1 Department of Neurology, The Affiliated Nanhua Hospital, Hengyang Medical School, University of South China, 421001 Hengyang, Hunan, China
2 Department of Intensive Care Unit, The Affiliated Nanhua Hospital, Hengyang Medical School, University of South China, 421001 Hengyang, Hunan, China
†These authors contributed equally.
Abstract
Background: Olfactory impairment has been reported in
patients with depression and in rodent models of depression. Olfactory epithelium
(OE) is the only peripheral neural tissue connected to the brain that has the
potential for self-renewal. We hypothesized the olfactory deficit during
depression may be related to the dysfunction of OE progenitor cells. The aim of
the present study was therefore to evaluate the expansion and neuronal
differentiation potency of cultured OE progenitor cells obtained from a rat model
of depression. Methods: Rats were exposed to chronic unpredictable mild
stress procedures to establish a depressive-like state. Depressive-like behavior
and olfactory sensing function were then evaluated and compared with control
rats. Primary OE progenitor cells were cultured in vitro. The
proliferation potency and survival of OE progenitor cells were assessed by
5-Ethynyl-2
Keywords
- depression
- olfactory epithelium
- progenitor cells
- cell expansion
- neuronal differentiation
Depression is a debilitating disorder with high morbidity and mortality. Despite
considerable research, the mechanisms underlying depression remain unclear. The
lack of access to living human brains is a major barrier to research in this
field. While autopsy studies can be informative, autopsy brains are a scarce
resource and are difficult to obtain. Moreover, results obtained from autopsy
brain tissue are biased due to the effects of disease course and drug therapy.
Hence, there is an urgent need for alternative ways to study depression.
Olfactory epithelium (OE) is located in the nasal cavity and is a unique neural
tissue comprised of abundant progenitor cells and olfactory sensory neurons (OSN)
that regenerate throughout life [1]. Although OE is located in the peripheral
part of the olfactory pathway, OSN can communicate with neurons in the olfactory
bulb via axons that extend through the cribriform plate. Thus, molecular
anomalies in the brain are always reflected in the olfactory pathway [2].
Moreover, OE can be readily obtained with minimal trauma. OE and
OE-derived progenitor cells are therefore emerging as promising resources for the
investigation of neuropsychiatric diseases. Aggravation of
Recent clinical evidence has shown that patients suffering from depression have difficulty in identifying odors [6, 7]. Moreover, olfactory performance is sensitive to antidepressant therapy, with improved olfactory scores in depressed patients being associated with the remission of clinical symptoms [8]. Another study reported that olfactory dysfunction implies a longer duration and course of depression, even though olfactory sensory function did not correlate positively with the severity of depression [9]. A neuroimaging study found the olfactory bulb had reduced volume in depressed patients compared to normal controls [10]. This clinical evidence for olfactory dysfunction may therefore provide new opportunities to investigate depression. However, the cause of olfactory dysfunction in depressed patients remains largely unclear. The hippocampus plays a pivotal role in stress regulation and cognitive processes, and altered neurogenic activity in the hippocampus has been associated with depression [11]. A postmortem study reported that depressed patients showed a decreased level of NeuN+ neurons in the hippocampus [12]. Enhancement of neurogenesis in the hippocampus may therefore protect against depression [13]. These studies suggest that olfactory sensing dysfunction during depression may be due to decreased neurogenesis of OE. Although peripheral neural tissue is exposed to environmental toxins, OE is capable of self-healing via increased proliferation and neuronal differentiation of progenitor cells. Poor quality of OE progenitor cells could therefore contribute to olfactory dysfunction in depression.
In the present study we evaluated olfactory function in the chronic unpredicted mild stress (CUMS)-induced rat model of depression. Primary OE progenitor cells were cultured and their proliferation, survival and neuronal differentiation potency was examined. We hypothesized that olfactory dysfunction during depression may be associated with decreased expansion and neuronal differentiation of OE progenitor cells.
All animal procedures were approved by the Ethics Committee of The Affiliated Nanhua Hospital. Male Sprague-Dawley (SD) rats was purchased from Hunan SJA Laboratory Animal Co., Ltd (Changsha, Hunan, China) aged 13–14 weeks (240–270 g) were maintained at 22 °C in a 12-hour light/dark cycle with free access to food and water, except when carrying out the CUMS procedure. Animals were randomly assigned to CUMS or control groups. The experimental groups were kept in isolation. After 48 hours of adaptation, rats were subjected to CUMS procedures consisting of a variety of unpredictable mild stressors. These included swimming in cold water (4 °C) or hot water (45 °C) for five minutes, cage tilting for 24 hours, water deprivation for 24 hours, fasting for 48 hours, shaking for 15 minutes, wet bedding for 24 hours, tail nip for two minutes, and inversion of the light/dark cycle. Subjects received one of these stressors at random each day for 21 consecutive days. The control group was given usual daily care and kept in groups. After the CUMS procedures, rats were subjected to behavioral tests as described below. Rats were then sacrificed by decapitation and the olfactory mucosa was collected.
