1 Department of Anesthesiology, Beijing Tongren Hospital, Capital Medical University, 100730 Beijing, China
Abstract
Hypoxic/ischemic brain injury remains a major clinical challenge, yet the cellular mechanisms linking oxygen-glucose deprivation/reperfusion (OGD/R) to opioid receptor regulation in human neurons are still not fully understood. The trafficking of μ-opioid receptors (MOR) and κ-opioid receptors (KOR) is a key regulator of neuronal survival under stress. Most studies to date in this field have employed rodent models. However, given the molecular and physiological differences between rodents and humans, this study employed human induced pluripotent stem cell (iPSC)-derived neurons to investigate opioid receptor trafficking during OGD/R, as well as the neuroprotective effects of Herkinorin.
Human iPSC-derived neurons were subjected to 2 h of OGD followed by 24 h of reoxygenation. Cells were treated with Herkinorin (0.1, 0.5, or 1 μM) during OGD/R. Apoptosis was assessed using flow cytometry, while the localization of MOR and KOR in membrane and cytoplasmic fractions was analyzed using western blotting. Western blotting was also used to quantify the expression of apoptosis-related proteins Bcl-2-associated X protein (Bax), B-cell lymphoma 2 (Bcl-2), and cleaved Caspase-3. Statistical comparisons were performed using one-way ANOVA with Tukey’s post hoc test or non-parametric equivalents.
OGD/R significantly increased neuronal apoptosis, upregulated pro-apoptotic Bax and cleaved Caspase-3, and downregulated anti-apoptotic Bcl-2. These changes were accompanied by altered distribution of MOR, but not KOR localization, specifically decreasing cytoplasmic MOR while maintaining membrane levels. Herkinorin, particularly at 1 μM, induced redistribution of MOR from the plasma membrane to cytoplasm, consistent with receptor internalization; it also significantly reduced apoptosis in a concentration-dependent manner. Furthermore, treatment with Herkinorin reversed the OGD/R-induced molecular changes by decreasing the expression of Bax and cleaved Caspase-3, while increasing that of Bcl-2. KOR trafficking remained largely unchanged under all conditions. Importantly, Herkinorin concentrations above 10 μM reduced neuronal viability, indicating a narrow therapeutic window.
Herkinorin exerts neuroprotective effects in human iPSC-derived neurons subjected to OGD/R, potentially by modulating MOR internalization and influencing mitochondrial-dependent apoptotic pathways. However, its efficacy is restricted to a limited dose range (0.1–1 μM), as higher concentrations are toxic. The receptor subtype-specific trafficking pattern observed in this study underscores the importance of human-relevant models for mechanistic and translational research on opioid receptors.
Keywords
- brain ischemia
- opioid receptors, mu
- opioid receptors, kappa
- receptor internalization
- induced pluripotent stem cells
- neuroprotection
- apoptosis
- Herkinorin
Hypoxic/ischemic brain injury is a major cause of long-term neurological
disability and mortality, with few effective therapies available. A persistent
barrier to the development of treatments is the low success rate for translation
of findings from preclinical studies to clinical therapies. While rodent models
have provided valuable mechanistic insights, single-cell transcriptomic and
spatial mapping studies have demonstrated substantial differences between rodent
and human brains in terms of cortical cell-type composition, gene expression
patterns, and laminar organization [1, 2]. Moreover, cross-species comparisons
reveal that brain-region transcriptomic similarity is particularly limited in
higher-order cognitive areas [2] that may influence injury responses and
pharmacological outcomes. Neurochemical systems also differ markedly between
species. Baseline levels of serotonin (5-HT), norepinephrine (NE), dopamine (DA),
and acetylcholine (ACh) in the prefrontal cortex vary significantly between
rodents and primates [3], potentially altering drug efficacy profiles. At the
gene level, the µ opioid receptor gene (OPRM1) in rodents exhibits
extensive alternative splicing, producing isoforms that differ from those in
humans [4, 5]. These isoform-specific variations can influence ligand binding,
signaling bias, and therapeutic effects, thereby limiting the direct
extrapolation of rodent pharmacology to human contexts. G protein-coupled
receptor (GPCR) signaling adds further complexity, as ligand-directed signaling
bias and
Human induced pluripotent stem cell (iPSC)-derived neurons offer a solution to such translational challenges. These cells retain a human genetic background, express human-specific receptor isoforms, and recapitulate patient-specific molecular phenotypes, showing high concordance with post-mortem brain molecular profiles [4, 10]. Our previous study showed that iPSC-derived neural cells express opioid receptors [11]. Therefore, iPSC-based neuronal models provide a physiologically relevant platform to investigate disease mechanisms and evaluate candidate therapeutics with greater translational potential.
In the current study, we hypothesized that Herkinorin exerts neuroprotective effects in human iPSC-derived neurons subjected to oxygen-glucose deprivation/reperfusion (OGD/R) by modulating MOR trafficking. To test this, we examined receptor trafficking patterns and apoptosis in iPSC-derived neurons under OGD/R, with and without Herkinorin treatment. This approach directly addresses the translational gap by combining a human-specific neuronal model with a mechanistic analysis of biased MOR agonism in hypoxic/ischemic injury.
