1 Mental Illness Research, Education, and Clinical Center, VA Palo Alto Healthcare System, Palo Alto, CA 94304, USA
2 Department of Psychiatry and Behavioral Sciences, Stanford University School of Medicine, Stanford, CA 94305, USA
Abstract
Despite the connections and clear importance of the cerebellum in motor function, research utilizing cerebellar neuromodulation for treatment of movement disorders is still underdeveloped. Here we conduct a systematic review to investigate non-invasive neurostimulation of the cerebellum and its potential impact on motor systems and its function. Our aim is to give a general review of each neurostimulation study focusing on the cerebellum as a treatment target in the past five years at time of search, in order to update the field on current findings and inspire similar cerebellar neurostimulation research without unnecessary repetition.
Using the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) guidelines, our search included articles over the past five years that evaluated neurostimulation of the cerebellum (e.g., transcranial magnetic stimulation, transcranial direct current stimulation, and transcranial alternating current stimulation, etc.). Inclusion criteria included: (1) neurostimulation (repetitive transcranial magnetic stimulation (rTMS), transcranial direct current stimulation (tDCS), transcranial alternating current stimulation (tACS)) of the cerebellum; (2) only original articles, and (3) outcomes focused on motor functions. Exclusion criteria included: (1) neurostimulation with the goal of targeting any brain structure other than the cerebellum and (2) reviews and conference abstracts.
The search revealed 82 distinct articles relevant to the research question. Included are 17 articles concerning rTMS, 56 articles concerning tDCS, and 9 articles concerning tACS. The majority of the studies are controlled trials of varying types, with 79, with two case studies and one pilot study.
Many studies showed significant effects on motor function and circuitry via non-invasive neurostimulation of the cerebellum. Common targets of cerebellar neurostimulation include visuomotor control, stroke rehabilitation for improvements in balance and coordination, and motor skill acquisition. The field is still exploring ideal parameters of neurostimulation for each disorder or function of interest. Future research areas should include the inclusion of individual anatomy, including functional connectivity, and improving stimulation selectivity.
Keywords
- neurostimulation
- cerebellum
- transcranial magnetic stimulation motor functioning
- transcranial direct current stimulation
- transcranial alternating current stimulation
The cerebellum has been a neglected topic in non-invasive neurostimulation, despite its link to a myriad of functions. Historically best known for error correction of motor movement, its connectivity also supports aspects of vision, language, attention, and various psychiatric and motoric conditions, such as Parkinson’s Disease. Although there is clear evidence the cerebellum is a significant node in neural networks involved in motor function, it is often left out as a piece of non-vital circuitry among motor and movement literature [1, 2]. Consequently, research on neurostimulation of the cerebellum is lagging behind neurostimulation of other motor brain regions. Despite the promise of cerebellar neurostimulation, there is not a consensus in the field of neurostimulation for applications in this area. A general review is needed to bring together results from various fields to illustrate the broad potential of cerebellar stimulation. Previous reviews have often focused on more specific disorders, or stimulation not only of the cerebellum. Here, we conduct a systematic review of non-invasive neurostimulation of the cerebellum choosing to focus on stimulation methods of transcranial magnetic stimulation (TMS), transcranial direct current stimulation (tDCS), and transcranial alternating current stimulation (tACS). We include only motor-related non-invasive neurostimulation of the cerebellum related to motor function in this review because motor dysfunction is a primary diagnostic feature for many clinical presentations.
The cerebellum’s impact on motor dysfunction occurs from its complex interconnectivity to various structures. Briefly, the cerebellum is fed information from the rest of the nervous system via separate inputs from the cortex, brain stem, spinal cord, and vestibular system [3]. The cerebellum also sends information to nearly every area of the brain, either functionally or via neuroanatomic connections [4, 5]. The cerebellum’s function as an adaptive controller depends on robust functional and structural connectivity to every other part of the brain [6]. Strong connections both to and from the cerebellum make it a logical target for neurostimulation given its connections to many regions and its involvement in a variety of disorders. Vital efferent connections for motor function in particular include the cerebellovestibular tract, the cerebelloreticular tract, the corticonuclear tract, and the cerebellorubral tract [7]. Afferent sources of the cerebellum include the dorsal spinocerebellar tract, the cuneocerebellar tracts, the olivocerebellar tract, the reticulocerebellar tract, and the vestibulocerebellar tract [8]. Structurally, the cerebellum is divided into three primary lobes, the anterior lobe, posterior lobe, and flocculonodular lobe, and into nine smaller lobules usually referred to by their respective roman numerals. Generally, these nine sub-lobules are associated with specific networks or functions, such as visuo-motor control, language, etc., although this can vary by individual or disease state [9]. Traditionally, as the view of the cerebellum was of motor modulation only, connections to non-motor regions have been neglected. Although in this article we focus on motor applications of cerebellar stimulation and subsequent connectivity, our hope is that this is taken as an example of the full potential of cerebellar stimulation. The motor cortex has been given extensive attention in the context of neurostimulation. In addition to serving as the stimulation target for studies of motor functioning, clinical TMS relies on the cortical excitability of the motor strip in order to calculate the therapeutic dosage for many clinical conditions [10]. While many studies have been undertaken focusing on motor cortex stimulation, the cerebellum, the backdoor of the brain, despite its blatant connections to the motor cortex has not been the focus.
Although non-invasive neurostimulation of the brain has ancient roots, stimulation targeting the cerebellum itself was only commonly experimented with since the 1970s [1]. Non-invasive neurostimulation generally is performed in either an excitatory or inhibitory manner, meant to either induce firing or prevent firing of the target neuron [11]. The effects of either can be complicated by subsequent targeting of an excitatory or inhibitory neuronal circuit [3]. Stimulation of the cerebellum is still not commonly done, even in motor applications where there are generally clear and easily defined and testable outcomes. Types of neurostimulation applied to the cerebellum include TMS, tDCS, and tACS [12]. TMS uses a wire coil to create a focused magnetic field to stimulate nerve cells at a targeted location, while tDCS and tACS use electric current delivered via electrodes on the skin to directly pass current through the skull and into the brain, resulting in the excitation or inhibition of neurons. All are thought to modify the nervous system by alteration of excitatory and inhibitory balances,either by direct firing or inhibition of neurons or priming of the system. Location of stimulation can also vary greatly between studies. An example figure of approximate electric fields based on different stimulation locations of TMS over the cerebellum is shown in Fig. 1. Within this study, we conducted a thorough and systematic review of the literature focusing on non-invasive neurostimulation of the cerebellum and its potential impact on motor function. This paper provides a general review of each neurostimulation study of the cerebellum related to motor function done in the last five years at time of search, to update the field on current findings and inspire similar cerebellar neurostimulation research without unnecessary duplication.
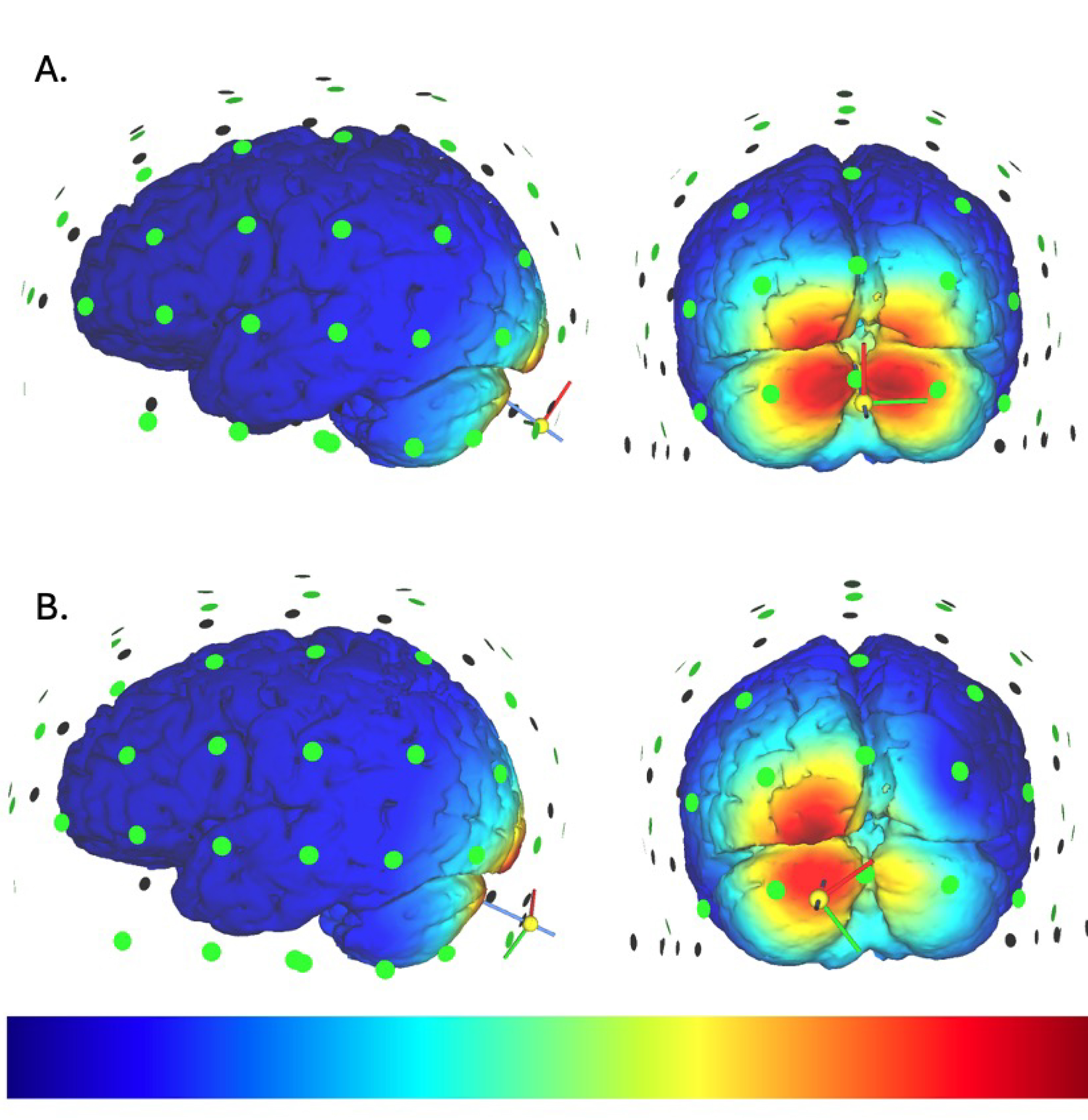 Fig. 1.
Fig. 1.
Views of the approximate electric field. Views based on (A). TMS stimulation at the inion with the handle pointing to the subjects’ left (B). TMS stimulation 3 cm to the left of the inion with the handle pointing oppositely from the green axis pointer. Simulations generated by SimNIBS 4.1.0 Version 4.1.0 developed by the Danish Research Centre for Magnetic Resonance (DRCMR) and the Technical University of Denmark (DTU) in Hvidovre, Denmark. These colormaps can be interpreted only as a very smooth approximation of the electric field, and does not determine exactly which neurons will be affected by the neurostimulation applied. TMS, transcranial magnetic stimulation.
Our search focused on articles evaluating neurostimulation of the cerebellum related to motor function, including transcranial magnetic stimulation, transcranial direct current stimulation, and transcranial alternating current stimulation, as well as articles that were included in searches for unspecified neurostimulation or neuromodulation. This search was limited to articles published in the last five years but did not limit the search according to study design, age, or sex of participants. This review follows Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) guidelines established by Page et al. (2021) [13]. The PRISMA checklist can be found in Supplementary Material. We systematically searched PubMed and ScienceDirect through September 2021 using the following keywords: transcranial magnetic stimulation AND cerebellum, transcranial direct current stimulation AND cerebellum, transcranial alternating current stimulation AND cerebellum, neuromodulation AND cerebellum, and neurostimulation AND cerebellum. After removing duplicate entries, two of the authors manually screened articles for relevance to this review’s focus on motor function. We did not include reviews or invasive neurostimulation methods, or non-human studies. The search strategy can be seen in Fig. 2.