Behavioral tests were performed after the CUMS procedure to evaluate depressive-like behavior and olfactory function. Each experiment was repeated three times and the average result recorded.
For the sucrose preference test, rats were first adapted to sucrose water before the test. Briefly, a bottle of 1% sucrose water and a bottle of purified water were given to each subject. Bottles were randomly fixed on each cage’s right or left side. The position of the two bottles was swapped 12 hours later. The sucrose preference test was carried out after 24 hours of adaptation. This was performed for 3 hours. The sucrose preference was calculated as: sucrose water consumption/total water consumption.
For the forced swimming test, rats were placed in a beaker filled with water at
25
For the open field test, rats were placed in a square container (100
The buried food-seeking test was used to assess olfactory function as previously described [14]. Briefly, each subject was food-deprived for 24 hours, then placed at the center of the test cage to search for a 2 g regular food chow pellet buried 8 centimeters beneath the bedding in a randomly chosen corner of the cage. The latency time, defined as the time between placing the rat into the cage and grasping the buried food with its forepaws and/or teeth, was recorded. If animals could not find the food pellet within 600 s, the test was terminated and a score of 600 s was given.
Following deparaffinization and rehydration, tissue slices were stained with hematoxylin for 5 minutes and then immersed five times in 1% acid ethanol. After several washes, slices were stained with eosin for 3 minutes and then dehydrated with graded alcohol and washed in xylene. Images were captured using a fluorescence microscope (Olympus, Tokyo, Japan).
OE was dissected from the olfactory mucosa under a dissecting microscope (SZX7, Olympus, Tokyo, Japan), cut into small pieces, digested in 0.125% Trypsin-ethylenediaminetetraacetic acid (EDTA) (Gibco, Anaheim, CA, USA) for 30 minutes, then centrifuged at 1400 rpm for 5 minutes and the cell pellet collected. To generate neurospheres, cells were resuspended and cultured in Dulbecco’s Modified Eagle’s Medium/Ham’s F 12 nutrient medium (DMEM/F12) supplemented with 50 ng/mL Epidermal Growth Factor (EGF) (PROSPEC, cyt-217, Rehovot, Israel), 50 ng/mL Recombinant Fibroblast Growth Factor 2 (FGF2) (PROSPEC, cyt-218), 1% ITS-X (Gibco, 51500056, USA), 2% B27 (Gibco, Anaheim, CA, USA), and 1% streptomycin/penicillin. The culture medium was changed every 4 days. For the purification of OE progenitor cells, the primary neurosphere was dissociated and the cells cultured again to generate secondary neurospheres. The fourth-generation neurospheres were collected for subsequent experiments. A commercially available Quantitative PCR mycoplasma detection kit was used for mycoplasma testing (Thermo Fisher, #4460623, Fisher, Waltham, MA, USA), no mycoplasma infection was found in the indicated cells. Nestin (a marker for neural stem cells) immunofluorescence staining was used for authentication of progenitor cells.
Purified progenitor cells dissociated from fourth-generation neurospheres were
seeded onto poly-L-lysine-coated coverslips. Cells were fixed and washed, blocked
for 60 minutes at room temperature, and incubated with primary antibody against
nestin (1:1000; #73349, Cell Signaling Technology, Danvers, MA, USA). The
samples were then incubated with a fluorescent secondary antibody (1:1000;
A23220, Abbkine, Wuhan, Hubei, China) for two hours in the dark at room
temperature. Finally, samples were counterstained with
4
OE progenitor cells were labeled with 5-Ethynyl-2
Cells were seeded onto a 96-well plate at 1
Cells were stained with PI (AP101, Multi Sciences, Hangzhou, Zhejiang, China) and Annexin (AP101, Multi Sciences) as per the manufacturer’s instructions. Cell apoptosis was then determined by flow cytometry methods.