Human neural stem cells (NSCs) were derived from human iPSC (Cell Inspire
Biotechnology, cat# iN-300, Shenzhen, China) and used between passages 2–4. The
supplier certificate indicated a high-purity NSC population (
Immunofluorescence staining was performed on neurons differentiated from NSCs to
confirm neuronal identity and to visualize opioid receptor expression. Neuronal
To model hypoxic/ischemic cellular damage in vitro, we utilized a
slightly modified version of previously described oxygen-glucose
deprivation/reoxygenation (OGD/R) procedures [12, 13]. Briefly, human
iPSC-neurons were washed twice with pre-equilibrated, glucose-free Dulbecco’s
Modified Eagle’s Medium (DMEM; Gibco, cat. no. 11966025, Thermo Fisher
scientific, Eugene, OR, USA) and then transferred to a modular airtight hypoxia chamber
(Billups-Rothenberg, Del Mar, CA, USA). The chamber was flushed for 15 min at a
constant flow rate (20 L/min) with a certified gas mixture of 95% N₂ and 5%
CO2, yielding an internal oxygen tension of
For reoxygenation, cells were returned to glucose-containing DMEM pre-equilibrated in a normoxic incubator (5% CO2, balance air; ~18–19% O2) and maintained for 24 h. pH stability was monitored using phenol red as an indicator, and the glucose concentration was restored to physiological levels (25 mM) at the start of reoxygenation. The selection of OGD and reoxygenation duration times was based on our previous systematic investigation [14].
Herkinorin (cat.no. 508018, Merck KGaA, Darmstadt, Germany) was dissolved in dimethyl sulfoxide (DMSO) to prepare a 10 mM stock solution. Working dilutions were prepared immediately before use by adding the stock directly into culture medium, ensuring the final DMSO concentration did not exceed 0.1% (v/v) in any group. Herkinorin is light-sensitive and prone to degradation. Stock solutions were therefore prepared in small batches, protected from light, and not stored for extended periods either as dry powder or dissolved in DMSO.
To determine the optimal concentration range, human iPSC-derived neurons at day 21 of differentiation were treated with Herkinorin at final concentrations of 0.05, 0.1, 0.5, 1, 10, and 50 µM for 72 h. Cell viability was assessed using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. The assay was previously validated for these cells by confirming a linear relationship between absorbance and cell number, and by verifying that 0.1% DMSO alone had no measurable effect on viability. Each concentration was tested in triplicate wells in each experiment, and repeated in three independent experiments.
Based on the viability results, concentrations of 0.1, 0.5, and 1 µM were selected for subsequent OGD/R experiments. Neurons were randomly allocated into the following groups: control (vehicle only), OGD/R, OGD/R + Herkinorin 0.1 µM, OGD/R + Herkinorin 0.5 µM, OGD/R + Herkinorin 1 µM.
In the treatment groups, Herkinorin was added 24 h prior to OGD/R induction. The OGD/R procedure consisted of 2 h of oxygen-glucose deprivation followed by 24 h of reoxygenation. After treatment, the apoptosis rate was measured by Annexin V-FITC/PI flow cytometry (BD FACSDiscover™ A8 Cell, BD Biosciences, Milpitas, CA, USA), and MOR and KOR levels in membrane and cytoplasmic fractions were quantified by Western blot.
Following treatment, apoptosis was assessed using Annexin V-FITC/PI staining (V13242, Thermo Fisher Scientific) according to the manufacturer’s instructions. Data were acquired on a BD FACSCanto II cytometer and analyzed with FlowJo software v10.8 (BD Biosciences, Ashland, OR, USA).
Membrane and cytoplasmic fractions were prepared using a Membrane and Cytoplasmic Protein Extraction Kit (Cat.no. KTP3005, Abbkine Scientific, Atlanta, GA, USA) containing protease/phosphatase inhibitors. Fraction purity was verified by Western blot detection of Na⁺/K⁺-ATPase and glyceraldehyde-3-phosphate dehydrogenase (GAPDH). Protein concentrations were determined by BCA assay (Cat. No. A55865, Thermo Fisher Scientific).
Equal amounts of protein (15 µg) were resolved by Novex™ (4–20%) and transferred to PVDF membranes (Cat.no. XP04200BOX, Thermo Fisher Scientific). Membranes were blocked in 5% non-fat milk/TBST for 1 h at room temperature and incubated overnight at 4 °C with the following primary antibodies: anti-Caspase-3 (1:5000, ab32351, Abcam), anti-cleaved Caspase-3 (1:500, ab32042, Abcam), anti-Bax (1:2000, ab18283, Abcam), anti-Bcl-2 (1:1000, ab32124, Abcam), anti-OPRM1 (1:1000, ab10275, Abcam), or anti-OPRK1 (1:1000, ab113533, Abcam). After washing, membranes were incubated with horseradish peroxidase (HRP)-conjugated secondary antibodies for 1 h. Bands were then visualized using ECL (34580, Thermo Fisher Scientific) and imaged on a ChemiDoc XRS+ system (Bio-Rad, Hercules, CA, USA). Densitometry was performed with Image J 1.54g (Wayne Rasband and contributors, National Institutes of Health, Bethesda, MD, USA), and normalized to appropriate loading controls.
Data are expressed as the mean
NSCs were induced to differentiate into mature neurons
for 14–21 days. Morphological changes were monitored by phase-contrast
microscopy, and neuronal identity was confirmed by immunofluorescence staining
(Figs. 1,2). Mature neurons displayed extensive neurite outgrowths and
characteristic soma morphology. Nuclei were counterstained with DAPI (blue), and
neurons exhibited strong immunoreactivity for the pan-neuronal marker
 Fig. 1.
Fig. 1.
