1 Laboratory of Pain and Analgesia, Department of Pharmacology, Institute of Biological Sciences, Federal University of Minas Gerais, 31270901 Belo Horizonte, Minas Gerais, Brazil
Abstract
The overall pain experience results from the balance between the nociceptive pathway and the body’s endogenous modulation of nociception. The interaction of these systems reduces nociception. Therefore, this study aimed to evaluate how the opioid and dopaminergic systems collaborate to inhibit pain at the peripheral level.
Swiss mice (30–40 g) had their pain sensitivity increased through paw administration of the prostaglandin E2 (2 μg). They then received opioid and dopaminergic receptor antagonists and agonists, along with an inhibitor of endogenous opioid peptide degradation and a dopamine (DA) reuptake inhibitor. The nociceptive threshold was measured using the paw withdrawal test. Groups were compared using one-way analysis of variance (ANOVA), with p < 0.05 considered significant.
The nonselective opioid receptor antagonist naloxone (50 μg/paw) and the selective κ nor-BNI (200 μg/paw; nor-Binaltorphimine) and δ naltrindole (60 μg/paw) receptor antagonists reversed the antinociception caused by peripheral administration of DA (80 ng/paw), but not the μ-opioid receptor antagonist CTOP (20 μg/paw; D-Phe-Cys-Tyr-D-Trp-Orn-Thr-Pen-Thr-NH2). The antinociception induced by a submaximal dose of DA (5 ng/paw) was enhanced by bestatin (400 μg/paw), an inhibitor of endogenous opioid peptide degradation. Conversely, peripheral antinociception from submaximal doses of the μ-, δ-, and κ-opioid agonists DAMGO (0.25 μg/paw; [D-Ala2, N-Me-Phe4, Gly5-ol]-Enkephalin), SNC 80 (5 μg/paw; (+)-4-[(αR)-α-((2S,5R)-4-Allyl-2,5-dimethyl-1-piperazinyl)-3-methoxybenzyl]-N,N-diethylbenzamide), and bremazocine (200 ng/paw) was increased by the DA reuptake inhibitor GBR 12783 (16 μg/paw; 1-(2-Diphenylmethoxyethyl)-4-(3-phenyl-2-propenyl)-piperazine). Furthermore, the antinociception from these agonists’ maximum doses was reversed by dopaminergic D2 (remoxipride, 4 μg/paw) and D3 (U99194, 16 μg/paw; 2,3-Dihydro-5,6-dimethoxy-N, N-dipropyl-1H-inden-2-amine) receptor antagonists, but not by the D4 (L-745, 870, 16 μg/paw; 3-(4-[4-Chlorophenyl]piperazin-1-yl)-methyl-1H-pyrrolo[2,3-b]pyridine trihydrochloride) receptor antagonist.
Overall, the data suggest that opioid-mediated antinociception depends on the activation of the dopaminergic system. This demonstrates that pain modulation can be enhanced through the interaction of these systems. Controlling pain at a peripheral level by activating endogenous pathways could be a promising approach to pain management.
Keywords
- peripheral antinociception
- dopamine
- opioid
- pain modulation
The opioid system is a key pain-relief mechanism that functions by inhibiting
pain signals. It involves three receptor types: µ,
Although opioid analgesics are some of the most powerful medications for treating severe pain, their long-term use can lead to analgesic tolerance, addictive behaviors, and other adverse effects such as respiratory depression and constipation [9, 10, 11]. Therefore, it is essential to find alternative drugs that provide effective pain relief with fewer side effects. A significant number of pre- and clinical investigations highlight the role of the dopaminergic system in modulating nociception. Dysfunction of dopaminergic neurotransmission may be associated with heightened pain sensitivity. Positron emission tomography examinations used to evaluate patients with fibromyalgia syndrome revealed decreased dopamine (DA) synthesis and release in presynaptic neurons [12, 13]. Additionally, clinical research indicates that administering levodopa, the precursor of DA, relieves pain in individuals with Parkinson’s disease [14, 15]. In preclinical trials, injecting levodopa into the intrathecal space and specific regions of the central nervous system produced an antinociceptive effect [16, 17]. Peripherally, intraplantar administration of DA reversed Prostaglandin E2-induced hyperalgesia in mice [18].
