1 Canadian Centre for Behavioural Neuroscience, Department of Neuroscience, University of Lethbridge, Lethbridge, AB T1K 3M4, Canada
2 Department of Psychology, Trent University, Peterborough, ON K9L 0G2, Canada
3 Regeron Pharmaceuticals, Bronx, NY 10591, USA
Abstract
The goal of these experiments was to determine which learning and memory system(s) were necessary for the retention of visual discriminations and subsequent acquisition of a second problem. The dorsal striatum should be involved in the acquisition and expression of this task based on previous work implicating this region in instrumental learning and memory processes. The perirhinal cortex has been implicated in learning and memory processes associated with visual information like objects, and pictures and may also play a role in the acquisition and/or retention of visual discriminations. As there is no clear spatial/relational component to the task, the hippocampus should not be involved.
Rats were trained on a two-choice visual discrimination task to criterion performance after which they received lesions to portions of the dorsal striatum (dorso-medial or dorso-lateral striatum) and medial temporal lobe (perirhinal cortex or hippocampus). After surgical recovery, the rats were tested for retention of the original discrimination, followed by training on a second problem on the same task.
The results showed that dorsal medial striatal lesions produced a retrograde deficit on picture discrimination, but dorsal lateral striatum lesions did not. Neither dorsal striatal lesion produced a deficit on acquisition of a second problem. Perirhinal cortex did not seem to make an essential contribution to the retention of the original discrimination or acquisition of the second problem. Surprisingly, subjects with hippocampal damage were severely impaired but eventually re-learned the discrimination. Damage to the hippocampus had no impact on acquisition of a second problem.
Taken together, the results of the present experiments show that the dorsomedial striatum and the hippocampus may support performance on this instrumental task if intact during acquisition but is not required for acquisition of a new problem. The implications of this pattern of results for our understanding of the organization of learning and memory in mammals is discussed.
Keywords
- instrumental learning
- visual discrimination
- Grice box
- hippocampus
- dorsal striatum
- perirhinal cortex
- multiple memory systems
- retrograde amnesia
- anterograde amnesia
The organization of learning and memory in the mammalian brain seems to consist of functionally and anatomical distinct systems [1]. Each of these systems has a central structure but is connected and interacts with a unique array of cortical and subcortical sites [1]. The central structures of these different systems include the amygdala, dorsal striatum, hippocampus, and perirhinal cortex. The amygdala and its related circuits have been repeatedly shown to be involved in both appetitive and aversive forms of classical conditioning [2, 3, 4, 5]. The dorsal striatum and related circuits have been implicated in different forms of instrumental learning [6]. The hippocampus and related circuits have been implicated in certain forms of context, spatial, and relational [7, 8, 9] learning and memory functions. The perirhinal cortex and related circuits are involved in perceptual and cognitive functions associated with visual and object recognition [10, 11].
The focus of the present study was to elucidate which of these learning and memory systems is involved in a simple visual discrimination task using pictures displayed on computer monitors. This task requires rats to swim to a hidden platform that is submerged in front of computer monitors that display the rewarded (S+) picture in a modified Grice box. Previous work using this paradigm have employed different types of recognition memory tasks like delayed matching to sample and simple discriminations demonstrated that both the hippocampus and perirhinal cortex make essential contributions.
A cursory analysis of this learning paradigm is that this is an instrumental task in which the subject learns to make a voluntary response (swim) towards a reinforced cue and learns to avoid a non-reinforced cue [12]. Based on this analysis, rats with damage to the dorso-medial striatum (DMS) and/or dorso-lateral striatum (DLS) should be impaired at the acquisition and/or expression of this task since both components of the dorsal striatum have been shown to be critical for the acquisition of different forms of instrumental learning [6].
Specifically, empirical and theoretical evidence suggests that DMS is important for cognitive control of instrumental behaviours while the DLS is essential for the acquisition and retention of stimulus-response habitual forms of instrumental learning [13]. A different line of evidence shows that the DMS is critical for the acquisition and retention of action-outcome associations which are thought to support the early stages of instrumental behaviours while the DLS has been shown to be essential for the acquisition and retention of habitual forms of instrumental learning in the later stages of conditioning [14]. Similarly, the perirhinal cortex (PRh) has been implicated in the acquisition of object recognition as well as simple visual discriminations using similar procedures and technology. The PRh might be seen as essential for the visual aspects of this learning task and portions of the dorsal striatum for the instrumental responding component.
The hippocampus (HPC) has been implicated in learning and memory processes largely in the spatial domain as well as other relational forms of learning [15]. The HPC is not usually thought to be required for simple discriminations or instrumental tasks [6].
The present experiments evaluated the effects of damage to different components of the dorsal striatum (DMS or DLS) as well as the PRh and HPC. The former regions have been implicated in various forms of instrumental learning and the latter in visually-based perceptual and cognitive processes. Based on previous work, the predictions are that damage to DMS and/or DLS will impair acquisition and retention of this instrumental version of a visual discrimination task. PRh and HPC dysfunction will have little or no effect on acquisition but may produce deficits in the retrograde direction.
The study was conducted in accordance with the Guidelines of the Canadian Council on Animal Care, and the protocol was approved by The University of Lethbridge Animal Welfare Ethics Committee (approval number: 0508).
Sixty male Long-Evans rats (University of Lethbridge colony; 350–450 g) were pair-housed in standard laboratory cages, kept on a 12:12 light-dark cycle (lights on at 07:30), and provided with food and water ad libitum. Each cage consisted of corncob bedding and had a black tube and shredded paper for enrichment. Environmental conditions in the rat colony room were held at a constant temperature of 21 °C, with 35% humidity. All rats were handled for 10 minutes a day for 5 days before the start of behavioural training.
