1 Institute of Higher Nervous Activity and Neurophysiology of the Russian Academy of Sciences, 117485 Moscow, Russia
2 Pushkin State Russian Language Institute, 117485 Moscow, Russia
3 Federal Research and Clinical Center of Intensive Care Medicine and Rehabilitology, 107031 Moscow, Russia
Abstract
Background: The study of tactile perception during a childhood is extremely important for understanding the social and communicative aspects of the child’s development. Tactile perception of stimuli with different valence can have different normative stages of development. Methods: In the present study, we examined changes in linear and nonlinear electroencephalogram (EEG) parameters during the presentation of pleasant (C-tactile optimal stroking), unpleasant (ice stroking), and neutral tactile stimuli in three groups of healthy volunteers: preschoolers 4 and 5 years, school-age children from 8 to 10 years, and adults from 20 to 40 years. Results: According to our findings, child maturation plays a significant role in the perception of pleasant and neutral tactile stimuli. Patterns of EEG dynamics related to tactile perception showed greater similarity between adult volunteers and school-aged children than preschoolers. Conclusions: Non-linear EEG parameters such as fractal dimension (FD), envelope mean frequency (EMF), and power spectral density (PSD) dynamics of the theta-rhythm were particularly sensitive to developmental changes in tactile perception. Hjorth complexity and peak alpha frequency (PAF) scores may serve as indicators of mature perception of С-tactile (CT)-stimuli.
Keywords
- C-tactile system
- school-aged children
- preschoolers
- adults
- EEG
- FD
- EMF
- PSD
- Hjort complexity
- PAF
Tactile perception is both the simplest and most important way for children to interact with the sensory world around them through physical contact [1]. The sense of touch is also the first sense that children develop while in the maternal womb [2]. During ontogenesis, a child’s tactile perception continues to develop and they actively explore the world through touch. Using their hands, the child learns to recognize and differentiate between different tactile stimuli, gaining knowledge of their biological, social, and cognitive meaning. According to Jones and Lederman (2006) [3], as early as 3 months of age, babies are able to distinguish between objects of different shapes by touch. By 6 months of age, they can also distinguish the hardness and texture of objects. In addition to the physical attributes of tactile stimuli, the child also learns to distinguish between their social and cultural aspects. He learns which touches of others are socially and culturally acceptable and which are not [4]. Moreover, touch of caregivers has been experimentally shown to promote the development of object categorization in other modalities in six- to nine month-olds [5].
Thus, as the child grows, initially biologically meaningful tactile stimuli acquire multiple cognitive, cultural, and social characteristics. The child learns to respond to these stimuli based on his or her own experiences.
While responses other unpleasant tactile stimuli, such as pain or excessive force or pressure or the sensation of a moving insect, undergo only minor changes to adapt to social and cultural aspects of the environment and reaching adult values to the early childhood, the ways in which a child responds to pleasant and neutral stimuli undergo significant changes [6, 7]. These changes depend largely on the environment in which the child is raised. Reactions to unpleasant and painful stimuli are usually instinctive and involve an attempt to avoid or alleviate unpleasant sensations. However, even reactions to stimulants such as fire or cold can be conditioned by sociocultural upbringing and depend on the unique personality traits of the individual [8, 9]. The perception of pleasant and neutral tactile stimuli, on the other hand, is highly dependent on social and cultural factors and develops as the child grows up and interacts with others [10]. It is the social and cultural traditions accepted in a particular community that form the so-called social distance and determine the acceptability of various tactile contacts between people [4, 11].
Differences in the development of perception of emotionally significant tactile stimuli with different valence can be explained by differences in the anatomy of their transmitting pathways and analyzers. For example, the sensation of pain is associated with receptor activation in primary afferent fibers, including both unmyelinated and myelinated fibers. Signals are then transmitted from the periphery to the posterior horns of the spinal cord, then through the brainstem and subcortical structures, eventually reaching the somatosensory cortex and limbic system [12]. Temperature sensitivity develops in a similar pattern, but the information is projected to different thalamic nuclei, eventually ending up in the insular and orbitofrontal cortex [13]. Conversely, gentle, and slow stroking, reminiscent of a caress, activates C-tactile (CT) receptors located on the human scalp. The resulting impulses travel along the spinothalamic tract and terminate in the insular cortex rather than the somatosensory cortex [14, 15, 16]. Perception of emotionally neutral features of tactile stimuli requires the formation of the cognitive skill of classifying stimuli according to certain parameters, including roughness, salience, stickiness, wetness, warmth, etc. [17, 18]. According to the literature, multiple receptors may be involved in the perception of the structure of a tactile stimulus [19], information from which flows to various cortical structures including somatosensory and motor areas, as well as temporal cortex [20, 21]. Therefore, it is important to separately examine the development of perception of emotionally salient stimuli with different valence (pleasant and unpleasant), as well as neutral stimuli, because they have clear anatomical differences and different dependence on sociocultural and personality factors. In this study, three stimuli (pleasant, neutral, and unpleasant) were carefully selected and presented under identical conditions to preschoolers, schoolchildren, and healthy adult volunteers. The study examined both the behavioral reactions of the subjects and the changes in electroencephalogram (EEG) activity when these stimuli were presented.
Although there have been no studies with a similar design to ours, and the number of available studies is extremely limited, some known patterns of tactile development in early life can be cited. Thus it has been established that adult-like pattern of haemodynamic responses to affective and non-affective touch can be recorded in two-year-old subjects [22]. It has also been shown that acuity and sensitivity decrease with age and that in children they are already behaviorally separate from affective touch [23]. The development of tactile skills in children is likely to have a two-way relationship with the development of sensitivity in other modalities, as the recognition of tactile pictures to touch improves with age and is mainly dependent on the development of shape discrimination skills [24], while the development of categorization skills in visual modality depends on the presence/absence of tactile support [5].
