1 Department of Neurology, First Affiliated Hospital of Xinxiang Medical University, Henan Joint International Research Laboratory of Neurorestoratology for Senile Dementia, Henan Key Laboratory of Neurorestoratology, 453100 Xinxiang, Henan, China
2 Department of Neurology, the Sixth Hospital of Wuhan, Affiliated Hospital to Jianghan University, 430015 Wuhan, Hubei, China
3 Department of Medical Record Management, First Affiliated Hospital of Xinxiang Medical University, 453100 Xinxiang, Henan, China
4 Department of Imaging, First Affiliated Hospital of Xinxiang Medical University, 453100 Xinxiang, Henan, China
5 Ann Romney Center for Neurologic Diseases, Brigham and Women’s Hospital, Harvard Medical School, Boston, MA 02115, USA
†These authors contributed equally.
Academic Editors: Emilia Salvadori and Hahn Young Kim
Abstract
Background: Matrix metalloproteinase-9 (MMP-9) is a significant protease required for synaptic plasticity, learning, and memory. Yet, the role of MMP-9 in the occurrence and development of cognitive decline after ischemic stroke is not fully understood. In this study, we used clinical data experiments to further investigate whether MMP-9 and genetic polymorphism are associated with post-stroke cognitive impairment or dementia (PSCID). Materials and Methods: A total of 148 patients with PSCID confirmed by the Montreal Cognitive Assessment (MoCA) 3 months after onset (PSCID group) were included in the study. The MMP-9 rs3918242 polymorphisms were analyzed using polymerase chain reaction coupled with restriction fragment length polymorphism, and the serum level of MMP-9 was measured using enzyme-linked immunosorbent assay (ELISA). The same manipulations have been done on 169 ischemic stroke patients without cognitive impairment (NCI group) and 150 normal controls (NC group). Results: The expression level of serum MMP-9 in the PSCID group and NCI group was higher compared to the NC group, and the levels in the PSCID group were higher than that in the NCI group (all p
Keywords
- ischemic stroke
- post-stroke cognitive impairment or dementia (PSCID)
- matrix metalloprotenase 9
- genetic polymorphism
- cognitive function
Vascular cognitive impairment (VCI) is a heterogeneous disease that involves cognitive decline characterized by disturbance of frontal or executive dysfunction [1]. VCI is usually a consequence of cerebrovascular disorders (cerebral infarction, cerebral hemorrhage, chronic cerebral hypoperfusion) and their risk factors (hypertension, diabetes, hyperlipemia, hyperhomocysteinemia, etc.) [1, 2]. Post-stroke cognitive impairment or dementia (PSCID; VCI after cerebral ischemic stroke) is very common and can affect different cognitive domains. Executive functions are the most commonly affected funtions [3]. Impairment in cognitive, especially executive functions, commonly appears within 3 months after stroke. According to various hospital-based studies [4, 5, 6], the prevalence of PSCID varies from 11.6% to 56.3%. Therefore, neurorestoration of cognitive impairment has gained increasing interest among researchers [7, 8, 9].
Matrix metalloproteinase-9 (MMP-9) is a member of the MMP family with 26 extracellular and intracellular matrix-degrading enzymes that regulate many zinc-binding proteolytic enzymes physiological processes, including activation of growth factors, tumor growth and metastasis, cleavage of zymogens, and remodeling of the extracellular matrix [7, 8]. MMP-9 is mainly produced by neurons, and its expression and activity have been detected in adult brain structure, such as the hippocampus, cortex, striatum, and cerebellum [9, 10]. In neurons, the MMP-9 expression is induced by neuronal activity under both physiological and pathological conditions, such as stroke [11, 12] and epilepsy [13]. Abnormalities and typically excessive MMP-9 gene expression have been associated with several central nervous system diseases [14, 15, 16]. Moreover, MMP-9 is involved in blood-brain barrier destruction [17], inflammatory reaction [18], atherosclerosis, and ischemic stroke [19], as well as in synaptic plasticity [20], learning, and memory [21]. Yet, the role of MMP-9 in the occurrence and development of PSCID is not fully understood.