The visual water task (see for specific details) was used to train rats on picture discriminations (Fig. 1). The visual water task is composed of a trapezoidal shaped metal pool of water measuring 17.5 cm in depth, with a hidden platform of 14 cm in height located at one end of the pool. The end wall of the tank was transparent. On one end of the trapezoid was a 45.7 cm long barrier that divided the end in half to create two arms for the rat to swim into with each half being 40.6 cm in width. On each side of the barrier, displayed through the transparent wall, were two 17-inch flat CRT Sony computer monitors (SONY, Tokyo, Japan) showing a black and white picture, one rewarded stimulus and one unrewarded stimulus 30.5 by 30.5 cm. Each picture stimulus displayed on the monitors had a near equal amount of luminance. The pictures used for the present experiments are shown in Fig. 2. The software used to manipulate different pictures and side location was developed by.
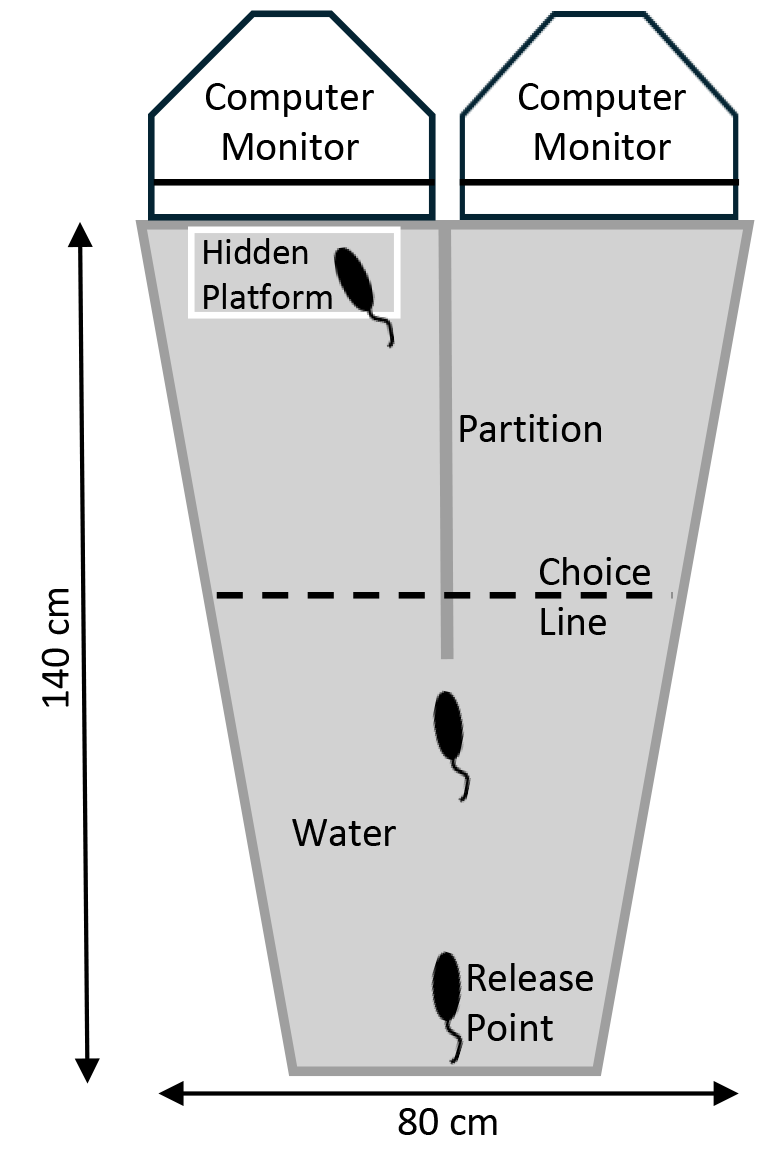 Fig. 1.
Fig. 1.
An overhead view of the Grice Box task. For this task, each rat was placed in the apparatus at the release point facing towards the computer monitors. The subjects were given 10 trials per day and had to associate one visual cue with a hidden platform for escape from the water, from another cue which was not reinforced. Once trained, the rats would typically swim to the middle partition and then decide which side to swim down. The choice line represents the point at which if the rat swam past was considered an error if it selected the wrong side.
 Fig. 2.
Fig. 2.
The visual cues that were associated with a platform or no platform. The top panel represents the cues used in problem 1 and the bottom panel are the cues used for problem 2.
For training, the 10-point star image was paired with the escape platform, while
the pill shape image was not paired with an escape platform. The location of the
images (left or right) and hence escape platform, was chosen using an ABBABA
sequence. Each trial started with the experimenter placing the rat into the Grice
box at the opposite end of the screens facing the two images used for the
discrimination and pressing the start button on the computer keyboard. Typically,
a rat would swim to the middle partition and then make a choice to go to the left
or right. For correct performance, the rats were required to approach the screen
which was reinforced with the submerged escape platform in front of the screen.
Errors were recorded when the rat swam 8 cm beyond the middle partition (choice
line) toward the non-reinforced screen, or if it initially went toward the
reinforced screen but turned around before reaching the platform and then swam to
the non-reinforced screen. The rat remained in the apparatus until it located the
hidden platform and then was immediately removed after doing so. The experimenter
would then stop the trial on the computer by indicating which side the rat
initially swam to. The experimenter was careful to watch and quickly correct for
a rat showing a swimming bias to one side (e.g., initially swimming to the left
side more than 4 times in a row) by setting the sequence of reinforcement to be
disproportionately opposite of the swim bias. Once the bias was corrected, the
regular reinforcement sequence was reinstated. Rats were given 10 trials per day
with an average inter-trial interval of 2 min. and were placed into a Plexiglas
holding cage in between trials. After completing the training trials, the rat was
returned to its home cage. Daily percent correct scores were calculated by
dividing the number of correct choices by the number of trials run for the day
for each subject. This was then used to make the group averages and
General surgical procedures were consistent throughout each experiment although the injections parameters and lesion technique varied among the groups. Surgeries were performed approximately 24 hr after the last day of behavioural training. Thirty minutes prior to the surgical procedure, the rats in Experiments 1 and 2 were administered Buprenorphine (0.03 mg/kg, Cat#137898 (WDDC) Vetergesic, Ceva Animal Health Inc., Cambridge, ON, Canada) subcutaneously as an analgesic. Surgery was conducted while rats were anesthetized with Isoflurane (4% with 2 L/min of oxygen for induction and 2% after surgical plane was established; Cat# 124125 (WDDC), Fresenius Kabi, Lake Zurich, IL, USA) in a standard stereotaxic apparatus. Once under anaesthesia, the hair from the top of the rat’s head was shaved and the skin cleaned with stanhexadine and alcohol. An incision was made in the scalp and periosteum along the midline. The fascia (periosteum) was cut laterally across the top of the skull and pushed to the edges of the surgical site with a sterile gauze swab. The skin was retracted with 2 mosquito forceps to expose the skull surface and trephining holes were drilled into the skull over each brain structure of interest. All infusions were done sequentially through a 30-gauge injection needle attached to a 10 µL Hamilton syringe (Model 701N, Cemented Needle, 32 gauge, 2 in., point style 3, Hamilton Company, Reno, NV, USA) via polyethylene tubing (PE-20). The specific lesion parameters used can be found in the methods sections for each experiment and all coordinates were in relation to bregma, the midsagittal suture, and skull surface.
