1 The Second Affiliated Hospital of Fujian University of Traditional Chinese Medicine, 350122 Fuzhou, Fujian, China
2 National-Local Joint Engineering Research Center of Rehabilitation Medicine Technology, Fujian University of Traditional Chinese Medicine, 350122 Fuzhou, Fujian, China
3 Rehabilitation Industry Institute, Fujian University of Traditional Chinese Medicine, 350122 Fuzhou, Fujian, China
4 College of Rehabilitation Medicine, Fujian University of Traditional Chinese Medicine, 350122 Fuzhou, Fujian, China
5 Traditional Chinese Medicine Rehabilitation Research Center of State Administration of Traditional Chinese Medicine, Fujian University of Traditional Chinese Medicine, 350122 Fuzhou, Fujian, China
†These authors contributed equally.
Academic Editor: Hongmin Wang
Abstract
Background: This study aimed to investigate the effects of electroacupuncture (EA) treatment at Zusanli (ST36) and Quchi (LI11) on cortico-striatal network connectivity after ischemia stroke by resting-state functional magnetic resonance imaging (fMRI). Methods: A rat model of middle cerebral artery occlusion (MCAO) was established. Rats were randomly assigned into a sham-operated control group (SC group, n = 8), untreated MCAO model group (MCAO group, n = 8), and MCAO group receiving EA treatment at ST36 and LI11 (MCAO + EA group, n = 8). Rats in the SC and the MCAO groups received no treatment. The MCAO + EA group was treated with EA from the 1st day to the 7th day after surgery. The behavioral tests including Zea Longa test and modified neurologic severity score (mNSS) for all rats were performed before and after treatment for MCAO + EA group. fMRI scans were performed after behavioral tests on the 7th day after surgery. Results: The neurologic severity scores estimated by Zea Longa and mNSS were significantly improved in the rat ischemic stroke model of MCAO within 1 week after EA treatment at acupoints ST36 and LI11. Besides, voxel-wise analysis showed that EA could increase the functional connectivity of the left striatum with the bilateral sensory cortex, bilateral motor cortex, left retrosplenial cortex, right cerebellum, bilateral hippocampus, bilateral auditory cortex, bilateral visual cortex, left parietal cortex, left cingulate gyrus, and left superior colliculus. Further graph theory analysis showed that EA significantly decreased the characteristic path length and increased the global efficiency of the cortico-striatal network. Conclusions: EA at ST36 and LI11 could improve the cortico-striatal network to impact the brain’s protective in MCAO, which is a potential treatment for ischemia stroke.
Keywords
- ischemic stroke
- functional connectivity
- striatum
- electroacupuncture
- motor
Stroke is a group of neurological disorders caused by cerebrovascular disease that occurs suddenly [1]. It has the characteristics of high incidence and disability rate all over the world, which seriously affects the quality of life of patients and brings a heavy burden to the family and society [2]. How to improve the motor function of stroke patients and improve the basic activities of daily living (BADL) is a problem worth paying attention to.
Movement production is a multi-step process including instructions sending out and processing via the cerebral cortex and subcortical regions, projecting down the spinal cord, innervating muscles and finally generating movement [3, 4]. Abnormal brain functional activity would lead to motor dysfunction [5]. As an important part of the basal ganglia, the striatum plays an important role in motor function, including the adjustment of the body’s voluntary movement, non-conscious movement, muscle tension, and fine movement [6]. It has been found that motor dysfunction in many diseases is associated with abnormal functional connectivity between the striatum and other brain regions [7, 8]. Besides, one previous study reported that the abnormal functional connectivity centered in the striatum may be one of the causes of motor dysfunction after stroke [9]. However, acupuncture on modulating the functional network of the striatum in the motor recovery of stroke still remains unclear.
Acupuncture is one important traditional Chinese medicine, which is thought to stimulate specific body regions (acupoints) to modulate meridian channels to treat human diseases, such as stroke [10, 11]. Electroacupuncture (EA) is developed from the traditional acupuncture technique and has the advantages of ease of use and stable stimulation parameters [12]. Zuansanli (ST36) and Quchi (LI11) are common acupoints that have been used to modulate motor function [13]. It demonstrated that EA at ST36 and LI11 has a modulating effect on the central nervous system to improve the motor function in human and animal models [14, 15, 16]. However, the underlying neural mechanisms remain unclear.
