows” the weaker one, making it less likely for the weaker stimulus to elicit a response. For example, suppose a loud sound (strong stimulus) and a faint light (weak stimulus) are presented together. In that case, the organism is more likely to associate the response with the loud sound rather than the faint light, in contradistinction to potentiation. Potentiation in adults is mainly observed in odor and taste aversion learning [149], but it has also been seen in autoshaping, maze learning, and contextual conditioning [150].
Kucharski and Spear [151] showed that potentiation is more pronounced in rat pups than adults. Eighteen-day-old rat pups showed improvement in odor conditioning when presented with an additional odor, whereas 60-day-old rats experienced a disadvantage in odor conditioning when presented with an additional odor. Additionally, 15-day-old rat pups showed less blocking in odor learning compared to adults.
In a study on mediated imitation, Spear and Kucharski [152] found that six-month-old infants remembered the train task for two weeks after learning, whereas their memory of the puppet task lasted only one day. When the train was presented immediately after training, they also remembered the puppet task for two weeks. Barr et al. [153] aimed to enhance the associative intensity of a potentiation stimulus by presenting a train as a memory cue. This resulted in the infants remembering both the train and puppet tasks for four weeks.
Memory retrieval after long delays strengthens them. Barr et al. [153] sought to enhance the train task strength by extending the retention period from 7 to 14 days, which is the maximum duration that 6-month-old infants can recall the train task. As expected, recalling the train memory 14 days after operant training strengthened it, and improved encoding of the puppet demonstration. Consequently, infants retained the train task for eight weeks instead of four, and the puppet task for six weeks instead of four.
The neuro-maturational hypothesis does not account for the finding that very young infants are able to remember the puppet imitation task for at least six weeks after just one 60-second performance. The paradigm posits that newborns cannot encode and retain a lasting memory of a singular demonstration for more than 24 h until the late-developing explicit memory system becomes functional [154]. Previous research indicated that potentiation is a primary process that substantially improves the encoding and long-term memory of new, predictive associations within the environment of infants.
Infant learning demonstrates exceptional capacities to establish intricate relationships and draw conclusions from an early age. A crucial element of this learning process is the establishment of associative chains and the ability for transitive inference. These cognitive processes enable newborns to associate distinct inputs into coherent sequences and to establish connections between parts that were not directly connected, indicating early and sophisticated cognitive capabilities.
Associative chains establish connections among a sequence of stimuli delivered consecutively, allowing children to develop an interconnected memory network. This phenomenon was investigated in trials where infants were subjected to various stimuli over successive days, and the duration of retention of these associations by the infants was then assessed.
With transitive inference, children demonstrate the ability to establish indirect associations between stimuli that were not directly matched during the learning process. This capability indicates that newborns may deduce links among items based on their associations with other stimuli, demonstrating a level of cognitive processing that was previously undervalued for such early developmental stages.
Two investigations revealed that 6-month-old infants possess an unusual ability to establish a complicated sequence of relationships. In both instances, infants established a sequence of concurrent connections that were progressively connected over several days. The initial study by Rovee-Collier and Cuevas [155] was based on an experiment by Dwyer et al. [156], where rats established an indirect linkage between two independent stimuli that had never co-occurred and were absent during the formation of the association.
Barr et al. [157] found that infants formed an association between puppets A and B after being pre-exposed to them for two consecutive days. Following this, they learned to kick to move a mobile in a specific context, creating a mobile-context association. The researchers predicted that when puppet A was later presented, it would reactivate the memory of puppet B, while the context would simultaneously activate the memory of the mobile. To ascertain whether the association had been established, they simulated a sequence of target actions on puppet B one day later and inquired about the duration of memory retention by the infant for these acts. If puppet B and the training mobile were not related, infants should delay imitation for only 24 h; conversely, if they were associated, infants should delay imitation for 2 weeks [157]. Infants exhibited deferred imitation for two weeks, whereas a control group with no relationship did not. Dwyer et al. [156] noted that six-month-old infants linked the memories of two never co-occurring absent stimuli. Their study also found that direct and indirect connections showed comparable efficacy.
One significant challenge in infant learning is managing infant associative learning. While their ability to form numerous connections is impressive, it can lead to an overload of associations, some of which might be redundant, irrelevant, or even counterproductive [158].
The significant downside of exuberant learning by newborns is their tendency to form excessive associations, many of which may be incorrect, irrelevant, or unhelpful. The challenge subsequently arises in determining how to eliminate the numerous superfluous relationships. Kraemer and Spear [159] compared this issue to the overproduction and pruning of superfluous synapses with brain maturation, and suggested the infantile propensity for abundant learning would probably be associated with mechanisms for the selective pruning of numerous non-relevant associations. The two processes noted were (1), accelerated forgetting in younger individuals, and (2) expedited extinction of the memory trace in younger individuals.
