1 College of Rehabilitation Sciences, Shanghai University of Medicine & Health Sciences, 201318 Shanghai, China
2 Rehabilitation Department, Shanghai Yangzhi Rehabilitation Hospital (Shanghai Sunshine Rehabilitation Center), 201620 Shanghai, China
3 Neuromodulation Therapy Department, Shanghai Health Rehabilitation Hospital, 201615 Shanghai, China
4 Medical Research and Education Department, Shanghai Health Rehabilitation Hospital, 201615 Shanghai, China
Abstract
Background: The clinical application of 10 Hz repetitive transcranil magnetic stimulation (rTMS) remains limited despite its demonstrated effectiveness in enhancing cortical excitability and improving cognitive function. The present study used a novel stimulus target [left dorsolateral prefrontal cortex + primary motor cortex] to facilitate the enhancement of cognitive function through the bidirectional promotion of cognitive and motor functions; Methods: Post-stroke cognitive impairment patients (n = 48) were randomly assigned to receive either dual-target, single-target, or sham rTMS for 4 weeks. Before and after 4 weeks of treatment, participants were asked to complete the Montreal Cognitive Assessment (MoCA) test, the Modified Barthel Index (MBI), the Trail-making Test (TMT), and the Digital Span Test (DST). In addition, the levels of brain-derived neurotrophic factor (BDNF) and vascular endothelial growth factor (VEGF) in serum were also measured. Results: After adjusting for pre-intervention (baseline) MoCA scores, the post-intervention MoCA scores varied significantly. After post-hoc analysis, differences existed between the post-treatment scores of the dual-target rTMS group and the sham rTMS group (the experimental group scores were significantly higher), and between those of the dual-target rTMS group and the single-target rTMS group (the dual-target rTMS scores were significantly higher). The serum VEGF levels of the dual-target rTMS group were significantly higher those that of the sham rTMS group. Conclusions: The present study presented data showing that a dual-target rTMS therapy is effective for Post-stroke cognitive impairment (PSCI). The stimulation exhibited remarkable efficacy, suggesting that dual-target stimulation (left dorsolateral prefrontal cortex+motor cortex (L-DLPFC+M1)) holds promise as a potential target for TMS therapy in individuals with cognitive impairment after stroke. Clinical Trial Registration: No: ChiCTR220066184. Registered 26 November, 2022, https://www.chictr.org.cn.
Keywords
- dorsolateral prefrontal cortex
- dual-target
- primary motor cortex
- post-stroke cognitive impairment
- repetitive transcranial magnetic stimulation
Stroke often contributes to lifelong disability and mortality [1]. The estimated incidence of stroke among Chinese individuals stands at 2022/10,000, with an annual morbidity rate of approximately 153.9/10,000 [2, 3]. Post-stroke cognitive impairment (PSCI) occurs within 6 mo after stroke, and is a common sequela after stroke [3]. According to previous research, the occurrence rate of PSCI among stroke patients in China has reached 80.97% [4].
Repetitive transcranial magnetic stimulation (rTMS) has been found to effectively regulate cortical excitability, thereby stimulating plasticity, and serving as a beneficial treatment option for PSCI [5, 6]. Several studies have reported beneficial effects of rTMS on PSCI. Wang et al. [7] concluded that high-frequency rTMS, combined with cognitive training, improves executive function in stroke patients. Kim et al. [8] also observed that high-frequency rTMS applied to the affected dorsolateral prefrontal cortex (DLPFC) produced improved cognitive functioning in stroke patients; they observed an increase in Digital Span Test (DST) score, and sequential and retrospective memory. The stimulation protocol is the primary emphasis of rTMS [9]. Most previous clinical studies of rTMS on improvement of PSCI have targeted the left DLPFC (L-DLPFC) due to its crucial involvement in regulating working memory and cognition [6, 10, 11]. Zhang et al. [12] performed rTMS stimulation on the L-DLPFC of PSCI patients (n = 20 in the rTMS group; n = 20 in the sham-stimulation group), using 5 Hz and an intensity of 80% Motion threshold (MT), and found that patients in the rTMS group had significantly higher Trail-making Test (TMT) scores after treatment than did the sham-stimulation group. However, the evidence supporting the efficacy of rTMS has been questioned; inconsistent results have been reported. Treatment failures may arise within studies due to variations in stimulus parameters [13, 14]. The potential association between the site of stimulation and this limitation should be further explored.
Patients with PSCI often have frontal-subcortical functional impairment [12], but the cognitive impairment caused by stroke is not limited to related brain regions within the cognitive network, it also involves sensorimotor-related brain regions. Studies have shown that brain activity in the frontoparietal network (FPN) is decreased in PSCI patients, suggesting that some cognitive function is impaired in stroke patients. Moreover, although there was no difference between Fugl-Meyer Assessment scale (FMA) and the Functional Comprehensive Assessment scale (FCA) scores, the functional Near-Infrared Spectroscopy (fNIRS) evaluation results showed that the activity of the sensorimotor network (SEN) area was significantly reduced, suggesting that the decreased activity of the SEN area may not be directly reflected in the patient’s motor function. The effect of reduced activity in SEN areas may be greater on cognitive function in stroke patients [12]. Therefore, it is imperative to integrate various stimulation sites when using rTMS in order to maximise the clinical benefits achieved through neuromodulation.
