1 Institute of Acupuncture and Moxibustion, Shandong University of Traditional Chinese Medicine, 250300 Jinan, Shandong, China
2 Innovation Institute of Chinese Medicine and Pharmacy, Shandong University of Traditional Chinese Medicine, 250300 Jinan, Shandong, China
3 Key Laboratory of Traditional Chinese Medicine Classical Theory, Ministry of Education, Shandong University of Traditional Chinese Medicine, 250300 Jinan, Shandong, China
4 Shandong Provincial Engineering Research Center for the Prevention and Treatment of Major Brain Diseases with Traditional Chinese Medicine, Shandong University of Traditional Chinese Medicine, 250300 Jinan, Shandong, China
†These authors contributed equally.
Abstract
Declining fertility due to ovarian dysfunction is a critical concern for population sustainability. Acupuncture shows potential for ovarian functional restoration, primarily mediated through its neuromodulatory effects on the hypothalamic-pituitary-ovarian (HPO) axis. Accumulating evidence from clinical and animal studies suggests that acupuncture modulates ovarian function through multiple neuroendocrine pathways. Specifically, acupuncture regulates hypothalamic activity by influencing the release of reproduction-associated neuropeptides (e.g., kisspeptin, neuropeptide Y, and gonadotropin-releasing hormone) and neurotransmitters (e.g., gamma-aminobutyric acid, serotonin, glutamate, and dopamine). Additionally, it may affect peripheral-to-central sensory nerve pathways as well as sympathetic and parasympathetic neural activity, which collectively enhance ovarian function. In this article, we comprehensively reviewed the neuroendocrine regulatory pathways through which acupuncture exerts its therapeutic effects in ovarian diseases, thereby providing a deeper understanding of the mechanisms involved and the directions for future research.
Keywords
- ovarian diseases
- acupuncture
- neuroendocrine
- hypothalamic-pituitary-ovarian (HPO) axis
Ovarian disorders refer to a category of pathological conditions affecting ovarian structure, function, or hormone secretion, which may lead to reproductive dysfunction, endocrine imbalances, and systemic complications. Delayed childbearing, exposure to environmental pollution, and the increasing incidence of malignant diseases in younger individuals are contemporary factors affecting female ovarian health [1]. Current clinical treatments for ovarian diseases include hormone therapy, ovulation induction with medications, insulin sensitizers, and other pharmacological approaches. Among these, estrogen and progesterone replacement therapy is the classic intervention for premature ovarian failure and ovarian insufficiency, and it is currently the first-line recommendation in major clinical guidelines [2]. However, long-term estrogen exposure may increase the risk of venous thrombosis and ovarian cancer. Discontinuation of hormone replacement therapy may lead to a recurrence of symptoms associated with ovarian function decline, and this underlying decline is often irreversible [3]. Assisted reproductive technologies utilize various cryopreservation techniques to protect reproductive cells and ovarian tissue, helping infertile couples achieve pregnancy [4]. However, following ovarian tissue cryopreservation and transplantation, significant follicular loss occurs due to ischemia, which shortens the lifespan of the transplanted tissue [5, 6]. Approximately 80% of follicles are lost during ovarian tissue cryopreservation and transplantation procedures. For women of advanced age or those with low ovarian reserve, ovarian cryopreservation may accelerate ovarian failure [7]. Although in vitro fertilization and intracytoplasmic sperm injection improve the chances of conception, they also increase the risk of scar tissue formation, infections, bleeding, and uterine rupture in future pregnancies, in addition to being costly [8, 9]. Currently, no single therapy stands out as superior, and a range of interventions, including acupuncture, are being tested worldwide.
As a minimally invasive modality, acupuncture carries minimal risks of physical
trauma or infection when performed under strict aseptic protocols, with reported
adverse events typically being mild and self-limiting [10, 11, 12, 13]. Clinical
observations up to May 2023 indicate that compared with clomiphene monotherapy,
adjunctive acupuncture therapy reduces the incidence of luteinized unruptured
follicle syndrome (LUFS) and ovarian hyperstimulation syndrome (OHSS) while
alleviating clomiphene-associated adverse effects such as nausea, vomiting,
headache, and dermatitis [14]. Nevertheless, methodological heterogeneity among
existing studies necessitates further validation of acupuncture’s safety profile
and adverse event rates in ovarian disorder management through rigorously
controlled trials. Acupuncture modulates the neuroendocrine function of the
hypothalamic-pituitary-ovarian (HPO) axis [15, 16] by influencing hypothalamic and
circulating neuropeptides (e.g., kisspeptin [17] and
The interaction between the central nervous system (CNS) and endocrine system helps maintain ovarian function, including follicular development, maturation, and ovulation [24]. The neuroendocrine control of ovarian function is regulated by the HPO axis, with multiple internal and external cues integrated through neural networks in the brain. Gonadotropin-releasing hormone (GnRH) released from the hypothalamus serves as the ultimate brain signal regulating reproduction, and neuropeptides in the hypothalamus play an indispensable role in the regulation of GnRH [17, 25]. Kisspeptin neurons in the arcuate nucleus co-express kisspeptin, neurokinin B (NKB), and Dynorphin (DYN) [26]. NKB belongs to the tachykinin family and primarily interacts with neurokinin-3 receptor to activate kisspeptin release [27]. DYN inhibits kisspeptin secretion by binding to the kappa opioid receptor [28]. The activating effect of NKB and inhibitory effect of DYN are essential for the coordination of kisspeptin secretion [27, 29]. Kisspeptin acts directly on GnRH neurons via G protein-coupled receptor 54 (GPR54), causing the pulsatile release of GnRH from the pituitary portal blood system to the anterior pituitary gland [30, 31]. Specific hormone-producing cells in the anterior pituitary release follicle-stimulating hormone (FSH) and luteinizing hormone (LH) into the bloodstream, thereby regulating gonadal activity [32]. However, dysregulated hypothalamic DYN secretion leads to increased GnRH pulses, which in turn induces excessive LH secretion, irregular follicular development, and ovulatory difficulties, causing disorders such as polycystic ovary syndrome (PCOS) and precocious puberty [33, 34]. Conversely, insufficient hypothalamic GnRH pulse secretion results in underdeveloped breasts, immature vulva, and primary amenorrhea, affecting the normal development and release of oocytes, and potentially leading to infertility or recurrent miscarriage [35]. Additionally, the abnormal central feedback effect of estradiol (E2) can disrupt the balanced operation of the HPO axis, resulting in reduced fertility and gradual decline in the functions of multiple organs [36, 37]. This issue has been discussed in greater detail in articles by Sang et al. [38] and Dufour et al. [39].
