1 Department of Pharmacology, School of Pharmaceutical Science, Ohu University, 963-8041 Koriyama, Fukushima, Japan
†These authors contributed equally.
Abstract
Ramelteon, a melatonin receptor agonist, has been reported to alleviate postoperative delirium (POD), although its efficacy remains controversial. The mechanisms of ramelteon’s effects are unclear and few animal studies have addressed POD-related behavioral and molecular changes. We investigated the specific postoperative behavioral and molecular changes that result from ramelteon pretreatment.
Ramelteon (0.03 or 0.3 mg/kg, p.o.) was given to mice once a day for 7 consecutive days before abdominal laparotomy under 2 h of isoflurane anesthesia. Postoperative locomotor activity was monitored for 7 days using s.c.-implanted Nano-tag devices in the dorsal region of aged mice (70–80 weeks). One day after surgery, a social interaction test was administered that used a habituation-discrimination paradigm to evaluate social recognition, specifically the ability to distinguish a novel aged intruder from a familiar young intruder after exposure to the latter. Working memory and related cognitive functions were evaluated using the Y maze and novel-object recognition tests. Cytokine levels and microglial activation in the prefrontal cortex and hippocampus were analyzed by western blotting 24 h post-surgery.
Isoflurane anesthesia for 2 h did not impair spontaneous alternation in the Y maze or performance in the novel-object recognition test. However, it induced prolonged hyperactivity and a decrease in social-recognition performance. Pretreatment with ramelteon at a dose of 0.3 mg/kg, but not 0.03 mg/kg, attenuated postoperative hyperactivity and preserved normal social recognition. Furthermore, ramelteon significantly reduced isoflurane-induced elevation of interleukin-1β in the prefrontal cortex but not in the hippocampus.
Isoflurane anesthesia combined with abdominal surgery was associated with prolonged hyperactivity and impaired social recognition, although other cognitive domains such as working memory appeared to remain unaffected. Ramelteon appeared to alleviate these behavioral and neuroinflammatory changes, suggesting its potential for preventing certain postoperative neurobehavioral alterations.
Keywords
- aged mice
- hyperactivity
- isoflurane anesthesia
- ramelteon
- social recognition deficit
In 2008, approximately 230 million individuals worldwide were reported to be undergoing surgeries requiring general anesthesia each year [1]. Among these, an estimated 26% experience transient neurobehavioral disturbances within the first week after surgery, and about 10% continue to show such symptoms even 3 mo later [2]. These postoperative disturbances are particularly prevalent in older adults, with incidence rates reported to reach up to 53% [3]. One of the clinically recognized conditions in this spectrum of symptoms is postoperative delirium (POD), which is characterized as an acute and fluctuating disturbance in mental status, typically occurring from the immediate postoperative period up to 5 days after surgery [4]. Clinically, POD is classified into hyperactive, hypoactive, and mixed subtypes [5], and patients often exhibit a range of symptoms, including cognitive impairment, disorganized thinking, anxiety, and sleep disturbances [6, 7]. The incidence of such conditions varies depending on factors such as age, type of surgery (e.g., abdominal, orthopedic, urological, and thoracic), and surgical setting (elective, emergency, or urgent), with reported rates ranging between 10% and 50% [8, 9]. Recent studies have focused on the impact of anesthetic agents, methods of administration, and duration of exposure as potential contributors to postoperative neurobehavioral changes [10, 11, 12]. These conditions are associated with increased postoperative morbidity, prolonged hospitalization, poor functional recovery, and reduced long-term survival [13]. Despite their clinical significance, the underlying mechanisms remain poorly understood. Currently, pharmacological treatments are limited, primarily due to insufficient knowledge of the pathophysiology of these postoperative neurobehavioral alterations [14].
A more comprehensive understanding of the pharmacological actions of anesthetics
may hold significant clinical relevance for the prevention and treatment of
postoperative neurobehavioral disturbances [12]. Accordingly, there is growing
interest in developing effective therapeutic strategies based on a better
understanding of the underlying mechanisms of such conditions. At present, oral
antipsychotic drugs are recommended as the first-line pharmacological treatment
for POD [15]. However, none have demonstrated robust therapeutic efficacy [16].
Surgery under isoflurane anesthesia has been shown to induce various
neurobehavioral impairments in mouse models, including hippocampal and striatal
dysfunction associated with sleep fragmentation [17]. Repeated anesthesia and
surgery do not impair learning and memory in middle-aged mice [18], but their
effects in aged animals remain unclear, particularly in relation to
region-specific neuroinflammatory responses and associated behavioral changes
[19]. Although many studies have focused on cognitive and memory impairments,
research addressing persistent postoperative hyperactivity in rodent models
remains limited, and the mechanisms underlying this phenomenon are largely
unknown. One previous study reported that isoflurane anesthesia induced
hyperlocomotion only during the first 10 min after the cessation of a 20-min
exposure [20], but that finding was based on a single study using young mice, and
the behavioral assessments were restricted to the immediate post-anesthesia
period. Recent evidence has indicated that general anesthesia, with or without
surgery, may contribute to the development of postoperative neurocognitive
disturbances, particularly in aged individuals, with neuroinflammation considered
to be a potential contributing factor [21]. In aged mice, surgical trauma
combined with anesthesia can disrupt the blood–brain barrier and activate
microglia, leading to elevated levels of proinflammatory cytokines, such as
IL-1
Recent studies have shown that ramelteon—a melatonin type 1 (MT1) and
type2 (MT2) receptor agonist—may improve certain behavioral and cognitive
functions in mice [24, 25]. Moreover, melatonin has been suggested to alleviate
impairments induced by long-term exposure to isoflurane by improving
circadian-rhythm resynchronization in aged mice [26, 27]. In clinical settings,
ramelteon has shown potential therapeutic effects in patients with delirium
[28, 29, 30]. In contrast, some reports have claimed that ramelteon does not
ameliorate delirium symptoms in the postoperative period [31, 32, 33, 34]. These
inconsistent findings may be attributable to the currently unclear mechanisms
underlying its potential neuroprotective effects, which have led to a lack of
consensus on the optimal timing of its administration, as well as to considerable
variability in patients’ surgical backgrounds. In light of the above, the present
study investigated whether a combination of 2 h of isoflurane anesthesia and
abdominal surgery induced postoperative changes in locomotor activity, social
recognition, and working memory in aged male mice (70–80 weeks old). We also
examined whether pretreatment with ramelteon mitigated these changes.
Furthermore, to explore potential molecular mechanisms underlying the observed
behavioral alterations, we assessed neuroinflammatory responses in the prefrontal
cortex and hippocampus, focusing on cytokines such as IL-1
We purchased 40 male C57BL/6J mice aged 32 weeks, 15 aged 48 weeks, 40 aged 54
weeks, and 15 aged 71 weeks from Charles River Laboratories (Yokohama, Japan).