Clinically, the frequency of rTMS stimulation is defined by high-frequency
stimulation (
We also examined the potential mechanism by which rTMS enhances the recovery of cognitive function after stroke, and the mechanism by which the double-target rTMS may be superior to the other groups. Vascular endothelial growth factor (VEGF) plays an important role in the vascular response in ischemic stroke, and can be used as one of the biomarkers for assessing the severity of the patient’s disease and the prognosis [20]. When brain tissue undergoes ischemic injury, the ischemic environment can stimulate the expression of VEGF [21], and the levels of VEGF is positively correlated with the recovery of neurological function in patients [22]. Brain-derived neurotrophic factor (BDNF) is a key factor involved in neuroplasticity and is the most widely distributed neurotrophic factor in the brain. After brain injury or stroke, BDNF can produce a variety of neuroprotective effects [23], including stabilisation of intracellular calcium homeostasis [24], maintenance of normal neuronal development and survival, and promotion of neuronal regeneration. It also is involved in the repair of damaged neurons, and it stimulates axon germination and neo-synaptic formation [25], which plays an important role in the prognosis, pathogenesis, and rehabilitation of stroke patients. Wang et al. [26] found that rTMS improved learning and memory abilities in demented rats; the mechanism may be related to the fact that rTMS enhances the expression of BDNF, synaptophysin mRNAs and proteins, and improves cognition by improving synaptic plasticity in the brain. Zong et al. [27] found that rTMS had a strong protective and repairing effect on the microvasculature around the infarct foci in rats; the expression of VEGF around the infarct foci was up-regulated after rTMS treatment. One study showed that after cognitive rehabilitation training combined with high-frequency rTMS treatment, patients had significantly higher serum levels of BDNF and VEGF, higher scores on the Montreal Cognitive Assessment (MoCA) than at pre-treatment, and improved auditory event-related potentials [28], so in the present study, we assessed the levels of both VEGF and BDNF in peripheral blood.
From December 2022 to October 2023, 57 patients were qualified at Shanghai Health Rehabilitation Hospital (Shanghai, China). 48 patients (including 18 women, representing 31.6% of the total sample) with a mean age of 67 years (Standard Deviation (SD) = 10.6) were ultimately recruited for participation in the study (Table 1). The inclusion criteria were: (1) age of patient, 60–75 years; (2) right-handed; (3) fulfilled the diagnostic criteria for stroke and cognitive impairment [29, 30, 31]; (4) a clear state of consciousness and stable vital signs; (5) at least one limb should function without any impediments; (6) with a cognitive impairment appearing after stroke, with a scoring range on the MoCA test of 9–26 points. The exclusion criteria were: (1) cognitive impairment stemming from either primary or secondary neurological disorder; (2) a history of epileptic seizures; (3) severe general impairment or concurrent medical conditions; (4) severe aphasia or cognitive impairment that might hinder the provision of informed consent or impede the behavioral measurements conducted during the study; (5) any contraindications to Transcranial Magnetic Stimulation (TMS) [32].
| Variables | Sham-rTMS | Single-target rTMS | Dual-target rTMS | p | |
| n = 18 | n = 15 | n = 15 | |||
| Sex, n (%) | 0.126 | ||||
| Male | 13 (72.2) | 12 (80.0) | 7 (46.7) | ||
| Female | 5 (27.8) | 3 (20.0) | 8 (53.3) | ||
| Age, years | 0.845 | ||||
| Mean (SD) | 69.06 (8.94) | 67.53 (10.63) | 67.20 (10.09) | ||
| Educational level, n (%) | 0.865 | ||||
| Primary school or less, n (%) | 2 (11.1) | 1 (6.7) | 1 (6.7) | ||
| Junior middle school, n (%) | 12 (66.7) | 9 (60.0) | 9 (60.0) | ||
| High school and above, n (%) | 4 (22.2) | 5 (33.3) | 5 (33.3) | ||
| Time since stroke onset (days) | 0.832 | ||||
| Mean (SD) | 75.42 (23.68) | 69.47 (27.84) | 74.63 (24.21) | ||
| Stroke type, n (%) | 0.097 | ||||
| Ischemic stroke, n (%) | 14 (77.8) | 14 (93.3) | 15 (100) | ||
| Hemorrhagic stroke, n (%) | 4 (22.2) | 1 (6.7) | 0 | ||
| Side of stroke, n (%) | 0.069 | ||||
| Left | 6 (33.3) | 7 (46.7) | 11 (73.3) | ||
| Right | 12 (66.7) | 8 (53.3) | 4 (26.7) | ||
| Comorbidities | |||||
| Hypertension, n (%) | 15 (83.3) | 12 (80.0) | 14 (93.3) | 0.557 | |
| Diabetes, n (%) | 7 (38.9) | 9 (60.0) | 10 (66.7) | 0.241 | |
| Coronary heart disease, n (%) | 2 (11.1) | 0 | 1 (6.7) | 0.892 | |
| Hyperlipidemia, n (%) | 1 (5.3) | 4 (21.1) | 1 (5.3) | 0.432 | |
| MoCA baseline | |||||
| Mean (SD) | 14.11 (4.03) | 18.40 (3.42) | 18.80 (2.57) | ||
| MBI baseline | 0.266 | ||||
| Mean (SD) | 37.11 (15.86) | 42.89 (14.29) | 45.11 (16.50) | ||
| TMT-A baseline | |||||
| Time (seconds) | 0.054 | ||||
| Mean (SD) | 137.06 (26.16) | 114.83 (31.28) | 121.95 (26.86) | ||
| TMT-B baseline | |||||
| Time (seconds) | 0.113 | ||||
| Mean (SD) | 207.13 (74.00) | 181.65 (65.65) | 228.84 (64.81) | ||
| DST baseline | |||||
| Number of Sequence | 0.245 | ||||
| Mean (SD) | 4.39 (2.22) | 5.17 (1.21) | 4.37 (1.34) | ||
| Number of Inverted sequence | 0.023 | ||||
| Mean (SD) | 1.56 (1.30) | 2.33 (1.00) | 2.53 (1.02) | ||
| BDNF baseline | |||||
| Mean (SD) | 10,040.98 (1362.26) | 13,858.89 (1521.27) | 14,178.61 (1487.54) | 0.404 | |
| VEGF baseline | |||||
| Mean (SD) | 86.81 (16.42) | 87.01 (15.73) | 95.00 (16.05) | 0.821 | |
rTMS, repetitive transcranil magnetic stimulation; SD, standard deviation; MoCA, Montreal Cognitive Assessment; MBI, Modified Barthel Index; TMT, Trail-making Test; DST, Digital Span Test; BDNF, brain-derived neurotrophic factor; VEGF, vascular endothelial growth factor.
