1 Department of Neurology, The Affiliated Hospital of Southwest Medical University, 646000 Luzhou, Sichuan, China
Academic Editor: Gernot Riedel
Abstract
Background: Camptocormia is one of the most common postural disorders
of Parkinson’s disease (PD) which has limited treatment options. In this review,
we summarize the efficacy of deep brain stimulation (DBS) for camptocormia in PD.
Methods: The PubMed (https://pubmed.ncbi.nlm.nih.gov/) and EMBASE
databases (https://www.embase.com/)
were searched for the terms “Parkinson Disease” and “camptocormia” in
combination with “deep brain stimulation”. We then explored the efficacy of DBS
for camptocormia by statistical analysis of the bending angle, the Unified
Parkinson’s Disease Rating Scale III (UPDRS-III) and L-dopa equivalent daily dose
(LEDD), and by evaluating the prognosis after DBS. Results: Twenty
articles that reported results for 152 patients were included in this review.
These comprised 136 patients from 16 studies who underwent subthalamic nucleus
deep brain stimulation (STN-DBS), and 13 patients from 3 studies who underwent
globus pallidus internus deep brain stimulation
(GPi-DBS). One study used both STN-DBS (2
patients) and GPi-DBS (one patient). After 3–21 months of follow-up, the mean
bending angle during the Off-period was significantly reduced compared to pre-DBS
(31.5
Keywords
- camptocormia
- Parkinson's disease
- deep brain stimulation
Camptocormia is a common postural deformity in Parkinson’s disease (PD), with a prevalence estimated to range from 3% to 18% [1]. It is described as forward flexion of the thoracolumbar spine, which aggravates in the standing position and disappears in the supine position. In addition to PD, other potential etiologies for camptocormia include axial myopathy, joint degenerative diseases, and atypical PD such as multiple system atrophy and progressive supranuclear palsy. However, the prevalence of camptocormia in PD (22.5%) is much higher than in other diseases [2]. Camptocormia may aggravate rapidly with the progression of PD and will be accompanied by dyskinesias, falls, severe back pain, difficulty in eating, and even respiratory failure, all of which significantly impact the quality of life and increase motor disability and care burden.
Camptocormia can be classified into lower
camptocormia (total camptocormia angle
The pathogenesis of camptocormia is unclear, with several potential contributing factors including dystonia, myopathy, proprioceptive disintegration, medication effects and soft tissue changes. Paraspinal dystonia is often observed in the early stage of camptocormia, followed gradually combined with focal myopathy and soft tissue changes [5].
Camptocormia usually appears in the advanced stage of PD, and has generally proved refractory to pharmacological treatment. Some studies have reported that dopaminergic drugs may even induce or aggravate camptocormia, especially high-dose and long-term use of levodopa and dopamine receptor agonists [6, 7]. The effectiveness of other therapeutic options such as lidocaine injection, botulinum toxin injection and rehabilitation training remains controversial. Complications from spinal surgery are common [5]. In recent years, deep brain stimulation (DBS) has been used to treat camptocormia in PD. Given the inconsistent results reported so far, we conducted this meta-analysis to assess the efficacy of DBS for camptocormia in PD patients.
We examined 46 articles from PubMed and 104 articles from EMBASE. The inclusion criteria were: (1) definitive diagnosis of PD and camptocormia, with the camptocormia related to PD; (2) Intervention with DBS; (3) English language study. The exclusion criteria were: (1) Review article; (2) Articles with missing or non-extractable data; (3) Duplicate articles or those with repeat clinical data. Twenty studies containing a total of 152 patients [3, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26] met all of the criteria and were included in the analysis. Three studies used globus pallidus internus deep brain stimulation (GPi-DBS), 16 used subthalamic nucleus deep brain stimulation (STN-DBS), and one study used both (Fig. 1). And the CRD number of the systematic review on the PROSPERO is 353766.