Total RNA from OE progenitor cells was extracted using TRIZOL reagent (Sigma-Aldrich, St. Louis, MO, USA). cDNA was synthesized using a commercially available kit (Takara, Kusatsu, Japan). The expression of mRNA was examined using the SYBR Premix EX Taq I kit (Takara, Japan) on a 7300 Plus Real-Time polymerase chain reaction (PCR) System (Thermo Fisher, Waltham, MA, USA). Thermocycling conditions were as follows: an initial 95 °C step for 30 s, followed by 40 cycles at 95 °C for 5 s, and 60 °C for 60 s. GAPDH was used as the internal reference. The primer sequences used are shown in Table 1.
| Target gene | Forward primer sequence | Reverse primer sequence |
| CAACTATGTGGGGGACTCGG | TGGCTCTGGGCACATACTTG | |
| NES | GTGACCCTTGGGTTAGAGGC | CTGGCAAAATGCCTTGGGTC |
| SOX2 | ACATGGCCCAGCACTACCAGA | ATCTCTCCCCTTCTCCAGTTCG |
| OLIG2 | TGGGGACGCTGTTTTTAGCA | CAGAACACCCCCTCCCAAAT |
| GAPDH | ACAGCAACAGGGTGGTGGAC | TTTGAGGGTGCAGCGAACTT |
Cells were homogenized using Radioimmunoprecipitation assay buffer (RIPA) buffer containing Phenylmethanesulfonyl fluoride (PMSF). Proteins were
