1 National University of Public Services, H-1083 Budapest, Hungary
2 Psychosomatic Outpatient Clinics, H-1037 Budapest, Hungary
3 Center for Neuropsychiatric Research of Traumatic Stress, Department of Psychiatry & UHSL, First Faculty of Medicine, and Department of Psychiatry, Faculty of Medicine Pilsen, Charles University, CZ-12108 Prague, Czechia
4 Neuroscience and Consciousness Research Department, Vision Research Institute, Lowell, MA 01854 USA
Academic Editor: Parisa Gazerani
Abstract
Here we present a complex hypothesis about the psychosomatic mechanism of serotonergic psychedelics. Serotonergic psychedelics affect gut microbes that produce a temporary increase of 5-HT by their host enterochromaffin cells (ECs). This increased 5-HT production—which is taken up and distributed by platelets—may work as a hormone-like regulatory signal that could influence membrane permeability in the host organs and tissues and in the brain. Increased plasma 5-HT levels could enhance permeability of the blood-brain barrier (BBB). Transiently increased permeability of the BBB allows for plasma 5-HT to enter the central nervous system (CNS) and be distributed by the volume transmission. Next, this gut-derived 5-HT could modulate excitatory and inhibitory neurotransmission and produce special network disintegration in the CNS. This transient perturbation of the normal neural hierarchy allows patients access to suppressed fear information and perform an emotional reset, in which the amygdale may have a key role.
Keywords
- classic psychedelics
- serotonin
- gut-brain axis
- platelets
- membrane permeability
- volume transmission
- amygdala
Serotonergic psychedelics (serotonergic hallucinogens) are a subclass of psychedelic drugs that bind and activate serotonin 5-HT2A receptors. Various studies have suggested that activation of the 5-HT2A receptor (a G-protein coupled receptor, coded by the HTR2A gene) has a key role in the effects of hallucinogenic drugs. 5-HT2A receptors are widely expressed in the human body, including in the gastrointestinal tract, platelets, and the nervous system [1]. There is growing evidence that serotonergic psychedelics can have important therapeutic effects on psychiatric disorders, but the underlying neurobiology and pharmacology of serotonergic psychedelics are currently not well understood [2, 3, 4, 5, 6].
Recently, we suggested that intestinal 5-HT, produced by enterochromaffin cells (ECs) and picked up by circulating platelets, may work as a hormone-like regulator and has a significant function in the regulation of membrane permeability in the intestine, brain, and other organs [7]. This notion may in part elucidate how gut dysbiosis could be linked to mental disorders.
After a brief review of the relevant literature about serotonin, classic psychedelics, gut-brain axis, platelets, blood-brain barrier (BBB) and volume transmission, and based on our previous articles’ conception [7, 8], we present a hypothesis about a possible mechanism for serotonergic psychedelics. We also point out briefly that the psychedelic-induced state bears similarities to the sleep-like state that may also contribute to a better understanding of the neuromolecular processes of psychedelics [9, 10].
Serotonin is an evolutionarily ancient molecule that can be found from nematodes to humans [11]. Serotonin (5-hydroxytryptamine, 5-HT) is a monoamine synthesized from tryptophan in two steps by tryptophan hydroxylase (TPH) and aromatic L-amino acid decarboxylase (AADC). TPH is a rate-limiting enzyme of 5-HT synthesis. There are two isoforms of TPH in vertebrates. TPH1 is primarily expressed in peripheral tissues mainly in intestinal enterochromaffin cells (ECs) and the pineal gland. TPH2 is expressed in peripheral myenteric neurons in the gut and in the neurons of raphe nuclei in the brainstem [12].
5-HT is not only known as a neurotransmitter, but it can also regulate nerve activity and various neuropsychological processes [13]. 5-HT also has hormonal, autocrine, and paracrine actions and can act as a growth factor [13]. About 95% of the body’s 5-HT is produced in the gut. In the gastrointestinal (GI) tract, 5-HT is produced by endocrine cells, called enterochromaffin cells (ECs), as well as mucosal mast cells and myenteric neurons [13, 14]. However, the most of the body’s 5-HT is produced by ECs in the gut.