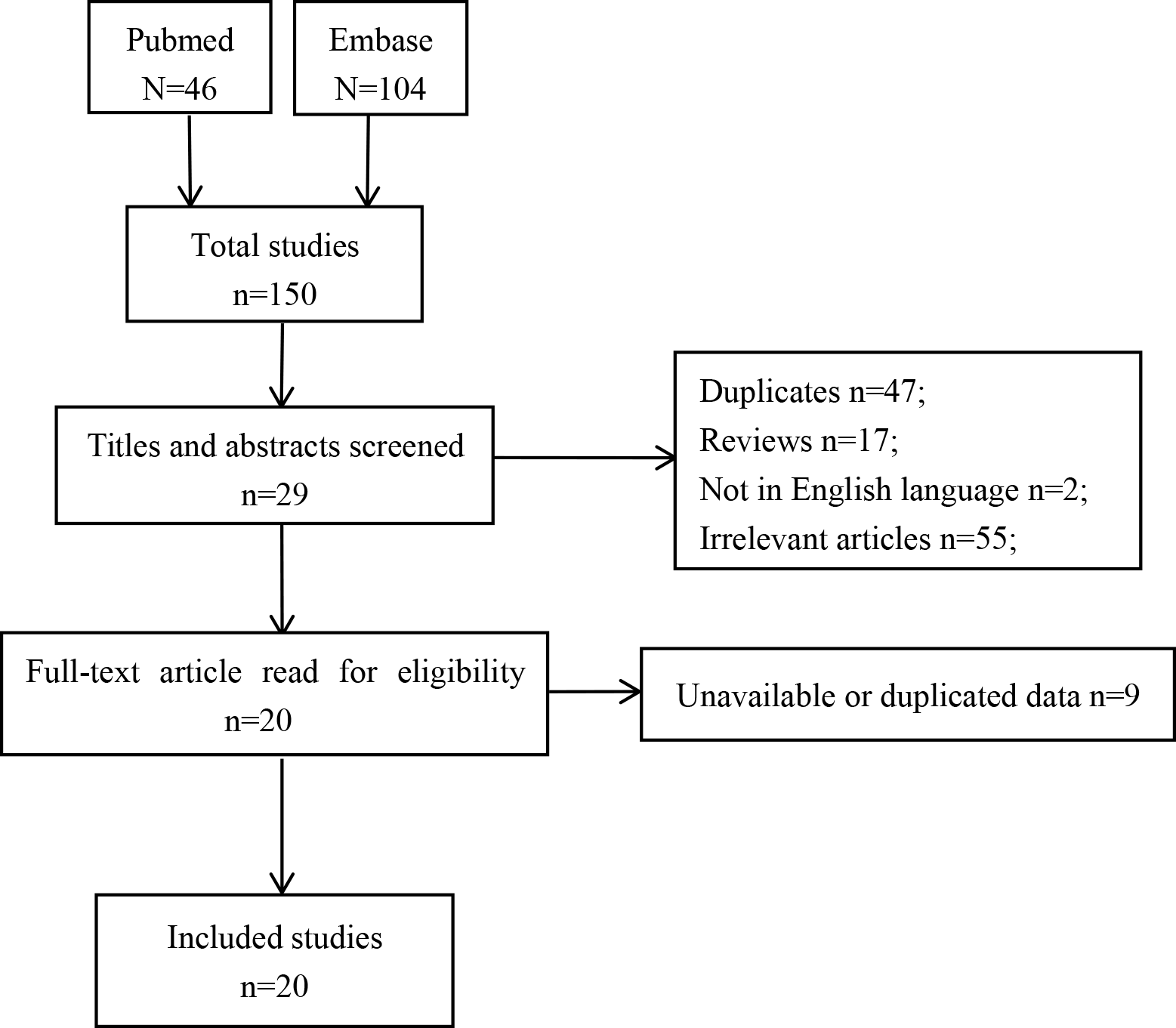 Fig. 1.
Fig. 1.Flowchart of study selection.
Information extracted from the selected papers included authors, year of publication, type of research, number of participants, age, gender, PD duration, camptocormia duration, bending angles (in both On and Off periods), the Unified Parkinson’s Disease Rating Scale III (UPDRS-III) score (in both On and Off periods), mean follow-up time, and the L-dopa equivalent daily dose (LEDD) assessed both pre- and post-DBS (Tables 1,2, Ref. [3, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26]).
