1 Namdaemun Sehwa Korean Medical Clinic, 04529 Seoul, Republic of Korea
2 Departments of Veterinary Anatomy and Animal Behavior, College of Veterinary Medicine and BK21 FOUR Program, Chonnam National University, 61186 Gwangju, Republic of Korea
3 Korean Medicine Data Division, Korea Institute of Oriental Medicine, 34054 Daejeon, Republic of Korea
4 Korean Convergence Medical Science, University of Science and Technology, 34113 Daejeon, Republic of Korea
5 Faculty of Traditional Medicine, Hue University of Medicine and Pharmacy, Hue University, 49000 Thua Thien Hue, Vietnam
6 Herbal Medicine Resources Research Center, Korea Institute of Oriental Medicine, 58245 Naju-si, Jeollanam-do, Republic of Korea
7 Center for Companion Animal New Drug Development, Jeonbuk Branch, Korea Institute of Toxicology, 56212 Jeongeup, Jeollabuk-do, Republic of Korea
8 Department of Oriental Medicine, College of Oriental Medicine, Dongshin University, 58245 Naju-si, Jeollanam-do, Republic of Korea
†These authors contributed equally.
Abstract
Background: Rheum tanguticum root, cataloged as
“Daehwang” in the Korean Pharmacopeia, is rich in various
anthraquinones known for their anti-inflammatory and antioxidant properties.
Formulations containing Daehwang are traditionally employed for treating
neurological conditions. This study aimed to substantiate the antiepileptic and
neuroprotective efficacy of R. tanguticum root extract (RTE) against
trimethyltin (TMT)-induced epileptic seizures and hippocampal neurodegeneration.
Methods: The constituents of RTE were identified by
ultra-performance liquid chromatography (UPLC). Experimental animals were grouped
into the following five categories: control, TMT, and three TMT+RTE groups with
dosages of 10, 30, and 100 mg/kg. Seizure severity was assessed daily for
comparison between the groups. Brain tissue samples were examined to determine
the extent of neurodegeneration and neuroinflammation using histological and
molecular biology techniques. Network pharmacology analysis involved extracting
herbal targets for Daehwang and disease targets for epilepsy from
multiple databases. A protein-protein interaction network was built using the
Search Tool for the Retrieval of Interacting Genes/Proteins (STRING) database,
and pivotal targets were determined by topological analysis. Enrichment analysis
was performed using the Database for Annotation, Visualization, and Integrated
Discovery (DAVID) tool to elucidate the underlying mechanisms.
Results: The RTE formulation was found to contain sennoside A,
sennoside B, chrysophanol, emodin, physcion, (+)-catechin, and
quercetin-3-O-glucuronoid. RTE effectively inhibited TMT-induced seizures at 10,
30, and 100 mg/kg dosages and attenuated hippocampal neuronal decay and
neuroinflammation at 30 and 100 mg/kg dosages. Furthermore, RTE significantly
reduced mRNA levels of tumor necrosis factor (TNF-
Keywords
- neurodegenerative disorders
- inflammation
- epilepsy
- Rheum tanguticum
- network pharmacology
Rheum tanguticum Maxim. ex Balf. is a perennial plant belonging to the genus Rheum L. of the Polygonaceae family [1]. Its roots, known as Daehwang (or Da Huang in Chinese), have a long history of medicinal use in China, Korea, and Japan [2]. In Korea, Daehwang is clinically administered for a range of conditions, including constipation, abdominal pain, diarrhea, jaundice, nosebleeds, conjunctivitis, sore throat, appendicitis, bruises, amenorrhea, and burns [3]. Formulations such as Daeseunggitang (Da Cheng Qi Tang), Dohaekseunggitang (Taohe Chengqi Tang), Jeodangtang (Di Dang Tang), and Sasimtang (San Huang Xie Xin Tang) all incorporate Daehwang. Various neurological symptoms, including perceptions of seeing a ghost, experiences related to mental illness, and chest tightness, are included in the Chinese clinical guidelines, Shanghanlun [4]. Existing literature consists of retrospective case reports focusing on the aftermath of stroke, depression [5, 6], and schizophrenia [7]. Recently, preclinical research regarding the efficacy of R. tanguticum in addressing various neurological disorders has been conducted. Rhubarb anthraquinones, such as emodin, aloe-emodin, chrysophanol, and physcion, are known to protect the central nervous system [8]. Daehwang has also been found to exert anti-neuroinflammatory and neuroprotective effects by inhibiting microglial activation [9]. A study employing the Morris water maze indicated that treatments using R. tanguticum could attenuate cognitive dysfunction in a rat model of Alzheimer’s disease [10]. Emodin, one of the anthraquinones, improved kainic acid-induced epileptic rats through regulation of multidrug resistance gene 1 [11]. Therefore, R. tanguticum shows promise both as an antiepileptic and a neuroprotective agent in the treatment of epilepsy and neurodegenerative conditions.
Epilepsy is a neurological disorder characterized by recurrent seizures arising from various etiological factors and complex initiation mechanisms [12]. Factors such as congenital brain anomalies, abnormal neuronal migration, birth trauma, intracerebral hemorrhage, intracranial inflammation, febrile seizures, hypoxia, hypoglycemia, hypocalcemia, brain injury, intracranial tumors, and cerebrovascular diseases are attributed to the onset of epilepsy [13]. In Korea, the incidence of epilepsy ranges from 50 to 70 cases per 100,000 individuals annually, ranking it as the third most prevalent neurological condition after dementia and stroke. Within 5 years after an initial seizure, approximately 80% patients are likely to experience a subsequent seizure, evolving into chronic active epilepsy. Roughly 30% of patients with epilepsy exhibit drug resistance [14, 15]. Consequently, the mean disease duration spans 10 years, with 20–30% of cases manifesting as lifelong conditions. Considering these circumstances, there is a pressing need for innovative therapeutic approaches for seizure management. Following the emergence of the epilepsy-epileptogenesis paradigm, the focus has shifted toward understanding the mechanistic links between inflammation and epilepsy [16, 17, 18]. In this context, our research aims to investigate the efficacy of natural products in trimethyltin (TMT)-induced epilepsy models.
The organotin compound, TMT exhibits specific toxicity in the hippocampus, an essential part of the brain’s limbic system [19]. TMT exposure is associated with a range of neurological symptoms, such as hypoactivity evolving into hyperactivity, hyperexcitability, ataxia, tremors, seizures, convulsions, memory deficits, and learning impairments, collectively termed TMT syndrome [20, 21]. Consequently, TMT is used to simulate neurodegenerative conditions, such as epilepsy and dementia in animal models [22, 23], and the hippocampal damage closely resembles that caused by convulsant agents or observed in certain human epilepsy cases [24]. The neurotoxic effects of TMT are attributed to multiple mechanisms, including apoptotic cell death, calcium dyshomeostasis, oxidative stress, and neuroinflammation [25]. Much like kainic acid, commonly used to study temporal lobe epilepsy and status epilepticus, TMT induces neuronal loss in the CA3 and CA1 regions of the hippocampus [26]. This loss is thought to result from either glutamate-dependent excitotoxicity or calcium overload [23]. When glutamate receptor antagonists are co-administered with TMT, excitotoxicity is reduced, suggesting potential neuroprotective benefits [27]. Therefore, TMT is a valuable tool for generating animal models of neurodegeneration associated with cognitive decline and temporal lobe epilepsy. This is because the neurodegenerative effects of TMT share key pathogenic features common to a broad array of neurodegenerative disorders, such as selective neuronal death and neuroinflammation [23, 28].