Recent research indicates that higher endogenous dopamine levels improve the cannabinoid agonist 2-AG (2-Arachidonoylglycerol)-induced peripheral antinociception. It is also important to understand that the interaction between the dopaminergic and endocannabinoid systems plays a key role in the inhibitory regulation of nociception at the peripheral level [19]. Similar to the endocannabinoid system, the opioid system appears to have an additive or synergistic effect with the dopaminergic system in the relief of pain. Activation of µ-opioid receptors in the periaqueductal gray (PAG) depends on the activation of dopaminergic receptors [20]. These receptors are widely found in the ventrolateral PAG and contribute to antinociception caused by systemically administered morphine [21, 22].
This study explores whether DA-induced peripheral antinociception involves engaging the opioid system and identifies which opioid agonists and receptors may trigger it. It also investigates if the dopaminergic system plays a role in opioid-mediated peripheral antinociception. The results indicate a possible approach to improve the effectiveness of lower opioid doses in pain management, reducing side effects, while also clarifying the peripheral mechanisms of dopaminergic antinociception.
Male Swiss mice (30–40 grams, 10–12 weeks old) from the Bioterism Center
(CEBIO) of the Federal University of Minas Gerais (UFMG) were used. The study
involved 340 animals, with five mice in each group (sample size of 5). The
animals were housed in open-top cages (OTCs) in groups of five per cage, using
plastic cages measuring 41
Hyperalgesic: Prostaglandin E2 (PGE2; Sigma, P-0409, St. Louis, MO,
USA; 2 µg/paw) dissolved in 10% ethanol in saline (NaCl 0.9%);