All mice were used in the experiment only after reaching at least 70 weeks of
age. In addition, 35 mice were reared at the animal facility of Ohu University by
maintaining C57BL/6J mice purchased from CLEA Japan (Tokyo, Japan) at 6 weeks of
age until they reached 70 weeks. The mice were housed in a controlled environment
(25
Ramelteon (Lot. No. RMT-322010-J1) was provided by Sanyo Chemical Laboratories
Co., Ltd. (Osaka, Japan). Isoflurane was purchased from Sigma-Aldrich (#792632,
St. Louis, MO, USA). The anti-mouse monoclonal antibody against IL-1
The mice were randomly assigned to one of the following groups: the 10-min
isoflurane anesthesia with surgery (sham control) group, the 2-h isoflurane
anesthesia with surgery group, or the ramelteon-treated 2-h isoflurane anesthesia
with surgery group (0.03, or 0.3 mg/kg). For the surgical procedure, mice were
initially anesthetized with 3% isoflurane in 100% air at a flow rate of 2
L/min for 30 min to perform a simple laparotomy. This was followed by maintenance
anesthesia with 1.5% isoflurane administered in a transparent anesthetic chamber
for up to 2 h, with additional isoflurane provided as needed. A longitudinal
midline incision was made through the skin, abdominal muscles, and peritoneum,
extending from the xiphoid process to 0.5 cm proximal to the pubic symphysis. A
segment of the small intestine was gently exteriorized using forceps and held for
1 min. The incision was then closed in layers using 5-0 Vicryl sutures. At the
end of the procedure, eutectic mixture of local anesthetics (EMLA) cream (2.5% lidocaine
and 2.5% prilocaine, Sato Pharmaceutical Co., Ltd, Tokyo, Japan) was applied to the incision sites. To alleviate
incision-related pain, the cream was reapplied every 12 h until behavioral
testing at 24 h post-surgery or for 48 h in mice used for locomotor-activity
assessment. The temperature of the anesthetic chamber was maintained using a
TCM-01 system (Brain Science Idea Co., Ltd., Osaka, Japan), with rectal
temperature controlled at 37
Based on a previous report that found the mean duration of delirium to be less
than 9 days (4.0
Melatonin is secreted at night in humans, and stimulation of melatonin receptors has been reported to be effective in promoting sleep [37]. The active phase of mice differs from that of humans by approximately 12 h; they are nocturnal and primarily active during the dark phase. In rodents, melatonin secretion has been reported to occur during this active period at night [38]. Therefore, in the present study, male C57BL/6J mice aged 70–80 weeks received an intraperitoneal administration of either vehicle (corn oil; Cat. No. 23-0320, Sigma-Aldrich) or ramelteon at a dose of 0.03 or 0.3 mg/kg, dissolved in corn oil immediately before use [39], once daily at 1800 h (2 h before the onset of the dark phase), for 7 consecutive days prior to surgery. For cognitive function tests, administration continued until the day before the test (i.e., the day of surgery). Additionally, the effects of ramelteon on activity levels after isoflurane anesthesia were assessed by administering it once daily at 1800 h for 7 days after isoflurane anesthesia.
At 70 weeks of age, the mice were randomly assigned to one of the following behavioral-testing groups: the 10-min isoflurane anesthesia with surgery (sham control) group, the 2-h isoflurane anesthesia with surgery group, or the ramelteon-treated 2-h isoflurane anesthesia with surgery group (0.03 or 0.3 mg/kg). Two investigators, blind to the group assignments, performed all behavioral tests. We recorded all behavioral tests using a web camera installed with the ANY-maze software (version 6.35; Muromachi Kikai Co., Ltd., Tokyo, Japan) to collect and analyze the data for all behavioral tests.
The social interaction test was performed 24 h after isoflurane anesthesia and
abdominal surgery. The test mouse was initially placed in an acrylic box (30 cm
A gray polyvinyl chlorid Y maze (YM-40M, Brain Science Idea Co., Ltd., Osaka, Japan) with
three arms, each measuring 50 mm (W)
As the evaluation in the current study had to be conducted 24 h after isoflurane
anesthesia with surgery, the object- recognition test was performed using a
modified version of the method described by Leger et al. [44]. An
acrylic box (internal dimensions: 300 mm [W]
After euthanizing the mice, the prefrontal cortex and hippocampus were harvested
for Western Blot analysis. Mouse prefrontal cortex and hippocampal tissues were
homogenized using a Dounce homogenizer (Cat. No. 357544, SANSYO Co., Ltd. Tokyo,
Japan), and the mixture was centrifuged at 500
All statistical analyses were performed using BellCurve software (version 3.20;
Social Survey Research Information Co., Ltd., Tokyo, Japan) [46]. A two-way
repeated measures analysis of variance (ANOVA; isoflurane
Because abnormal increases or decreases in locomotor activity are commonly
observed after surgery [5], and the mean duration of postoperative behavioral
disturbances has been reported to be less than 9 days (Fig. 1A, 4.0
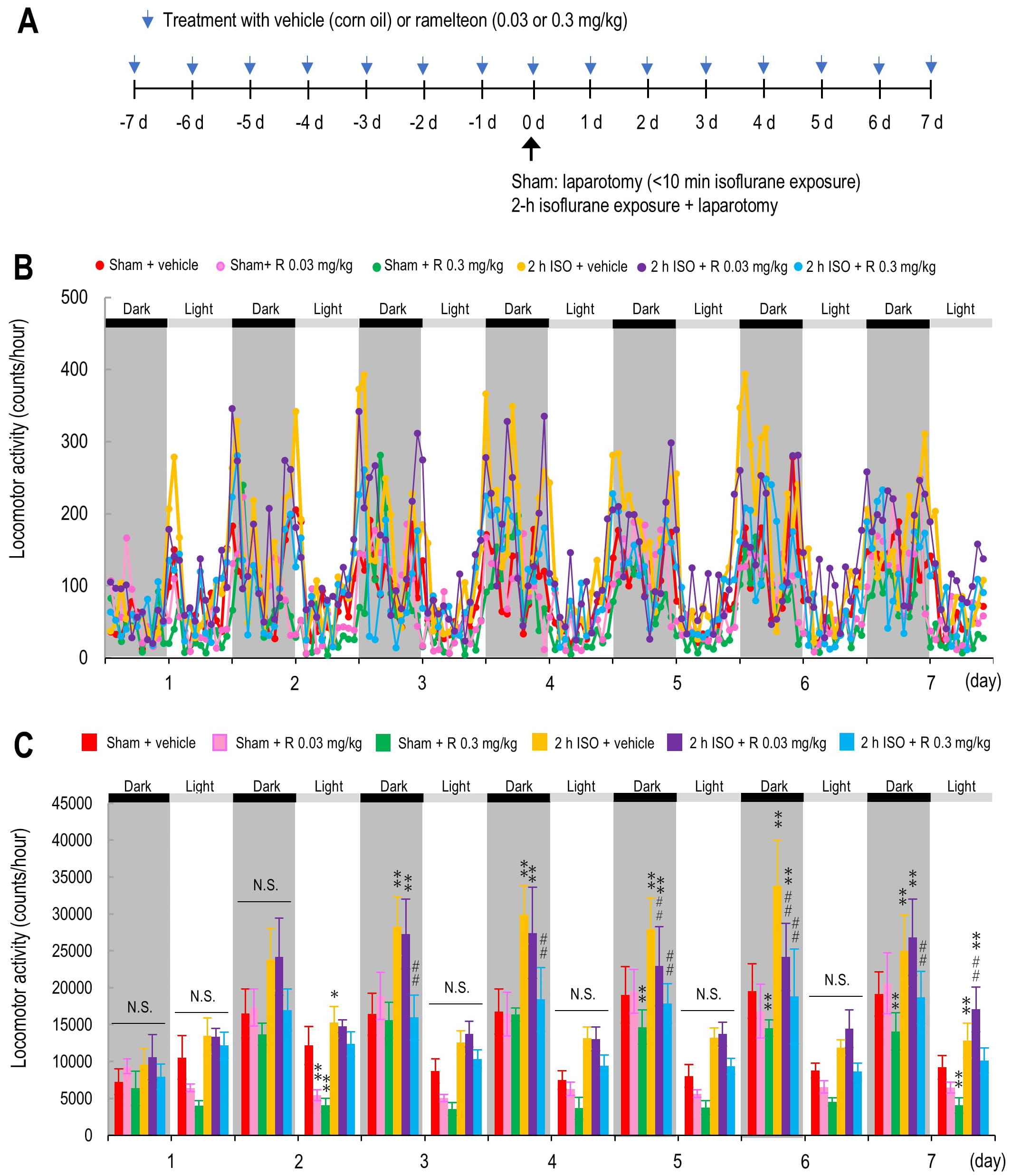 Fig. 1.