| Authors | Years | Study Type | Method | Participants | Mean age (Years) | Gender (M/F) | Duration of PD (Years) | Duration of Camptocormia (Months) | Evidence Level | Follow-up (Months) |
| Lai et al. [3] | 2021 | Retrosp obs cohort | STN | 10 | N | N | N | N | III | 6.0 |
| Lai et al. [8] | 2021 | Retrosp obs cohort | GPi | 11 | N | N | N | N | III | 7.3 |
| Liang et al. [9] | 2020 | Prosp trail | STN | 15 | 62.5 |
7/8 | 10.5 |
25.2 |
II | 6 |
| Schlenstedt et al. [26] | 2019 | Retrosp obs cohort | STN | 27 | N | N | N | N | III | 6–12 |
| Sakai et al. [10] | 2017 | Retrosp obs cohort | STN | 14 | 51.9 |
8/6 | 13.1 |
40.8 |
III | 6 |
| Yamada et al. [11] | 2016 | Prosp trail | STN | 17 | 66.4 |
7/10 | 12.9 |
48.2 |
II | |
| Schulz-Schaeffer et al. [12] | 2015 | Retrosp obs cohort | STN | 25 | 67.1 |
21/4 | 15.4 |
62.4 |
III | 6–12 |
| Umemura et al. [13] | 2010 | Case series | STN | 8 | 65.1 |
2/6 | 15.5 |
N | IV | 12 |
| Sako et al. [14] | 2009 | Case series | STN | 6 | 51.2 |
2/4 | 9.0 |
N | IV | N |
| Soares et al. [15] | 2019 | Case series | STN | 2 | 65.5 |
1/1 | 10.5 |
5.3 |
IV | 8–12 |
| Roediger et al. [16] | 2019 | Retrosp obs cohort | STN | 3 | N | N | N | N | III | 15.4 |
| Pandey et al. [17] | 2016 | Case report | STN | 1 | 58 | 1/0 | 7 | 60 | IV | 3 |
| Ekmekci et al. [18] | 2016 | Case report | STN | 1 | 51 | 0/1 | 10 | N | IV | 6 |
| Lyons et al. [19] | 2012 | Case report | STN | 1 | 63 | 0/1 | 19 | 228 | IV | 3 |
| Asahi et al. [20] | 2011 | Case series | STN | 4 | 62.8 |
2/2 | 11.5 |
62.4 |
IV | 25.8 |
| Capelle et al. [21] | 2011 | Case series | STN | 2 | 69.0 |
2/0 | 13.5 |
N | IV | 21 |
| Yamada et al. [22] | 2006 | Case report | STN | 1 | 71 | 0/1 | 11 | N | IV | 3 |
| Hellmann et al. [23] | 2006 | Case report | STN | 1 | 53 | N | 25 | 228 | IV | 10 |
| Capelle et al. [21] | 2011 | Case report | GPi | 1 | 64 | 1/0 | 10 | N | IV | 21 |
| Thani et al. [24] | 2011 | Case report | GPi | 1 | 57 | 0/1 | 13 | 24 | IV | 14 |
| Micheli et al. [25] | 2005 | Case report | GPi | 1 | 62 | 1/0 | 9 | 2 | IV | 3 |
| Obs, observational; Prosp, prospective; Retrosp, retrospective; PD, Parkinson’s disease; STN, subthalamic nucleus; GPi, globus pallidus internus; F, female; M, male; N, not available. | ||||||||||
| Authors | Method | Bending angle (Off) | Bending angle (On) | UPDRS-III (Off) | UPDRS-III (On) | LEDD | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Pre-DBS | Post-DBS | Pre-DBS | Post-DBS | Pre-DBS | Post-DBS | Pre-DBS | Post-DBS | Pre-DBS | Post-DBS | ||
