1 Department of Neurology, Soonchunhyang University Seoul Hospital, Soonchunhyang University School of Medicine, 04401 Seoul, Republic of Korea
Abstract
Background: The purpose of this study was to determine the detailed
characteristics of dizziness in patients with de novo Parkinson’s
disease (PD) and the clinical implications of dizziness. Methods:
Ninety-three people with de novo PD were enrolled between July 2017 and
August 2022 for this retrospective study. Using each representative scale,
various motor and non-motor symptoms were assessed. In addition, clinical
manifestations of dizziness in those patients, including its presence, type,
frequency, and duration of occurrence, were investigated. Results:
Thirty-nine patients with de novo PD reported dizziness, with presyncope
being the most common (38%). The most common frequency was several times a week
(51%). The most common duration was a few seconds (67%). Multivariable logistic
regression analysis showed that dizziness was more common in women than in men
{odds ratio (OR): 3.3601, 95% confidence interval (CI): 1.0820–10.4351,
p = 0.0361}. Dizziness was significantly related to non-motor symptoms
of low global cognition (OR: 0.8372, 95% CI: 0.7285–0.9622, p =
0.0123) and severe autonomic dysfunction (OR: 1.1112, 95% CI: 1.0297–1.1991,
p = 0.0067). A post-hoc analysis revealed that dizziness was only
associated with cardiovascular dysautonomia (adjusted OR: 10.2377, 95% CI:
3.3053–31.7098, p
Keywords
- autonomic dysfunction
- cardiovascular dysautonomia
- cognitive impairment
- dizziness
- sex
- Parkinson's disease
Parkinson’s disease (PD) is a representative neurodegenerative disorder with motor symptoms that include tremor, bradykinesia, rigidity, and/or gait disturbance [1]. Dopaminergic degeneration in the substantia nigra is known to be a key pathologic burden associated with such motor symptoms in patients with PD [2]. In addition, non-motor symptoms (NMSs) are strongly suspected to be related to non-dopaminergic degenerations of the noradrenergic, serotonergic, and cholinergic systems as well as dopaminergic degeneration beyond the substantia nigra [3]. Not only motor symptoms, but also NMSs can significantly impact the quality of life of patients with PD. Therefore, studying these motor and non-motor symptoms is necessary to deepen our understanding of the pathophysiology of PD and to better treat patients with PD.
It has been widely reported that some important NMSs, which include sleep disorders, loss of smell, depression, and constipation, could be present before development of the motor symptoms of PD [4]. Such NMSs not only provide pathophysiological evidence of disease progression, but also help clinicians diagnose PD easier or earlier in real clinical settings. Besides, clinicians have occasionally seen patients with PD complaining of dizziness at very early stages or even at a de novo state [5, 6]. A previous study reported that dizziness could precede diagnosis by about five years, and that it could be regarded as a potential NMS in the prodromal stage of PD [7]. Interestingly, another report recently documented that dizziness in patients with PD might not be associated with orthostatic hypotension [5], inferring that dizziness is not directly linked to cardiovascular dysfunction or orthostatic hypotension. However, the clinical differences between patients with de novo PD who complain of dizziness, and those who do not, have yet to be well understood. Therefore, the aim of this study was to investigate the clinical characteristics and the relevance of dizziness in drug naïve patients with de novo PD, focusing on the very early stages while excluding the adverse effects of dopaminergic medications, which might induce dizziness or orthostatic hypotension.
