1 Department of Neurology, The First Hospital of Anhui University of Science and Technology (Huainan First People's Hospital), 232000 Huainan, Anhui, China
2 Department of Neurology, Fu Yang Fifth People's Hospital, 236000 Fuyang, Anhui, China
3 Department of Neurology, Hefei Hospital Affiliated to Anhui Medical University, The Second People's Hospital of Hefei, 230000 Hefei, Anhui, China
4 Department of General Medicine, The First Affiliated Hospital of Anhui Medical University, 230000 Hefei, Anhui, China
Abstract
To explore the relationship between YKL-40 level, telomere length, and different subtypes of insomnia disorder.
A total of 145 individuals suffering from insomnia were enrolled and divided into four groups according to the insomniac subtypes: difficulty initiating sleep, early morning awakening, difficulty maintaining sleep, and mixed symptoms. Eighty healthy controls were also collected at the same time. Peripheral leukocyte genomic DNA was extracted, relative telomere lengths were measured using the real-time quantitative polymerase chain reaction method, and YKL-40 levels were determined using enzyme-linked immunoassay. Logistic regression modeling was used to analyze the correlation between different insomnia subtypes, YKL-40 level, and telomere length.
People with telomere lengths in the lowest tertile were more likely to have trouble falling asleep (odds ratio (OR) 2.13, 95% confidence interval (CI) 1.22–3.63; p = 0.03) and had a higher frequency of mixed symptoms (OR 1.49, 95% CI 1.30–2.81; p = 0.04). People in the highest tertile of YKL-40 level had an increased chance of waking up early (OR 2.98, 95% CI 1.54–5.33; p = 0.01) and more mixed symptoms (OR 1.47, 95% CI 1.22–2.79; p = 0.02). Furthermore, using receiver operating characteristic curve analysis, the area under the curve of YKL-40 level and telomere length was 0.806 and 0.746, respectively.
Telomere length in patients with difficulty initiating sleep and mixed symptoms was significantly shortened and the level of YKL-40 in people who have early morning awakening and mixed symptoms was significantly increased. Our findings provide the first evidence that leukocyte telomere length and YKL-40 level are individually linked to mixed symptoms.
Keywords
- insomnia disorder
- telomere length
- YKL-40
- difficulty initiating sleep
- early morning awakening
- difficulty maintaining sleep
Chronic insomnia disorder is defined by the International Classification of Sleep Disorders diagnostic manual, 3rd edition (ICSD-3), as a complaint of trouble falling asleep, staying asleep, or waking up excessively early with related daytime consequences, even with adequate opportunities and conditions for sleep [1].
Chronic insomnia disorder describes a persistent state of insomnia that lasts longer than 3 months and is defined by symptoms that appear more than 3 nights per week [2]. The clinical features of insomnia include trouble falling asleep, poor sleep maintenance, early awakenings, worse sleep quality, and shorter overall sleep duration along with associated fatigue, mood disorders, and decline in cognitive function. Approximately 30–35% of people globally report having signs of insomnia [3], and comparable prevalence figures are seen in epidemiological research conducted in other nations.
Information about the physiology of insomnia has been attained through several approaches, including investigating genetic, molecular, and cellular processes; brain circuitry; physiologic causes; sleep behaviors; and self-reporting. These findings have resulted in multiple models of insomnia. Sleep-wake characteristics, including the length and timing of sleep, are inherited and controlled by several genes. Research on humans and animals has also linked genetic processes to insomnia [4]. Aging has attracted the attention of both researchers and the general public. A cross-sectional study of the Canadian Longitudinal Study on Aging has shown that insomnia disorder may increase the risk of further cognitive decline [5]. Anxiety and sadness are directly linked to insomnia, and those who struggle with insomnia experience various mental health issues, such as concentration, attention, or memory deficits, which are often reported as the first symptom to a doctor [6]. Multiple investigations have linked sleeplessness to cognitive decline and the potential to accelerate aging [7, 8, 9]. The variability in biological aging across individuals influences the vulnerability to sleeplessness.
Findings from the Women’s Health Initiative Study showed that greater levels of late-differentiated CD8+ T cells as well as a higher epigenetic age of blood tissue were linked to insomnia symptoms [10]. Signs of insomnia are linked to accelerated aging, and inadequate sleep has been linked to a decrease in the number of naïve T cells [10].