In the current study, with the aim to determine whether EA treatment at ST36 and LI11 could improve functional connectivity of the cortico-striatal network after stroke, we used the resting-state functional magnetic resonance imaging (fMRI) to observe the changes of cortico-striatal network and applied graph theory to estimate its features in rat ischemic stroke model with middle cerebral artery occlusion (MCAO) after EA treatment.
Twenty-four healthy male Sprague Dawley (SD) rats, weighting 250
The experiment protocol is shown in Fig. 1. The MCAO rat model was established by the method described in the study of Longa et al. [17]. The rats were anesthetized with 3% isoflurane during surgery. The left common carotid artery (CCA) was taken and the left internal carotid arteries (ICA), external carotid arteries (ECA) were isolated carefully. The artery was then ligated near the bifurcation of the ICA and ECA. A small opening was made at the site that was 3 mm distal to the CCA ligation, subsequent to blocking the blood flow in the ICA with an artery clip. The middle cerebral artery was occluded by insertion of a nylon filament (diameter 0.24 mm). After 2 hours of ischemia, the nylon filament was carefully pulled out to establish reperfusion. The rats in the SC group only separated the vessels, but without ligation or insertion of wires. The Laser Doppler Flowmetry (Biopac Systems, Goleta, CA, USA) was used for observation. The MCAO model was considered successful, only when cerebral blood flow dropped to less than 80% of baseline. Throughout the procedure, the rectal temperature of all rats were maintained at 37 ℃ until anesthesia was restored.
 Fig. 1.
Fig. 1.
Experimental protocol. The MCAO and MCAO + EA were modeled at the beginning. The MCAO + EA group was treated with EA from the 1st day to the 7th day after surgery. Rats in the SC and the MCAO groups received no treatment. The behavioral tests including Zea Longa test and mNSS for all rats were performed before and after treatment for MCAO + EA group. fMRI scans were performed after behavioral tests on the 7th day after surgery. All the rats were sacrificed after the fMRI scan. EA, electroacupuncture; fMRI, functional magnetic resonance imaging; MCAO, middle cerebral artery occlusion; mNSS, modified neurologic severity score; SC, sham-operated control.
After surgery, SC group and MCAO group did not receive any treatment, while MCAO + EA group received 30 minutes EA treatment each day for 7 consecutive days. The needle was directly pierced with a depth of about 2–3 mm at ST36 and LI11. The stimulation parameters were set as follows: dense disperse waves of 1/20 Hz, current of 2 mA, peak voltage of 6 V. The rats were quiet without restlessness, auricle slight flapping or local muscle contraction as the degree. The EA treatment performed using stainless steel acupuncture needles (0.3 mm diameter, Huatuo, Suzhou Medical Appliance Factory, Suzhou, China).
Neurologic deficits were evaluated via Zea Longa [17] and modified neurologic severity score (mNSS) [18] on the 1st and 7th day after surgery. Zea Longa was scored as follows: 0 point, no symptom of neurological impairment; 1 point, rats could not fully extend the contralateral forelimb; 2 points, the body turned to the hemiplegia side while walking; 3 points, the body leaned to the hemiplegia side when walking; 4 points, the rat could not be self-issued with loss of consciousness [17]. mNSS includes four aspects (motor, sensory, balance, reflex) with a total score of 18, and 0 indicates no impairment of neurological function [18]. The higher the score of the two behavioral tests, the more severe impairment of neurological function. All behavioral tests were conducted by two investigators who were blinded to the experiment protocols.
Each group of rats received resting-state fMRI scanning at the 7th day after MCAO. Rats were anesthetized with 1%–3% isoflurane (mixed with 20% oxygen and 80% air) inhaled for 5 minutes, and then were further anesthetized via intramuscular injection of dexmedetomidine hydrochloride (2 mL:200
Echo planar imaging sequence (EPI) was applied to acquire fMRI data with the following parameters: repetition time = 2000 ms, echo time = 28 ms, field of view = 32*32 mm
fMRI data were preprocessed using Statistical Parametric Mapping (SPM12, https://www.fil.ion.ucl.ac.uk/spm/) and DATA Processing Assistant for Resting-state fMRI (DPARSF, http://www.restfmri.net/forum/DPARSF). Preprocessing steps included enlarging 10 times of the voxel size, slice-timing correction, realignment for head motion correction, normalization into the standard rat brain atlas, and smoothing with a full-width half maximum of 4 mm. Data were excluded if head movements exceeded 1.0 mm of maximum translation in the x, y, z directions or 2.0 degree maximum rotation in the three axes. Then, the smoothed images were detrended and filtered (0.01–0.08 Hz).