In addition to neuropeptides, neurotransmitters such as GABA, glutamate (Glu), and serotonin (5-HT) help regulate GnRH secretion in the hypothalamus [40, 41, 42]. In red seabream, it has been well established that 5-HT, GABA, and neuropeptide Y promote the release of GnRH [43]. Inhibition of the GABA content in the median eminence of female monkeys by either antisense oligodeoxynucleotides or dinucleotides against GAD67 mRNA, which encodes glutamic acid decarboxylase 67, acutely inactivates GABA type A receptors or reduces GABA mRNA levels, leading to immediate GnRH release and early onset of menarche and ovulation [44]. Additionally, dopamine shares many signaling components with GnRH receptors. While GnRH activates phospholipase C [45], dopamine is known to exert inhibitory effects by preventing the production of inositol 1,4,5-trisphosphate and diacylglycerol, thus counteracting the GnRH-induced increase in intracellular calcium concentration and reducing protein kinase C and mitogen-activated protein kinase (MAPK) activity [46]. Interestingly, MAPK activity is considered a mediator of GnRH-induced stimulation of gonadotropin subunit gene transcription [47]. Furthermore, Glu activates N-methyl-D-aspartate receptors [48], protein kinases (calcium/calmodulin-dependent protein kinase) [49], phosphatidylinositol 3-kinase [50], and MAPK pathways [51]. These pathways collectively promote the synthesis and release of GnRH. Notably, the dynamic balance between central neurotransmitters and endocrine cells in regulating sex hormone levels and the occurrence of ovarian function involves Glu, which inhibits the release of hypothalamic and blood GnRH [52], while GABA and 5-HT promote GnRH release [43]. These findings elucidate the pivotal role of GnRH dysregulation-induced gonadal steroid imbalance within the HPO axis in the pathogenesis of ovarian disorders, while highlighting the dynamic interplay between hormone receptor expression profiles and disease progression.
Previous studies have focused on the regulation of GnRH secretion by acupuncture, which influences the release of LH and FSH by the pituitary gland [17, 53, 54]. However, there has been little research on the effects of acupuncture on the upstream regulation of GnRH. GnRH secretion is regulated by the three key neurotransmitters secreted by the upstream neurons (kisspeptin, neuropeptide Y (NPY), and DYN) [39], and by various other neurotransmitters such as GABA, Glu, 5-HT, and dopamine (DA) [55, 56, 57, 58, 59]. The following section summarizes the role of acupuncture in regulating the neuroendocrine balance in the HPO axis from the perspective of neuropeptides and neurotransmitters.
Kisspeptin, encoded by the Kiss1 gene, acts on the hypothalamus and is a key neuropeptide involved in the regulation of GnRH and reproductive processes [30, 60]. Studies have shown that acupuncture at Guanyuan (CV4) and Zigong (EX-CA1) can regulate the GnRH/kisspeptin-GPR54 signaling pathway in the hypothalamus of ovariectomized female rats [61]. This mechanism mimics the effect of administering small doses of E2 to increase the secretion of GnRH, GPR54, and kisspeptin-1 in the hypothalamus of ovariectomized rats, thereby exciting the hypothalamic kisspeptin-GPR54 signaling pathway [31]. This promotes the expression of corticotropin-releasing hormone mRNA and release of corticotropin-releasing hormone in the hypothalamus, leading to increased peripheral blood E2 levels and improved functional activity of the HPO axis in ovariectomized rats [62]. This may be one of the mechanisms underlying the improvement of ovarian dysfunction by acupuncture. Additionally, after electroacupuncture treatment, increased kisspeptin protein expression was detected in the ovaries of PCOS rats, and the levels of serum sex hormones (LH and FSH), ovarian granulosa cells, and secretory function of theca cells were improved [37, 63]. This suggests that the improvement in ovarian function in PCOS rats by electroacupuncture may be related to the kisspeptin/kiss1r system. Thus, acupuncture can promote the secretion of kisspeptin in the hypothalamus, activate the HPO axis, and stimulate the release of hormones such as GnRH, LH, and FSH, thereby facilitating follicular development and ovulation, which plays a therapeutic role in the treatment of ovarian diseases (Fig. 1 and Table 1, Ref. [20, 64, 65, 66, 67, 68, 69, 70, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81]).