Ethical approval was obtained from the Medical Ethics Committee of Shanghai University of Medicine & Health Sciences on October 27, 2022 (2022-E4-6100-21-201034-03-210302197009090947). The Ethics Committee of Chinese Registered Clinical Trials has reviewed this study and has granted its approval. The registration number is ChiCTR220066184. Each participant provided written informed consent.
The flow chart of the procedures is illustrated in Fig. 1.
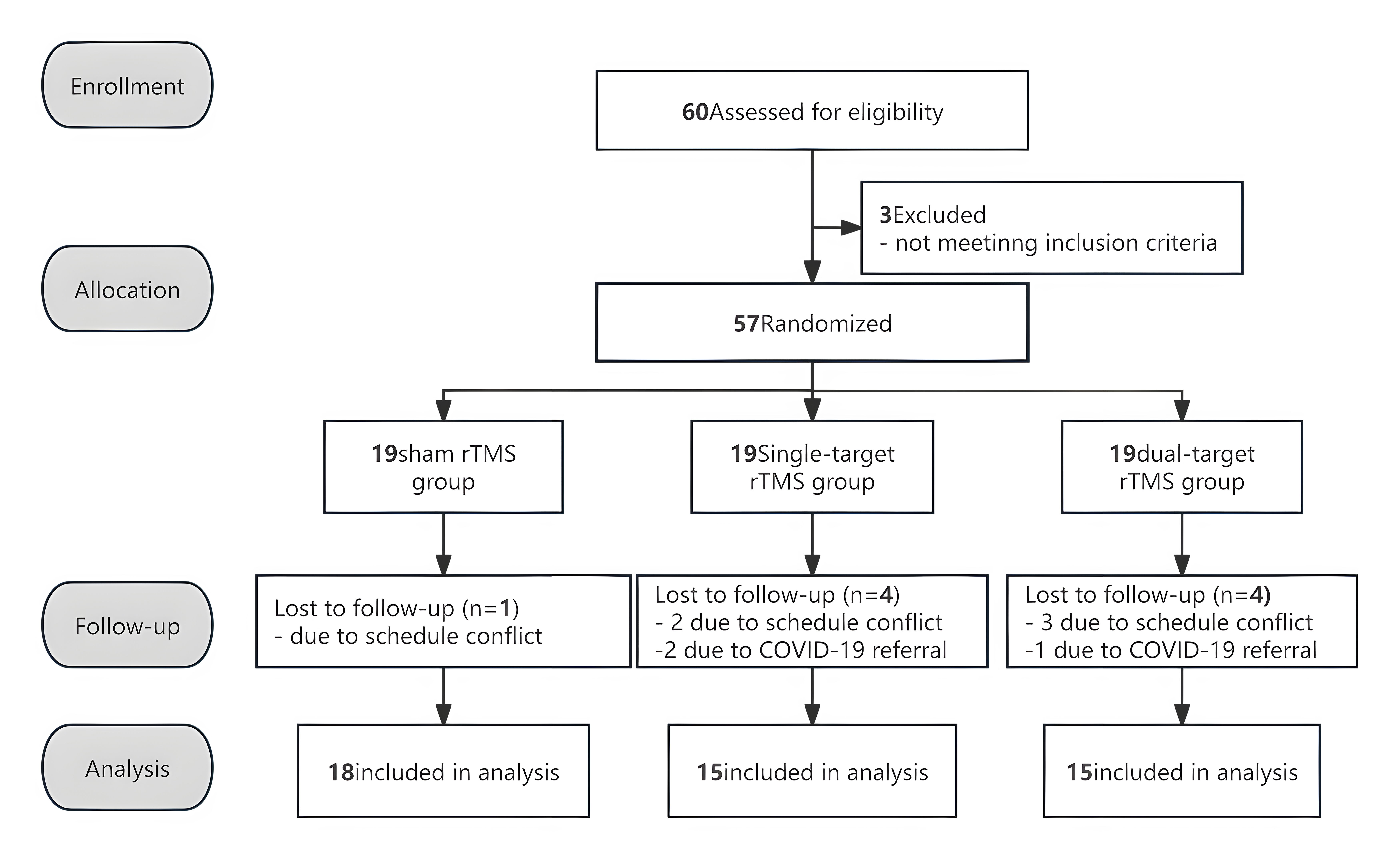 Fig. 1.