The telomere, a unique DNA-protein composite structure at the end of each
chromosome that contains the repeated nucleotide sequence “TTAGGG”, is essential
for maintaining the integrity and stability of the chromosomal end [11]. Telomere
length represents one of the most recognized markers of cell senescence because
telomeres shorten with age, resulting in chromosome instability and cell
senescence or apoptosis [12]. The attrition rate of telomeres has been an
attractive study topic because it more accurately reflects a person’s
physiological and functional state than their chronological age. Moreover, new
research has unequivocally shown that telomere shortening is associated with
age-related disorders, such as cardiovascular diseases [13], cancer [14], and
Alzheimer’s disease [15]. In a study of 925 individual questionnaires [16],
individuals were categorized as short (less than 6 hours), average (6 to 8
hours), and long (more than 8 hours) sleepers based on their reported the length
of sleep. This study suggested that only insomnia disorder, insomnia disorder
total sleep time
YKL-40 is a lectin that binds to chitin and is a member of the glycosyl hydrolase family 18. It is also known as chitinase 3-like protein 1 or human cartilage glycoprotein 39 [17]. The 1997 discovery of the human gene encoding YKL-40 led to its location on chromosome 1q31-q32 [18]. The protein lacks enzymatic function and it’s specific biological role is unknown. Many different types of cells, such as macrophages, neutrophils, synoviocytes, chondrocytes, fibroblast-like cells, smooth muscle cells, and tumor cells, produce and release YKL-40. It is crucial for tissue damage, inflammation, tissue regeneration, and remodeling reactions [19]. Sleep loss elevates inflammation and microglial activation [20]. In bipolar disorder, mean YLK-40 levels are much greater compared with those of controls [21]. Recent researchhas suggested that elevated brain, cerebrospinal fluid, and serum levels of YKL-40 are linked to cognitive impairment and dementias [22, 23]. YKL-40 is associated with cell age, which means that it may be involved in the process of aging [24]. At present, research on telomeres, YKL-40, insomnia, and aging is attracting much attention, and the relationships between YKL-40, sleep, and cognition are worth exploring.
We therefore aimed to further assess the correlation between YKL-40 level and telomere length, and difficulty initiating sleep (DIS), early morning awakening (EMA), difficulty maintaining sleep (DMS), and mixed symptoms (MS) in a case-control research design. We also explored the effects of sleep upon the correlation between YKL-40 and telomere length in patients, cognitive impairment in patients with different subtypes of insomnia, and YKL-40 level and telomere length as potential biomarkers of insomnia disorder.
Initially, 145 insomnia sufferers were chosen from January 2021 to July 2023
from the Neurology Unit at The First Affiliated Hospital of Anhui University of
Science and Technology (Huainan First People’s Hospital), Anhui province. Those
patients underwent polysomnography (PSG) examination and all patients met the
Diagnostic and Statistical Manual of Mental Disorders, 5th edition (DSM-5) [25],
criteria for insomnia symptoms (dissatisfaction with sleep time or quality was
the main complaint), accompanied by one or more of the following symptoms:
difficulty in starting sleep, difficulty in maintaining sleep, or early
awakening; impaired daytime functioning; sleep disturbances causing clinically
significant discomfort or impairment of social, occupational, educational,
learning, behavioral, and other important functional domains; insomnia with sleep
difficulties
A total of 145 patients meeting the enrollment criteria were grouped according to sleep complaint, including 25 in the difficulty initiating sleep group, 35 in the early morning awakening group, 35 in the difficulty maintaining sleep group, and 50 in the mixed symptoms group. Written informed consent was obtained from all patients or their families/legal guardians for the publication of any potentially identifiable images or data included in this article. This program was approved by The First Affiliated Hospital of Anhui University of Science and Technology (First People’s Hospital of Huainan) Ethics Committee (approval number 2021-KY-214-001).
The following information was collected: general information (name, age, sex, years of education, body mass index (BMI), etc.), clinical data (course duration month, current illness, past, personal and family history, laboratory test results, etc.), and depression, anxiety, cognitive function, and sleep assessment scores according to the Hamilton Depression Rating Scale-17 (HAMD-17) [26], Hamilton Anxiety Scale-14 (HAMA-14) [27, 28], Montreal Cognitive Assessment (MoCA) [29], Athens Insomnia Scale (AIS) [30], Insomnia Severity Index (ISI) [30], and Pittsburgh Sleep Quality Index (PSQI) [31, 32].
All patients in the case group (1) met the DSM-5 criteria for insomnia; (2) were aged 18–80 years; (3) had normal verbal communication abilities and the capacity to complete the scale tests; (4) had not taken any sedatives or other psychotropic drugs within 1 month prior to the visit; (5) had no history of organic diseases in the liver, kidney, or brain; and (6) provided signed informed consent indicating that their participation in the investigation was voluntary.
The exclusion criteria included (1) secondary insomnia associated alongside
organic diseases, mental disorders, and nervous system diseases; (2) physical
diseases, such as of the heart, lung, liver, kidney, and autoimmune system,
pathogenic microbial infections, tumors, or any psychiatric disorders; (3)
ingestion of high levels of caffeine (
Polysomnography (PSG) is a technique used to diagnose sleep disorders and is also an important experimental technique in sleep research. In this study, subjects underwent routine sleep monitoring overnight using the Australian Compumedics Siesta 802 series PSG system (SN180457, Compumedics Limited, Abbotsford, Australia). The acquired sleep data were analyzed using the ProFusion Sleep software (version 3, Compumedics Limited, Abbotsford, Australia). Subjects avoided lunch and alcohol consumption on the day of examination. Before the examination, their hair, face, and skin behind the ears were washed, avoiding the use of skin care products and conditioners. At 7:30 pm, the sleep monitoring room was installed with electrodes, and debugging instruments were worn to adapt to the guide connection and sleep environment in the monitoring room. No coffee, Coca-Cola, or other stimulating drinks were allowed before going to bed. Electrode placement and technical parameters were according to the American Association of Sleep Medicine Sleep and Related Events Interpretation Manual (2007 edition). The main focus of this study was the sleep electroencephalogram (EEG), providing an analysis of sleep progression, including total sleep duration (TST), sleep latency (SL), waking time after sleep (WASO), and sleep efficiency (SE). The PSG system was wired to a recording system.