5-HT, released in the gut from ECs, can regulate several gastrointestinal functions such as vasodilation, secretion, peristalsis, pain perception, nausea, etc. by activating 5-HT receptors on intrinsic and extrinsic afferent nerve fibers [15]. Much of the serotonin from the ECs is taken up by platelets and stored in their dense bodies (together with ATP, ADP, and Ca2+) and distributed throughout the body, similar to a hormone and released upon their activation, which is involved in diverse physiological functions [16, 17, 18].
5-HT has key roles in diverse normal physiological conditions and in pathological processes such as aggression; depression; schizophrenia and anxiety; sexual behavior and mood; cognitive functions; memory and learning; sleep; appetite; body temperature regulation; gastrointestinal regulations; glucose homeostasis and lipid metabolism; bone metabolism; vascular and immune processes including chemotaxis, leukocyte activation, proliferation, and cytokine secretion; hematological diseases; energy and apoptosis; age-related processes; among others [12, 17, 19, 20, 21, 22, 23, 24, 25, 26, 27].
5-HT has a surprisingly strong lipid membrane affinity [28]. Recently, Dey et al. [29] found that when 5-HT binds to the membrane, it can directly change membrane properties and cellular function, which is independent of 5-HT receptors. Other studies have also shown that 5-HT can modulate vascular permeability [30, 31]. In addition, Engberg et al. [32] found that small membrane-binding molecules as neurotransmitters can influence essential membrane properties. The authors found that 5-HT can bind to biologically relevant membrane models that induce a significant structural change in its domain size. As a result, the conformation of membrane proteins can be changed by altered membrane properties that modify membrane signaling processes (for example, via modification of the secondary structure of G protein-coupled receptors).
There are approximately one trillion anucleated platelet cells derived from megakaryocytes in the blood. Platelets are not only mediators of homostasis and thrombin generation, but also have essential roles in various physiological and pathophysiological processes such as homeostasis, inflammation, innate and adaptive immunity, angiogenesis and tissue repair, cardiovascular disease, and cancer, among others [33, 34]. It is currently accepted that platelets work as immune cells with an incredible number of functional roles [35, 36].
The major granule types of platelets are
Several ATP-binding cassette (ABC) transporters and solute carriers (SLCs) have been found in platelets [39]. The platelet agonists, like thrombin, ADP, PAF, epinephrine, and 5-HT, interact with their 5-HT2A receptors on platelets [40]. On the platelet surface, there are serotonin transporters (SERT) and 5-HT2A receptors [41, 42, 43, 44]. SERT is an important mechanism that regulates plasma 5HT levels [45]. Platelet SERTs can quickly re-uptake secreted 5-HT from dense granules and contribute to 5-HT release during platelet activation [46].
Platelets express various CXC chemokine receptors and pattern recognition receptors (PRRs) such as Toll-like receptors (TLRs) that detect pathogen associated molecular patterns (PAMPs) from pathogens. TLRs on circulating platelets permit platelets to bind pathogens [47]. This allows platelets to efficaciously kill pathogens or present them to cells of the immune system. Platelet-derived CXC chemokines are important mediators of inflammation, immune defense, and repair processes following vascular or tissue damage [48, 49].
Platelets can communicate with other cells in different ways, such as direct cell-cell interactions by membrane receptors or indirectly through the release of various soluble factors from their granules, and the release of microparticles. Microparticles are enriched in non-coding microRNAs (miRNAs) that suppress mRNA translation through multiple mechanisms and are potent regulators of gene expression [50, 51].
Activated platelets can also release some of their mitochondria, and these extracellular mitochondria can induce paracrine or endocrine responses, interact with neutrophils, and trigger neutrophil adhesion to the endothelial wall [52, 53].
Activated platelets can mediate T-cell functions (by platelet factor 4 (PF4, CXCL4)-RANTES or serotonin.) and can also activate peripheral blood B cells and increase production of immunoglobulins [33, 54, 55].
Platelets also serve as peripheral models for neuronal 5-HT dynamics [56, 57].
Several proteins have been identified in both neurons and platelets, such as
serotonin transporters and receptors, reelin, amyloid-beta precursor protein
(APP), and brain-derived neurotrophic factor (BDNF), among others. Platelets also
contain chemokines and cytokines, and neurotransmitters (5-HT, dopamine,
epinephrine, histamine, and gamma-aminobutyric acid (GABA)) [58, 59]. Platelets
and neurons have similar secretory vesicles that store gamma-aminobutyric acid
(GABA), glutamate, dopamine, 5-HT, epinephrine, calcium, adenosine
5
Activated platelets can synthesize pro-inflammatory mediators such as platelet-activating factor (PAF), prostaglandins (PGs), and thromboxanes. Activated platelets release 5-HT and PAF that have important functions in the regulation of neuroinflammation, hemorrhage, and neuronal plasticity after traumatic brain injury [60].