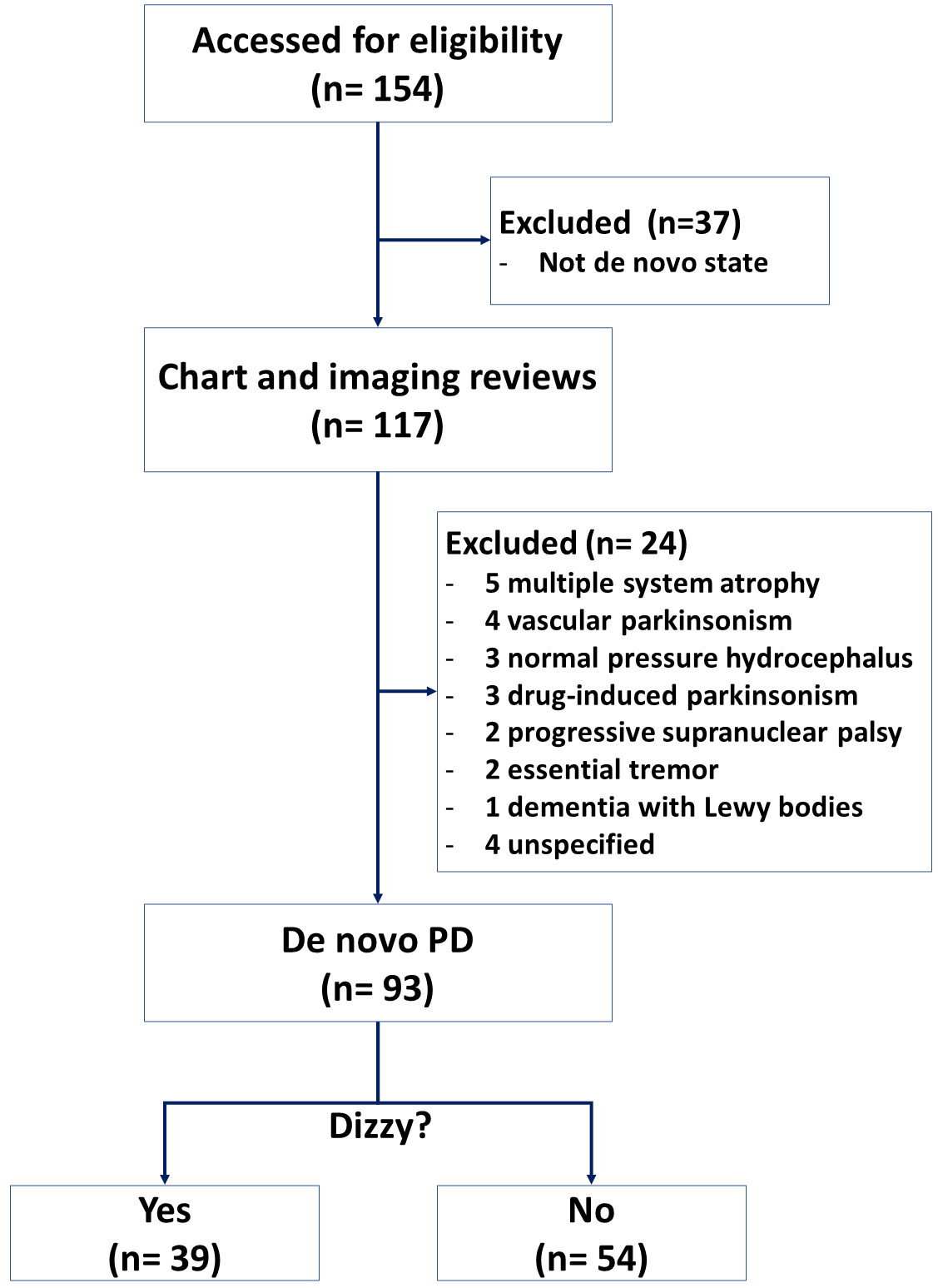
In our movement disorders clinic, any patients with parkinsonism have been evaluated through clinical assessments and neurological evaluations including both brain magnetic resonance imaging (MRI) and dopamine transporter imaging (DAT) since 2017. This retrospective study enrolled patients with de novo PD clinically diagnosed not only by the UK Brain Bank criteria [8], but also by the typical pattern of striatal dopaminergic depletion on DAT scanning [9, 10]. Among them, those who were suspicious of atypical parkinsonism or secondary parkinsonism were excluded from this study. Of a total of 154 patients with parkinsonism, only 93 patients were finally included between July 2017 and August 2022 for this study. The remainder had neither a de novo PD state at the time of registration nor a clinical diagnosis of PD during the follow-up (Fig. 1). This retrospective study was approved by the ethics committee of our hospital with a waiver of informed consent (No. 2022-12-003).