Rapid forgetting is a fundamental technique by which newborns navigate the numerous associations they establish. The process of forgetting is not only indicative of an evolving memory system, but also functions adaptively. By quickly forgetting, newborns can efficiently eliminate redundant or irrelevant associations, thus refining and reinforcing significant connections.
It is well known that newborns exhibit a faster rate of forgetting than adults. In numerous species, such as frogs [160], chicks [161], mice [162], rats [163], puppies [164], monkeys [165], and humans [166], younger animals exhibit more rapid forgetting than older animals with the lengthening of the retention interval, despite comparable retention following brief test delays. The accelerated forgetting by newborns is commonly characterized as a memory deficit, attributed to either diminished or less robust encoding, an incapacity to retain memories in storage, or an impairment of retrieval [166, 167]. Human neonates show limited long-term memory retention of events, which is often attributed to the neuro-maturational paradigm positing that explicit memory systems mature later in development [168]. Moreover, some researchers argue that forgetting in early life may have adaptive value, allowing infants to prioritize relevant information as their cognitive systems develop [169].
Rapid forgetting serves as an adaptive mechanism to manage the vast amount of information encountered by infants. It is not necessarily a memory deficit, but rather an evolutionarily selected strategy that helps infants adapt to their rapidly changing environment. This accelerated forgetting allows infants to shed excessive, potentially irrelevant, or inappropriate associations [170, 171].
The retention of a new connection is enhanced and its forgetting prevented when a related cue is presented to the infant, hence facilitating memory retrieval [166, 172]. Furthermore, retention is extended when the encoding and retrieval duration is increased [173]. The advantage of memory retention by retrieval after an extended delay is most pronounced in younger infants. Accessing memory after conclusion of the forgetting function enhances memory by 136% at 6 months, 67% at 9 months, 62% at 12 months, and just 40% at 15 months [171]. Even after the connection has been lost, its memory can be resurrected if an individual encounters the identical signals present during the original encoding, although there is a temporal limit to this possibility.
The neuro-maturational model fails to explain these findings. The hypothesis posits that the brain of an early infant lacks the maturity necessary to encode, retain, and recover long-term memories prior to the conclusion of the first year.
The process of exuberant learning and rapid forgetting continues until the end of the first year of life, but does not last indefinitely. As infants approach their first birthday, their learning processes begin to undergo significant changes. The ability to form numerous associations begins to wane, and the mechanisms for managing these associations become more refined [155].
The transition marks the conclusion of the exuberant learning period, and the brain then begins to favor more stable and long-term connections rather than the rapid formation and pruning of myriad associations. This shift is crucial for the development of more complex cognitive abilities and the stabilization of memory [172].
Research indicates that by around 9 months of age, the ability of infants to form exuberant associations starts to decline. This decline is an adaptive response that allows for more effective learning and memory retention [173].
Cuevas et al. [174] found that infants aged six months were able to associate two puppets that had been pre-exposed simultaneously, but not when they were pre-exposed sequentially. In contrast, 9-month-old infants could relate two puppets regardless of whether they were pre-exposed sequentially or simultaneously. Interestingly, 12-month-old infants showed the ability to associate two puppets that were pre-exposed sequentially, but not simultaneously. Consequently, the ease with which newborns establish vibrant associations between concurrently offered stimuli diminishes after 9 months postpartum. Cuevas noted that modifying the number of trials in the sequential pre-exposure condition influenced SPC solely at the transitional age of 9 months. At this time, an increase in trials extended the interstimulus interval necessary for infants to establish a sequential association, while a reduction in trials completely hindered their capacity to form such an association.
The phase of exuberant learning in human infants concludes around 9 months, aligning with the transition from experience-based perceptual tuning to perceptual narrowing, and the presumed shift from the implicit memory system to the explicit memory system [155]. The typical age at which these three shifts occur is not coincidental. All transitions illustrate the same phenomenon: a developmental shift in the learning and memory abilities of early infants. The phase of exuberant learning and the initial stage of perceptual tuning [155, 174] examine how rapidly developing but still immature organisms integrate the fundamental aspects of their environment with the survival-supporting relationships formed while adapting to a rapidly changing ecology. Due to the fast changes in their niches during early development, immature newborns must rapidly acquire essential relationships. The timing of the changeover for both intervals has been objectively established. Conversely, the neuroanatomical foundations for implicit memory have not been explicitly examined in human neonates [174]. The timing of the transition to the explicit memory system has been deduced from the appearance of behaviors linked to different neuroanatomical components of the explicit memory system in adulthood.