The rigorous guidelines provided by the nucleic acid extraction kit (CW2087S, Shanghai Yes Service Biotech, Inc., Shanghai, China) were followed for extracting DNA, and relative telomere length was measured by quantitative real-time polymerase chain reaction using a Roche Light Cycler II polymerase chain reaction (PCR) 480 (Roche Molecular Systems, Inc, Rotkreuz, Switzerland) [33].
Primer sequences and heat cycling protocols have been previously described [34]. Procedures were carried out by technicians who were blinded to the results.
Human serum YKL-40 level was determined using enzyme-linked immunoassay (ELISA) employing the Rayto RT-6100 microplate analyzer (V513815, Shanghai Chuangyi Science and Education Equipment Co., Ltd, Shanghai, China). The procedure was carried out in accordance with the human cartilage glycoprotein 39 (HCgp-39) ELISA detection kit (YQ-737628E, Shanghai Research & Development Biotechnology Co., Ltd, Shanghai, China).
SPSS Statistics 23.0 (IBM-SPSS Statistics, Chicago, IL, USA) was used to examine
the dataset, and p
In addition, the predictive accuracy of YKL-40 level and telomere length for insomnia disorder was evaluated through the receiver operating characteristic (ROC) curve and area under the curve (AUC). The specificity (the odds of detecting a negative test in a negative population, also known as the true negative rate) was also calculated.
This case-control study found that, while there were no statistically
significant differences between the cases and controls regarding age, sex, and
BMI index, those suffering from insomnia were significantly more inclined to
experience anxiety, sadness, and cognitive impairment. The AIS, PSQI, and ISI
scores of the cases were substantially greater compared with those of the
controls (p
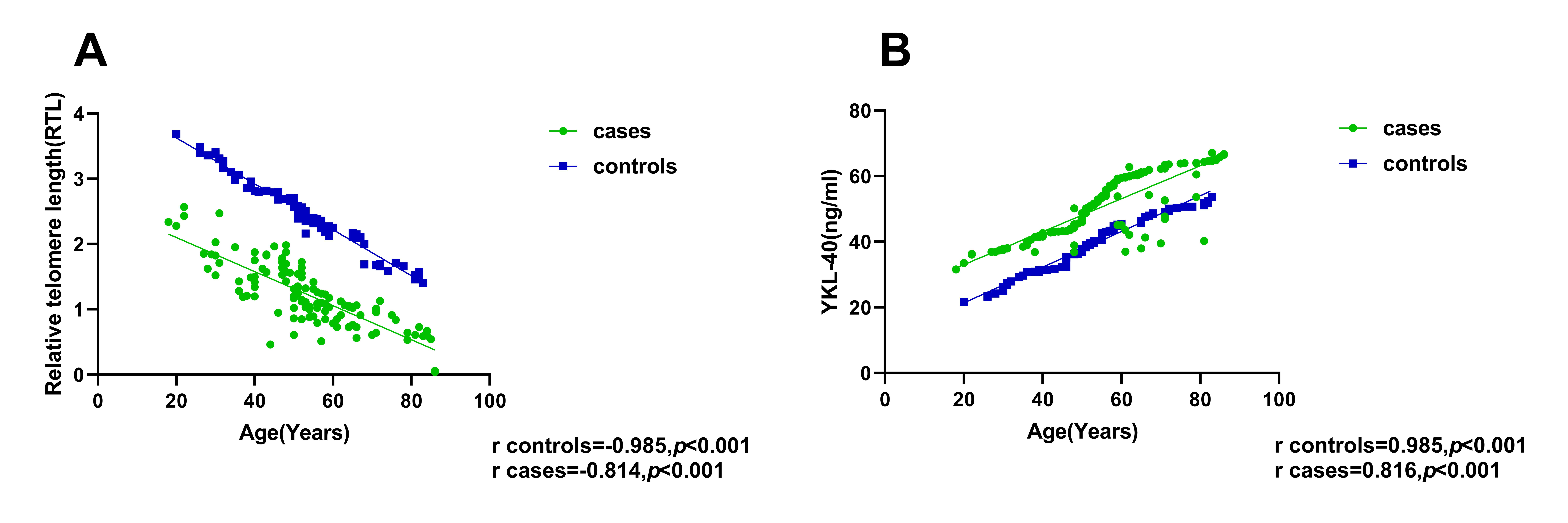 Fig. 1.
Fig. 1.