When planning the research, we assumed that traditional EEG markers were not effective to predict the emotional response to tactile stimulation, while the features of non-linear dynamics of the EEG and complex approach of analysis were more sensitive to the emotional aspects of tactile perception [25]. As was previously showed non-linear features of the EEG were one of the most sensitive and useful approaches to recognize emotions based on electroencephalography [26]. The ability to distinguish emotional valences independently on sensory modality demonstrated Higuchi fractal dimension, Hillbert envelope frequency, entropy of the EEG and the Hjorth parameters [26, 27]. Regarding tactile stimulation, some researchers reported, that unlike to linear indices, the nonlinear features, including Higuchi’s dimension and Hjorth parameters could be used to distinguish the difficult-to-detect subtleties of frictional interaction between skin and object surface and could be useful to detect different levels of roughness during passive tactile stimulation [25, 28, 29, 30]. Other studies had indicated that non-linear measures, including envelope frequency, Hjorth complexity (HC), and fractal dimension (FD), when combined with spectral characteristics and peak alpha frequency (PAF), can serve as an effective tool for evaluating the subjective pleasantness of tactile stimulation [31, 32]. The EEG studies taking into account wide range of frequency bands or used cluster analysis significantly improved sensitivity to the different parameters of tactile stimulation including force and velocity and perceived sensations including pain and touch [33, 34].
Currently, there is limited research comparing how children of different age groups perceive pleasant and unpleasant tactile sensations. In addition, most child development research on unpleasant sensations uses stimuli that are considered conditionally neutral, including surfaces that are unpleasant to touch or brushes with stiff bristles, to label unpleasant sensations [35].
We aimed to investigate the developmental changes of the EEG in response to
CT-fibers optimal stroking, neutral tactile stimuli triggered mostly
alpha-beta (A
Three age groups of subjects participated in the study. Thirty-one healthy typically developing preschoolers aged 4 to 5 years (right-handed, 16 males) and 30 healthy typically developing schoolchildren aged 8 to 10 years (right-handed, 15 males) participated in the study. Also, 30 healthy adult volunteers between 20 and 40 years of age (right-handed, 14 males, mean age 29.3) participated in the experiment. All participants took the Wechsler intelligence test (see Table 1).
| Group | Number | Age (y) | Sex (F:M) | WISC-V * |
| Adults | 30 | 30.2 |
14:16 | 112.8 |
| Preschoolers | 31 | 4.6 |
16:15 | 109.2 |
| Schoolchildren | 30 | 9.1 |
15:15 | 110.7 |
In adults the Wechsler Adult Intelligence Scale was used; WISC-V Test (Wechsler Intelligence Scale for Children) was used in children.
* the total values are presented. F, female; M, male.
All participants (and parents of children) were interviewed before study by clinical psychologist and physician to exclude the development, psychological or neurological disorders, to check handedness, and to describe the design of the experiment and the type of stimuli. Exclusion criteria were as follows: history of anxiety, depressive, personality, or psychotic disorders, post-traumatic stress disorder (PTSD), sleep disorders, attention-deficit hyperactivity disorder (ADHD), and other trauma-related mental disorders; autism/social communication disorders; language dysfunction; tactile hypersensitivity; and ethnic minorities, abnormalities during EEG registration, left-handed or ambidexter.
Prior to the study, both children and their parents received an introduction to the experimental procedures and had the opportunity to familiarize themselves with tactile stimulation several days before the experiment. If any of the participants showed intolerance to cold or other tactile stimuli, the study was not conducted for their safety and comfort. The study was performed following the ethical standards reported in the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. The Ethics Committee approved the study protocol of the Pushkin State Russian Language Institute (Moscow, Russia) (protocol code 17-3-24-118, date of approval 15.07.22). All individuals provided written informed consent after receiving a complete description of this study.
Subjects were presented with 3 types of stimuli (Fig. 1A):
(1) slow stroking with a soft brush (2–5 cm/s) with medium pressure;
(2) slow stroking with a bristle brush (2–5 cm/s) with medium-strong pressure;
(3) slow stroking with a specialized ice brush (2–5 cm/s).