It is not surprising that the implicit memory system cannot explain the phase of exuberant learning, since the features of the implicit memory system were identified based on the learning and memory capabilities of amnesic individuals who had experienced a period of intense learning during infancy [175]. While the significance of developmental alterations in the brain mechanisms underpinning learning and memory is universally acknowledged, Spear noted that “an ontogenetic change in the neurophysiological mechanisms responsible for all learning and memory seems unlikely” [176].
Although the neuro-maturational model explains many aspects of memory development, it does not fully account for the exuberant learning observed in early infancy. An alternative perspective suggests that a universal learning process, such as quick mapping, may explain how infants rapidly acquire information. This model posits that while fundamental memory processes change as the brain matures, the core memory mechanism remains constant. Empirical evidence from studies on human neonates supports the idea that early infancy is marked by a unique phase of rapid learning. The neuro-maturational model does not fully encompass this viewpoint.
Multiple factors may interfere with the rapid cognitive development that is characteristic of the prenatal and early postnatal periods. Maternal smoking and cardiometabolic conditions during pregnancy have been associated with adverse cognitive and behavioral outcomes in offspring, including attention deficit hyperactivity disorder (ADHD), behavioral problems, and learning and memory impairments. For example, elevated maternal fasting glucose and triglycerides have been associated with an increased risk of hyperactivity and behavioral problems in children [177, 178, 179].
However, the interpretation of such associations is complicated by potential confounding variables, such as shared genetic predispositions and environmental factors. Genetic education studies suggest that family effects may partially explain the observed outcomes [177, 178]. While there is evidence supporting an association between prenatal exposures and later child development, further longitudinal and genetically-informed research is needed to clarify causal mechanisms.
Socioeconomic status (SES) plays a significant role in shaping early brain development through both structural and functional pathways. Studies show that higher SES is associated with greater brain volumes in regions such as the prefrontal cortex, which supports cognitive and emotional regulation [180]. Conversely, lower SES is linked to elevated maternal stress, which may negatively influence neurocognitive outcomes [181]. Infants from low-income families also tend to show reduced frontal gamma power, which is an early neural marker associated with language and attention development [182].
While SES is a powerful predictor of developmental outcomes, it is also important to consider the broader cultural and contextual factors. Oversimplifying the influence of SES carries the risk of reinforcing stereotypes and overlooking the resilience and variability within low SES families [183].
Although there is no universal agreement among cognitive scientists, it is recognized that memory systems serve diverse functions and operate under fundamentally different rules. Declarative (or explicit) memory encompasses the majority of what we typically associate with “memory” or “remembering”, involving the explicit recognition or recollection of names, locations, dates, and events. In contrast, non-declarative memory includes a range of unconscious competencies, such as priming, the acquisition of habits and skills, and certain conditioning modalities [184]. An important feature of non-declarative memory is that its influence manifests as changes in behavior or performance, while the underlying experiences remain unconsciously inaccessible [185].
Declarative memory is characterized by its rapidity, adaptability, and susceptibility to errors, such as memory trace deterioration and retrieval failures. Non-declarative memory, on the other hand, is generally slower, arising from gradual learning processes, and is considered more dependable but inflexible [186]. The differentiation between these memory types is crucial for developmental biologists, as they rely on distinct brain substrates that exhibit varying developmental trajectories [187].
Non-declarative memory is supported by brain regions such as the cerebellum, neocortex, and striatum [188]. These regions mature early, enabling infants to develop this capability from a young age. The development of non-declarative memory systems is influenced by the dynamic interplay of various brain structures, with the prefrontal cortex undergoing significant maturation during early childhood, facilitating increasingly sophisticated learning and memory [11, 126, 188]. Additionally, neuroplasticity, the ability of the brain to reorganize and adapt its neural connections in response to various experiences, learning, and environmental changes, is a defining feature of early childhood, allowing infants to adapt to their environments and learn from their experiences.
During infancy, the brain undergoes substantial development, particularly in areas related to learning and memory, such as the cortex, hippocampus, and prefrontal cortex [189]. Declarative memory, which involves the conscious recall of facts and events, relies heavily on the hippocampus and associated cortical areas. The hippocampus is integral to the formation and consolidation of these memories, beginning its development during fetal stages and continuing its maturation postnatally. The prefrontal cortex also plays a crucial role in supporting declarative memory by helping infants to focus on relevant stimuli and ignore distractions [190].