In platelets and in the brain cortex, significant similarities were revealed between the characteristics of the 5-HT2A receptor or SERT structure [61, 62, 63]. Since selective serotonin reuptake inhibitors (SSRIs) block the reuptake of 5-HT into neurons as well as the uptake of 5-HT into platelets, various researchers use platelets as peripheral models for neuronal 5-HT dynamics, mainly in depression and schizophrenia, and for monitoring the effect of antidepressants [59, 64, 65, 66].
In recent decades, it has become increasingly evident that the gastrointestinal microbiota and the genome of the gut microbiota (microbiome) play essential roles in maintaining health (homeostasis). The gut-brain axis (GBA) provides, as a continuous bi-directional communication system, the flow of information between the gastrointestinal tract and the brain. Bidirectional communication within the GBA can be mediated through the vagus nerve, via the systemic nervous system by the release of hormones, microbial metabolites (short-chain fatty acids, amino acids), and neurotransmitters, and through the immune system by the production of cytokines [67, 68].
Recently, Spencer et al. [69] found that large populations of excitatory and inhibitory neurons in the enteric nervous system (ENS) produce temporally synchronized bursts of activity that are independent of the central nervous system. Nowadays, the ENS is considered as the “second brain” because it can work independently of the brain. In a new research, Morarach et al. [70] have defined 12 different kinds of neurons in the ENS of mice. Wei et al. [71] demonstrated that indole, a tryptophan metabolite, produced by tryptophanase-expressing intestinal microbes, induced neurogenic effects in the adult mouse hippocampus. This result provides a possible elucidation of how gut-brain communication is translated into brain cell renewal through molecules produced by gut microbes that stimulate neurogenesis in the adult brain.
Diverse psychological and environmental stresses can perturb the healthy functioning of the gastrointestinal microbiota system that causes dysbiosis. Dysbiosis has been associated with various diseases such as irritable bowel syndrome (IBS), cardiovascular diseases, obesity, arthritis, diabetes, kidney disease, allergy, asthma, metabolic syndrome, among others [72, 73, 74, 75, 76, 77, 78]. Antidepressants can also disturb the gut microbiome and intestinal microbial 5-HT synthesis and cause dysbiosis [15, 79, 80, 81, 82, 83, 84, 85, 86]. Relatively rarely, antidepressants can also induce visual and auditory hallucinations [87, 88, 89, 90].
Dysbiosis also has a key role in the development of neuropsychiatric, neurodevelopmental, and neurodegenerative disorders such as schizophrenia, depression, autism, attention deficit-hyperactivity disorder (ADHD), and Parkinson’s disease, among others [91, 92, 93, 94, 95]. In mice, commensal microbiota can influence the levels of brain-derived neurotrophic factor expression in the cortex and hippocampus, as well as the postnatal development of the hypothalamic-pituitary-adrenal (HPA) stress response [96]. In addition, perturbed gut microbiota can change neurotransmitter modulation in the brain and, as a consequence, can also alter mood, cognition, behavior, and memory mechanisms [97, 98, 99, 100].
We should consider that in evolution, the ENS evolved before the central nervous system (CNS) and is considered a “second brain” that can operate independently of the brain and spinal cord [101]. ENS may perform implicit learning and memorization, so it may work like a little brain in the gut [101]. Gut microbes are part of our non-conscious system that can regulate behavior [102]. Post-natal gut microbial colonization takes place in parallel with cognitive development that lasts throughout our entire lives [102].