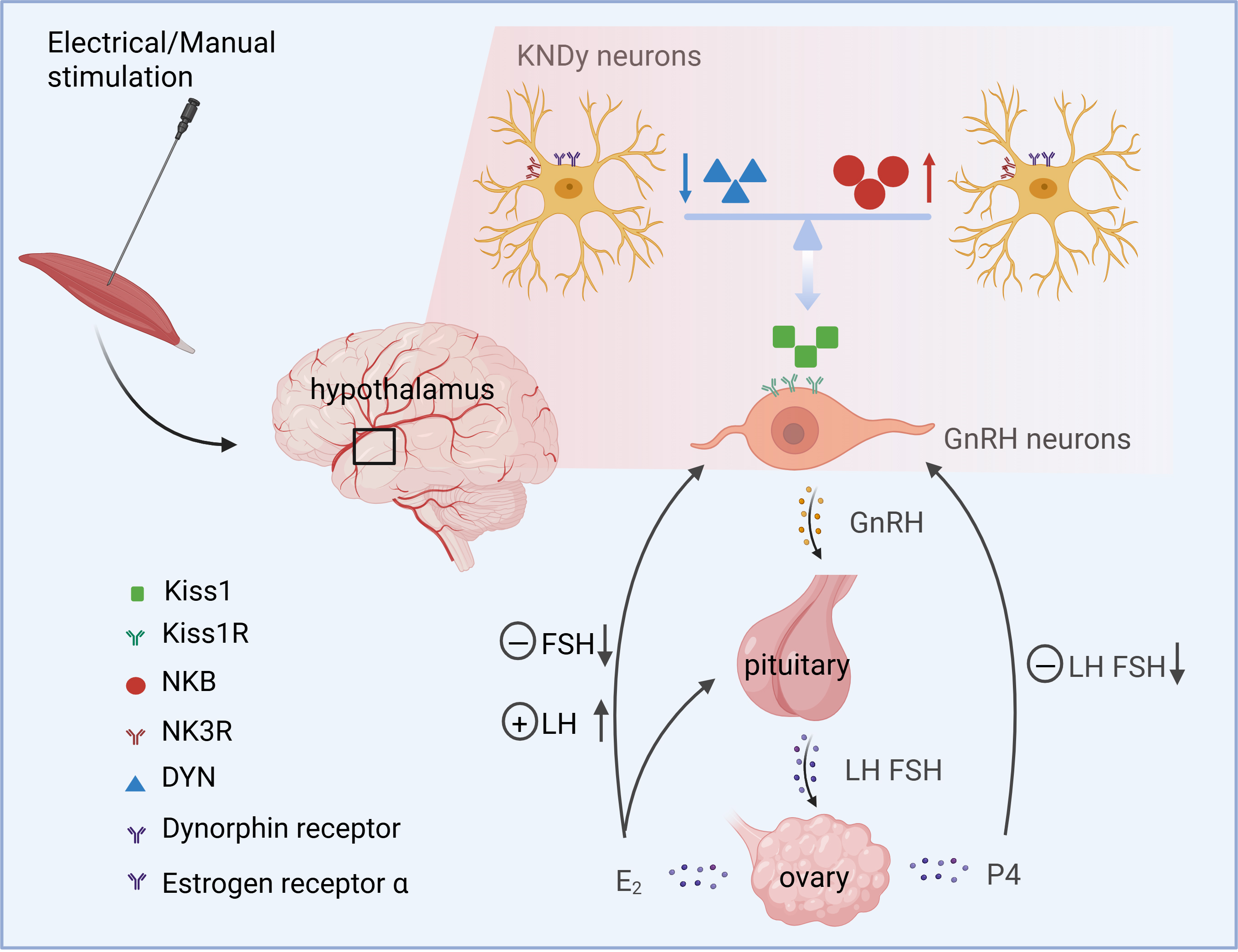 Fig. 1.
Fig. 1.
Acupuncture modulates ovarian neuroendocrine regulation through
the HPO axis. Acupuncture modulates NK3 and Dynorphin receptors on
kisspeptin/neurokinin B/dynorphin (KNDy) neurons in the hypothalamus, thereby
regulating GnRH release and influencing LH, FSH, progesterone (P4), and estradiol
(E2) levels in the hypothalamic-pituitary-ovarian (HPO) axis. Figure created with BioRender (https://www.biorender.com).