Correlations between age and relative telomere
length (RTL) and YKL-40 concentration. (A) Leucocyte telomere length and
aging showed a strongly inverse relationship in both the control group
(association coefficient r = –0.985, p
| Cases (n = 145) | Controls (n = 80) | Statistics | p | |
| Age, years | 46.01 |
44.39 |
t = 1.04 | 0.30 |
| Male, n (%) | 30 (21.13%) | 17 (21.25%) | 0.92 | |
| BMI, kg/m2 | 24.02 |
23.32 |
t = 1.54 | 0.12 |
| AIS | 11 (9–14) | 0 (0–0) | Z = –5.823 | |
| PSQI | 13 (12–17) | 0 (0–1) | Z = –5.749 | |
| HAMA-14 | 8 (4–10) | 1 (0–2) | Z = –5.444 | |
| HAMD-17 | 10 (6–13) | 2 (1–2) | Z = –5.559 | |
| MoCA | 17 (15–20) | 22 (20–27) | Z = –5.325 | |
| ISI | 15 (12–19) | 0 (0–1) | Z = –5.824 |
Abbreviations: BMI, Body Mass Index; AIS, Athens Insomnia Scale; PSQI, Pittsburgh Sleep Quality Index; HAMA-14, Hamilton Anxiety Scale-14; HAMD-17, Hamilton Depression Rating Scale-17; ISI, Insomnia Severity Index; MoCA, Montreal Cognitive Assessment.
There were no notable variations within sex, age, disease duration, years of education, and HAMA-14, HAMD-17, MoCA, AIS, and PSQI scores between the four groups (Table 2). Among the four subtypes, SE showed significant group differences, compared with the MS group, with the EMA and DMS groups showing higher SE (Table 3).
| Characteristics | DIS (n = 25) | EMA (n = 35) | DMS (n = 35) | MS (n = 50) | p |
| Age, years | 46.67 |
45.87 |
47.01 |
45.34 |
0.7 |
| Male, n (%) | 6 (24) | 7 (20) | 7 (20) | 10 (20) | 0.98 |
| BMI, kg/m2 | 23.92 |
24.15 |
24.38 |
23.77 |
0.7 |
| Course duration, years | 8 (6.25, 10.75) | 9 (8, 10.5) | 9 (6, 12) | 10 (8, 15) | 0.11 |
| Years of education | 15 (11, 17.5) | 14.5 (10, 19) | 13 (9, 15) | 15.5 (9, 20) | 0.5 |
| HAMA-14 | 10 (6, 15) | 9 (6, 14) | 11 (7, 17) | 10 (5.5, 16) | 0.3 |
| HAMD-17 | 12 (5, 17) | 10.5 (5, 15) | 11 (6, 17) | 10.5 (7, 14) | 0.4 |
| MoCA | 19 (14, 22) | 20 (15, 22) | 19 (14, 22) | 20.5 (14, 23) | 0.6 |
| AIS | 12.5 (7, 16) | 11.5 (7.5, 16) | 13 (8, 17) | 12 (7, 18) | 0.3 |
| PSQI | 15 (11.5, 20) | 15.5 (10, 19) | 13 (9, 19) | 16 (12, 22) | 0.2 |
Abbreviations: BMI, body mass index, DIS, difficulty initiating sleep; EMA, early morning awakening; DMS, difficulty maintaining sleep; MS, mixed symptoms.
| Item | DIS | EMA | DMS | MS | p |
| TST (min) | 388.1 |
369.9 |
375.7 |
358.8 |
0.55 |
| SL (min) | 35.3 (23, 43) | 18.4 (12.2, 23) | 16.9 (10.9, 19.7) | 17.8 (11.3, 20.9) | 0.34 |
| WASO (min) | 125.3 (44.8, 181.2) | 77.3 (40.2, 173.5) | 92.2 (48.9, 156.8) | 119.9 (53.6, 198.8) | 0.13 |
| SE (%) | 71.5 (58.5, 83.1) | 82.8 (61, 89.8)# | 83.2 (60.5, 90.2)# | 68.7 (52.3, 78.2) | 0.04 |
Abbreviations: TST, total sleep time; SL, sleep latency; WASO, wake after sleep
onset; SE, sleep efficiency; EEG, electroencephalogram. #In contrast to the
mixed symptoms category, p
Multivariate testing adjusted for age, sex, BMI, course duration, years of
education, and HAMA-14, HAMD-17, MoCA, AIS, and PSQI scores, showed that people
with telomere lengths in the lowest tertile were much more likely to have a
subtype of difficulty initiating sleep (OR 2.13, 95% CI 1.22–3.63; p =
0.03) and have mixed symptoms (OR 1.49, 95% CI 1.30–2.81; p = 0.04)
compared with those in the top tertile (Table 4). Shorter telomeres did not
significantly correlate with the likelihood of waking up early or having trouble