There is a continuous dynamic interaction between the microbiome and the GI system in which 5-HT is a key signaling molecule [103]. The microbiota is required for the maturation and modulation of the ENS in a 5-HT-dependent manner [104, 105]. Various studies suggested a key role of intestinal bacteria in the regulation of 5-HT within the gut [18, 106, 107, 108]. Wikoff et al. [109] revealed that the plasma 5-HT levels were 2.8-fold higher in conventionalized mice compared with their germ-free (GF) counterparts. Mandić et al. [107] found that Clostridium ramosum promoted 5-HT secretion from ECs. In experiments by Reigstad et al. [110], short-chain fatty acids produced by gut microbes promoted colonic 5-HT production through an effect on ECs. Yano et al. [18] found that the gut microbiota has a central function in promoting levels of colon and blood 5-HT, basically by increasing the 5-HT synthesis of host ECs. Explicitly, the authors found that the microbiota can modulate high levels of peripheral 5-HT, 64% of colonic and 49% of serum concentrations. These results support the concept that the microbiota can regulate 5-HT metabolism mostly by affecting host colonic ECs. The synthesis of gut-derived 5-HT in ECs is controlled by indigenous spore-forming bacteria, mainly Turicibacter sanguinis and Clostridial species that are present in the human microbiota [18]. The indigenous microbiota modulates hippocampal levels of 5-HT, supporting the role of the microbiota in regulation of the brain’s serotonergic system [111].
The BBB is a complicated network of vasculature comprising microvessel endothelial cells with various enzymes, efflux pumps, and transporters [112]. Although the BBB is referred to as a “barrier”, the term is misleading because it allows for bidirectional cell and substance exchange [113].
Platelet activating factor (PAF) is a G-protein coupled receptor that is a ubiquitous phospholipid inflammatory mediator that performs autocrine and paracrine functions [114]. PAF is released by various cells, like platelets, monocytes/macrophages, neutrophils, and endothelial cells. PAF can modulate the function of the brain microvascular endothelial cells and temporarily and reversibly increase the permeability of the BBB [115].
Under physiological conditions, the concentration of PAF is very low in the circulation. In contrast, under inflammation, the concentration of circulating PAF essentially increases, which contributes to inflammation-mediated tissue injury, including BBB breakdown [116]. PAF binds to its receptor that mobilizes Ca2+ that activates cellular signaling pathways such as phospholipase-C-mediated signaling and induces platelet arachidonic-acid release and 5-HT secretion, which transiently increases the permeability of the BBB [116, 117, 118, 119].
In experiments by Bulat and Supek [120, 121], rats were injected intravenously with 5-HT that produced a conspicuous increase in 5-HT in the brain. Nakatani et al. [122] demonstrated that augmented brain serotonin could cross the BBB through the 5-HT transporter from the brain to the circulating blood. Recently, Young et al. [123] reported that SERT is present in the BBB of the rat, suggesting that 5-HT can enter into the CNS. Young et al. [123] emphasized that because the SERT can function bidirectionally, we must consider whether 5-HT could be transported from the bloodstream to the CNS.
Extracellular vesicles (EVs) are lipid bilayer enclosed microvesicles or exosomes of cells that are released by all types of cells into the extracellular space [124]. EVs serve cell-to-cell communication and facilitate the exchange of a wide variety of molecules between adjacent or distant cells. Platelet-derived EVs (pEVs) can release numerous soluble mediators that are in general stored in platelet granules, such as vascular endothelial growth factor (VEGF), platelet factor 4 (PF4), Willebrand factor (vWF), serotonin, among others [125, 126]. Although platelets cannot pass through tissue barriers, their EVs can enter lymph, bone marrow, and synovial fluid. Puhm et al. [124] proposed that pEVs may be transferred across tissue barriers such as the BBB, especially under inflammatory conditions.
Latest experiments found that platelets can enter the CNS parenchyma and directly interact with neuronal cells [127]. When platelets are activated in the CNS, they release various pro-inflammatory mediators, neurotrophic factors, and neurotransmitters, which stimulate neuronal electrical and synaptic activity and promote the development of new synapses and axonal reconstruction near the site of damage [127]. Kopeikina and Ponomarev emphasize [127]: “Platelets and their secreted factors could affect many cell types involved in the regulation of BBB integrity including endothelial cells, astroglia, and pericytes”.
It can be seen that 5-HT could be transported from the bloodstream to the CNS by various proposed mechanisms. Namely, by the help of pEVs [126]; by the help of SERT that is present in the BBB [123]; by means of increased plasma 5-HT level that can increase permeability of microvessel endothelial cells as well as the BBB [118, 128, 129, 130]; by PAF that modulates the function of the brain microvascular endothelial cells—that are the major component of the BBB—and increases the permeability of the BBB [115, 128]; or platelets may enter CNS parenchyma and directly interact with neuronal cells [127].