So far, only a few studies reported on the role of MMP-9 in VCI and its progression to dementia [22, 23, 24]. Even some studies have not found correlation with MMP-9 polymorphism and cognition [25, 26, 27]. In this study, we use enzyme-linked immunosorbent assay (ELISA) to detect serum MMP-9 levels and their association with PSCID. Furthermore, genotyping assays were performed to determine the relationship between the MMP-9 rs3918242 polymorphism and susceptibility of PSCID in a Chinese population.
A total of 2351 acute ischemic stroke patients consecutively enrolled in the
Department of Neurology, the First Affiliated Hospital of Xinxiang Medical
University, Henan, China, between June 2013 and September 2018 were screened. The
main inclusion criteria were: (1) ischemic stroke confirmed by imaging (computed
tomography (CT) or magnetic resonance imaging (MRI)) within 72 hours of symptom
onset, according to the criteria of the 10th Edition of International
Classification of Diseases (ICD-10); (2) unrelated Han Chinese people and a
Mini-Mental State Examination (MMSE) score of illiterate
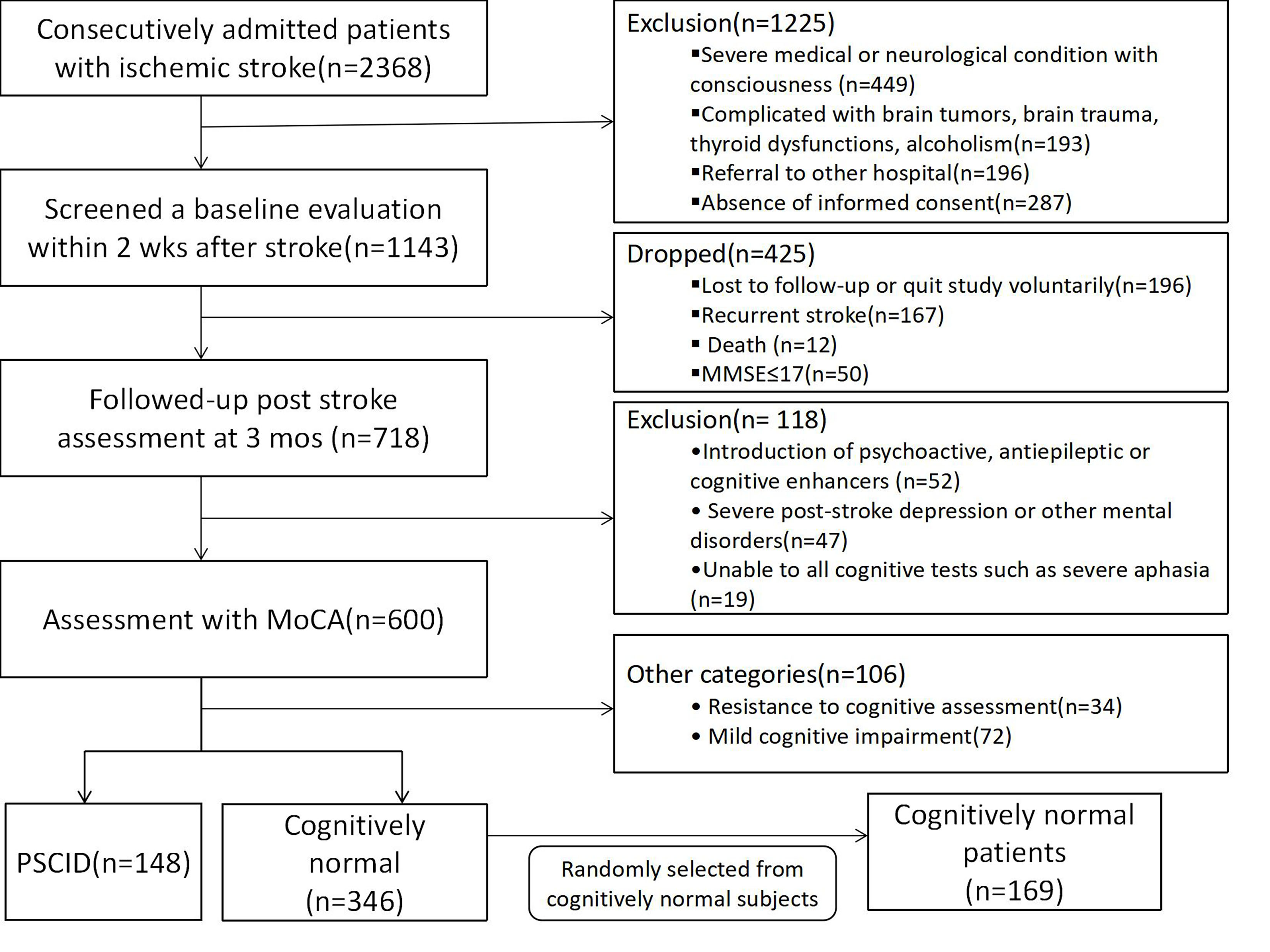 Fig. 1.