The visual paired comparison (VPC) task was introduced by Robert Fantz [191]. This task involves presenting infants with pairs of pictures, where one picture is familiar, and the other is novel. The infants’ preference for looking at the novel picture over the familiar one is taken as evidence of recognition memory. In the VPC task, two identical stimuli are initially presented together. Then, a novel stimulus is displayed alongside one of the previously presented stimuli. Infants demonstrate memory for the familiar item by spending greater time examining the novel stimulus. This method has been employed extensively to examine visual recognition memory in infancy, providing significant insights into early cognitive development.
Changes in allocating attention to known and novel stimuli are generally investigated across brief intervals, with durations spanning from seconds to many minutes. Rose et al. [192] indicated that from the age of 3 to 12 months, duration of the novelty reaction extended from 5–10 seconds to 10 minutes. Nonetheless, these delays do not signify the maximum capacity of baby recognition memory [155]. For instance, infants aged 5 months identify facial stimuli after 2 weeks. Visual attention has been utilized to study memory retention over periods ranging from one to three months. Over extended durations, the recognition evidence changes, leading to increased visual attention towards familiar stimuli. This shift in attention distribution is considered indicative of the varying importance of memory traces over development. The basic premise is that with a recent memory trace, infants can bypass the processing of familiar stimuli and instead concentrate on new stimuli. As the memory trace diminishes, they direct their attentional resources to rebuild the trace for the previously familiar stimulus [193].
While attentional preference approaches assess alterations in the reactions of infants to familiar stimuli, it remains uncertain whether they gauge the same form of recognition as shown by adults when they openly acknowledge prior exposure to a certain stimulus. Mandler [194] proposed that experiments on newborn recognition memory are more comparable to adult priming than to studies of recognition memory. Adults with amnesia exhibit normal priming despite significant impairments in recognition memory. Snyder [195] posited that increased focus on novel stimuli may come from an inherent characteristic of the visual system rather than a mere recognition reaction. She proposed that repetition suppression, characterized by diminished neural responses in the occipital-temporal visual pathway due to stimulus repetition, accounts for the shift in attention from an old stimulus to a new one. This reasoning aligns with the assertions of Nelson [168], who proposed that attention to novel stimuli during early development may be influenced more by the frequency of presentation than by their inherent novelty. In conclusion, while alterations in the allocation of infant attention due to prior exposure may stem from recognition memory, such judgments are not necessary for eliciting the response [196]. Therefore, they should not be presumed, particularly in light of various alternative explanations.
Deferred imitation was initially proposed by Piaget [197] as a key indicator of the emergence of symbolic reasoning in infancy. Commencing in the mid-1980s, this methodology was formulated as an assessment of memory capacity in newborns and young children [198]. It utilizes props to execute a singular activity or a multi-step sequence, after which the infant or young child was asked to duplicate, either instantly (elicited imitation), after a delay (delayed imitation), or both.
Much has been learned from the work of Bauer and associates [199] regarding the conditions under which learning and testing occur during deferred imitation and support the formation of declarative memories, rather than non-declarative memories. This leads to mnemonic behaviors that reflect the characteristics of the declarative memories. Initially, performance improves with multiple experiences [199], but infants can also acquire knowledge and retain it from a single encounter [200]. Declarative memory is known for its rapid learning. Additionally, the content of memories formed through imitation-based tasks can be expressed through language. As children develop linguistic abilities, they can describe multi-step sequences they experienced as preverbal infants [201, 202, 203, 204]. The memory traces formed by tasks requiring imitation show flexibility. Infants retain memories even when (a), the objects used during retrieval differ in shape, size, color, or material composition from those used during encoding [205, 206, 207, 208]; (b) the room’s appearance at retrieval differs from that at encoding [209, 210]; (c) encoding and retrieval occur in different environments [211, 212]; and (d) the individual prompting the recall differs from the person who exhibited the actions [211]. Infants as young as 9 to 11 months show evidence of flexible extension of event knowledge [213, 214]. Imitation-based activities successfully meet the “amnesia test” [215] trialed on persons with amnesia. This is characterized by impaired declarative memory processes compared to control participants, as assessed by an imitation-based task that incorporated multi-step sequences. While typical adults replicated the model’s activities even after a delay, amnesic patients performed inadequately, exhibiting no improvement compared to control participants who had not witnessed the presented events. Older children and young adults who experienced amnesia due to pre- or perinatal injuries exhibit diminished performance on imitation-based activities [216]. These findings support the notion that, despite imitation-based tasks being nonverbal, they still use declarative memory.