| Yamada et al. [11] | STN | 84.0 |
54.8 |
59.6 |
51.9 |
42.4 |
18.8 |
23.5 |
16.5 |
612.7 |
378.9 |
| Sakai et al. [10] | STN | N | N | 55.5 |
38.7 |
45.5 |
41.5 |
23.8 |
19.5 |
658.9 |
302.9 |
| Schlenstedt et al. [26] | STN | 42.2 |
35.5 |
35.2 |
32.9 |
N | N | N | N | N | N |
| Liang et al. [9] | STN | 42.0 |
8.5 |
14.6 |
7.1 |
55.1 |
24.3 |
N | N | 965.7 |
459.0 |
| Lai et al. [3] | STN | 51.4 |
28.4 |
N | N | N | N | N | N | N | N |
| Lai et al. [8] | GPi | 42.9 |
28.5 |
N | N | N | N | 62.9 |
N | N | N |
| Schulz-Schaeffer et al. [12] | STN | N | N | 53.2 |
34.3 |
N | N | 22.5 |
15.2 |
1044.0 |
561.0 |
| Umemura et al. [13] | STN | N | N | N | N | 50.6 |
20.4 |
28.6 |
N | 616.5 |
264.3 |
| Sako et al. [14] | STN | 73 |
17.0 |
N | N | 48.3 |
15.0 |
41.0 |
15.0 |
N | N |
| Case reports. [15, 16, 17, 18, 19, 20, 21, 22, 23] | STN | N | N | 42.5 |
29.0 |
46.2 |
23.8 |
33.5 |
17.9 |
674.2 |
475.5 |
| Case reports. [21, 24, 25] | GPi | N | N | N | N | 41.3 |
25.0 |
N | N | 708.3 |
675.0 |
| DBS, deep brain stimulation; STN, subthalamic nucleus; GPi, globus pallidus internus; LEDD, L-dopa equivalent daily dose; UPDRS-III, the Unified Parkinson’s Disease Rating Scale III; N, not available. | |||||||||||
The quality of articles was evaluated according to the following criteria: (I)
Randomized controlled trial; (II) Prospective observational or case-controlled
study; (III) Retrospective study; (IV) Case report or series (
Review Manager software (version 5.3, The Nordic Cochrane Centre, Copenhagen,
Denmark) and SPSS Statistics (version 17.0, IBM SPSS Inc, Chicago, IL, USA)
were used for statistical analysis. Mean differences and 95% confidence
intervals for the variables are presented as forest plots, with the Chi-squared
and I
Details of the 20 studies and 152 participants included in this meta-analysis
are listed in Tables 1,2. The patients were comprised of 55 males, 45
females and 52 with unknown gender. STN-DBS was employed as the intervention in
16 studies (136 patients) and GPi-DBS (13 patients) in three studies. One study
used both STN-DBS (2 patients) and GPi-DBS (one patient). Comparisons of the
clinical data between pre- and post-DBS periods are shown in Figs. 2,3.
After an average of 3–21 months follow-up post DBS, the mean bending angle
assessed during the Off-period was markedly lower than during the pre-operative
period (31.5
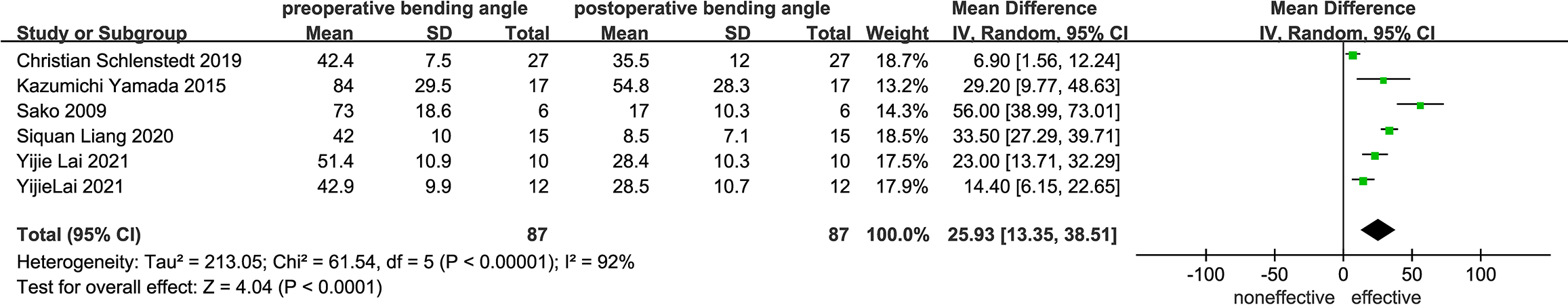 Fig. 2.
Fig. 2.Comparison bending angle between pre- and post-DBS (Off-period). DBS, deep brain stimulation.