Immunofluorescence staining of human iPSC-neurons. (A) Light microscopy image of neurons. (B) Negative control showing fluorescence without primary antibody. (C) DAPI staining highlighting cell nuclei. Scale bar = 100 µm. iPSC, induced pluripotent stem cell; DAPI, 4′,6-diamidino-2-phenylindole.
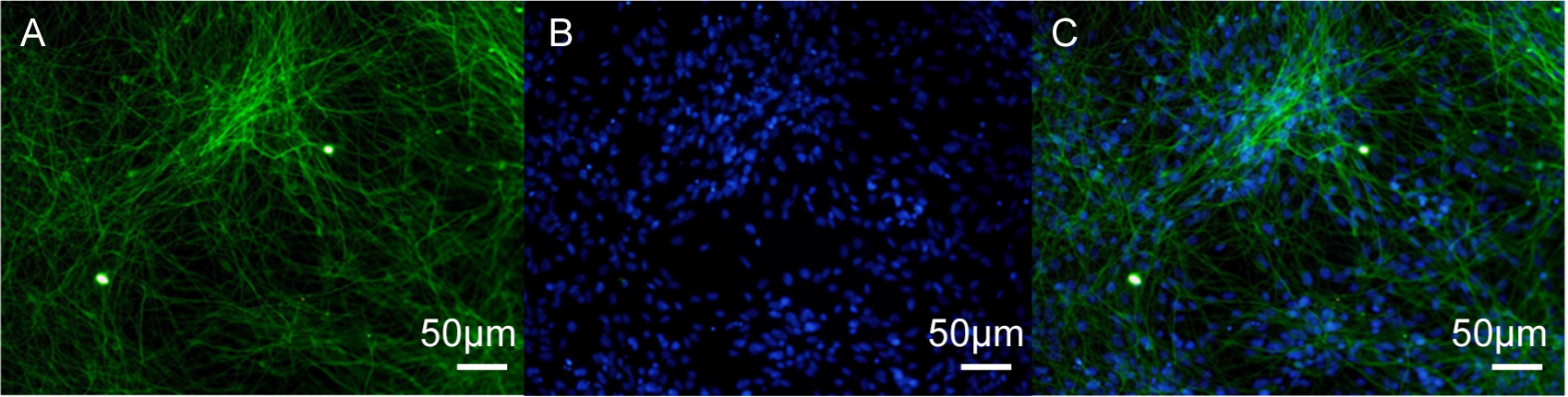 Fig. 2.
Fig. 2.
TUBB3 immunofluorescence staining in human iPSC-neurons. (A)
Neurons stained with the TUBB3 antibody, indicating beta-III tubulin. (B) Cell
nuclei stained with DAPI. (C) Merged image showing co-localization of TUBB3 and
DAPI. Scale bar = 50 µm. TUBB3,
The expression of MOR (µ opioid receptors) and KOR (
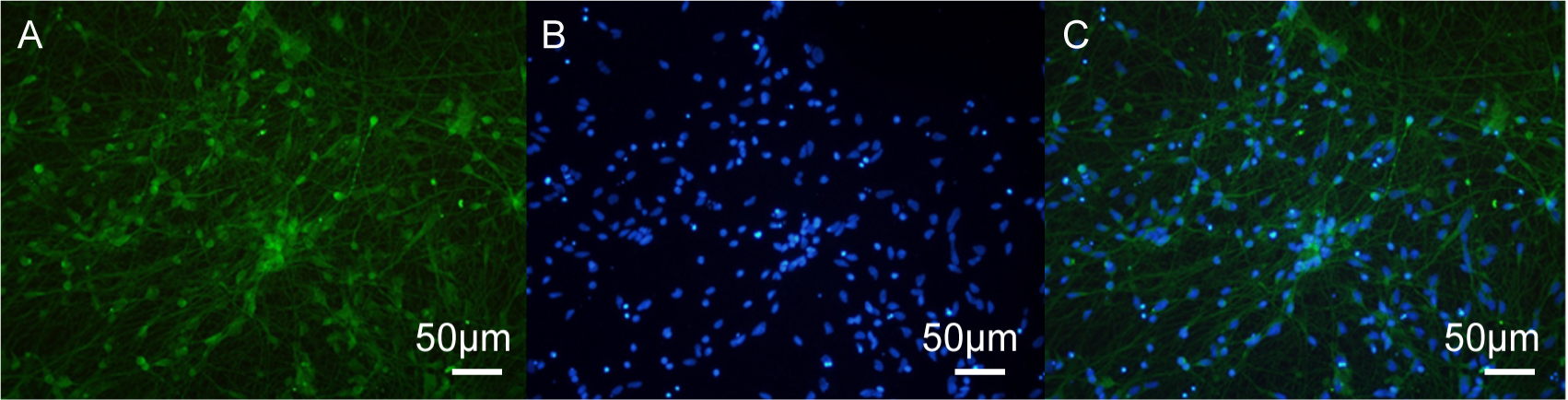 Fig. 3.
Fig. 3.
MOR immunofluorescence staining in human iPSC-neurons. (A) Immunofluorescence image of MOR (µ opioid receptors). (B) DAPI staining of nuclei. (C) Merged image showing co-localization of MOR and DAPI. Scale bar = 50 µm.