opioid system drugs: Naloxone (NX; Sigma, N-109; 12.5, 25, and 50
µg/paw) non-selective opioid receptor antagonist, dissolved in
saline; CTOP (D-Phe-Cys-Tyr-D-Trp-Orn-Thr-Pen-Thr-NH2; Tocris, 1578, Bristol, UK;
20 µg/paw) selective µ-opioid receptor antagonist, dissolved
in saline; Naltrindole (NTD; Tocris, 0740; 15, 30, and 60 µg/paw)
selective
The pressure paw withdrawal method was used to determine the nociceptive
threshold [25, 26]. During testing, mice were placed on their ventral side on a
table, with the paw being tested positioned so that its plantar surface rested on
the compressive surface of the analgesimeter (37215, 37216, Ugo-Basile, Gemonio,
VA, Italy). This device applied gradually increasing pressure (grams/second)
until the paw was withdrawn. The pressure (grams) at which the paw withdrew was
defined as the mechanical nociceptive threshold. A cutoff of 150 grams was set to
prevent potential injury. The threshold was calculated by averaging three
consecutive measurements. The
Initially, the nociceptive threshold was measured before administering any drug
(basal) using the paw pressure test. At 0 h, hyperalgesic Prostaglandin E2 (PGE2)
was administered. Literature indicates that PGE2 hyperalgesia peaks at 180
minutes after administration [27]. Therefore, all other drugs were given so their
peak effects aligned with this time, when the nociceptive threshold was measured
again (180 minutes). The analysis focused on the change (
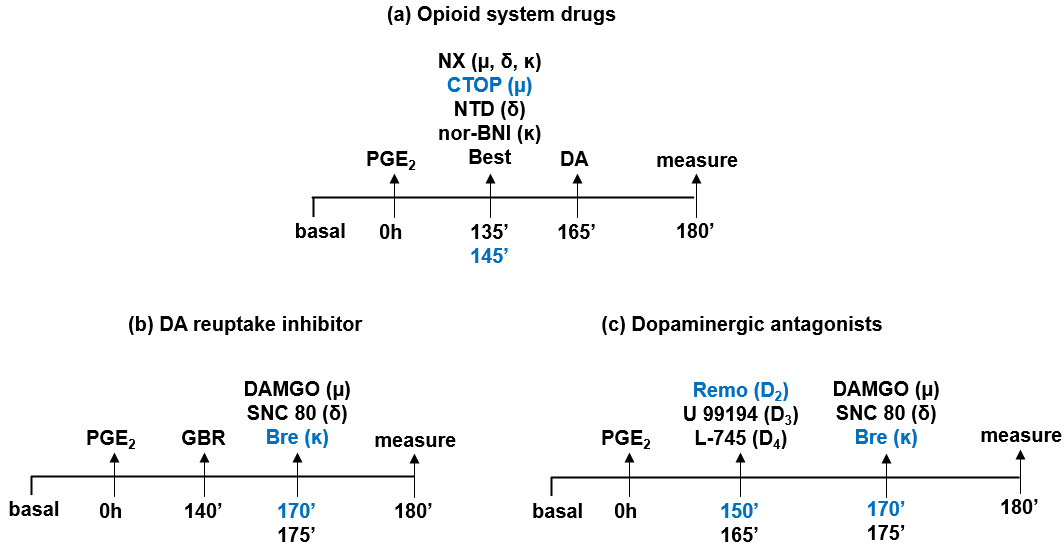 Fig. 1.
Fig. 1.