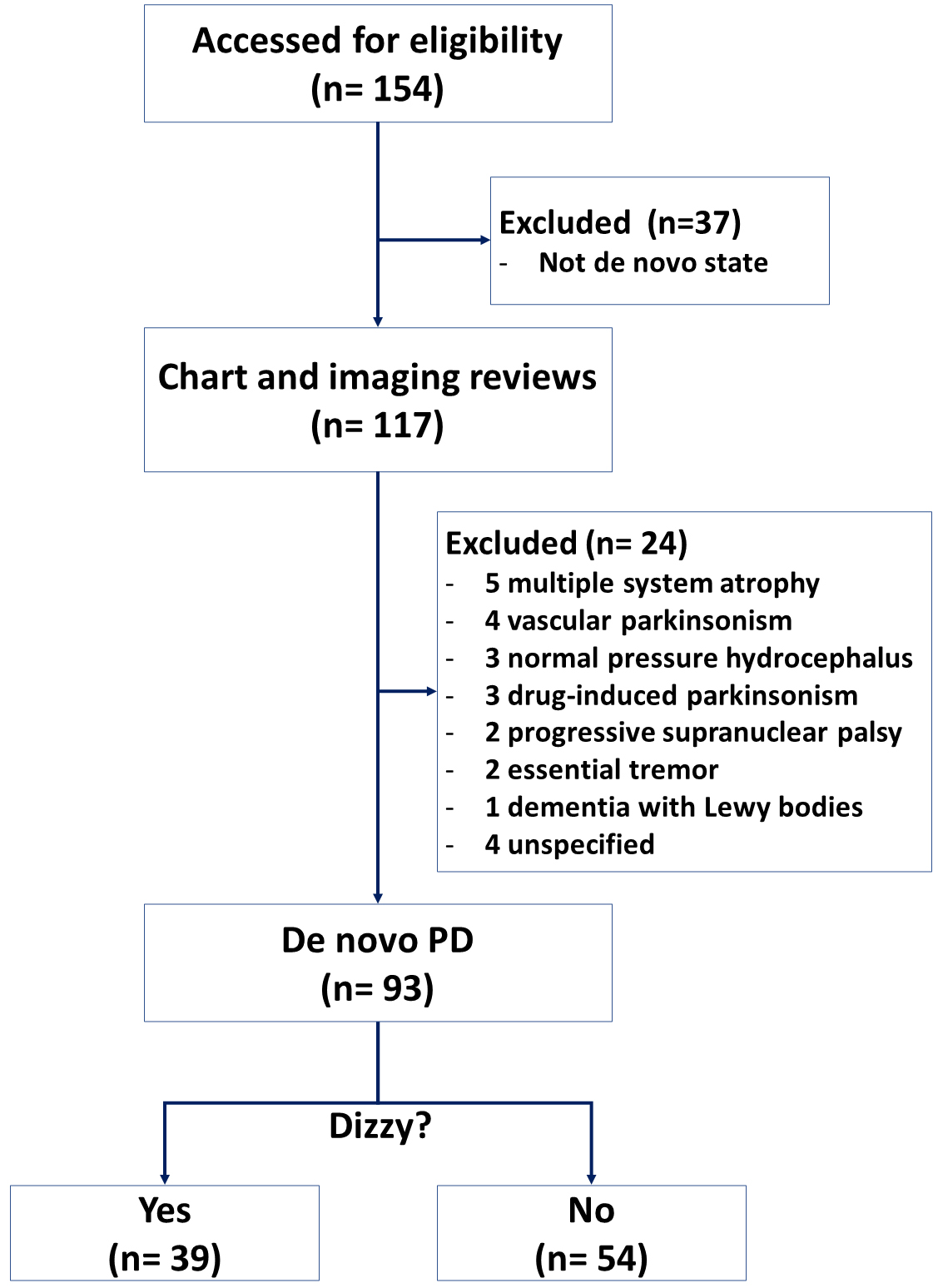 Fig. 1.
Fig. 1.Flow chart of the study. PD, Parkinson’s disease.