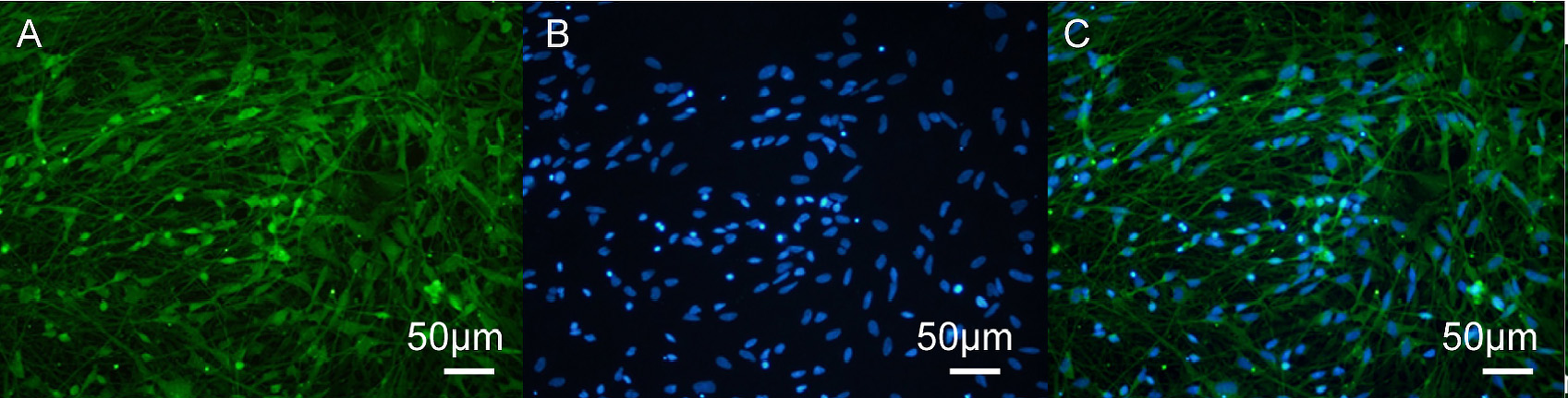 Fig. 4.
Fig. 4.
KOR immunofluorescence staining in human iPSC-neurons. (A)
Immunofluorescence image of KOR (
Cell viability following Herkinorin treatment was assessed by MTT assay (Fig. 5). Human neurons were incubated for 72 h with Herkinorin at 0.01, 0.05, 0.1,
0.5, 1, 10, or 50 µM. Concentrations up to 1 µM did not significantly
reduce cell viability compared to the control, whereas 10 µM and 50
µM caused significant decreases in viability (p
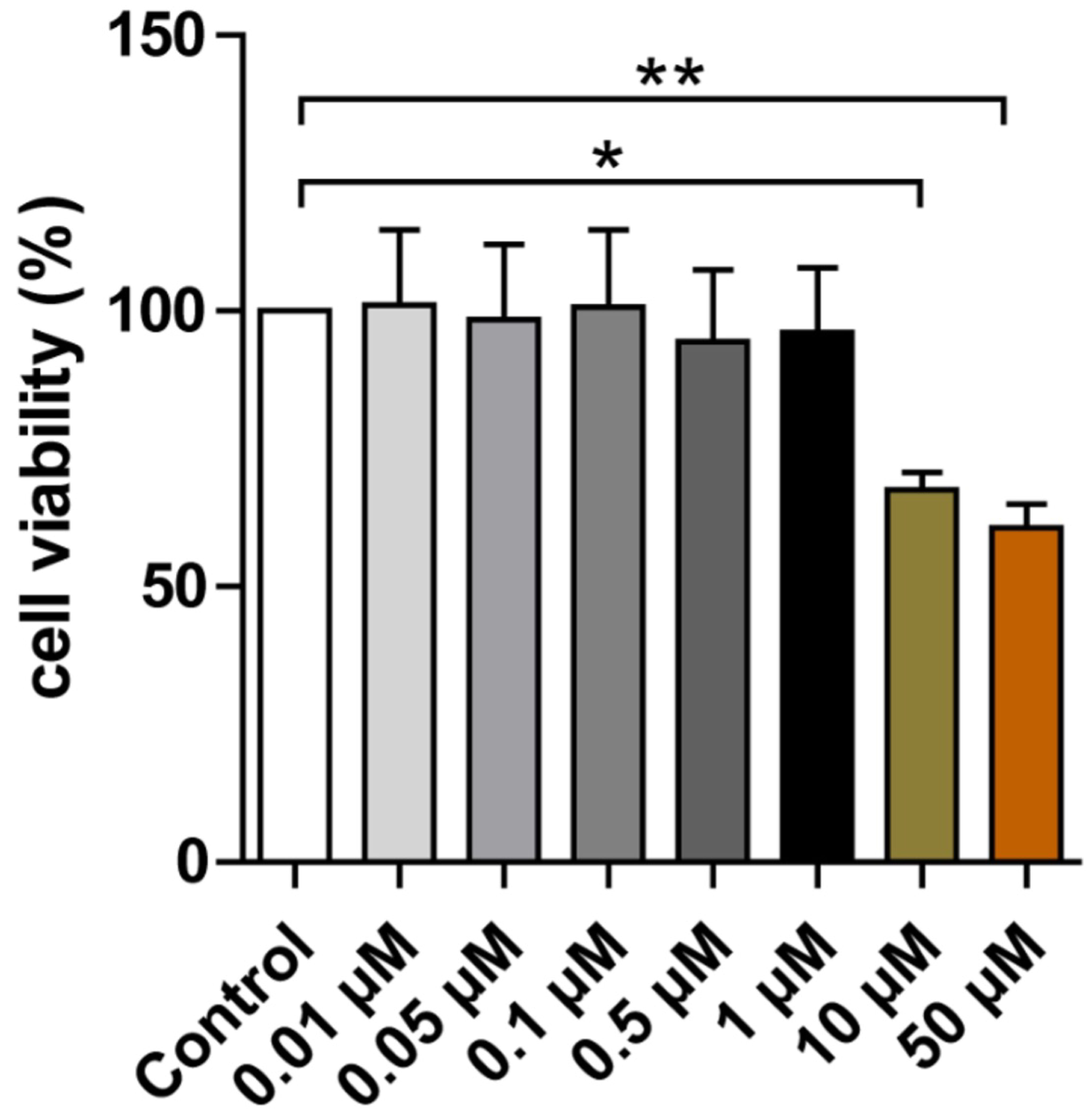 Fig. 5.