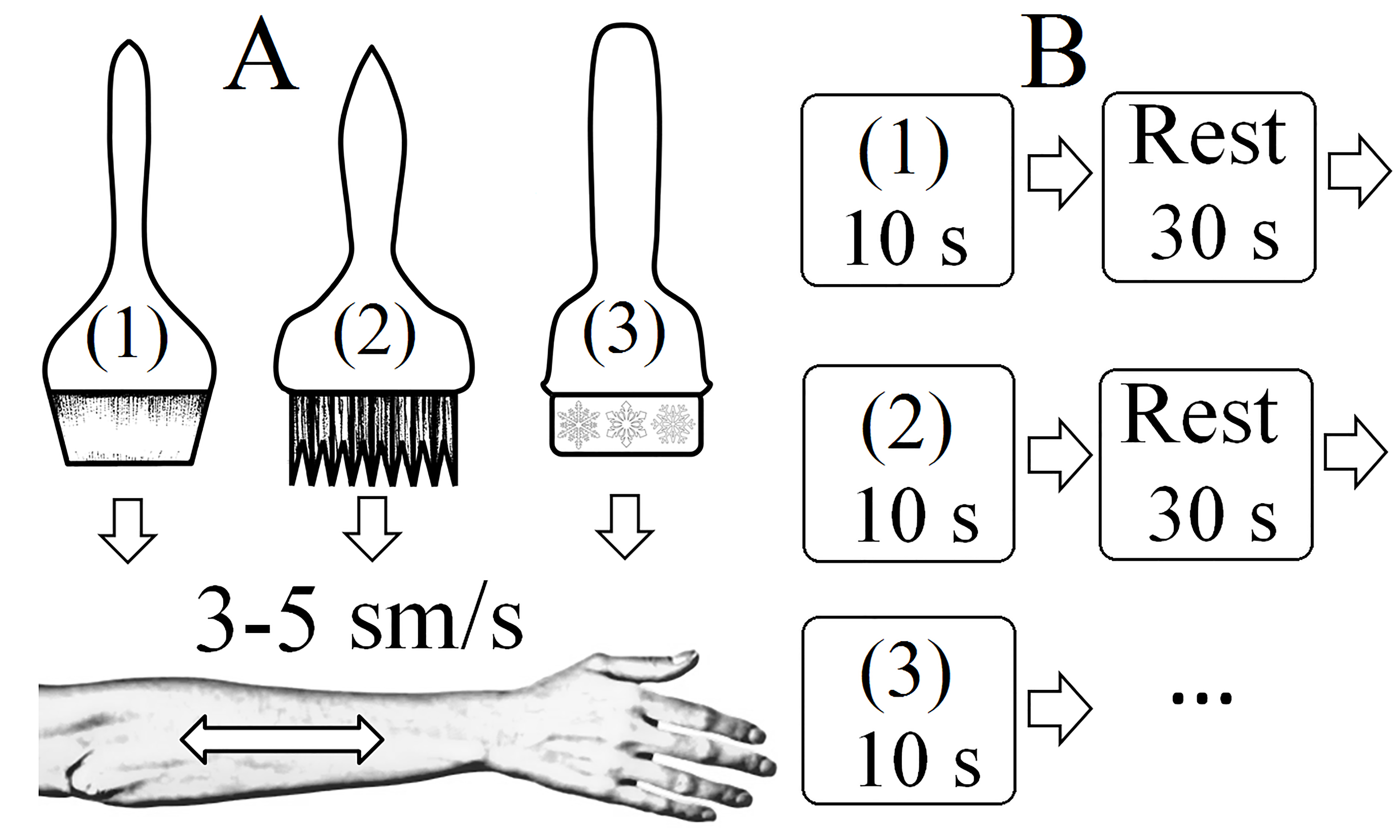 Fig. 1.
Fig. 1.
Paradigm and stimuli. (A) Types of stimuli presented (three types of brushes) and paradigm presentation scheme. (1) soft brush (pleasant stimulus); (2) hard brush (neutral stimulus); (3) ice brush (unpleasant stimulus). (B) Paradigm diagram (1)-(3) - stimuli types.
The stimuli were selected basing on our previous studies, demonstrated which type of stimuli induced pleasant or unpleasant response in children in adults [21] and the pilot study described below.
The pilot study consisted only on stimuli assessment and recruited 38 healthy adults from 20 to 40 years old (20 males, mean age 30.6). Participants assessed the stroking of nine stimuli types (three pleasant, three unpleasant and three neutral), which included two surfaces of soft brush, two surfaces of bristle brush, neurological wheel, hot bags, ice, vibration, textile surface. The stimuli were presented with slow velocity and varied pressure (from medium to strong). The stimuli were assessed by the following scale: pleasantness, pain, arousal, and tickling. As a result, three stimuli were selected: soft brush with artificial squirrel texture with medium pressure (most pleasant stimulus with mean pleasantness 7.7), bristle artificial plastic brush with medium-strong pressure (as most neutral stimuli with mean pleasantness 4.9) and the ice with medium pressure (as most unpleasant stimulus with mean pleasantness 2.1)
The experimental study began with EEG resting state 1–1.5 min minutes with eyes opened and 30–60 seconds with eyes closed, then followed with 1 min the eyes opened recording. At the initial stages of our study, we found that preschoolers had difficulties to keep eyes closed during tactile stimulation, that is why the resting state EEG with eyes opened was selected as a control or background condition for all groups of participants. All stimulation was presented during eyes opened and we compared it only with eyes opened recording. During pilot recordings we had to exclude the presentation of stimuli with eyes closed (preschoolers had difficulties to keep eyes closed during tactile stimulation) and the closed eyes condition was used only for a formal EEG assessment.
The pressure force was selected in the pilot study so that the stimuli differed
from each other as much as possible in terms of emotional valence: the pressure
and stiffness of the brush were selected to be the most pleasant in one case and
the most neutral or even unpleasant in the other. Based on the results of the
pilot experiment, a soft 50 mm wide squirrel brush and a very stiff bristle brush
also 50 mm wide were selected. As an unpleasant tactile stimulus, an ice with a
stimulating silicone surface of 5
To eliminate the discomfort experienced by most preschoolers in the eyes-closed situation, we included tactile stimulation for participants while they sat with their eyes open in a darkened room. To prevent anticipation of the upcoming stimulus, we used a cloth screen. The stimuli’ onset and offset were marked manually using EEG-registration interface. Additionally, we used video directed to area of stimulation and the EEG registration module to control and correct the timing and duration of the events.
Two experimenters participated in data collection: a doctor and a clinical psychologist. The doctor was involved in fixing the EEG helmet and presenting tactile stimulation. The clinical psychologist assessed the consistency of the video recording and EEG recording and was responsible for the subjective assessment of the stimuli by children and adults. Always right hand was stimulated.
The experimenter was trained to present the stimulus under identical conditions (same speed and pressure) with video demonstrated correct pressure and velocity. The speed was additionally controlled by the video: the velocity of each stroking was measured and it did not go beyond 2–5 cm/sec. The stroking was back-and-forth movement. The 15 sm of the arm surface was stimulated with continuous movement.