The onset of symptoms in neurodegenerative diseases is frequently linked to neuroinflammation. Proinflammatory elements, including activated glial cells and microglia, have garnered significant interest as potential therapeutic targets for individuals with epilepsy [29]. An analysis of the dementia drug development pipeline in 2023 delineated the roles of candidate therapies aimed at amyloid, epigenetics, inflammation/immunity, metabolism/bioenergetics, neurogenesis, neurotransmitter receptors, and oxidative stress [30]. Therefore, this study sought to assess the regulatory efficacy of R. tanguticum in animal models of seizures and hippocampal neurodegeneration induced by TMT.
Identification of complex molecular pathways remains a substantial challenge in the context of herbal medications. Unlike single-compound agents, herbal medications usually comprise multiple components, complicating the task of elucidating specific action mechanisms [31]. Network pharmacology has recently emerged as a new approach for identifying compound-target pathways related to particular diseases, offering a systematic and holistic perspective. Despite advances in clarifying the actions of various herbs and pharmacological agents through network pharmacology’s “multi-target–multi-pathway” frameworks, the molecular mechanisms accounting for the effectiveness of R. tanguticum in epilepsy treatment remain undefined.
In this study, the therapeutic potential of R. tanguticum for epilepsy was evaluated by integrating experimental research with network pharmacology. Initially, a mouse model was employed to investigate the protective effects against TMT-induced hippocampal degeneration. Subsequently, a network pharmacological analysis was conducted to comprehensively evaluate the regulatory mechanisms involved in epilepsy treatment, focusing on potential active compounds and target genes (Fig. 1).
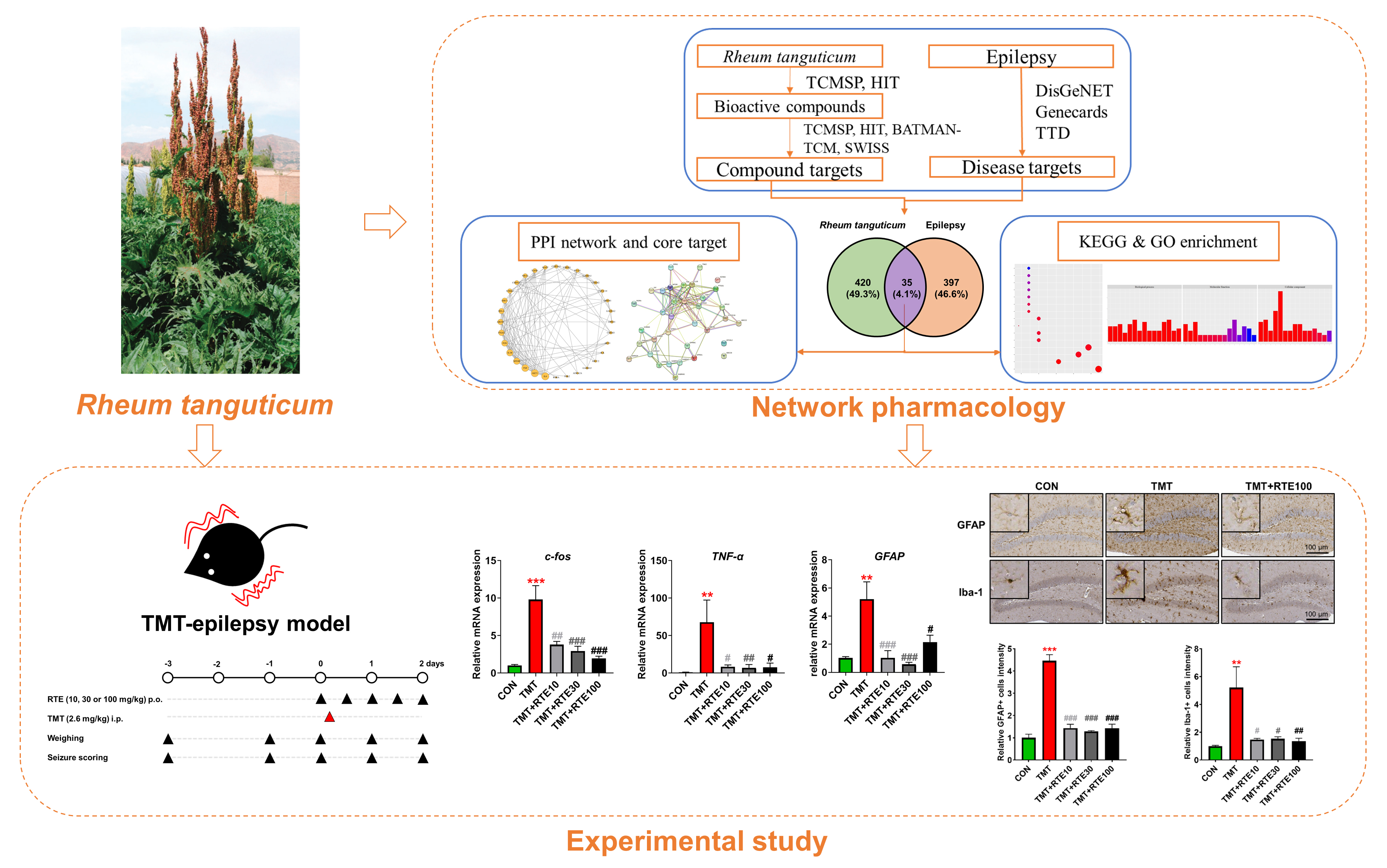 Fig. 1.
Fig. 1.Flowchart for investigating therapeutic potential of
Rheum tanguticum in epilepsy treatment. TCMSP, traditional chinese
medicine systems pharmacology database and analysis platform; HIT,
herbal-ingredient-target platform; BATMAN-TCM, bioinformatics analysis tool for
molecular mechanism of TCM; TTD, therapeutic target database; PPI,
protein-protein interaction; KEGG, Kyoto encyclopedia of genes and genomes; GO,
gene ontology; TMT, trimethyltin; RTE, Rheum tanguticum root extract;
TNF-
Decoction has been one of the most widely used methods for extracting herbal
ingredients in traditional Chinese medicine. We extracted R. tanguticum
using the decoction method as used in Geijigadaehwang-tang, whose
neuroprotective effects were previously confirmed [32]. R. tanguticum
was commercially acquired from Nanumherb Co., Ltd. (Gyeongbuk, Korea; product
number: HA1900240302; origin: Shaanxi, China; plant parts used: root and
rhizome). The R. tanguticum root extract (RTE) was prepared by
performing cooling reflux extraction for 2.5 h using 1.0 L distilled water as the
solvent at 100
For the ultra-performance liquid chromatography (UPLC; Acquity UPLC, Waters, MA,
USA) analysis, RTE (2 mg/mL) and standard compounds (0.2 mg/mL) were solubilized
in water. The standard compounds used were sennoside B, sennoside A,
chrysophanol, physcion (Ministry of Food and Drug Safety, Cheongju, Republic of Korea),
emodin, (+)-catechin (Sigma-Aldrich, St. Louis, MO, USA), and
quercetin-3-O-glucuronoid (ChemFace, Wuhan, Hubei, China). The mobile phase
consisted of 0.1% formic acid in water (A) and 0.1% formic acid in acetonitrile
(B). Chromatography began with an initial 5% proportion of solvent B, which was
then linearly increased to 50% over a span of 20 min. The samples were loaded on
an Acquity UPLC C18 column (1.7
Eight-week-old male C57BL/6 mice were sourced from Central Lab Animal Inc.
