1 Department of Traditional Chinese Medicine, Children’s Hospital of Fudan University, 201102 Shanghai, China
Abstract
The insulin-like growth factor (IGF) system plays a vital role in regulating gonadotropin-releasing hormone (GnRH), whether the IGF2 can act on the GnRH neurons during the pubertal period is unclear.
Central precocious puberty (CPP) rats were induced by danazol, and when the rats met the first diestrus, they were euthanized and tissues were collected. GT1-7 cells were cultured and treated with 0, 1, 10 ng/mL IGF2 for 4 hours and the changes in GnRH were measured. Mice were injected intracerebroventricularly with IGF2 (15 ng/g, 5 μL) or with the same dose of phosphate buffered saline (PBS), after eight hours, they were euthanized and tissues collected.
CPP rats had increased expression of IGF2 and GnRH mRNA and their respective proteins in the preoptic area (POA) of the hypothalamus. Treatment of GT1-7 cells with 10 ng/mL of IGF2 increased GnRH mRNA and protein expression, and GnRH concentration in the culture medium. Injection of IGF2 protein into the lateral ventricle of mice increased the expression of GnRH mRNA and protein in the POA.
IGF2 may upregulate the synthesis of GnRH during the pubertal period, and may also take part in the pathology of CPP.
Keywords
- gonadotropin-releasing hormone
- insulin-like growth factor 2
- central precocious puberty
- GT1-7
Precocious puberty is clinically defined as the appearance of secondary sexual characteristics in males before the age of nine years or in females before the of seven and a half years. Central precocious puberty (CPP) is associated with the early activation of the gonadotropin-releasing hormone (GnRH) generator [1]. The cell bodies of GnRH neurons are mainly distributed in the hypothalamic preoptic area (POA). A small number of neurons are found in the mediobasal hypothalamus (MBH), which can extend to the median eminence-arcuate (ME-ARC) and release GnRH [2]. In our study, we divided the hypothalamus into the POA and the MBH by performing a separation 2 mm posterior to the optic chiasm. The MBH includes the ARC and the ME. Some studies have suggested that the peak production of peripheral blood luteinizing hormone (LH) is associated with the activation of GnRH neuron perikarya in the POA and morphological changes in GnRH neurons in the ME [3, 4]. The ME is the area where neuroendocrine substances are released from nerve terminals into the capillary networks of the primary plexus. It acts as a neuroendocrine transducer, with a dense network of capillaries formed between the ME and the tuberal part of the pituitary, neuroendocrine substances can enter the pituitary in the form of neuropeptides [5]. The ME-ARC structure in the MBH plays a crucial role in the release of GnRH. When the MBH is isolated using special techniques, stimulation of the MBH can promote GnRH release, but no peak in LH is observed in the peripheral blood [6]. Conversely, isolation of the POA does not affect the peak [7]. This may be because GnRH neuron cell bodies are located in the POA, and isolating the MBH disrupts the connection between the POA and MBH, preventing the maintenance of GnRH pulsatile release. These findings indicate that the ME-ARC structure in the MBH plays a primary regulatory role in GnRH release but is not sufficient to maintain its pulsatile rhythm [5].
The insulin-like growth factor (IGF) system includes insulin, IGF1, and IGF2 as ligands and insulin receptor-A (INSR-A), INSR-B, IGF1R, INSR/IGF1R hybrids, and IGF2R as receptors [8]. Many studies have shown that insulin and IGF1 can act on the hypothalamus, regulating the onset of puberty [9, 10, 11, 12, 13, 14]. Daftary and Gore [10] found that the expression of IGFR increases with age in both the POA and MBH, indicating that the IGF1-IGF1R system plays an important role in reproductive system development. IGF1 could directly regulates GnRH neurons, continuous intracerebroventricular injection of IGF1 in female rats could advance puberty [11], and delayed puberty was observed in GnRH-IGF1R-Knock out (GnRH-IGF1R-KO) mice [12]. Besides Bálint et al. [13] also discovered that IGF1 can indirectly regulate GnRH by modulating gamma-aminobutyric acid (GABA) neurons.
IGF1 and IGF2 are highly conserved across different species and possess homologous sequences with IGF-like structures. However, IGF2 has a unique E domain segment in addition to its shared sequence with IGF1. The function of this segment is currently under investigation [15, 16]. Both IGF1 and IGF2 can bind to receptors such as the IGF1R, but only IGF2 binds specifically to the IGF2R receptor, which is associated with the clearance of IGF2 [8]. While IGF1 and IGF2 are expressed throughout most tissues over a lifetime, their spatiotemporal specificities differ. The concentration of IGF2 in fetal circulation is several times that of IGF1, with IGF1 increasing during adolescence in normal children, while IGF2 remains relatively stable [16]. Although mice with IGF1 or IGF2 knockouts exhibit abnormalities in gonadal development and delayed puberty, current research on IGF2 has predominantly focused on its impact during the fetal period [17, 18]. A study conducted in 1995 showed that the secretion of GnRH in GT1-7 cells was increased after treatment with IGF2 [19], and a clinical study from 2018 indicated a correlation between IGF2 and CPP [20]. Our investigation revealed a synchronous elevation of IGF2 and GnRH in a precocious puberty model. Consequently, we aimed to validate whether the elevated IGF2 during adolescence could lead to abnormal GnRH regulation. To further verify the role of IGF2 in the regulation network of GnRH during the postnatal period, we examined changes in IGF2 in the POA and the MBH of the hypothalamus of CPP rats. Subsequently, we treated GT1-7 cells with recombinant IGF2 protein and performed lateral ventricle injections in mice to further validate whether IGF2 mediated the synthesis and secretion of GnRH in the hypothalamus during the postnatal period.