A presentation marker was presented at the beginning and end of stimulus presentation. Each unpleasant stimulus was presented 10 times for 10 s, and each pleasant or neutral stimulus was presented 20 times for 10 s in a randomized order, with an inter-stimulus interval of 30–40 s (Fig. 1B).
After the main phase of the study and a short break, each stimulus was presented once, and subjects (including both adults and children) were asked to rate the following parameters on a scale of 1 to 9: pleasantness, pain, arousal, and tickling.
EEG was acquired using a 19-channel EEG amplifier Encephalan with the recording
of polygraphy channels (Poly4, Medicom MTD, Taganrog, Russia). The
sampling rate was 250 Hz. The amplifier bandpass filter was nominally set to
0.05–70 Hz. AgCl electrodes (Fp1, Fp2, F7, F3, Fz, F4, F8, T3, C3, Cz, C4, T4,
T5, P3, Pz, P4, T6, O1, and O2) were placed according to the International 10–20
system. The electrodes placed on the left and right mastoids served as joint
references under unipolar montage. The vertical electrooculogram (EOG) was recorded with AgCl cup
electrodes (Poly4, Medicom MTD, Taganrog, Russia) placed 1 cm above and below the left eye, and the horizontal EOG was
acquired by electrodes placed 1 cm lateral from the outer canthi of both eyes.
The electrode impedances were kept below 10 k
Each subject’s EEG recordings consisted of 3 minutes of resting state EEG slices with eyes closed (approximately 30 seconds) and eyes open (2–2.5 minutes), followed by approximately 30 minutes of EEG with tactile stimulation. To eliminate unwanted artifacts such as oculomotor, electrocardiogram (ECG), and muscle interference, independent component analysis was applied to the EEG recordings using EEGLab 14_1_2b software (Matlab toolbox, Swartz Center for Computational Neuroscience, San Diego, CA, USA) [36]. Visual analysis was then performed and any remaining artifacts were manually removed, ensuring that no more than 5 seconds of the background recording and no more than 1% of the EEG recordings contained any labels.
The resulting data were fragmented according to stimulus type (labeled as
Presentation markers). Further analysis of the EEG data included these fragments.
To calculate linear and nonlinear EEG parameters, 10-second intervals for each
type of tactile stimulation were used (10
All EEG characteristics (see below) were calculated for each electrode and then averaged over the frontal (F3, F4, Fz), central (C3, C4, Cz) and parietal areas (P3, P4, Pz, O1, O2).
Power Spectral Density (PSD) of EEG was analyzed using Matlab R2018b program (Matsoft, Natvik, MA, USA). Fast Fourier transform (FFT) was used to analyze the power spectra of the studied EEG fragments. The EEG spectrum was estimated for each studied condition. The obtained power spectra were averaged in 1 Hz steps in the range from 2 to 20 Hz (2–3 Hz, 3–4 Hz, … 19–20 Hz). Logarithmic power values were used for further statistical analysis.
The PAF identification was conducted by exploring the
source-level power spectra at scalp electrodes using a fast Fourier transform
(FFT) for a continuous EEG segment of 220 s. Alpha peak frequency was identified
as the center of gravity frequency within the 7–13 Hz band. The center of
gravity frequency refers to the “weighted sum of spectral estimates divided by
alpha power”:
Fractal dimension D2 was calculated for the entire frequency range (2–20 Hz). The mathematical computation of fractal dimension D2 was performed using the Higuchi method [37], based on the transition from the original sequence to sequences obtained by decimation, i.e., taking every k-th observation. For each selected value of k, k such independent series were generated, differing in the shift of the starting point. For each of them, the curve length was computed as the sum of absolute differences between data points, normalized to the length of the original curve. The arithmetic mean of these calculated lengths was then computed across all k series taken with a step size of k, resulting in an averaged estimation of the length L(k). The FD was calculated using tool prepared in C++ environment.
For series with a fractal dimension D, the function for series with a fractal
dimension D, the function
The Envelope Mean Frequency (EMF) was calculated using tool prepared in C++ environment. We calculated the EEG signal envelope using the Hilbert transform and analyzed the envelope frequency in the 2–20 Hz frequency range. To calculate the Hilbert envelope frequency, the Hilbert transform y(t) of the function x(t) was first calculated using the following formula.
The envelope was calculated as
and then was subjected to Fourier transformation to calculate its spectrum. In addition, the average frequency of the Hilbert envelope will be calculated as follows:
This frequency (
We calculated the HC parameter in the 2–20 Hz range:
HC =
Statistical analysis was performed using Statistica13 software (StatSoft Inc,
Hamburg, Germany). To compare the effect of tactile stimulation (three types of
stimuli and resting state) on EEG dynamics between groups, we used repeated
measures analysis of variance (ANOVA) with Bonferroni correction (p
We found no significant group differences between the subjective evaluation of
stimuli on the scales: the pleasantness of each stimulus (the analysis was made
separately for subjective assessment of pleasant, unpleasant, and neutral
stimuli) differed neither between children, nor between children and adults
(p
| Adults | ||||
|---|---|---|---|---|
| Pleasantness | Pain | Arousal | Tickling | |
| Soft brush | 6.9 |
1.2 |
3.8 |
2.8 |
| Bristle brush | 4.9 |
2.9 |
4.2 |
1.1 |
| Ice brush | 1.2 |
5.9 |
7.0 |
1.0 |
| Preschoolers | ||||
| Pleasantness | Pain | Arousal | Tickling | |
| Soft brush | 6.6 |
1.8 |
5.0 |
3.3 |
| Bristle brush | 5.7 |
4.2 |
4.7 |
1.5 |
| Ice brush | 1.3 |
7.0 |
6.9 |
1.1 |
| Schoolchildren | ||||
| Pleasantness | Pain | Arousal | Tickling | |
| Soft brush | 6.5 |
1.4 |
4.9 |
3.0 |
| Bristle brush | 4.5 |
3.7 |
4.9 |
1.3 |
| Ice brush | 1.1 |
6.1 |
6.7 |
1.0 |
The bold font represents differences in variability of subjective assessments between groups. SD, standard deviation.