subjected to 10% Sodium Dodecyl Sulfate PolyAcrylamide Gel Electrophoresis (SDS-PAGE) and transferred onto
Polyvinylidene Fluoride (PVDF) membranes. After blocking for one hour,
the membrane was incubated separately with primary antibodies against
All data were expressed as the mean
A series of tests for depressive-like behavior was performed to evaluate the rat model of depression. As shown in Fig. 1, rats subjected to CUMS procedures exhibited reduced glucose consumption (p = 0.008; Fig. 1A), increased immobility (p = 0.017; Fig. 1B), and decreased body rearing (p = 0.003; Fig. 1C) compared to the control group. Moreover, depressive-like rats showed poorer olfactory function, as demonstrated by the buried food-seeking test (p = 0.007; Fig. 1D). These results indicate the state of depression was correlated with reduced olfactory performance in a rat model.
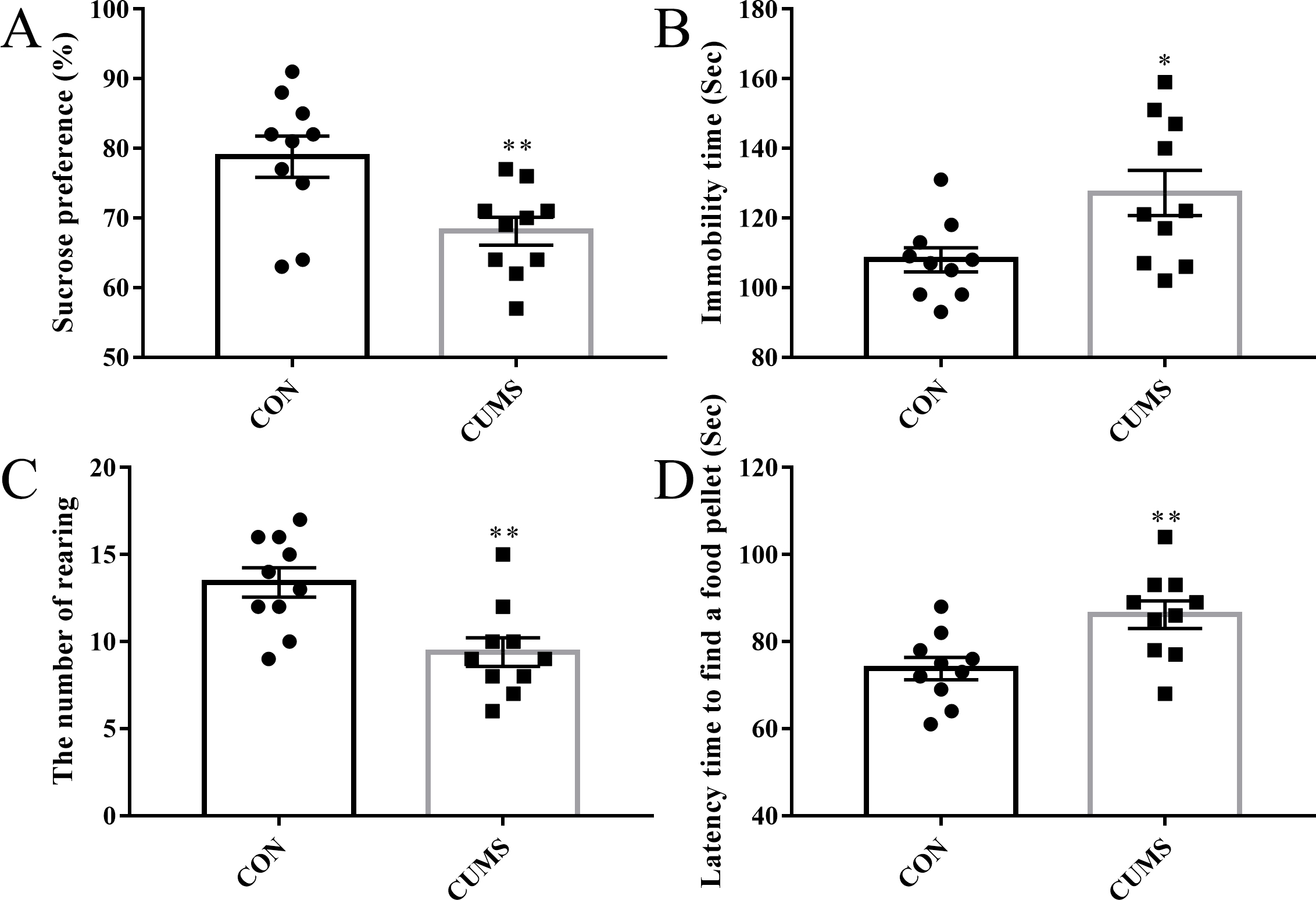 Fig. 1.
Fig. 1.Evaluation of depressive-like behavior and olfactory sensing
function. (A) Sucrose preference test. (B) Immobility time recorded in the