(Seoul, Republic of Korea) and given a week to acclimatize. Female mice demonstrate variable
social behavior dependent on their estrus cycle [33]; hence, their use in
neuroscience research necessitates documenting estrus stages and modifying test
protocols accordingly [34]. To preclude such variables from influencing the data,
only male mice were utilized in this study. The animals were housed under the
following controlled environmental conditions: 23
All experimental procedures were conducted in accordance with the protocols
approved by the Institutional Animal Care and Use Committee at Dongshin
University (approval no.: DSU2023-04-03). The mice were divided into the
following five groups: group 1, control; group 2, TMT; group 3, TMT+RTE (10
mg/kg); group 4, TMT+RTE (30 mg/kg); and group 5, TMT+RTE (100 mg/kg). The RTE
dosages were determined based on both the no-observed-adverse-effect-level
(NOAEL) of rhubarb for chronic toxicity [35] and the 10% Daehwang
content in Geijigadaehwang-tang [32], as previously determined in our study
regarding neuroprotective efficacy. On day 0, TMT (Wako, Osaka, Japan) was
administered intraperitoneally at a dosage of 2.6 mg/kg after diluting in saline.
RTE was initially administered orally 1 h before TMT injection and subsequently
twice daily, considering pharmacokinetics of the rhubarb anthraquinones [36].
Monitoring activities, including RTE administration, seizure assessment, and
weight measurement, were performed daily between 09:30 and 10:30. RTE doses were
administered at 09:30 and 15:30. On day 2, when neurotoxicity and clinical
symptoms were most manifested, the mice were euthanized [25, 37] by intravenously
injecting a combination of alfaxalone (Rompun
Seizure behavior was assessed using a bright box (dimensions: 40
The procedure for quantitative reverse transcription-polymerase chain reaction
(qRT-PCR) analysis is extensively documented in the literature [39]. Hippocampal
tissue was processed to extract total RNA using the Hybrid-R Kit (GeneAll
Biotechnology, Seoul, Republic of Korea). The NanoDrop ND-2000 system (Thermo Fisher
Scientific, Waltham, MA, USA) was used to assess the concentration of RNA.
Reverse transcription of 1 µg RNA was performed using the
PrimeScript RT master mix according to the manufacturer’s guidelines (Takara,
Tokyo, Japan). qRT-PCR was conducted in triplicate using the CFX96TM Real-Time
system (Bio-Rad, Hercules, CA, USA) and SYBR Green qPCR master mix (MBiotech,
Hanam, Korea). The
The immunohistochemistry technique has been previously described in detail [39]. Sections of deparaffinized tissue were treated with primary antibodies, namely rabbit anti-doublecortin (DCX; Cell Signaling Technology, Beverly, MA, USA), GFAP (Cell Signaling Technology), and ionized calcium-binding adaptor molecule 1 (Iba-1; Wako Pure Chemical Industries, Ltd., Osaka, Japan). After washing with phosphate-buffered saline (PBS), the sections were incubated using biotinylated anti-rabbit IgG (Vector Laboratories, Newark, CA, USA). The Vector ABC Elite kit (Vector Laboratories) was used for detection of specific binding with a 3,3-diaminobenzidine tetrahydrochloride solution. The Motic Easyscan Digital Slide Scanner (Motic, Hong Kong, China) was used to analyze the stained sections. The ImageJ software was used for quantification of staining intensity in the hippocampus (NIH, Bethesda, MD, USA).
Compounds associated with R. tanguticum were combined from UPLC analysis and
databases, including Traditional Chinese Medicine Systems Pharmacology Database
and Analysis Platform (TCMSP, https://tcmsp-e.com/tcmsp.php), version 2.3 [40]
and Herbal-Ingredient-Target Platform (HIT, http://hit2.badd-cao.net/), version
2.0 [41] using “Da huang” or “Dahuang” as search terms. The retrieved
compounds’ synonym names, Chemical Abstracts Service (CAS) numbers, PubChem
compound IDs (CID), and International Chemical Identifier (InChIKey) numbers were
then entered in the PubChem database (https://pubchem.ncbi.nlm.nih.gov/) to
determine the chemical structures and ensure the compounds’ compatibility for
subsequent analysis [42]. Redundant compounds across databases and those lacking
PubChem database information were excluded from further analysis. Subsequently,
the compounds’ oral bioavailability (OB) and drug-likeness (DL) parameters were
acquired from the TCMSP to assess their pharmacokinetic profiles. Compounds
demonstrating an OB value
The active compounds of R. tanguticum and their corresponding target genes were retrieved from multiple databases, namely TCMSP version 2.3 [40], HIT version 2.0 [41], Bioinformatics Analysis Tool for Molecular mechANism of TCM (BATMAN-TCM, http://bionet.ncpsb.org/batman-tcm) [44], and SWISSTargetPrediction (http://www.swisstargetprediction.ch/) [45]. Compound names, CIDs, and CAS numbers were entered in the HIT 2.0 system of the TCMSP (https://tcmsp-e.com/tcmsp.php) and HIT (http://hit2.badd-cao.net/) databases to extract target gene information. For BATMAN-TCM, the CIDs of compounds were submitted by selecting predicted potential genes, including known associates, with a score_cutoff set at 20. The SWISS Target Prediction required a probability filter threshold of 0.1 to determine relevant genes. The collected target genes were then cross-referenced for accuracy regarding gene IDs and nomenclature using the UniProt database (https://www.uniprot.org/), focusing on the species “Homo sapiens”.
Subsequent steps involved identifying epilepsy-associated disease targets using DisGeNET version 7.0 (https://www.disgenet.org/) [46], Genecards version 5.9 (https://www.genecards.org/) [47], and the Therapeutic Target Database (TTD, https://db.idrblab.net/ttd/) [48]. Criteria for selection included all disease targets from TTD and those with a gene-disease association score exceeding 0.1 in DisGeNET as well as targets with a relevance score surpassing 10 in Genecards. Common targets between R. tanguticum compounds and epilepsy were earmarked for further analysis.
To examine the protein interactions among the overlapping targets, a protein-protein interaction (PPI) network was constructed. This was facilitated by the Search Tool for the Retrieval of Interacting Genes/Proteins (STRING, https://string-db.org/) database, with the species set to “H. sapiens” and a medium confidence score threshold of 0.4 [49]. The network topology was subsequently analyzed using Cytoscape 3.9.0 (U.S. National Institute of General Medical Sciences, Bethesda, MD, USA), where centrality of the nodes was quantified by the degrees, indicative of the number of linkages per node [50].
To elucidate the biological processes and molecular pathways implicated in the treatment of epilepsy by R. tanguticum, enrichment analysis involving Gene Ontology (GO, https://geneontology.org/) and Kyoto Encyclopedia of Genes and Genomes (KEGG, https://www.genome.jp/kegg/) pathways was conducted using the Database for Annotation, Visualization, and Integrated Discovery (DAVID, https://david.ncifcrf.gov/). In this analysis, an adjusted cut-off p-value of 0.01 was applied after Benjamini’s correction [51]. The GO analysis addressed the molecular functions, cellular components, and biological processes associated with the targets, while the KEGG pathway analysis explored the targets’ roles in diverse pathways and processes. A comprehensive “herb-compound-target-pathway” network was also constructed using Cytoscape 3.9.0 to delineate the action mechanisms of RT [50].