Fig. 5.
Effect of Herkinorin on the viability of human iPSC-neurons.
Treatment with 10 µM and 50 µM Herkinorin significantly reduced cell
viability compared with the control group. Data are expressed as the mean
Apoptosis was quantified by Annexin V–FITC/PI flow cytometry. Representative
plots (Fig. 6A–C) and the corresponding quantitative analysis (Fig. 6D) show a
significant increase in apoptosis in the OGD/R group compared to the control
group, which was attenuated by Herkinorin in a concentration-dependent manner
(0.1 µM: 18.2%
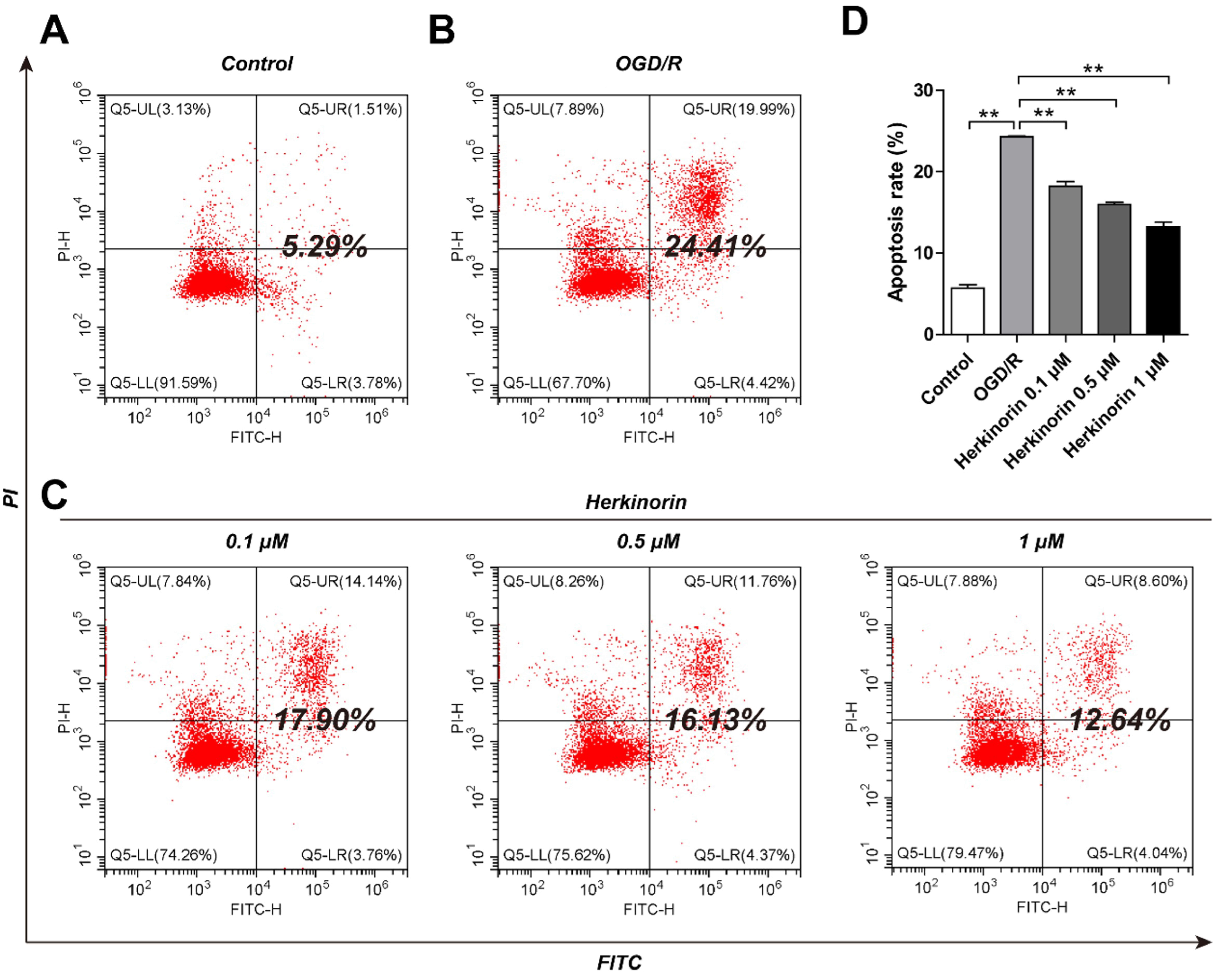 Fig. 6.
Fig. 6.