staying asleep. After secondary analysis, when telomere length was considered as
an ongoing variable, a 1 SD reduction in leukocyte telomere length was linked to
an approximately 48% greater susceptibility to difficulty initiating sleep and a
42% increased susceptibility to having mixed symptoms (p
| Tertiles of telomere length | p trend | Per 1 SD decrease | p | ||||
| Highest tertile | Middle tertile | Lowest tertile | in Ln-transformed telomere length | ||||
| ( |
(1.12–1.98) | ( | |||||
| Controls (n = 80) | 46 | 23 | 11 | ||||
| Early morning awakening (EMA) (n = 35) | 7 | 10 | 18 | ||||
| ORs (95% CI) | |||||||
| No adjustment | 1.0 | 0.82 (0.63–1.14) | 1.18 (0.91–1.64) | 0.14 | 1.13 (1.07–1.31) | 0.046 | |
| Model I | 1.0 | 0.81(0.56–1.73) | 1.07 (0.84–1.68) | 0.25 | 1.09 (0.91–1.28) | 0.31 | |
| Model II | 1.0 | 0.85 (0.60–1.65) | 1.08 (0.82–1.76) | 0.41 | 1.08 (0.95–1.32) | 0.35 | |
| Difficulty maintaining sleep (DMS) (n = 35) | 8 | 11 | 16 | ||||
| ORs (95% CI) | |||||||
| No adjustment | 1.0 | 0.74 (0.39–1.17) | 1.25 (0.69–1.91) | 0.07 | 1.17 (0.95–1.52) | 0.17 | |
| Model I | 1.0 | 0.81 (0.37–1.95) | 0.92 (0.42–1.87) | 0.88 | 1.10 (0.82–1.47) | 0.58 | |
| Model II | 1.0 | 0.75 (0.37–1.67) | 0.87 (0.42–1.90) | 0.79 | 1.07 (0.77–1.66) | 0.70 | |
| Difficulty initiating sleep (DIS) (n = 25) | 5 | 7 | 13 | ||||
| ORs (95% CI) | |||||||
| No adjustment | 1.0 | 1.02 (0.67–1.09) | 1.19 (0.98–2.07) | 0.07 | 1.18 (0.98–1.60) | 0.08 | |
| Model I | 1.0 | 1.17 (0.99–2.35) | 1.39 (1.10–2.80) | 0.05 | 1.29 (1.06–1.75) | 0.04 | |
| Model II | 1.0 | 1.53 (1.14–2.41) | 2.13 (1.22–3.63) | 0.03 | 1.48 (1.24–1.96) | 0.04 | |
| Mixed symptoms (MS) (n = 50) | 9 | 14 | 27 | ||||
| ORs (95% CI) | |||||||
| No adjustment | 1.0 | 1.02 (0.73–1.91) | 1.05 (0.82–1.98) | 0.24 | 1.04 (0.75–2.00) | 0.053 | |
| Model I | 1.0 | 1.18 (0.81–1.79) | 1.31 (0.92–1.83) | 0.23 | 1.24 (1.01–1.98) | 0.04 | |
| Model II | 1.0 | 1.22 (1.08–1.85) | 1.49 (1.30–2.81) | 0.04 | 1.42 (1.29–2.38) | 0.03 | |
The threshold tertile values of leucocyte telomere length (relative telomeric
repeat sequences/single-copy genes (T/S) ratio) were calculated via the control
category, alongside
Model I: adjustment for age, sex, body mass index, course duration, years of education, and HAMA-14, HAMD-17, MoCA, AIS, and PSQI scores.
Model II: adjustment for the previously listed factors as well as total sleep duration, sleep latency, wakefulness following the start of sleep, and effectiveness of sleep.
Abbreviations: OR, odds ratio; CI, confidence interval; SD, standard deviation.
In order to investigate the altered impacts of age, sex, anxiety, depression, and cognitive impairment, we carried out in-depth analyses by assessing the factors’ interactions (Table 5). When it came to trouble falling asleep, people were more likely to be female (OR 1.44, 95% CI 1.12–1.87) than male (group not included) for every 1 SD drop in leukocyte telomere length. Association between difficulty initiating sleep and mixed symptoms were positive when anxiety, depression, and cognitive impairment were present. The interaction between cognitive impairment and difficulty initiating sleep (pinteraction = 0.03) and mixed symptoms (pinteraction = 0.04) were statistically noteworthy (Table 5).