| Acupuncture type | Animal type | Acupoint | Disease | Target | Alterations in signaling | Reference |
| EA | Rat | CV3, CV4 | PCOS | Ovary and serum | AMH↓ FSH↑ E2↑ T↓ | [64] |
| EA | Rat | ST29, SP6 | PCOS | Serum | FSH↑ LH↓ T↓ | [65] |
| EA | Rat | ST29, SP6 | PCOS | Serum | P↑ | [20] |
| EA | Rat | ST29, SP6 | PCOS | Serum | T↓ FAI↓ LH↓ LH/FSH↓ AMH↓ INHB↓ FINS↓ E2↑ FSH↑ SHBG↑ | [66] |
| EA | Rat | ST29, SP6, LR3, PC6 | PCOS | Serum | P↑ E2↑ DHT↓ | [67] |
| MA | Rat | CV4, RN3, CV6, SP6, EX-CA1 | PCOS | Serum | LH↓ AMH↓ FSH↑ T↓ E2↑ | [68] |
| EA | Rat | ST29, SP6 | PCOS | Serum | T↓ LH↓ FSH↑ LH/FSH↓ | [69] |
| Mox | Rat | BL23, CV4, CV8 | POI | Serum | E2↑ FSH↓ LH↓ AMH↑ | [70] |
| EA | Rat | CV4, ST36, SP6 | POF | Serum | E1↑ E2↑ E3↑ TSH↑ T↓ DHEA↓ 17-OHP↓ 21D↓ | [71] |
| EA | Rat | CV4, EX-CA1, CV6 | PCOS | Serum | FSH↑ LH↑ T↑ E2↑ P↑ | [72] |
| Mox | Rat | CV4, RN12, BL23 | DOR | Serum | E2↑ AMH↑ FSH↓ LH↓ | [73] |
| MA | Rat | Option 1: CV4, SP6, CV3, ST29; Option 2: GB20, DU16, BL10, GB12; Option 3: CV4, SP6, BL23, LR3, GV20; Option 4: CV4, SP6 | POF | Serum | E2↑ FSH↑ LH↑ | [74] |
| MA | Rat | CV4, SP6, ST36, BL20, BL23, EX-CA1 | POF | Serum | E2↑ FSH↓ LH↓ | [75] |
| EA | Rat | ST36, SP6, PC6 | PCOS | Serum | E2↓ T↓ P↓ LH↓ FSH↓ | [76] |
| Mox | Rat | CV4, SP6 | POF | Serum | FSH↓ LH↓ E2↑ | [77] |
| Mox | Rat | ST36 | 10 months | Serum | FSH↑ E2↑ | [78] |
| EA | Mice | CV4, ST36, SP6 | POF | Serum | E1↑ E2↑ E3↑ | [79] |
| EA | Rat | CV4, EX-CA1, SP6 | Thin endometrium | Uterus | ER↑ PR↑ | [79] |
| EA | Rat | SP6, LR3, ST36 | Controlled ovarian hyperstimulation | Uterus | ER↑ PR↑ | [80] |
| EA | Mice | CV4, SP6 | SAMP8 | Serum | E2↑ GnRH↓ LH↓ FSH↓ | [81] |
EA, electroacupuncture; MA, Manual-acupuncture; Mox, moxibustion; CV4, Guanyuan;
SP6, Sanyinjiao; ST36, Zusanli; CV3, Zhongji; ST29, Guilia; GB20, Fengchii; DU16,
Fengfu; BL10, Tianzhu; GB12, Wangu; BL23, Shenshu; LR3, Taichong; GV20, Baihui;
DU2, Yaoshu; KI3, Taixi; EX-CA1, Zigong; LI4, Hegu; LR3, Taichong; PC6, Neiguan;
BL20, Pishu; ST25, Tianshu; ST29, Guilai; T, testosterone; FSH,
follicle-stimulating hormone; LH, luteinizing hormone; INS, insulin; AMH,
anti-Müllerian hormone; INHB, inhibin B; DHT, dihydrotestosterone; FGLU,
fasting blood glucose; FINS, fasting blood insulin; HOMA-IR, homeostasis model
assessment-insulin resistance; E1, estrone; E3, estriol; TSH,
thyroid-stimulating hormone; T, testosterone; DHEA, dehydroepiandrosterone;
17-OHP, 17-hydroxyprogesterone; 21D, 21-deoxycortisol; SHBG, sex hormone-binding
globulin; P, progesterone; FAI, free androgen index; PCOS, polycystic ovary
syndrome; POI, premature ovarian insufficiency; POF, premature ovarian failure;
Neuropeptide Y (NPY), which is widely distributed in the central nervous (e.g.,
hypothalamus and amygdala) and peripheral nervous systems, is closely associated
with metabolic cues and fertility regulation [82] (Table 2, Ref.
[15, 17, 63, 83, 84, 85, 86, 87]). During labor, NPY levels continuously increase from
birth until cervix dilatation occurs. Additionally, a surge of immunoreactive NPY
is released from the portal vein into the pituitary portal circulation before
puberty, followed by a surge of LH [88]. Notably, acupuncture has been reported
to exert multifaceted therapeutic effects in ovarian disorders through NPY.
Mechanistic studies demonstrate that EA significantly enhances
hypothalamic-ovarian axis function in PCOS rodent models by downregulating
hypothalamic NPY mRNA and NPY2 receptor expression. This, in turn, suppresses
sympathetic hyperactivity and reduces adipose tissue NPY overexpression, thereby
ameliorating polycystic ovarian morphology and endocrine dysregulation [83, 84].