The different lesion techniques for each brain region were selected for the following reasons. The quinolinic acid lesion was selected for the dorsal striatal regions because it has been shown to be the most effective in damaging neurons in that region versus other neurotoxins like N-methyl-D-asparate (NMDA). NMDA lesions have been shown to be very effective in damaging neurons in the HPC and it is much more affordable than others, like ibotenic acid, and thus was selected. Finally, we used an electrolytic approach to damage the perirhinal cortex as this region is difficult to damage with neurotoxins because of spread of the toxin to adjacent brain region issues. For the latter, if a deficit was reported the results would have been difficult to interpret as both neurons in perirhinal cortex as well as fibers of passage coursing through this cortical region could contribute to the deficit, but because no deficit was found we are confident with the veracity of the finding.
Following the lesions, the scalp incision was closed using suturing thread. All subjects were given a post-surgical subcutaneous injection of Metacam (1 mg/kg; Cat#114424 (WDDC), Boehringer Ingelheim, Burlington, ON, Canada) for pain relief and to reduce inflammation. Rats were given a post-surgical intraperitoneal (i.p.) injection of Diazepam (5 mg/kg; Cat# 127045 (WDDC), Sandoz Canada, Boucherville, QC, Canada) to control seizure activity. Additional doses of Diazepam were administered as needed if there were overt signs of seizure activity during surgical recovery. The same surgical procedures were used for the Sham rats except that no damage was done to their brain, and instead of drilled holes, bone scoring was etched across the surface of the skull. The rats were allowed to recover for a minimum of 7 days before behavioural testing.
Upon completion of behavioural testing, all rats were sacrificed by receiving an i.p. overdose of sodium pentobarbital (320 mg/kg; Euthanyl Cat# 127189 (WDDC), Bimeda-MTC Animal Health Inc., Cambridge, ON, Canada) and were perfused transcardially with 0.1 M PBS followed by 4% paraformaldehyde. Their brains were excised and stored in a 4% paraformaldehyde solution for 24 hours and then transferred to a 30% sucrose PBS solution. Later, frozen brains were sectioned coronally at 40 µm Every fifth section was mounted on gelatin-coated slides (1% gel) and stained with cresyl violet. The stained sections were examined through a light microscope (Leica, Wetzlar, Germany) to examine the extent of the lesions.
This experiment assessed the effects of neurotoxic lesions of the DMS or DLS on retention and re-acquisition of picture discriminations. We also tested the effects of these lesions on the acquisition of a new picture set problem on the same training apparatus. These regions were selected because they have been implicated in instrumental learning and memory processes and should be required for the retention of this task.
Rats were pre-trained on the task until reaching asymptotic performance (see general methods). Based on behavioural performance, rats were divided into three surgical groups (SHAM, DMS, DLS).
Quinolinic acid (2,3-Pyridinedicarboxylic acid, Cat# P63204, Sigma, Oakville,
ON, Canada) was dissolved in 0.1 M phosphate-buffered saline to a concentration
of 30 mg/mL and titrated to pH 7.4 with sodium hydroxide. Quinolinic acid was
infused at 0.175 µL/min over 1 min and 20 s per site and was then
left to diffuse for 2 min. Eight rats received neurotoxic DLS lesions. The
coordinates of the infusion sites (mm) were (anterior/posterior, AP) 1.6, 0.5, –0.8, (medial/lateral, ML
Twenty-four rats were trained on the picture discrimination task and their
performance can be seen in Fig. 3A. For illustrative and statistical analysis,
the rats were separated into their pre-DLS, pre-DMS, or pre-SHAM groups. Rats
showed a slow acquisition curve of learning to pair the 10-point star with the
escape platform. The rats showed the typical behaviours previously reported of
scrabbling against the wall, attempting to jump out of the apparatus, and holding
on to the middle partition before eventually swimming directly to the end with
the picture. As training progressed the rats’ performance improved and they would
swim directly to the middle partition and look at both pictures before choosing
to swim towards one of them, or they would swim directly to the picture. An ANOVA
with repeated measures indicated a significant effect of Day [F(23,483) =
18.34, p
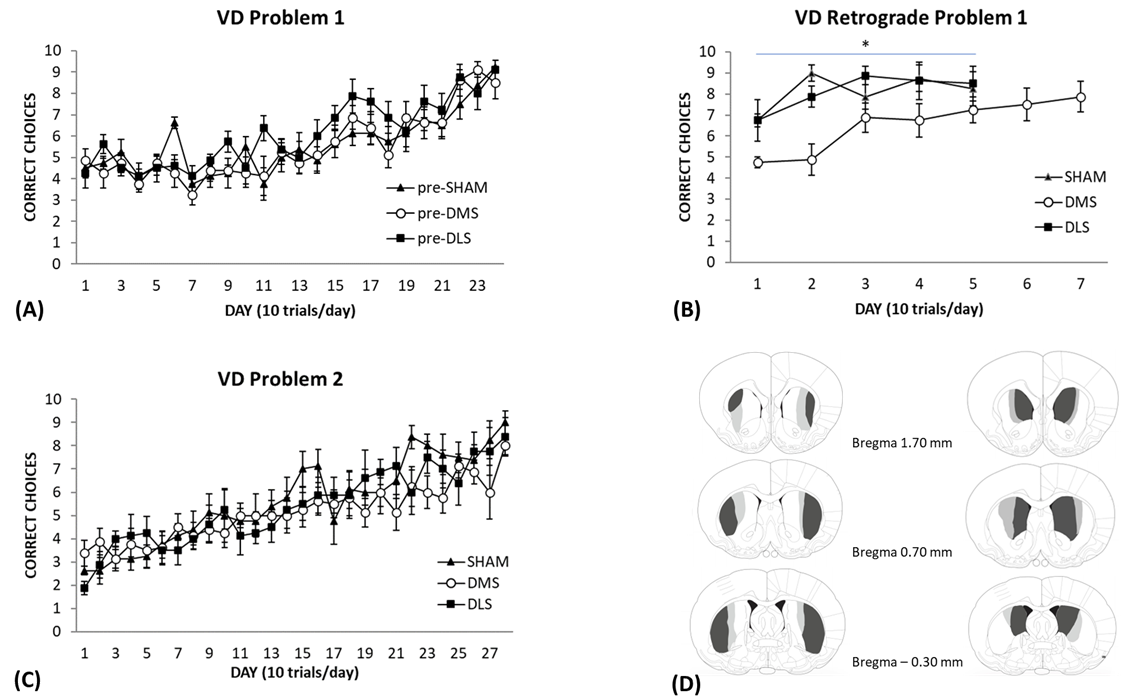 Fig. 3.