Effects of Herkinorin on apoptosis in OGD/R-treated human
iPSC-neurons. (A) Control group. (B) OGD/R group showing increased apoptosis
rate. (C,D) Treatment with 0.1, 0.5, or 1 µM Herkinorin significantly
reduced apoptosis induced by OGD/R. Data are expressed as the mean
Western blot analysis demonstrated that OGD/R induced pronounced pro-apoptotic
signaling in human iPSC-neurons (Fig. 7). Specifically, densitometric
quantification revealed that OGD/R significantly increased the expression of Bax
(3.65
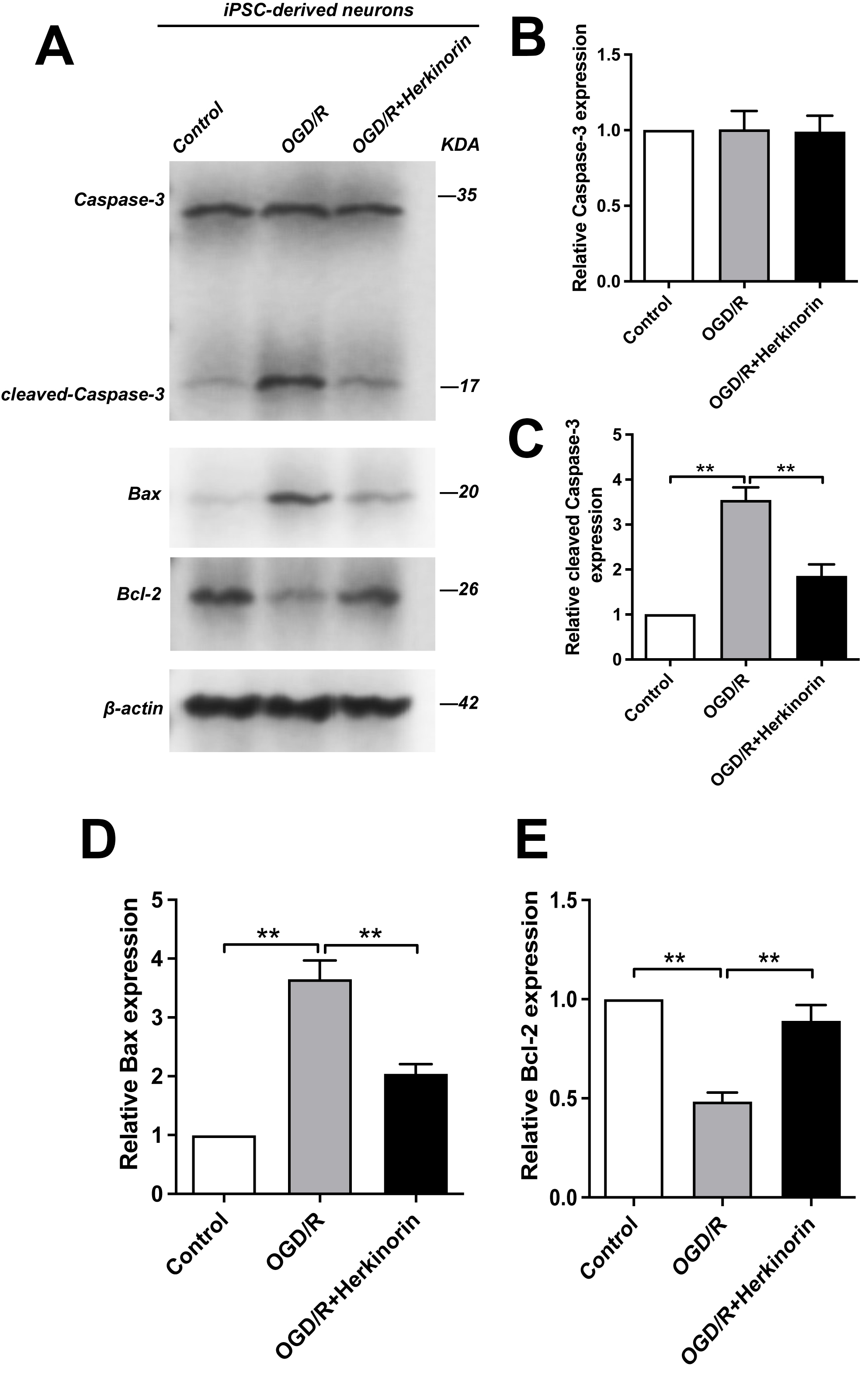 Fig. 7.
Fig. 7.