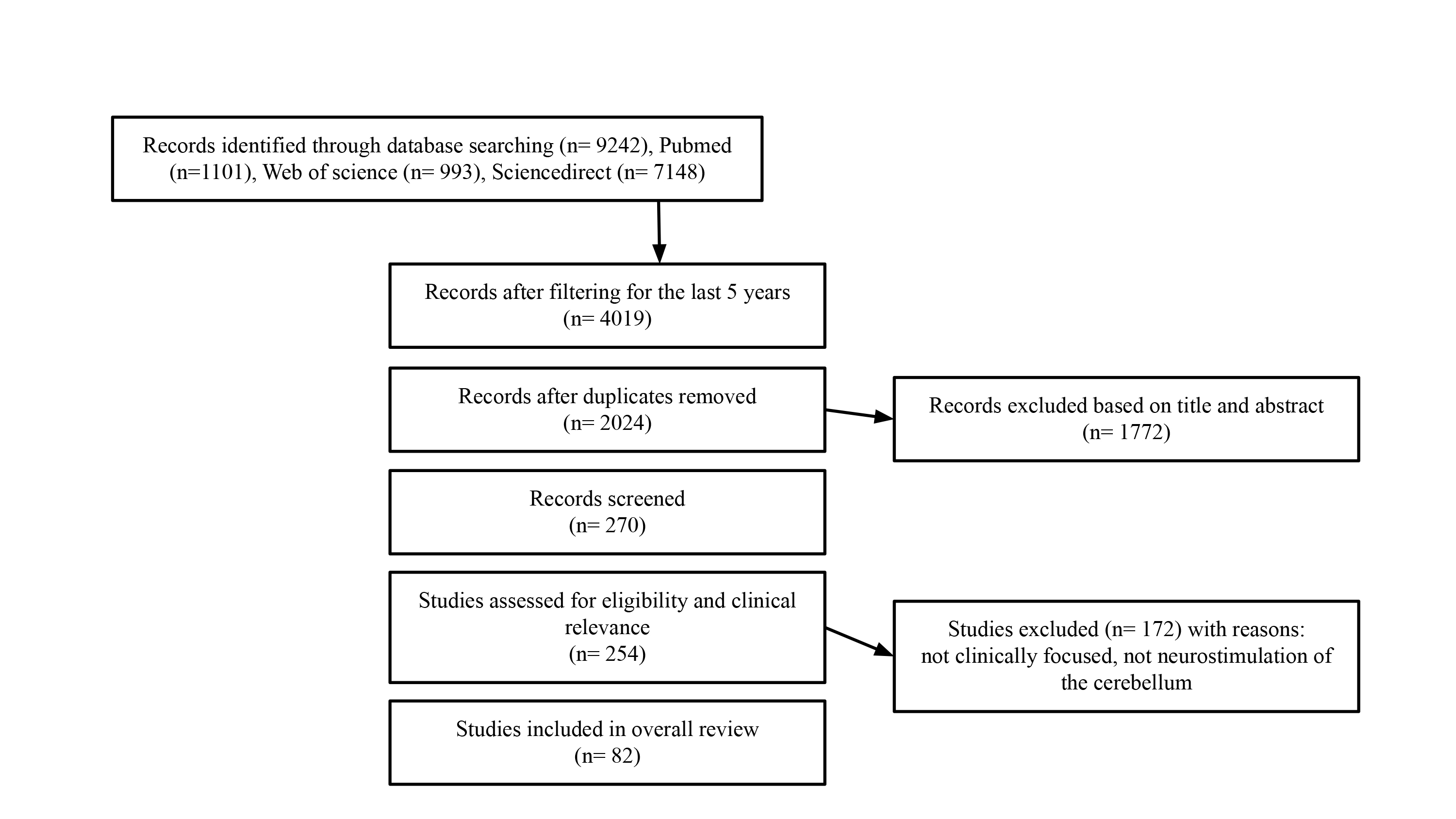 Fig. 2.
Fig. 2.
Search strategy and number of included papers at each step. 82 total studies are included in the review following Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) guidelines (the PRISMA checklist can be found in Supplementary Material).
The following inclusion criteria were used to determine if studies were eligible for inclusion: (1) neurostimulation (repetitive transcranial magnetic stimulation, rTMS; tDCS; tACS) of the cerebellum related to motor function and (2) original articles. The exclusion criteria included the following: (1) neuromodulation solely targeting any brain structure other than the cerebellum (i.e., neurostimulation of the cerebellum had to be the main focus of the study); (2) reviews and conference abstracts. Several independent investigators performed the literature search and selection, and conferred to include or exclude studies with uncertainties. 82 distinct articles relevant to the research question. Included are 56 articles concerning tDCS, 9 articles concerning tACS, and 17 articles concerning rTMS.
The search revealed 82 distinct articles relevant to the research question. Included are 56 articles concerning tDCS, and 9 articles concerning tACS (Table 1, Ref. [14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66, 67, 68, 69, 70, 71, 72, 73, 74, 75, 76, 77, 78]), 17 articles concerning transcranial magnetic stimulation (Table 2, Ref. [79, 80, 81, 82, 83, 84, 85, 86, 87, 88, 89, 90, 91, 92, 93, 94, 95]). The majority are controlled studies of varying types, along with two case studies and one pilot study. We have divided our review findings into specific functional areas, including visuomotor, reflexes, mood and dystonia, whole body control, motor skill acquisition, gait, hand and grip, and excitability and inhibition. Many of the included studies overlapped these categories and were placed by best fit.
| Ref | N (treatment, control) | Participant Type | Method | Stimulation type | Cathodal/Anodal | Electrode Position | Target | Intensity | Duration | Findings | Blind | Active, Sham, Control |
| Akremi et al., 2022 [74] | 10, 10 | Children between ages 10–16 and confirmed medical diagnosis of DCD. | tDCS | Concurrent | Anodal | (1) A 35 cm2 (7 |
Motor sequence learning and upper limb coordination | 2 mA | 20 min | They found that this stimulation reduced the number of errors made during the motor sequence task although it did not impact coordination or learning observed outside of the stimulation task. | DB | Active, sham |
| Azarpaikan et al., 2021b [56] | 48, 16 | Healthy participants. | tDCS | Offline | Anodal | The 5 |
Bimanual adaptive motor tasks | 1.5 mA | 15 min | atDCS over the cerebellum leads to more improvement in motor performance and transfer in a bimanual coordination task than atDCS over the right parietal. | DB | Active, sham |
| Azarpaikan et al., 2021a [55] | 105, 15 | Right-handed healthy individuals with no history of neurological or psychiatric problems. Age range = 20–31 yrs. | tDCS | Both | Anodal | Cathode electrode was placed on the ipsilateral deltoid muscle in all groups. Anode was placed on the right posterior parietal cortex in groups 1, 2, and 3, and on the cerebellum in groups 4, 5, and 6. | Modulation of oscillatory brain activity | 1.5 mA | 15 min | anodal parietal and cerebellar tDCS cause changes in brain wave frequencies. | No | Active, sham |
| Azarpaikan et al., 2020 [73] | 48, 16 | Healthy participants. | tDCS | Both | Anodal | stimulation was applied to the cerebellum with a direct current of 1.5 mA for 15 min. | Two-hand coordination | 1.5 mA | 15 min | The during-tDCS and after-tDCS groups achieved greater improvements in speed/accuracy than the before-tDCS and sham-tDCS groups. | SB | Active, sham |
| Baharlouei et al., 2020 [29] | 32 | Healthy older adults, above age 60 yrs. | tDCS | Offline | Anodal | (1) In the M1 tDCS group, the anode was centered 1 cm behind the vertex, and the return electrode was placed on the forehead. (2) In the cerebellar tDCS group, the anode was centered 1 cm below the inion of the occipital bone, and the return electrode was fixed over the right shoulder. | Balance | 2 mA | 20 min | Anodal tDCS of the cerebellum and Ml could improve the postural balance indices in healthy older adults. These two techniques exerted similar effects on static balance. | DB | Active, sham |
| Ballard et al., 2019 [42] | 44 | Healthy, young adults, ages 18–20 yrs. | tDCS | Offline | Both | The electrode placement for both experiments was focused on the right lateral posterior cerebellum. The “spiral out” approach was used for cathodal stimulation, and the “spiral in” approach was used for anodal stimulation. | Sequence learning | 2 mA | 20 min | Cathodal stimulation has a positive influence on learning while anodal stimulation has the opposite effect, relative to sham. | SB | Active, sham |
| Beyer et al., 2017 [48] | 90 | Healthy, young participants- Experiment 1: mean age = 23.5, Experiment 2: mean age = 23.6, Experiment 3: mean age = 23.3 yrs. | tDCS | Offline | Both | The stimulating electrode was centered 3 cm lateral to the inion over the right cerebellar hemisphere. The reference electrode was placed in a vertical position on the ipsilateral deltoid muscle in Experiment 1 and on the buccinator muscle in Experiment 2. | Conditioned eyeblinks | 2 mA | 20 min | No clear polarity-dependent effects of cerebellar tDCS on CR acquisition and timing as previously described; cerebellar tDCS during extinction did not modulate extinction or reacquisition. | DB | Active, sham |
| Bocci et al., 2018 [24] | 20 | Right-handed healthy volunteers with no history of neurological didsorders. Mean age = 25 yrs. | tDCS | Both | Both | Anode: centered on the median line 2 cm below the inion, with its lateral borders about 1 cm medially to the mastoid apophysis; Cathode: over the right shoulder. | Hand blink reflex | 2 mA | 20 min | Did not find an effect of cathodal cerebellar tDCS, but found that anodal cerebellar tDCS at 2 mA for 20 minutes significantly dampened the magnitude of the hand blink reflexes | SB | Active, sham |
| Bocci et al., 2020 [41] | 4 | Patients with Huntingtons’s Disease (HD), mean age = 46.5 yrs. | tDCS | Offline | Anodal | Anode: applied on the median line, 2 cm below the inion, with lateral borders about 1 cm medially to the mastoid apophysis; Cathode: over the right shoulder. | Dystonia | 2 mA | 20 min | Found that clinical scores improved, in particular symptoms related to dystonia. These effects lasted approximately 4 weeks. | DB | Active, sham |
| Bodranghien et al., 2017 [70] | 20 | Healthy participants, mean age = 24 yrs. | tDCS | Offline | Anodal | The anode (size: 5 |
Self-paced finger movements | 1.5 mA | 20 min | No significant change in entropy ratios and laterality coefficients (LC) after atDCS of the cerebellum in the 14 channels. | SB | Active, sham |
| Chothia et al., 2016 [40] | 12 | Healthy adults; age range = 23–57 yrs. | tDCS | Concurrent | Anodal | Anode: Left parietal lobe, 3 cm lateral from the mid-central scalp (Cz). | Whole body control | 2 mA | 15 min | Reduced excitation in the descending pathway without inhibiting cervical propriospinal neurons. | SB | Active, sham |
| Cathode: Left buccinator. | ||||||||||||
| Craig & Doumas, 2017 [37] | 16 | Both older and young adults. | tDCS | Concurrent | Anodal | Anodal electrode was placed on the median line 2 cm below the inion. The reference electrode was placed on the right buccinator muscle. | Balance difficulty for younger adults | 2 mA | 20 min | Younger adults had only offline and not task effects, while for older adults both stimulation over the cerebellum and M1 were found to increase performance, but only during stimulation. | DB | Active, sham |
| Doeltgen et al., 2016 [75] | 27 | Right-handed healthy adults without a neurological or musculoskeletal disorder affecting upper limbs. | tDCS | Concurrent | Anodal | One electrode positioned 3 cm lateral and 1 cm inferior to the inion (right lateral cerebellum) and the other over the ipsilateral buccinator muscle. | Cerebellar inhibition | 2 mA | 20 min | Reduced cerebellar brain inhibition, but did not impact short afferent inhibition or impact reflexes. | DB | Active, sham |
| Ehsani et al., 2017 [36] | 14, 15 | Healthy older adults, mean age = 65.79 yrs. | tDCS | Both | Anodal | Active (anode) and the returning (cathode) electrodes were placed over the cerebellum (1 cm below inion of occipital bone) and right arm. | Postural control in older adults | 1.5 mA | 20 min | Found that postural sway was significantly lessened, and balance scores significantly improved after anodal tDCS over the cerebellum. | DB | Active, sham |
| Emadi et al., 2020 [30] | 90 | Healthy college students with no history of neurological or musculoskeletal disorders. | tDCS | Both | Both | For anodal stimulation, the anode was placed 3 cm laterally to the inion over the cerebellar hemisphere ipsilateral to the participant’s dominant leg, and the cathode was placed over the ipsilateral buccinator muscle. For cathodal stimulation, the opposite montage was used (i.e., anode over the buccinator muscle and cathode over the cerebellar hemisphere). | Balance control | 2 mA | 20 min | Sway was helped with visual feedback, and effects remained longer when cathodal tDCS was applied, implying it supports the short term maintenance of the positive effects of visual feedback therapy for balance. | DB | Active, sham |
| Fernandez et al., 2017 [66] | 14 | Right-handed healthy adults between the ages 23–37 yrs. | tDCS | Both | Cathodal | The centre of the cathodal electrode was positioned 1 cm below the inion and 3 cm to the right, while the anodal electrode was placed over the right buccinator muscle. | gait rhythm | 2 mA | 20 min | Did not observed any detrimental effects when stimulation was applied during walking test. | DB | Active, sham |
| Ferrucci et al., 2019 [46] | 40 | Healthy participants, mean age = 26.65 yrs. | tDCS | Both | Anodal | The active electrode was centered on the median line 2 cm below the inion with its lateral borders about 1 cm medially to the mastoid apophysis (over the cerebellum) and the reference electrode over the right deltoid muscle. | Motor skill acquisition | 2 mA | 20 min | Did not find that cerebellar tDCS had any impact on spatial navigation skills in a group of healthy subjects. | N/A | Active, sham |
| Fleury et al., 2021 [17] | 16, 11 | Right-handed healthy participants with no neurological disorders and have never experienced prisms before the experiment. | tDCS | Concurrent | Cathodal | The cathodal electrode was placed over the right cerebellum (1 cm below and 4 cm right to the inion) and the anodal electrode was placed over the right deltoid muscle. | learning and visuomotor skills | 2 mA | 21 min | Participants in the c-tDCS group showed (1) altered error reduction and pointing trajectories during exposure, mainly during the first trials; (2) increased magnitude but reduced robustness of pointing after-effects; and crucially, (3) slightly altered transfer of after-effects to the throwing task. | SB | Active, sham |
| Foerster et al., 2017 [34] | 15 | Right-handed and right-footed healthy females, aged 21–24 yrs. | c-tDCS | Offline | Both | The electrodes were placed on the right cerebellar hemisphere (3 cm lateral to the inion) and over the deltoid muscle in the right arm. | Balance performance | 1 mA | anodal: 13 min cathodal: 9 min | Found an impairment of performance during a dynamic balance task due to cathodal cerebellar tDCS, but not an improvement of balance performance during the anodal condition. | DB | Active, sham |
| Giustiniani et al., 2021 [64] | 18 | Right-handed healthy participants, mean age: 25.2 yrs. | tACS | Concurrent | Anodal | The centre of the active electrode was placed over the right cerebellar hemisphere (1 cm under and 3 cm right to the inion). The centre of the return electrode was placed over the ipsilateral buccinator muscle. | Motor skill acquisition | 2 mA | n/a | Gamma stimulation was found to impair participant performance in repeated tasks, but not randomized tasks. | DB | Active, sham |