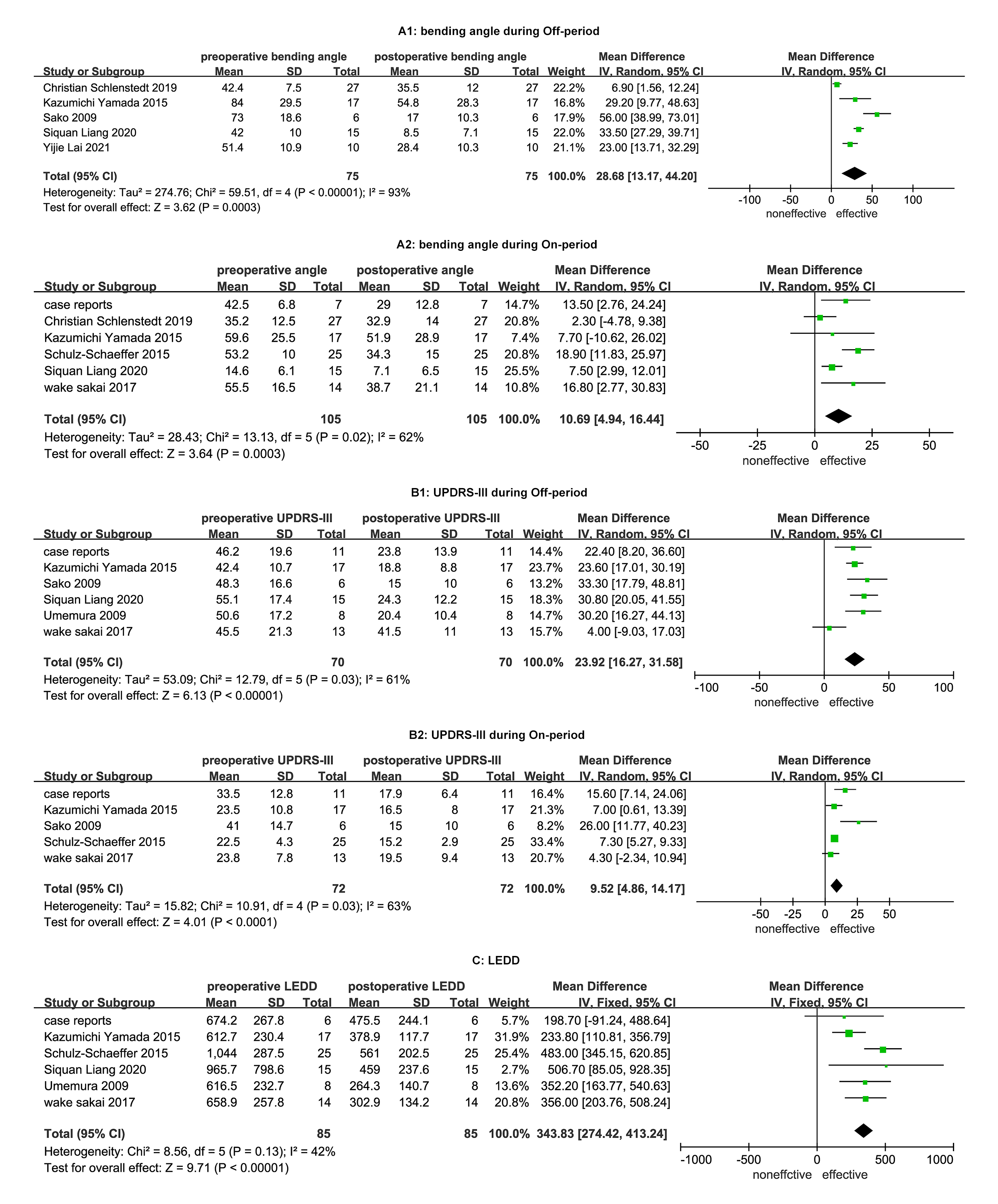 Fig. 3.
Fig. 3.Comparisons of clinical data between pre- and post-STN-DBS. A1: comparison bending angle between pre- and post-STN-DBS during Off-period; A2: comparison bending angle between pre- and post-STN-DBS during On-period; B1: comparison UPDRS-III between pre- and post-STN-DBS during Off-period; B2: comparison UPDRS-III between pre- and post-STN-DBS during On-period; C: comparison LEDD between pre- and post-STN-DBS. DBS, deep brain stimulation; STN, subthalamic nucleus; UPDRS-III, the Unified Parkinson’s Disease Rating Scale III; LEDD, L-dopa equivalent daily dose.
Subgroup analysis was performed to explore the effectiveness of STN and GPi-DBS.