Motor symptoms were measured using both the Unified Parkinson’s Disease Rating Scale (UPDRS) part III and the Hoehn and Yahr (HY) stage. Tremor, rigidity, bradykinesia, and postural instability and gait difficulty (PIGD) subscores were also calculated. For statistical analysis, motor subtypes were derived and classified into two groups, a tremor-dominant type and a non-tremor dominant type (intermediate and PIGD), as previously described [8, 11]. Global cognitive function was assessed using the Korean version of Montreal Cognitive Assessment (MoCA-K) [12]. Anxious mood and depressive mood were studied with the Korean version of the Beck Anxiety Inventory (BAI) [13] and the Korean version of the Beck Depression Inventory (BDI) [14], respectively. Fatigue symptom was evaluated using the Parkinson’s Disease Fatigue Scale (PFS) [15]. Various autonomic symptoms were assessed with the Korean version of the Scale for Outcomes in Parkinson’s Disease-Autonomic (SCOPA-AUT) [16]. In addition, the clinical characteristics of dizziness including frequency, duration, and type were surveyed as previously described [6, 17]. For each patient with parkinsonism, a routine question about dizziness was asked, which was, “Do you experience dizziness?” If the patient answered “yes”, the detailed characteristics of the dizziness, including frequency, duration, and nature, were further evaluated. The information collected on dizziness was categorized into different groups based on the following criteria: The frequency of dizziness was classified as occurring more than once a day (several times a day), more than once a week (several times a week), or more than once a month (several times a month). The duration of dizziness was classified into seconds (from a few seconds to tens of seconds), minutes (from a few minutes to tens of minutes), or hours (from a few hours to an entire day). The clinical features of dizziness were also classified into the following categories: (i) vertigo (whirling or spinning); (ii) presyncope (orthostatic dizziness or impending faint); (iii) disequilibrium (loss of balance or unsteadiness when walking); or (iv) non-specific type (lightheadedness or unclassified). During the initial evaluation of parkinsonism, clinical assessments for dizziness, SCOPA-AUT, and cognitive function were also conducted.
The distribution of data for dizziness characteristics is presented by
percentage. Student t-test for continuous variables and chi-squared test
for categorical variables were used to compare the data of two groups. To predict
dizziness in patients PD, univariable and subsequent multivariable logistic
regression analyses were performed. In the multivariable logistic regression
analysis, variables with a p-value of less than 0.1 from the univariable
logistic analysis were included, using the stepwise selection method. A value of
p
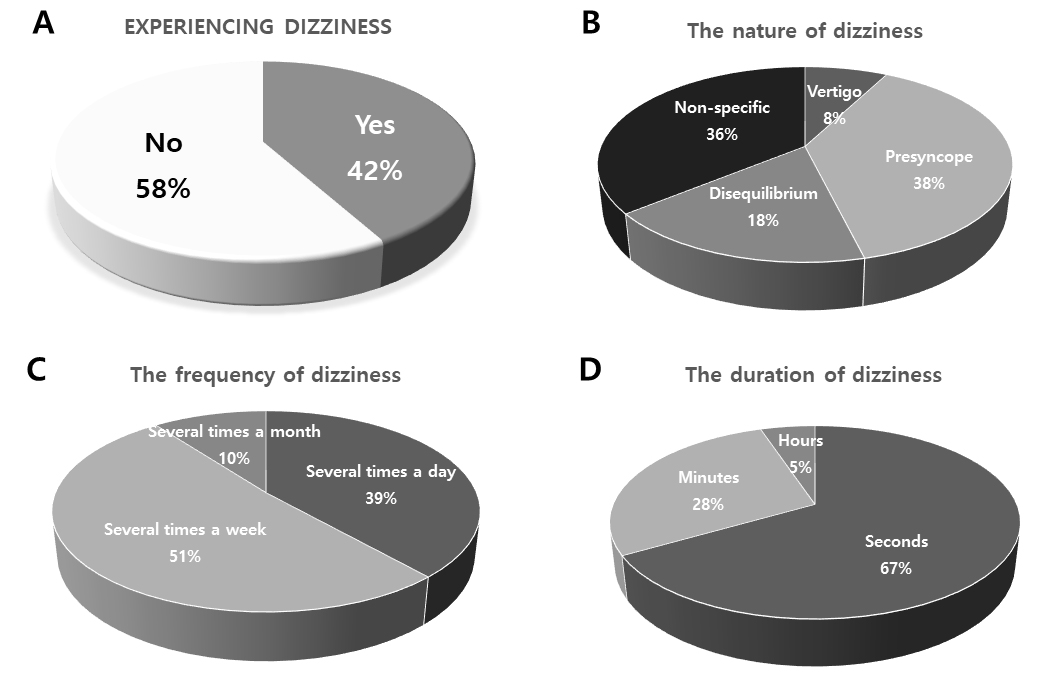
About 42% (39 out of 93) of patients with a de novo state of PD reported a feeling of dizziness in our cohort (Fig. 2A). In addition, the clinical characteristics of such dizziness were illustrated according to the nature, frequency, and duration of dizziness (Fig. 2B–D). The most common type of dizziness was presyncope (38%), followed by non-specific type (36%), disequilibrium (18%), and vertigo (8%). The most common frequency of dizziness attack was several times a week (51%), followed by several times a day (39%) and several times a month (10%). The most common duration of dizziness was a few seconds (67%), followed by a few minutes (28%) and a few hours (5%).