Fig. 1.
Gross locomotor activity averaged in 2-hour intervals from Day 1
to Day 7. (A) Schematic diagram of the experimental design. (B,C) Locomotor
activity levels (B) and daily averaged gross locomotor activity (C) from Day 1 to
Day 7 in the following groups: vehicle-treated sham group (red; n = 7), ramelteon
0.03 mg/kg-treated sham group (pink; n = 5), ramelteon 0.3 mg/kg-treated sham
group (light green; n = 5), vehicle-treated 2-hour isoflurane exposure group
(yellow; n = 6), ramelteon 0.03 mg/kg-treated 2-hour isoflurane group (purple; n
= 6), and ramelteon 0.3 mg/kg-treated 2-hour isoflurane group (light blue; n =
6). Horizontal dark gray bars indicate the dark phase; horizontal light gray bars
indicate the light phase of the day. p
| Day | Phase | F (5, 95) | p value | Group 1 | Group 2 | z value | p value | F value |
| 0.5 | Dark | 0.2925 | 0.9160 | Sham | Sham + R 0.3 mg/kg | 0.8040 | 0.9658 | |
| Sham + R 0.03 mg/kg | 1.9150 | 0.3975 | ||||||
| 2 h ISO | 2.2200 | 0.2369 | ||||||
| 2 h ISO + R 0.3 mg/kg | 0.6697 | 0.9847 | ||||||
| 2 h ISO + R 0.03 mg/kg | 3.1952 | 0.0225 | * | |||||
| 2 h ISO | 2 h ISO + R 0.3 mg/kg | 1.4939 | 0.6670 | |||||
| 2 h ISO + R 0.03 mg/kg | 0.9397 | 0.9344 | ||||||
| 2 h ISO + R 0.3 mg/kg | 2 h ISO + R 0.03 mg/kg | 2.4337 | 0.1542 | |||||
| 1 | Light | 1.663 | 0.1511 | Sham | Sham + R 0.3 mg/kg | 5.9218 | p |
** |
| Sham + R 0.03 mg/kg | 3.7415 | 0.0041 | ** | |||||
| 2 h ISO | 2.8278 | 0.0612 | ||||||
| 2 h ISO + R 0.3 mg/kg | 1.5982 | 0.5997 | ||||||
| 2 h ISO + R 0.03 mg/kg | 2.6757 | 0.0891 | ||||||
| 2 h ISO | 2 h ISO + R 0.3 mg/kg | 1.1849 | 0.8417 | |||||
| 2 h ISO + R 0.03 mg/kg | 0.1466 | 1.0000 | ||||||
| 2 h ISO + R 0.3 mg/kg | 2 h ISO + R 0.03 mg/kg | 1.0383 | 0.9029 | |||||
| 1.5 | Dark | 2.118 | 0.0699 | Sham | Sham + R 0.3 mg/kg | 2.5666 | 0.1150 | |
| Sham + R 0.03 mg/kg | 0.7297 | 0.9775 | ||||||
| 2 h ISO | 6.9969 | p |
** | |||||
| 2 h ISO + R 0.3 mg/kg | 0.4604 | 0.9973 | ||||||
| 2 h ISO + R 0.03 mg/kg | 7.3409 | p |
** | |||||
| 2 h ISO | 2 h ISO + R 0.3 mg/kg | 6.2988 | p |
** | ||||
| 2 h ISO + R 0.03 mg/kg | 0.3314 | 0.9994 | ||||||
| 2 h ISO + R 0.3 mg/kg | 2 h ISO + R 0.03 mg/kg | 6.6302 | p |
** | ||||
| 2 | Light | 2.462 | 0.0383 | Sham | Sham + R 0.3 mg/kg | 7.3290 | p |
** |
| Sham + R 0.03 mg/kg | 6.1490 | p |
** | |||||
| 2 h ISO | 2.9509 | 0.0444 | * | |||||
| 2 h ISO + R 0.3 mg/kg | 0.1795 | 1.0000 | ||||||
| 2 h ISO + R 0.03 mg/kg | 2.4826 | 0.1387 | ||||||
| 2 h ISO | 2 h ISO + R 0.3 mg/kg | 2.6705 | 0.0902 | |||||
| 2 h ISO + R 0.03 mg/kg | 0.4512 | 0.9976 | ||||||
| 2 h ISO+ R 0.3 mg/kg | 2 h ISO + R 0.03 mg/kg | 2.2194 | 0.2372 | |||||
| 2.5 | Dark | 4.100 | 0.0021 | Sham | Sham + R 0.3 mg/kg | 0.7631 | 0.9727 | |
| Sham + R 0.03 mg/kg | 2.2293 | 0.2327 | ||||||
| 2 h ISO | 11.3613 | p |
** | |||||
| 2 h ISO + R 0.3 mg/kg | 0.4305 | 0.9980 | ||||||
| 2 h ISO + R 0.03 mg/kg | 10.3745 | p |
** | |||||
| 2 h ISO | 2 h ISO + R 0.3 mg/kg | 11.3628 | p |
## | ||||
| 2 h ISO + R 0.03 mg/kg | 0.9509 | 0.9312 | ||||||
| 2 h ISO + R 0.3 mg/kg | 2 h ISO + R 0.03 mg/kg | 10.4119 | p |
$$ | ||||
| 3 | Light | 1.794 | 0.1215 | Sham | Sham + R 0.3 mg/kg | 4.6530 | p |
** |
| Sham + R 0.03 mg/kg | 3.3345 | 0.0149 | * | |||||
| 2 h ISO | 3.7046 | 0.0046 | ** | |||||
| 2 h ISO + R 0.3 mg/kg | 1.5435 | 0.6352 | ||||||
| 2 h ISO + R 0.03 mg/kg | 4.7767 | p |
** | |||||
| 2 h ISO | 2 h ISO + R 0.3 mg/kg | 2.0825 | 0.3035 | |||||
| 2 h ISO + R 0.03 mg/kg | 1.0331 | 0.9047 | ||||||
| 2 h ISO + R 0.3 mg/kg | 2 h ISO + R 0.03 mg/kg | 3.1156 | 0.0282 | * | ||||