| Variables | Difficulty initiating sleep | Mixed symptoms | ||||||
| Case/control | OR (95% CI) | p | pinteraction | Case/control | OR (95% CI) | p | pinteraction | |
| Sex | ||||||||
| Male | 6/17 | 1.04 (0.92–1.59) | 0.21 | 0.07 | 10/17 | 1.14 (0.95–1.81) | 0.32 | 0.35 |
| Female | 19/63 | 1.44 (1.12–1.87) | 0.007 | 40/63 | 1.73 (1.18–2.64) | 0.04 | ||
| Age, years | ||||||||
| 15/51 | 1.13 (0.95–2.19) | 0.32 | 0.09 | 32/51 | 1.53 (1.12–2.24) | 0.008 | 0.07 | |
| 10/29 | 1.05 (0.90–1.73) | 0.45 | 18/29 | 1.61 (1.22–2.85) | 0.005 | |||
| Anxiety | ||||||||
| No | 3/79 | 1.46 (1.16–1.88) | 0.005 | 0.24 | 6/79 | 1.23 (1.01–1.79) | 0.04 | 0.38 |
| Yes | 22/1 | 1.67 (1.26–2.22) | 0.04 | 44/1 | 1.53 (1.12–1.94) | 0.007 | ||
| Depression | ||||||||
| No | 4/79 | 1.53 (0.99–2.44) | 0.12 | 0.35 | 7/79 | 1.18 (1.04–1.84) | 0.004 | 0.41 |
| Yes | 21/1 | 1.51 (1.15–1.89) | 0.04 | 43/1 | 1.23 (1.08–1.95) | 0.006 | ||
| Cognitive impairment | ||||||||
| No | 2/78 | 1.84 (1.41–2.55) | 0.04 | 0.03 | 5/78 | 1.55 (1.18–2.25) | 0.01 | 0.04 |
| Yes | 23/2 | 1.36 (1.12–1.92) | 0.01 | 45/2 | 1.76 (1.21–2.64) | 0.02 | ||
ORs (95% CI) and p values were calculated alongside multivariate logistic regression analyses following adjustment for conventional vascular risk variables, which includes age; sex; body mass index; course duration; years of education; HAMA-14, HAMD-17, MoCA, AIS, and PSQI scores; total sleep duration; sleep latency; wake following sleep onset; and sleep effectiveness, except for the stratification element. pinteraction was calculated by the assessment of interactions.
Multivariate testing adjusting for age, sex, BMI, course duration, years of education, and HAMA-14, HAMD-17, MoCA, AIS, and PSQI scores, individuals within the highest tertile of YKL-40 level had a significantly elevated risk for the early morning waking subtype [OR 2.98, 95% CI 1.54–5.33; p = 0.01] and mixed symptoms (OR 1.47, 95% CI 1.22–2.79; p = 0.02) compared with individuals in the lowest tertile. There was no discernible correlation found between YKL-40 level, risk of difficulty initiating sleep, and difficulty maintaining sleep (Table 6).
| Tertiles of YKL-40 concentration | p | ||||
| Lowest tertiles | Middle tertiles | Highest tertiles | |||
| ( |
(41.7–50.83) | ( | |||
| Controls (n = 80) | 47 | 28 | 5 | ||
| Difficulty initiating sleep (DIS) (n = 25) | 5 | 9 | 11 | ||
| ORs (95% CI) | |||||
| No adjustment | 1.0 | 0.88 (0.63–1.24) | 1.18 (0.91–1.64) | 0.14 | |
| Model I | 1.0 | 0.91 (0.66–1.73) | 1.27 (0.84–2.68) | 0.15 | |
| Model II | 1.0 | 0.95 (0.60–1.75) | 1.18 (0.82–1.96) | 0.07 | |
| Difficulty maintaining sleep (DMS) (n = 35) | 7 | 10 | 18 | ||
| ORs (95% CI) | |||||
| No adjustment | 1.0 | 0.84 (0.39–1.27) | 1.25 (0.79–1.98) | 0.07 | |
| Model I | 1.0 | 0.91 (0.47–1.95) | 0.92 (0.62–1.97) | 0.88 | |
| Model II | 1.0 | 0.95 (0.57–1.87) | 1.07 (0.72–2.90) | 0.19 | |
| Early morning awakening (EMA) (n = 35) | 6 | 9 | 20 | ||
| ORs (95% CI) | |||||
| No adjustment | 1.0 | 1.12 (0.97–1.49) | 1.19 (0.98–2.07) | 0.06 | |
| Model I | 1.0 | 1.17 (0.96–2.15) | 1.47 (1.09–2.82) | 0.04 | |
| Model II | 1.0 | 1.93 (1.14–3.41) | 2.98 (1.54–5.33) | 0.01 | |
| Mixed symptoms (MS) (n = 50) | 10 | 14 | 26 | ||
| ORs (95% CI) | |||||
| No adjustment | 1.0 | 1.08 (0.73–1.96) | 1.11 (0.85–1.98) | 0.14 | |
| Model I | 1.0 | 1.18 (0.91–2.79) | 1.51 (1.12–2.83) | 0.03 | |
| Model II | 1.0 | 1.21 (1.11–1.95) | 1.47 (1.22–2.79) | 0.02 | |
The threshold values of tertile of YKL-40 concentration were calculated via the
control category, alongside
Model I: adjustment for age, sex, body mass index, course duration, years of education, and HAMA-14, HAMD-17, MoCA, AIS, and PSQI scores.
Model II: modification for the previously listed factors as well as total sleep duration, sleep latency, wakefulness following the start of sleep, and effectiveness of sleep.