forced swimming test. (C) The number of rearing in the open field test. (D)
Latency time to find food in the buried food-seeking test. n = 10 per group.
*p
OE was isolated from olfactory mucosa, which is composed of the upper OE and the lower lamina propria (Fig. 2A). Neurosphere formation (an aggregated form of neural progenitor cells) is a key feature of neural progenitor cells. OE biopsies were collected for generating neurospheres, which emerged after culturing for 4 days in sphere formation medium (Fig. 2B). For the purification of progenitor cells, primary neurospheres were dissociated and cultured to generate secondary neurospheres. Fourth-generation neurospheres were then collected for the following experiment. Purified progenitor cells derived from fourth-generation neurospheres stained positively for Nestin (a marker of neural progenitor cells) using immunofluorescence (Fig. 2C). These results demonstrate the successful establishment of an in vitro model of OE progenitor cells.
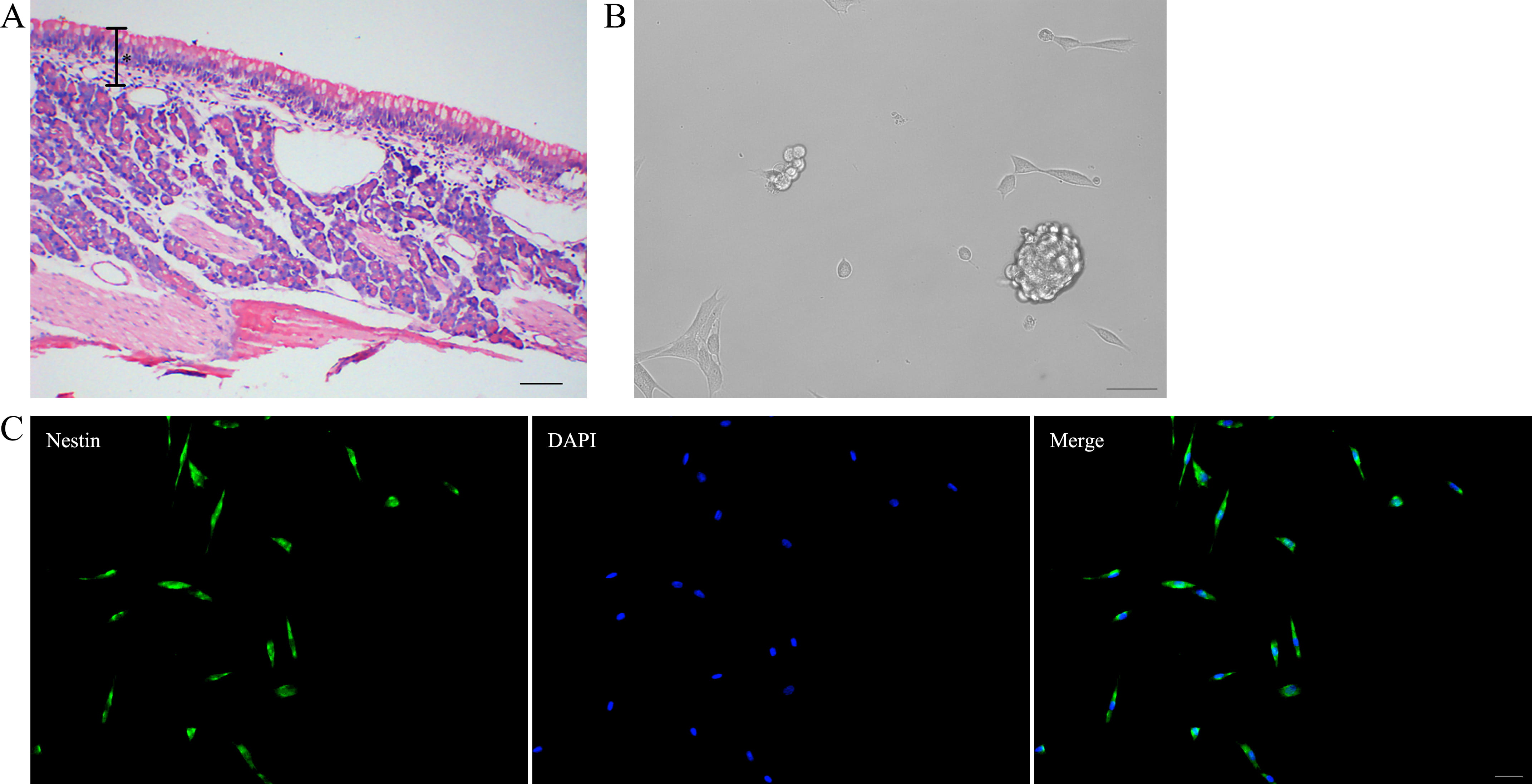 Fig. 2.