Event and autobiographical memory include phenomena ranging from a flag waving in the wind, to intricate and temporally extensive occurrences such as being kidnapped. For current purposes, we adopted the description by Nelson ([217], p. 11): “Events involve individuals engaging in intentional activities, manipulating objects, and interacting with one another to attain a specific outcome”. This definition omits basic physical alterations like flag-waving, as they lack actors participating in intentional actions. Conversely, the term encompasses the activities individuals partake in throughout a typical day, and the distinctive experiences that ultimately shape our identities. This concept delineates the aspects to be recalled regarding events, specifically actors, acts, objects, and the sequence in which these components amalgamate to accomplish particular objectives.
Personal memories of past events influence present behavior and serve as a framework for future planning. Events and their narratives serve as significant teaching instruments: we acquire knowledge by reading and listening to accounts of historical occurrences and their impact on the world. Ultimately, recollections of our participatory experiences are intrinsically defining, as our identity is shaped by our past actions and experiences. We utilize our past experiences to elucidate our current conduct and to inspire future decisions. Consequently, the capacity to recall prior events is essential for both adult and developing individuals.
The defining characteristic of memories related to past events is particularly important for autobiographical memory. As the term implies, autobiographical memories are about oneself. According to Bauer et al. [218], aside from the primary feature of self-relevance, autobiographical memory can be viewed as a “family resemblance” category and is distinguished by its unique features [219]. Especially vivid or prototypical autobiographical memories are of distinctly personal and relevant past events that can be pinpointed to a specific time and place and are expressed verbally. Their recall involves a “re-living” of the past experience. Memories that could also be autobiographical but are less prototypical might lack one or more of these characteristic features. They could be of recurring events [220], might not be shared verbally [221], might be less vivid, or there might not be an awareness of the memory’s source [222, 223]. Viewing autobiographical memory in this manner, as a “mental natural kind” [224], allows for its development to be envisioned under the umbrella of event memory.
Bauer and Fivush [224] contends that, in addition to the singular distinguishing quality of self-relevance, autobiographical memory may be seen as a “family resemblance” category, delineated by its distinctive traits. In particular, “good” or archetypal autobiographical memories pertain to distinctly personal, significant, and specific past events that may be situated in a precise time and location, and are articulated vocally. Their retrieval entails “re-living” the experience as a previous event. Autobiographical memories that are less prototypical may lack one or more defining characteristics: they may pertain to recurrent events [221], may not be communicated verbally [188], may consist of less vivid recollections, or may lack awareness of the memory’s source [222, 223]. By regarding autobiographical memory as a “mental natural kind” [224], it becomes feasible to conceptualize its evolution within the larger framework of event memory.
The creation, preservation, and retrieval of event memories in adults relies on multi-component brain networks encompassing temporal and cortical regions [225]. Following an event, numerous sensory and motor signals from numerous brain regions converge on parahippocampal components in the temporal lobes, including the entorhinal cortex. The hippocampus, a structure within the temporal lobe, consolidates these components to form a lasting, cohesive memory trace [226]. Cortical regions function as long-term memory storage, whereas prefrontal regions are involved in retrieval following a delay. Consequently, long-term memory retrieval necessitates the involvement of many cortical regions, such as the prefrontal cortex and temporal structures, along with preserved interconnections between them [225, 227].
Elements within the temporal-cortical network that are involved in declarative memory are known to mature early in humans [228]. For example, the majority of cells constituting the hippocampus are generated early in gestation and are functionally integrated by the conclusion of the prenatal development. In most of the hippocampus, neurons begin forming connections early in development, reaching the adult level of synapse quantity and density by around six months after birth. Nonetheless, the dentate gyrus of the hippocampus, an essential conduit between the cortex and the hippocampus, does not reach its complete cellular complement until postnatally [229]. Hippocampal volume and myelination continue to expand beyond the preschool years and into adolescence [230].
The association areas also experience an extended developmental trajectory [231]. All six cortical layers become discernible only in the seventh month of fetal development [231]. The synaptic density in the prefrontal cortex increases significantly from 8 months postnatally, reaching its zenith between 15 and 24 months [232]. Pruning commences in late childhood, with final adult levels attained in late adolescence or early adulthood [233]. During the intervening years, alterations occur in cell size and in the lengths and branching patterns of dendrites [234]. Changes in the prefrontal cortex, including myelination, continue throughout adolescence. Adult levels of specific neurotransmitters are not achieved until the second and third decades of life [235].
Developmental alterations in the declarative memory network affect the efficacy and efficiency of information encoding and stabilization for long-term storage, the reliability of storage, and the ease of retrieval [234, 235]. Environmental supports in the first year of life influence the formation and stabilization of neural circuits that underlie learning and memory, affecting the efficiency of information encoding, consolidation, and later retrieval [236]. Delayed maturation of the prefrontal cortex can influence every stage of the memory retrieval process [237], and delayed maturation of the dentate gyrus may result in suboptimal consolidation of new knowledge. If encoding is disturbed, consolidation is diminished. With impaired consolidation, diminished levels of information are available for consolidation, and hence less information will be retained [238]. With less information retained, a corresponding reduction in retrieval capacity will occur [238]. Disparities in the volume of retrievable information will become increasingly evident over time, as interference and decay exacerbate the deterioration of the already compromised trace [234].