The research protocol was approved by the Ethics Committee of Children’s Hospital of Fudan University ([2020] 211).
The danazol-induced precocious puberty model is a classic model with high stability and operational convenience. Many studies have shown that in rats with precocious puberty induced by danazol, there is an increase in GnRH expression in the hypothalamus [21, 22, 23]. This is also key evidence indicating that the precocious puberty model is a CPP model [24]. In our study, 18 female Spraque-Dawley (SD) rats (5 days old) were randomly divided into three groups, with six rats in each group: normal group (N), solvent control group (C), and model group (M). At 5 days of age, the model and solvent control groups received a single subcutaneously injection with danazol (300 ng/25 µL, MedChemexpress, HY-B1029, Austin, TX, USA) and solvent (ethanol:ethylene glycol = 1:1) respectively in the morning. Starting from the age of 15 days, the time of vaginal opening was recorded, and vaginal smears were performed when vaginal opening was observed. Different periods of the estrous cycle were identified based the method published by Cora et al. [25]. At the first estrus phase following a complete estrous cycle in the Model group rats, we euthanized the rats from the N, C and M groups using 10% urethane (51-79-6, Aladdin, Shanghai, China) at a random ratio of 1:1:1.
The rats were anesthetized with 10% urethane and blood, hypothalamus, uterus, and ovaries were collected. Hypothalamic tissue was dissected into the POA and the MBH. The POA was dissected from 1 mm anterior of the optic chiasm to the border of the optic chiasm, and the MBH was dissected from the border of the optic chiasm to the mammillary bodies. All detailed methodology is described in Fig. 1.
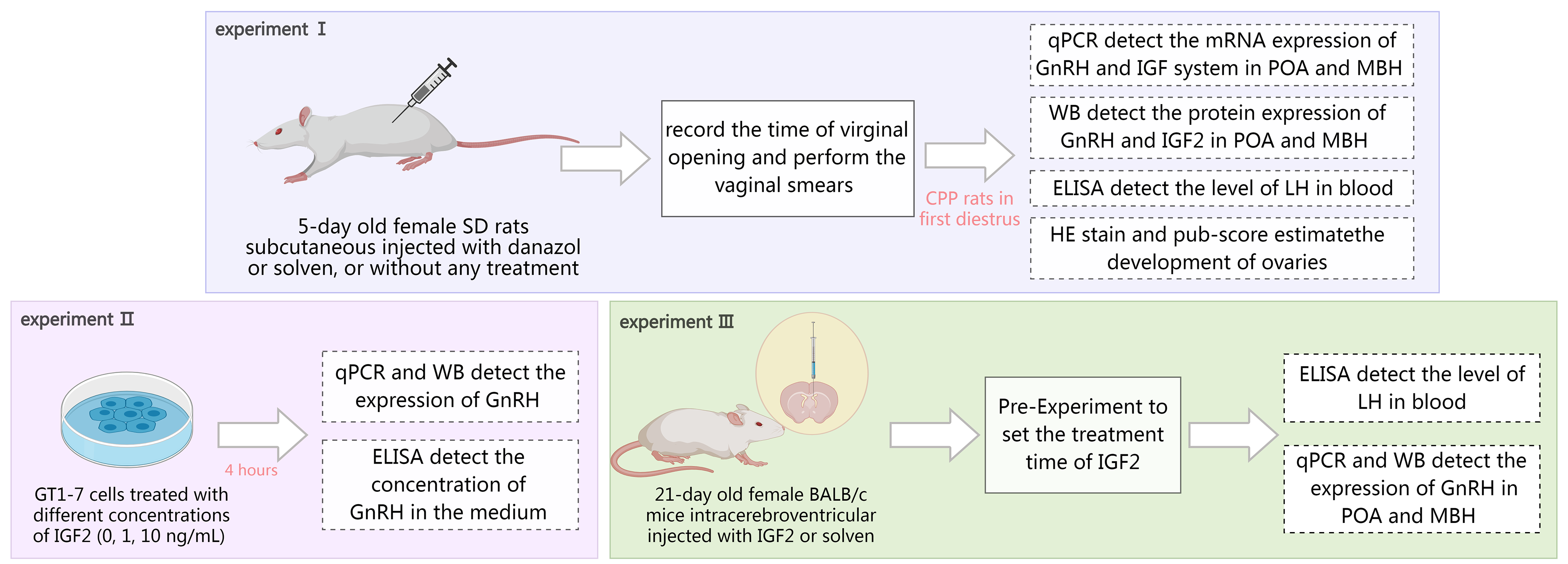 Fig. 1.
Fig. 1.
Experiments design for the research. SD rats, Spraque-Dawley rats; qPCR, quantitative polymerase chain reaction; GnRH, gonadotropin-releasing hormone; IGF, insulin-like growth factor; POA, preoptic area; MBH, mediobasal hypothalamus; ELISA, enzyme-linked immunosorbent assay; LH, luteinizing hormone; HE, hematoxylin and eosin; WB, Western Blot.