Fig. 1.
The CONSORT flowchart visually outlines the recruitment, group allocation, treatment allocation, follow-up procedures, and analytical methods in clinical trials. rTMS, repetitive transcranil magnetic stimulation.
Fig. 2 shows the experimental design. After recruiting PSCI patients, they were randomly assigned to one of three groups for 20 treatments (4 weeks, 5 consecutive days, and 2 days off per week). All patients received routine rehabilitation treatment (40 min/day, 5 days/week, for 4 weeks), with one-on-one physical and occupational therapy provided by qualified rehabilitation therapists. The experimental rTMS groups formed were (a) the dual-target group for real L-DLPFC + real M1 stimulation; (b) single-target group for real L-DLPFC + sham M1 stimulation; and (c) sham-stimulation group for sham L-DLPFC + sham M1 stimulation. Outcome measures were obtained twice: initially for a baseline prior to the intervention, and after 4 weeks of rehabilitation and rTMS intervention.
 Fig. 2.
Fig. 2.
Experimental design. BDNF, brain-derived neurotrophic factor; VEGF, vascular endothelial growth factor; DLPFC, dorsolateral prefrontal cortex; M1, motor cortex.
The general methods of allocation and blinding used previously [32]. The detailed methods were as follows. An investigator (XJW) used a computer-generated random-number table to randomly allocate patients to the three groups. The allocation information was disclosed to rTMS physical therapists and doctors (YL and YWW), via phone call by statisticians who conducted the rTMS stimulation sessions on patients (XJW). The physical therapists involved in rTMS were not engaged in any other research-related tasks. The researchers responsible for evaluating, collecting, and processing outcome data were blinded to the allocation of treatments. Treatment assignments were also concealed from patients [33].
The rTMS treatment uses a magnetic stimulator with an 8-shaped coil (MagProX100,
Tonica, Farum, Denmark). The DLPFC region was targeted for
stimulation using the “standard 5 cm” method, which involves transitioning 5 cm
forward from the left M1 region. The stimulus intensity of each group was 80% of
the resting motor threshold. The resting motor threshold was the minimum
stimulating power necessary to elicit an overt motor response (motor evoked
potential, significantly greater than 50 µV) in the contralateral abductor
pollicis brevis, on at least 5 of 10 trials [34]. The dual-site stimulation was
performed sequentially. The L-DLPFC is stimulated first, then the ipsilesional
M1. During the actual procedure, we used a new and more rigorous method for sham
stimulation. The coil was positioned at the identical scalp location and operated
with identical parameters as those used for the dual-target group [32]. The
handle was oriented downwards, and the coil was tilted by 90° in the
axial plane [9]. The edge of the coil was kept in contact with the scalp to
produce audible clicking noises [35, 36, 37]. This approach ensured that the
sham-stimulation procedure closely resembled the real stimulation, but without
producing significant neural effects. Fig. 3 shows rTMS intervention details.
Stimulation parameters: L-DLPFC: 10 Hz, 80% resting motor
threshold (RMT), 2000 pulses (5 s
 Fig. 3.
Fig. 3.
The details of repetitive transcranil magnetic stimulation (rTMS) intervention. RMT, resting motor threshold; L-DLPFC, left dorsolateral prefrontal cortex.
All assessments were done twice, pre-treatment (baseline) and post-treatment. The main evaluation indicator were MoCA scores. The version we chose is Montreal Cognitive Assessment Beijing Version (MoCA-BJ), a tool that evaluates the overall cognitive function [38, 39]. The secondary results included the evaluation of the basic activities of daily life by the Modified Barthel Index (MBI) [40], visual attention, executive function with the Trail-making Test (TMT) [41, 42] and attentional function with the Digital Span Test (DST) [43]. Additionally, serum samples were collected to assess the levels of BDNF and VEGF. At baseline, blood specimens were collected before rTMS stimulation. After 4 weeks, blood specimens were collected the day after completing rTMS stimulation. For the blood draw, the patient started fasting at 18.00; 5 mL of peripheral blood were collected the next morning [44]. The blood specimen was centrifuged, and serum was extracted and then stored at –80 °C. The quantification of BDNF and VEGF serum levels was accomplished with enzyme-linked immunosorbent assay (ELISA) kits (Multi-Sciences, Hangzhou, Zhejiang, China). Strict adherence to the provided instructions was observed throughout the procedure. A single technician, blind to the treatment status of the patients, conducted all measurements. Each sample was duplicated and the results were averaged to ensure precision and consistency.