The development of memory in infants is a multifaceted process that evolves significantly over time. An infant’s capacity to encode, consolidate, and retrieve memories is intricately linked to the maturation of their neural structures [226, 227].
Early in development, the temporal structures, particularly the hippocampus, begin to form and establish connections. This early formation is crucial for the initial stages of memory processing [239]. However, the dentate gyrus, a part of the hippocampus critical for linking the cortex with the hippocampal region, does not reach its full complement of cells until after birth [240]. This delayed development implies that infants may have limited capacity for memory consolidation in the first few months of life.
As infants grow, the hippocampus continues to develop, with significant increases in volume and myelination extending into adolescence. These changes enhance the efficiency of memory processes over time. The association areas of the brain play a role in integrating sensory information. They also develop gradually, with all six cortical layers becoming distinct by the seventh prenatal month, and synaptic density peaking postnatally [241].
The prefrontal cortex area of the brain is associated with the control of advanced cognitive functions, including planning and decision-making, and undergoes a prolonged period of maturation [242]. The synaptic density in this region increases significantly during the first two years of life, but pruning to adult levels does not begin until late childhood. This extended development period affects the overall efficacy and reliability of memory encoding and retrieval processes.
The protracted maturation of these neural structures means that the memory capabilities of infants improve progressively. Early memories are often less vivid and stable due to ongoing development of the brain’s memory-related regions. As the brain matures, these processes become more efficient, leading to more reliable and detailed memory formation [243].
The declarative memory network influences the effectiveness of information encoding and stabilization necessary for long-term retention, memory storage reliability, and the ease of retrieval [243, 244, 245]. The delayed maturation of the prefrontal cortex influences every stage of a memory trace, from initial encoding through to consolidation and retrieval. Delayed maturation of the dentate gyrus is consequential, since it may result in suboptimal consolidation of new knowledge.
When information processing is inefficient, the consequences for memory are significant. If the encoding process is impaired, the amount of information that can be consolidated and stored is greatly reduced. As a result, less information is saved, and therefore, less will be available for retrieval in the future. Over time, the gap in the amount of retrievable information will grow, as processes like decay and interference further weaken the already degraded memory traces.
Ineffective early-stage memory encoding in infancy can have consequences on cognitive development and later life. Several key points are outlined here. Impaired Cognitive Development: ineffective memory encoding can lead to difficulties in learning and retaining new information, which can affect overall cognitive development [246]. Attention deficits: poor memory encoding can affect the development of attention systems, leading to issues with sustained and selective attention [247]. Language development: memory plays a crucial role in language acquisition, and ineffective encoding can result in delays or difficulties in language development [248]. Emotional and social development: early memory encoding is linked to the development of emotional regulation and social skills [249], and ineffective encoding can lead to challenges in these areas. Childhood amnesia: ineffective early-stage memory encoding can contribute to childhood amnesia, where individuals have little to no recollection of their early childhood experiences [250].
Prefrontal cortical developmental changes are likely to influence age-related variations in the efficiency of information encoding in young infants. Three-month-old newborns require a longer duration to encode a stimulus compared to six-month-old infants [251]. Researchers have employed event-related potentials (ERPs) to assess changes in encoding processes associated with development [252, 253]. ERPs are electrical oscillations in the brain that occur in response to a specific stimulus. These potentials are measured using EEG and are used to assess changes in brain activity associated with various types of stimuli, including familiar and novel stimuli. ERPs are valuable in the study of cognitive processes, including memory encoding and recognition, as they provide insights into how the brain processes and responds to different stimuli. Bauer and colleagues [254] conducted a longitudinal study to explore the connection between encoding and long-term recall. The ERPs of infants were recorded while they viewed photographs of props from multi-step sequences they had recently encountered, mixed with images of props from unfamiliar sequences. The response amplitudes to stimuli encoded at 10 months were significantly greater than those observed in the same infants at 9 months, but no differences were observed in responses to novel stimuli. The disparities in encoding were associated with variations in recall. One month after each ERP recording, long-term retention of the sequences was evaluated by imitation. The infants demonstrated better recall of the sequences presented at 10 months compared to those shown at 9 months.