Fig. 2.Identification of olfactory epithelium (OE) progenitor cells.
(A) HE staining of the olfactory mucosa. * indicates the OE portion of olfactory
mucosa. Scale bar is 100 µm. (B) Neurosphere formation. Scale bar is 100
µm. (C) Purified progenitor cells derived from fourth-generation
neurospheres express nestin. Scale bar is 50 µm. HE, hematoxylin-eosin; DAPI, 4
EdU labeling was used to evaluate the proliferative potential of purified OE
progenitor cells (Fig. 3A). Compared to controls, OE progenitor cells derived
from depressive-like rats showed reduced proliferative potential
(p
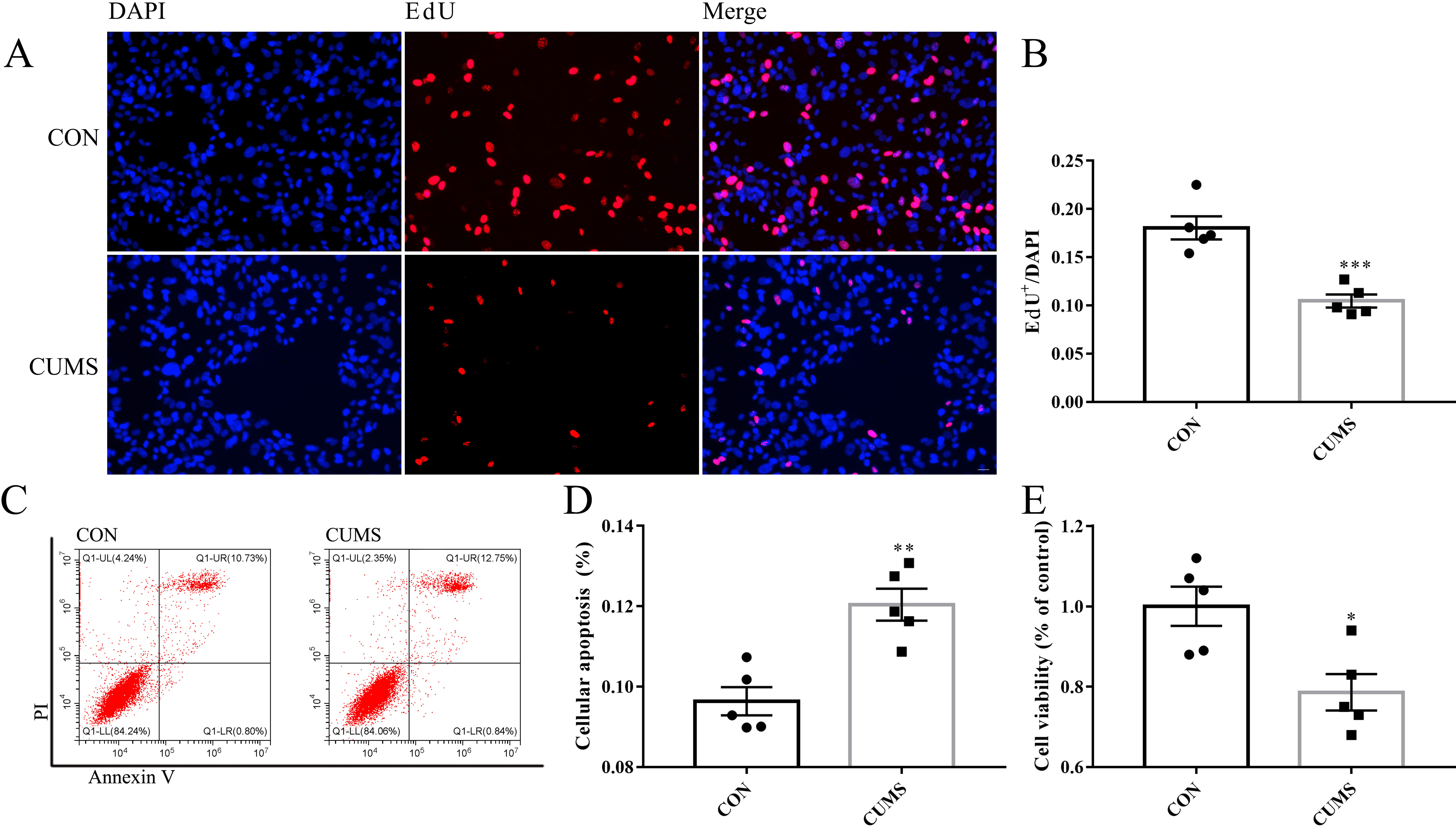 Fig. 3.
Fig. 3.Proliferation and survival ability of OE progenitor cells. (A)
EdU labeling for the measurement of proliferation potency. Scale bar is 20
µm. (B) Ratio of EdU
To study neuronal differentiation, purified OE progenitor cells were cultured
for 3 days in DMEM/F12 supplemented with fetal bovine serum and B27. OE
progenitor cells obtained from depressive-like rats showed reduced mRNA
expression of
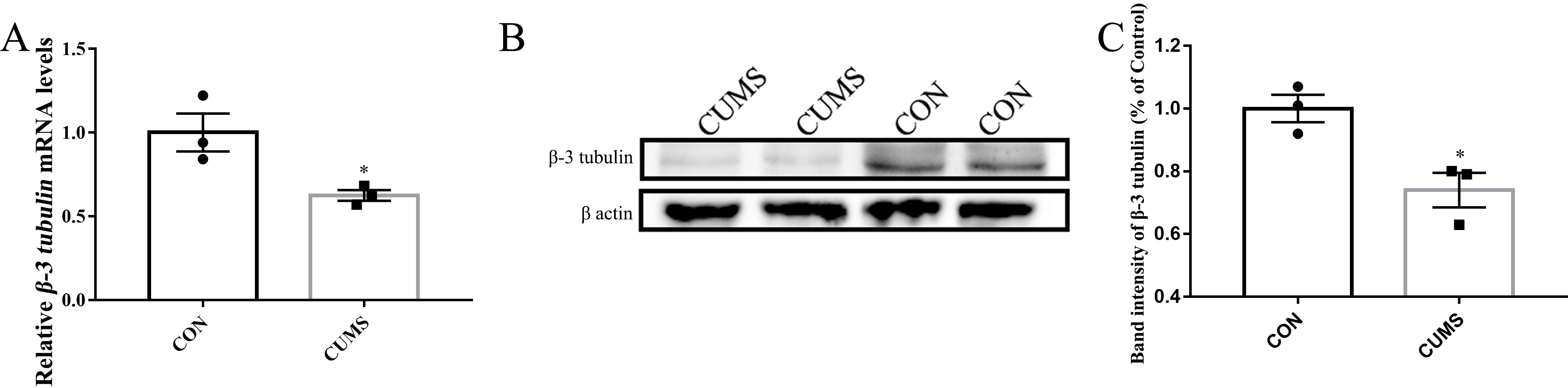 Fig. 4.