| 3.5 | Dark | 4.440 | 0.0011 | Sham | Sham + R 0.3 mg/kg | 0.3643 | 0.9991 | |
| Sham + R 0.03 mg/kg | 0.3181 | 0.9995 | ||||||
| 2 h ISO | 12.5594 | p |
** | |||||
| 2 h ISO + R 0.3 mg/kg | 1.5879 | 0.6064 | ||||||
| 2 h ISO + R 0.03 mg/kg | 10.1619 | p |
** | |||||
| 2 h ISO | 2 h ISO + R 0.3 mg/kg | 10.5724 | p |
## | ||||
| 2 h ISO + R 0.03 mg/kg | 2.3103 | 0.1989 | ||||||
| 2 h ISO + R 0.3 mg/kg | 2 h ISO + R 0.03 mg/kg | 8.2621 | p |
$$ |
**p
| Day | Phase | F (5, 95) | p value | Group 1 | Group 2 | z value | p value | |
| 4 | Light | 1.608 | 0.1654 | Sham | Sham + R 0.3 mg/kg | 3.4815 | 0.0095 | ** |
| Sham + R 0.03 mg/kg | 1.1122 | 0.8741 | ||||||
| 2 h ISO | 5.4155 | p |
** | |||||
| 2 h ISO + R 0.3 mg/kg | 1.8384 | 0.4444 | ||||||
| 2 h ISO + R 0.03 mg/kg | 5.2916 | p |
** | |||||
| 2 h ISO | 2 h ISO + R 0.3 mg/kg | 3.4470 | 0.0106 | * | ||||
| 2 h ISO + R 0.03 mg/kg | 0.1194 | 1.0000 | ||||||
| 2 h ISO + R 0.3 mg/kg | 2 h ISO + R 0.03 mg/kg | 3.3276 | 0.0152 | * | ||||
| 4.5 | Dark | 2.434 | 0.0402 | Sham | Sham + R 0.3 mg/kg | 3.9452 | 0.0020 | ** |
| Sham + R 0.03 mg/kg | 0.4191 | 0.9983 | ||||||
| 2 h ISO | 8.5581 | p |
** | |||||
| 2 h ISO + R 0.3 mg/kg | 1.1150 | 0.8730 | ||||||
| 2 h ISO + R 0.03 mg/kg | 3.7327 | 0.0042 | ** | |||||
| 2 h ISO | 2 h ISO + R 0.3 mg/kg | 9.3211 | p |
## | ||||
| 2 h ISO + R 0.03 mg/kg | 4.6498 | p |
## | |||||
| 2 h ISO + R 0.3 mg/kg | 2 h ISO + R 0.03 mg/kg | 4.6713 | p |
$$ | ||||
| 5 | Light | 1.771 | 0.1262 | Sham | Sham + R 0.3 mg/kg | 3.8557 | 0.0028 | ** |
| Sham + R 0.03 mg/kg | 2.1535 | 0.2678 | ||||||
| 2 h ISO | 4.9593 | p |
** | |||||
| 2 h ISO + R 0.3 mg/kg | 1.2485 | 0.8102 | ||||||
| 2 h ISO + R 0.03 mg/kg | 5.4375 | p |
** | |||||
| 2 h ISO | 2 h ISO + R 0.3 mg/kg | 3.5758 | 0.0070 | ** | ||||
| 2 h ISO + R 0.03 mg/kg | 0.4608 | 0.9973 | ||||||
| 2 h ISO + R 0.3 mg/kg | 2 h ISO + R 0.03 mg/kg | 4.0366 | 0.0015 | ** | ||||
| 5.5 | Dark | 5.550 | p |
Sham | Sham + R 0.3 mg/kg | 4.5769 | p |
** |
| Sham + R 0.03 mg/kg | 2.4600 | 0.1457 | ||||||
| 2 h ISO | 13.6269 | p |
** | |||||
| 2 h ISO + R 0.3 mg/kg | 0.7124 | 0.9798 | ||||||
| 2 h ISO + R 0.03 mg/kg | 4.3981 | p |
** | |||||
| 2 h ISO | 2 h ISO + R 0.3 mg/kg | 13.8177 | p |
## | ||||
| 2 h ISO + R 0.03 mg/kg | 8.8931 | p |
## | |||||
| 2 h ISO + R 0.3 mg/kg | 2 h ISO + R 0.03 mg/kg | 4.9246 | p |
$$ | ||||
| 6 | Light | 1.433 | 0.2195 | Sham | Sham + R 0.3 mg/kg | 3.8471 | 0.0029 | ** |
| Sham + R 0.03 mg/kg | 2.0172 | 0.3386 | ||||||
| 2 h ISO | 2.9833 | 0.0407 | * | |||||
| 2 h ISO + R 0.3 mg/kg | 0.1438 | 1.0000 | ||||||
| 2 h ISO + R 0.03 mg/kg | 5.4158 | p |
** | |||||
| 2 h ISO | 2 h ISO + R 0.3 mg/kg | 3.0133 | 0.0375 | * | ||||
| 2 h ISO + R 0.03 mg/kg | 2.3440 | 0.1858 | ||||||
| 2 h ISO + R 0.3 mg/kg | 2 h ISO + R 0.03 mg/kg | 5.3573 | p |
** | ||||
| 6.5 | Dark | 2.404 | 0.0424 | Sham | Sham + R 0.3 mg/kg | 4.6122 | p |
** |
| Sham + R 0.03 mg/kg | 1.2940 | 0.7860 | ||||||
| 2 h ISO | 5.6154 | p |
** | |||||
| 2 h ISO + R 0.3 mg/kg | 0.4443 | 0.9977 | ||||||
| 2 h ISO + R 0.03 mg/kg | 7.3083 | p |
** | |||||
| 2 h ISO | 2 h ISO + R 0.3 mg/kg | 5.8393 | p |
## | ||||
| 2 h ISO + R 0.03 mg/kg | 1.6313 | 0.5780 | ||||||
| 2 h ISO + R 0.3 mg/kg | 2 h ISO + R 0.03 mg/kg | 7.4706 | p |
$$ | ||||
| 7 | Light | 2.376 | 0.0445 | Sham | Sham + R 0.3 mg/kg | 4.6657 | p |
** |
| Sham + R 0.03 mg/kg | 2.4917 | 0.1360 | ||||||
| 2 h ISO | 3.4721 | 0.0098 | ** | |||||
| 2 h ISO + R 0.3 mg/kg | 0.8710 | 0.9520 | ||||||
| 2 h ISO + R 0.03 mg/kg | 7.4996 | p |
** | |||||
| 2 h ISO | 2 h ISO + R 0.3 mg/kg | 2.5065 | 0.1316 | |||||