Age-related variations in encoding persist beyond the age of one year. Compared to 15-month-old infants, 12-month-olds require additional trials to master multi-step events to a level that signifies complete encoding of the data. Consequently, 15-month-olds exhibit slower attainment of the criterion compared to 18-month-olds [255]. During preschool years, age-related alterations in encoding are indicated by variations in short-term memory “span”, as assessed by tests measuring the recall of numbers or words. At the age of two years, children can typically retain around two units of information. By the time they reach the age of five to seven, their memory capacity increases to four and five units, respectively.
Memory consolidation during infancy is a critical process that transforms recently acquired information into durable memories [256]. This process is significantly influenced by sleep, which plays a vital role in enhancing memory retention and overall cognitive development. With memory consolidation, the brain reprocesses and strengthens newly learned information during sleep, making it more stable and enduring [257]. Short-term memories must be transformed into long-term memories [256].
Sleep, especially naps, enhances memory retention in infants. Infants who take a prolonged nap shortly after learning new behaviors demonstrate significantly better memory performance compared to those who do not nap [257]. Sleep, therefore, facilitates the consolidation of memories acquired from various learning experiences. For example, studies have shown that 6- and 12-month-old infants who napped after learning retained their memories better than those who remained awake [258]. The timing of sleep relative to learning is crucial. Infants who sleep shortly after encoding new information exhibit improved retention of that information over time [258]. As infants grow, the mechanisms of memory encoding and consolidation evolve. In early childhood, processes such as encoding and consolidation contribute significantly to age-related differences in long-term declarative memory [188]. The ability to remember specific events improves with age, as evidenced by developmental changes in the capacity of infants to imitate learned actions over time [211].
Memory system development in infants is associated with maturation of brain structures such as the hippocampus, which is crucial for memory processing. Maturation of these structures is believed to enhance the efficiency of memory storage and contribute to improved cognitive outcomes as children transition out of frequent napping patterns [259].
Additionally, brain astrocytic microdomains are involved in memory consolidation by facilitating protein recycling necessary for synaptic strengthening, which is essential for long-term memory storage [260]. Memory consolidation and storage in infancy are profoundly affected by sleep and the maturation of specific brain regions. Sleep not only aids in the retention of newly learned information, but also plays a critical role in the overall development of cognitive abilities during early childhood. Understanding these processes is vital for promoting effective learning strategies in infancy and early childhood education settings.
Age-related encoding differences are not the only cause of age-related declarative memory patterns. Despite statistically controlling encoding levels [261], matching [193] or aligning children of varying ages to the same learning criterion [201], older children exhibit superior long-term recall compared to younger children. This indicates that younger children remain susceptible to forgetting, even after a memory has been effectively stored. Increased sensitivity presumably arises from the relative underdevelopment of the structures and connections necessary for consolidation of long-term memories.
Evidence suggests that consolidation and storage mechanisms contribute to variability in long-term memory retention throughout the first year of life. Bauer and colleagues [262] utilized ERPs as well as behavioral measures to examine (a), the effects of instantaneous ERPs on event encoding, (b) consolidation and storage examined by ERPs after one week, and (c) long-term recall by postponed imitation one month later. Collectively, the infants demonstrated encoding in response to familiar and novel stimuli, even though there was a disparity in recall, which correlated with differences in consolidation and storage.
Infants who were unable to recall the events after one month also failed to recognize the familiar props after one week. Conversely, those who remembered the events after one month showed effective consolidation and retention after one week. The two groups of infants showed no differences in encoding, and individual differences in encoding did not significantly predict long-term memory. However, effective consolidation and storage over one week accounted for 28% of the variance in recall one month later. The mechanisms of consolidation and storage continue to influence individual differences in memory during the second year of life. For infants aged 20 months, the information retained 48 h after exposure to events explained 25% of the variance in recall after one month [263].
Memory retrieval in infants is influenced by several factors, including cognitive development and the biological basis of memory processes. Research indicates that infants can recognize previously encountered stimuli, suggesting that basic forms of memory retrieval are present even at a very young age [264]. By around 6 to 7 months, infants demonstrate the ability for recall memory, although specific experiences from infancy may not be retrievable in adulthood due to phenomena like infantile amnesia [265].
Attention and memory system interaction is crucial for effective memory retrieval. The attention of infants can affect how they process and store information, which in turn influences their ability to retrieve memories later on. Studies suggest that understanding these interactions can provide insights into the cognitive development of infants and how they learn from their environments [264]. As infants grow, they develop cognitive control over their thoughts and actions, which plays a significant role in retrieval processes. This development transitions from reactive forms of control, where infants retrieve information as needed, to more proactive forms, where they maintain relevant information in anticipation of its use [266].