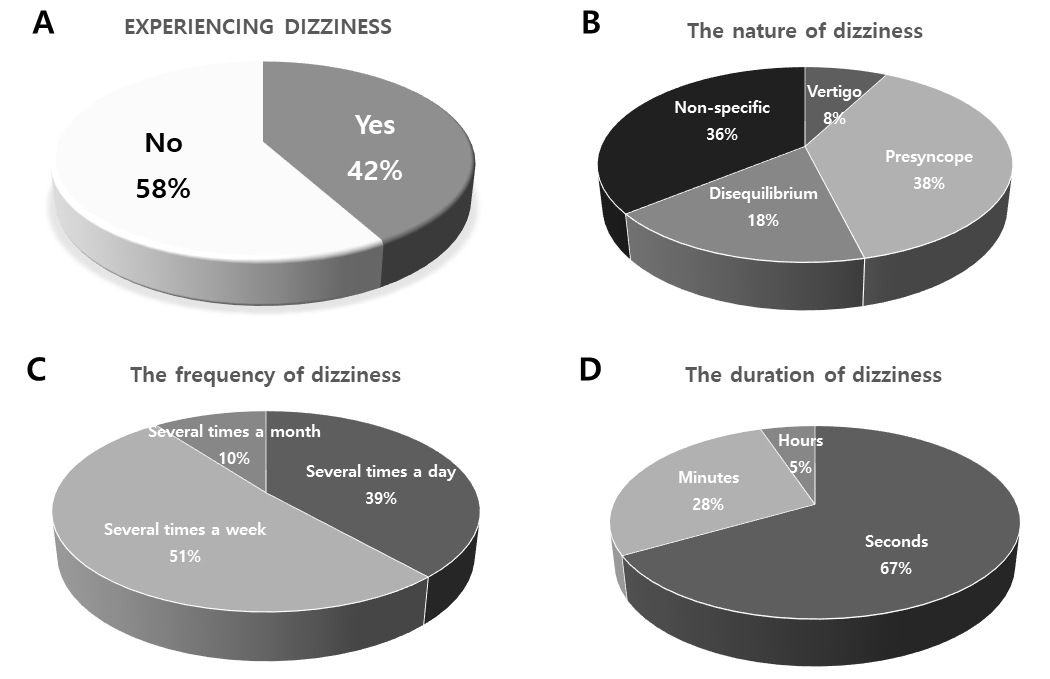 Fig. 2.
Fig. 2.Characteristics of dizziness in patients with de novo Parkinson’s disease. (A) Experience of dizziness. (B) Nature of dizziness. (C) Frequency of dizziness. (D) Duration of dizziness.
Table 1 summarizes the demographics and clinical features of de novo
Parkinsonian patients with or without dizziness. Of note, dizziness was reported
more in female patients than in male patients (p = 0.0177). However,
dizziness showed no significant difference according to other baseline
demographics or comorbidities. There were no significant differences in motor
symptoms including their subscores and phenotypes between those with dizziness
and those without dizziness. However, compared to those without dizziness, those
with dizziness showed more severe non-motor symptoms of depression (p =
0.0317), anxiety (p = 0.0014), and fatigue (p = 0.0099). In
addition, gastrointestinal (p = 0.0393) and cardiovascular (p
| Variable | Dizziness in de novo PD | p-value | ||
| Yes (n = 39) | No (n = 54) | |||
| Age, yr | 69.8 |
71.7 |
0.3239 | |
| Sex -Female | 27 (69.2%) | 24 (44.4%) | 0.0177 | |
| BMI (kg/m |
23.5 |
23.6 |
0.8577 | |
| Level of education, yr | 8.8 |
10.0 |
0.2731 | |
| Disease duration, yr | 1.4 |
1.3 |
0.4147 | |
| Follow-up duration, yr | 1.7 |
1.9 |
0.4758 | |
| UPDRS-III score (motor) | 25.5 |
23.7 |
0.938 | |
| Tremor score | 3.1 |
2.9 |
0.8275 | |
| Rigidity score | 5.0 |
5.0 |
0.9245 | |
| Bradykinesia score | 10.9 |
8.6 |
0.0756 | |
| PIGD score | 3.5 |
2.7 |
0.1668 | |
| Hoehn and Yahr stage | 2 (1−3) | 2 (1–3) | 0.6249 | |
| Motor subtype | ||||
| TD group | 13 (33.3%) | 27 (50.0%) | 0.1103 | |
| Non-TD group | 26 (66.7%) | 27 (50.0%) | ||
| MoCA-K (global cognition) | 21.6 |
23.7 |
0.0513 | |
| BDI (depression) | 11.3 |
8.0 |
0.0317 | |
| BAI (anxiety) | 9.2 |
4.7 |
0.0014 | |
| PFS (fatigue) | 48.0 |
39.2 |
0.0099 | |
| SCOPA-AUT (total score) | 14.5 |
11.3 |
0.0573 | |
| Gastrointestinal (GI) domain | 3 (0–16) | 2 (0–9) | 0.0393 | |
| Urinary (UR) domain | 7.3 |
7.1 |
0.8541 | |
| Cardiovascular (CV) domain | 1 (0–6) | 0 (0–2) | ||
| Thermoregulatory (TR) domain | 1 (0–3) | 0 (0–6) | 0.1585 | |