Moreover, molecular profiling reveals that EA modulates prostaglandin
PGF2
| Acupuncture type | Animal type | Acupoint | Disease | Target | Alterations in signaling | Reference |
| EA | Rat | CV6, SP6, ST36 | PCOS | Hypothalamus (ARC) and serum | kisspeptin↓ GnRH↓ LH↓ E2↑ | [86] |
| EA | Rat | CV4 | PCOS | Hypothalamus (ARC) and serum | kisspeptin↓ GnRH↓ DYN↓ Kiss1↑ Ar↑ E1↑ Kiss1r↑ T↓ LH↓ | [17] |
| EA | Rat | SP6, ST36, CV6 | PCOS | Hypothalamus (ARC) and serum | NPY↓ GnRH↓ | [84] |
| EA | Rat | The abdominal and hindlimb muscles, in somatic segments corresponding to the innervation of the ovaries | PCOS | Mesenteric adipose tissue | NPY↓ GnRH↓ NGF↓ ADRB3↓ | [83] |
| EA | Rat | CV4, SP6 | OHSS | Ovary and serum | PGE2↓ COX-2↓ PKA/CREB↓ | [85] |
| EA | Rat | CV4 | PCOS | Ovary and serum | LH↓ T↓ kisspeptin↓ E2↑ | [63] |
| EA | Women | — | POF | Ovary | [15] | |
| EA | Rat | GB26 | PCOS | Hypothalamus and serum | kisspeptin↓ LH↓ E2↓ T↓ | [87] |
CV6, Qihai; GB26, Daimai; ST36, Zusanli; FSH, follicle-stimulating hormone; GnRH,
gonadotropin-releasing hormone; DYN, Dynorphin; Ar, androgen receptor; OVX,
ovariectomized; SAMP8, senescence-accelerated
mouse-prone 8; ADRB3, beta(3)-adrenergic receptor; NGF, nerve growth factor; NPY,
neuropeptide Y; OHSS, ovarian hyperstimulation syndrome;
The hypothalamus releases GnRH in a pulsatile manner to act on the anterior pituitary, which constitutes the final common pathway for the central regulation of fertility [90] (Tables 1,2). Research has found that electroacupuncture at the CV4 acupoint can promote the expression of GnRH and Kiss1 mRNA, and significantly increase serum LH and E2 levels. An increase in LH and FSH recruits and stimulates the growth and maturation of follicles, whereas insufficient levels of LH and FSH disrupt the growth of dominant follicles, leading to the impaired development of primordial follicles [17]. However, excessive expression can stimulate androgen synthesis in ovarian follicular cells, causing hyperandrogenism, which leads to hirsutism, acne, and alopecia in patients with PCOS. Acupuncture at CV4 can reverse the serum levels of LH and FSH in patients with PCOS [91]. In an ovariectomized rat model, electroacupuncture increased hypothalamic GnRH mRNA expression and mRNA levels of pituitary GnRH receptor, thereby correcting the dysfunction of the HPG axis [92]. In addition, acupuncture CV4, Qihai (CV6), Zhongji (CV3), bilateral Dahe (KI12), bilateral Shenshu (BL23), bilateral Zusanli (ST36), bilateral Sanyinjiao (SP6) and bilateral Taichong (LR3) in combination with GnRH analogue improved clinical pregnancy rates and improves pregnancy outcomes in IVF-ET RIF patients [93]. Thus, acupuncture at specific acupoints bidirectionally affects hypothalamic GnRH release and hormone levels, resulting in effective treatment of ovarian disorders and improved success rates of assisted reproductive technologies.
Current research evidence demonstrates that
Neurotransmitters such as norepinephrine (NE), GABA, 5-HT, Glu, and dopamine may
interfere with the pulsatile secretion of GnRH, leading to dysregulation of the
HPO axis. A randomized controlled trial involving patients with PCOS demonstrated
that 16 weeks of EA significantly elevated serum NE levels while reducing 5-HT
concentrations. Although GABA levels showed a downward trend, no statistically
significant intergroup difference was observed [19]. Sequencing of the anterior
ventral periventricular nucleus (AVPV) of the hypothalamus in
early-reproductively senescent female rats showed that EA significantly affected
neuroactive ligand-receptor interactions, glutamatergic, and
| Acupuncture type | Animal type | Acupoint | Disease | Target | Alterations in signaling | Reference |
| EA | Women | CV3, CV6 | Perimenopause | Serum | NE↑ 5-HT↓ GABA↓ | [19] |
| EA | Rat | CV4, SP6 | Reproductive aging | Brain and serum | LH↑ hormone receptor Pgr↑ adrenoceptor Adra1a↑ neurotransmitter transporters Slc17a6↑ Slc32a1↑ glutamate decarboxylase Gad2↑ Kiss1↑ | [99] |
| MA | Rat | CV4, SP6, EX-CA1 | Menopausal | Serum | ACTH↑ 5-HT↓ NE↓ | [100] |
| EA | Rat | CV4, CV3, SP6, EX-CA1 | OVX | Serum | GABA↑ |
[101] |
DA, Dopamine; 5-HT, 5-hydroxytryptamine; NE,
Noradrenaline;
One of the main mechanisms of acupuncture involves stimulation of receptors at the acupuncture site, transmission of neural impulses through the corresponding afferent nerves to the dorsal horn of the spinal cord, and then to the hypothalamus, eventually influencing neurotransmitter release and modulating the function of the HPO axis [102]. Meanwhile, acupuncture activates the sympathetic and vagal pathways, which is one of the mechanisms that regulate the neuroendocrine system and maintains the balance of ovarian hormone levels [103]. We summarized the schematic diagram of the complex pathway of action of acupuncture stimulation from peripheral-central-ovarian as shown in Fig. 2.