Timeline of experiments. The baseline nociceptive threshold was measured before administering any drug. All drugs were injected into the right hind paw via intraplantar injection. Prostaglandin E2 (PGE2; 2 µg) was injected into the paw at 0 hours. The final nociceptive threshold was measured after 180 minutes. (a) Naloxone (NX; 12.5, 25, and 50 µg/paw), naltrindole (NTD; 15, 30, and 60 µg/paw), and nor-BNI (50, 100, and 200 µg/paw) were administered along with a maximum dose of dopamine (DA; 80 ng/paw). Bestatin (Best; 400 µg/paw) was administered with a submaximal dose of DA (5 ng/paw). (b) GBR 12783 (16 µg/paw) was administered alongside submaximal doses of DAMGO (0.25 µg/paw), SNC 80 (5 µg/paw), and bremazocine (Bre; 200 ng/paw). (c) Remoxipride (Remo; 4 µg/paw), U 99194 (16 µg/paw), and L-745,870 (16 µg/paw) were administered together with a submaximal dose of DAMGO (1 µg/paw), SNC 80 (20 µg/paw), and Bre (600 ng/paw).
Data were analyzed with Prism software version 8.0.2 (GraphPad, Boston, MA,
USA), and normality was confirmed using the Shapiro-Wilk test. Our primary
outcome variable was continuous; therefore, we compared means between the control
and treated groups using one-way analysis of variance (ANOVA). Pairwise
comparisons were assessed with a Bonferroni post hoc correction for multiple
comparisons, and effects were expressed as mean difference
To assess the role of the opioid system in DA-induced antinociception, animals
were initially pre-sensitized with intraplantar injections of the hyperalgesic
agent Prostaglandin E2 (PGE2; 2 µg/paw). The administration of DA
(80 ng/paw) reversed hyperalgesia and restored the nociceptive threshold to
baseline levels. DA-mediated antinociception was reversed by naloxone (NX; 12.5,
25, and 50 µg/paw), a non-selective opioid receptor antagonist, in a
dose-dependent manner (Fig. 2a). Similarly, naltrindole (NTD; 15, 30, and 60
µg/paw), a selective
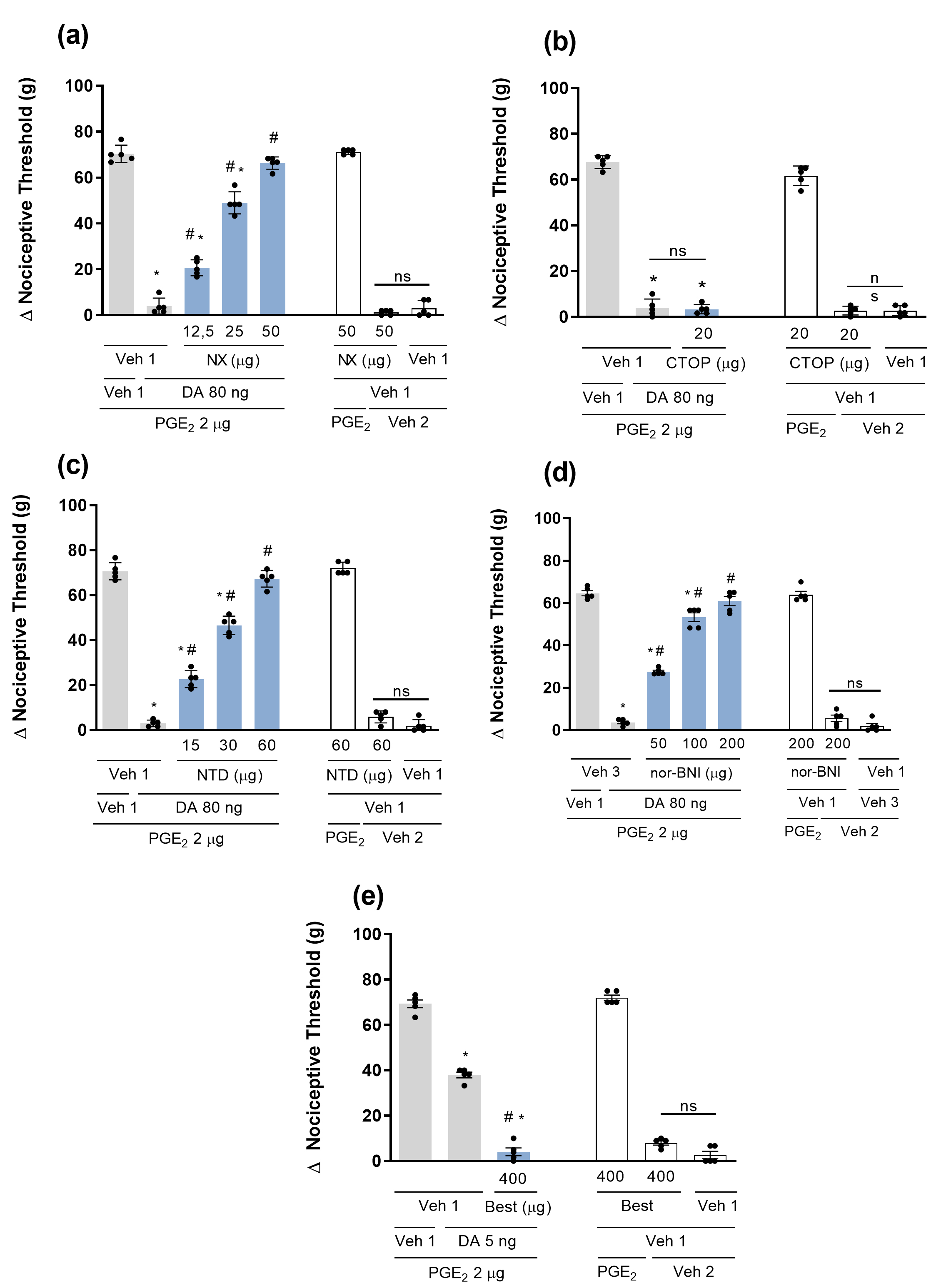 Fig. 2.
Fig. 2.