Fig. 3.
Visual Discrimination (VD) performance in rats with SHAM,
DMS or DLS lesions. (A) Rats were trained on the visual discrimination to
problem set 1. Each day represents an average of 10 trials (
Retention
A week following surgery, the rats were placed back into the apparatus to assess
their retention of the original picture discrimination. As displayed in Fig. 3B,
the SHAM and DLS lesion groups showed a brief decrease in their choice accuracy
performance. The rats that had DMS lesions showed a significant decline in
performance on the first couple of days of retention testing, but their
performance improved thereafter. An ANOVA with repeated measures revealed
significant effects of Group [F(2,21) = 6.20, p = 0.008] and Day
[F(4,84) = 7.36, p
The rats were then trained on a second problem set using novel pictures as the
stimuli to ascertain whether a lesion to the DLS or DMS would produce a different
pattern of performance. The learning curve of the second problem set can be seen
in Fig. 3C. In the beginning of training, the rats’ scores were below 50% which
suggests that the new pictures were saliently different from the original
pictures. Furthermore, although the rats knew the procedural elements of the task
(ie/swimming towards one of the pictures to find the escape platform), it still
took them approximately the same amount of time to acquire the new problem. The
results of the ANOVA showed a significant effect of Day [F(27,567) = 15.64,
p
Histology
Of the Sixteen rats that underwent DLS or DMS surgery, none were removed from the study. The location and extent of the smallest and largest DLS and DMS lesions are shown in Fig. 3D. The lesion extent was very similar to our previous work with almost all subjects having a large lesion and one in each group having a slightly smaller lesion. All lesions included both the anterior and posterior portions of the structure (Fig. 3D (left side)). Two lesions in this group were slightly larger on one side and two lesions showed minor cortical thinning unilaterally. The DMS lesions showed extensive neuronal damage to the medial portions of the striatum and large expansions of the lateral ventricles. Two lesions in this group were larger on one side and another showed unilateral cortical thinning. Representations of the lesions are shown in Fig. 3D (right side). The light grey and dark grey areas represent the largest lesion, and the dark grey area represents the smallest lesion.
The goal of this experiment was to assess the effects of electrolytic lesions of the PRh on learning and remembering a visual discrimination task. We also tested the effects of these lesions on the acquisition of a new problem on the same task. This region was selected because there is a significant body of research implicating it in visual learning and memory processing.
The procedures were identical to those used in Experiment 1, except that rats were divided into two groups (PRh and SHAM) based on acquisition performance.
Rats received electrolytic lesions of the perirhinal cortex (PRh, n =
10), or sham surgery (SHAM, n = 8). A scalp incision was made, and the
muscle overlying the temporal skull was displaced. A portion of skull overlying
the target area was removed using a hand-held drill. A bipolar stainless-steel
electrode insulated with Teflon except for approximately 1 mm at the tip was used
to deliver the electric current (1.5 mA for 10 s). The electrode was angled at
10° to the vertical plane at five sites per hemisphere through the PRh.