The results are presented as the mean
The chemical constituents of RTE were characterized using UPLC under optimized chromatographic conditions. The analysis revealed the presence of (+) catechin, sennoside B, quercetin-3-o-glucuronoid, sennoside A, physcion, emodin, and chrysophanol, which were consistent with standard references when subjected to identical analytical conditions (Fig. 2). In addition, the quantitative analysis results were summarized in Table 1.
 Fig. 2.
Fig. 2.Composition analysis of RTE. (A) UPLC chromatogram of RTE (2.0 mg/mL) at 280 nm. (B) UPLC chromatogram of standard compounds (0.2 mg/mL) at 280 nm. (1) (+) catechin (tR 3.20 min); (2) sennoside B (tR 4.64 min); (3) quercetin-3-o-glucuronoid (tR 4.81 min); (4) sennoside A (tR 5.14 min); (5) physcion (tR 9.49 min); (6) emodin (tR 11.29 min); and (7) chrysophanol (tR 12.82 min). RTE, Rheum tanguticum root extract; UPLC, ultra-performance liquid chromatography.
| Calibration equation | r |
Content* (mg/g, %) | |
| (+) catechin | y = 19366x – 1839.1 | 1.00 | 25.51 |
| sennoside B | y = 20111x – 5010.0 | 1.00 | 7.17 |
| quercetin-3-o-glucuronoid | y = 20027x – 9564.7 | 0.99 | 10.76 |
| sennoside A | y = 24097x – 3675.5 | 0.99 | 5.07 |
| physcion | y = 57438x – 1720.2 | 0.99 | 1.32 |
| emodin | - | - | trace |
| chrysophanol | - | - | trace |
*, Content results are shown as mean
Fig. 3A illustrates the experimental schedule, as detailed in section 2. We
evaluated the protective effects of RTE against seizures induced by TMT toxicity
using groups of 8–9 mice. RTE was administered orally to the mice 1 h before TMT
administration and subsequently, twice daily. After TMT administration, the mice
demonstrated aggressive behavior, reduced body weight, convulsions, and seizure
activity from the first day. A marked reduction in body weight was observed in
the TMT group on the first 2 days (day 0: 21.4
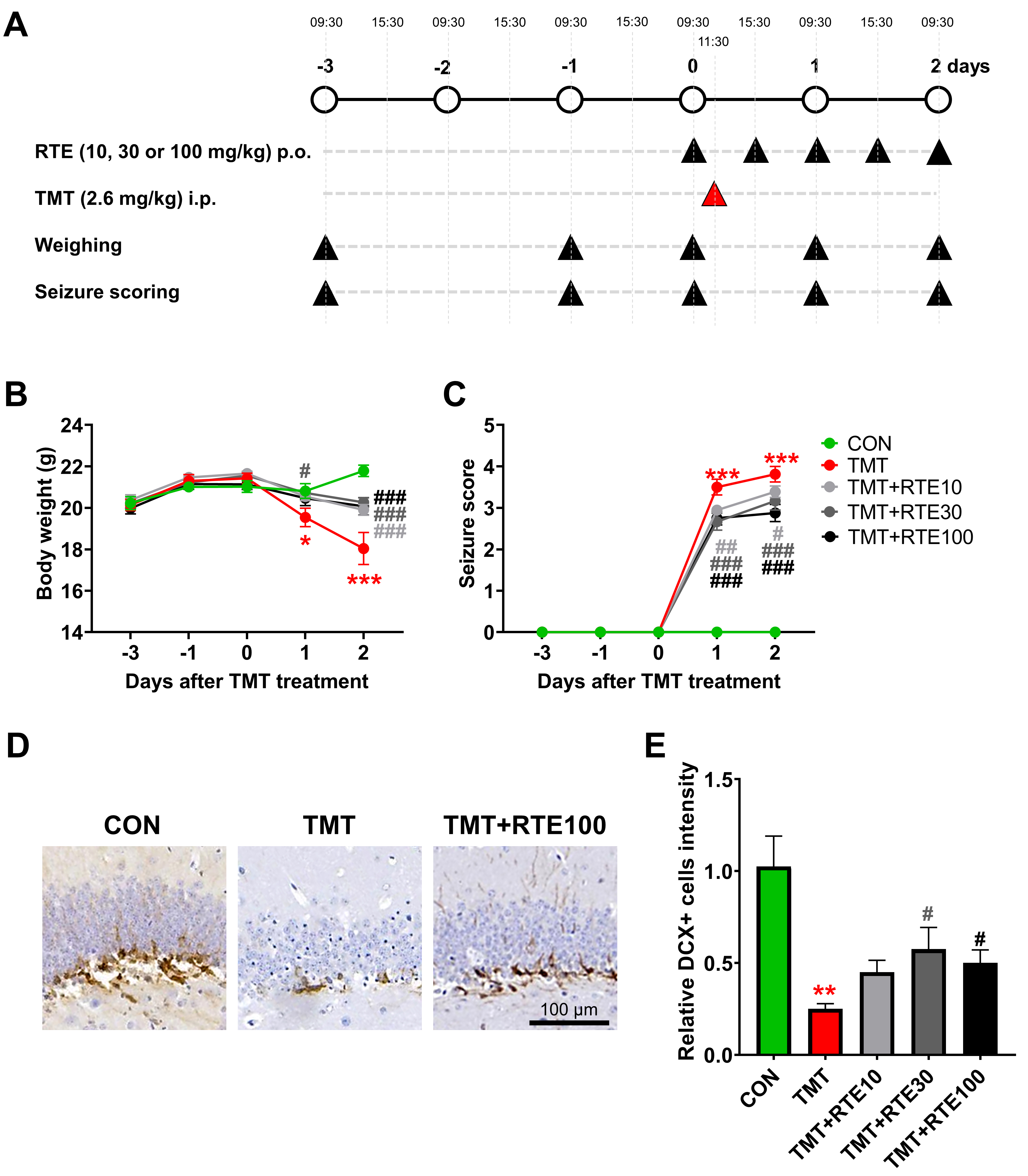 Fig. 3.
Fig. 3.Anticonvulsive effects of RTE on TMT-induced seizures.