| Pupillomotor (PM) domain | 0 (0–6) | 0 (0–6) | 0.8426 | |
| Orthostatic hypotension |
7/35 (20%) | 7/44 (15.9%) | 0.6432 | |
| Common comorbidity | ||||
| Diabetes mellitus, yes | 12 (30.77%) | 13 (24.07%) | 0.4788 | |
| Hypertension, yes | 17 (43.59%) | 27 (50.00%) | 0.5443 | |
| Coronary disease, yes | 5 (12.82%) | 7 (12.96%) | 0.9842 | |
Data are shown as n (%) or mean
Orthostatic blood pressure
To evaluate risk factors for dizziness in de novo PD, univariable and subsequent multivariable logistic regression analyses were conducted for various clinical characteristics (Table 2). The odds ratio (OR) and 95% confidential interval (95% CI) for variables were also obtained. We found that dizziness in those with de novo PD was significantly associated with female (OR: 3.3601, 95% CI: 1.0820–10.4351, p = 0.0361), lower score of MoCA (OR: 0.8372, 95% CI: 0.7285–0.9622, p = 0.0123), and higher score of total SCOPA-AUT (OR: 1.1112, 95% CI: 1.0297–1.1991, p = 0.0067).
| Variable | Univariable | Multivariable | ||||
| OR | 95% CI | p-value | OR | 95% CI | p-value | |
| Age, yr | 0.9937 | 0.9473–1.0423 | 0.7955 | |||
| Sex-Female | 2.8750 | 1.1152–7.4120 | 0.0288 | 3.3601 | 1.0820–10.4351 | 0.0361 |
| BMI (kg/m |
0.9643 | 0.8227–1.1304 | 0.6542 | |||
| Level of education | 0.9272 | 0.8487–1.0129 | 0.9272 | |||
| Disease duration, yr | 1.4250 | 0.8709–2.3317 | 0.1586 | |||
| UPDRS-III score | 1.0135 | 0.9719–1.0570 | 0.5302 | |||
| Hoehn and Yahr stage | 1.9641 | 0.6713–5.7468 | 0.2178 | |||
| Motor subtype | 2.2857 | 0.9009–5.7992 | 0.0818 | – | – | – |
| MoCA-K | 0.8727 | 0.7859–0.9690 | 0.0108 | 0.8372 | 0.7285–0.9622 | 0.0123 |
| BDI | 1.0665 | 1.0004–1.1370 | 0.0487 | – | – | – |
| BAI | 1.0944 | 1.0161–1.1788 | 0.0173 | – | – | – |
| PFS | 1.0325 | 1.0036–1.0622 | 0.0272 | – | – | – |
| SCOPA-AUT | 1.0864 | 1.0201–1.1570 | 0.0099 | 1.1112 | 1.0297–1.1991 | 0.0067 |
| Orthostatic hypotension | 0.7778 | 0.2443–2.4766 | 0.6706 | |||
| Diabetes mellitus | 0.6612 | 0.2420–1.8061 | 0.4197 | |||
| Hypertension | 1.7250 | 0.8487–1.0129 | 0.2369 | |||
| Coronary disease | 1.0197 | 0.2520–4.1258 | 0.9781 | |||
Significant p values are in bold. OR, odds ratio; CI, confidence interval.