The coordinates (mm) were (AP) –3.5, –4.5, –5.5, –6.5, –7.5; (ML
Eighteen rats were trained on the picture discrimination task as previously
described. The learning curve for the rats, divided into their pre-SHAM or
pre-PRh groups, can be seen in Fig. 4A. The rats that were subsequently
eliminated following histological verification of lesion target were not included
in this descriptive or statistical analysis, and therefore the PRh group had
n = 7, and the SHAM group n = 8. The results indicated a
significant effect of Day [F(23,299) = 9.54, p
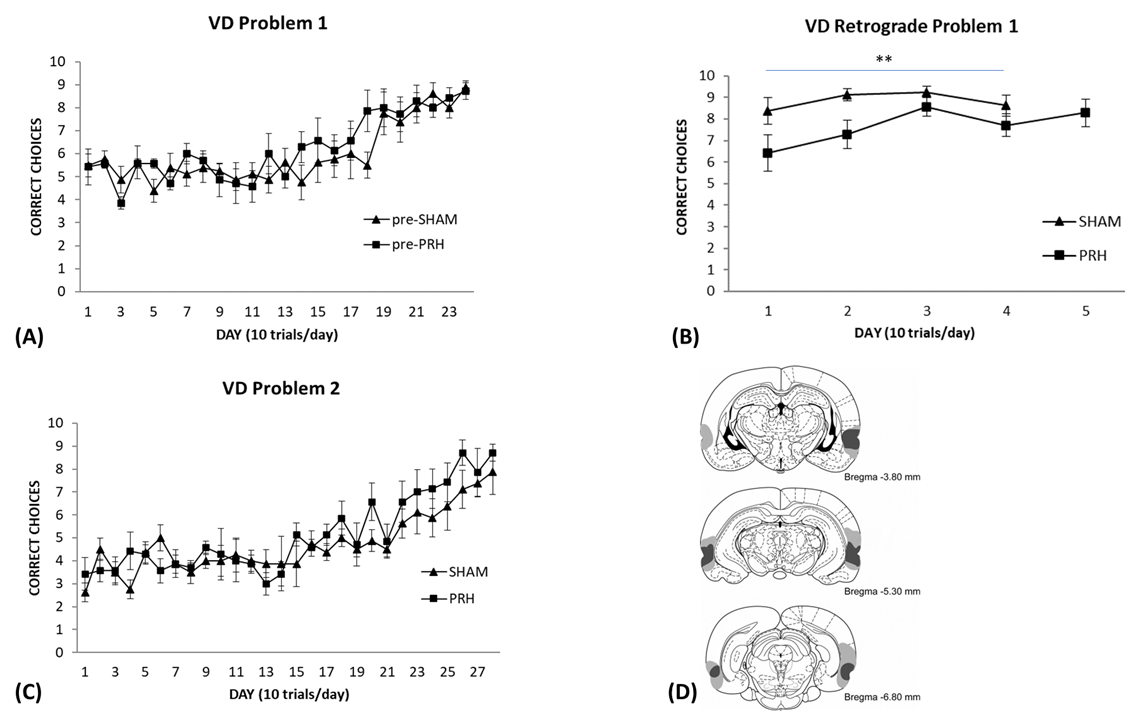 Fig. 4.
Fig. 4.
Visual Discrimination performance in rats with SHAM or PRH
lesions. (A) Rats were trained on the visual discrimination to problem set 1.
Each day represents an average of 10 trials (
Retention
As can be seen in Fig. 4B, the SHAM group performed better than the PRh group during the retention test of the original picture discrimination. An ANOVA with repeated measures performed on the first four days of retention testing revealed significant effects of Group [F(1,13) = 9.36, p = 0.009] and Day [F(3,39) = 3.03, p = 0.04], but no interaction [F(3,39) = 0.82, p = 0.49]. Although the PRh group were initially impaired compared to controls, their discrimination choices did improve quite rapidly compared to how long it took them to learn the discrimination initially, suggesting that they still had the representation available to them.
These groups were then trained on a second picture set and their performance is
illustrated in Fig. 4C. Both groups swam to the middle partition and looked at
each picture before swimming towards one of them; a strategy that was used while
they were learning the first picture discrimination. As can be seen, the groups
showed a slow and gradual increase in the number of correct choices they made. In
fact, the PRh group were a bit better than the SHAM group towards the end of
training, although not significantly. An ANOVA indicated a significant effect of
Day [F(27,351) = 10.79, p
Histology
Of the eighteen rats that underwent surgery, three were eliminated due to incomplete PRh lesion. The location and extent of the smallest (enclosed dark grey area) and largest (dark grey and light grey area) PRh lesions are shown in Fig. 4D. One PRh rat was excluded from the experiment because it had extensive damage outside the target area and two other rats were excluded because PRh damage was only found in one hemisphere. All rats remaining in the PRh group sustained substantial and nearly complete, bilateral damage to the PRh. However, 1 rat had sparing in the anterior region of the PRh in one hemisphere. Minor bilateral damage to the lateral entorhinal cortex was also found in all PRh rats. Damage to the anterior portion of the postrhinal cortex occurred in 3 rats bilaterally and in 1 rat unilaterally. In 4 rats, bilateral damage was found in the temporal association cortex and minimal damage was found unilaterally in 2 rats. The lateral amygdala was slightly damaged unilaterally in 1 rat. No damage was found in the hippocampus.
This experiment assessed the effects of neurotoxic lesions of the HPC on learning and remembering a visual discrimination task. We also tested the effects of these lesions on the acquisition of a new problem on the same training apparatus. This region was of interest because it is not supposed to be involved in simple instrumental learning tasks but some work using a variant of this task reported impaired retention, normal re-acquisition rates, and normal acquisition of a new problem following HPC damage [16].
The behavioural training procedures were identical to those used in Experiment 1, except that rats were divided into two groups (HPC and SHAM) based on behavioural performance.