The GT1-7 cell line was generously donated by Professor Pamela Mellon (San Diego, CA, USA) and transported by Professor Feng Tao (Beijing, China). The cell line has been authenticated using short tandem repeat analysis and has completed mycoplasma contamination testing, which indicates no mycoplasma contamination. Based on the previous study, we selected concentrations of 0, 1, and 10 ng/mL for cell treatment [26]. The ratio of cell culture medium is as follows: 10% fetal bovine serum (F8318, Sigma, St. Louis, MO, USA), 1% dual antibody (15140-122, Gibco, St. Louis, MO, USA), and 89% high glucose Dulbecco’s modified Eagle medium (DMEM) (C11995500BT, Gibco, St. Louis, MO, USA), cultured in an environment of 5% CO2 at 37 °C. When the cells reached approximately 80% confluence, the culture medium was replaced with DMEM, and the cells were treated with varying concentrations of IGF2 protein (792-MG, R&D Systems, Minneapolis, MN, USA) (0, 1, 10 ng/mL), for 4 hours. After the treatment was completed, the culture medium was centrifuged at 3000 rpm for 20 minutes. The supernatant was frozen and stored at –80 °C. The samples were then tested using a GnRH enzyme-linked immunosorbent assay (ELISA) kit (E-EL-0071, Elabscience, Wuhan, Hubei, China). The cells were washed three times with phosphate buffered saline (PBS), and RNA or protein was extracted following the protocols for RNA and protein extraction. The morphology of the cells 48 hours after resuscitation was shown in Fig. 2.
 Fig. 2.
Fig. 2.
The morphology of the cells 48 hours after resuscitation
(100
To better apply the results of the in vitro experiments to the in vivo experiments, mice were used for subsequent experiments. First, we performed a preliminary experiment using two female BALB/c mice in each group to determine the treatment time. Then 12 female BALB/c mice were divided into two groups: a PBS control (P) group and an IGF2 treatment (T) group. After intraperitoneal injection of tribromoethanol (1.25%, 0.2 mL/10 g, Nanjing aibei Biotechnology, Nanjing, Jiangsu, China), IGF2 or PBS was injected into the right lateral ventricle, the intracerebroventricular injection was performed as previously described by Kim et al. [27]. The treatment group (T) was injected with IGF2 (15 ng/g, 5 µL, 792-MG, R&D, Minneapolis, MN, USA), the PBS control group (P) was injected with the same dose of PBS. Eight hours after the intracerebroventricular injection of IGF2, the mice were euthanized with tribromoethanol (75-80-9, Aladdin, Shanghai, China), and the hypothalamus and blood were collected. The boundary delineation of the POA and MBH was the same as in Experiment 2.
The ovarian slices were prepared using the paraffin-embedded section process,
with a thickness of 5 µm. Ovarian slices were stained with hematoxylin and
eosin (HE). The development levels of ovaries were estimated using a scoring
method (pub-score), published by Gaytan et al. [28]. The morphological
changes of follicles at different stages and the pub-score scoring process are
shown in Fig. 3. In this study, scores greater than or equal to eight were rated
as eight points. Organ coefficients of the ovaries and uterus were calculated
using the following formula: organ coefficient = wet organ weight (g)/body weight
(g)
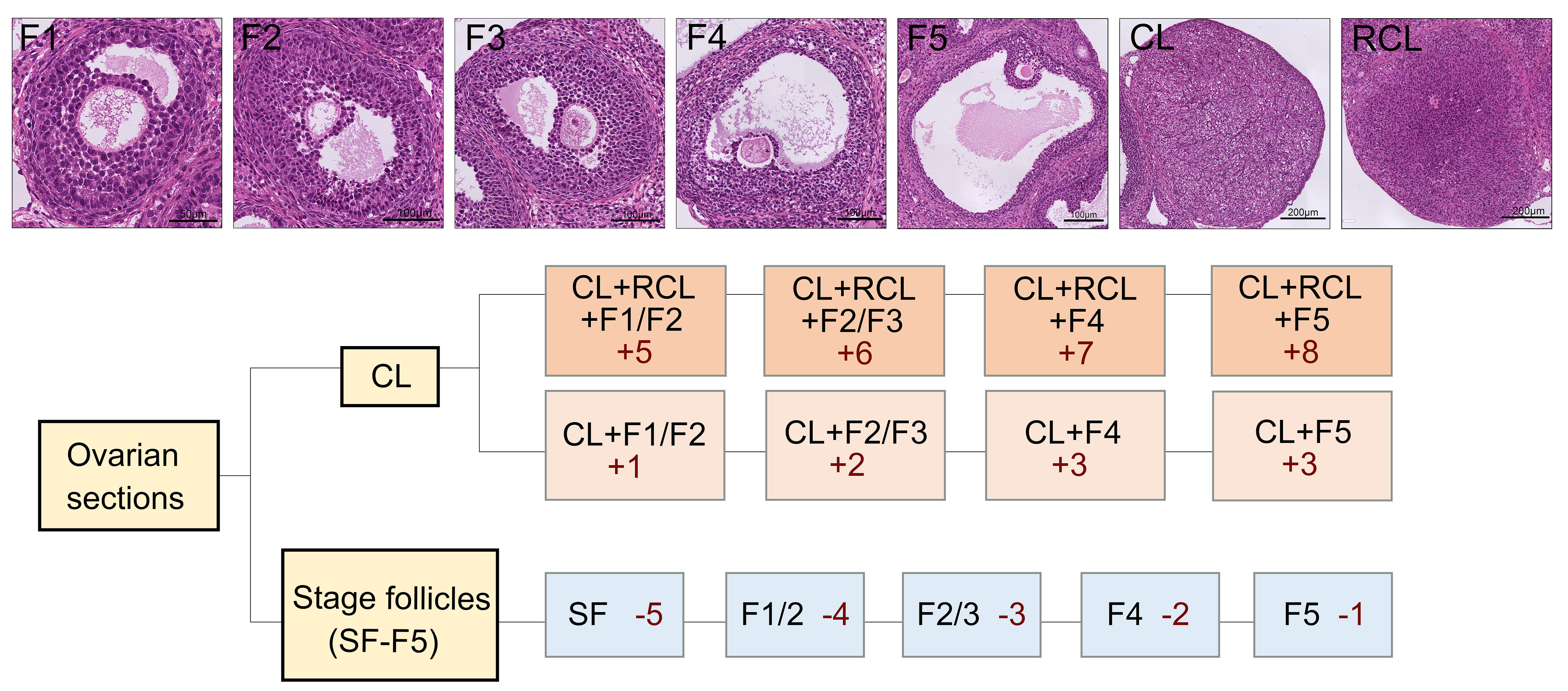 Fig. 3.