The AUC of YKL-40 concentration and telomere length were 0.806 and 0.746 (p
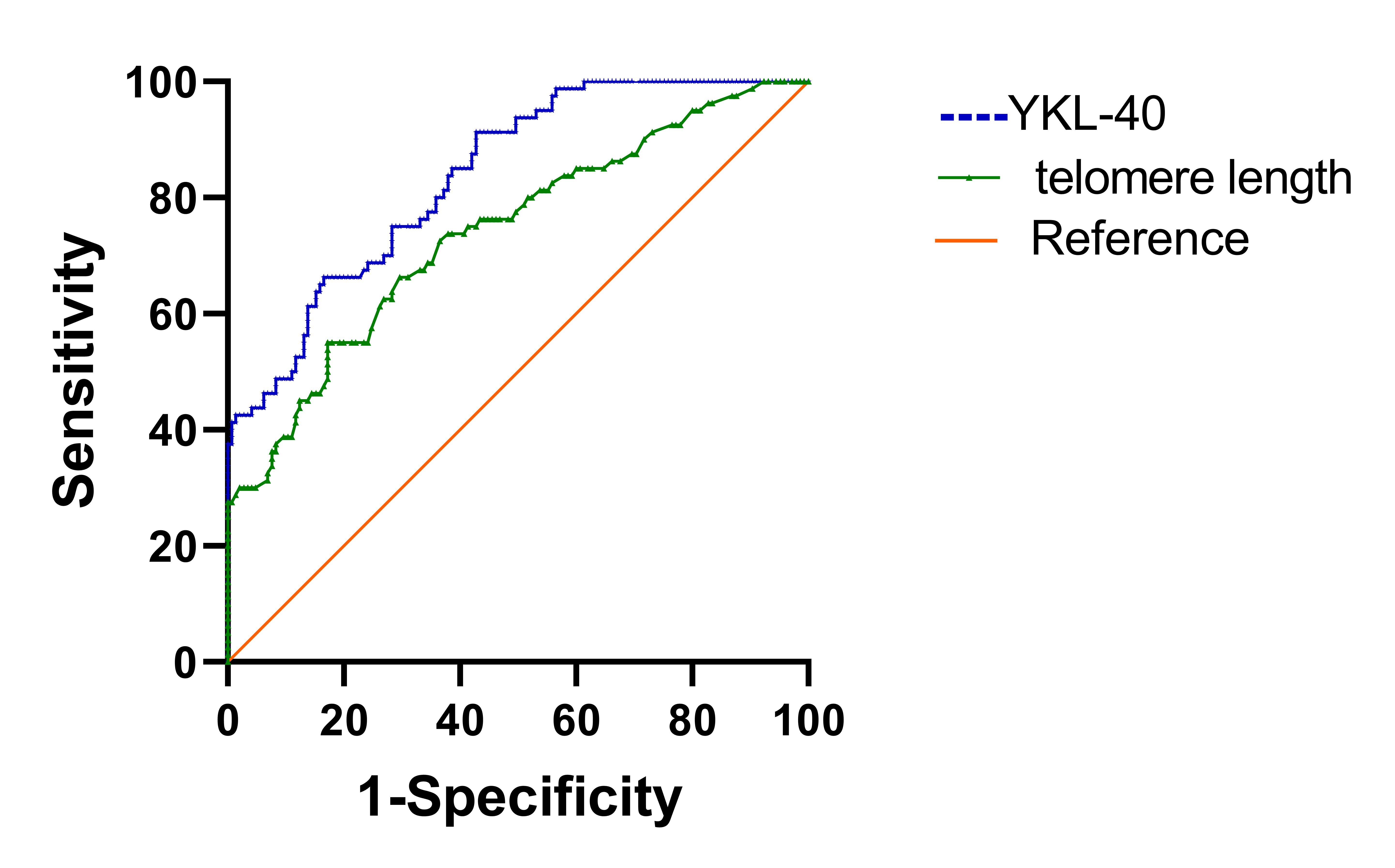 Fig. 2.
Fig. 2.
Receiver operating characteristic curve and area under the curve
(AUC) for predicting insomnia disorder. The
AUC of YKL-40 concentration and telomere length were 0.806 and 0.746 (p
Our findings in this case-control study demonstrated a significant correlation, irrespective of other risk factors including anxiety and depression, between shorter leukocyte telomere length (LTL) and the risk for trouble falling asleep and mixed symptoms. Moreover, cognitive impairment appeared to be associated with difficulty initiating sleep and mixed symptoms. There was no discernible variation within telomere length across early morning awakening/difficulty maintaining and controls. Together, these results suggest that telomere shortening plays diverse and important functions in age-related illnesses.
It has long been believed that telomere length serves as a reliable predictor of
age. Shorter telomere length has been associated with insomnia in those 70–88
years old, but not in younger adults. This finding raises the possibility that
clinically severe sleep disruptions accelerate cellular aging, particularly in
later life [35]. A study of 925 individuals with self-reported sleep duration
suggested that total sleep time
Because telomeres contain a lot of guanine, they are vulnerable to oxidative damage. This may be demonstrated by the production of 8-hydroxy-2-deoxyguanosine (8-oxodG), a crucial indicator of oxidative stress that accelerates telomere shortening [43]. Oxidative stress could lead to DNA damage, as shown by the presence of 8-oxodG and a decrease of telomeric-repeat binding factor 1 (TRF1) and telomeric-repeat binding factor 2 (TRF2), which play a role in proper telomere replication as well as T-loop formation, resulting in excessively shortened telomere lengths [44]. For a possible link between insomnia and telomere length, one study showed that insomnia might lead to increased levels of oxidation, which might shorten telomere length [45]. In addition, the hypothalamic-pituitary-adrenal axis is disrupted by chronic sleep deprivation and abnormal cortisol secretion, which decreases telomerase activity, shortening the telomere [46, 47].