Based on the above, the view that serotonin cannot cross the BBB may be re-examined.
Classical serotonergic psychedelics create altered states of consciousness that involve a change in sensory perception, mood and, thinking, including the perception of reality and self-esteem. In the last few decades, various studies have suggested that serotonergic psychedelics can have beneficial effects on psychiatric disorders like mood and depressive symptoms, anxiety, post-traumatic stress disorder (PTSD), alcoholism, or inflammatory diseases [131, 132, 133, 134, 135, 136].
Hallucinogens are naturally occurring chemicals, including mescaline (3,4,5-trimethoxyphenethylamine), psilocybin (magic mushrooms), and DMT (N,N-dimethyltryptamine) or synthetic compounds, such as lysergic acid diethylamide (LSD), which can induce alterations in human consciousness, emotion, and cognition.
Studies agree that serotonergic psychedelics are mediated primarily by activation of the brain’s serotonin 5-HT2A receptors and that 5-HT2A receptor activation is necessary for most of the psychoactive effects [137, 138, 139, 140, 141]. However, there is evidence that interactions with other receptor sites also play a role in the psychopharmacological and behavioral effects of serotonergic hallucinogens [142, 143, 144]. Serotonergic psychedelics have a significant effect on the major neuronal populations that regulate excitatory and inhibitory neurotransmission [145]. Psychedelic agents directly activate some of the excitatory neurons expressing 5-HT2A and then other cell types, which include subpopulations of inhibitory somatostatin and parvalbumin GABAergic interneurons and astrocytes that create distinct and regional responses [146]. Nevertheless, the effects of serotonergic psychedelic drugs on neurotransmission and cortical networks have been partially elucidated to date [147].
Although serotonergic hallucinogens can produce numerous alterations in the brain networks [148], here we focus on the amygdala and the default mode network (DMN), since the amygdala gets input from all sensory modalities and is a key structure in the emotional brain, and the DMN is considered as the backbone of cortical integration [149, 150].
The DMN is a group of anatomically separated areas in the brain. DMN’s typical electrophysiological manifestations are strong low-frequency oscillations, coherent during the resting state, that are mainly activated when individuals are focused on their internal mentalstates, such as self-referential processing, interoception, autobiographical memory retrieval, or imagining the future [150, 151].
The classical regions of the DMN are the posterior cingulate cortex and retrosplenial cortex; ventromedial, anteromedial, and dorsal prefrontal cortex; temporal pole; middle temporal gyrus; hippocampus and parahippocampal cortex; amygdala and the posterior parietal cortex.
There are several studies about the function of the amygdala as related to affective dysfunctions in many psychiatric disorders [152, 153, 154, 155, 156, 157]. The human amygdala gets input from all sensory modalities, and the visual modality is the most significant for emotional aspects of social interactions. The amygdala is a key structure in the emotional brain, with diverse affective processes [158]. It seems that the amygdala may be a core brain network, and signals from the amygdala reach around 90% of the prefrontal cortex (PFC) [159, 160]. Furthermore, emotional stimulation can be processed without awareness that activates the amygdala, which has an impact on human behavior [161].
A recent review by De Gregorio et al. [162] concluded that psilocybin and LSD can modulate functional brain connectivity. Mueller et al. [163] investigated the acute effects of LSD and found a significant negative correlation between LSD-induced amygdala response to fearful stimuli and the LSD-induced subjective drug effects. Bershad et al. [164] studied the effects of a single low dose of LSD on healthy subjects. The authors found increased amygdala seed-based connectivity with the right angular gyrus, right middle frontal gyrus, and the cerebellum, and decreased amygdala connectivity with the left and right postcentral gyrus and the superior temporal gyrus. It seems that very low doses of LSD produce insignificant subjective changes but can alter brain connectivity within the limbic network.
Grimm et al. [165] investigated psilocybin’s acute effects on the amygdala in 18 healthy subjects. They found that psilocybin decreased the connectivity between the amygdala and the striatum during angry face discrimination. In addition, the happy face discrimination decreased the connectivity between the amygdala and the frontal pole. There was no effect on discrimination against fearful faces. According to the authors, psilocybin acts as a modulator of the amygdala’s major connectivity hubs. Roseman et al. [166] investigated the effects of psilocybin on 20 patients with moderate to severe, treatment-resistant depression with two separate dosing sessions with psilocybin. They observed that psilocybin with psychological support produced increased responses in the right amygdala to emotional faces in patients. However, SSRIs have the opposite effect. This suggests that while SSRIs reduce negative emotions, psilocybin allows patients to confront and work through them.