The higher SD in the perception of pleasant tactile stimulation (CT-optimal stroking) was accompanied by a deviation in subjective evaluations among adults. Fig. 2 depicts the distribution of subjective assessments in children in adults for the scale pleasantness. While in children the distribution of subjective pleasantness from stroking with a soft brush was normal (Fig. 2), in adults’ subjective evaluations of pleasant stimuli could be divided into pleasant and neutral (Fig. 2c). Moreover, the variability in subjective pleasantness of pleasant stimulus in healthy adults was associated to the EEG dynamics (Fig. 2d,e), namely the decrease in PAF during pleasant stimulation compared to the resting state (see section “Correlation Analysis”).
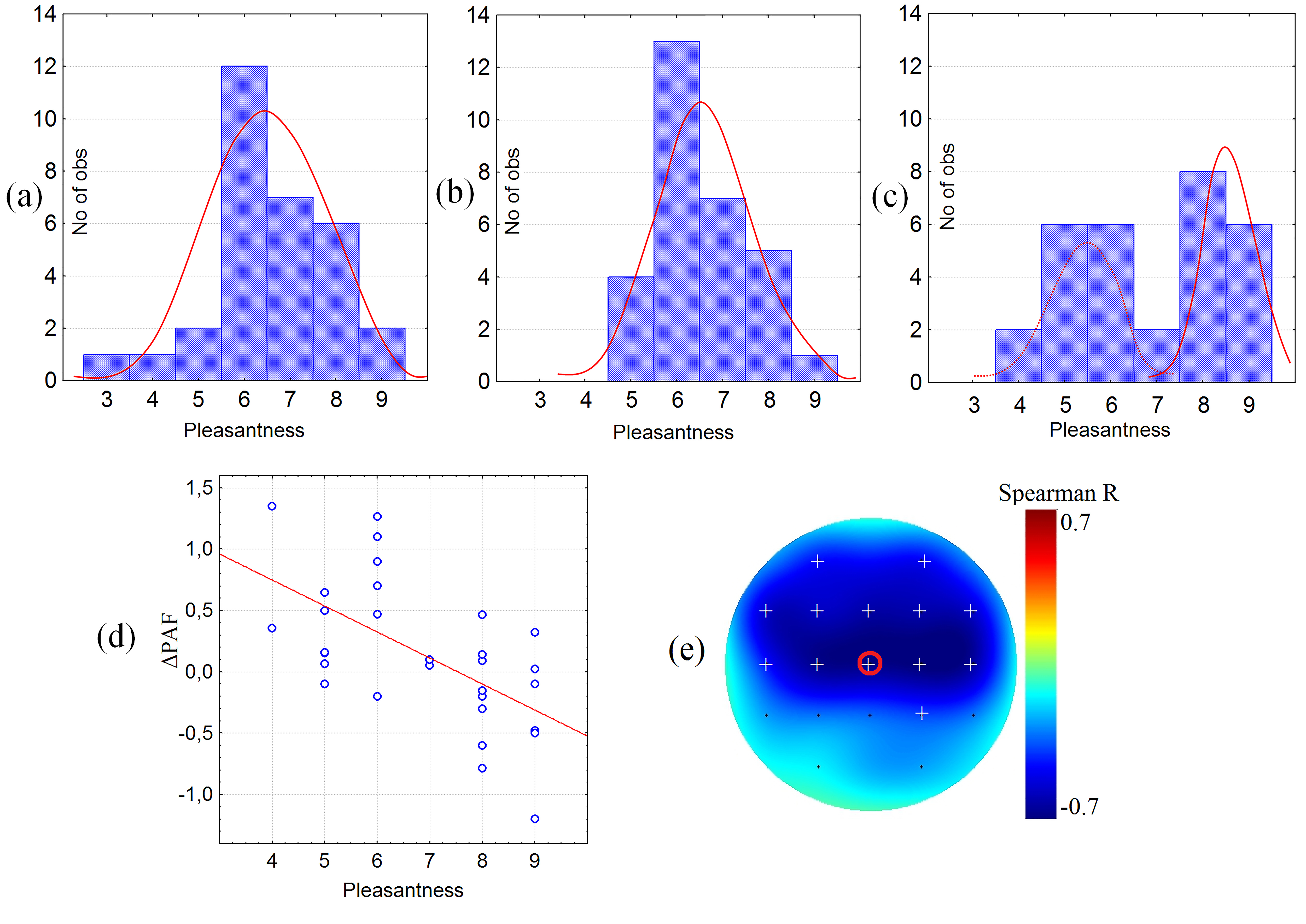 Fig. 2.
Fig. 2.
The subjective assessments. The subjective assessment of pleasant stimulus by scale “Pleasantness” in three groups of participants. (a–c) plots depict number of participants (ordinate axis) assessed the stimulus by the corresponded rate (abscissa axis, from 1 to 9 by scale “Pleasantness”): (a) preschoolers, (b) school children, (c) adults; (d) the scatterplot of the correlation between subjective pleasantness (scale from 1 to 9) of soft brush and dynamics of peak alpha frequency (PAF) (Hz) compared to the rest in adults (Spearman rank correlation r = –0.65, p = 0.0001 in Cz electrode—marked in red). (e) the topography of significant correlation between subjective pleasantness of soft brush and dynamics of PAF compared to the rest in adults. (The white crosses depict the significant correlations after Bonferroni correction. The red circle marked the electrode (Cz) for which the correlation plot was depicted.)