| Grami et al., 2022 [22] | 21 | Right-handed healthy participants, mean age: 28 yrs. | tDCS | Offline | Anodal | The anodal electrode (size: 5 |
Brain networks | 1.5 mA | 20 min | Observed a behavioral increase in task accuracy when compared to sham, and an increase in connectivity between the central executive network, salience network, and lobule VII of the cerebellum. | SB | Active, sham |
| Hulst et al., 2017 [20] | 20 | Right-handed individuals with cerebellar degeneration; age range: 30–74 yrs. | tDCS | Offline | Both | The anodal electrode was placed on the position of the right cerebellar cortex, with the center of the anodal electrode 3 cm lateral to the inion, and the cathodal electrode was placed on the right buccinator muscle. | Cerebellar degeneration | 2 mA | 22 min | Did not find a clinical response in this subject population. | SB | Active, sham |
| Iannone et al., 2019 [27] | 1 | Female participant with generalized dystonia; 45 yrs. | tDCS | Offline | Anodal | Electrode placement on the scalp was determined using the International 10–20 EEG system (surface 35 cm2). | Mood and Dystonia | 2 mA | 30 min | After 10 tDCS sessions there was also improvement in mood with normalization of Beck Depression Inventory scores. There were no complications in spite of the implanted STN-DBS leads. | N/A | No |
| Inukai et al., 2016 [39] | 16 | Healthy male participants; mean age: 21. | tDCS | Offline | Both | In Experiment 1, the center of the stimulation electrode was placed 2 cm below the inion and the other electrode on the forehead. In Experiment 2, the center of stimulation electrode was also placed 2 cm below the inion and the other electrode on the right buccinator muscle. | Whole Body Control | 2 mA | 20 min | Reported a lowering of center of gravity sway during cathodal tDCS for both return electrode conditions. | SB | Active, sham |
| Jackson et al., 2019 [54] | 42 | Right-handed males who are not participating in a throwing sport; mean age: 25 yrs. | tDCS | Concurrent | Anodal | Anode 3 cm right of the inion; cathode right buccinator muscle. | Motor learning | 2 mA | 25 min | Complex overhand throwing task that cerebellar tDCS enhanced motor learning via decline in errors. | DB | Active, sham |
| Jalali et al., 2018 [76] | 34 | Right-handed healthy young individuals; mean age 22 yrs. | tDCS | Concurrent | Anodal | The anodal electrode was placed over the right cerebellar cortex, 3 cm lateral to the inion. The cathodal electrode (reference) was placed over the right buccinator muscle. | GABA and glutamate with MRS | 2 mA | 25 min | Did not find group changes of GABA and glutamate due to cerebellar tDCS. However, their application of tDCS did coincide with motor memory retention and this was correlated with a decrease in cerebellar glutamate. | SB | Active, sham |
| John et al., 2017 [69] | 14, 14 | Right-handed individuals with pure cerebellar degeneration; mean age: 50.8 yrs. | tDCS | Offline | Anodal | For cerebellar stimulation the anodal electrode was placed over the right cerebellar cortex, with the center of the electrode being 3 cm lateral to the inion, and the cathodal electrode was placed on the right buccinator muscle. The anodal electrode for M1 stimulation was centered over the area of the left primary motor cortex. | Cerebellar degeneration | 2 mA | 25 min | No observation of any change in grip strength in the patient group or controls. | SB | Active, sham |
| Jones et al., 2021 [45] | 48 | Right-handed undergraduate students; mean age: 21.4 yrs. | tDCS | Concurrent | Both | The anode electrode was centered at 10–20 location F10, and the cathode electrode was centered at 10–20 location T5, also known as P7. | Motor skill acquisition | 2 mA | 30 min | c-tDCS did not significantly increase performance on a complex category learning task. | DB | Active, sham |
| Jongkees et al., 2019 [47] | 72 | Right-handed, healthy undergraduate students between 18 and 30 yrs. | tDCS | Concurrent | Both | Three electrodes of 35 cm2 (5 cm |
Motor task learning | 1 mA | 20 min | Anodal tDCS demonstrated increased response latencies, but only when the previously trained sequence and a transfer sequence had to be performed in the same experimental block. | No | Active, sham |
| Kamali et al., 2019 [72] | 16 | Right-handed participants with 2 to 3 years of experience in pistol shooting. | tDCS | Concurrent | Anodal | The anode electrode (35 cm2) was placed over the right cerebellar area (CB2), 1 and 3 cm below and lateral to the inion. The cathode electrode (16 cm2) was placed over the left dorsolateral prefrontal cortex (dlPFC). | Motor visualization | 2 mA | 20 min | Found that tDCS improved the average shooting score and improved accuracy when anodal stimulation was applied at 1 mA for 20 minutes over the right cerebellum, 1 cm below and 3 cm lateral to the inion with a cathode electrode over the left dlPFC. | DB | Active, sham |
| Katagiri et al., 2021 [32] | 36 | Healthy adult participants; mean age: 21.8 yrs. | tDCS | Offline | Both | One electrode was positioned over the cerebellum (2 cm below the inion of the occipital bone). The other electrode was centered on the supraorbital region. | Whole body control | 2 mA | 20 min | Found that anodal tDCS induced cerebellar inhibition that was correlated with learning of postural control. | TB | Active, sham |
| Kenville et al., 2020 [35] | 20 | Healthy right-handed participants; mean age: 23.29 yrs. | tDCS | Offline | Anodal | The anode (35 cm2, current density: 0.057 mA/cm2) was placed either over the bilateral M1 leg area or the bilateral cerebellum, with the cathode (reference; 100 cm2, current density 0.020 mA/cm2) placed on the medial part of the supraorbital bone (tDCS of bilateral M1 leg area) or the right musculus buccinator (tDCS of bilateral cerebellum). | Whole body control | 2 mA | 20 min | tDCS over M1 and the cerebellum has been observed to increase maximum voluntary contraction force, | DB | Active, sham |
| Koganemaru et al., 2020 [68] | 14 | Healthy, right-handed young participants with no history of neurological illness or gait disorders; mean age: 20–23 yrs. | tACS | Concurrent | N/A | The electrode (5 |
Gait rhythm | 2 mA | 10 min | Significantly entrained their gait rhythm. | No | Active, sham |
| Kumari et al., 2020 [62] | 30 | Healthy individuals aged 18 years or above. | tDCS | Concurrent | Anodal | The anodal electrode was placed 3 cm lateral to the inion to position it over the cerebellar hemisphere ipsilateral to the dominant leg, which was placed on the fast belt of the split-belt treadmill 20. The cathode was placed over the ipsilateral buccinator muscle. | Task adaptation | 2 mA | 15 min | Increased maintenance of learned walking patterns, implying a role in anodal tDCS of the cerebellum in motor adaptation. | DB | Active, sham |
| Küper et al., 2019 [71] | 48 | Healthy individuals aged 18 years or above. | tDCS | Concurrent | Both | For cerebellar stimulation, one electrode was centered 3 cm laterally to the inion and the other electrode was placed on the right buccinator muscle. | Cerbellar cortex activity | 1.8 mA | 20 min | Cathodal c-tDCS leads to a disinhibition of the dentate nucleus, whereas anodal c-tDCS may have the opposite effect. | No | Active, sham |
| Leow et al., 2017 [50] | 72 | Healthy right-handed individuals; mean age: 22.2 yrs. | tDCS | Concurrent | Anodal | For all groups, the anodal electrode was placed over the scalp area estimated to overly the right cerebellar cortex (3 cm lateral to the inion), and the reference electrode was positioned on the skin area overlying the right buccinator muscle. | Sensorimotor adaptation | 1.5 mA | 40 min max. | tDCS increased implicit remapping during sensorimotor adaptation, irrespective of preparation time constraints. | No | Active, sham |
| Liebrand et al., 2020 [61] | 25 | Healthy right-handed individuals; mean age: 22.6 yrs. | tDCS | Concurrent | Anodal | For lM1 tDCS, the anodal electrode was centered at FC3 and the cathodal electrode at CP3. For sham, electrodes were placed either with lM1 or rCB montage. | Motor learning | 1 mA | 20 min | Right cerebellar tDCS led to improved sequence performance in Mid-Late time windows and learning-specific increase in activity from neuromodulation. | SB | Active, sham |
| Liew et al., 2018 [15] | 111 | Healthy right-handed individuals that had not previously participated in a visuomotor adaptation study. | tDCS | Concurrent | Anodal | Cerebellar (CB) stimulation utilized a bipolar electrode montage with the anode placed over the right cerebellar cortex, located 3 cm to the right of the inion, and the reference electrode placed over the right buccinator muscle. dlPFC stimulation utilized a canonical bipolar electrode montage with the anode placement corresponding to the F3 position in the international 10–20 EEG system and reference placement over the right contralateral supraorbital region. | Visuomotor adaptation | 2 mA | 25 min | Different neural regions can be engaged to improve visuomotor adaptation, but shows that each region’s effects are highly context-dependent and not clearly dissociable from one another. | DB | Active, sham |
| Lipp et al., 2020 [49] | 100 | Healthy young individuals; age range: 19–35 yrs. | tDCS | Offline | Anodal | For cerebellar stimulation, one electrode was centered 1–2 cm caudal and 3–4 cm lateral to the inion ipsilateral to the US (that is, on the right side). The other electrode was placed over the ipsilateral (right) buccinator muscle. | Extinction | 1–2 mA | 20 min | No direct effects of dlPFC or cerebellar tDCS were demonstrated on extinction. | DB | Active, sham |
| Mamlins et al., 2019 [23] | 120 | Healthy right-handed individuals; age range: 19–31 yrs. | tDCS | Offline | Both | The active electrode was placed 3 cm lateral to the inion with its center on the same level as the inion. The return electrode was positioned on the ipsilateral buccinator muscle. | Adaptation | 2 mA | 19 min | Unable to support previous findings of modulatory cerebellar tDCS effects in reaching adaptation tasks. | DB | Active, sham |
| Marotta et al., 2021 [21] | 45 | Healthy right-handed students; age range: 18–35 yrs. | tDCS | Offline | Anodal | The anode was placed over one of the two target areas according to the international EEG 10–20 system for EEG electrode placement. For tDCS over the left PMc (Experiment 1), the anode was placed on FC5, which approximates the scalp location overlying the left PMc. | Causal role of PMc and cerebellum | 1–2 mA | 25 min | Anodal tDCS over the PMc reduced the feeling of agency in the active congruent condition, while it enhanced proprioceptive drift when applied over the cerebellum. | DB | Active, sham |
| Matsugi & Okada, 2020 [77] | 11 | Healthy individuals with no history of epilepsy or other neurological diseases; mean age: 21.2 yrs. | tDCS | Offline | Anodal | The center of the active electrode was placed at 1 cm below and 3 cm to the right of the inion, and the reference electrode was placed on right deltoid muscle. | Spinal motoneuron pool excitability | 2 mA | 15 min | H-reflex ratio was significantly increased by anodal c-tDCS, reduced by cathodal c-tDCS, and not affected by sham c-tDCS, indicating that the effect of c-TMS on the H-reflex is modulated by c-tDCS in a polarity-specific manner. | SB | Active, sham |
| Miyaguchi et al., 2020 [60] | 30 | Healthy right-handed individuals; mean age: 21 yrs. | tACS | Concurrent | N/A | Two active electrodes (5 |
Motor learning | 1 mA, 70 Hz | 1 min/8 trials | No | Active, sham | |