Fig. 1.Flow chart of participants’ selection.
After the onset of 3 months [28], 583 patients underwent a follow-up evaluation,
and 486 patients were assessed with global cognitive functions by Changsha
version of the Montreal Cognitive Assessment
(MoCA-CS) [29] and according to the flow chart [30, 31]. Yu K.H’s protocol was
referred (Fig. 1) [32]. Age- and gender-matched cognitively normal patients after
ischemic stroke (MoCA
All subjects were evaluated based on MMSE and MoCA (Fig. 1). MoCA was scored
based on a 30-point scale with 7 cognitive subtests: visual-spatial executive,
naming, attention, language, abstraction, delayed recall, and orientation. The
MoCA test was conducted to rectify the educational level of the bias correction,
and subjects with education
Unified and standardized survey questionnaire terminologies were used in a quiet environment without interferences. The neuropsychological assessments for each patient were completed by one psychological surveyor on the same day.
The blood samples were collected during cognitive assessment between three
months and half a year after stroke. And the blood sample were collected in
EDTA-containing tubes and separated the serum immediately and frozen at –80
The MMP-9 rs3918242 polymorphisms were performed by polymerase chain reaction (PCR) coupled with restriction fragment length polymorphism, as described in our previous studies [19].
The collection, processing and storage of blood sample are same as DNA extraction. The process of measuring MMP-9 is according to the MMP-9 ELISA Kit’s (Wuhan Fine Biotech Co., Ltd.; Wuhan; China) protocol.
SPSS 24.0 (SPSS Inc., Chicago, IL, USA) was used for statistical processing. The
measurement data in the results were expressed as mean
There were significant differences in diabetes, hyperhomocysteinemia, serum
MMP-9 levels, and MoCA scores between the three groups (all p
| Variables | Groups | F/Z/χ |
p | |||
| PSCID | NCI | NC | ||||
| (n = 148) | (n = 169) | (n = 150) | ||||
| Age [years] | ||||||
| 73 (49.32) | 88 (52.07) | 73 (48.67) | ||||
| 75 (50.68) | 81 (47.93) | 77 (51.33) | 0.42 | 0.81 | ||
| Gender | ||||||
| Females | 98 (66.22) | 107 (63.31) | 100 (66.67) | |||
| Males | 50 (33.78) | 62 (36.69) | 50 (33.33) | 0.47 | 0.79 | |
| Educations [years] | ||||||
| 102 (68.92) | 118 (69.82) | 102 (68.00) | ||||
| 46 (31.08) | 51 (30.18) | 48 (32.00) | 0.12 | 0.94 | ||
| NIHSS |
8.59 |
8.37 |
- | 0.34 | 0.56 | |
| Hypertension | ||||||
| No | 52 (35.14) | 63 (37.28) | 54 (36.00) | |||
| Yes | 96 (64.86) | 106 (62.72) | 96 (64.00) | 0.16 | 0.92 | |
| Diabetes mellitus | ||||||
| No | 94 (63.51) | 129 (76.33) | 113 (75.33) | |||
| Yes | 54 (36.49) | 40 (23.67) | 37 (24.67) | 7.68 | 0.02 | |
| Hyperlipidemia | ||||||
| No | 128 (86.49) | 154 (91.12) | 141 (94.00) | |||