Statistical analysis was performed using SPSS Software 25 (IBM-SPSS Statistics,
Chicago, IL, USA) [45]. A paired t-test or Wilcoxon rank-sum test was
used for intragroup comparison. Intergroup comparision was done by one-way
analysis of variance (ANOVA) or Kruskal-Wallis H test, then a post-hoc analysis
was conducted. If there was a difference at baseline, such as in MoCA scores or
DST Inverted sequence scores, pre-treatment data were used in a covariate
analysis to eliminate the effect of baseline differences. The level of
statistical significance was set at p
Sixty right-handed patients underwent qualification screening; 3 did not meet the selection criteria. Fifty-seven patients were randomly assigned to three treatment groups, with n = 19/gp. Nine patients did not complete the research project. Forty-eight, therefore, were included in the analysis as shown in the patient flow chart (Fig. 1). Table 1 lists the demographic and clinical characteristics of the sample.
Table 2 shows that after adjusting for Pre-intervention MoCA scores, the post-Intervention MoCA scores of different groups varied after intervention (F = 4.645, p = 0.015). Compared with the baseline, the MoCA scores of all groups were higher at 4 weeks. Post-hoc analysis revealed that the dual-target rTMS group score was significantly higher than the sham rTMS group score (p = 0.021), and the dual-target rTMS group score was significantly higher than the single-target rTMS group score (p = 0.008), upon the completion of the treatment protocol (Fig. 4).
| Group | Pre-Intervention | Post-Intervention (Before correction) | Post-Intervention (After correction) |
| Mean (SD) | Mean | ||
| Sham-rTMS | 14.00 |
16.00 (3.74) | 18.58 |
| Single-target rTMS | 18.00 |
19.87 (3.14) | 18.50 |
| Dual-target rTMS | 18.00 |
21.53 (3.20) | 19.80 |
#p
aRepresents a statistically significant difference compared to the
dual-target rTMS group, p
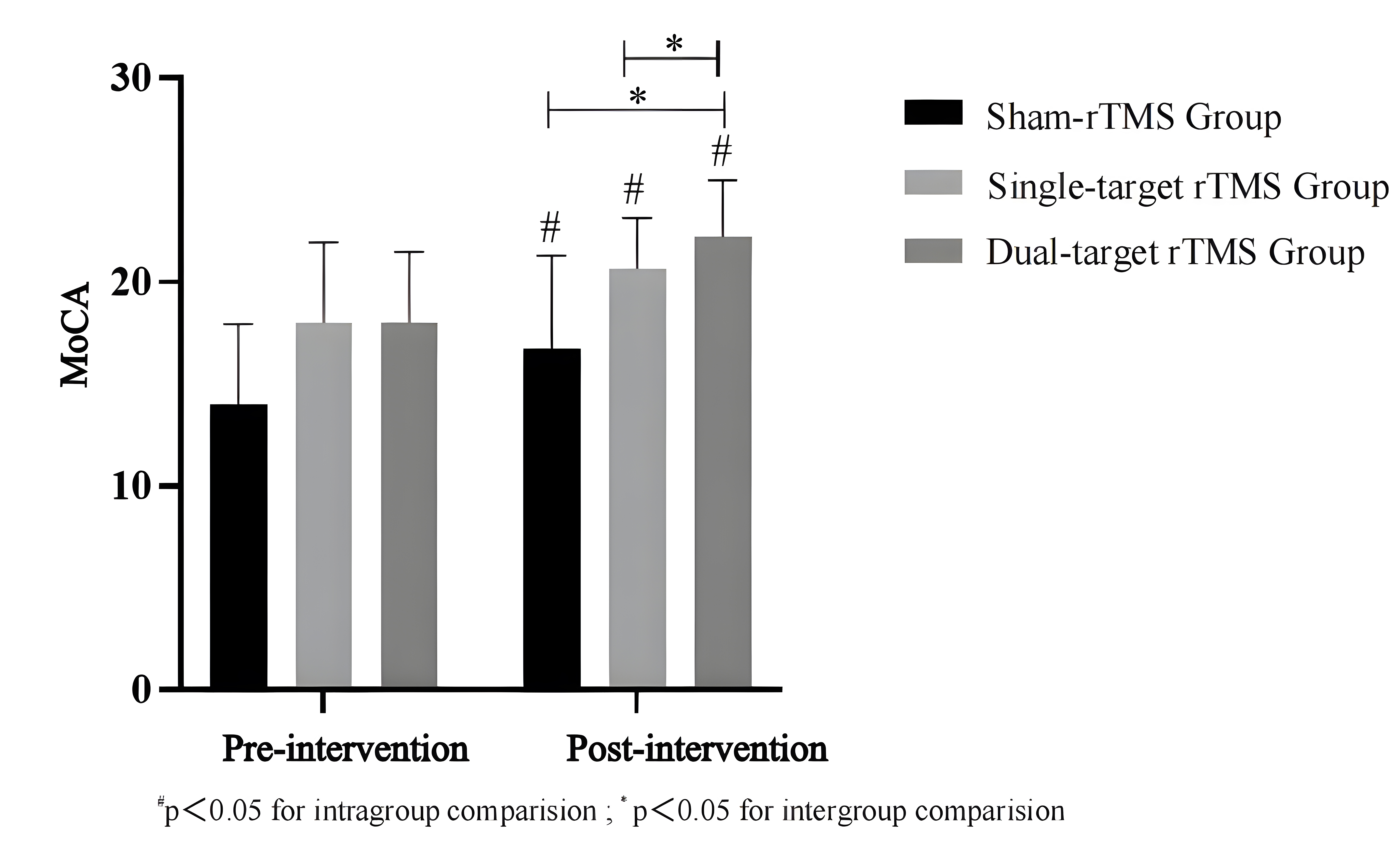 Fig. 4.