One hour prior to surgery, all rats were given Phenobaritol i.p. as an
anticonvulsant (30 mg/kg; Cat #115189, Sandoz, Basel, Switzerland). The
hippocampal lesions were produced by 7 bilateral intracranial microinfusions of
NMDA (7.5 mg/mL; Cat# M3262, Sigma, Oakville, ON, Canada) dissolved in 0.1 M
phosphate buffered saline (PBS). The coordinates (mm) for the hippocampal lesion
were: (AP) –3.1, –4.1, –5, –5, –5.8, –5.8, –5.8; (ML
Eighteen rats were trained on the picture discrimination task as described
previously. Fig. 5A displays the rats’ performance on the task separated into
their pre-SHAM or pre-HPC groups. One rat was subsequently eliminated following
histological verification of lesion target and is not included in this
descriptive or statistical analysis, and therefore the HPC group had n =
9, and the SHAM group n = 8. As can be seen both groups learned to swim
towards the positive visual stimuli indicating the escape platform in the same
amount of training. Therefore, any differences observed during the retention test
of problem set one was not due to differences in their original learning. The
results indicated a significant Day effect [F(20,300) = 17.77, p
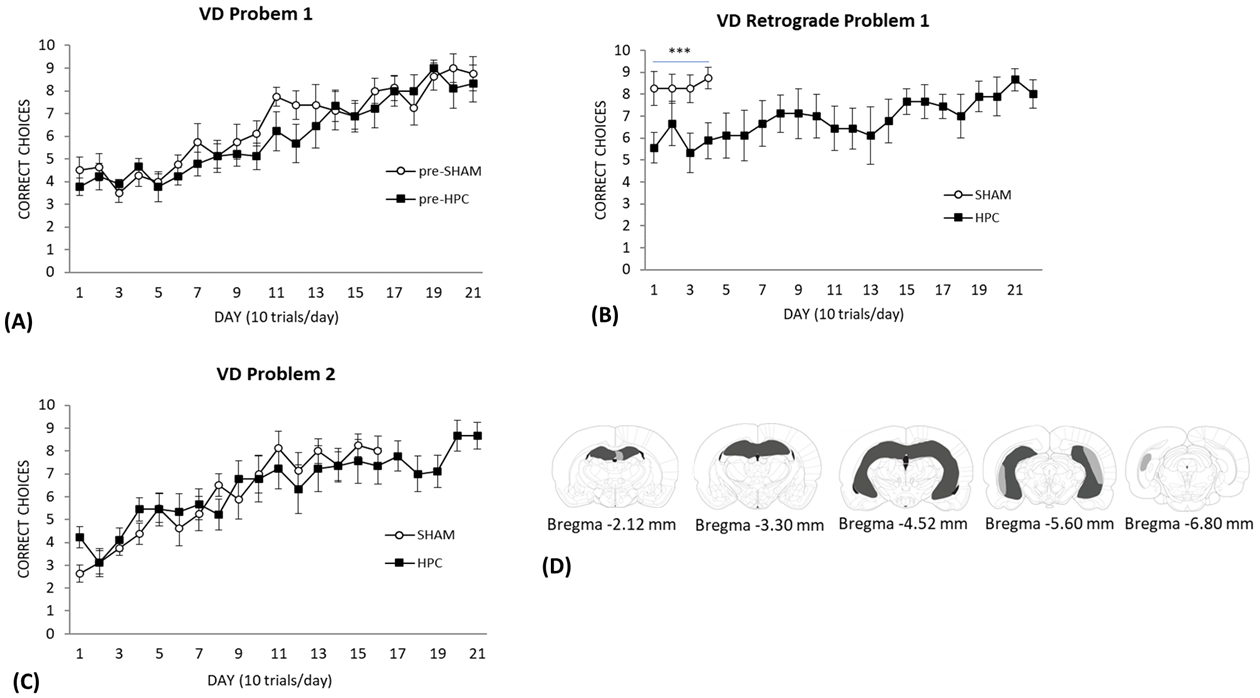 Fig. 5.
Fig. 5.
Visual Discrimination performance in rats with SHAM or HPC
lesions. (A) The results of the visual discrimination training to problem set 1.
Each day represents an average of 10 trials (
Retention
As can be seen in Fig. 5B, the SHAM control group performed better than the HPC
group during the retention test of the original picture discrimination. An ANOVA
with repeated measures performed on the first 4 days of retention testing
confirmed a significant effect of Group [F(1,15) = 9.33, p
These groups were then trained on a second picture set and their performance is
illustrated in Fig. 5C. As can be seen, both groups showed a slow and gradual
increase in the number of correct choices made and their learning curve was not
different from each other. An ANOVA performed on the first 16 days needed by the
SHAM group to reach criterion indicated a significant effect of Day
[F(5.806,87.095) = 14.89, p
Histology
Of the ten rats that underwent complete HPC lesion (dorsal and ventral) surgery, one rat was excluded due to extensive overlying cortical damage and thalamic damage. Fig. 5D shows the smallest (enclosed dark grey area), and largest (dark grey and light grey area) hippocampal lesion included in the study. The lesions included both anterior and posterior portions of the hippocampus and were large. Some minor sparing of neurons was found in some of the specimens. Five hippocampal lesions showed minor sparing of the dentate gyrus, CA3, and CA1 regions in the most anterior regions of the dorsal hippocampus. 4 of these brains showed bilateral sparing and one showed unilateral sparing. However, in all cases most of the dorsal hippocampal neurons were absent. In all the specimens, neurons in the intermediate and ventral regions were largely destroyed. Some minor sparing of neurons was found in some of the specimens in the most posterior ventral hippocampal region. Five specimens showed bilateral sparing of CA1 and CA3 and two showed unilateral sparing in this most posterior region of hippocampus proper. One specimen showed unilateral cortical thinning at one of the injection sites in the ventral portions of the brain. None of the brains showed any signs of neuronal death in the subicular, entorhinal, or perirhinal cortical regions.
The present experiments investigated the contributions of different neural systems implicated in learning and memory functions to the retention, re-acquisition and acquisition of an instrumental visual discrimination task.
The pattern of results indicated that damage to the DLS had no effect on retention of this instrumental task or on acquisition of a second problem. In contrast, damage to the DMS following learning of the first problem produced an impairment on retention, but had no effect on acquisition of the second problem. The results also showed that the PRh made little or no contribution to the retention of the task or in acquisition of a second problem. Finally, rats with damage to the HPC showed severe impairments in the retention of the first problem but showed no impairments in the acquisition of a new problem. This pattern of deficits following damage to portions of the dorsal striatum or components of the medial temporal lobe (PRh, HPC) were not predicted and a discussion of why this pattern of effects was found is discussed below.
Previous empirical and theoretical work shows a critical role for these brain regions in instrumental learning [1]. The DMS has been implicated in cognitive control of behaviour [13] as well as action-outcome associations in which the subject learns that a particular instrumental or voluntary response results in a particular outcome (i.e., reward). This association is thought to guide accurate responding early in instrumental training [17]. The DLS has been implicated in stimulus-response associative learning [6] and/or habitual forms of instrumental learning [18]. The former, involves associations between neutral stimuli and specific voluntary responses that are repeatedly reinforced. The latter is a form of instrumental learning that occurs later in training following a lot of reinforced trials in which the responding becomes insensitive to outcome devaluation, although the term “habit” has been used in both formulations.