(A) Schematic diagram of the experiment demonstrating neurodegeneration induced
by TMT. (B) Clinical seizure behavior scoring. (C) Body weight scoring. (D,E) DCX
immunohistochemistry results (mean
The severity of seizures was found to have intensified on the second day
relative to that of those on the first day, which conforms to the results of
previous studies [25, 37]. Initially, the TMT group exhibited a seizure severity
score of 3.5
A significant relationship exists between the severity of seizures and neuronal
death [52]. DCX-positive immature progenitor neurons in the dentate gyrus (DG)
substantially decreased 2 days after TMT administration—from 1.00
Fig. 4 illustrates the marked elevation in mRNA expressions of c-fos
(9.8
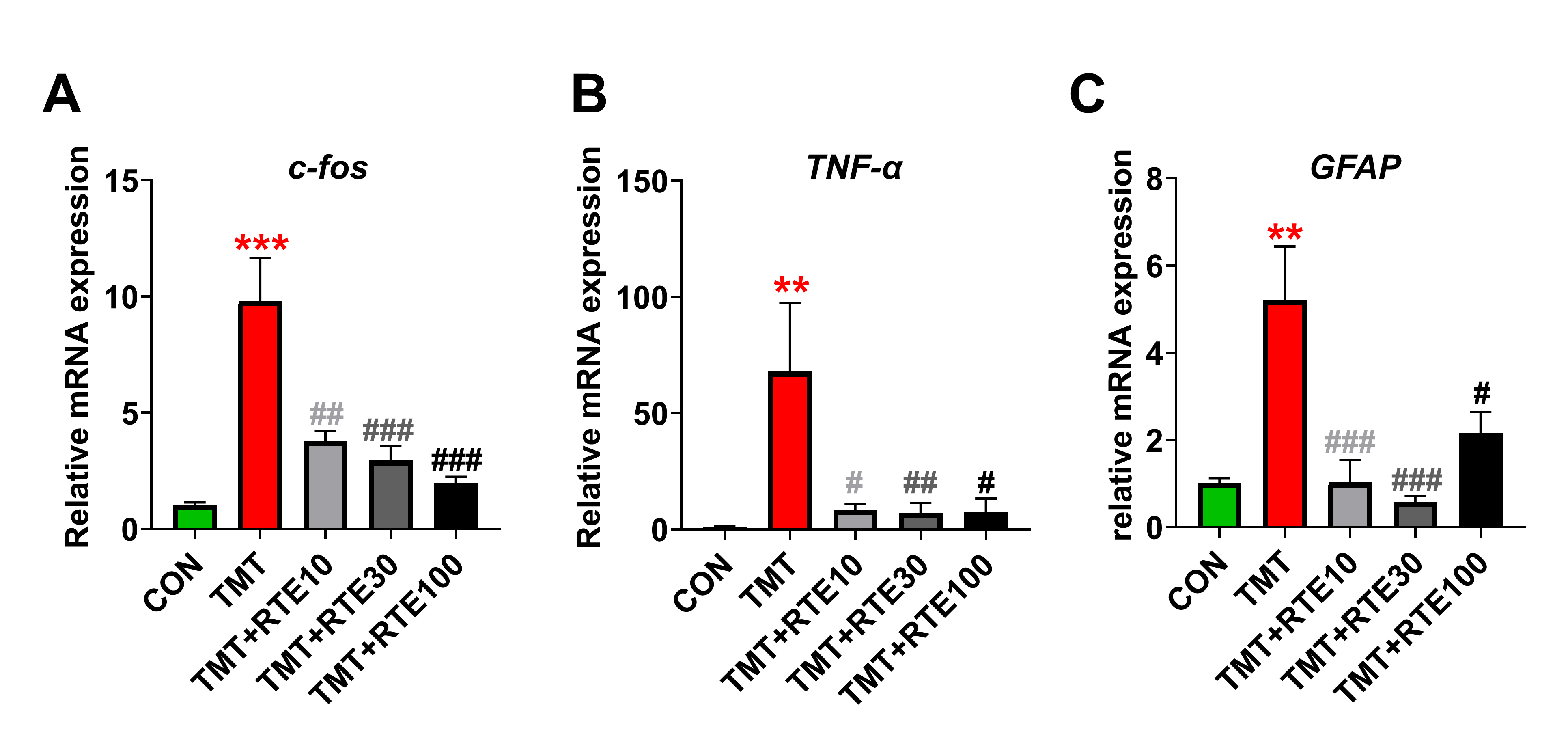 Fig. 4.
Fig. 4.Inhibition of (A) c-fos, (B)
TNF-
Representative photomicrographs presented in Fig. 5A show GFAP-positive
astrocytes and Iba-1-positive microglia in the DG of the hippocampus in the
control, TMT, and TMT+RTE mice. In the hippocampus, cells positive for both GFAP
and Iba-1 exhibited a satellite-like morphology characteristic of mature
astrocytes and microglia. The immunoreactivities of GFAP and Iba-1 were
noticeably more pronounced in TMT-treated mice (GFAP: 4.46
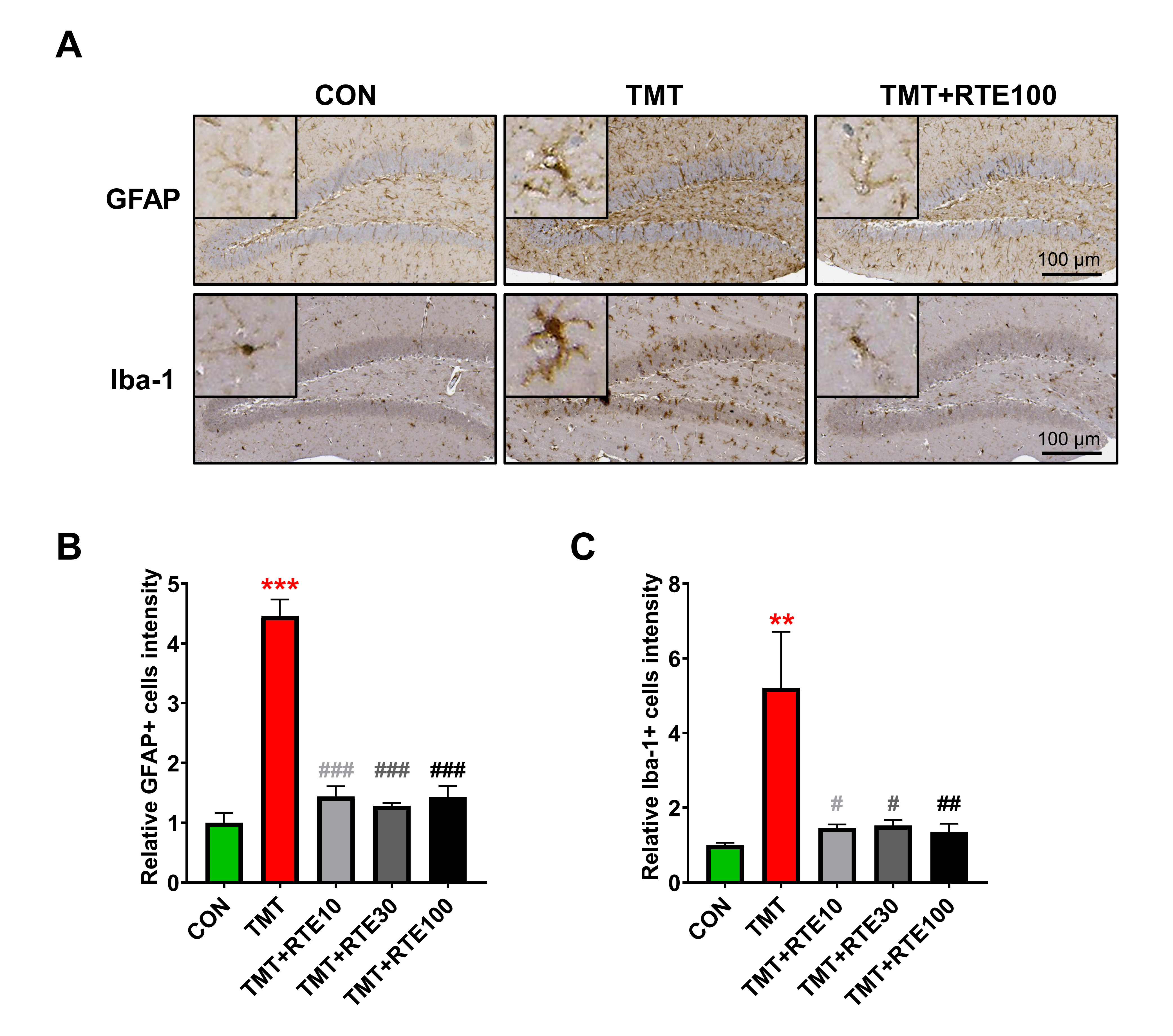 Fig. 5.