As a post-hoc analysis, logistic regression analysis for subdomains of the
SCOPA-AUT for the relationship of dizziness was performed after adjusting for
age, sex, disease duration, MoCA-K, the UPDRS part III score, and the HY stage
(Table 3). It was found that dizziness in patients with de novo PD was
significantly associated with a higher score of cardiovascular dysautonomia (adj
OR: 10.2377, 95% CI: 3.3053–31.7098, p
| Variable | Univariable | Multivariable | ||||
| OR | 95% CI | p-value | OR* | 95% CI | p-value | |
| Gastrointestinal (GI) domain | 1.0131 | 0.8332–1.2318 | 0.8962 | |||
| Urinary (UR) domain | 1.0093 | 0.8602–1.1843 | 0.9093 | |||
| Cardiovascular (CV) domain | 9.5472 | 3.0125–30.2565 | 0.0001 | 10.2377 | 3.3053–31.7098 | |
| Thermoregulatory (TR) domain | 1.3490 | 0.7664–2.3745 | 0.2995 | |||
| Pupillomotor (PM) domain | 0.8734 | 0.9387–1.1984 | 0.5983 | |||
* Adjusted for age, sex, disease duration, MoCA-K, the UPDRS part III score, and the HY stage. Significant p values are in bold. HY, Hoehn and Yahr.
If a patient with PD is experiencing dizziness, a comprehensive evaluation to determine the underlying cause is crucial. Treatment will depend on the specific cause identified. In our daily clinical practice, we occasionally encounter patients, even those with de novo PD, who complain of dizziness, which appears to be non-specific. Based on these clinical experiences, we hypothesized that dizziness could be one of the non-motor symptoms in PD, like constipation or depression. Specifically, we aimed to clarify the frequency and clinical presentation of dizziness in patients with PD in the de novo state.
Up to 30% of the elderly occasionally feel dizzy in their daily lives, while up to about 46~68% of patients with PD feel dizzy [6, 18]. However, the prevalence of dizziness should be interpreted with caution since many previous studies were conducted on patients with a med-on state [6, 18, 19], implying that a significant number of parkinsonian patients might show drug-induced adverse effects including dizziness, not a symptom of the disease itself. Our study investigated the presence of dizziness and its characteristics only in the drug naïve state of patients with de novo PD. As a result, we found that about 42% of patients with de novo PD had dizziness. By excluding adverse effects of taking anti-parkinsonian drugs, we could uncover the clinical impact of dizziness on other motor or non-motor symptoms in patients with PD itself, especially during very early stages of the disease.
Dizziness is a general term that could be distinguished into four categories according to its presumed etiologies [17]. In the current study, we showed that the types of dizziness in patients with de novo PD were diverse. The most common type of dizziness in patients with de novo PD was presyncope or orthostatic dizziness (38%). All other types of dizziness accounted for 62% (36% with lightheadedness or non-specific type, 18% with disequilibrium or feeling unbalanced, and 8% with vertiginous type). Of note, we figured out that dizziness was not associated with orthostatic hypotension (Table 1). In line with our results, Park et al. [19] also showed that about half of PD patient with dizziness did not have an orthostatic hypotension. Moreover, Klanbut et al. [20] demonstrated that orthostatic hypotension in some patients with PD was asymptomatic. On the other hand, Park and Kang recently showed that vestibular dysfunction might be involved in the pathogenesis of dizziness in PD [5]. Collectively, we could suspect that dizziness itself is not directly linked to orthostatic dizziness in patients with PD. It seems that the underlying mechanism of dizziness in PD can be multidimensional, rather than due to a single reason. Accordingly, the precise mechanisms for the relationship between dizziness and PD remain to be further elucidated.
Intriguingly, our study revealed that dizziness was significantly linked to women with de novo PD than to men with this disorder (Tables 1,2). It is currently unclear if women complain more of dizziness because they are women since it is generally accepted that dizziness tends to increase with age, and is reported higher in women [21, 22]. Besides, there is another possibility of there being other reasons for why women with PD complain of more dizziness. Some sex differences in motor symptoms and non-motor symptoms including depression and pain have been investigated in subjects with PD [23, 24]. The mechanism of sex-specific differences in PD is known to be associated with various players including hormonal, neuroendocrinal, genetic, and biochemical/molecular factors [25]. However, to the best of our knowledge, such a sex difference for dizziness in PD has yet to be evaluated. Further studies are required to address this issue.