Effects of Herkinorin on apoptosis-related protein expression
under OGD/R conditions in iPSC-derived neurons. (A) Western blot analysis of
Caspase-3, cleaved Caspase-3, Bax, and Bcl-2 expression in control, OGD/R, and
OGD/R + Herkinorin groups. (B–E) Quantitative analysis of protein expression
presented as relative fold changes: (B) Caspase-3, (C) cleaved Caspase-3, (D)
Bax, and (E) Bcl-2. Data are expressed as the mean
Opioid receptor trafficking was investigated by isolating membrane and
cytoplasmic protein fractions. Fraction purity was validated using Na⁺/K⁺-ATPase
(membrane marker) and GAPDH (cytoplasmic marker). Equal amounts of protein (15
µg) were loaded, and all blots included molecular weight markers. Western
blot analysis revealed significant changes in the distribution of µ opioid
receptor (MOR) in OGD/R group as compared to the Control group (Fig. 8A). In
OGD/R neurons, cytoplasmic levels of MOR decreased significantly (0.49
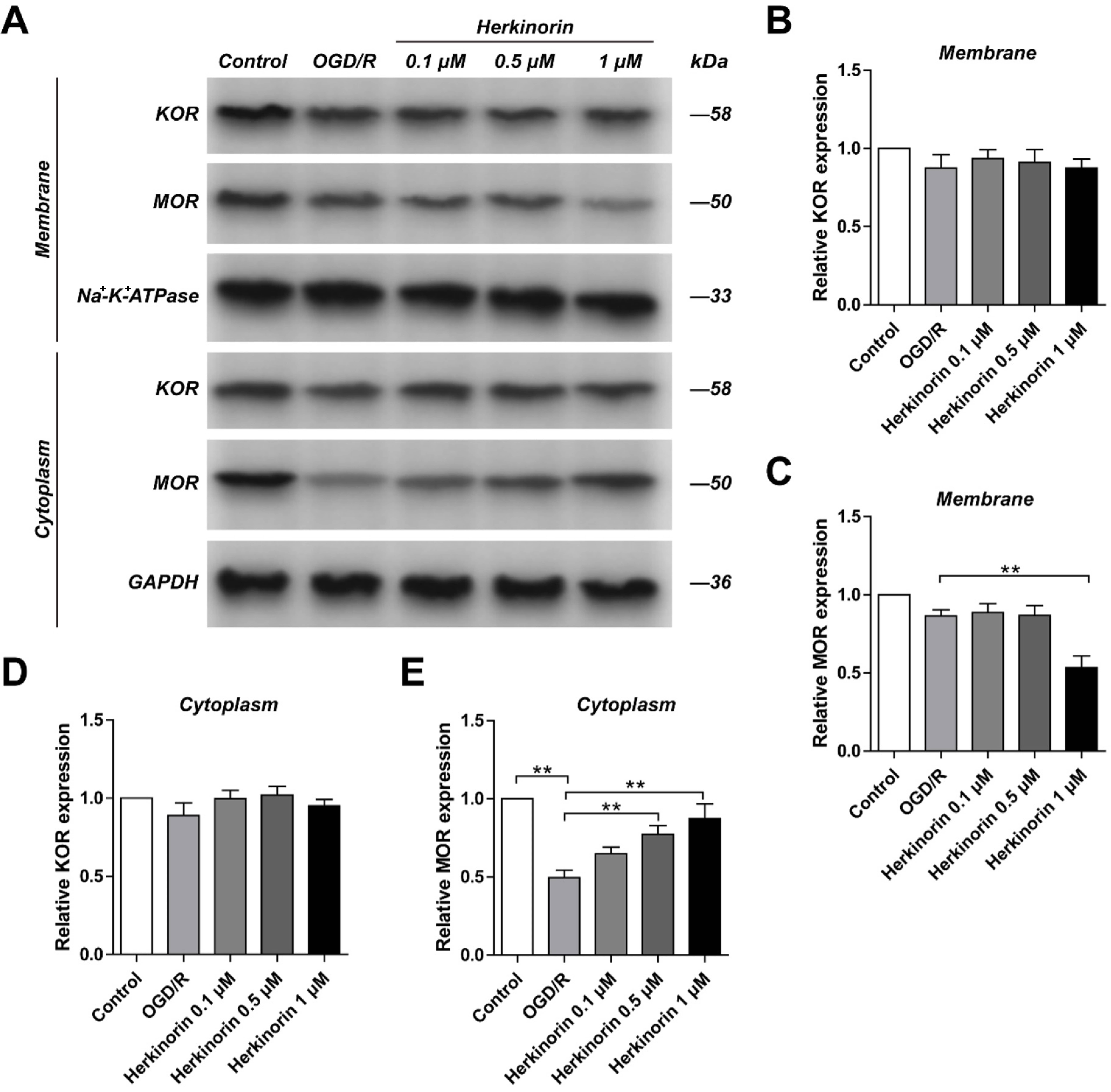 Fig. 8.
Fig. 8.
Effects of OGD/R and Herkinorin on opioid receptor expression in
human iPSC-neurons. (A) Western blot analysis of µ opioid receptor (MOR)
and
Over recent decades, animal models have played a crucial role in neuroscience,
particularly for in vitro and in vivo modeling of disease.
These models have been instrumental in elucidating various pathophysiological
events that might otherwise have remained unknown. However, the biological and
genetic discrepancies between species have been a significant barrier in the
development of effective treatments for many neurological and neurodegenerative
diseases. Approximately 90% of drugs that show efficacy in animal models
ultimately fail in human clinical trials [15]. The utilization of human tissues,
derived mainly from autopsies or fetal specimens, for cell culture models is
limited by the scarcity of these resources and the challenges in scaling up
primary cultures. Human iPSC-based models offer efficient platforms for
elucidating pathological mechanisms and drug screening in inflammatory diseases.
iPSC-derived neurons and glial cells are critical for probing the molecular
mechanisms underlying hypoxic or ischemic injury and for exploring potential
treatments [16, 17]. The integration of human iPSC-derived brain organoids with
cell culture methods to assess the effects of OGD/R is a promising approach for
stroke research [18]. Luminescent neurospheres derived from human iPSCs are
particularly suited for high-throughput screening aimed at discovering new
treatments for acute stroke [19]. Furthermore, brain organoids based on human
iPSCs serve as a novel research platform for anti-stroke drug development, and
the use of iPSC-induced human cortical organoids to model OGD enables detailed
investigation of the mechanisms underlying cerebral ischemia [20, 21]. De Paola
et al. [21] developed a novel human 3D self-assembled iPSC-derived model
(human cortical organism, hCO) to examine ischemic effects caused by OGD. These
authors found that hCOs exhibited neuronal death and a reduction in neuronal
network complexity after being subjected to OGD. Although OGD is commonly
utilized to simulate hypoxic or ischemic injury, existing studies report
considerable variability in the severity of injury [22]. Previous OGD research
predominantly utilized primary rodent neuronal cells or genetically altered cell
lines in non-physiological states, such as the SH-SY5Y cell line which is known
for its genetic abnormalities. Hence, there is a pressing need to develop
non-neoplastic, in vitro human cell-based models. In 2020, Juntunen
et al. [23] established the first in vitro human stroke model
using neurons differentiated from human iPSCs. The lack of a standardized OGD
protocol to accurately replicate hypoxic or ischemic injury in vitro is
also noteworthy. Experimental OGD durations have varied from 1 to 24 h, sometimes
followed by a reperfusion period, while oxygen levels in hypoxic conditions have
ranged between 0% and 8% [23]. Our earlier study demonstrated that neurons
derived from human iPSCs are capable of expressing µ-opioid receptors [11].