| Miyaguchi et al., 2018 [14] | 20 | Healthy right-handed individuals; mean age: 21.5 yrs. | tACS | Concurrent | N/A | The electrodes were placed on the scalp over the left M1, right cerebellar cortex region and right cheek. The center of the M1 electrode was placed on the scalp over the left M1 hot spot. The center of the right cerebellar cortex electrode was placed 2.0 cm below and 3.0 cm laterally to the inion. The reference electrode was placed the right cheek to minimize any unintended effect of other cortex. | Motor performance | 1.0 mA, Gamma (70 Hz), beta (20 Hz) | 30 s/12 trials | Subjects with poorer motor performance, tACS with gamma oscillations applied over the M1 and contralateral cerebellar hemisphere improved their performance. | N/A | Active, sham |
| Miyaguchi et al., 2019a [25] | 20 | Healthy right-handed individuals; mean age: 21.3 yrs. | tACS | Concurrent | N/A | Two active electrodes (5 |
Motor performance | 1.0 mA, 70 Hz | 30 s/10 trials | The effect of gamma tACS over the M1 and the cerebellar hemisphere has phase specificity. | N/A | Active, sham |
| Miyaguchi et al., 2019b [59] | 20 | Healthy right-handed individuals; mean age: 20.7 yrs. | tACS | Concurrent | N/A | Two active electrodes (5 |
Motor learning | 1 mA–2 mA, 70 Hz | 30 s/10 trials | Task error was found to be lower under the anti-phase condition, but not the in-phase condition when compared to sham. | No | Active, sham |
| Mizuguchi et al., 2018 [53] | 24 | Healthy male participants; mean age: 23 yrs. | tDCS | Offline | Both | The target electrode was placed over the right cerebellum, 3 cm lateral to the inion, and the reference electrode was placed over the right buccinator muscle. | Task performance | 2 mA | 20 min | Improvement did not differ among stimulation conditions. The magnitude of improvement was associated with an individual’s level of task performance only under cathodal tDCS condition | DB | Active, sham |
| Naro et al., 2017 [67] | 25 | Healthy individuals; mean age: 30 yrs. | tACS | Concurrent | Anodal | The active electrode (25 cm2) was positioned over the right cerebellar hemisphere in the same position used for DCC (and brief tDCS), the reference over the ipsilateral buccinator muscle. | Excitability and gait control | 2 mA, 3000 cycles at 10 Hz, 15,000 at 50 Hz, and 90,000 at 300 Hz | 5 min | Paradigm allowed eliciting CBI from lower limbs; and (2) our approach allowed exploring some aspects of cerebellar networks related to the lower limbs. | DB | Active, sham |
| Newstead et al., 2018 [26] | 44 | Healthy individuals; no significant differences of gender, handedness and age. | tDCS | Offline | Both | One electrode is positioned over the left dlPFC (F3 position according to the 10–20 electrode system), whilst the opposing electrode is positioned over the contralateral lobe of the cerebellum. | Mood and Dystonia | 2 mA | 12 min | Results revealed significantly elevated mood in participants following both single and repeated tDCS conditions compared to sham, with increased mood from repeated c-tDCS. | SB | Active, sham |
| Petti et al., 2017 [78] | 8 | Healthy right-handed individuals; mean age: 30.1 yrs. | tDCS | Offline | Both | One electrode was applied over the cerebellum 3 cm to the right of inion. The second electrode was positioned on the ipsilateral deltoid muscle. | Brain networks | 2 mA | 20 min | Cathodal stimulation appeared to have minimal changes from sham, however anodal stimulation was observed with lateral synchronization in the sensorimotor area, as well as network segregation in sensory-motor rhythms. | No | Active, sham |
| Poortvliet et al., 2018 [38] | 14, 14 | Healthy young adults. | tDCS | Offline | Anodal | The anode was positioned over the cerebellum with the center of the electrode located approximately 1.5 cm below the inion. The reference electrode was positioned centrally on the forehead to avoid lateralized current flow. | Standing balance | 1 mA | 20 min | Improved postural steadiness during a platform vibration task. | DB | Active, sham |
| Rauscher et al., 2020 [31] | 40 | Healthy middle-aged participants between ages 50–65 yrs. | tDCS | Concurrent | Anodal | An electrode of the size of 5 cm |
Whole body balance | 2.8 mA | 19 min 30 sec | “Significant learning effects were found in all participants. Balancing performance and learning rate was significantly less in the group of middle-aged adults compared to our previous group of young adults. No significant effects of cerebellar tDCS were observed”. | DB | Active, sham |
| Rodriguez-Ugarte et al., 2018 [18] | 14 | Healthy individuals between 23 and 38 yrs. | tDCS | Offline | Anodal | The anode over the primary motor cortex in Cz and the cathode over the right cerebro-cerebellum (two centimeters right and one centimeter down of the inion). | Motor visualization | 0.4 mA | 15 min | The stimulation group compared to sham achieved higher accuracy results for the 5 consecutive days of the experiment. | SB | Active, sham |
| Rodriguez-Ugarte et al., 2018 [19] | 10 | Healthy individuals between 20 and 39 yrs. | tDCS | Concurrent | Anodal | A second anode was used over Cz to supply a current slightly higher than the one in the anode over the right cerebrocerebellum (1 cm right and 1 cm down from the inion). | Motor visualization | 0.3–0.2 mA | 15 min | Active tDCS group reaching higher task accuracy levels faster than the non-stimulation group, although both groups eventually reached the same level. | SB | Active, sham |
| Samaei et al., 2017 [58] | 15, 15 | Healthy older adults, mean age = 68.7 yrs. | tDCS | Concurrent | Anodal | The active anode electrode was placed over ipsilateral cerebellum (1 cm below inion of occipital bone and 1 cm medial to mastoid process). The returning electrode was placed over right arm. | Motor learning | 2 mA | 20 min | RT was significantly increased after 35 minutes and 48 hours in sham tDCS group, which indicates a lack of short-term and long-term offline learning in older adults. AtDCS on cerebellar region produced more short-term and long-term offline improvement in RT compared to sham tDCS. | DB | Active, sham |
| Schubert et al., 2021 [65] | 22 | Healthy right-handed individuals, age range: 18–38 yrs. | tACS | Both | N/A | For rCB tACS, one electrode was placed on the right mandibula and the other 1 cm below and 3 cm right to the inion. For lM1 tACS, one ring-shaped electrode was placed around electrode FC3 and one around CP3 rendering the current flow as precisely as possible to C3. | Motor Sequence Learning | 1 mA at 10 Hz | 20 min | 10 Hz tACS applied to right cerebellum showed learning-related |
SB | Active, sham |
| Seyed Majidi et al., 2017 [43] | 30 | Healthy right-handed individuals; age range: 19–31 yrs. | tDCS | Concurrent | Anodal | In all participants of the prefrontal group, the anodal electrode was placed over the left prefrontal cortex (Fp3) and the reference (cathodal) electrode was placed over the right supraorbital region (on the forehead). In all participants of the cerebellar group, the anodal electrode was placed on the right cerebellar hemisphere (3 cm lateral to the inion) and the reference electrode was placed on the right buccinator muscle. | Probabilistic classification learning | 2 mA | 20 min | No differences in performance and learning were observed between subjects receiving sham stimulation or anodal stimulation over either the right cerebellum or the left prefrontal cortex. | No | Active, sham |
| Shimizu et al., 2017 [57] | 82 | Healthy young adults with a mean age of 21.28 yrs. | tDCS | Concurrent | Both | The active electrode was centred 2 cm below the inion in order to target the cerebellum. The reference electrode was placed on the cheek ipsilateral to the dominant hand in order to keep the path of current flow similar relative to the hand being used. | Fine motor sequence learning | 2 mA | 20 min | Polarity-specific effects on fine motor sequence learning and generalization; an inhibitory effect of cathodal tDCS was found during practice, anodal tDCS enhanced transfer performance. | No | Active, sham |
| Steiner et al., 2020 [33] | 48 | Healthy individuals; age range: 20–29 yrs. | tDCS | Concurrent | Both | The cerebellar electrode (7 cm height |
Balance time | 2.8 mA | n/a | No significant tDCS effects on learning the balance task were observed. | N/A | Active, sham |
| Summers et al., 2018 [52] | 14 | Healthy individuals; mean age: 28.8 yrs. | c-tDCS | Concurrent | Anodal | Active electrode placement was intended to target the vermal region and bilateral hemispheres of the cerebellum. The reference electrode (50 |
Motor Training | 2 mA | 20 min | c-tDCS applied during task training is capable of modulating or interfering with practice-related changes in corticospinal excitability without disrupting performance improvement. | DB | Active, sham |
| van der Vliet et al., 2018 [51] | 117 | Healthy right-handed individuals | tDCS | Concurrent | Both | 5 |
Motor skill learning | 2 mA | 15 min | Carriers of the BDNF Val66Met polymorphism were more likely to be learners. Within the group of learners, anodal tDCS supported eyeblink conditioning in BDNF Val66Met non-carriers, but not in carriers. | DB | Active, sham |
| Verhage et al., 2017 [44] | 40 | Healthy right-handed college students; age range: 20–31 yrs. | tDCS | Anodal | The target electrode was placed over the right cerebellum 3 cm lateral to the inion, and reference electrode was placed over the ipsilateral buccinator muscle. | Learning | 1.5 mA | 20 min | cerebellar tDCS does not modulate performance and learning on an implicit categorization task. | SB | Active, sham | |
| Weightman et al., 2020 [16] | 162 | Healthy younger adults; age range: 18–29 yrs. Healthy older adults; age range: 66–84 yrs. | tDCS | Both | Anodal | For M1 stimulation the anodal electrode was positioned over the ‘hand area’ of the left motor cortex, and the cathode electrode was placed over the contralateral supraorbital ridge. For cerebellar stimulation the anode was centred over the right cerebellar cortex, 3 cm lateral to the inion and the cathode was placed on the superior aspect of the right trapezius muscle. | Motor adaptation in tasks dominated by movement of the distal versus proximal upper limb | 2 mA | 17 min | Stimulation of the cerebellum can selectively improve motor adaptation using arm movements, whereas M1 tDCS selectively improves adaptive performance when using the hand/fingers. This effect was found in two separate age groups. | SB | Active, sham |
| Wessel et al., 2020 [63] | 15 | Healthy young right-handed participants; age range: 18–35 yrs. | tACS | Both | N/A | The active electrode was placed 3 cm lateral to the inion and the return electrode over the ipsilateral buccinator muscle. | Acquisition | 2 mA | 20 min | Active stimulation did not enhance skill acquisition or retention. Minor effects on striato-parietal FC were present. | DB | Active, sham |
| Yosephi et al., 2018 [28] | 65 | Older adults with high fall risk; ages: 60–85 yrs. | tDCS | Both | Anodal | The active (anode) and the returning (cathode) electrodes were placed bilaterally over the cerebellum (1 cm below inion of occipital bone) and the right buccinator muscle. | Balance | 2 mA | 20 min | Postural training with M1 or bilateral cerebellar atDCS, especially bilateral cerebellar atDCS, can significantly improve postural control or balance in older adults with high fall risk. Two weeks of postural training alone or two weeks cerebellar atDCS alone is not a sufficient intervention. | DB | Active, sham |
Abbreviations: tDCS, transcranial direct current stimulation; DCD, developmental coordination disorder; DB, double-blind; SB, single-blind; c-tDCS, cerebellar transcranial direct current stimulation; atDCS, anodal transcranial direct current stimulation; EEG, electroencephalogram; tACS, transcranial alternating current stimulation; rCB, right cerebellum; BDNF, brain-derived neurotropic factor; CR, conditioned response; STN-DBS, subthalamic nucleus deep brain stimulation; N/A, not applicable; GABA, gamma-aminobutyric acid; MRS, magnetic resonance spectroscopy; TB, triple blind; FC, functional connectivity; US, unconditioned stimulus; PMc, premotor cortex; C-TMS, Cerebellar transcranial magnetic stimulation; DCC, double-containment coil; CBI, cerebellar inhibition; CP, centroparietal; RT, resting threshold.