An increase in the theta rhythm (4–8 Hz) compared to the resting state was found in the central regions of both schoolchildren and adults, but only when an unpleasant tactile stimulus was presented. In contrast, preschoolers showed a decrease in the theta rhythm specifically for unpleasant stimuli (F(4, 168) = 15.645, p = 0.0000).
A significant decrease in alpha rhythm power in the range of 8–10 Hz was observed in schoolchildren when presented with all types of stimuli in frontal and central areas. However, in preschoolers, a significant decrease was observed only for pleasant and unpleasant tactile stimulation (F(4, 168) = 14.109, p = 0.0000). In adults, a significant decrease in alpha rhythm frequency in the range of 8–10 Hz was observed during both neutral and unpleasant tactile stimulation compared to the resting state in frontal, central and parietal areas (Fig. 3).
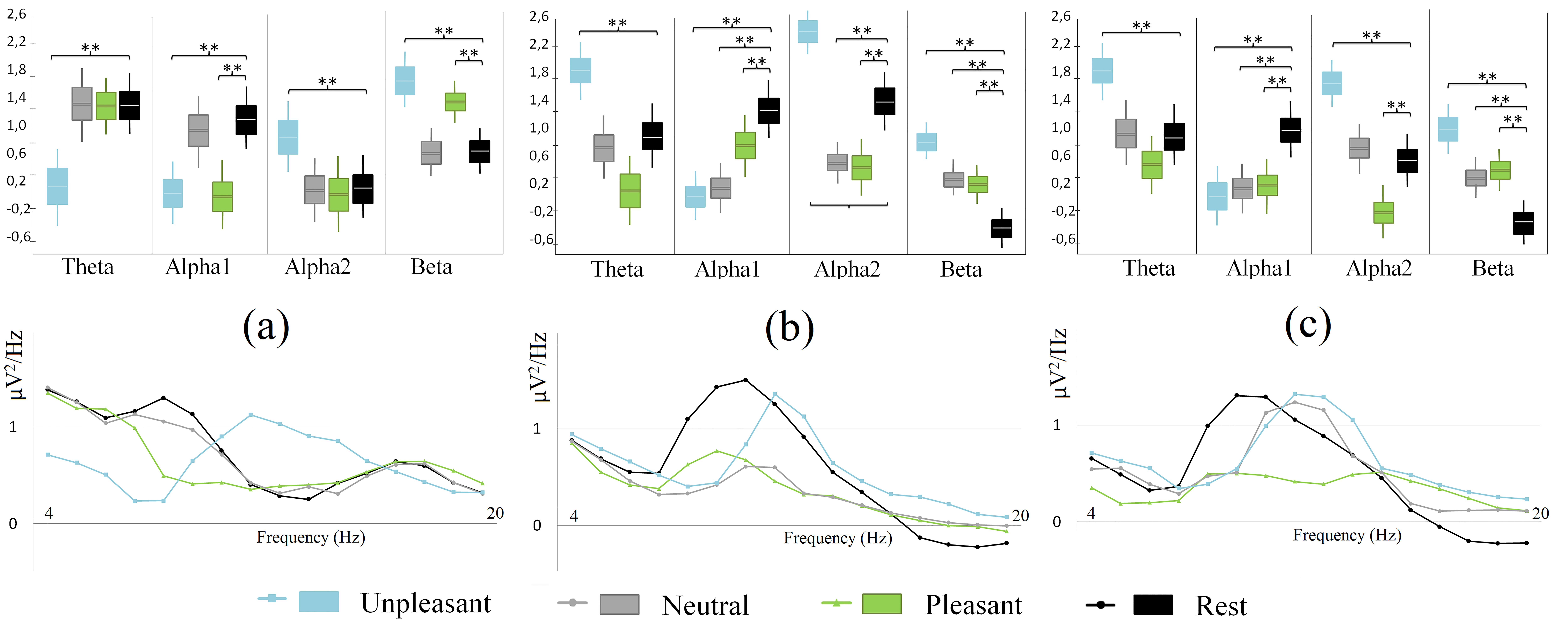 Fig. 3.
Fig. 3.
Power spectral density. Average electroencephalogram (EEG) power values in three groups of
participants. Values were calculated in the frequency ranges theta (4–8 Hz),
alpha1 (8–10 Hz), alpha2 (10–13 Hz) and beta (16–20 Hz) and power spectral
density (PSD) curves with step 1 Hz (4–5, 5–6… 19–20 Hz) when presented
with unpleasant, neutral, and pleasant tactile stimuli, as well as at rest. ** p
In schoolchildren and preschoolers, a significant increase in EEG power at 10–12 Hz was observed only during unpleasant stimuli compared to the resting state in central and parietal areas. A decrease in alpha2-rhythm power with same topography was observed in schoolchildren only when pleasant tactile stimuli were presented. In adults, EEG power at 10–12 Hz was significantly higher only for unpleasant stimuli, and a decrease in alpha2 rhythm was observed for pleasant and neutral stimuli (F(4, 168) = 12.346, p = 0.0000) incentral and parietal areas.