The environment also plays a critical role in memory retrieval. Cues that are present during the encoding of memories can facilitate retrieval later on. For instance, familiarity with a specific environment can enhance performance when recalling information learned in that context [265]. Memory retrieval in infancy is a complex process that involves cognitive development, attention, environmental cues, and the maturation of brain structures responsible for memory. Understanding these factors provides a clearer picture of how infants learn and remember, highlighting the importance of early experiences in shaping cognitive abilities.
Long-term memory storage retrieval relies heavily on effective functioning of the prefrontal cortex, a brain region that experiences an extended developmental trajectory. Consequently, retrieval processes have historically been seen as the primary contributor to developmental disparities in long-term recollection [267, 268]. Nonetheless, there is scant data available to assess the role of retrieval processes, as most studies present alternate potential sources of age-related discrepancies. For example, without assessing the contributions of early-stage encoding and consolidation processes, it is inappropriate to attribute age-related differences in long-term recall primarily to retrieval-related processes. Furthermore, relying only on one recall test makes it difficult to determine whether a memory is intact but inaccessible because of the cues provided (retrieval failure), or if the memory trace has been degraded (consolidation/storage failure).
Bauer and Hertsgaard [200] evaluated children aged 13-, 16-, and 20-months across various delays, ranging from 1 to 12 months. To eliminate encoding processes as a potential cause of developmental differences in long-term recall, the encoding levels of 13- and 16-month-old infants, and 16- and 20-month-old infants, were matched. Additionally, the children underwent two recall trials and were assessed for relearning to examine potential test-induced improvements in retrieval. Despite these measures, the younger children retained less information than the older children, and showed lower levels of relearning. These findings strongly suggest that consolidation and storage, rather than retrieval processes, are the main contributors to developmental changes.
Brainerd and colleagues [171, 269] reached similar conclusions for preschool-aged and older children. Their research investigated encoding discrepancies as a factor in age-related effects by ensuring that participants achieved a criterion level of learning before introducing a delay. Numerous test trials were conducted to evaluate the roles of storage and retrieval failure. A consistent observation was that the majority of age-related variation in children’s recall was due to memory failures during consolidation and/or storage, rather than retrieval. Furthermore, the findings suggested no variation in retrieval failure rates during childhood, despite reductions in storage failure rates. These results indicate that consolidation and storage processes remain significant sources of developmental change during this period.
The assertion that processes like encoding and consolidation significantly influence long-term recall does not imply that developmental changes in the prefrontal cortex are irrelevant to memory formation during infancy and childhood. Rather, the function of developmental alterations in the prefrontal cortex is likely to differ from prior assumptions. The significant impact of the development of prefrontal structures mostly pertains to processes that precede retrieval, rather than the retrieval processes themselves. Encoding and consolidation are inter-related processes involving cortical and medial temporal regions. Consequently, alterations in both these regions are significant. Furthermore, the definitive storage locations for long-term memories are the association cortices. The prefrontal cortex is believed to have a crucial role in the retention of information on the spatial and temporal aspects of events and experiences, which are the distinguishing characteristics of individual episodes. Consequently, developmental alterations in the prefrontal cortex may primarily facilitate more effective encoding, consolidation, and storage, with a secondary contribution being to enhance retrieval processes.
In conclusion, the early years of life are crucial for brain development and cognitive growth. During this period, the foundations for learning and memory are established through the intricate interplay of various brain structures. We have emphasized the importance of understanding the neuroanatomy of infant learning and retention to elucidate how these processes evolve and contribute to lifelong cognitive capabilities.
We also reviewed the complex processes of brain development, learning, and memory in the fetus and during the first two years of life postpartum. We have highlighted that neural connections and key brain structures start to form during the fetal stage and continue to develop after birth. This paper discusses how the fetus, infants, and toddlers absorb stimuli from their environment and develop learning and memory capabilities. We have also reviewed recent research findings, presented insights into the development of learning and memory in the fetus and infants, and compared changes in learning and memory with EEG findings measured in early childhood. Overall, we underscore the significance of early experiences in shaping neural pathways and cognitive functions, and the interplay between genetic and environmental influences in shaping developmental outcomes.
GL: Conceptualization, Validation, Investigation, Resources, Writing-Original Draft, Writing-Review and Editing, Visualization, Supervision, Project Administration; RA: Conceptualization, Validation, Investigation, Writing-Original Draft, Writing-Review and Editing, Visualization. AD’A: Conceptualization, Writing-Original Draft, Writing-Review and Editing, Supervision. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest. Gerry Leisman is serving as one of the Editorial Board members of this journal. We declare that Gerry Leisman had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Bettina Platt.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