Fig. 5.Histopathological changes in GFAP and Iba-1 after RTE
administration on TMT-induced glial activation (A–C) GFAP and Iba-1
immunohistochemistry results expressed as mean
Within R. tanguticum, a total of 6 compounds were identified
through UPLC analysis, while 92 were cataloged in the TCMSP database, and 31 in
the HIT database. Compounds were considered for absorption, distribution,
metabolism and excretion (ADME) profile analysis based on an OB value
| No. | Molecular ID | Molecule Name | OB (%) | DL | PubChem ID |
| 1 | MOL002235 | Eupatin | 50.8 | 0.41 | 5317287 |
| 2 | MOL002251 | Mutatochrome | 48.64 | 0.61 | 5281246 |
| 3 | MOL002259 | Physciondiglucoside | 41.65 | 0.63 | 442762 |
| 4 | MOL002260 | Procyanidin B-5,3 |
31.99 | 0.32 | 162845209 |
| 5 | MOL002268 | Rhein | 47.07 | 0.28 | 10168 |
| 6 | MOL002276 | Sennoside E_qt | 50.69 | 0.61 | 162899601 |
| 7 | MOL002280 | Torachrysone-8-O-beta-D-(6 |
43.02 | 0.74 | 163001298 |
| 8 | MOL002281 | Toralactone | 46.46 | 0.24 | 5321980 |
| 9 | MOL002288 | Emodin-1-O-beta-D-glucopyranoside | 44.81 | 0.8 | 11968447 |
| 10 | MOL002293 | Sennoside D_qt | 61.06 | 0.61 | 135397905 |
| 11 | MOL002297 | Daucosterol_qt | 35.89 | 0.7 | 5742590 |
| 12 | MOL002303 | Palmidin A | 32.45 | 0.65 | 5320384 |
| 13 | MOL000358 | Beta-sitosterol | 36.91 | 0.75 | 222284 |
| 14 | MOL000471 | Aloe-emodin | 83.38 | 0.24 | 10207 |
| 15 | MOL000554 | Gallic acid-3-O-(6 |
30.25 | 0.67 | 162995045 |
| 16 | MOL000096 | (-)-Catechin | 49.68 | 0.24 | 73160 |
| 17 | C1125 | Baicalin | 40.12 | 0.75 | 64982 |
| 18 | C1114 | Baicalein | 33.52 | 0.21 | 5281605 |
| 19 | C1241 | Cianidanol | 54.83 | 0.24 | 9064 |
OB, oral bioavailability; DL, drug-likeness.
The integration of data from the TCMSP, HIT, and Swiss target databases led to
the prediction of 455 target proteins associated with the 19 active compounds in
R. tanguticum. From epilepsy-related databases, 432 specific targets
were extracted. Cross-referencing these datasets revealed 35 common targets
between R. tanguticum and epilepsy, indicating their potential as
targets for R. tanguticum’s antiepileptic effects (Fig. 6A, Table 3).
These targets were ranked by their predicted significance as follows: IL-6, AKT1,
TNF, MTOR, IL-1
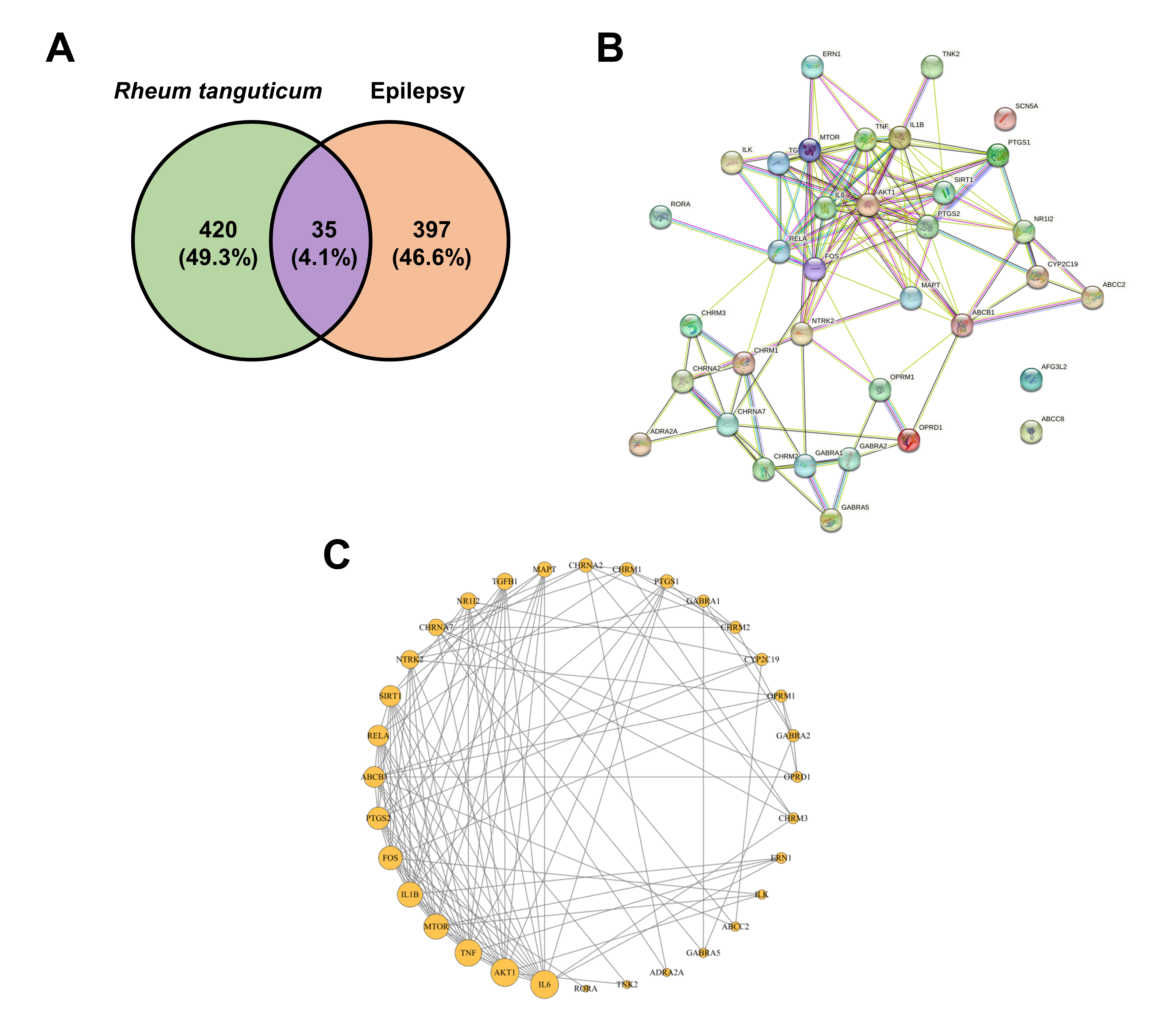 Fig. 6.