| 2 h ISO + R 0.03 mg/kg | 3.8810 | 0.0026 | ## | |||||
| 2 h ISO + R 0.3 mg/kg | 2 h ISO + R 0.03 mg/kg | 6.3875 | p |
$$ |
**p
Since pretreatment with ramelteon at 0.3 mg/kg attenuated the prolonged
hyperactivity observed after 2-h isoflurane anesthesia and surgery, we further
examined the effects of ramelteon pretreatment on the effect on social
recognition and interaction 24 h after 2-h isoflurane exposure combined with
laparotomy, as well as the effect of ramelteon pretreatment (0.3 mg/kg) on social
recognition after 2-h isoflurane anesthesia combined with laparotomy. Three-way
repeated measures ANOVA revealed a significant interaction effect among
isoflurane, ramelteon, and time on social interaction (as measured by the number
of entries into the zone in the presence of the young mouse; Fig. 2A)
(F41,49 = 7.24, p
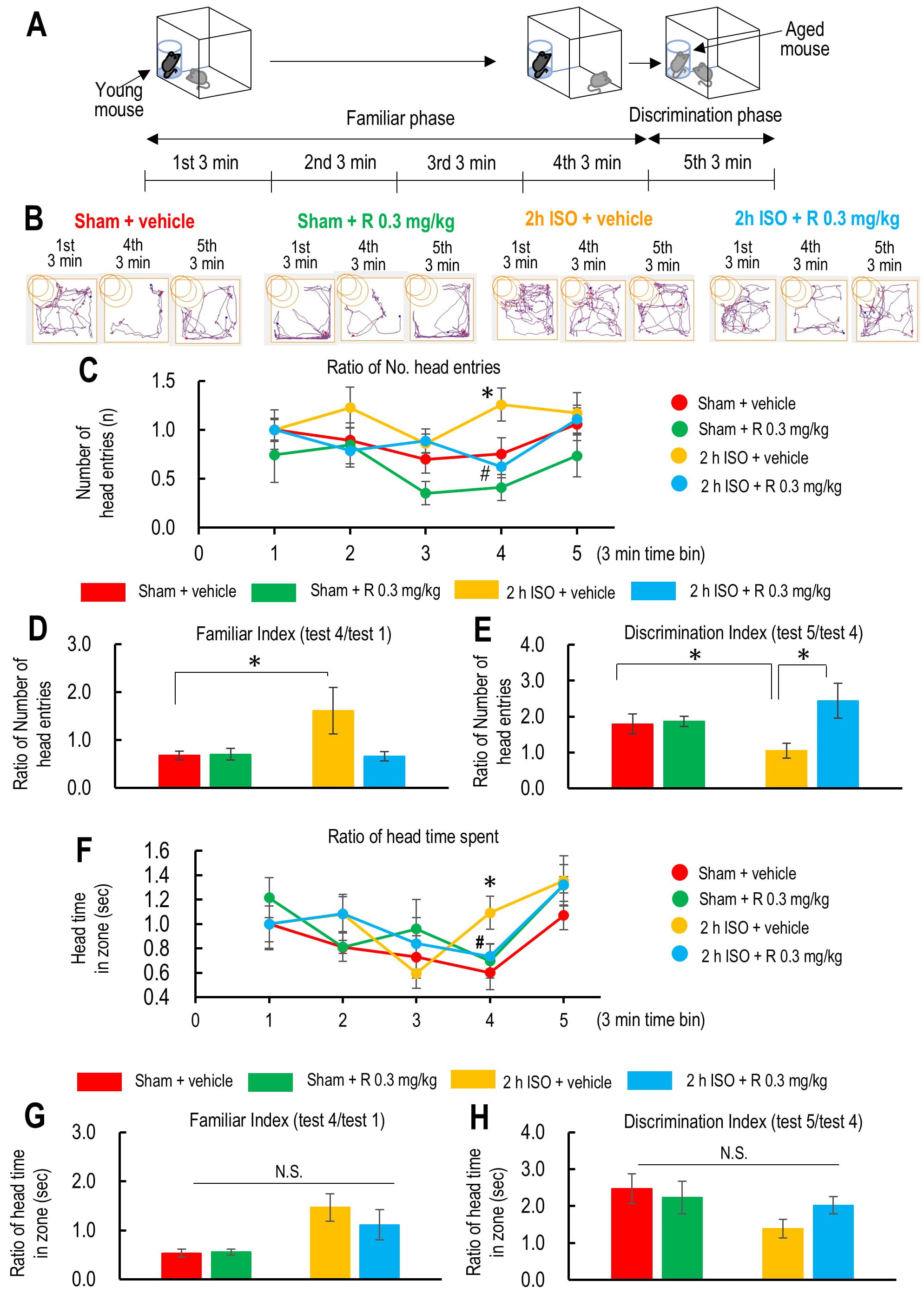 Fig. 2.
Fig. 2.
The effect of ramelteon on the social interaction test in aged
mice after 2-hour isoflurane exposure and laparotomy. (A) Schematic drawings
represent the protocol for the social interaction test. (B) Movement trajectories
of the initial and final 3 minutes of approach to the young mouse and the
trajectory of the mouse approaching the novel-aged mouse. (C) Time course of the
ratio of the total number of mouse head entries into the zone where the young
mouse is present in the beaker placed at the corner of the test box in the
vehicle-treated sham group (red; n = 8), ramelteon 0.3 mg/kg-treated sham group
(light green; n = 5), vehicle-treated 2-hour isoflurane exposure group (yellow; n
= 9), and ramelteon 0.3 mg/kg-treated 2-hour isoflurane exposure group (light
blue; n = 8). (D) Ratio of the number of mouse head entries into the zone where
the young mouse is present during test 4 against test 1 (test 4/test 1) as a
familiar score. (E) Ratio of the number of mouse head entries into the zone where
the aged mouse is present during test 5 (test 4/test 1) against that into the
zone where the young mouse is present during test 4 as a discrimination score.
(F) Time course of the ratio of the time spent in the zone where the young mouse
is present in the beaker placed at the corner of the test box. (G) Ratio of the
time spent in the zone where the young mouse is present during test 4 against
test 1 (test 4/test 1) as a familiar score. (H) Ratio of the time spent in the
zone where the aged mouse is present during test 5 (test 4/test 1) against that
in the zone where the young mouse is present during test 4 as a discrimination
score. p
A two-way ANOVA (isoflurane
Finally, we measured the habituation/discrimination scores as described above.
However, there was no significant interaction between isoflurane and ramelteon on
time spent in the zone with the young mouse, as measured by the habituation score
(F1,29
To assess whether the 2-h exposure to isoflurane combined with laparotomy alone,
or in combination with pretreatment with ramelteon at 0.3 mg/kg, affects
hippocampus-dependent spatial working memory, aged mice were subjected to the
Y-maze test 24 h after surgery. The test was conducted over a 10-min period,
excluding the initial 2-min habituation phase; the remaining 8-min exploration
phase was used to assess spatial working memory. A one-way ANOVA revealed that
neither prolonged isoflurane anesthesia nor ramelteon pretreatment resulted in
differences in spontaneous alternation behavior from that of the
vehicle-pretreated sham group (F3,28 = 1.57, p = 0.223; Fig. 3A).
Total distance traveled was also unaffected by either treatment (F3,28
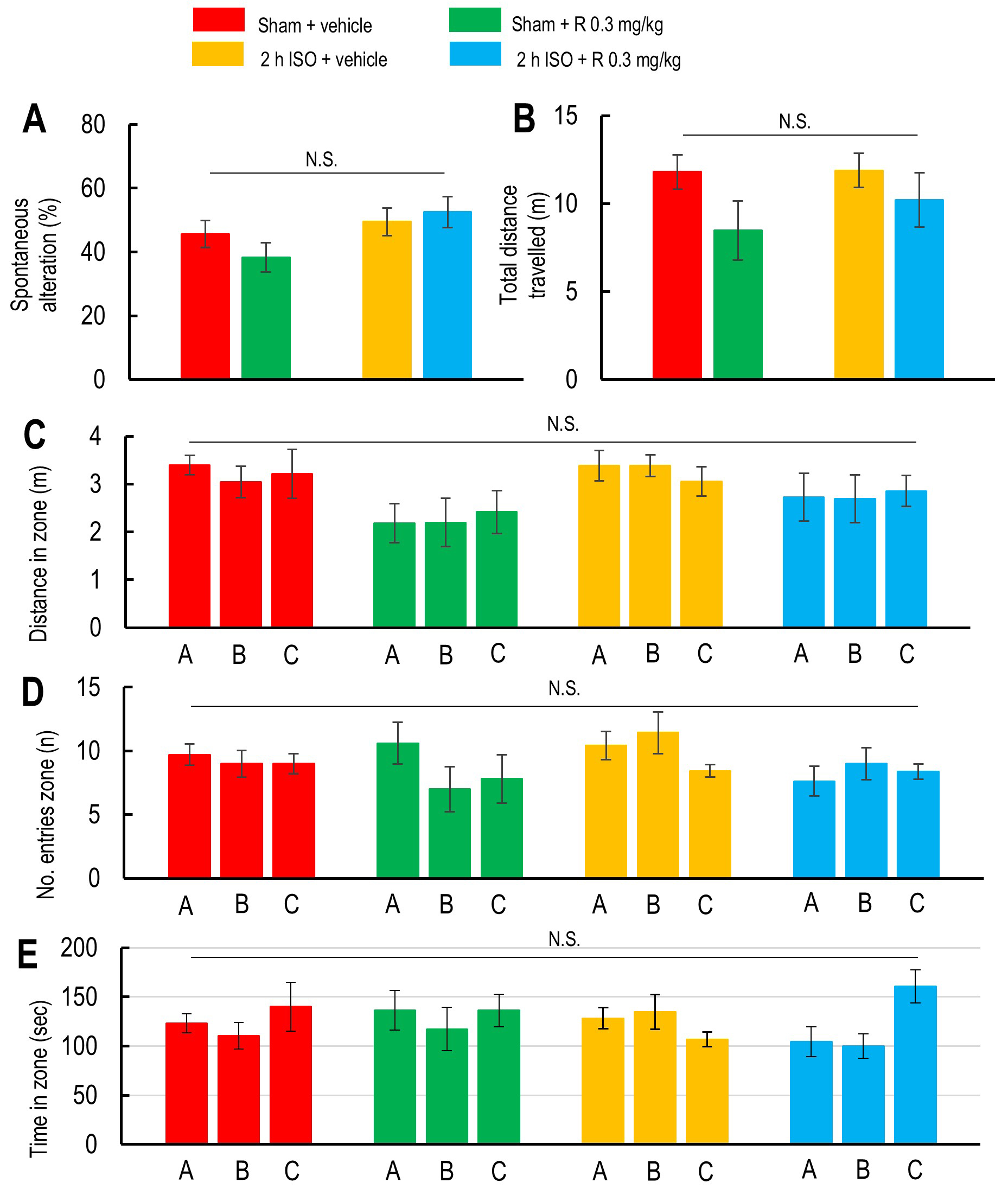 Fig. 3.