Functional connectivity was evaluated using seed-based correlational analysis. The time courses of the voxels in the left striatum was averaged to use as the reference time courses. Pearson’s correlation coefficients between the reference time course and the time course of every voxel in the whole brain were calculated.
A total of 15 brain regions reported in the voxel-based analysis, including bilateral hemispheres, were selected for graph theory analysis. Graph theoretical approaches were applied to characterize the undirected weighted network of striatum using the Gretna software (https://www.nitrc.org/projects/gretna/). According to graph theory, the node of the network was represented by the brain region, the edge of the network was defined as the connectivity of each pair of nodes, and the weight of the edge was defined as the absolute value of correlation coefficients. To assess the global network properties, the global efficiency and characteristic path length were calculated. The characteristic path length is defined as the average of the shortest path lengths between any pair of nodes in the network [19]. Characteristic path length is inversely related to global efficiency and is a measure of the capacity for information transfer in the network [20].
Statistical analyses were performed using the Statistical Package of the Social Science (SPSS) software, version 24.0 (IBM SPSS Statistics for Windows, IBM Corp., Armonk, NY, USA). The experimental data conforming to the normal distribution were described by mean
The assessment examined the neurological deficits of the rats via the Zea Longa and mNSS. Before intervention, the Zea Longa and mNSS scores in the MCAO group were significantly higher than those in the SC group (p
| SC | MCAO | MCAO + EA | Z | p | |
| Pre-treatment | 0 | 2 (2–3)*** | 2 (2–2.75) | 17.97 |
|
| Post-treatment | 0 | 2 (2–2.75)*** | 1 (1–1)†† | 20.28 |
|
| EA, electroacupuncture; SC, sham-operated control group; MCAO, middle cerebral artery occlusion control group; MCAO + EA, MCAO with EA treatment group. *** p †† p |
|||||
| SC | MCAO | MCAO + EA | F | p | |
| Pre-treatment | 0 | 14.38 |
14.38 |
734.72 |
|
| Post-treatment | 0 | 12.25 |
10.63 |
556.01 |
|
| EA, electroacupuncture; SC, sham-operated control group; MCAO, middle cerebral artery occlusion control group; MCAO + EA, MCAO with EA treatment group. *** p † p |
|||||
The differences of the functional connectivity of the left striatum were estimated among the three groups. Compared with the SC group, the MCAO group showed that the functional connectivity of the left striatum with the bilateral sensory cortex, bilateral motor cortex, left retrosplenial cortex, right dorsal thalamus, right cerebellum, right hippocampus, bilateral auditory cortex, bilateral visual cortex, and lateral amygdala was decreased (Table 3, Fig. 2A). In contrast, the MCAO + EA group showed that the functional connectivity of the left striatum with the bilateral sensory cortex, bilateral motor cortex, left retrosplenial cortex, right cerebellum, bilateral hippocampus, bilateral auditory cortex, bilateral visual cortex, left parietal cortex, left cingulate gyrus, and left superior colliculus was increased (Table 3, Fig. 2B).
| Brain region | MCAO |
MCAO + EA |
||
| Clusters | t-value | Clusters | t-value | |
| Striatum left | 400 | –7.0338 | 278 | 6.2249 |
| Striatum right | 385 | –6.6577 | 35 | 4.8181 |
| Sensory cortex left | 18 | –4.2347 | 360 | 5.9202 |
| Sensory cortex right | 154 | –5.0759 | 123 | 5.9281 |
| Motor cortex left | 13 | –4.0053 | 42 | 5.1772 |
| Motor cortex right | 51 | –5.4477 | 43 | 4.7989 |
| Retrosplenial cortex left | 23 | –4.9827 | 51 | 6.1194 |
| Dorsal thalamus right | 15 | –4.999 | - | - |
| Cerebellum right | 35 | –4.911 | 36 | 5.3971 |
| Piriform cortex right | 65 | –5.1252 | - | - |
| Hippocampus left | - | - | 34 | 5.8503 |
| Hippocampus right | 11 | –5.2207 | 19 | 5.1278 |
| Auditory cortex left | 26 | –4.1564 | 13 | 4.0961 |
| Auditory cortex right | 22 | –5.5066 | 11 | 4.3337 |
| Visual cortex left | 32 | –4.5233 | 132 | 7.5369 |
| Visual cortex right | - | - | 62 | 5.7978 |
| Entorhinal cortex left | 64 | –5.6032 | - | - |
| Entorhinal cortex right | 42 | –5.7427 | - | - |
| Amygdaloid body left | 25 | –6.6209 | - | - |
| Amygdaloid body right | 23 | –4.6629 | - | - |
| Parietal association cortex left | - | - | 19 | 4.877 |
| Cingulate gyrus left | - | - | 10 | 4.3132 |
| Superior colliculus left | - | - | 10 | 4.5954 |
| EA, electroacupuncture; SC, sham-operated control group; MCAO, middle cerebral artery occlusion; MCAO + EA, MCAO with EA treatment group. p |
||||
 Fig. 2.