Fig. 4.
Comparison of Montreal Cognitive Assessment (MoCA) scores before and after treatment for the three groups.
Table 3 shows the MBI, TMT and DST scores after intervention. Compared with the baseline, the MBI, TMT, and MoCA scores of the dual-target rTMS group and single-target rTMS group improved after 4 weeks of rTMS treatment and cognitive therapy.
| Sham-rTMS | Single-target rTMS | Dual-target rTMS | F | p | |
| MBI | 46.56 |
55.40 |
55.13 |
2.484 | 0.093 |
| TMT-A time | 134.29 |
100.80 |
98.47 |
7.811 | 0.006* |
| TMT-B time | 215.31 |
152.00 |
203.53 |
2.130 | 0.131 |
| DST sequence | 5.11 |
6.00 |
5.27 |
1.825 | 0.171 |
| DST inverted sequence | 1.89 |
2.87 |
3.07 |
6.209 | 0.004* |
#p
There were no noteworthy disparities in the baseline MBI measurements of the
three study groups. After 4-week intervention, MBI increased significantly in
both dual-target rTMS group (55.13, 95% confidence interval (CI), 48.00 to
62.27; p
3.3.2.1 TMT-A Time
The TMT-A time scores improved from baseline, significantly, over 4 weeks in both the dual-target rTMS group (p = 0.005) and the single-target rTMS group (p = 0.012), but not in the sham-rTMS group (p = 0.582) (Table 3). The between-group post-treatment scores differed significantly among groups (F = 5.779, p = 0.006). Post-hoc analysis showed that, after treatment, the dual-target rTMS group exhibited significantly lower scores than did the sham rTMS group (p = 0.013), and the single-target group scores were significantly lower than those of the sham rTMS group (p = 0.023).