Participation of the opioid system in dopamine-mediated
peripheral antinociception. Effect of both nonselective and selective
antagonists for µ,
To investigate the role of endogenous DA in opioid-induced antinociception, it
was used a dopamine reuptake inhibitor GBR 12783. The results show that
PGE2-induced hyperalgesia (2 µg/paw) was partially reversed by
administering submaximal doses of the µ-,
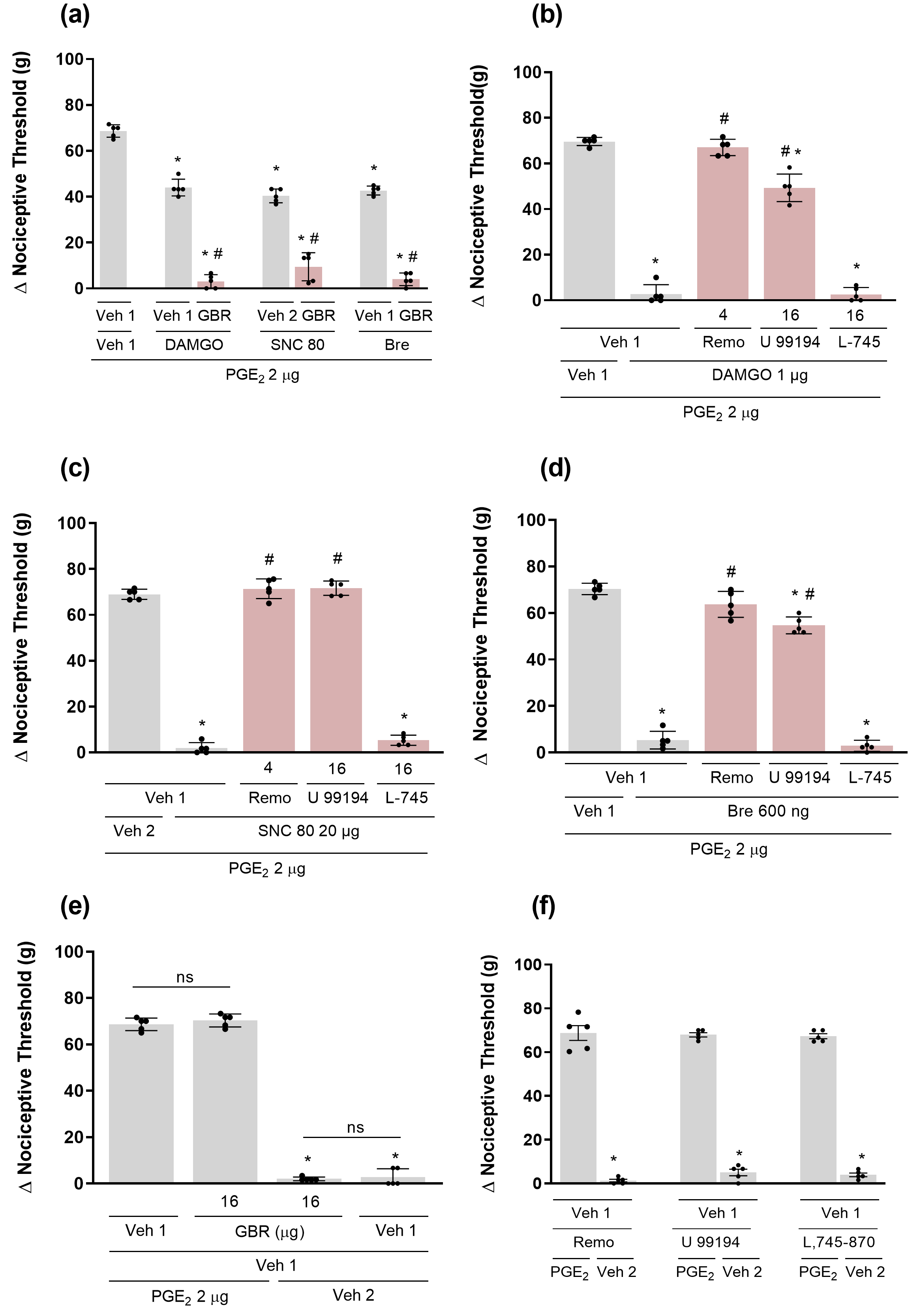 Fig. 3.
Fig. 3.