| Ref | N (treatment, control) | Participant Demographics | Method | Stimulation type | Stimulation Location | Hz | %MT | Sessions; Duration | Pulses/Session | Target | Findings | Blind | Active, sham, control |
| Du et al. 2018 [90] | 20 | Healthy individuals; ages 20–62 yrs | TMS | Both | Left Motor Cortex (M1): Motor evoked potentials (MEPs), Posterior Cerebellum: the middle of the bilateral Crus I/II, Left Prefrontal Cortex (PFC): The junction of the middle and anterior thirds of the middle frontal gyrus, corresponding to the junction between posterior regions of Brodmann area (BA) 9 and the superior section of BA 46. | single pulses | 80% and 120% | 60 pulses/block for each intensity | Single pulse/each block ranged from 4 to 10 s | Motor coordination and cognition | Cerebellar evoked prefrontal synchronization was positively associated with working memory but negatively associated with coordinated rapid finger tapping. | N/A | Active, sham |
| Ferrari et al., 2021 [81] | 20 | Healthy college students; mean age = 23.1 yrs | rTMS | Both | Left and Right Primary Motor Cortex (M1): MEPs in contralateral FDI muscle, Left Posterior Cerebellum: specifically, the Crus II area. | 1 Hz | 100% | 15 min/2 sessions | 900 pulses | Motor cortical excitability | The posterolateral (left) cerebellum modulates motor cortical response to negative emotional stimuli. This implies a fascinating interaction between cerebellar impact on mood and motor function. | No | Active/sham |
| Hassan et al., 2019 [92] | 15, 15 | (1) Individuals with hepatic encephalopathy (HE); mean age = 64.1. yrs. (2) Healthy participants; mean age = 60.3 yrs | TMS | Both | Cerebellum: Conditioning stimulus applied at the midpoint between the mastoid process and the inion. Contralateral M1: Test stimulus applied over the vertex. Right FDI Muscle: Used for recording MEPs. | single pulses | 95% | 10 trials per condition, intertrial interval of 10 |
70 trials total | Hepatic encephalopathy | Lowered cerebellum inhibition was achieved via TMS, correlating with disease severity, and implying a connection of disease state with GABAergic neurotransmission in the cerebellum. | No | No |
| Koch et al., 2020 [79] | 36 (study 1) 12 (study 2) | Study (1) Healthy individuals; mean age = 26.2 yrs. Study (2) Healthy individuals; mean age = 25.6 yrs | cTBS/iTBS | Both | Theta Burst Stimulation (TBS): Applied over the lateral cerebellum, specifically 1 cm inferior and 3 cm right to the inion. TMS/EEG Recordings: Applied over the left primary motor cortex (M1). | 50 Hz | 90% | 2 sessions (study 2) | cTBS: three-pulse bursts, repeated every 200 ms for 40s. iTBS: 2 s train repeated 20 times, every 10 s for total of 190 s | Visuomotor adaptation | Found that intermittent theta burst improved adaptation, with fewer errors observed. In contrast, continuous theta burst stimulation slowed learning, specifically the rate of error reduction. | N/A | Active, sham |
| Matsugi et al., 2020 [89] | 14 | Healthy individuals; mean age = 19.5 yrs | TMS | Concurrent | Primary Motor Cortex (M1) Stimulation: Applied to the left M1 at the hotspot for the right FDI, with the coil orientation directed from anterior to posterior. Cerebellar Stimulation: Applied to the right cerebellar hemisphere, positioned 1 cm below and 3 cm to the right of the inion, with the coil oriented to induce an upward current. | single pulses | 90% | 10 trials/4 conditions | 20–40 ms after single-pulse | Cortical silent period (cSP) | cSP was significantly shorter in 1 |
No | No |
| Matsugi & Okada, 2017 [93] | 14 | Study (1) Healthy individuals; mean age = 19.7 yrs. Study (2) Healthy individuals, mean age = 22.9 yrs | tSMS | Offline | Motor Cortex (M1): Applied to the left M1 at the hotspot for the right FDI, with the figure-of-eight coil oriented to direct current from anterior to posterior. tSMS Over Cerebellum: Applied to the right cerebellum, with the cylindrical magnet positioned 1 cm below and 3 cm to the right of the inion. The magnet’s south pole was used, and it was held in place for 15 minutes using a stand with a clip. | single pulses | 90% | 10 consecutive stimuli | ISI of 0, 10, 20, 30, 40, 50, 60, 70, 80 ms | Cerebellar brain inhibition (CBI) | TMS delivered to the cerebellar hemisphere transiently reduces cerebellar inhibitory output but does not affect the excitability of the contralateral motor cortex. | SB | Active, sham |
| Mirdamadi & Block, 2021 [85] | 54 | Healthy young adults; ages 18–33 yrs | cTBS | Both | Motor Cortex (M1): Stimulation at the FDI hotspot on the left M1 with the figure-of-eight coil oriented to induce PA current. S1: Stimulation 1 cm posterior and 2 cm lateral to the left FDI hotspot on the left S1 with the coil handle 45° from the midline. Cerebellum (CB): Stimulation 3 cm lateral and 1 cm inferior to the inion on the right cerebellum with the coil handle pointing superiorly. | 5 Hz | 70% | 3 sessions for 10, 120, or 150 trials | Three pulses at 50 Hz, repeated at 5 Hz for 40 s, for a total of 600 stimuli | Proprioception | A cerebellar stimulation group demonstrated offline decrements in proprioceptive function. | No | sham |
| Odorfer et al., 2019 [86] | 16, 16 | Individuals with idiopathic cervical dystonia (CD) | cTBS | Both | M1: Stimulation at the left M1 “hotspot” for the right FDI with the coil handle pointing backward. PMd: Stimulation 2 cm anterior and 1 cm medial to the M1 hotspot with the coil handle pointing backward. Cerebellum: Stimulation 3 cm lateral and 1 cm inferior to the inion with the coil handle pointing upwards. | 50 Hz | 80% | 3 sessions/8 min. | 600 pulses/2 conditions for a total of 40 s | Cervical dystonia | The cortical silent period was decreased in patients with dystonia, and that stimulation of the lateral cerebellum impacting finger movements may impact disinhibition via purkinje cells. | SB | Active, sham |
| Pauly et al., 2021 [95] | 20 | Experiment (1) Healthy individuals; mean age = 27 yrs. (2) Healthy individuals; mean age = 27 yrs | rTMS, cTBS, and tDCS | Concurrent | M1: Left M1 “hotspot” for right FDI, with a 70 mm figure-of-eight coil, handle directed posteriorly. PMd: Left PMd, 2 cm anterior and 1 cm medial to M1, with a 25 mm figure-of-eight coil, handle directed posteriorly. Cerebellum: Right cerebellum, 3 cm lateral and 1 cm inferior to the inion, with a 70 mm figure-of-eight coil, handle directed upwards. | 1 Hz, 50 Hz | 90% | varied by method | varied by method | Plasticity | Cerebellar 1 Hz rTMS increases net corticospinal excitability and facilitatory interactions in cerebello-M1 and PMd-M1 pathways, whereas cerebellar PAS reduces corticospinal excitability. | No | Active, sham |
| Porcacchia et al., 2019 [87] | 12, 13 | (1) Individuals with cervical dystonia (disease duration: 8 |
iTBS and cTBS | Concurrent | M1: Left M1, at the “hotspot” for right FDI muscle, using a 70 mm figure-of-eight coil. Cerebellum: Right cerebellum, 3 cm lateral and 1 cm inferior to the inion, using a 70 mm figure-of-eight coil. | single pulses | 90% | 2 sessions | 20 trials for 6 conditions | Isolated cervical dystonia | In control subjects, they were able to induce increases and decreases in motor cortex excitability, where the same effect was not observed in the dystonia patient group. | No | No |
| Richard et al., 2017 [88] | 22 | Healthy individuals; mean age = 29.5 yrs. | TBS | Concurrent | Supplementary Motor Area (SMA)- determined through neuro-navigation and MRI-based localization. Coil Position: Midline with handle backward for posterior-to-anterior current direction. Cerebellum: Lobule VIII of the cerebellum. | 50 Hz | 80% | repeated every 200 ms continuously for 40 s | 600 pulses/three-pulse bursts | Gait initiation | Cerebellar stimulation was found to increase the co-activation of relevant muscle groups, along with decreasing execution duration when compared to sham. | DB | sham |
| Rurak et al., 2022 [82] | 59 | (1) Healthy younger adults; age range = 18–32 yrs. (2) Healthy older adults; age range = 60–82 yrs. | TMS | Concurrent | M1 Stimulation Site: Left primary motor cortex, determined by the site eliciting consistent MEPs in the FDI muscle. Coil Position: Figure-of-eight coil placed tangentially with a posterior-to-anterior current direction. Cerebellum Stimulation Site: Right cerebellar hemisphere, 3 cm lateral and 1 cm inferior to the inion, targeting the hand motor region. | 5 kHz | 90% | 4 experimental blocks with 24 trials | 2.5 min blocks |
Motor control | There was no difference found between unilateral and bilateral, however there was a statistical difference between unilateral stimulation and control. | DB | No |
| Shin et al., 2019 [83] | 1 | One 57 year old female with isloated focal leg dystonia. | TMS | Offline | Cerebellum Stimulation Position: Right cerebellum, 3 cm lateral and 1 cm inferior to the inion, aligning with the hand motor region of the cerebellar cortex. | 1 Hz | 80% | For 5 days, 600 stimulations per day | 20 trains with a duration of 30 s each, separated by 10 s | Focal leg dystonia | Dystonia was partially improved after stimulation delivered at 90% RMT 3 cm lateral and 1 cm inferior to the inion. | No | No |
| Spampinato et al., 2017 [80] | 32 | Healthy individuals; mean age = 23.9 yrs. | TMS | Concurrent | M1 Excitability: Right FDI Muscle: Stimulation over the motor representation using a figure-eight coil. Right TA Muscle: Stimulation over the motor representation (Experiment 1). CBI Measurements: Brainstem Motor Threshold: Over the inion with a double-cone coil. Conditioning Stimulus: 3 cm lateral to the inion over the cerebellum. Test Stimulus: Over the motor cortex (M1) for the right FDI or TA using a figure-eight coil. | single pulses at 5 s intervals | 5% below the brainstem motor threshold to the cerebellum | 11 sessions | 20/session | Motor learning | CBI changes only for the effector involved in the movement; learning-related changes in cerebellar–M1 connectivity reflect a somatotopy-specific interaction. | No | No |
| Tanaka et al., 2018 [94] | 29 | (1) Healthy individuals; mean age = 26.6 yrs. (2) Healthy individuals; mean age = 24.6 yrs. | TMS | Concurrent | Primary Stimulation Site: Left M1 (Motor Cortex) over the representation of the right FDI muscle using a figure-eight coil. Conditioning Stimulus (CS): Applied to the cerebellum 3 cm lateral to the inion using either a figure-eight or double-cone coil. Test Stimulus (TS): Delivered over the left M1 to elicit MEPs in the right FDI muscle. | single pulses | 90% | 10 trials per condition | 1 pulse per trial | Cerebellar brain inhibition | The cerebellum exerts facilitatory control over M1 excitability during imaginary muscle contraction but not during imaginary muscle relaxation. | N/A | N/A |
| Yildiz et al., 2018 [91] | 26 | (1) Individuals with multiple system atrophy-cerebellar subtype (MSA-C); predominated cerebellar dysfunction); mean age = 56.7 yrs. (2) Individuals with Alzheimers Disease (AD); mean age = 80.6 yrs. (3) Healthy individuals; mean age = 53.4 yrs. | rTMS | Offline | Left Motor Cortex (M1): Stimulation to elicit MEPs with a figure-of-eight coil, current from posterior to anterior. Cerebellum: Stimulation with a figure-of-eight coil, placed between the inion and mastoid process, current directed downward. Median Nerve: For peripheral conditioning, stimulating just above the motor threshold. | 1 Hz | 90% | single session/within 30 min | 600 pulses | Cortical excitability | After application of rTMS over the cerebellum in MSA-C patients, the maladaptive disinhibition and RT results showed an improvement compared to their previous results. | DB | No |
| Zabihhosseinian et al., 2020 [84] | 8, 8 | Healthy individuals; aged between 18 and 40 yrs. | TMS | Concurrent | First Stimulation (Cerebellum): Delivered by a double-cone coil over the ipsilateral cerebellum, 5 ms before the second stimulation. Second Stimulation (M1): Delivered by a figure-of-eight coil over the contralateral M1. Conditioning Stimulus: Applied over the cerebellum. Test Stimulus: Applied over M1 with the coil positioned to maximize the MEP amplitude in the target muscle. | single pulses | 40, 50, 60, 70, and 80% | 2.5-h session/15 TS and 15 CS-TS trials | 10 stimulations at each level | Motor performance | Both groups improved in accuracy following acquisition and retention, but the control group improved significantly more versus lower improvements for the fatigue group. | N/A | Control |
Abbreviations: TMS, transcranial magnetic stimulation; rTMS, repetitive transcranial magnetic stimulation; iTBS, intermittent theta burst stimulation; MEP, motor evoked potential; cTBS, cerebellar theta burst stimulation; FDI, first dorsal interosseous; RMT, resting motor threshold; ISI, interstimulus interval; tSMS, transcranial static magnetic field stimulation; PMd, dorsal premotor cortex; PAS, paired associative stimulation; CS-TS, conditioning stimulus- test stimulus.