3.3.2.2 TMT-B Time
The three groups exhibited no between-group significant differences either before or after the intervention measures were implemented. The TMT-B time scores improved significantly at 4 weeks in both the dual-target rTMS group (p = 0.037) and single-target group (p = 0.021), but not in the sham-rTMS group (p = 0.656) (Table 3).
3.3.3.1 DST Sequence
Prior to and after the intervention, there were no significant differences
between-group differences. The DST-sequence scores improved significantly after 4
weeks of treatment in all three groups: the dual-target rTMS group (p
3.3.3.2 DST Inverted Sequence
The DST inverted-sequence scores improved significantly after 4 weeks of treatment in both the dual-target rTMS group (p = 0.028) and the single-target rTMS group (p = 0.028), but not in the sham-rTMS group (p = 0.111) (Table 3). Post-hoc analysis of between-group differences in post-treatment scores revealed significantly higher post-treatment scores in DST inverted-sequence scores in the dual-target rTMS group than in the sham rTMS group (p = 0.007), and higher scores in the single-target rTMS group than in the sham rTMS group (p = 0.030) (Table 3).
The comparison of baseline BDNF and VEGF levels in patients in three groups
showed no significant differences (p
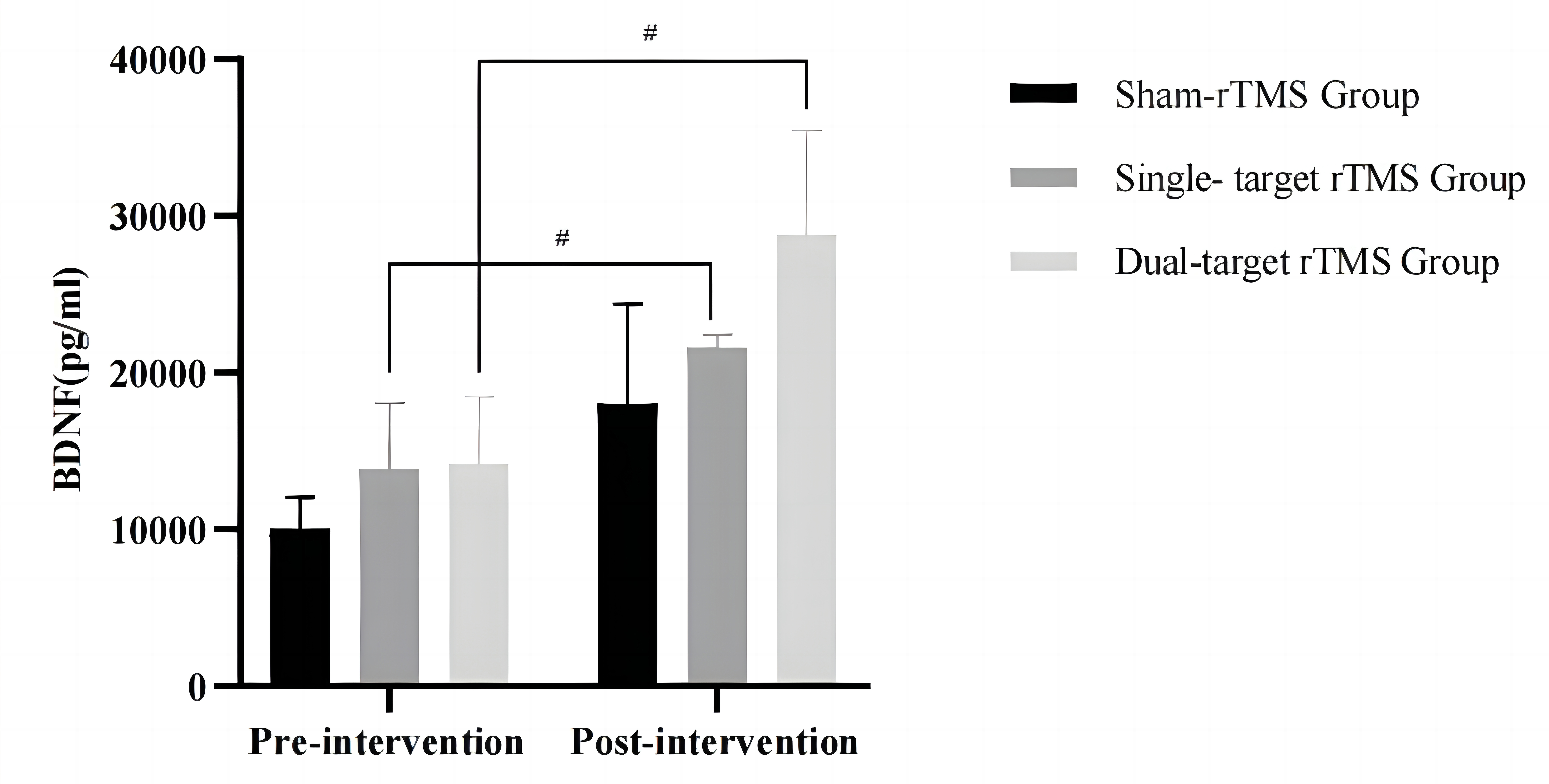 Fig. 5.
Fig. 5.
Comparison of BDNF levels of the three groups before and after
treatment. #p
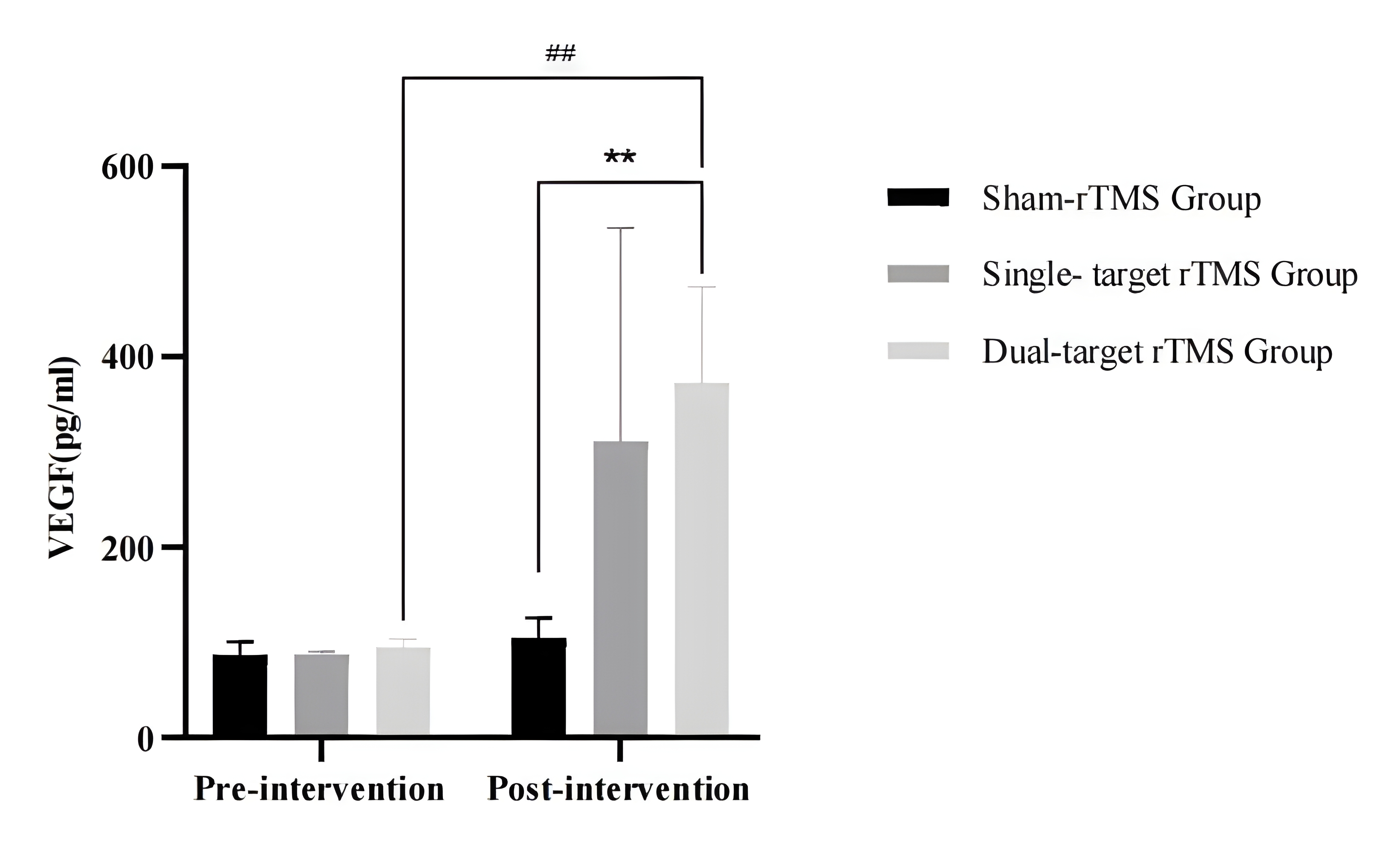 Fig. 6.