An increase in beta rhythm in the range of 16–20 Hz was observed in
preschoolers only when presented with pleasant and unpleasant tactile stimuli
compared to the resting state in central and frontal areas. In schoolchildren and
adults, this increase in beta rhythm was observed for all stimulus types
(Wilcoxon Matched Pairs Test, p
In adult volunteers, a significant increase in PAF in central and parietal
regions was observed during the presentation of neutral and unpleasant tactile
stimulation compared to the resting state. The data were depicted in Fig. 4a.
However, no significant differences in PAF were found when pleasant tactile
stimulation was presented (mixed stimuli group (3) stimuli (4) effect F(6, 264) =
18.234, p
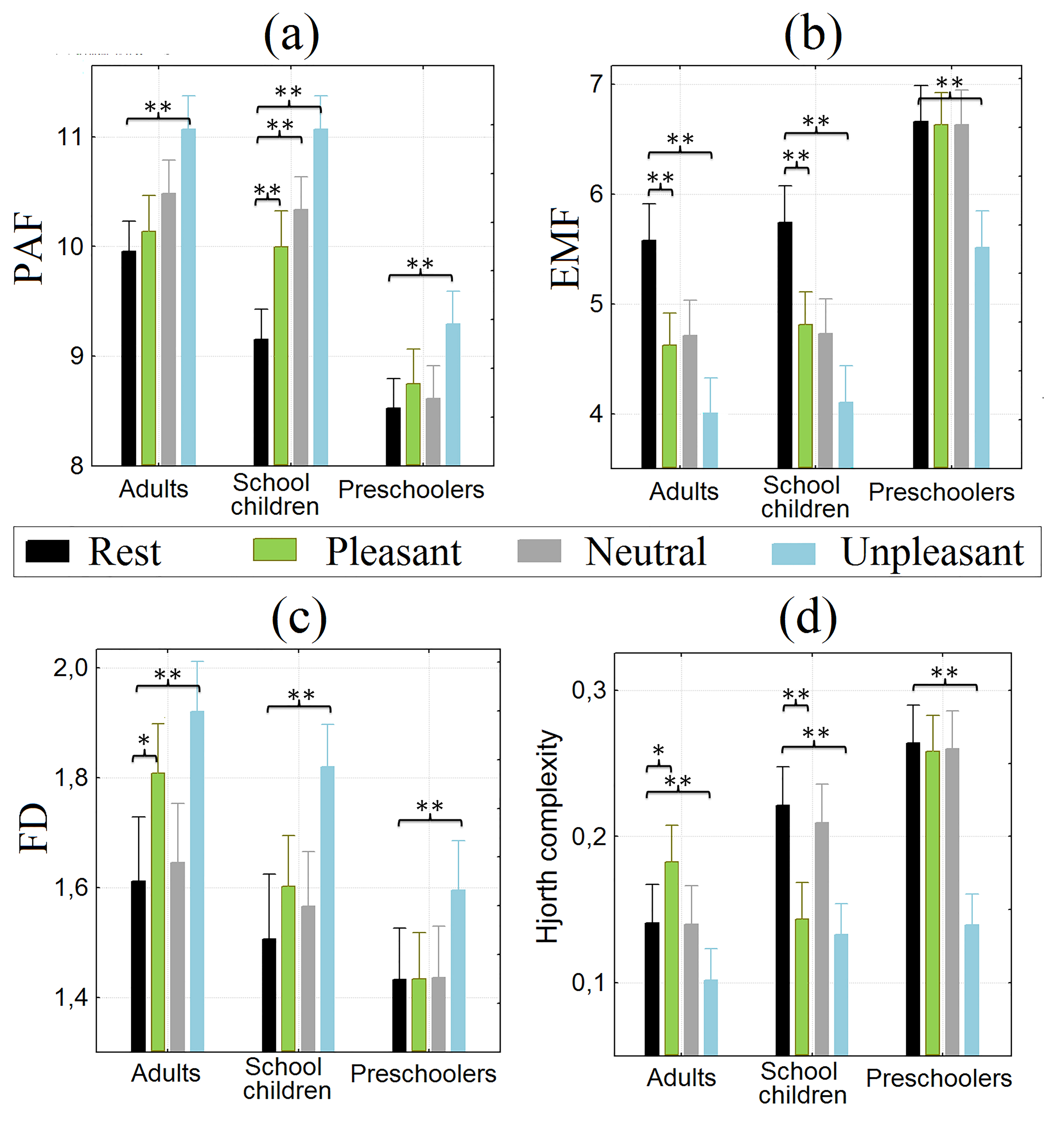 Fig. 4.
Fig. 4.
Peak alpha frequency and non-linear EEG features. (a) Peak alpha frequency (PAF) at rest and
during tactile stimulation. Mean and deviations (
In adults, as in schoolchildren, there was a decrease in EEG power in the
central and parietal regions during all types of tactile stimulation (F(6, 264) =
14.466, p
Healthy adult participants demonstrated the increase of the FD during pleasant and unpleasant tactile stimulation compared to the resting
state in the central and parietal regions (Fig. 4c). Similar (but less pronounced
for the pleasant stimuli) changes of the FD were observed in schoolchildren (F(6,
264) = 7.941, p
Adults showed an increase in the signal complexity index - EEG entropy (HC) for pleasant tactile stimulation and a decrease for unpleasant
stimuli in central and parietal regions (F(6, 264) = 6.620, p
Correlation analysis revealed a significant association between the decrease in PAF during pleasant stimulation compared to the resting state in the group of healthy adults (Spearman rank correlation r = –0.65, p = 0.0001) in central and frontal regions (Fig. 2d). No significant correlation between PAF dynamics and subjective pleasantness of stimuli was found in any of the pediatric groups.