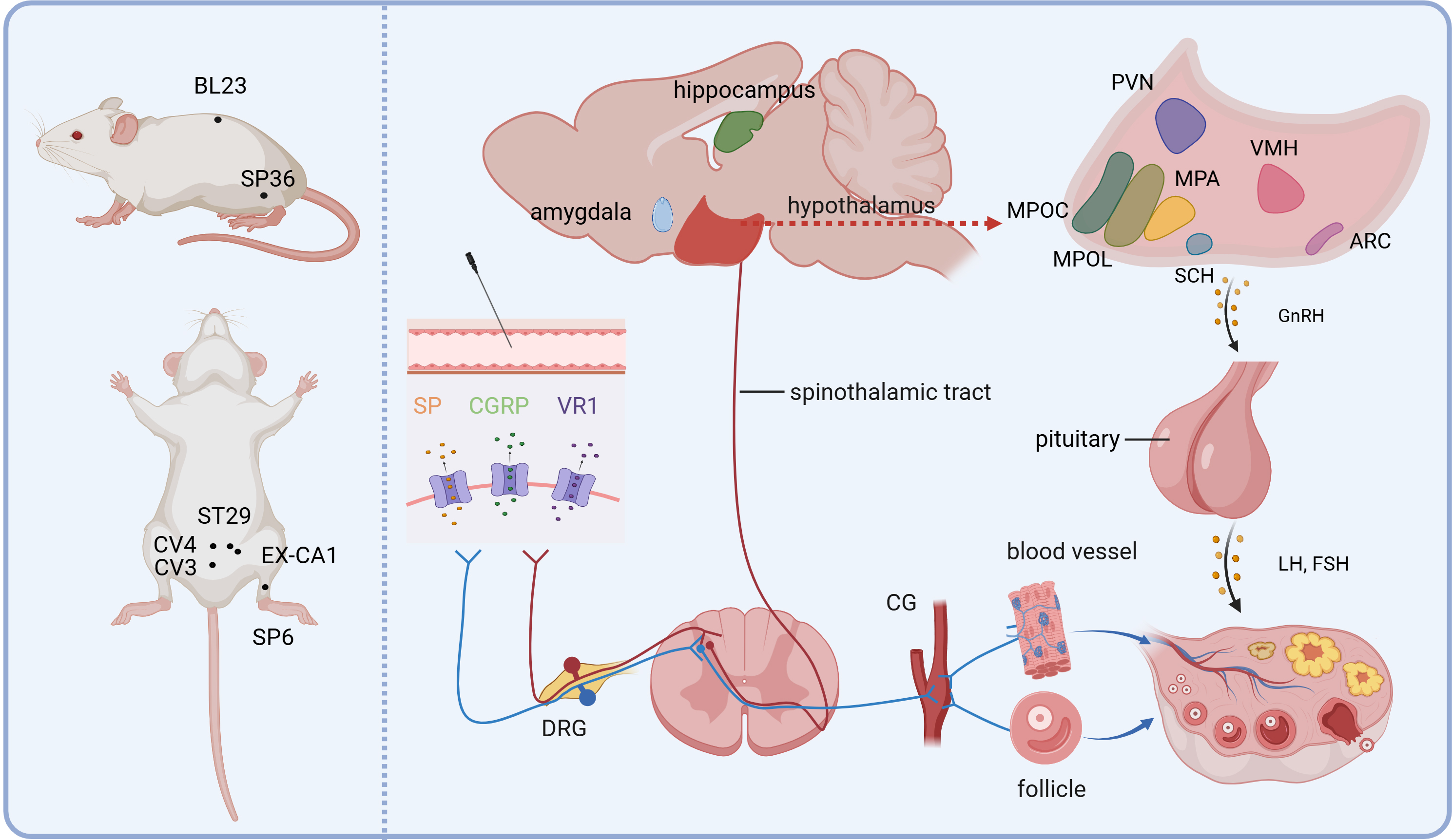 Fig. 2.
Fig. 2.
Peripheral-central-target-organ afferent pathways in the regulation of ovary by acupuncture. The primary acupoints used for treating female reproductive disorders are identified on the left. The peripheral-central neural conduction mechanisms involved in acupuncture stimulation are illustrated on the right. Figure created with BioRender (https://www.biorender.com). SP, Substance P; CGRP, calcitonin gene-related peptide; VR1, Vanilloid receptor subtype 1; DRG, dorsal root ganglion; CG, coeliac ganglia; PVN, Paraventricular Hypothalamic Nucleus; VMH, Ventromedial Hypothalamic Nucleus; MPA, medial preoptic area; MPO, medial preoptic nucleus; MPOL, medial preoptic nucleus, medial part; ARC, arcuate hypothalamic nucleus; SCH, suprachiasmatic nucleus.
Acupuncture involves biomechanical interactions between the inserted needle and
the multi-layered tissue microenvironment of acupoints, which includes skin,
muscle, and connective tissue. During needle manipulation, mechanical signals are
generated through dynamic tissue responses such as collagen fiber deformation,
elastic stretching of the fascial network, and interstitial fluid redistribution.
This may be one of the mechanisms by which the needling effect is produced. These
responses may trigger distinct mechanical conduction pathways depending on the
needling technique (e.g., lifting-thrusting, twisting, or electroacupuncture).
This cause is one of the mechanisms by which the effects of needling are
produced. For example, a study demonstrated that acupuncture at the CV4 acupoint
activates both myelinated
The spinal cord plays a pivotal role in transmitting sensory information during acupuncture, acting as a ‘hub’ that links the peripheral and central nervous systems [109]. Studies indicate that the spinal nerve segments related to the reproductive system are primarily located in the T10–L2 and L6–S2 regions [110, 111, 112]. After injecting PRV into the ovary, PRV expression was detected in the dorsal root ganglia of the T12–L2 spinal segments [113]. In an experimental study using Evans blue dye to detect plasma extravasation points in the skin of rats with ovarian inflammation, researchers found that ovarian inflammation can lead to the appearance of plasma extravasation points in specific areas of the body surface. These areas not only correspond to the spinal ganglion segments that innervate the ovaries but also are concentrated in acupoint regions such as CV4-EX-CA1 and BL23-GV4, which are known to be used for treating ovarian diseases [102]. These findings suggest that the therapeutic effects of acupoints on ovarian dysfunction may arise from neuroanatomical convergence, where disease-sensitive acupoints share spinal segmental innervation with ovarian regulatory pathways.