In the present experiments, damage to two components of the dorsal striatum had different effects on retention of the picture discrimination task, disruption of the DMS impaired and DLS had no effect on retention performance. Damage to neither region impaired acquisition of the second problem on the task. These effects were surprising as it was predicted that either the DMS or DLS would produce some kind of impairment on this task. This prediction was made because the task used in these experiments fits the description of an instrumental task and should have resulted in impairments in groups of rats with damage to these striatal regions. The task requires the subject to swim (instrumental response) to a reinforced visual cue (stimulus) which results in a positive outcome (escape from water).
One way to try to understand the present results is to compare the predictions made by two theories of dorsal striatal function. We predicted a deficit in the anterograde and retrograde direction following DLS damage and no deficit in subjects with DMS damage [1]. Balleine and colleagues [14] would predict that damage to neither region would produce an impairment as the other system can take control if the other is rendered dysfunctional, essentially switching control from the goal-directed to the habit system or vice versa. For the anterograde results, the latter prediction was borne out. However, both theoretical positions had difficulty accounting for the complete pattern of results reported here.
First, we’ll focus on the lack of impairments of the DLS lesion on any component of the visual discrimination task. From our perspective, one explanation of this lack of effect is the nature of this instrumental task. Based on our analysis of the literature, the DLS is required for instrumental tasks in which “The DLS is essential in bringing movements that lead to reinforcement under specific stimulus and temporal control. This is brought about by repeated, reinforced experience in which significant quantitative (accuracy) and qualitative (fine-tuning) improvement of the motor response. These experiences can ultimately lead to discriminative automaticity and motor habits. The mechanism underlying this type of learning would be excitatory, associative conditioning specific to the reinforced cue.” [13]. We have also argued that instrumental tasks with discrete responses are more likely to engage and ultimately require the DLS and related circuits [13, 19] versus tasks with general approach responses required to obtain reinforcement. The water-based visual discrimination task used here requires the subjects to use the same general approach response to both cues. It is possible that a neural network centered on the amygdala [20] could support learning on this task, particularly in the absence of the DLS or DMS as this system supports simple conditioned approach responses to reinforced cues via classical conditioning mechanisms. Similarly, on this task, the subjects are not required to respond to discrete conditional cues at a particular time, the discriminative cues are statically present during the entire trial period and the subject can make a choice. The point we are making here is that subtle differences in task design can impact which learning and memory system is recruited and/or required for high levels of performance [21].
The rats with DMS damage induced following training produced an impairment. This suggests that the task is mediated at least in part by a neural network centered on the DMS and this system is in control of discriminative behaviour when asymptotic performance is reached. However, the anterograde lesion results show that the DMS is not necessary for normal learning to occur if it is absent during acquisition. On the surface this supports the Balleine prediction. However, the retrograde experiment is more problematic for this theoretical position. Their theory would predict that by the end of pre-training, when the subjects are intact, the DLS habit system should be controlling behaviour but it is the rats given retrograde DMS lesions that were impaired suggesting that the task is potentially always under cognitive control [13] or goal-directed representations.
Upon observing the subjects perform this task, it appears that the task remains “cognitive” as there is little automaticity or habitual responding that develops during training. Often, the rats would approach the choice point and hold onto the vertical divider and scan back and forth between the computer screens sampling the cues in a manner like vicarious trial and error learning [22], a pattern of behaviour during learning long associated with cognitive processing and HPC [23]. Furthermore, once the DMS was damaged the DLS should have been able to support discriminative behaviour on the task [17]. One way to interpret our results is that the intact animal uses a goal-directed solution throughout training. However, when the DMS or HPC are rendered dysfunctional in the retrograde direction these subjects can recruit the DLS to support re-learning of this instrumental task. Further work is required to evaluate some of these predictions and discrepancies, including the use of devaluation procedures.
One approach to addressing these issues would be to go back to the data set and look at other outcome measures like response latency and decision-making pauses at the choice point that might indicate impairments in goal-directed behaviour, habit learning, conditioned inhibition, and decision-making processes. A definitive answer to some of the questions raised above are difficult to determine based on the current experiments because the data was recorded manually throughout the training procedure. Thus, we are unable to go back and look at these different response measures. A computer controlled the stimuli presentations but did not collect any choice data. Similarly, no video recording was used either. Future research using this kind of paradigm can look at these different response measures to try and tease apart the different types of behavioural strategies and errors.
Another interesting issue that emerges from our data set can be found during training of the first and then second visual discrimination problem. It is somewhat puzzling that it takes equally long to learn the second task. This is strange because presumably the action-outcome contingency should have already been established in the first problem, so why is there no apparent savings for problem two that has the same action-outcome contingency? Our current hypothesis is that the response-outcome association is not formed independently in the original problem. The representation is likely a stimulus-response-outcome association and during the second problem a new one must be formed from scratch incrementally. To our knowledge there is currently little or no work on these important issues in the literature.
Large lesions of the PRh had little or no effect on the acquisition and expression of the visual discrimination task. These effects were not surprising despite previous work that shows a critical role for this brain region in visually based perceptual abilities and associated cognitive processes based on visual information [24]. The lack of effect of PRh cortex lesions is probably due to the nature of the task used in these experiments.
It seems that several factors determine the sensitivity of a task to PRh dysfunction [10, 11, 25, 26, 27]. One factor is the complexity of the to-be discriminated or remembered visual items or objects. The more similar the visual stimuli are, the more sensitive the task will be to damage to this region or whether the stimuli are 2-or 3D. The second factor is whether there is a delay between the sample and choice items during training and testing. Finally, the PRh memory system seems critical for tasks in which an item is presented and then must be remembered one time, essentially a single episode or event. The task used in the present experiments does not have any of these features and no effects of PRh lesions were found even though the task was a visually based discrimination. However, it is important to note that there are inconsistencies in the literature on these issues including demonstrations of acquisition deficits on a simple two-choice instrumental discrimination like the one used in the present study [28].
Rats with neurotoxic damage to the HPC showed severe impairments in the retrograde direction. However, they could acquire a new problem in the anterograde direction.