Involvement of the dopaminergic system in peripheral opioid
effects antinociception. Effect of a dopamine reuptake inhibitor (a) GBR 12783
(16 µg/paw) and dopamine receptor antagonists D2 Remoxipride (Remo; 4
µg/paw), D3 U 99194 (16 µg/paw), and D4 L-745,870 (16
µg/paw) on antinociception mediated by µ,
This study used a pharmacological approach to clarify how DA and opioids promote peripheral antinociception by examining how these two systems interact in suppressing pain at the peripheral level. The increase in nociceptive response was caused by PGE2, which sensitizes primary afferent neurons and leads to hyperalgesia to mechanical stimulation [27]. Previous studies have shown that DA blocks the effects of PGE2 in this model by activating D2 family receptors [18]. In addition to the classic roles assigned to the dopaminergic system [28], scientific literature increasingly emphasizes DA’s role in pain modulation [5, 29, 30]. Meanwhile, opioid drugs are well-established in medicine for treating severe pain. Although highly effective, these drugs are linked to analgesic tolerance, addiction, and physical dependence [11]. Therefore, developing new drugs with good analgesic effects and fewer side effects, or drugs that can enhance existing analgesics’ effectiveness while reducing adverse effects, is a promising strategy.
Although dopaminergic analgesia is well documented, its mechanisms are not fully understood. Therefore, our study aimed to examine the influence of the opioid system on DA-mediated antinociception at the peripheral level. The interaction between the opioid and dopaminergic systems in producing analgesia in the central nervous system has already been described, as these systems are closely connected anatomically [31]. The periaqueductal gray (PAG) plays a key role in nociception, often linked to opioid activity [32]. Dopaminergic neurons in this region may be involved in opioid analgesia [33]. Additionally, antinociception caused by activating D2 receptors in the PAG requires the activation of opioid receptors [20].
At the peripheral level, we observed that antinociception mediated by DA
administration was reversed by antagonism of
Another important finding from our study was that intermediate antinociception
caused by DA was enhanced by bestatin, a drug that prevents the breakdown of
endogenous opioids. Since different opioid peptides prefer specific receptors, we
can infer from our results that, in our model, the opioids involved in
dopaminergic antinociception are likely enkephalins and dynorphins, because these
peptides can activate
In the next part of our study, we explored how endogenous DA influences opioid
antinociception. The opioid system operates through three receptor types:
µ,
The results of this study, along with the literature review, highlight the critical role of the dopaminergic system in pain modulation, both centrally and peripherally. Additionally, considering the additive and/or synergistic interactions between D2 receptors and opioid receptors, D2 agonists may serve as adjuvants to enhance analgesic effects and decrease the adverse effects of opioids. Overall, these findings suggest that targeting the dopaminergic system could offer a fresh approach for more effective management of pain. This study has some limitations. First, only the mechanical withdrawal test was employed, and additional methods such as the thermal test were not included. Second, only male animals were used in the present experiments. Potential sex differences, as well as the inclusion of additional behavioral tests, will be considered in future studies.
This study showed how the body’s own pain relief systems work together to influence the blocking of pain signals at the peripheral site. Our data suggest that the opioid system is involved in the peripheral pain relief caused by DA, and that the dopaminergic system contributes to the analgesia produced by opioids. Understanding these mechanisms provides a promising path for creating new treatments that could be more effective than traditional options.
The data that support the findings of this study are available from the corresponding author upon reasonable request.
BFGQ: Investigation, Methodology, Writing – original draft; WCPB: Investigation, Methodology; ALI: Investigation, Methodology; FCSF: Investigation, Methodology; TRLR: Investigation, Formal analysis, Resources; IDGD: Investigation, Resources, Formal analysis, Conceptualization, Writing review & editing, Supervision. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The Animal Experimentation Ethics Committee at the Federal University of Minas Gerais approved this study under protocol numbers 196/2018 and 129/2021, and followed ARRIVE 2.0 guidelines. Use Committee and the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Not applicable.
This work was supported by grants from Comissão de Aperfeiçoamento de Pessoal do Nível Superior (CAPES), Conselho Nacional de Pesquisa (CNPq), and Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.