Acupoint specificity implies that different spinal nerve segments are associated with different acupoints and thereby potential therapeutic applications in various types of disorders. Clinically, acupoints commonly used to treat ovarian disorders are predominantly located in the lumbar-abdominal region and lower limbs, including CV4, SP6, ST36, and EX-CA1 [114]. For instance, studies investigating the nerve segment distribution of the CV4 point in rats found that an injection of cholera toxin B subunit-coupled horseradish peroxidase (HRP) into the CV4 point and subsequent acupuncture led to labeled cells in bilateral T11–L3 dorsal root ganglia, with the highest number of labeled cells observed in the L2 segment, suggesting that L2 is the primary pathway for signal transmission from the CV4 acupoint to the spinal cord [115]. Studies have also indicated that the nerve segments for SP6 are between L2 and S3 and those for ST36 are between L4 and S3, which are proximal to the nerve segments governing reproductive organs, and exert comprehensive regulatory effects on the neuroendocrine network [23]. The nerve segments for EX-CA1 are between T12 and L1, and can effectively modulate the HPO axis in endocrine disorders [23, 116].
Furthermore, electroacupuncture at different points can target different aspects of the same disease, for instance, electroacupuncture at ST36 exhibits advantages in improving hormone levels and synthesis of sex hormone-binding globulin, whereas that at SP6 shows relatively pronounced improvements in ovarian tissue morphology and androgen receptor expression. Electroacupuncture at CV4 demonstrated significant benefits in both areas, and has considerable therapeutic advantages [117]. In conclusion, the neuroanatomical precision of acupuncture manifests through its segment-specific regulation of ovarian steroidogenesis. Selective acupoint stimulation achieves segmental neuromodulation within thoracolumbar spinal circuits, establishing an evidence-based approach for optimizing ovarian hormonal homeostasis.
The neural activity in brain regions is closely associated with reproductive hormone levels. EA precisely regulates neuroendocrine signaling of the HPO axis and can treat ovarian lesions. The pathogenesis of primary dysmenorrhea, premenstrual syndrome (PMS), and premature ovarian insufficiency (POI) is associated with abnormalities in the central brain area where EA regulates the HPO axis. Using resting-state functional magnetic resonance imaging to observe brain changes in patients with diminished ovarian reserve (DOR), the study found increased Amplitude of low-frequency fluctuations (ALFF) values in the left inferior temporal gyrus (ITG) and middle temporal gyrus (MTG) in DOR patients, along with decreased ALFF values in the bilateral superior frontal gyrus (SFG), middle frontal gyrus (MFG), and left inferior frontal gyrus (IFG) compared to healthy controls. Acupuncture treatment increased ALFF values in the left SFG, MFG, and supplementary motor area (SMA), and enhanced functional connectivity between the left SFG and the bilateral calcarine sulcus and lingual gyrus [118]. In patients with PMS, a decrease in functional connectivity density was noted in the dorsolateral prefrontal cortex and anterior part of the inferior frontal gyrus, with increased connectivity in the medial prefrontal cortex. Furthermore, connectivity between the prefrontal cortex and thalamus was reduced, indicating impaired cooperation between the right ventricle and right prefrontal cortex, which may affect HPO function [119]. Differences in functional connectivity between the hypothalamus and whole brain areas in patients with premature ovarian insufficiency and healthy volunteers, and increased connectivity between the right ventricle and left anterior frontal gyrus, right anterior frontal gyrus, and left insula after acupuncture treatment in patients with premature ovarian insufficiency were observed [120]. Thus, acupuncture can modulate the strength of the functional connectivity in the hypothalamus and/or whole brain regions to improve reproductive endocrine function.
Animal experiments have provided a more detailed description of the brain regions involved in acupuncture modulation of the HPO axis. Two weeks after ovariectomy, the distribution of Fos protein-labeled neurons was observed in brain regions, including the medial preoptic nucleus (MPN), lateral preoptic nucleus, and suprachiasmatic nucleus. Electrical acupuncture at CV4 resulted in abundant specific Fos-labeled cells in the lateral preoptic nucleus, paraventricular nucleus (PAV [ventral part]), and suprachiasmatic nucleus, especially in the arcuate nucleus (ARC), PAV, and MPN, with no labeling observed in the MPN [121]. Furthermore, acupuncture at Danzhong (CV17), Zhongwan (RN12), and CV4 stimulated the neurons associated with unknown sexual arousal in the medial preoptic area, ARC, and ventral part, with the highest stimulation observed in the medial preoptic area, followed by the arcuate and paraventricular nuclei [23]. Studies on HRP virus tracing and GnRH immunohistochemistry indicate substantial overlap between HRP-traced neurons associated with sexual arousal stimulation and sites expressing positive GnRH immunoreactivity [115]. These findings highlight that the hypothalamus is the pivotal brain region in the acupuncture treatment of ovarian disease, with potential central targets in the amygdala and hippocampal regions. Acupuncture can restore the HPO axis function by modulating cooperative interactions between the hypothalamus and multiple brain regions.