Fig. 2.
Brain functional connectivity of the left striatum among the three groups. Detailed regions in the images show decreased functional connectivity with the left striatum in (A) MCAO group compared with SC group and increased functional connectivity with the left striatum in (B) MCAO + EA group compared with MCAO group (p
In the present study, we investigated the effect of EA on the topology using graph theory network analyses. The global efficiency of the MCAO group was significantly decreased (p
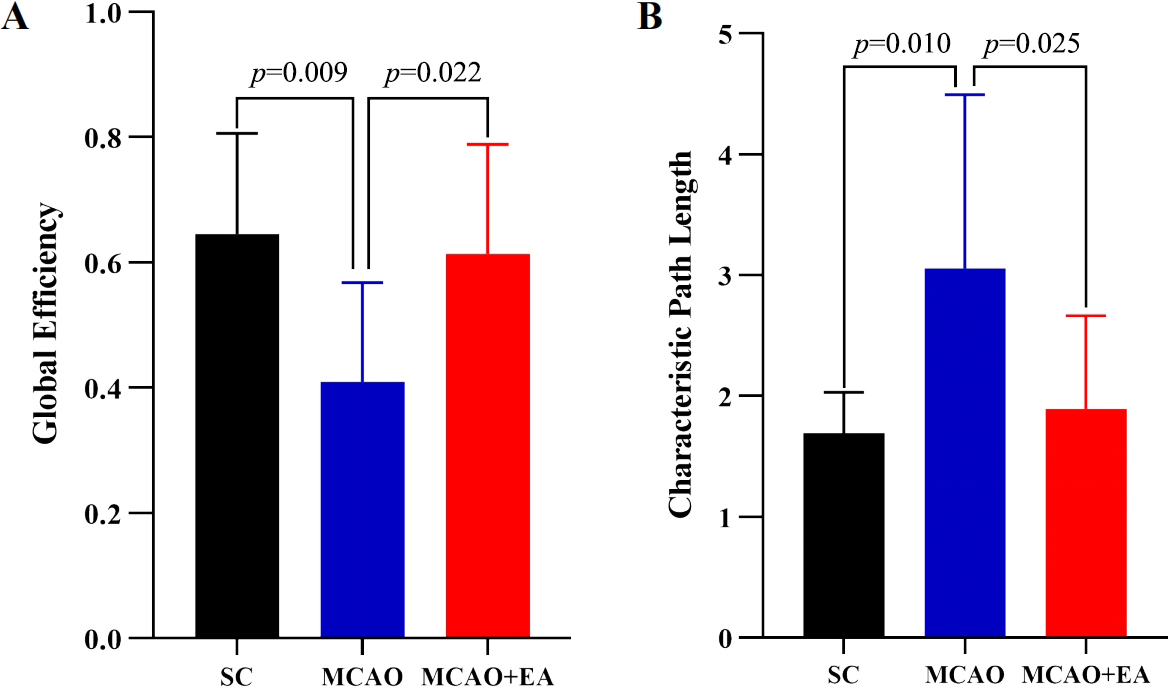 Fig. 3.
Fig. 3.
Global graph theoretic measures. Global effificiency (A) and characteristic path length (B) in the SC, MCAO and MCAO + EA groups.
Motor dysfunction is a common problem after ischemic stroke. Acupuncture, as a traditional Chinese medicine treatment, has been proven to have a positive effect on stroke [21, 22]. In this study, we found that compared with the untreated MCAO group, the EA treatment at ST36 and LI11 can significantly reduce the scores of Zea Longa and mNSS, which indicates that EA can improve the neurological deficits in MCAO group. Meanwhile, the result showed that EA could increase the functional connectivity between the striatum and the sensory cortex, motor cortex, striatum and so on in the MCAO group. The graph theory analysis showed that the global efficiency was significantly increased and the characteristic path length was significantly decreased after EA treatment compared to the MCAO group.