Fig. 3.
Morphological changes of follicles at different stages and the scoring process of pub-score. The different forms of follicles and corpus luteum correspond to different scores. According to the scoring process, the developmental stage of the ovaries can be assessed. F1, follicles 1; CL, corpus luteum; RCL, recurrent corpus luteum; SF, small follicles. F1: scale bar = 50 µm; F2, F3, F4, F5: scale bar = 100 µm; CL, RCL: scale bar = 200 µm.
Serum LH level and GnRH concentration in the culture medium were measured using ELISA kits (E-EL-M3053, Elabscience, Wuhan, Hubei, China) according to the manufacturer’s instructions.
The TRIzol method was performed to extract RNA. The concentration and purity of RNA were measured, and reverse transcription was performed using the PrimeScript™ RT Master Mix (RR036A, TAKARA, Kyoto, Japan). Quantitative polymerase chain reaction (qPCR) was performed using the SYBR Premix Ex Taq™ II system (RR820A, TAKARA, Kyoto, Japan). The primer sequences are listed in Table 1.
| Gene | Primer sequences (from 5′ to 3′) |
| GAPDH-F (rat) | GCAAGTTCAACGGCACAG |
| GAPDH-R (rat) | GCCAGTAGACTCCACGACAT |
| GAPDH-F (mice) | AGGTCGGTGTGAACGGATTTG |
| GAPDH-R (mice) | TGTAGACCATGTAGTTGAGGTCA |
| GnRH-F (rat) | GGGCAAGGAGGAGGATCAAAT |
| GnRH-R (rat) | CGCAGATCCCTAAGAGGTGAA |
| GnRH-F (mice) | AGCACTGGTCCTATGGGTTG |
| GnRH-R (mice) | GGGGTTCTGCCATTTGATCCA |
| IGF1-F | GCACTCTGCTTGCTCACCTT |
| IGF1-R | CTTGGTCCACACACGAACTG |
| IGF1R-F | CACATCCTGCTCATCTCCAA |
| IGF1R-R | AAGAGGTCTCCCAGGCTCTC |
| IGF2-F | TACCTCTCAGGCCGTACTTCC |
| IGF2-R | TCCAGGTGTCGAATTTGAAGA |
| IGF2R-F | TTGCTATTGACCTTAGTCCCTTGG |
| IGF2R-R | AGAGTGAGACCTTTGTGTCCCCAC |
GAPDH, glyceraldehyde-3-phosphate dehydrogenase; GnRH, gonadotropin-releasing hormone; IGF1, insulin-like growth factor 1; IGF1R, insulin-like growth factor 1 receptor; IGF2, insulin-like growth factor 2; IGF2R, insulin-like growth factor 2 receptor.
Protein extraction was performed by radioimmunoprecipitation assay (RIPA) buffer. The protein concentration was
determined by the BCA kit (P0012S, Beyotime Biotechnology, Shanghai, China), 12% sodium dodecyl sulfate
polyacrylamide gel electrophoresis (SDS-PAGE) gels were prepared, and the samples were loaded. The antibodies were
rabbit anti-IGF2 (1:500, Ab9574, Abcam, Cambridgem, UK), rabbit anti-GnRH
(1:1000, abs120521, Absin, Shanghai, China), and rabbit anti-
Data analysis was performed using GraphPad software (8.0, GraphPad Software, San Diego, CA, USA). One-way analysis of variance (ANOVA) or t-test were used when the data followed a normal distribution. A nonparametric test was used when the data did not follow a normal distribution. The statistics depicted in the figures were shown in the supplement.