Fig. 6.Target analysis of Rheum tanguticum for epilepsy treatment from various databases. (A) Intersection of 35 targets between R. tanguticum and epilepsy. (B) Protein-protein interaction network intensity of the 35 overlapping targets. (C) Topology analysis of the protein-protein interaction network. Targets are represented by circular nodes, and larger nodes denote higher significance.
| No. | Symbol | Uniprot ID | Description |
| 1 | IL-6 | P05231 | Interleukin-6 |
| 2 | AKT1 | P31749 | RAC-alpha serine/threonine-protein kinase |
| 3 | TNF | P01375 | Tumor necrosis factor |
| 4 | MTOR | P42345 | Mammalian target of rapamycin |
| 5 | IL-1 |
P01584 | Interleukin-1 beta |
| 6 | FOS | P01100 | Protein c-fos |
| 7 | PTGS2 | P35354 | Prostaglandin G/H synthase 2 |
| 8 | ABCB1 | P08183 | ATP-dependent translocase ABCB1 |
| 9 | RELA | Q04206 | Transcription factor p65 |
| 10 | SIRT1 | Q96EB6 | NAD-dependent protein deacetylase sirtuin-1 |
The 35 shared targets were subjected to KEGG pathway and GO term enrichment
analysis using the DAVID database. A total of 34 pathways with significant
enrichment having an adjusted p-value
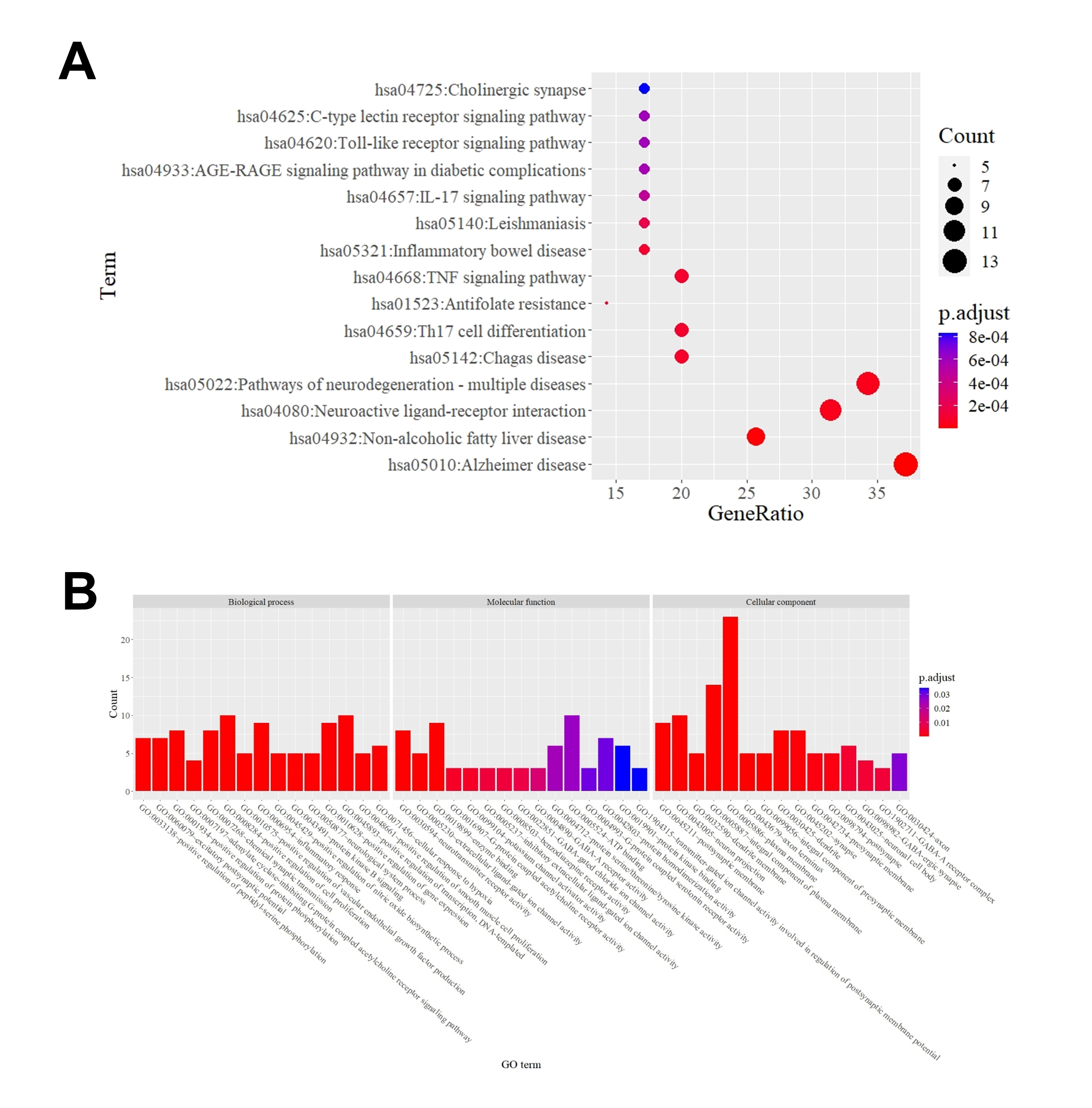 Fig. 7.
Fig. 7.Enrichment analysis of Rheum tanguticum for epilepsy treatment using (A) KEGG and (B) GO pathways. GO, Gene Ontology; KEGG, Kyoto Encyclopedia of Genes and Genomes.
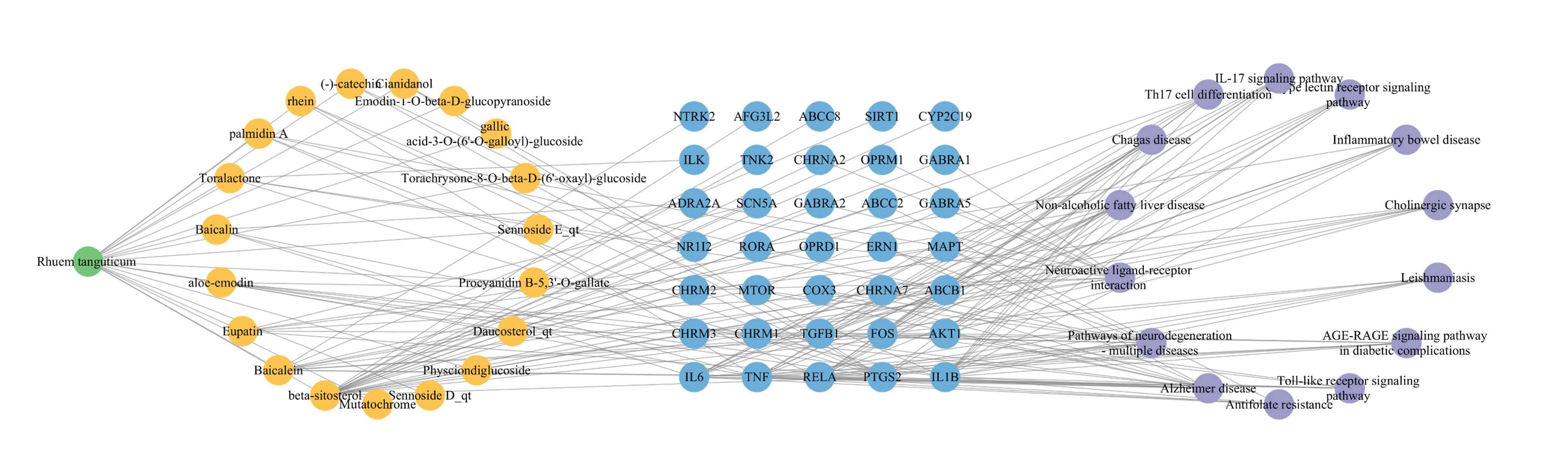 Fig. 8.
Fig. 8.Network herb-compound-target-pathway of Rheum tanguticum in treating epilepsy. The herb is represented by the green node, compounds by orange nodes, targets by blue nodes, and pathways by purple nodes.