| Yes | 20 (13.51) | 15 (8.88) | 9 (6.00) | 3.49 | 0.18 | |
| Hyperhomocysteinemia | ||||||
| No | 62 (41.89) | 93 (55.03) | 86 (57.33) | |||
| Yes | 86 (58.11) | 76 (44.97) | 64 (42.67) | 8.36 | 0.02 | |
| Tobacco smoking | ||||||
| Never | 95 (64.19) | 108 (63.91) | 96 (64.00) | |||
| Yes | 53 (35.81) | 61 (36.09) | 54 (36.00) | 0.00 | 1.00 | |
| Alcohol intake | ||||||
| Never | 78 (52.70) | 102 (60.36) | 90 (60.00) | |||
| Yes | 70 (47.30) | 67 (39.64) | 60 (40.00) | 2.33 | 0.31 | |
| TOAST classification | ||||||
| Large-artery atherosclerosis | 52 (35.14) | 59 (34.91) | - | |||
| Small vessel occlusion | 64 (43.24) | 71 (42.01) | - | |||
| Cardioembolism | 10 (6.76) | 12 (7.10) | - | |||
| Other determined etiology | 13 (8.78) | 16 (9.47) | - | |||
| Undetermined etiology | 9 (6.08) | 11 (6.51) | - | 0.11 | 1.00 | |
| MMP-9 (mg/dL) |
299.07 |
266.56 |
197.75 |
88.14 | ||
| MoCA |
20.27 |
28.62 |
29.50 |
337.74 | ||
| *p BMI, Body mass index; NIHSS, National Institutes of Health Stroke Scale; TOAST, Trial of Org 10172 in Acute Stroke Treatment. | ||||||
With diabetes mellitus, when hyperhomocysteinemia and serum MMP-9 level were used as independent variables, and PSCID as dependent variables, logistic regression analysis showed that diabetes mellitus, hyperhomocysteinemia, and elevated serum MMP-9 level were the main risk factors of PSCID (OR = 1.77, 95% CI: 1.07–2.93; OR = 1.88, 95% CI: 1.18–3.56; OR = 1.01, 95% CI: 1.01–1.02) (Table 2).
| Variables | Beta value | Standard error | Wald value | OR | 95% CI | p |
| Diabetes mellitus (%) | 0.63 | 0.24 | 4.91 | 1.77 | 1.07–2.93 | 0.03 |
| Hyperhomocysteinemia (%) | 0.57 | 0.26 | 7.10 | 1.88 | 1.18–3.56 | 0.01 |
| MMP-9 (mg/dL) | 0.01 | 0.0003 | 51.31 | 1.01 | 1.01–1.02 |
The Spearman correlation analysis showed that the serum MMP-9 level was
negatively correlated with the total score of MoCA scale in patients with PSCID
(p
| Variables | Fraction (mean |
MMP-9 (ng/mL) | |
| r | p | ||
| Visual spatial executive | 2.74 |
–0.401 | 0.000 |
| Naming | 2.59 |
–0.230 | 0.005 |
| Attention | 4.27 |
–0.470 | 0.000 |
| language | 1.99 |
–0.255 | 0.002 |
| Abstraction | 1.18 |
–0.006 | 0.944 |
| Delayed recall | 2.66 |
–0.165 | 0.045 |
| Orientation | 4.84 |
–0.130 | 0.116 |
| Total | 20.27 |
–0.517 | 0.000 |
The genotype frequency of MMP-9 rs3918242 in the NC group conformed to
Hardy-Weinberg equilibrium (p
| MMP-9 rs3918242 | Genotype % | Allele % | Hardy-Weinberg equilibrium | ||||
| TT | TC | CC | T | C | p | ||
| PSCID (%) | 77 (52.03) | 45 (30.40) | 26 (17.57) | 199 (67.23) | 97 (32.77) | 14.22 | 0.001 |
| NCI (%) | 109 (64.50) | 43 (25.44) | 17 (10.06) | 261 (77.22) | 77 (22.78) | 12.95 | 0.002 |
| NC (%) | 102 (68.00) | 40 (26.67) | 8 (5.33) | 244 (81.33) | 56 (18.67) | 2.22 | 0.329 |
| 14.57 | |||||||
| p | 0.006 | ||||||