In our study, the model group showed early vaginal opening (p
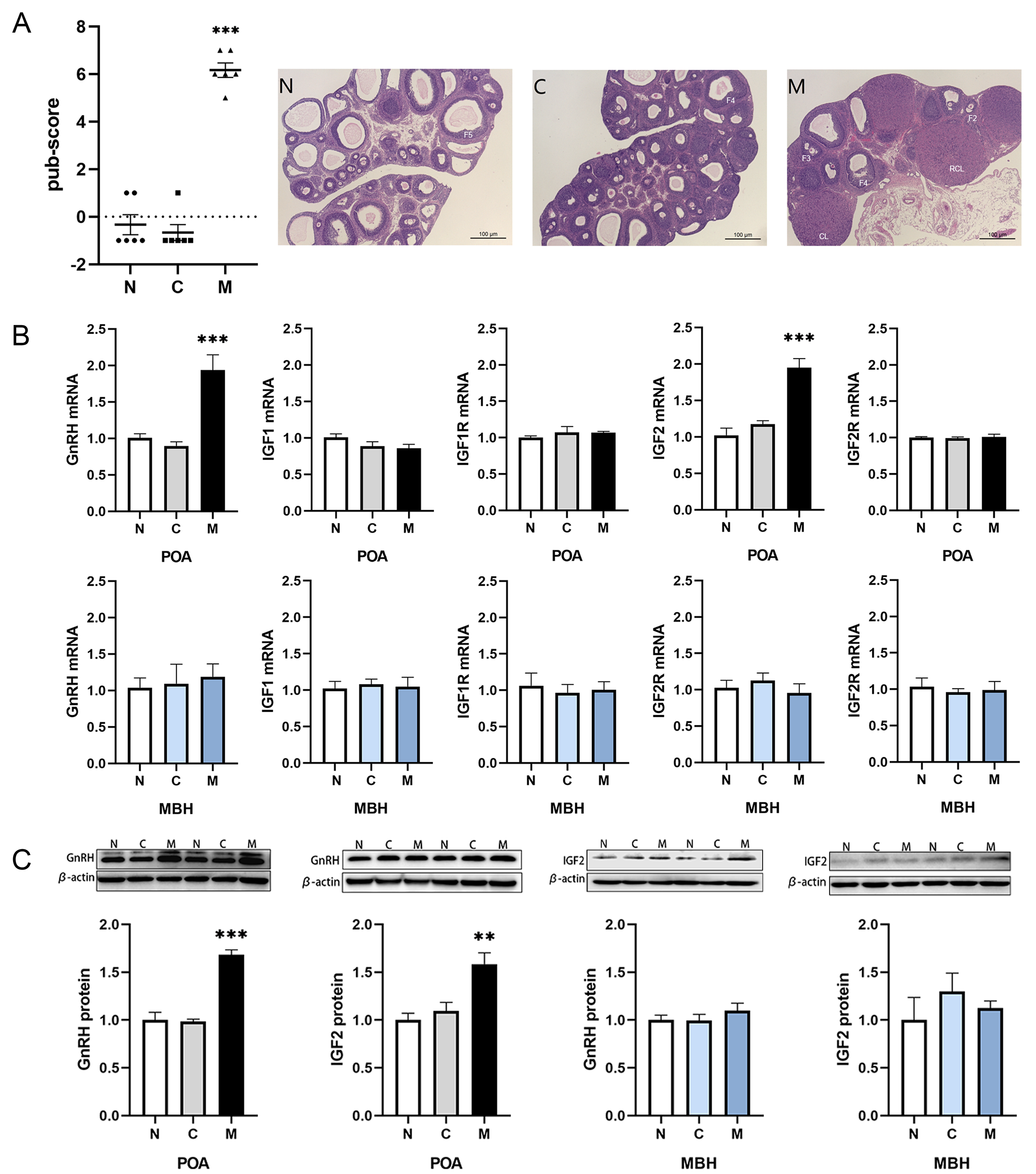 Fig. 4.
Fig. 4.
Expression of the IGF system in rats with CPP. (A) The
pub-score ratings of each group of rat ovaries and representative ovarian
sections. Scale bar = 100 µm. (B) The mRNA expression levels of GnRH, IGF1, IGF1R, IGF2, and IGF2R in
the hypothalamus of rats in the different groups. (C) The protein expression
levels of GnRH and IGF2 in the hypothalamus of rats in the different groups. Data
are presented as mean and SEM, compared to the control group. **p
| Group | Time of virginal opening (PND) | Ovarian coefficient (×104) | Uterus coefficient (×104) | LH level in serum (mIU/mL) |
| N | 33.83 |
3.16 |
13.27 |
23.42 |
| C | 33.67 |
3.48 |
14.50 |
20.91 |
| M | 22.50 |
4.18 |
14.71 |
51.45 |
Data are presented as mean and standard error of the mean (SEM), compared to the
control group, **p
Rats subcutaneously injected with danazol, at the age of 5 days, exhibit early
vaginal opening, elevated LH levels and more mature gonads [30]. In most previous
studies, the GnRH levels in the POA and the MBH were not examined separately. We
found that during the first diestrus phase in the model group, there was an
increase in GnRH mRNA expression in the POA (p
The IGF family is strongly associated with the occurrence of precocious puberty.
In this study, we examined the mRNA expression of IGF1, IGF1R, IGF2, and IGF2R in
different regions of the hypothalamus of rats with danazol-induced precocious
puberty. During the first diestrus phase in precocious puberty rats, we found
changes only in the mRNA expression of IGF2 in the POA (p
After discovering the upregulating trend in the mRNA levels of the IGF2 in the
POA of the precocious puberty model (both p values
GT1-7 is a commonly used hypothalamic cell line in reproductive research, representing GnRH neurons. GT1-7 cells not only synthesize and secrete GnRH but also exhibit pulsatile release characteristics. In a 1999 study, it was shown that the effects of IGF1 and IGF2 on GT1-7 cells vary with increasing concentration and duration of stimulation. When the concentration was too high or the stimulation time was too long, both IGF1 and IGF2 inhibited GnRH secretion from GT1-7 cells [26].