These results suggest that the retention of this task is highly dependent on the HPC and related circuits when it is intact during learning [29]. However, if the hippocampus is damaged before acquisition there is no noticeable impairment. This suggests that there are at least two solutions to this simple visual discrimination task. To get an acquisition deficit on this task you might have to disrupt more than one of these central structures of these learning and memory systems. This strategy has been used to some success in several laboratories [30, 31].
One final point concerning HPC and anterograde learning deficits is that the absence of anterograde amnesia is observed in rats with prior experience in the task (second problem). It is possible that naive rats might have a deficit [32]. Further research is required to assess this idea. The retrograde deficits (HPC and DMS) are suggestive of a default representation based on a synergistic interaction between HPC and DMS. This idea is based on the demonstration that damage to either region produces a retrograde deficit on this visual discrimination task and there are clear functional interactions between these two regions on spatial navigation/memory tasks [33].
However, this does not help explain the lack of anterograde impairments following damage to both these regions or why the HPC would be involved in the first place. The reason for this dependency on the HPC is not immediately obvious. Rats with HPC damage can acquire most simple discrimination tasks or even show enhanced acquisition (for review see [1]). Retrograde lesions of the HPC, in many instances, produce impairments in simple discrimination tasks [16] suggesting that the HPC might encode critical contextual information for task retention.
Overall, the pattern of effects suggests that the default representation supporting learning on this task is mediated by the HPC and DMS but in their absence another system can compensate. One theory may be helpful in this instance. The “heterarchic theory” of the organization of learning and memory in the mammal suggests that the HPC sits at the top of a heterarchy of multiple learning and memory networks. According to the theory, if the HPC is intact it is always involved in learning and memory processes particularly early in training [29]. This theory has some difficulty in accounting for the deficits because some of the factors that should lessen the dependence on the HPC posited in the theory are part of the visual discrimination task, used in this study, including repeated training sessions and a lack of pattern separation/completion component to the task [34].
Another potential explanation for the dependence of this task on HPC function in the retrograde direction is that success on this discrimination might be highly dependent on conditioned inhibition processes linked to the ventral HPC. From our point of view, the acquisition of simple discriminations is interesting because asymptotic performance is guided by excitatory conditioning (pavlovian and instrumental) to the reinforced cue but also by inhibitory conditioning that accrues to the non-reinforced cue. This conditioned inhibition seems to be mediated by a neural circuit that includes the ventral HPC and potentially portions of the ventral striatum and is context specific [31, 35, 36]. Removal of the HPC following training on the visual discrimination would abolish the influence of this conditioned inhibition representation and potentially produce a retrograde impairment. It is also possible that performance of this task might be more dependent on inhibitory conditioning to the non-reinforced cue. Further research is required to assess the contributions of excitatory versus inhibitory conditioning on this task.
A final point, during the initial days of retention testing/re-acquisition we observed that HPC lesioned rats tended to immediately swim along the walls of the apparatus towards the discriminative cues and didn’t go to the middle partition before deciding where to go. This suggests that the cognitive processes associated with successful discriminative behaviour were absent in the HPC lesioned rats.
Any experimental brain manipulation that causes a deficit on a learning task can be because the manipulation is impairing learning and memory processes or it is affecting supports for learning and memory like sensory, motor, and motivation. This has been an important aspect of our research program for decades. One reason we believe this is not the case in the present experiments is that none of the lesions impacted acquisition of the tasks. As acquisition would require sensory, motor, and motivational supports, this pattern of results suggests that the lesions did not impact those functions. Further, we have invested a significant amount of time and resources showing the specificity of the learning and memory effects following these kinds of lesions by designing experiments that exclude these other interpretations of lesion/behavioural work. These approaches include double and triple dissociations [37] in which the sensory, motor, and motivational aspects of the task are the same, but the mnemonic requirements are different. We have also modified instrumental tasks to reduce the motor demands and ensure equal reinforcement experiences in work assessing the contributions of the different regions of the dorsal striatum to stimulus-response learning [19]. Even under these conditions, rats with neurotoxic lesions of the dorso-lateral striatum, but not dorso-medial striatum, were impaired at a stimulus-response task.
The role of various brain regions implicated in learning and memory processes in the retention, re-acquisition, and acquisition of a visually-based instrumental task was assessed. Rats with lesions of the DLS, DMS, PRh, and HPC were tested. The results showed that none of these brain regions, on their own, are essential for the acquisition of this task. Retrograde lesions of the DMS and HPC resulted in retention and reacquisition deficits on the task suggesting a role of these regions when they are intact during acquisition. The implications of this work for our understanding of the organization of memory in the mammal are significant.
Data is available upon reasonable request to the corresponding author.
RM (Conceptualization; Methodology; Validation; Resources; Writing-Original Draft; Writing – Review & Editing; Supervision; Project administration; Funding acquisition); JK (Validation; Investigation; Data Curation); EZ (Validation; Investigation; Data Curation, Writing – Review & Editing); RS (Conceptualization; Writing – Review & Editing); HL (Validation; Investigation; Data Curation, Writing – Review & Editing); FS (Validation; Investigation; Data Curation, Writing – Review & Editing); NH (Methodology; Validation; Investigation; Data Curation, Formal Analysis; Writing – Review & Editing). All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
The study was conducted in accordance with the Guidelines of the Canadian Council on Animal Care, and the protocol was approved by The University of Lethbridge Animal Welfare Ethics Committee (approval number: 0508).
We authors would like to thank Courtney Lamb for behavioural testing and the University of Lethbridge Animal Care Staff for diligent care of the subjects.
This work was funded by a Natural Sciences and Engineering Research Council of Canada grant (RGPIN-2020-06929) awarded to RJM.
The authors declare no conflict of interest. Robert J. McDonald is serving as one of the Editorial Board members of this journal. We declare that Robert J. McDonald had no involvement in the peer review of this article and has no access to information regarding its peer review. Full responsibility for the editorial process for this article was delegated to Gernot Riedel. Fraser Sparks’ affiliation is with Regeron Pharmacueticals. He has no conflict of interest to declare as Regeron had no involvement with this research study.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.