Fig. 3.
Effects of 2-hour isoflurane exposure with laparotomy and ramelteon pretreatment on working memory assessed by Y-maze spontaneous alternation. (A,B) Spontaneous alternation percentage (A) and the total distance traveled during the test (B) in the vehicle-treated sham group (red; n = 7), ramelteon 0.3 mg/kg-treated sham group (light green; n = 5), vehicle-treated 2-hour isoflurane exposure group (yellow; n = 7), and ramelteon 0.3 mg/kg-treated 2-hour isoflurane exposure group (light blue; n = 8). Total distance traveled in each arm (C), total number of entries into each arm (D), and total time spent in each arm (E) were analyzed to assess cognitive performance and general activity in the vehicle-treated sham group (red), ramelteon 0.3 mg/kg-treated sham group (light green), vehicle-treated 2-hour isoflurane exposure group (yellow), and ramelteon 0.3 mg/kg-treated 2-hour isoflurane exposure group (light blue). N.S. indicates no significant difference.
Next, we investigated whether non-spatial recognition memory was influenced by
2-h isoflurane exposure with laparotomy and pretreatment of ramelteon at a dose
of 0.3 mg/kg in aged mice after surgery. One-way ANOVA indicated that neither 2-h
isoflurane exposure nor ramelteon pretreatment (0.3 mg/kg) affected locomotor
activity during the open field test (F3,25
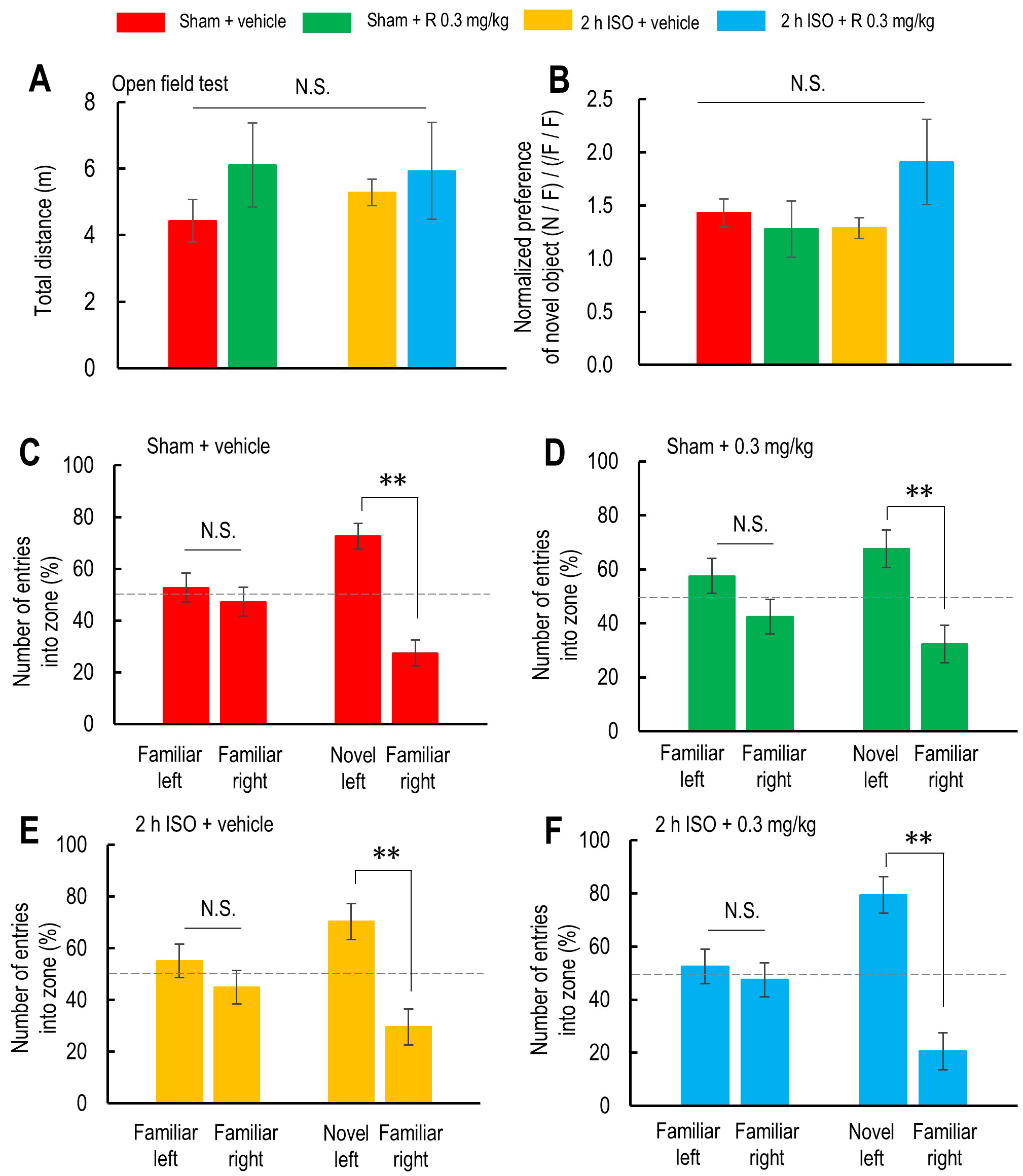 Fig. 4.
Fig. 4.
Effects of 2-hour isoflurane exposure with laparotomy and
ramelteon pretreatment on performance in the novel object recognition test.