In our study, we found that after stimulation with 10 ng/mL IGF2 for 4 hours,
the mRNA and protein levels of GnRH in GT1-7 cells increased (p
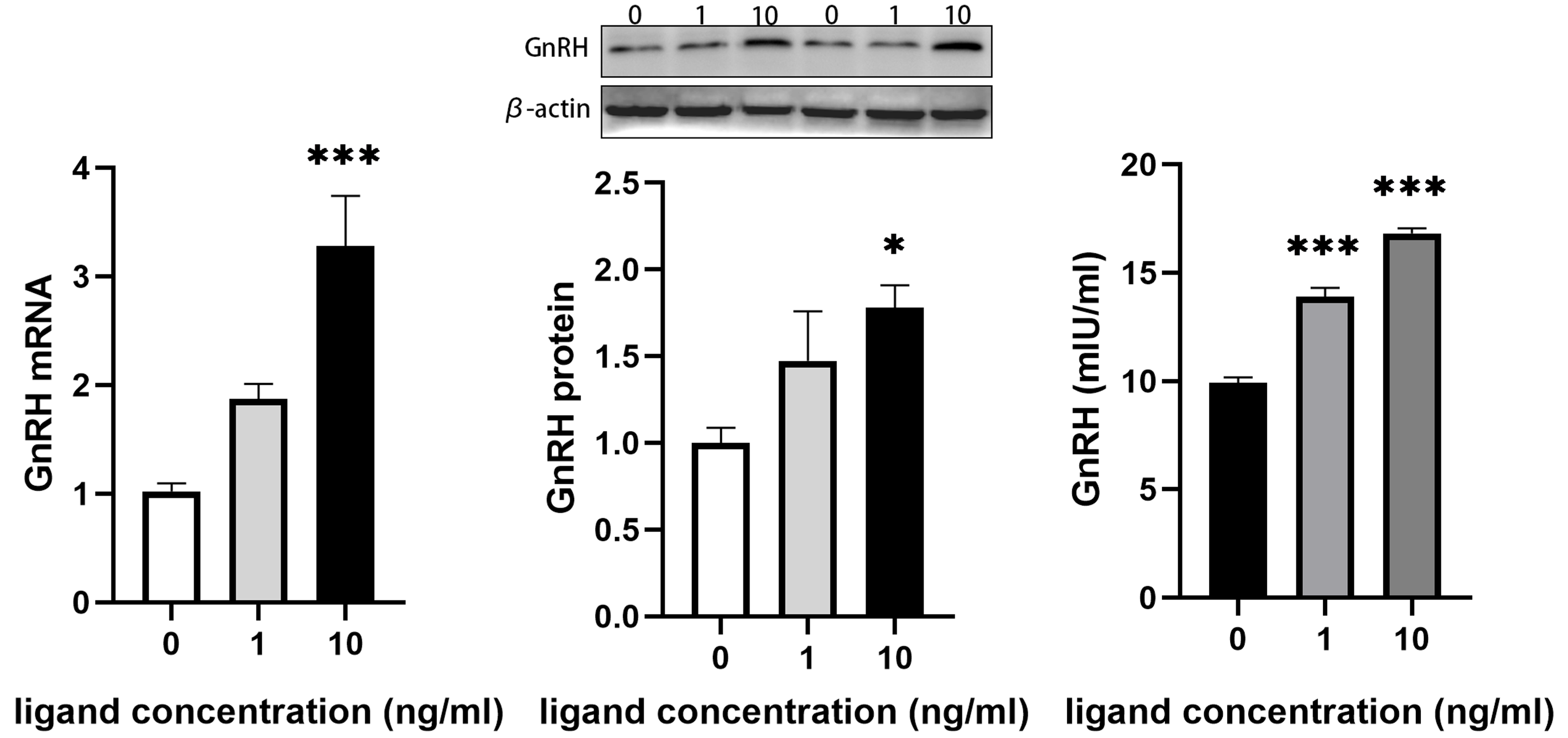 Fig. 5.
Fig. 5.
GnRH synthesis and secretion in GT1-7 cells treated with
different concentrations of IGF2 for 4 hours. Data are presented as mean and
SEM, compared to the control group (0 ng/mL), *p
To further elucidate the role of IGF2 on GnRH, we performed a preliminary experiment to determine the optimal treatment (Fig. 6A). Subsequently, we administered a higher dose of IGF-2 via intracerebroventricular injection into the mouse brain and conducted tissue sampling after 8 hours. Our results revealed that after 8 hours, IGF2 elevated the LH levels (p = 0.039), and exerted a stimulatory effect on GnRH mRNA in the hypothalamus (p = 0.041) (Fig. 6B), with a more pronounced difference observed at the protein level (p = 0.005) (Fig. 6C). The detailed values can be found in Supplementary Table 3. Notably, this stimulatory effect was specific to GnRH expression in the POA and did not extend to the MBH. This specificity may be associated with the precise regulatory role of IGF2 on GnRH, suggesting a preference for regulating the perikarya of GnRH neurons and promoting GnRH secretion through the stimulation of GnRH synthesis.