Acupuncture treatment of ovarian disorders involves complex neuroendocrine interactions. Acupuncture can influence the autonomic nervous system (ANS) and subsequently regulate the HPO axis by stimulating specific acupoints [122]. The sympathetic nerve fibers of the ovaries primarily originate from the celiac ganglion. A portion of these fibers projects to the ovarian blood vessels through the ovarian plexus, while another portion passes through the ovarian nerve (SON) to innervate the ovarian follicles [95]. Acupuncture can modulate ovarian blood flow by stimulating ovarian sympathetic nerve reflexes, and ongoing acupuncture treatment positively affects hyperandrogenemia, menstrual cycles, and ovulation rates in women with PCOS [123]. Studies have shown that electroacupuncture increases ovarian blood flow and mean arterial pressure. After severing the ovarian sympathetic nerves, the ovarian blood flow response and mean arterial pressure are significantly decreased, indicating that the electroacupuncture-induced increase in ovarian blood flow was mediated by a reflex through the ovarian sympathetic nerves [124]. Additionally, SON removal reduces serum E2 and progesterone levels, and the number of developing follicles. When unilateral SON denervation is performed, the number of follicles decreases in the denervated ovary, whereas compensatory follicle numbers increase in the innervated ovary [20].
In addition to sympathetic innervation via the SON and ovarian plexus, the ovary
receives parasympathetic innervation through the vagus nerve [125]. Research has
shown that electroacupuncture modulates the vagus nerve to stimulate
This review is the first to summarize the mechanism underlying the therapeutic effects of acupuncture in ovarian disease from the perspective of neuroendocrine and neural pathways. Acupuncture modulates GnRH pulses, thereby balancing GnRH and its downstream hormones (LH, FSH, and E2). It regulates the release of hypothalamic neuropeptides and neurotransmitters, thus modulating hormone levels in the HPO axis and maintaining reproductive homeostasis. Furthermore, acupuncture involves two main neural pathways. The first one is the somatosensory nerve pathway, where signals travel from acupoints to the dorsal root ganglia and spinal cord and then to the central structures (such as the preoptic area, ARC, and paraventricular hypothalamic nucleus (PVN)) before acting on the ovaries. The other pathway involves the autonomic nervous system, including the sympathetic and vagal nerves, which improve blood flow to the ovaries, inflammation, and hormone levels, thus regulating HPO axis function. These mechanisms suggest promising therapeutic targets for acupuncture in the treatment of ovarian diseases.
This study acknowledges several limitations: (1) Mechanistic research dimensionality constraints: while existing studies have confirmed acupuncture’s multi-pathway regulation of the HPO axis through neuropeptides (Kisspeptin and NPY), neurotransmitters (GABA and 5-HT), and the autonomic nervous system, the molecular mechanisms and specific regulatory networks remain to be systematically elucidated. It is recommended to integrate cutting-edge methodologies—such as neurotropic viral tracing (e.g., transsynaptic pseudorabies virus), TESOS-based three-dimensional ultrastructural imaging, and whole-brain functional connectivity mapping (fcMRI)—to systematically delineate the precise structural and functional networks bridging acupoints, central neural circuits, and ovarian physiology. (2) Deficient cross-system synergy mechanisms: current investigations on neuroendocrine regulatory mechanisms in ovarian disorders, particularly interaction patterns under distinct pathological conditions, are predominantly hormone-centric. It is imperative to leverage convergent methodologies, such as organ-on-a-chip-based neuron-ovary co-cultures and CRISPR-based screening in conditional knockout models, to decode the multidimensional crosstalk between neuroendocrine circuits and immune effectors mediating acupuncture-driven amelioration of ovarian pathologies. (3) Insufficient experimental standardization: current animal studies investigating acupuncture for ovarian dysfunction exhibit significant methodological heterogeneity, such as diverse POI modeling approaches (e.g., cyclophosphamide, cisplatin, busulfan) with inconsistent dosage regimens and treatment durations, coupled with the prevalent use of fixed acupoint combinations and suboptimal stimulation parameters in acupuncture interventions. To address these limitations, we recommend establishing pathology-specific standardized modeling guidelines (e.g., POI vs. PCOS) and implementing integrated evaluation frameworks incorporating multi-omics biomarkers (anti-Müllerian hormone (AMH), ovarian transcriptomic profiles) and functional imaging modalities such as ultrasound-based dynamic follicular monitoring. By implementing these comprehensive strategies, we will be able to systematically and multidimensionally analyze the regulatory mechanism of acupuncture on ovarian diseases.
E2, estradiol; FSH, follicle-stimulating hormone; LH, luteinizing hormone; CNS, central nervous system; NE, neuroendocrine; GnRH, gonadotropin releasing hormone; NKB, neurokinin B; DYN, Dynorphin; KOR, kappa opioid receptor; GPR54, G protein-coupled receptor 54; PCOS, polycystic ovarian syndrome; HPG, hypothalamic-pituitary-gonadal; GABA, gamma-aminobutyric acid; Glu, glutamate; 5-HT, serotonin; MAPK, mitogen-activated protein kinase; DA, dopamine; CRH, corticotropin-releasing hormone; HPO, hypothalamic-pituitary-ovarian;
YJC designed the research study. YB, JLY and ZZ conceptualized and designed the study, designed the figure, performed the literature searches and drafted the manuscript. SX, FNC, YJZ and YCX collected and sorted references and provided help and advice on the table and figure. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This study was supported by National Key R&D Programme Funded Projects (No.2022YFC3500405), Shandong University of Traditional Chinese Medicine Doctoral Student Quality Improvement and Innovation Project (YJSTZCX2024004), and the Open Research Fund of the Key Laboratory of Classical Theory of Traditional Chinese Medicine, Ministry of Education.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