The logistic regression analysis showed TC genotype carrying MMP-9 rs3918242 (OR
= 3.91, 95% CI: 1.63–9.38) and CC genotype (OR = 2.79, 95% CI: 1.12–7.00)
were associated with a significantly increased risk of PSCID (p
| MMP-9 rs3918242 | Groups | OR (95% CI) * | p | Groups | OR (95% CI) * | p | ||
| PSCID (%) | NCI (%) | PSCID (%) | NC (%) | |||||
| TT | 77 (52.03) | 102 (68.00) | 1.0 (Ref.) | - | 77 (52.03) | 109 (64.50) | 1.0 (Ref.) | - |
| TC | 45 (30.40) | 40 (26.67) | 3.91 (1.63–9.38) | 0.002 | 45 (30.40) | 43 (25.44) | 2.03 (1.02–4.05) | 0.044 |
| CC | 26 (17.57) | 8 (5.33) | 2.79 (1.12–7.00) | 0.028 | 26 (17.57) | 17 (10.06) | 1.51 (0.71–3.22) | 0.282 |
| * Adjusted for Diabetes mellitus, Hyperhomocysteinemia. | ||||||||
The relationship between MMP-9 rs3918242 and PSCID risk of was further analyzed and stratified by demographic characteristics such as diabetes mellitus and hyperhomocysteinemia (Table 6). The logistic regression analysis showed that diabetes mellitus and hyperhomocysteinemia in the TCCC genotype of MMP-9 rs3918242 were associated with increased risk of PSCID (OR = 1.26, 95% CI: 1.14–2.66; OR = 1.24, 95% CI: 1.02–2.69). In addition, hyperhomocysteinemia increased the risk of PSCID (OR = 1.19, 95% CI: 1.10–2.38).
| Variables | PSCID | NC | OR (95% CI) | p | PSCID | NCI | OR (95% CI) | p | ||||
| TT | TC+CC | TT | TC+CC | TT | TC+CC | TT | TC+CC | |||||
| Diabetes mellitus | ||||||||||||
| No | 51 | 43 | 86 | 43 | 1.65 (0.87–3.14) | 0.13 | 51 | 43 | 73 | 40 | 1.59 (0.84–3.02) | 0.06 |
| Yes | 26 | 28 | 23 | 17 | 1.26 (1.14–2.66) | 0.01 | 26 | 28 | 29 | 8 | 1.69 (0.90–3.56) | 0.37 |
| Hyperhomocysteinemia | ||||||||||||
| No | 47 | 15 | 53 | 40 | 1.51 (0.71–3.16) | 0.27 | 47 | 15 | 58 | 28 | 2.37 (0.86–4.82) | 0.22 |
| Yes | 30 | 56 | 56 | 20 | 1.24 (1.02–2.69) | 0.00 | 30 | 56 | 44 | 20 | 1.19 (1.10–2.38) | 0.00 |
Our data suggested that TC and CC genotypes of MMP-9 rs3918242 polymorphism were associated with a significantly increased risk of PSCID in a Han Chinese population, further suggesting an association between rs3918242 variation in the MMP-9 gene and PSCID. We also found that the TC genotype of rs3918242 increased the risk of PSCID compared to the TT genotype. Our results support the hypothesis that MMP-9 gene polymorphisms are associated with PSCID. In addition, our data suggested that diabetes mellitus, hyperhomocysteinemia, and MMP-9 serum levels were associated with PSCID. However, there was no significant association between PSCID and other demographic features (such as age, gender, hypertension, hyperlipidemia, tobacco smoking, alcohol intake, as well as TOAST classification). Moreover, MMP-9 in the serum was negatively correlated with MoCA score, especially visual-spatial executive, naming, attention, language, and delayed recall.