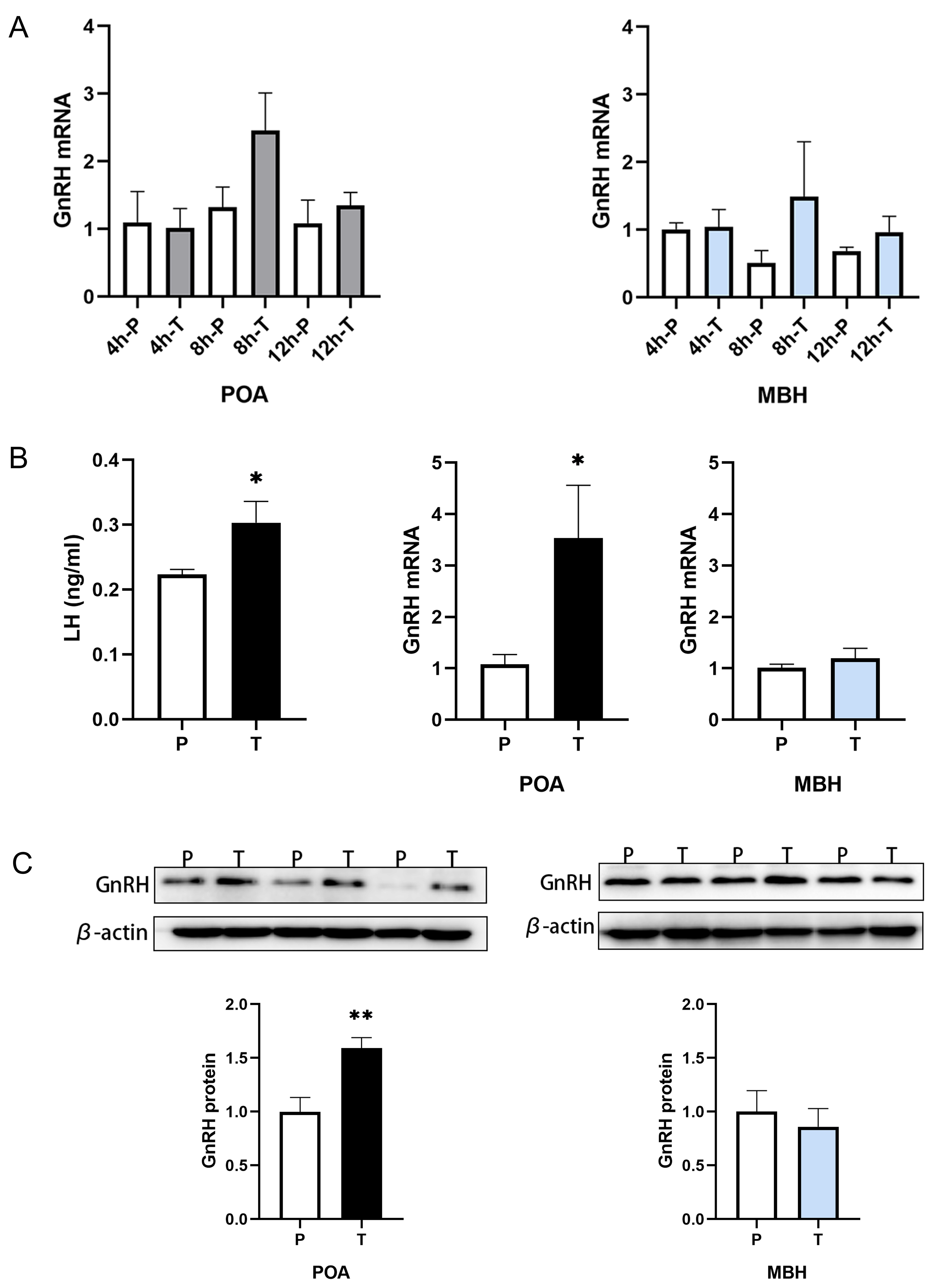 Fig. 6.
Fig. 6.
The expression of GnRH of hypothalamus in mice injected
intracerebroventricularly with IGF2. (A) The pre-experiment of
intracerebroventricular injection of IGF2. (B) The LH level and GnRH mRNA of mice
injected intracerebroventricularly with IGF2 for 8 hours. (C) The protein level
of mice injected intracerebroventricularly with IGF2 for 8 hours. Data are
presented as mean and SEM, compared to the P group, *p
In our study, we divided the hypothalamus into two parts, the POA and the MBH, which contain the ME-ARC. Various factors can form a GnRH regulatory network that participates in the initiation of puberty. The IGF system is involved in the onset of puberty. Circulating IGF1 in rats shows an increasing trend before puberty and insulin, IGF1 and IGF2 can cross the blood-brain barrier (BBB) and combine their receptors, which are expressed on neurons in the POA and the MBH [32, 33].
The danazol-induced precocious puberty model is a classic CPP model. By assessing the mRNA expression levels of IGF1, IGF1R, IGF2, and IGF2R in the hypothalamus, we found that IGF2 expression was increased in the POA of the hypothalamus, similar to the trend observed for GnRH. This suggests that IGF2 acts on GnRH synthesis during the pubertal period, so we performed in vitro and in vivo experiments in mice to further confirm this hypothesis. To further elucidate the effect of IGF2 on GnRH synthesis and secretion, we stimulated GT1-7 cells with 0, 1, or 10 ng/mL IGF2 for 4 hours and found that IGF2 participates in the regulation of multiple stages of GnRH synthesis and secretion. A previous study showed that IGF2 can cross the BBB [33], indicating that it may also can perfuse to the GnRH neurons by intracerebroventricular injection. To clarify the effect of IGF2 on GnRH, we injected IGF2 into the lateral ventricles of prepubertal mice. Our results showed that IGF2 upregulated the expression of GnRH in the POA, but not in the MBH. It is currently believed that the GnRH neuron perikarya in the POA is primarily involved in GnRH synthesis and that molecules in this region mainly influence the final secretion of GnRH by regulating its synthesis [34]. In contrast, a small number of GnRH neurons in the MBH and regulation of GnRH synthesis in this area may not have as significant an impact on GnRH synthesis as in the POA. GnRH neurons primarily project to the ME; therefore, it is thought that molecules in the MBH can be related to the regulation of GnRH neuron release or the morphological changes in GnRH neuron terminals [35]. Excitatory signals can promote GnRH secretion by reshaping the axons of GnRH neurons or by directly upregulating GnRH release [36]. Miller and Gore [37] found that even though the GnRH neuron perikarya located in the POA expressed more IGF1, in the hypothalamus it was more inclined to act directly on GnRH neuroterminals in the ME, thus directly regulating GnRH secretion. In our study, we found that danazol-induced CPP rats showed a synchronized increase in GnRH and IGF2 expression in the POA, whereas there was no such trend in the MBH. Intracerebroventricular injection of IGF2 in mice led to an increase in GnRH expression in the POA, whereas GnRH expression in the MBH remained unchanged. Therefore, our preliminary assumption was that IGF2 may have a greater tendency to act on the perikarya of GnRH neurons in the POA, thereby promoting GnRH synthesis. However, this differs from the results shown by Hiney et al. [38], who collected ME from rats, incubated them with IGF2 and found that the concentration of GnRH increased in the medium. This discrepancy may be due to the different rodent species, time of tissue collection, and study design. The levels of GnRH and LH exhibit strong fluctuations. Additionally, in experiments involving the separation of the MBH from other parts of the rat brain, peaks in LH were not observed [6]. However, in experiments so called “POA deafferentation”, adult rats were still able to ovulate [7]. The study by Barr and Barraclough [39] also found that on the day of estrus in rats, when peripheral blood LH peaks, GnRH in the MBH might be at a low level. We observed a similar phenomenon in our experiment. We believe this might be related to the following points: in our cell experiments, IGF2 also promotes the release of GnRH. Due to the rapid release of GnRH in the ME, this may result in no change in GnRH protein expression in the MBH. Secondly, the regulatory effects of IGF2 on the POA and MBH might differ in strength and duration, with a potentially shorter effect time on the MBH. Therefore, we may have missed the time point when GnRH levels increased. Due to the limitations of our study’s duration, we cannot fully clarify IGF2’s effects on the MBH. The lack of observed increase in GnRH protein expression in the MBH does not mean that IGF2 does not regulate it; a longer-term observational study may be necessary. Our in vivo experiments are still preliminary and can only suggest a promotional but temporary effect of IGF2 on GnRH synthesis. Whether it can activate the hypothalamic-pituitary-gonadal (HPG) axis requires more evidence.