For the STN-DBS trials, the mean post-operative bending angles during both the
Off- and On-periods were significantly reduced compared to the pre-operative
period (32.1
The correlation analysis between clinical data and decreased angle of camptocormia after DBS were listed in Table 3. Pearson correlation analysis showed that the decease in bending angle after DBS was negatively correlated with the duration of camptocormia (R = –0.433, p = 0.013, Table 3), whereas positively associated with the pre-bending angle (R = 0.352, p = 0.03, Table 3).
| Decreased angle after DBS in On-period | Decreased angle after DBS in Off-period | |||
| Pearson Correlation | p-value | Pearson Correlation | p-value | |
| Age | –0.241 | 0.107 | –0.275 | 0.095 |
| Duration of PD | 0.128 | 0.398 | –0.269 | 0.102 |
| Duration of CC | –0.104 | 0.493 | –0.433* | 0.013* |
| LEDD | –0.062 | 0.748 | –0.404 | 0.136 |
| Pre-bending angle (On) | 0.157 | 0.298 | –0.241 | 0.184 |
| Pre-bending angle (Off) | 0.036 | 0.845 | 0.352* | 0.030* |
| Pre-UPDRS-III (On) | –0.023 | 0.939 | –0.070 | 0.895 |
| Pre-UPDRS-III (Off) | 0.036 | 0.852 | 0.081 | 0.728 |
| DBS, deep brain stimulation; PD, Parkinson’s disease; CC, Camptocormia; LEDD, L-dopa equivalent daily dose. | ||||
Camptocormia is a common postural disorder that generally occurs during the advanced stage of PD. DBS is an effective treatment that can alleviate many motor and non-motor symptoms of PD patients in clinical practice. This meta-analysis found that the average post-operative UPDRS-III scores and mean LEDD were lower than during the pre-operative period, thus supporting the efficacy of DBS for the treatment of motor symptoms.
The mean post-operative bending angles following both STN-DBS and GPi-DBS were also found to be significantly lower than in the pre-operative period, indicating that DBS can markedly improve camptocormia in PD patients. The underlying pathogenesis of camptocormia is still unclear, although the central pathophysiological mechanism is thought to play an important role in the occurrence and progression of this condition [28, 29]. DBS may send high-frequency stimulation to reduce the firing frequency on GPi/STN, thereby inducing disinhibition of motor thalamic nuclei and ultimately exciting the motor cortex [30]. Therefore, we speculate the improvement of camptocormia following DBS occurs mainly through above mentioned central pathophysiological mechanism that alleviates dysregulation of the basal ganglia and dystonia [29].
We found that the decreased bending angle after DBS was negatively correlated
with the duration of camptocormia, whereas positively associated with the
pre-bending angle. This concurs with previous studies that found camptocormia
duration of
In this meta-analysis, STN-DBS was observed to show a similar decrease in the mean bending angle as GPi-DBS. Previous studies have also reported similar improvement of motor symptoms in PD patients [31, 32]. However, GPi and STN each have advantages and disadvantages. For example, STN-DBS may be associated with a greater reduction of dopaminergic medication and alleviation of non-motor symptoms than GPi-DBS, but also with a higher risk of cognitive deterioration over time. On the other hand, patients with more severe dyskinesia or gait disorders may benefit more from GPi-DBS [31, 32]. Some authors have also suggested superiority of GPi-DBS for PD [33]. Therefore, the choice of whether to use GPi or STN for PD patients with camptocormia may depend on other motor and non-motor symptoms, such as tremor, dyskinesia, gait, cognition and mood.
This meta-analysis has several limitations. Firstly, the sample size was relatively small. Secondly, only English language studies were included, which might give rise to some bias. Thirdly, the included studies may have heterogeneity in camptocormia, PD characteristics and follow-up time.
DBS can improve camptocormia in PD patients. Patients in the early stage of camptocormia and with a greater bending angle may benefit more from DBS.
PD, Parkinson’s disease; DBS, deep brain stimulation; LEDD, L-dopa equivalent daily dose; STN-DBS, subthalamic nucleus deep brain stimulation; GPi-DBS, globus pallidus internus deep brain stimulation; TCC, total camptocormia; UCC, upper camptocormia.
FW and XG participated in design, literature selection, data extraction, statistical analysis, and drafting the manuscript. LH and HZ performed study selection and statistical analysis. All authors also participated in analyzing results, revising the manuscript, and approving the final version of this manuscript.
Not applicable.
We would like to thank all participants in this study.
This research received no external funding.
The authors declare no conflict of interest.