Numerous studies have investigated the relationships between MMP-9 gene polymorphisms and ischemic stroke. However, these results remain debatable. A meta-analysis showed that MMP-9 (-1562C/T) gene polymorphism was not associated with ischemic stroke [34]. In addition, Wang et al. [35] showed that the MMP-9 gene rs3918242 and rs17577 polymorphisms are not significantly correlated with ischemic stroke risk. Contrary, another meta-analysis suggested that MMP-9 (-1562C/T) polymorphisms might be a risk factor for ischemic stroke [36]. Also, several recent studies have reported the significant association between MMP-9 and cerebral ischemic stroke [15, 37, 38]. Gao et al. [37] showed that the rs3787268 locus in the MMP-9 gene might increase the risk of ischemic stroke in a southern Chinese Han population. Moreover, Hao and colleagues [16] conducted a similar study with 317 patients and found that MMP-9 rs3918242 polymorphism is correlated with an elevated risk of ischemic stroke. In addition, Li et al. [15] observed a significant association between the MMP-9 rs3918242 polymorphism and the risk of ischemic stroke, which is consistent with our previous study [19].
Preclinical studies showed that MMP-9 undergoes high expression and activation in brain tissue after a major complication of acute and chronic stroke [24, 39], suggesting that MMP-9 may participate in the acute brain injury and edema resulting from neuroinflammation [40, 41, 42]. On the other hand, secondary disruption to the deep white matter, the blood-brain barrier (BBB), and demyelination at the cerebral brain [24, 39, 43, 44], have an important role in the pathogenesis of vascular cognitive impairment and vascular dementia [22, 43]. Some clinical trials have reported elevated MMP-9 levels in the serum of patients with stroke [45, 46]. The high levels of MMP-9 in acute ischemic stroke confirm the involvement of this metalloproteinase in the regulation of inflammation in stroke [47, 48]. Meanwhile, animal studies suggested that MMP-9 has a role in cognitive functions, especially learning and memory [21, 49, 50, 51]. Therefore, the expression level of MMP-9 can be dispensable for PSCID. An increasing number of reports indicate that MMP-9 levels are in cerebrospinal fluid [22, 39, 41, 52] and peripheral blood [23, 53] of patients with PSCID. Furthermore, our data showed higher serum MMP-9 levels in patients with PSCID compared to normal controls and patients without cognitive impairment, which is consistent with the previous study. Furthermore, the serum level of MMP-9 was negatively correlated with the MoCA score, especially visual-spatial executive, naming, attention, language, and delayed recall.
This study has a few limitations. It has a relatively small sample size. Also, changes in MMP-9 expression at different development periods of PSCID were not analyzed. It is widely accepted that a large sample size is a key issue in genetic polymorphisms association studies of complex traits generally and cognitive dysfunction specifically [54]. In addition, it is unlikely that a polymorphism in a single gene would have a profound effect on the risk of PSCID which may be caused by many risk factors. Furthermore, the distribution and expression of MMP-9 in the pathogenesis and progression of post-stroke cognitive impairment or dementia are dynamic. According to the previous studies [21, 22, 53, 55], MMP-9 expression levels are associated with PSCID, although they are not exactly equal in serum, cerebrospinal fluid, and brain tissue after BBB disruption. So, multi-center, multi-source, large sample size studies investigating PSCID and dynamic monitoring of MMP-9 are needed to further confirm these findings.
Our data and previous studies suggest that MMP-9 level and MMP-9 rs3918242 gene polymorphism are associated with post-stroke cognitive impairment or dementia (PSCID). MMP-9 has a role in the development of post-stroke cognitive impairment or dementia (PSCID); yet, the mechanisms of action need to be further explored.
JZhao and SL designed the study and wrote the protocol. FY and XP enrolled the patients and wrote the paper. QingL, FW and ZX were in charge of follow-up of patients. RC, DJ, JZhang, MW and QiongL interpreted the patients’ image data. SJ and SL help in manuscript revision and interpretation of the results.
The study was approved by the Ethics Committee of the first affilicated hospital of Xinxiang Medical University. The ethical statement No. is EC-022-016. All subjects gave written informed consent in accordance with the Declaration of Helsinki.
Not applicable.
Natural Science Foundation of Henan Province (182300410389); Joint construction project of Henan Medical Science and technology research plan (LHGJ20190437); Xinxiang Medical University Scientific Research Innovation Support Program Project (YJSCX202185Y).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