Fig. 4.OE progenitor cells derived from depressive-like rats showed
reduced potential for neuronal differentiation. (A) Relative mRNA expression of
To further evaluate the expression of mRNA related to neural stemness, we performed RT-PCR of cultured OE progenitor cells. Compared to the control group, OE progenitor cells obtained from rats exposed to CUMS showed reduced expression of mRNA related to neural stemness (NES: p = 0.002, SOX2: p = 0.04, OLIG2: p = 0.42; Fig. 5).
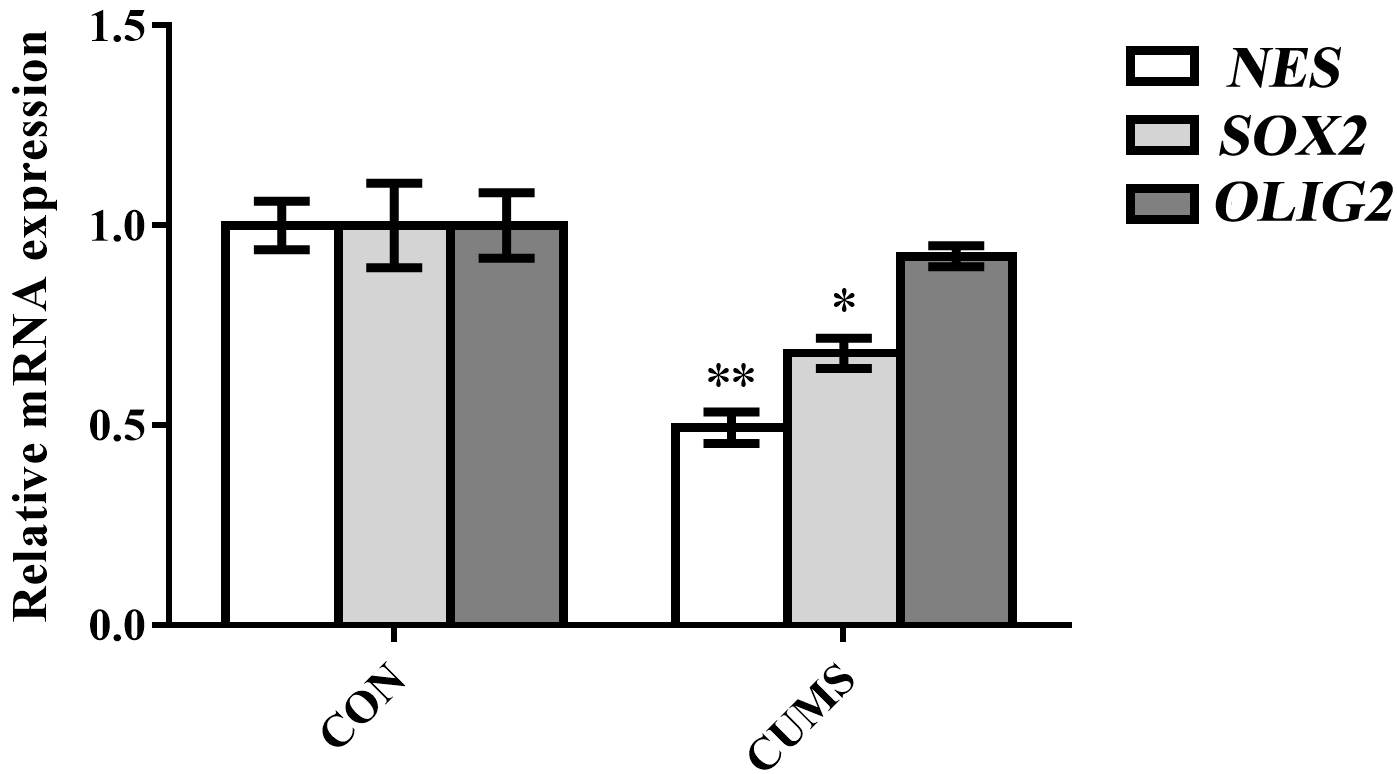 Fig. 5.
Fig. 5.Relative mRNA expression associated with neural
stemness in OE progenitor cells. n = 3 per group. *p
In the present study the state of depression in CUMS-induced depressive-like rats was found to correlate with olfactory dysfunction. We subsequently investigated the expansion and neuronal differentiation potency of primary cultured OE progenitor cells. As expected, OE progenitor cells derived from rats exposed to CUMS exhibited decreased potency for proliferation, survival, and neuronal differentiation. In addition, reduced mRNA expression associated with neural stemness was also observed in OE progenitor cells derived from depressive-like rats.