Using human iPSC-derived neurons to model OGD/R, the present study assessed the
effects of Herkinorin on apoptosis-related proteins and the distribution of MOR.
We found that OGD/R increased the expression of pro-apoptotic Bax and cleaved
Caspase-3, while decreasing that of anti-apoptotic Bcl-2. Moreover, Herkinorin at
1 µM reversed these changes. Herkinorin was also observed to alter MOR
localization, reduce membrane-associated MOR, and increase cytoplasmic MOR in a
concentration-dependent manner. Our data support previous evidence that MOR
activation confers neuroprotection by modulating mitochondrial apoptotic pathways
[24], while extending these findings to human iPSC-derived neurons. This model
has greater physiological relevance than rodent neurons or immortalized cell
lines [16, 17, 23, 25]. The Herkinorin-induced reduction in Bax and cleaved
Caspase-3 expression, coupled with increased Bcl-2, suggests a shift toward an
anti-apoptotic profile, consistent with MOR-mediated pro-survival signaling. In
the current study we limited the experimental concentration range of Herkinorin
to 0.1–1 µM, as higher concentrations (
A notable point of contention in the literature is whether Herkinorin induces
MOR internalization. While some studies have reported no
The observed reciprocal changes in membrane and cytoplasmic MOR suggest
internalization, which could enable protective intracellular signaling. However,
we did not directly measure
Herkinorin, a derivative of salvinorin A and a potent agonist of MOR and KOR,
may not cause the significant tolerance or dependence that is typically
associated with traditional opioids. Recent research suggests that Herkinorin
produces a dose-dependent, antinociceptive effect in a rat pain model, indicating
its potential as a novel analgesic with a lower risk of dependence or tolerance
[28]. However, research on the neuroprotective effects of Herkinorin is limited,
with only a few studies hinting at such properties [14, 29]. Further
investigations are needed to better understand the neuroprotective capabilities
of Herkinorin and the associated mechanisms. Our study revealed distinct
alterations in the distribution of MOR between membrane and cytoplasmic fractions
under OGD/R, particularly after Herkinorin treatment. OGD/R alone did not
significantly affect membrane MOR, but 1 µM Herkinorin reduced the membrane
expression of MOR while increasing cytoplasmic levels. This reciprocal change
suggests ligand-induced internalization, whereby MOR translocates from the
membrane to the cytoplasm, potentially engaging protective intracellular
signaling. Alternatively, OGD/R may promote cytoplasmic MOR degradation, with
Herkinorin preventing this process, or enhancing redistribution. These
dose-dependent effects were most pronounced at 1 µM and warrant further
investigation using live-cell trafficking assays and pathway-specific analyses.
However, it is important to emphasize the narrow therapeutic window observed in
our study. At concentrations above 10 µM, Herkinorin reduced cell
viability. Thus, the observed neuroprotection is confined to a limited
concentration range (0.1–1 µM), which may pose challenges for clinical
translation. Overall, our results indicate that OGD/R and Herkinorin treatments
lead to a redistribution of MOR between the cytoplasm and membrane in neuronal
cells. This may influence mitochondrial function and thereby affect cell
survival. Conversely, some studies have found that Herkinorin does not induce
In the present study, we observed that OGD/R failed to induce detectable KOR
internalization in human iPSC-derived neurons, even though MOR redistribution was
evident following Herkinorin treatment. This finding may be explained by several
factors. First, differences between models could play a key role. Previous
observations of OGD-induced KOR internalization were mostly in rodent or
immortalized neuronal cell lines (e.g., Neuro2A cells stably expressing tagged
KOR) in which receptor expression levels, membrane composition, and intracellular
trafficking machinery differ substantially from those of human iPSC-derived
neurons [30]. Second, KOR internalization is known to require specific
In summary, Herkinorin reduced OGD/R-induced apoptosis and modulated MOR distribution in human iPSC-derived neurons. Future work should focus on strategies to expand its therapeutic range, such as structural modification of Herkinorin analogs, controlled delivery systems, or combination therapies designed to mitigate toxicity while preserving efficacy. These findings highlight the potential of physiologically relevant human neuronal models for studying opioid receptor biology under ischemia-like conditions. They also underscore the need to consider experimental context when interpreting conflicting data on membrane trafficking. The future use of physiological iPSC-derived neural cells to simulate hypoxic or ischemic injury in the context of OGD should lead to further valuable insights in this field.
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
XC and GW designed the study. ZJ, YY, and XL developed culture cells and performed immunofluorescence, flow cytometry, and Western blot analyses. Data were analyzed by ZJ and XL. The manuscript was written by ZJ and XC. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This work was supported by Beijing Natural Science Foundation-7212019.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.