We found specific EEG markers that correspond to age-related (developmental) changes in the perception of tactile stimuli with different valence. It is noteworthy that the response to unpleasant tactile stimulation in children of different ages and adults did not differ significantly. At the same time, healthy adults and schoolchildren showed similar EEG changes associated with tactile perception under neutral and unpleasant stimulation, whereas in preschool children only unpleasant stimuli caused EEG changes similar to those in adults. The earlier perceptual development of unpleasant stimuli perception is required for the child survival and safety. According to the previous studies made in other modalities and the infants of 2–3 months could distinguish facial expressions of fear and anger, however ability to recognize the positive or neutral emotions growth up the 4–6 months [38, 39, 40]. The ability to laugh and to recognize other’s laughter also develops later than for crying due to more difficult social function of laugher consisted in promoting social bonding and influencing social group dynamics [41, 42]. EEG response to unpleasant stimulus was accompanied by an increase in PAF and FD, and a decrease in EMF. These indices were sensitive to emotional or affective responses to stimuli. Previous studies have linked increases in FD and PAF to states of higher mental effort such as working memory, arousal level, and cognitive or emotional processing of stimuli [43, 44, 45, 46]. Thus, the observed changes in both children and adults indicate an effective response to ice stimulation, which was perceived as unpleasant and even painful, as manifested in both EEG changes and behavioral responses. Neutral stimuli requiring higher cognitive activity for recognition and evaluation elicited reduced responses in preschoolers, consistent with children’s difficulties in perceiving and analyzing tactile stimuli [47, 48].
Regarding the perception of pleasantness of stroking, our study showed that
adult healthy volunteers rated the pleasantness of stimuli more differentially.
Adults who reported greater pleasantness of CT-stroking also showed a decrease in
PAF, indicating relaxation. We have previously shown that
decreased PAF is associated with greater subjective pleasantness of touch and
increased salivary oxytocin levels following tactile stimulation. Conversely,
when participants were unable to relax due to fatigue, overexcitement,
experimental conditions, or hypersensitivity to tactile stimuli, an increase in
PAF was observed when presented with any type of stimuli. Divergent
shifts in alpha rhythm frequency depending on the type of emotional reaction are
well-known and described in various studies with healthy adults. For instance,
Kostyunina and Kulikov (1996) [49] found significant differences (p
At the same time the perception of pleasant and neutral tactile stimuli could be
also associated to not only to the social and individual factors, but also with
the experimental conditions. The slow stroking could be one of the mechanisms to
trigger CT-fibers [15]; however, as was showed in previous studies it’s not
necessary but not sufficient. The gentle forces [52] and a neutral (typical skin)
temperature of stroking are required to induce CT-afferents response [53]. In
spite we attempted to control the forces of stroking we couldn’t avoid mixing
CT-fibers and other coetaneous receptors (in particularly A
The ice stimulation due to the cold temperature let us to avoid the stimulation
of CT-receptors. Moreover, the response to unpleasant thermal or painful stimuli
could be opposed to pleasant stimuli due to the pain-reducing effect of
A
Finally, it is worth discussing the preschooler-specific decrease in the theta rhythm PSD observed during the perception of unpleasant tactile stimuli, in contrast to the observed increase in theta power in school-age children and adults. This difference in EEG dynamics during the presentation of unpleasant tactile stimuli is the only difference that distinguishes adults from children. Given that the decrease in theta power in response to emotionally meaningful stimuli is more characteristic of patients with impaired consciousness [22], we hypothesized that this EEG change is one of the indicators of developmental changes in EEG dynamics during tactile perception.
The obtained data for the developmental changes of perception of pleasant, unpleasant and neutral tactile stimuli, as well as their EEG correlates could play an important role diagnosing autism spectrum disorder (ASD). In particular, alterations of tactile perception were found individuals with ASD independently of ages and the severity of symptoms [57, 58]. The further research of tactile perception in children with ASD taking into account factors of age dynamics, pleasantness of stimuli and revealed in this study EEG markers will improve diagnostic methods for developmental disabilities.
This study has potential limitations including relatively small sample of participants and age restrictions. In particular, we did not examine the tactile perception of children younger than 4 years of age or older than 10 years of age. The presentation of an unpleasant tactile stimulus (ice) could potentially affect the perception of other tactile stimuli as well as the overall emotional state. To compensate for this limitation, we made relatively long pauses (30 seconds) between stimuli during the study. We had no ability to prepare neurographic testing of sensory nerve fibers and to control the type of skin receptors which were stimulated.
The perception of pleasant and neutral tactile stimuli in children and adults appeared to be more dependent on the child’s maturation. EEG dynamics related to tactile perception showed greater similarity between the responses of adult volunteers and school-aged children compared to preschoolers. Nonlinear EEG parameters such as FD, EMF and PSD dynamics of the theta rhythm were sensitive to changes in the development of tactile perception. Hjort complexity and PAF scores can be used as indices of maturity of perception of CT-stimuli.
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Conceptualization, Methodology, Formal Analysis, Investigation, Writing – Original Draft Preparation: GP; Data Curation, Writing – Review & Editing, Supervision: GP and LM. Both authors read and approved the final manuscript. Both authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was conducted in accordance with the principles of biomedical ethics formulated in the 1964 Helsinki Declaration and its subsequent updates and was approved by the Institutional Ethics Committee of the Pushkin State Russian Language Institute (protocol code 17-3-24-118, date of approval 15.07.22). All participants submitted voluntary written informed consent signed by him or her for the publication of any potentially identifiable images or data included in this article.
Electrophysiological studies were carried out using equipment of the Research Resource Center of IHNA and NPh RAS for functional brain mapping.
The study was supported by grant of the Russian Science Foundation, project № 22-15-00324, “Social tactile contacts and their role in psycho-emotional rehabilitation”.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