Despite anhedonia, we observed reduced olfactory sensing function in depressive-like rats. This could be due to the fact that emotional and olfactory processing are both regulated by the shared brain area, such as the amygdala [15]. An earlier study suggested that rats exposed to CUMS showed a reduced number of OSN in the OE [16]. As this peripheral neural tissue has the potential to regenerate throughout life, we speculated that the reduced number of OSN may contribute to the dysfunction of OE progenitor cells. To further test this idea, we evaluated the quality of OE progenitor cells in primary culture. The neural progenitor cell type is capable of multi-lineage differentiation and self-renewal. In the current study, OE progenitor cells had the ability to differentiate into neuron-like cells, as detected by RT-PCR and immunoblotting. Moreover, OE progenitor cells derived from depressive-like rats showed reduced ability for proliferation and neuronal differentiation. Other studies have also reported cellular dysfunction of OE progenitor cells obtained from patients with schizophrenia, Parkinson’s disease, or bipolar disorder [17, 18]. Previous studies in this active research area have suggested that decreased neurogenesis in the hippocampus might be responsible for anhedonia in depression. We have provided further evidence that decreased neurogenesis in the OE may be partly responsible for the olfactory dysfunction during depression. The genes associated with neural stemness are crucial for the maintenance of neural progenitor cells [19]. The present study also found that mRNA expression associated with neural stemness was reduced in OE progenitor cells obtained from depressive-like rats. Together, these results indicate that a poor quality of OE progenitor cells may contribute to the impairment of olfactory sensing function during depression.
OE progenitor cells are emerging as a robust tool for the pathobiological analysis of neuropsychiatric diseases. These cells have many advantages over other stem cell models. First, OE can be obtained dynamically with minimal trauma. Therefore, various pathological alterations in OE progenitor cells may correspond better to the different disease conditions. Second, unlike induced pluripotent stem cells or induced neuronal cells, OE progenitor cells are obtained directly from neural tissue and do not need genetic reprogramming, thus more closely resembling the natural state. Third, as the unique peripheral tissue connected to the olfactory pathway, OE progenitor cells may serve as a window to explore pathobiological changes in the neurocircuitry of the OE, olfactory bulb, and olfactory cortex. Lastly, the OE progenitor cell model should be more convincing for the exploration of disease-specific changes since it has the same genetic background as the subject. Experimental studies examining protein localization/abundance, epigenetic markings, and gene expression have already reported disease-specific changes in several neuropsychiatric diseases using this in vitro cell model [20]. Of note, long non-coding RNA (lncRNA)-microRNA (miRNA)-messenger RNA (mRNA) network is emerging as potential diagnostic biomarkers for depression [21], we speculate that olfactory progenitor cells may be a robust tool for this field.
Olfactory signals are regulated by both internal and external stimuli [22]. Internal stimuli such as neurotransmitters, paracrine signals and hormones can affect the survival of olfactory sensory neurons and progenitor cells. Further research is needed to identify the initial factors in the brain that reduce the expansion and neuronal differentiation potency of OE progenitor cells. External stimuli such as environmental pathogens and toxicants that irritate nasal inflammation may also impact the OE microenvironment, thereby affecting olfactory sensing function. An epidemiological study found that patients who suffer chronic nasosinusitis have a higher prevalence of depression [23]. The relationship between environmental factors and depression will be interesting to explore in future work.
There are several limitations to this study. First, additional phenotypic and histochemical studies of OE and OE-derived progenitor cells from depressive patients may allow more convincing conclusions to be drawn. Second, comparison of the differentially expressed genes in OE progenitor cells between depressed subjects and normal controls is needed to further elucidate the mechanism of olfactory impairment. Finally, the extent to which rodent models of depression can accurately reflect the pathophysiological changes occurring in depressed patients requires further investigation [24].
This study suggests that olfactory dysfunction and anhedonia occur simultaneously in a rat model of depression. Importantly, these results indicate that olfactory dysfunction during depression may be due to the reduced expansion and neuronal differentiation potential of OE progenitor cells. OE progenitor cells may be a robust and feasible tool for the investigation of depression.
Data available upon reasonable request.
LC, YX, JC and YL designed the research study. LC and YX performed the research. LC and YX conducted experiments. JC analyzed the data. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The animal study protocol was approved by the Institutional Review Board of The Affiliated Nanhua Hospital (protocol code: 2023-1LY-01).
The authors would like to thank Z. Peng and H. Zhang for their technical assistance.
This work was supported by Key Project of Hunan Provincial Health Commission (NO. 202203074157).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