YKL-40 is a novel indicator of inflammation and produced by macrophages, neutrophils, synoviocytes, chondrocytes, fibroblast-like cells, smooth muscle cells, and tumor cells [19]. It was demonstrated that both cultured macrophages and astrocytes under neuroinflammatory conditions expressed this protein abundantly. Previous studies of neurodegenerative dementias diseases, such as Creutzfeldt-Jakob disease, Alzheimer’s disease, dementia with Lewy bodies, vascular dementia, and Parkinson’s disease, showed that YKL-40 constitutes a disease-specific indicator of neuroinflammation [20, 23, 48, 49, 50]. However, there has been very little research on sleep and YKL-40. So far, there have been no studies on the different clinical symptoms of insomnia and YKL-40. Our findings in the present study indicated a favorable correlation across higher YKL-40 levels and early morning awakening and mixed symptoms. Considering that sleep deficiency may cause the activation of microglia and astrocytes, and the stimulation and release of many pro-inflammatory factors such as interleukin and tumor necrosis factor [51, 52]. A possible explanation is that insomnia triggers neuroinflammation accompanied by elevated YKL-40 levels.
In the present study, the well-known association between age and telomere length
(r = –0.814, p-values
Studies have shown that low SE, sleep disruption, and insufficient sleep may cause poor cognitive function [54, 55, 56, 57], and a meta-analysis showed that longer LTL is connected to higher brain volume and higher global cognition by [58].
Consistently, in the present study, we found that cognitive impairment was statistically significantly related to difficulty initiating sleep and mixed symptoms.
Treatment for insomnia may slow down the aging process as it can lead to cognitive impairment and dementia, both of which can accelerate biological aging [7, 8, 9, 59]. Moreover, telomere length and YKL-40 concentration have been shown to be biomarkers of aging [12, 24]. We therefore studied the relationship between cognitive impairment, insomnia disorder, and biomarkers of aging, i.e., telomere length and YKL-40 level. Larger samples are needed in the future to investigate the underlying mechanisms. Since telomere length is continuously shortened as cells divide, shortened telomere length may reflected active cell proliferation, which may be induced by oxidative stress and chronic inflammation. There is evidence that amyloid plaques in the brain are accompanied by neuroinflammatory states characterized by the activation of astrocytes and microglia and release of pro-inflammatory mediators. Patients with cognitive dysfunction show activation of peripheral immune states associated with this inflammation of the brain; thus, shortened telomere length may also be an indicator of cognitive dysfunction [60]. Insomnia may disrupt the inflammatory cascade and increase oxidative stress, thereby increasing the levels of inflammatory cytokines and oxides [44], which could cause DNA damage, ultimately leading to telomere shortening [61].
One strength of the current study was that chronic insomnia was subdivided into four subtypes. The harmonization of sleep measures and covariates was an additional strength. The limitations are that, firstly, this case-control study was unable to identify causation between chronic insomnia, telomere attrition, YKL-40 level, aging, and cognitive impairment; therefore, a large-scale prospective cohort study is needed. Secondly, this study measured sleep through self-report. Therefore, the scoring in each scale might have been subject to the individual’s emotions and there might have been some recall bias. Finally, the sample size was relatively small; therefore, larger sample sizes are needed in future studies.
In this case-control study, telomere length decreased with age and YKL-40 level increased with age. Telomere length within patients suffering difficulty initiating sleep and mixed symptoms was significantly shortened. The level of YKL-40 within sufferers with early morning awakening and mixed symptoms was significantly increased. Cognitive impairment was associated with difficulty initiating sleep and mixed symptoms.
All data generated or analyzed during this study are included in this published article.
MZ contributed to the study conception and design, drafting of the manuscript. JL contributed to analysis of data and critical revision of the manuscript. PPL, YW and CYR contributed to experimental design, data statistics and manuscript revision. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
This program has been approved by The First Affiliated Hospital of Anhui University of Science and Technology (First People’s Hospital of Huainan) Ethics Committee (approval number: 2021-KY-214-001). Written informed consent was obtained from all patients or their families/legal guardians for the publication of any potentially identifiable images or data included in this article. We confirm that we have read the journal’s position on ethical publishing issues and confirm that this report is consistent with these guidelines. None of the patient’s information was leaked and the patient gave full informed consent.
We are grateful to the technicians and staff of the Genetics Laboratory in Huainan City for the data collected and management.
Anhui University of Science and Technology university-level project (QN2019124) and Anhui University of Science and Technology medical special cultivation project (YZ2023H1A002).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