Our study revealed that injecting IGF2 during the prepubertal period increases the synthesis of GnRH in the hypothalamus of mice, promoting the secretion of LH in peripheral blood. Therefore, we believe that IGF2 may not only play a role in the development of the gonads during the fetal stages, as reported in previous studies, but may also have a regulatory effect on GnRH during the pubertal period [16, 17, 18]. However, this effect may occur centrally in the nervous system, making detection in the peripheral blood challenging. Alternatively, this regulatory effect may only occur under pathological conditions, with the regulatory impact potentially overshadowed by the control exerted by IGF1 under physiological conditions. However, our research only demonstrates that IGF2 plays a certain regulatory role in the synthesis of GnRH, but it still cannot prove that IGF2 can promote pubertal development. This requires a more extended study, along with evidence showing that IGF2 accelerates the development of the reproductive glands during puberty.
Moreover, in this study, researchers mentioned that mice with GnRH defects exhibit reduced levels of IGF2 [40]. We observed an increase in IGF2 levels. This regulatory effect may not necessarily be unidirectional, because GnRH levels can influence IGF2 levels. Our experiments focused on verifying the regulatory effect of IGF2 on GnRH, without investigating the reciprocal regulation of GnRH on IGF2. GnRH may also regulate IGF2.
IGF2 binds to the IGF1R receptor, inducing phosphorylation changes in the receptor and regulating gene expression within the cells. To some extent, this molecular mechanism overlaps with the action of IGF1 [41]. Elevated IGF2 levels in tumor-related studies can lead to overexpression of the phosphoinositide 3-kinase/protein kinase B (PI3K/AKT) and mitogen-activated protein kinases/extracellular signal-regulated kinase (MAPK/ERK) pathways associated with cellular energy metabolism [42]. These pathways have been verified to be involved in the synthesis and secretion of GnRH in GnRH cells [43]. Additionally, IGF2R, a specific receptor for IGF2, appears to have a stronger correlation with the clearance of IGF2. Although our study did not identify changes related to IGF2R receptors, the limited sampling time prevented us from conclusively stating that the expression levels of IGF2R and IGF2 are unrelated. Thus, the IGF2R may also play a role in precocious puberty models. Furthermore, as an imprinted gene, IGF2 possesses characteristics associated with other imprinted genes. Mutations inherited from the father in the IGF2 gene can lead to Beckwith-Wiedemann Syndrome (BWS) [44]. The relationship between the characteristics of IGF2 imprinted genes and reproductive development requires further investigation.
Numerous regulatory molecules collectively form a complex regulatory network upstream of GnRH, including pivotal players such as kisspeptin [45]. In our study, we investigated only the regulatory effects of IGF2 on GnRH. We have formulated the hypothesis that not only is the cellular mechanism of IGF2 on GnRH neurons worthy of further exploration, but the role of IGF2 in the regulatory network of GnRH and its interactions with other regulatory molecules requires further investigation.
IGF2 may be involved in the regulatory mechanism of GnRH secretion during the pubertal period. In the precocious puberty model, a synchronous elevation of IGF2 and GnRH in the hypothalamus can be detected. IGF2 may increase the release of hypothalamic GnRH by directly regulating its synthesis in the hypothalamic POA, thereby activating the HPG axis. The effect of IGF2 on reproductive development may not be confined to physiological conditions or the fetal period.
All data reported in this paper will also be shared by the lead contact upon request.
YHW and XLD designed the research study. XLD performed the research and writing the original draft. YHW reviewed the manuscript. YYH and XHH help and advice on the methodology and data curation. JY and WS help and advice on the design of the research study. YTL help to perform the research. WS and YTL help and advice on the writing of manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The research protocol was approved by the Ethics Committee of Children’s Hospital of Fudan University ([2020] 211).
We gratefully acknowledge the assistance and instruction from professor Mellon of the University of California and professor Tao Feng of the Beijing Academy of Agriculture and Forestry Sciences for sharing the GT1-7 cells line.
This research was supported by grants from the National Natural Science Foundation of China (grant numbers 81273804 and 82104932).
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/j.jin2311208.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.