Zea Longa and mNSS were used to assess the neurological deficits of all rats. It showed that EA lasting for 7 days could improve the motor function of ischemia stroke, which was consistent with the previous studies [15]. EA treatment improved motor function after ischemic stroke in rats that may be related to the modulation of astrocytes, the improvement of abnormal synaptic activity, neuron metabolism/energy deficiency and the promoting of the repair of damaged brain tissue [23, 24]. Our previous studies have also confirmed that EA at ST36 and LI11 can stimulate neuronal activity in motion-related areas of rats with ischemia reperfusion injury and promote the recovery of motor function in rats [25, 26].
Striatum is the intermediate brain region in which the cerebral cortex transmits to the basal ganglion, and is mainly responsible for the integration and coding of neural information, and plays a critical role in regulating movement initiation and cessation, maintaining the coordination and accuracy of movement [27, 28]. In this study, the MCAO group showed decreased functional connectivity between the left striatum and sensory cortex, motor cortex, cerebellum after ischemia stroke, while EA at ST36 and LI11 could significantly increase the functional connectivity of the left striatum with these motor-related brain regions. Motor cortex is the area of the brain engaged in the planning, control and execution of autonomous movements, which governs the movement of various parts of the body [29]. Sensory cortex plays an important role in regulating motor processing, such as gaze and orientation. Besides, sensory cortex, together with the motor cortex, plays a crucial role in the integration of movement [30]. The cerebellum is an important movement regulation center under the cerebral cortex, which plays a role in maintaining body balance, regulating muscle tone and coordinating movement [31, 32]. Motor cortex, sensory cortex and striatum are related to the cortico-striatal brain circuits, which are associated with motor function processing and sensorimotor integration [33, 34]. When the cortico-striatal brain circuits were abnormal, there was paroxysmal dystonia and postural adaptation problem [35, 36]. Besides, several studies have shown that the visual cortex, auditory cortex, and retrosplenial cortex take part in movement, such as balance maintenance, motor planning, and motor control. The study also found that EA increased the functional connectivity between the left striatum and the visual cortex, auditory cortex, and retrosplenial cortex, which might contribute to the improvement of motor function after stroke [37, 38, 39].
Further graph theory analysis to investigate the parameters of cortico-striatal network in MCAO after EA. It showed that compared to the MCAO group, the MCAO + EA group had significantly higher global efficiency and significantly lower characteristic path length after received EA treatment. The global efficiency is mainly related to the network’s ability to transfer information between nodes in parallel through multiple edges [40], while the characteristic path length indicates the length of nodes that need to be crossed for the information to reach the endpoint [41]. It suggests that EA of ST 36 and LI11 can improve the information transfer efficiency of cortico-striatal network after ischemic stroke.
Some limitations should be pointed out. First, although anesthesia has been widely used in animal studies of functional magnetic resonance imaging, brain functional activity might be affected by anesthesia. Further study should be conducted to verify the results in the awake state. Besides, the study was conducted only in rats, and the results still need to be confirmed clinically. In addition, the study only investigated the effect of EA at ST36 and LI11 in ischemic stroke. Further study should be done to investigate the different effects between EA at ST36 and atLI11.
In conclusion, this study suggests that EA at ST36 and LI11 could improve motor function via increasing the functional connectivity between the ipsilateral striatum and brain regions that involved in motor function, which might provide a potential non-pharmacologic therapeutic method for ischemic stroke.
SL, LY, and TT contributed to conception and design of the study. LY, TT, YL, and MY collected the data. LY, TT, and WL performed the statistical analysis. LY and TT wrote the first draft of the manuscript. SL revised the manuscript. All authors contributed to manuscript revision, read, and approved the submitted version.
All experiments were in accordance with the requirements of the Animal Experiment Ethics Committee of Fujian University of Chinese Medicine (Code: FJTCM IACUC 2020079).
Not applicable.
This work was supported by grants from the Scientific Research Foundation for the High-level Talents, Fujian University of Traditional Chinese Medicine (X2019002-talents), Science and technology platform construction project of Fujian science and Technology Department (Grant No.2018Y2002) and National Natural Science Foundation of China (82004441).
The authors declare no conflict of interest.