Notably, we found that the presence of dizziness in patients with de novo PD was significantly related to not only lower scores of MoCA-K, but also higher scores of SCOPA-AUT (specifically, cardiovascular dysautonomia). Recent evidence has demonstrated that dizziness in older adults might be an important risk factor of cognitive impairment or dementia [26]. Moreover, in line with our findings, Lee and colleagues [27] have reported that dizziness in the elderly with parkinsonism including PD, normal pressure hydrocephalus, and multiple system atrophy is significantly associated with both cognitive impairment and postural instability. Accordingly, they supposed that postural instability might be a crucial factor of dizziness [27]. However, our results did not reveal any association between dizziness and postural instability probably because our study population was limited to those with a de novo state of PD, implying that most participants had little problem of postural instability. Postural instability significantly contributes to the disease progression of PD. Therefore, it is reasonable to infer that another important determinant of dizziness in patients with de novo PD may exist beyond postural instability. Further, a recent review has illustrated a possible linkage between cognitive dysfunctions and cardiovascular dysautonomia [28]. Moreover, another review has suggested that orthostatic hypotension-induced cerebral hypoperfusion might cause cognitive decline in patients with PD [29]. Collectively, we could deduce that dizziness in patients with de novo PD might be regarded as an indicative symptom for cardiovascular dysautonomia as well as cognitive impairment.
The SCOPA-AUT questionnaire consists of 25 items that assess various aspects of autonomic dysfunction, including cardiovascular function, gastrointestinal function, and urinary function. The cardiovascular domain of the SCOPA-AUT questionnaire comprises three items related to orthostatic dizziness [16]. However, dizziness itself was not identical to orthostatic dizziness, since dizziness in patients with PD could be presented as being of various types [6, 17], which have already been described. Moreover, the investigation of the characteristics of subjective dizziness and the SCOPA-AUT scale was separately conducted in the current study. Our study primarily focused on examining the clinical correlation between subjective complaints of dizziness and motor/non-motor symptoms in patients with de novo PD. Further follow-up studies are needed to investigate the association between cardiovascular dysfunction or orthostatic dizziness and clinical features of patients with de novo PD.
This study has several shortcomings. First, this was a retrospective study with a relatively small sample size. Novertheless, we could draw meaningful conclusions through the multivariable logistic regression analyses. However, caution should be exercised in extrapolating the findings of this study, and further larger-scale prospective studies are necessary. Second, it is important to note that the SCOPA-AUT questionnaire is an indirect method of assessing autonomic dysfunction, although the SCOPA-AUT questionnaire is a validated and reliable tool for evaluating autonomic dysfunction in patients with PD. Therefore, additional tests and evaluations may be necessary for a complete assessment of the relationship between dizziness and autonomic dysfunction in patients with de novo PD. Third, as this study is a retrospective study, we only investigated the medical history and could not perform a thorough evaluation of all medications. Therefore, we could not investigate the association between various medications, including alpha-blockers, diuretics, and calcium channel blockers, which can cause orthostatic hypotension. In addition, this study conducted various surveys on symptoms such as dizziness and autonomic abnormalities to obtain clinical information, so it may include recall bias from the participants.
In conclusion, we found that a considerable number of patients with de novo PD complained of dizziness and that their characteristics of dizziness varied. Such dizziness was more prevalent in women than in men, and was significantly associated with cognitive decline and cardiovascular dysautonomia. Thus, clinicians should monitor these clinical symptoms in patients with PD who complain of dizziness and provide appropriate management early.
PD, Parkinson’s disease; BMI, body mass index; UPDRS-III, Unified Parkinson’s Disease Rating Scale part III; PIGD, postural instability and gait disorder; TD, tremor-dominant; MoCA-K, Korean version of Montreal Cognitive Assessment; BAI, Beck Anxiety Inventory; BDI, Beck Depression Inventory; PFS, Parkinson’s Disease Fatigue Scale; SCOPA-AUT, Scales for Outcomes in Parkinson’s Disease-Autonomic Dysfunction; GI, gastrointestinal; CV, cardiovascular.
The data underlying this article will be shared on reasonable request to the corresponding author.
KYK designed the research study. JY, ROK, and EJL performed the research. KYK, JY, ROK, and EJL analyzed the data. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
All procedures were performed in accordance with ethical standards of the institution and/or the national research committee as well as with the 1964 Helsinki Declaration and its subsequent amendments. This study was retrospective and was approved with waiver of individual informed consent by the ethics committee of our Institutional Review Board (IRB No. 2022-12-003).
We thank all staffs in our department of Neurology for their kind and enthusiastic support.
This study was supported by Soonchunhyang University Research Fund.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.