There is a fundamental discrepancy between the mechanisms of SSRIs that can decrease negative emotions and psilocybin that allows patients to achieve an emotional breakthrough [164]. Furthermore, there is an inconsistency between the results by Roseman et al. [167] and those by Kraehenmann et al. [168], since in the latter, acute treatment with psilocybin reduced amygdala activity during emotion processing, which was associated with an increase in positive mood in healthy subjects. The study by Kraehenmann et al. [168] may suggest similarities between the mechanisms of antidepressants and psilocybin. However, Roseman et al. [167] assume that this interpretation is not likely because their pre- versus post-resting-state fMRI outcomes support the notion [169] that the post-acute changes observed just one-day after a psychedelic experience are very different to those found during the acute psychedelic state. Since psilocybin acutely decreases but post-acutely increases DMN integrity [169], it can produce a transient disintegration within the normal network by reducing connectivity between the frontal cortex and the lower brain areas [170]. That is, psychedelics could create a transient perturbation of the normal neural hierarchy by reducing top-down control and increasing bottom-up information transfer [170, 171].
Studies have revealed that the serotonin system can regulate DMN connectivity. Namely, genetic polymorphisms of the 5-HT1A receptor can modulate the activity and functional connectivity of the DMN [172, 173, 174]. Abnormal connectivity in the DMN has been associated with major depressive disorder (MDD). However, there are contradictory results in studies regarding increased or decreased functional connectivity within the DMN in MDD [175, 176, 177, 178]. In addition, patients with depression present increased amygdala responses to fearful faces.
In the latest study, Shao et al. [179] found that a single dose of psilocybin produced an immediate and long-lasting increase in connections between neurons in mice. Namely, the authors demonstrated increases in the number of dendritic spines and in their size within 24 hours of the administration of psilocybin in frontal cortical pyramidal cells (Dendrites and the dendritic spines of neurons have key roles in the connectivity of the brain and are the locus of long-term synaptic plasticity, which is associated with learning and memory.). These changes were still present a month later. In addition, mice subjected to stress have shown behavioral improvements and increased neurotransmitter activity after a single dose of psilocybin.
According to Carhart-Harris et al. [180], psychedelics reduce the stability and integrity of brain networks and simultaneously reduce the degree of separateness or segregation between them. Namely, psychedelics cause network disintegration and desegregation [180]. These findings suggest that psychedelics significantly influence brain complexity and connectivity and competition of neural assemblies, which play a significant role in the mechanisms of conscious experience [4]. Winkelman suggested [181] that psychedelics enhance access to information that is normally unconscious and may become conscious and available through visual symbolic processes that use image-schemas to integrate knowledge.
In important experiments by Carhart-Harris et al. [169], nineteen patients with resistant major depression got 10 mg psilocybin, followed 1 week later with 25 mg psilocybin. Functional magnetic resonance imaging (fMRI) was performed at baseline and post-treatment at 1 day after the 25-mg dose. They found an increased DMN integrity in patients one-day post treated with psilocybin that is essentially different to that revealed during the acute psychedelic state. The authors suggested that this mechanism can be compared to a “reset” process in which the acute modular disintegration of the DMN allows subsequent reintegration and restoration of normal operation. Carhart-Harris told the BBC News website [182] about their experiments [169]: “Patients were very ready to use this analogy. Without any priming they would say, ‘I’ve been reset, reborn, rebooted’, and one patient said his brain had been defragged and cleaned up”.
It is possible that psychedelics may produce a special functional connectivity in the CNS, mainly within the DMN, with transient perturbation of the normal neural hierarchy—reduced connectivity between the frontal cortex and the lower brain areas—that makes it possible for patients to accomplish emotional resolution, in which the amygdala can have a key role [166, 171, 183]. Roseman et al. [184] proposed that there are essential differences between the therapeutic mechanisms by which SSRIs mitigate negative emotions and those by psilocybin, since the latter allows patients to confront and work with negative emotions.