Visuomotor tasks, or those relating to perception of environment and the necessary motor responses as well as visualization of movement, are classic tasks to recruit and test cerebellar functioning. In a visuomotor task, applied tACS at 1 mA at either gamma (70 Hz) or beta (20 Hz) to M1 and/or the right cerebellar hemisphere, only finding improvement in task performance for the gamma stimulation condition, particularly for lower performers [14].
Liew et al. (2018) [15] attempted to show that anodal tDCS over the
cerebellum would improve implicit learning over a visuomotor adaptation task,
with significant effects on target error and implicit learning, which
interestingly was worse during vertical conditions than horizontal screen task
conditions. Notably, these results were highly variable between individuals.
Another group compared a younger (18–29 years) and older cohort (66–84 years)
in a visuomotor rotation task, with 2 mA stimulation applied by 5
Marotta et al. (2021) [21] studied the concept of bodily
self-perception with a moving rubber hand illusion. Stimulation was applied by a
pair of 5
Koch et al. (2020) [79] focused on a combination of learning and visuomotor skills, and used theta burst stimulation over the cerebellum before a visuomotor adaptation task. They tested both continuous and intermittent theta burst stimulation (iTBS) and found that intermittent theta burst improved adaptation, with fewer errors observed [79]. In contrast, continuous theta burst stimulation slowed learning, specifically the rate of error reduction. Spampinato et al. (2017) [80] delivered a conditioning pulse of TMS to the cerebellum before a pulse over the motor cortex in a group of 32 subjects to measure cerebellar inhibition (CBI). The conditioning stimulus was delivered 3 cm lateral to the inion and 5 ms before a test stimulus targeting the contralateral M1 representation of first dorsal interosseous (FDI) or tibialis anterior (TA). They found that a conditioning pulse not only impacted the targeted muscle of the right hand, but also the inhibition/excitation balance of distal muscles such as the leg.
Given the complexity of the visuomotor network, and its functioning being foundational for motor learning, it makes sense that overall potential benefit is mixed. It appears that there are limited to no gains when applying this to healthy individuals in the context of improved visuomotor functioning. Although this is the case, instances of increased errors, lower functioning, or neuroanatomic impairments, the overall process can be improved. On the other hand, the literature suggests that there are benefits to specific aspects of the process (i.e., the motor component).
Reflexes are automatic motor responses after the exposure of specific types of external stimulation (e.g., physical touch, puff of air). While many of them fade over the course of development, when primitive reflexes remain, motor dysfunction tends to be prominent [96]. Bocci et al. (2018) [24] did not find an effect of cathodal cerebellar tDCS on a hand blink reflex; however, found that anodal cerebellar tDCS at 2 mA for 20 minutes significantly dampened the magnitude of the hand blink reflexes, interestingly implying the involvement of the cerebellum in defensive reflexes. Miyaguchi et al. (2019) [25] demonstrated that during tACS over both left M1 and the right cerebellar hemisphere of either 1 mA or 2 mA at 70 Hz, lower performers improved more in a visuomotor task during the stimulation conditions than sham. Taken together, it appears that cerebellar stimulation can influence the magnitude of reflexes. This can be a good first step to treat conditions in which reflexive responses are dysregulated, such as in gait disorders, ataxia, nystagmus, and others.
Mood disorders remain a common and difficult to treat area of neuropsychiatric conditions. Cerebellar neurostimulation is not traditionally considered as a treatment option, however, many studies show promise in treating mood as well as accompanying motor complaints. Cerebellar transcranial direct current stimulation (c-tDCS) was used to assess improvements in mood in clinically healthy patients [26]. Results revealed significantly elevated mood in participants following both single and repeated inhibitory tDCS conditions compared to sham, with increased mood from repeated c-tDCS. C-tDCS has also been evaluated in its ability to alleviate dystonic movements and improve mood in a case study involving a participant who previously underwent deep brain stimulation (DBS) [27]. Along with improvement in dystonic movements, they also found a significant reduction of depression symptoms on the Beck Depression Scale. This study suggests fronto-cerebellar tDCS influences mood in those with depression secondary to other conditions. Ferrari et al. (2021) [81] using cerebellar rTMS to investigate whether the cerebellum influences the modulation of motor cortical excitability in response to emotional stimuli found increased motor evoked potentials during the viewing of fearful faces compared to neutral faces. Their findings suggest the posterolateral (left) cerebellum modulates motor cortical response to negative emotional stimuli.
These studies suggest a fascinating interaction between cerebellar impact on mood and motor function, and show that TMS and tDCS may have similar effects under the right conditions. The associated positive impact appears to be present in both non-clinical and clinical populations. Overall, it appears that this may be an exciting frontier for neuromodulation researchers.
Cerebellar stimulation has been applied to many motor applications, including a variety of studies attempting to improve total body balance, posture, and lower limb strength. One study found that bilateral anodal tDCS over the cerebellum improved postural stability indices, greater than that of M1 stimulation alone [28]. Another found equal benefit in balance in older adults for cerebellar and M1 tDCS in both the mediolateral and anteroposterior balance directions [29]. Emadi Andani and colleagues [30] delivered stimulation over the cerebellar hemisphere ipsilateral to the dominant leg for 20 min at 2 mA in a group of 90 participants [31]. They tested cathodal, anodal, and sham stimulation along with differences in provisional or visual balance feedback. Sway was lessened with visual feedback, and effects remained longer when cathodal tDCS was applied, implying it supports the short-term maintenance of the positive effects of visual feedback therapy for balance, or learning how visual feedback can contribute to balance more quickly than without neurostimulation [30].
One group focused on learning of a whole-body balance task in a group of 40
subjects between 50 and 65 years old, where learning occurred but was not
statistically improved by tDCS applied at 2.8 mA through 5
Steiner et al. (2020) [33] looked at the effect of 2.8 mA tDCS cerebellar stimulation centered on the inion with return electrodes over the buccinator muscles, and found no effect on dynamic balance tasks or learning thereof. Foerster et al. (2017) [34] found an impairment of performance during a dynamic balance task due to cathodal cerebellar tDCS, but not an improvement of balance performance during the anodal condition. Stimulation was applied at 1 mA over the right cerebellar hemisphere and the deltoid muscle in the right arm, where anodal stimulation was applied for 13 minutes, and cathodal for 9 minutes. Anodal tDCS over M1 along with the cerebellum was observed to increase maximum voluntary contraction force, a measure of physical fitness, when compared to sham in a group of 25 healthy subjects. Stimulation was applied at 2 mA for 20 minutes over the bilateral cerebellum [35]. Ehsani et al. (2017) [36] also looked at postural control in older adults. They found that postural sway was significantly lessened, and balance scores significantly improved after anodal tDCS over the cerebellum at 1.5 mA for 20 minutes [36].
Craig & Doumas (2017) [37] compared anodal tDCS over the cerebellum in younger and older adults, with a higher level of balance difficulty for younger adults. In younger adults, stimulation 2 cm below the inion at 2 mA for 20 minutes had only offline and not task effects, while older adults receiving stimulation over the cerebellum and M1 were found to increase performance, but only during stimulation [37]. Another group also found that tDCS of the cerebellum improved postural steadiness during a platform vibration task when delivered at 1.5 cm below the inion at 1 mA for 20 minutes [38].
Inukai et al. (2016) [39] compared tDCS over the cerebellum, 2 cm below the inion at 2 mA for 20 minutes, with return electrodes in two separate experiments on the forehead and right buccinator muscle for sham, cathodal, and anodal stimulation. They reported a lowering of center of gravity sway during cathodal tDCS for both return electrode conditions. Chothia et al. (2016) [40] found that anodal tDCS of the cerebellum at 2 mA for 15 minutes reduced excitation in the descending pathway without inhibiting cervical propriospinal neurons.
In a study measuring cerebellar inhibition on the motor cortex, a factor important for motor control, dual site TMS was used to measure inhibition in younger subjects versus older adults [82]. They tested differences in improvement in balance in the elderly via bilateral, unilateral, or sham stimulation. There was no difference found between unilateral and bilateral, however there was a statistical difference between unilateral stimulation and control.
In summary, tDCS and TMS stimulation at combined motor and cerebellar locations tended to yield improved motor control, relative to sham or other stimulation locations. Notably, the effect was generally short-lived without post-stimulation effects. Taken together, these protocols only entailed a few sessions which may have not been enough for lasting change. The path forward may rely on confirming the most successful stimulation locations, patterns, and intensities as well as including individual anatomy factors.