The primary objective of pharmacotherapy in the management of epilepsy is to achieve complete remission of epileptic seizures. Medications targeting the reduction of neuronal excitation or the augmentation of inhibitory processes through various mechanisms, such as gamma-aminobutyric acid (GABA)-receptor enhancement, inhibition of glutamate excitation, or the modulation of voltage-gated sodium and calcium channels, are widely utilized [54]. Nevertheless, studies indicate that over 30% persons with epilepsy exhibit resistance to antiepileptic drugs [55]. Furthermore, the prolonged use of these medications is often associated with somatic, neurological, psychiatric, and neonatal adverse effects, posing significant challenges to epilepsy pharmacotherapy [56]. This situation underscores the urgent need for research regarding natural substances that possess antiepileptic properties through novel mechanisms.
Our study demonstrates that RTE mitigates the severity of TMT-induced epileptic seizures and associated weight loss in a dose-dependent manner (Fig. 3B,C). R. tanguticum, which is one of the source plants for Daehwang, a recognized medicinal in China, Korea, and Japan, was utilized in this investigation. Hepatotoxicity of Daehwang has been previously reported [57, 58], so clinical use requires toxicity control, such as compliance with appropriate dosage and use of hot water extraction method. The recommended dosages for Daehwang are documented in the pharmacopeias of these nations, and it is available commercially as a raw component for herbal preparations. The neuroprotective effects of R. tanguticum as a part of the formulation, Geijigadaehwang-tang has been studied previously [32], and our findings confirm its antiepileptic and neuroprotective potential. However, variations in the therapeutic effects may arise depending on the geographical location of the source and processing of the plants used as Daehwang, which warrants further research. Moreover, the chronic toxicity assessments of rhubarb extract have indicated a NOAEL of 94 mg/kg/day, reflecting a range of biological safety in several contexts [35]. Our findings showed no significant change in body weight for dosages from 10 to 100 mg/kg/day, with RTE notably inhibiting the weight loss associated with TMT-induced neurotoxicity (Fig. 3B).
Therefore, we conducted a component analysis to verify the quality standards and
identify the constituents of R. tanguticum. Sennoside A, sennoside B,
chrysophanol, emodin, physcion, (+)-catechin, and quercetin-3-O-glucuronide were
identified as components in RTE by UPLC (Fig. 2). These findings conform to the
results of the component analyses of rhubarb and Daehwang performed in
previous studies [57, 58]. Various research efforts have underscored the
neuroprotective properties of these constituents. For example, sennoside
derivatives have shown potent antioxidative effects against oxidative stress
induced by hydrogen peroxide, light, and radiation, mitigating the production of
reactive oxygen species production and neutralizing free radicals [59, 60].
Chrysophanol has been noted for its efficacy in reducing inflammation in focal
ischemic brain injuries and decreasing proinflammatory cytokines, including
TNF-
Additional active compounds, including (-)-catechin, sennoside D, sennoside E, and aloe-emodin, which are isomers of those verified by the UPLC technique were identified by the in silico PPI analysis (Table 2). The compound, rhein is primarily known for its neuroprotective effects, which are attributed to its antioxidant and anti-inflammatory properties [67, 68]. Similarly, baicalein has been shown to be effective in a range of epilepsy models, including temporal lobe and posttraumatic epilepsy ones [69, 70]. Owing to the lack of available standards, only a subset of the RTE components were identified by UPLC. However, compounds similar to rhein and baicalein, though not directly identified, may be present in RTE. In silico methods help estimate the other RTE constituents that may contribute to seizure inhibition and neuroprotection. Therefore, despite discrepancies between computer simulations and component analysis, both strands of evidence corroborate the therapeutic potential of R. tanguticum root in treating epilepsy.
TMT is known to be associated with neurodegeneration in the central nervous system, especially in the hippocampus. The pathogenic mechanisms of TMT involve several processes, including oxidative stress, neuroinflammation, neuronal death, and regeneration [25]. While the precise mechanisms remain unclear, glutamate-mediated excitotoxicity and calcium dysregulation by TMT play important roles in the pathophysiology of epilepsy [71]. In our study, mice exposed to TMT exhibited aggressive behavior and seizures, which resemble symptoms of degenerative brain disorders [28]. When RTE was administered to these mice, an amelioration of these clinical symptoms was observed on the first and second days after treatment (Fig. 3B,C).
DCX is found in proliferating neural progenitors and neuronal precursors during adult neurogenesis and serves as a reliable marker for neurogenesis in adult brains [72]. Adult mouse hippocampi typically exhibit high DCX levels. However, a previous study showed that DCX expression markedly reduced after a 2–4-day exposure to TMT, with notable recovery observed on the 10th day after exposure [73]. We found that adult neurogenesis significantly reduced 2 days after TMT treatment, although RTE administration prevented this neuronal degeneration (Fig. 3D,E). Additionally, neuronal death has been identified as a critical factor in epileptogenesis [52]. Our study suggests that RTE treatment mitigates TMT-induced neuronal degeneration and epilepsy-like behavior.
C-fos is recognized as a marker of neural activity and is associated with cellular development, differentiation, transformation, and death [74, 75]. After a seizure, c-fos activation is noticeable in the brain [76]. Previous studies have shown increased c-fos expression following TMT administration, which is correlated with the intensity of seizure behaviors that was highest 2 days after administration [38]. In our study, RTE lowered the mRNA expression of c-fos, thus mitigating seizure behaviors (Fig. 4A). Additionally, within the PPI network, both TNF (degree = 15) and Protein c-fos (FOS, degree = 13) were identified as central proteins in the context of epilepsy treatment (Fig. 6).
TNF-
The morphological analysis of the PPI network extended beyond c-fos,
TNF-
The implications of this study for clinical practice are indirect and preliminary, offering foundational insights for translational research in treating neurodegenerative conditions. While we did not investigate the effect of varied concentrations of Daehwang extract for extended treatment durations, our findings lay the groundwork for subsequent experimental investigations involving these compounds.
In summary, this study identified the components of RTE and demonstrated its in vivo anti-toxic activity against TMT damage. Concurrently, RTE was observed to attenuate the clinical symptoms of seizures, reduce hippocampal neurogenesis, and increase glial activation. Complementary to these findings, network pharmacology approaches revealed that 19 active compounds in RTE, interacting with 35 distinct targets, are integral to its antiepileptic efficacy. These compounds appear to exert their beneficial effects by modulating pathways associated with neurodegeneration and TNF signaling. Collectively, our findings suggest that RTE may possess therapeutic promise for conditions such as epilepsy and neurodegenerative diseases by counteracting inflammatory and oxidative pathways.
Data generated or analyzed during this study are included in this published article.
JC, SL, JSK, and SIL conceived and designed the experiment. SK, MNT, SMR, DHK, YEL, SWC, and SJ performed the experiments. JC, SMR, CM, SL, and JSK analyzed and interpreted the data. JC, SMR, JSK, CM, and SIL contributed reagents, materials, analysis tools or data. JC, SK, JSK, MNT, and SIL wrote the paper. SWC, JSK, and SIL wrote the review and editing. SWC acquired grant funding. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
All experimental procedures were conducted in accordance with the protocols approved by the Institutional Animal Care and Use Committee at Dongshin University (Approval No.: DSU2023-04-03).
Not applicable.
This work was supported by a National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (No. 2021R1A2C1009604).
The authors declare no conflict of interest. Changjong Moon is serving as one of the Editorial Board members/Guest editors of this journal. We declare that Changjong Moon had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Jesús Pastor and Gernot Riedel.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.