(A,B) Total distance traveled during the test (A) and normalized preference for
the novel object (B) in the vehicle-treated sham group (red; n = 7), ramelteon
0.3 mg/kg-treated sham group (light green; n = 5), vehicle-treated 2-hour
isoflurane exposure group (yellow; n = 7), and ramelteon 0.3 mg/kg-treated 2-hour
isoflurane exposure group (light blue; n = 6). (C–F) Number of entries into the
object zones during the first 5 minutes (with two identical objects) and the last
5 minutes (with one familiar and one novel object) in the vehicle-treated sham
group (C), ramelteon 0.3 mg/kg-treated sham group (D), vehicle-treated 2-hour
isoflurane exposure group (E), and ramelteon 0.3 mg/kg-treated 2-hour isoflurane
exposure group (F). p
Finally, we investigated the effect of 2-h isoflurane exposure with laparotomy
and ramelteon pretreatment on cytokines and microglial activation in the
prefrontal cortex and hippocampus. Since ramelteon at 0.3 mg/kg did not affect
social recognition or working memory in the vehicle-treated sham control mice, we
examined its effects only in the 2-h isoflurane anesthesia with laparotomy group
by comparing protein levels of cytokines, markers of microglial activation, and
brain-derived neurotrophic factor (BDNF), a marker of synaptic plasticity, among
the vehicle-treated sham group, the vehicle-treated isoflurane group, and the
ramelteon-treated isoflurane anesthesia with laparotomy group. IL-1
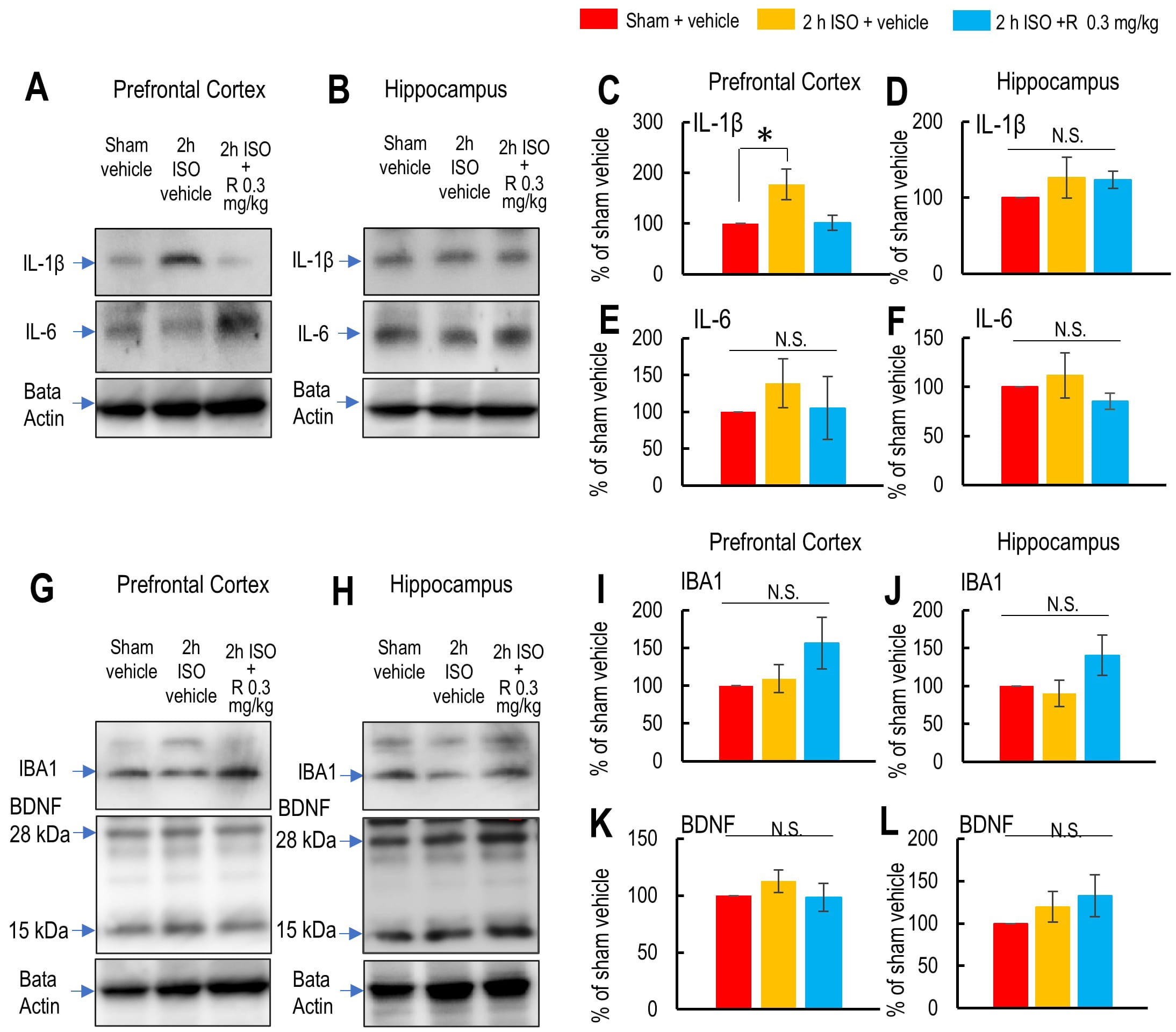 Fig. 5.
Fig. 5.
Effects of 2-hour isoflurane exposure with laparotomy and
ramelteon pretreatment on protein levels of cytokines, activated microglia, and
brain-derived neurotrophic factor (BDNF) in the prefrontal cortex and
hippocampus. (A,B) Representative western blot images showing IL-1
We then examined whether microglial activation was induced by 2-h isoflurane
exposure with laparotomy by assessing the IBA-1 protein levels in the prefrontal
cortex and hippocampus. IBA-1 protein levels were not affected by 2-h isoflurane
exposure, suggesting that significant microglial activation was not observed in
either region (IBA-1 in prefrontal cortex: F2,17 = 1.808, p =
0.120; IBA-1 in hippocampus: F2,8
Additionally, we assessed whether the levels of BDNF, a key protein involved in
synaptic plasticity, were affected by 2-h isoflurane exposure. BDNF levels were
not altered by 2-h isoflurane exposure in either the prefrontal cortex or
hippocampus (BDNF in prefrontal cortex: F2,11
Postoperative memory impairment and cognitive dysfunction have been observed in various rodent models, including aged mice, with several underlying neuronal mechanisms proposed to account for these impairments, such as neuroinflammation, synaptic dysfunction, and disturbances in neurotransmitter systems [17, 18, 48]. However, the mice used in the present study demonstrated prolonged hyperactivity after isoflurane exposure, which may represent a phenomenon related to, but distinct from, the clinical features of POD. POD can manifest in at least two forms: a hypoactive form, characterized by drowsiness and reduced motor activity, and a hyperactive form, characterized by agitation, aggression, restlessness, and the potential for self-harm or harm to others. Both forms are associated with core features of delirium, including disturbances in attention, awareness, and cognition [49]. Postoperative hyperactive delirium, a specific subtype of POD, affects approximately 15% of older patients undergoing non-cardiac surgeries, such as hip-fracture repair, under regional anesthesia [50]. POD is often observed starting from postoperative Day 1 and can persist for up to 1 week [51]. Although many studies have reported cognitive dysfunction after inhalational anesthesia in mice and rats, no studies to date have documented sustained hyperactivity over several days post-surgery in rodent models, despite numerous clinical reports on POD and hyperactivity [49, 52]. Intracranial surgery in mice under isoflurane anesthesia has been reported to increase locomotor activity during the initial peak of the dark phase; however, locomotor activity usually decreases from postoperative Day 2 [53]. Additionally, volatile anesthetics have been shown to induce hyperactivity during the induction phase but produce full anesthesia at higher concentrations [54, 55]. However, these phenomena are not considered to represent POD. In our study, a 2-h exposure to isoflurane significantly increased locomotor activity for over 7 days after surgery, particularly during the dark phase, which corresponds to the active period in mice. Notably, this prolonged hyperactivity was not observed in younger mice (6–10 weeks old; Supplementary Fig. 7) after the same isoflurane exposure. These findings suggest that our model may represent a specific form of postoperative hyperactivity in aged mice, but further studies are needed to clarify its direct relevance to POD.