Fig. 6.
Comparison of VEGF levels of the three groups before and after
treatment. ##p
Our research has established the practicality of using a 20-session treatment regimen of 10-Hz rTMS, targeted at dual regions (L-DLPFC and ipsilesional M1), in conjunction with traditional rehabilitation therapy, for the effective treatment of patients with PSCI. Overall cognitive function was significantly enhanced by the cumulative impact of bilateral high-frequency rTMS, surpassing both the single-target rTMS and sham rTMS in efficacy. Dual-target 10-Hz rTMS, when combined with traditional rehabilitation methods, is anticipated to yield more favorable outcomes in the management of PSCI patients.
The improvement effect on specific cognitive domains is not obvious. The findings of research examining the impact of rTMS on executive function remain inconclusive. Certain studies have reported favorable outcomes associated with the use of single-target rTMS. For instance, Wang Lu et al. [7] reported that the combination of high-frequency rTMS with cognitive training was found to effectively enhance executive function among stroke patients. Kim et al. [8] observed that high-frequency rTMS applied to ipsilesional DLPFC resulted in significant improvement in cognitive function among stroke patients. They also observed an increase in both forward and reverse DST scores, suggesting that stimulation of DLPFC with 10-Hz rTMS had a definite therapeutic effect on post-stroke execution impairment [8]. The present study showed that there was significant improvement in TMT-A time and DST inverted sequence in the dual-target rTMS group after treatment.
Cognitive function is closely related to a patient’s ability to function in
daily life. Cognitive impairment slows down the processing speed of the central
nervous system when patients face external information, which is manifested as
decreased executive ability, weakened reaction ability, and poor self-care
ability [46, 47]. Our findings show that all three research groups improved over
their pre-treatment baselines (p
There are several candidate mechanisms by which rTMS can be beneficial in PSCI therapy: (a) rTMS can exert significant influence on various neurobiological processes, including the modulation of metabolic activity in both stimulated and remote brain regions; (b) by the alteration of oxidative stress levels; (c) by activation of neurotransmitter systems; (d) by enhancing the plasticity of synapses; (e) through long-term potentiation; and (f) through the connectivity and functionality of neuronal networks [51, 52, 53, 54]. Additionally, according to recent reports, 20-Hz rTMS has been demonstrated to elevate regional cerebral blood flow in the left DLPFC, in addition to other regions such as the cingulate gyrus, basal ganglia, hippocampus, amygdala, and insula [54, 55]. In our research, an increase in serum BDNF and VEGF suggests that curative strategies may be beneficial through increasing synaptic plasticity and promoting the transmission function between synapses, which facilitates learning and the release of neurotrophic factors.
At present, research on the utilisation of rTMS in the management of PSCI is still in the exploratory stage, and there is little research in China on the application of dual-target rTMS. In the present experiment, a comparison was made between sham-rTMS stimulation, single-target rTMS, and dual-target rTMS. Based on the findings of this study, the potential exists to devise an innovative bilateral rTMS approach, which can serve as a therapeutic tool for neurorehabilitation by enhancing cognitive function among stroke survivors.
The current investigation is subject to several constraints. First, it will be necessary in the future to conduct research on a larger sample size of patients to ascertain the precise impact of bilateral rTMS on various stroke subtypes. Second, there was a lack of post-treatment follow-up. Third, there may be subjective errors in the cognitive assessment scales used to assess the effectiveness of treating cognitive impairment. In the future, we will apply techniques such as electroencephalography, positron emission tomography (PET), and functional magnetic resonance imaging (fMRI) to evaluate the degree of cognitive impairment and further explore the mechanism by which rTMS enhances cognitive function in individuals with cognitive impairment. Fourth, the degree of cognitive impairment of the patients included in this study was not limited to mild or moderate. Doing so would have avoided the cognitive differences observed in the patients’ baseline scores. Fifth, the method of sham-stimulation in our study may not have provided a realistic-enough sensation. Recently we learned that a coil has been especially devised for false stimulation that can make similar sounds, but has no actual stimulation effect. The sham-stimulation method should be more realistic in future studies.
The present study presented data showing that a dual-target rTMS therapy is effective for PSCI. The stimulation exhibited remarkable efficacy, suggesting that dual-target stimulation (L-DLPFC+M1) holds promise as a potential target for TMS therapy in individuals with cognitive impairment after stroke.
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
BSX and CRL designed the research study. HW performed the research. YWW and YL provided help on investigation and methodology. XJW analyzed the data. BSX wrote the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Ethical approval have been obtained for Medical Ethics Committee of Shanghai Health & Medicine College on October 27, 2022 (2022-E4-6100-21-201034-03-210302197009090947).
The authors are grateful to the support from Shanghai University of Medicine & Health Sciences and Shanghai Health Rehabilitation Hospital.
This research was funded by Shanghai Health Rehabilitation Hospital, grant number E4-6100-21-201,034.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