Motor skill acquisition is highly relevant not only for normal adaptation and function but also for recovery post injury or disorder. Neurostimulation of the cerebellum may play a role in the rate or improvement of motor learning for a variety of conditions. For the below studies, if neurostimulation conditions are not included in the interest of space they can be found in Table 1. Shin et al. (2019) [83] found that in a single subject case study with isolated focal leg dystonia due to an acute cerebellar infarction who underwent 600 stimulations of rTMS for 5 days at 1 Hz on the right cerebellum, dystonia was partially improved after stimulation delivered at 90% resting motor threshold (RMT) 3 cm lateral and 1 cm inferior to the inion. One group found that neck fatigue impacted the extent to which TMS over the ipsilateral cerebellum impacted skill acquisition, concluding that motor fatigue may play a role in this circuit learning [84]. Theta burst magnetic stimulation can also be used for cerebellar stimulation. One group used continuous theta-burst stimulation (cTBS) over the left primary somatosensory cortex (S1), right lateral cerebellum, or a sham condition. Both the S1 and cerebellar stimulation conditions demonstrated interruptions in learning, the S1 group in impaired proprioceptive function and the cerebellar group in decrements in proprioceptive function. Stimulation was delivered at three pulses at 50 Hz, repeated at 5 Hz for 40 s at 70% resting motor threshold [85]. Odorfer et al. (2019) [86] used continuous bilateral continuous theta-burst stimulation, with each side stimulated individually, at 80% active motor threshold bilaterally for 40 seconds. They found that the cortical silent period was decreased in patients with dystonia, and that stimulation of the lateral cerebellum impacting finger movements may impact disinhibition via purkinje cells [86]. Porcacchia et al. (2019) [87] applied a paired-pulse transcranial magnetic stimulation over the right cerebellum and left motor area to a group of 12 subjects with isolated cervical dystonia. In control subjects, they were able to induce increases and decreases in motor cortex excitability, where the same effect was not observed in the dystonia patient group, implying a need for an intact circuit to modify [87]. Bocci et al. (2020) [41] applied 2 mA anodal cerebellar transcranial direct current stimulation (cTDS) to four subjects with Huntington’s Disease and found that clinical scores improved, in particular symptoms related to dystonia. These effects lasted approximately 4 weeks [41].
Cerebellar function has been found to be tied to acquisition and maintenance of
motor skills, which is a separate focus from that of performance during the task
itself. C-tDCS was used to assess cognitive learning among healthy participants
during motor tasks. One study investigated motor-sequence-learning and found
cathodal stimulation to the lateral posterior cerebellum positively influenced
learning a new motor sequence, while anodal stimulation had the opposite effect
[42]. Regarding probabilistic and implicit categorized learning tasks among
healthy participants [43, 44], authors found c-tDCS did not significantly impact
learning on these complex tasks compared to sham. Similarly, a 2021 study found
that c-tDCS did not significantly increase performance on a complex category
learning task [45]. Ferrucci et al. (2019) [46] did not find that
cerebellar tDCS had any impact on spatial navigation skills in a group of healthy
subjects. Jongkees et al. (2019) [47] observed that anodal tDCS of the
cerebellum, compared to cathodal and sham, demonstrated increased task response
latencies, indicating behavioral interference, possibly revealing effects of
anodal cerebellar tDCS on motor task learning. Stimulation for all conditions was
applied with a 5
In a group of 100 healthy subjects, five stimulation groups were tested for delay during eyeblink conditioning; anodal tDCS of the cerebellum, cathodal tDCS of the cerebellum, anodal tDCS of the dorsolateral prefrontal cortex (dlPFC), cathodal tDCS of the dlPFC, and sham. Renewal effects were found for all subjects except those that received anodal tDCS of the dlPFC during extinction. No effects for dlPFC or cerebellar tDCS were shown for extinction. As stated by the authors the methods laid out in this study may not adequately test cerebellar involvement in neural extinction [49]. Leow et al. (2017) [50] tested implicit remapping of sensorimotor processes during adaptation and found that cerebellar anodal tDCS improved this skill when applied at 1.5 mA to the right cerebellum.
Evidence of TMS improvement in motor learning topics implies interaction of TMS
with an existing neural adaptation mechanism, such as that involved with brain-derived
neurotropic factor (BDNF). One group looked at subjects with the BDNF Val66Met polymorphism
as it is associated with motor skill learning. Subjects performed an eyeblink conditioning
task and received sham, anodal, or cathodal tDCS, or participated in a
vestibulo-ocular reflex adaptation task and received sham and anodal tDCS.
Stimulation at 2 mA was delivered through saline-soaked 5
Summers et al. (2018) [52] also examined cerebellar activity, its
modulation with tDCS and the effect on task training. Anodal stimulation was
applied at 2 mA for two 15 min sessions and tracking accuracy along with motor
excitability was measured [52]. Effects on corticospinal excitability were noted,
without an effect on performance. Mizuguchi et al. (2018) [53] applied 2
mA cerebellar tDCS during a dart throwing task. Improvement was seen with only
cathodal and not anodal or sham tDCS, and only for lower performers with cathodal
stimulation, supporting the idea of a ceiling effect for neural stimulation.
Another study found in a complex overhand throwing task that cerebellar tDCS
enhanced motor learning via decline in errors. They studied a total of 42 total
subjects, with stimulation of 25 min, 2 mA, anode 3 cm to the right of the inion,
cathode to the right buccinator muscle ipsilateral to the right hand with 5
One group was concerned with the effects of anodal tDCS on bimanual adaptive motor tasks. They found that bilateral cerebellar stimulation 2.5 cm from the inion resulted in faster movement and lower error compared to sham and parietal stimulation [55]. The same group studied anodal tDCS applied to the cerebellum at 1.5 mA 3 cm lateral to the inion for 15 min, was observed to increase alpha, beta, and sensorimotor rhythm power bands as observed by electroencephalogram (EEG), along with an increase in offline learning [56]. Shimizu et al. (2017) [57] found that anodal tDCS enhanced motor learning performance, and cathodal tDCS inhibited learning. Stimulation was applied 2 cm below the inion at 2 mA [57]. Another group was interested in online, during practice of a new motor skill, and offline, after training sessions, motor learning in a group of older adult subjects. Anodal stimulation was applied at the cerebellum at 2 mA for 20 minutes, and only for 30 seconds in the sham group. Stimulation on the cerebellum was associated with lower error rate compared to sham and a faster reaction time, although reaction time was not significant [58].
Miyaguchi et al. (2019) [59] studied the phase relationship of tACS
specific improvement of motor learning via interference with cerebellar circuits.
They applied stimulation with 5
Delving into mechanisms, Liebrand et al. (2020) [61] found that tDCS over the right cerebellum was accompanied by functional magnetic resonance imaging (fMRI) measured increased activity in right M1, left cerebellar lobule VI, left inferior frontal gyrus, and right inferior parietal lobule. With slightly different results than many studies, Kumari et al. (2020) [62] found that anodal cerebellar tDCS at 2 mA for 15 minutes 3 cm lateral to the inion during a split-belt treadmill task did not increase task adaptation, however it did increase maintenance of learned walking patterns, still implying a role in anodal tDCS of the cerebellum in motor adaptation.
Wessel et al. (2020) [63] studied the application of 50 Hz tACS over the left cerebellum during grip force motor skill learning task, further investigated by TMS stimulation and the resulting resting state fMRI connectivity in a group of 15 subjects. tACS stimulation was applied over the left cerebellum 3 cm lateral to the inion at 2 mA for 20 minutes, with a return electrode over the ipsilateral buccinator muscle. Double-pulse TMS stimulation was performed with a posterior to anterior orientation at 80% motor threshold over the first dorsal interosseous muscle. Statistical analyses did not find an effect of tACS on task learning, and TMS influenced fMRI analyses over the motor circuits also did not display an effect. Another group compared gamma (50 Hz) to delta (1 Hz) stimulation tACS over the right cerebellum during a reaction time right hand task. Gamma stimulation was found to impair participant performance in repeated tasks, but not randomized tasks [64]. Schubert et al. (2021) [65] applied 10 Hz tACS to left M1 and right cerebellum during a learning task, observing diminished learning after cerebellar stimulation compared to sham but not M1 stimulation.
Overall, these studies support a possible role of cerebellar neurostimulation, both TMS and tDCS or tACS during the initial stages of motor learning and conditioning, although stimulation location is still to be optimized and is likely highly disorder dependent as not all studies show a significant effect. Concurrent motor cortex and cerebellar stimulation likely impact motor learning through synchronized activity in these regions, or improved connection between the two. Exact stimulation locations over the cerebellum are highly varied, and with current stimulation technology the stimulation area may not be focal enough to determine the exact “best” stimulation location, if there is one. Individual functional connectivity and anatomy needs to be included to improve stimulation techniques further.
Gait refers to a walking pattern, usually focused on arm and step velocity, length, time, width, and symmetry. Gait stability and adjustment to different circumstances can be modified via cerebellar neurostimulation. One group studied gait via applied cathodal tDCS to the right cerebellar hemisphere during an adaptive walking task and found a disruption in gait adaptation when stimulation was applied at 2 minutes at 2 mA 1 cm below the inion and 3 cm to the right of the inion with an anodal electrode over the right buccinator muscle [66]. Interestingly, they found non-independent processing of spatial and temporal domains during gait in the right cerebellum, facets which are often evaluated independently and should be evaluated together during complex task study.
Naro et al. (2017) [67] tested different cerebellar transcranial alternating current stimulation frequencies over the right cerebellar hemisphere. They found that the frequencies of 50 and 300 Hz, but not 10 Hz, modulated M1-leg excitability [67]. However, they did not observe a gait perturbation for any group. TACS over the left cerebellum 3 cm left of the inion has been observed to reinforce a gait rhythm, delivered at the approximated frequency of the individual gait at 2 mA. Subjects also switched to a reversed gait frequency when the stimulation phase was inverted, all compared to sham. TACS may be able to modulate rhythm generation via cerebellar circuits [68].
Continuous theta burst transcranial magnetic stimulation of the cerebellum has been used to study gait initiation. Cerebellar stimulation was found to increase the co-activation of relevant muscle groups, along with decreasing execution duration when compared to sham. Stimulation was performed at an individual level with a neuro-navigation system, bilaterally targeting lobule VIII with the handle pointing vertically. 600 stimuli were delivered at 80% motor threshold in three pulse bursts at 50 Hz, every 200 ms [88].
In these studies, TMS, tACS, and tDCS were all found to potentially impact processing of gait via neurostimulation of the cerebellum. Many of the stimulation locations, for example 1 cm below the inion vs. 3 cm left of the inion may have overlapping stimulation locations based on the electric field dependent on exact stimulation tool, strength, distance of cerebellum from scalp (which may be entirely different from the commonly used motor threshold based on distance of the motor cortex from the scalp), and many other factors.
Neurostimulation of the cerebellum has also been used to study hand coordination and force of grip. John et al. (2017) [69] applied anodal tDCS (2 mA for 22 minutes) to the lateral cerebellum in a group of patients with cerebellar degeneration, without observation of any change in grip strength in the patient group or controls. Another group looked specifically at EEG rhythms in the brain after applying 1.5 mA anodal tDCS to the lateral cerebellum halfway between the mastoid and the inion. They found a decrease in the delta band at electrodes F2 and T8, but no change in entropy ratios or laterality coefficients in a 14 channel portable EEG headset [70].
Küper et al. (2019) [71] studied 48 participants while they completed a finger tapping task in a magnetic resonance imaging (MRI) scanner with real time tDCS stimulation in the scanner. Stimulation was performed at 1.8 mA with an electrode 3 cm lateral to the inion with the other on the right buccinator muscle and checked for location within the MRI for lateral cerebellum coverage. They did not observe an increase in MRI signal in the cerebellum for either anodal, cathodal, or sham stimulation, however cathodal stimulation was observed to increase the signal in the dentate nuclei, while anodal stimulation showed the opposite in a trending manner confirming the possibility of excitatory or inhibitory effects being induced by cerebellar stimulation of opposite charge. Kamali et al. (2019) [72] studied improvement of pistol shooting via tDCS targeting of the cerebellum and dorsolateral prefrontal cortex. Compared to sham, they found that tDCS improved the average shooting score and improved accuracy when anodal stimulation was applied at 1 mA for 20 minutes over the right cerebellum, 1 cm below and 3 cm lateral to the inion with a cathode electrode over the left dlPFC [72]. Another group compared anodal tDCS of the cerebellum at 1.5 mA before, during, and after a two-handed coordination task also compared to sham. The during-task and after-task stimulation groups observed significant improvements to the before-task and sham stimulation groups [73]. Akremi et al. (2022) [74] applied anodal transcranial direct current stimulation for 20 minutes at 2 mA to a group of 10 children with developmental coordination disorder. They found that this stimulation reduced the number of errors made during the motor sequence task although it did not impact coordination or learning observed outside of the stimulation task [74].
Matsugi et al. (2020) [89] observed that cerebellar TMS shortened the cortical silent period resulting from TMS over the motor cortex by approximately 30 ms. Stimulation was delivered to the cerebellum with a double cone coil 1 cm below and 3 cm to the right of the inion at 90% of the resting motor threshold [89].