In the present study, we found that social recognition, as assessed by the social-interaction test, was impaired in mice after 2-h isoflurane exposure with laparotomy, but not in mice that underwent a sham laparotomy. The prefrontal cortex has been suggested to play a key role in regulating social interactions, including social approach and recognition [56, 57]. Reduced excitatory synaptic transmission in the pyramidal neurons of the supragranular layer of the prefrontal cortex has been shown to lead to impaired social interaction [58]. Reciprocal connections between the thalamic area and the prefrontal cortex also play an important role in social recognition by encoding social information [57]. Moreover, N-methyl-D-aspartic acid (NMDA) and amino-3-hydroxy-5-methyl-4-isoxazolepropi- onic acid (AMPA)/kainate glutamate receptors in the prefrontal cortex have been implicated in social-recognition memory [59], and synaptic plasticity in this region is crucial for cognitive behaviors [60, 61]. Therefore, elevated neuronal excitability in pyramidal neurons of the supragranular layer of the prefrontal cortex contributes to social recognition and memory.
In the present study, 2-h isoflurane exposure increased the protein level of
IL-1
Although many studies have reported hippocampus-dependent memory impairments and
increased cytokine levels in the hippocampus after long-term isoflurane exposure
[66, 67, 68, 69, 70, 71], the current study found no changes in hippocampal cytokine levels,
including IL-1
Our molecular analyses revealed a region-specific neuroinflammatory response,
characterized by significantly increased IL-1
In the current study, we found that pretreatment with ramelteon for 7 days
before to 2-h isoflurane exposure prevented hyperlocomotor activity for 7 days,
social recognition deficits, and an increase of IL-1
Additional strategies targeting different mechanisms of POD should include
controlling the depth of sedation during anesthesia administration. In line with
the controversial results of ramelteon treatment, due to the multifaceted and
heterogeneous nature of POD, replication of the full spectrum of its clinical
manifestations in a single animal model remains challenging. Likewise, it is
unlikely that a single pharmacological intervention can comprehensively
ameliorate all associated symptoms. Therefore, it is essential for future
research to delineate which specific aspects of POD are represented by each
animal model and to identify targeted pharmacological strategies that can
selectively mitigate those symptoms. Such an approach would facilitate a more
nuanced understanding of POD pathophysiology and support the development of
tailored therapeutic interventions. In line with this, accumulating
pharmacological evidence suggests that the neuroprotective and anti-inflammatory
effects of melatonin are primarily mediated by MT1 and MT2 receptors,
which are expressed in POD-relevant brain regions such as the prefrontal cortex
and hippocampus. For instance, luzindole, a non-selective MT1/MT2
antagonist, has been widely used to investigate receptor-mediated effects of
melatonin in the central nervous system [79]. Similarly, the selective MT2
antagonist cis-4-Phenyl-2-propionamidotetralin (4P-PDOT) has been shown to block
melatonin-induced modulation of neuroinflammation and cognition in rodents [80].
Although our study did not include MT-receptor antagonists, the observed
protective effects of ramelteon—especially its suppression of IL-1
Repeated exposure to isoflurane has been investigated in animal models because single exposures often did not produce robust or reproducible cognitive impairments in rodents [81, 82, 83]. However, the results of these studies have been inconsistent, particularly regarding the extent of cognitive deficits caused by repeated anesthetic exposure in aged animals. Although some studies reported no impairments in spatial memory or psychomotor performance in aged mice [19, 82, 83], others suggested potential adverse effects on cognitive functions depending on factors such as age, duration of exposure, and interval of exposure. For example, repeated exposures to isoflurane even facilitated spatial learning in young adult mice [81]. Those findings suggested that increasing the frequency or duration of anesthetic exposure does not necessarily exacerbate cognitive dysfunction. Furthermore, our current design incorporates surgical stress combined with a single isoflurane exposure in aged mice, which may better replicate the clinical conditions that produce postoperative cognitive dysfunction. Thus, although repeated-exposure models may offer additional insights, a single exposure, combined with surgery, remains a relevant and valid approach to studying postoperative neurobehavioral changes. Nevertheless, future studies that include repeated exposures would be valuable for further exploring the effects of anesthetic duration and frequency on postoperative cognitive dysfunction.
Another limitation is that the present study did not include an anesthesia-only control group. Our paradigm was designed to reflect realistic perioperative conditions, in which general anesthesia is typically administered for surgical procedures as is the case in many studies using mice [18, 72, 84, 85, 86]. However, it is important to consider the potential impact of prolonged anesthesia alone. Previous studies have shown that anesthesia alone does not consistently result in cognitive or behavioral impairments [85, 87]. On the other hand, it has also been reported that isoflurane anesthesia indeed alters the brain metabolites of mice immediately after anesthesia [88]. Those findings suggest that the behavioral changes observed in our study may have stemmed primarily from the combination of surgical stress and prolonged anesthesia. Nonetheless, including an anesthesia-only group in future studies would help clarify the individual contribution of anesthesia to postoperative neurobehavioral outcomes, particularly in aged brains.
A 2-h isoflurane anesthesia exposure, combined with abdominal surgery, induced
prolonged hyperactivity and impaired social recognition in aged mice, without
impairing working memory functions; both of these cognitive processes involve not
only the hippocampus but also other brain regions such as the perirhinal cortex.
Pretreatment with ramelteon 0.3 mg/kg prevented these deficits and suppressed
IL-1
ANOVA, analysis of variance; BDNF, brain-derived neurotrophic factor; IBA, ionized calcium-binding adapter molecule 1; IL, interleukin; ISO, isoflurane; MT, melatonin; NIH, National Institutes of Health; N.S., not significant; 4P-PDOT, cis-4-Phenyl-2-propionamidotetralin; POD, postoperative delirium; PVDF, polyvinylidene fluoride; SDS-PAGE, sodium dodecyl sulfate-polyacrylamide gel electrophoresis; SEM, standard error of the mean; TEMED, N,N,N’,N’-Tetramethylethylenediamine; Tukey HSD test, Tukey honestly significant difference test; WB, western blotting.
Data are available from the corresponding author upon a reasonable request and with the permission of KS.
ET, MA, RM, and KS performed experiments and acquired and analyzed all data. KS designed the experiments, performed the experiments, and analyzed all the data. KS conceived and designed the experiments and wrote the first draft of the manuscript. ET, MA, RM, and KS analyzed and interpreted all data. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This study was approved by the Animal Care Committee of Ohu University (Approval Nos. 2022-34, 2023-14, and 2024-29). All animal procedures were conducted in accordance with the guidelines of the Animal Care Committee of Ohu University. The three Rs principles (Replacement, Reduction, and Refinement) were implemented throughout the present study by adopting the principles of laboratory animal care to minimize distress and utilizing the minimum number of required animals for all experiments. This study was carried out in compliance with the ARRIVE guidelines. We strictly adhered to the principles of laboratory animal care to minimize animal stress and use the minimum number of animals necessary for the experiments.
We thank Fuka Ohnuma for technical support for western blotting and gratefully acknowledge the help of past and present laboratory members.
This work was supported by Lead Chemical Co., Ltd. (Number of Sponsored Research Agreement (SRA) is RML171201S) and a research grant from the School of Pharmaceutical Sciences, Ohu University (KS).
The authors declare the following financial and personal relationships that could be perceived as potential competing interests: KS has received research funding from Lead Chemical Co., Ltd. All other authors declare no conflicts of interest. No employees of Lead Chemical Co., Ltd. were involved in data analysis, peer review selection, or any editorial decisions related to this manuscript.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/JIN42403.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