Neurostimulation of the cerebellum may have useful effects on grip and reaction time. The findings of Kamali et al. (2019) [72] increasing reaction time in a group of healthy subjects, even if effects are slight, are fascinating as improving performance in a healthy group has often been found to be more difficult than improving impaired performance, such as in aging.
As an adaptive controller, the cerebellum plays a large role in not only direct modification of specific neural activity, but also in general excitation and inhibition, and therefore plasticity, both within the cerebellum and beyond [97]. The following articles are concerned with neurostimulation of the cerebellum along this theme. These articles may be related to previous sections (motor, language, etc.) but are primarily concerned with excitation and inhibition and so are placed in their own section.
Doeltgen et al. (2016) [75] demonstrated that anodal tDCS at 2 mA for 20 minutes over the cerebellum (3 cm lateral and 1 cm inferior to the inion) reduced cerebellar brain inhibition, but did not impact short afferent inhibition or impact reflexes. Jalali et al. (2018) [76] applied tDCS to the cerebellum and then investigated gamma-aminobutyric acid (GABA) and glutamate with magnetic resonance spectroscopy (MRS). Although they reported high levels of variability, they did not find group changes of GABA and glutamate due to cerebellar tDCS [76]. However, their application of tDCS did coincide with motor memory retention and this was correlated with a decrease in cerebellar glutamate.
Cerebellar to prefrontal cortex connectivity plays a role in many functions such as motor coordination and cognition, and can be modified by TMS observed by EEG and magnetic resonance spectroscopy, to measure GABAergic inhibition, as demonstrated by Du et al. (2018) [90]. They report that cerebellar evoked prefrontal synchronization was positively associated with working memory but negatively associated with coordinated rapid finger tapping, possibly suggesting competing resources for ideal performance in each category.
Applying these principles to a patient group, Yildiz et al. (2018) [91] compared low-frequency application of TMS over the cerebellum in subjects with multiple system atrophy, cerebellar subtype (MAS-C), Alzheimer’s disease, and healthy controls. They found that in the MAS-C group cerebellar TMS, maladaptive motor cortex disinhibition was lessened, and reaction times improved, with no effect in the Alzheimer’s or control groups. Hassan et al. (2019) [92] applied TMS to the cerebellum with the goal of depressing cerebellar activity in persons with hepatic encephalopathy. Lowered cerebellum inhibition was achieved via TMS, correlating with disease severity, and implying a connection of disease state with GABAergic neurotransmission in the cerebellum. In a study measuring cerebellar inhibition on the motor cortex, a factor important for motor control, dual site TMS was used to measure inhibition in younger subjects versus older adults [82]. They tested differences in improvement in balance in the elderly via bilateral, unilateral, or sham stimulation. There was no difference found between unilateral and bilateral, however there was a statistical difference between unilateral stimulation and control.
Matsugi & Okada (2017) [93], in a different approach, used a compact cylindrical NdFEb magnet to induce transcranial static magnetic field stimulation to decrease cerebellar brain inhibition in a transient manner, without impacting resting motor threshold or motor evoked potentials. Spinal excitability can also be modulated by cerebellum neurostimulation. Matsugi & Okada (2020) [77] also found that the H-reflex ratio, or the response of the right soleus muscle by electrical stimulation of the right tibial nerve, was increased by anodal cerebellar tDCS, reduced by cathodal stimulation, both compared to sham. The same group applied TMS over the cerebellum along with TMS of M1 compared to TMS of only M1 during visualization of muscle contraction and relaxation [94]. They found that the cerebellum can be shown to exert control over M1 excitability during contraction, but not relaxation implying a priming role.
Pauly et al. (2021) [95] compared 1 Hz TMS, theta-burst stimulation, paired associative stimulation, and tDCS, all applied to the cerebellum. Paired associative stimulation was found to reduce cortical excitability in the motor cortex, while TMS increased motor thresholds and increased cerebellum to motor cortex pathways. Unlike other studies, they did not observe an impact of tDCS or cTBS. Petti et al. (2017) [78] was concerned with cerebellar tDCS on brain networks. They applied stimulation over the right cerebellar hemisphere and evaluated EEG activity and network organization [78]. Cathodal stimulation appeared to have minimal changes from sham, however anodal stimulation was observed with lateral synchronization in the sensorimotor area, as well as network segregation in sensory-motor rhythms.
The reported changes in excitation and inhibition likely have effects beyond just the regions studied. Given the interconnectedness of the cerebellum with the rest of the brain and its role as an adaptive controller, it follows reason that stimulation of specific regions of the cerebellum would modify excitability or inhibition in connected regions, whether that is in motor thresholds, accessible working memory, or direct measurements of glutamate and other neurotransmitters. These studies emphasize the variable effect of cerebellar stimulation, and how the adaptive circuitry and connectivity must be taken into account.
Non-invasive stimulation of the cerebellum can have a myriad of effects on motor function and circuitry. The applicability of these techniques in functional areas from balance to recovery after stroke are clear. Despite abundant evidence, neurostimulation of the cerebellum is still not employed to its full potential, and the field is barely beginning to realize the possibilities of neurostimulation of the cerebellum as a door to non-invasive modification of the rest of brain function.
Many of the treatments for the disorders and circuit malfunctions detailed above include stimulation at similar general locations in the cerebellum, such as over the inion, the right hemisphere of the cerebellum, or 2 cm below the inion. Many do not include individual differences in functional connectivity or anatomy. Taking advantage of the cerebellum’s innate interconnectedness with the rest of the brain as well as its role as an adaptive controller or error modulator is a logical step for noninvasive neuromodulation of many disorders, not just those detailed here. The studies detailed above clearly indicate potential for cerebellar stimulation for a variety of disorders, including gait recovery, motor adaptation, reaction time, mood associated with motor disorders, and many more. It is shown that it is possible to improve motor learning rate and retention, improve gait especially in elderly populations, and add to inherent balance ability for a short time after stimulation is concluded.
The density and interconnectedness of the cerebellum itself allows for modification of various circuits by just a slight adjustment of neurostimulation target. The use of a common stimulation site for a myriad of disorders, such as 3 cm lateral to the inion, implies that stimulation of the cerebellum is acting via circuitry of the cerebellum itself, and not bypassing its inherent functions. Individual factors must also be taken into account, where subject specific functional or structural connectivity may impact response to cerebellar stimulation. This is of particular concern when individuals have conditions which are directly associated with motor symptoms not associated with specific cerebellar impairment. Evidence of cerebellar neurostimulation improving motor function via cerebellar targeting is promising for non-motor function to be improved by cerebellar targeting, as evidenced by the interaction of mood and motor symptoms seen above [26, 27]. The study of motor systems with cerebellar neurostimulation also provides the opportunity to impact the fields of self perception and proprioceptive drift [21].
Neurostimulation of the cerebellum and response of individual disorders to neurostimulation is dependent on adequate targeting of the circuits of interest. If a specific disorder requires excitatory neurostimulation at an individual specific location for a certain amount of time, neurostimulation studies may see a lack of results if they either (1) missed the target location, (2) delivered inhibitory stimulation rather than excitatory, (3) did not deliver stimulation for an adequate period of time to introduce neural remodeling, along with potential biological complications such as the subject’s neural system lacking sufficient innate plasticity. Part of the mechanism of neurostimulation in many of these studies depends on the Hebbian learning rule of firing together and wiring together. Especially with the goal of modulating motor circuits neurologically distant from the cerebellum, a full understanding of the circuitry of the disorder is necessary.
As the effect of cerebellar stimulation is through circuit connections, for example impacting neural activity in the cerebral cortex and thalamus via stimulation of the cerebellum routed through the dentate nucleus for stroke rehabilitation [98]. Given this mechanism via circuit modulation and the widespread connectivity of the cerebellum, the potential applications of cerebellar neurostimulation are enormous, applying not only to motor applications but cognitive, learning, social, and affective functions. As an example in visuomotor cases, some of which are detailed here, cerebellar stimulation may modify motor function by targeting of the dorsolateral premotor cortex via network connections including the putamen, the superior parietal lobule, supplementary motor area and primary motor cortex [99]. Neural changes due to cerebellar neurostimulation can be seen in study such as Liebrand et al. (2020) [61], where putamen-cerebellar connectivity was modulated by cerebellar tDCS. Spampinato et al. (2017) [80] demonstrated strengthening of M1-cerebellar connectivity in a somatotopic specific way, impacting not only the hand target of interest but also untargeted leg muscles. Grami et al. (2022) [22] focused on reconfiguration of large-scale neural networks, showing that cerebellar neurostimulation can impact regions involved in both motor execution and motor visualization.
Overall, less is known regarding how altering the stimulation protocol influences outcomes, particularly in the context of cerebellar stimulation. In the context of single pulse TMS protocols, cortical excitability is often evidenced by immediate behavioral demonstrations with repeated sessions given in hopes of initiating more lasting behavior change. For 1 Hz TMS protocols (i.e., low frequency protocols), studies focused on neuronal inhibition resulting in decreases in observable behaviors such as ameliorating muscle contractions, a hallmark feature of dystonia, or reducing overactive functional connections. When considering high frequency TMS protocols, such as 5 Hz or theta burst stimulation (TBS), the overall goal is increasing activity in certain areas or pathways. What is challenging is that regardless of the specific protocol being used, there is a significant gap in the literature regarding what is necessary in order to maintain the durability of these effects. Summarizing and sharing findings in a concise way can help move the field forward to optimize the parameters to ensure desired outcomes and lasting change. Although promising, there is much research to be done to optimize cerebellar neurostimulation. Ceiling effects must always be taken into account, found for every stimulation location the cerebellum being no exception. Factors such as motor fatigue are also important, as the cerebellum does not operate outside the limits of the system [84].
The cerebellum is an extremely useful modulation tool that must be used within the limits of the specific function of interest. For example, stimulation may be able to increase grip control and coordination, but it cannot increase grip strength beyond the muscles and motor cortex which it is connected to. Neurostimulation of the cerebellum may be most useful in cases of reduced function related to network dysfunction rather than optimizing normal function. As an adaptive controller, much of the cerebellar role comes into play with learning novel tasks, maintaining new skill acquisition, and adjusting excitability/inhibition of circuits and therefore circuit function generally. Learning itself is not a simple task, and complex interactions between acquisition, offline and online learning, and skill maintenance can be studied through neurostimulation of the cerebellum. Many of the studies above report a particular effect on learning during stimulation itself, however in some studies including younger subjects increased offline effects were observed, likely due to an adjustment of the excitability/inhibition balance in the involved circuits.
Further insights into cerebellar function and neuromodulation potential will be revealed as the field moves into the possibility of stimulation being conducted within the scanner as demonstrated by Küper et al. (2019) [71]. Although steps forward in the field of cerebellar neurostimulation are promising, we have yet to fully understand and utilize the potential of this door into the brain. Future considerations in the study of cerebellar stimulation include the inclusion of functional and direct connectivity at an individual level, improving selectivity of neurostimulation techniques applied, including visualizations of the electric field for each stimulation condition, which will also improve selection of varying stimulation sites, as well as careful consideration of the target groups and whether stimulation may be helpful in their specific case.
TMS, transcranial magnetic stimulation; CBI, cerebellar inhibition; iTBS, intermittent theta burst stimulation; MEP, motor evoked potential; dmPFC, dorsomedial prefrontal cortex; rTMS, repetitive transcranial magnetic stimulation; tDCS, transcranial direct current stimulation; tACS, transcranial alternating current stimulation.
Data sharing is not applicable as no data were generated or analyzed.
KW: Visualization, Investigation, Writing, original draft, Writing, review & editing. NW: Investigation, Writing, original draft, Writing, review & editing. BW: Writing, review & editing. MG: Writing, review & editing. NR: Writing, review & editing. SG: Investigation, Writing, original draft, Writing, review & editing. HA: Investigation. MM: Visualization, Investigation, Writing, original draft, Writing, review & editing. BW, MG, NR, and HA were also responsible for collecting and sorting references and additionally responsible for designing and drawing the figures/tables. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
This work was supported by the Sierra Pacific Mental Illness Research, Education, and Clinical Center (MIRECC) at the VA Palo Alto Health Care System. For full details of the program sites please see the associated website at the Sierra Pacific (VISN 21) MIRECC - MIRECC / CoE (va.gov).
This research received no external funding.
The authors declare no conflict of interest.
Supplementary material associated with this article can be found, in the online version, at https://doi.org/10.31083/j.jin2310195.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.