Recently, we pointed out [171] that classical psychedelics increase the vividness of autobiographical memories and often encourage the recall and re-experiencing of autobiographical memories, i.e., the visual effects of psychedelics may play a key role in resetting fears [169]. These recalled memories are often coupled with strong negative or positive emotional intensity that had been avoided and/or forgotten before the experience [185]. From this perspective, psychedelics might be helpful in the memory processing of traumatic memories and stressful experiences.
The inconsistencies in neuroimaging studies about classical psychedelics [167, 169] are probably due to numerous factors. Many aspects must be considered in neuroimaging studies regarding the neural mechanisms of classic psychedelics, such as the type of classic psychedelic, oral or intravenous administration, optimal dose, dose duration and frequency, acute effect or post-acute effect, individual (biopsychosocial) factors, healthy volunteers or depressed patients, the method of measurement and its accuracy, among others.
We should also emphasize that studies support the notion that SSRIs work via normalization of the amygdala response to emotional stimuli. Namely, SSRIs can normalize amygdala reactivity by increasing responses to positive emotional stimuli and decreasing responses to negative emotional stimuli [186, 187, 188, 189].
Psychedelics induce acute effects that promote DMN disintegration and hyperconnectivity between brain areas that allow centers that do not normally communicate with each other [169, 190]. These acute biomolecular mechanisms also induce significant epigenetic changes, which can have an effect on synaptic plasticity (synaptic rewiring) and facilitate long-term changes in brain neurochemistry [179, 190, 191]. Thus, a single administration of psychedelics can establish long-lasting effects with lasting beneficial outcomes.
Serotonin syndrome (SS or serotonin toxicity) is a potentially life-threatening adverse drug reaction produced by excessive serotonergic agonism in central and peripheral nervous system serotonergic receptors [192]. Symptoms of SS include altered mental status, autonomic instability, and neuromuscular abnormalities. Antidepressants, such as SSRIs (fluoxetine, paroxetine, and fluvoxamine), require several weeks of chronic dosing before the benefit is felt, which increases the risk of adverse effects such as SS [193]. In contrast, serotonergic psychedelic agents have a rapid and long-lasting antidepressant effect [194]. Psychedelic agents may induce rapid synaptic plasticity, which may be a key mechanism by which they can exert long-term antidepressant effects [194]. In the published literature, the majority of clinical reports of SS almost always include combinations of two or more serotonergic agents, i.e., different types of antidepressants and other serotonergic drugs such as antibiotics, opioids, antihistamines, and atypical antipsychotics [195, 196]. Namely, SS typically occurs with a serotonergic drug overdose or in combination with drugs that can increase intrasynaptic serotonin [195]. However, serotonergic psychedelics alone almost never induce SS [197].
To this day, the classical paradigm prevails that basic communication in the neural system takes place through synaptic transmission. Numerous studies and experiments support that volume transmission (VT) is also a major and widespread mode of intercellular communication in the CNS [198, 199, 200, 201, 202, 203, 204, 205, 206, 207, 208].
VT intercellular communication takes place in the extracellular fluid in the brain and in the cerebrospinal fluid (CSF) [204]. Exosomes may be the primary vesicular carriers of volume transmission and important neurotransmission regulators [202, 209]. Exosomes are extracellular vesicles (ECVs) with a diameter of between 40 and 100 nm that are mainly derived from endosomes. All cells can secrete exosomes that work as mediators of near and long-distance intercellular communication. They can transfer lipids, proteins, receptors, miRNAs, RNA, DNA, among others. At the soma–dendritic level, neurons may communicate via VT through both extrasynaptic exocytosis and an ECV-mediated manner [209]. Since the BBB is a complex vascular network containing microvessel endothelial cells with various enzymes, efflux pumps, transporters, etc. [115], exosomes released from BBB microvascular endothelial cells may be a possible route of entry of plasma 5-HT and other molecules into the CNS [204].
Central monoamines such as dopamine, norepinephrine, and serotonin perform VT in the mammalian CNS, which could modulate glutamate and GABA neurons via diffusion and flow in the extracellular fluid and CSF [210, 211]. According to Fuxe and Borroto-Escuela [212], the integration of VT and synaptic transmission via receptor-receptor interactions in heteroreceptor complexes may be essential for CNS communication, which is also important in psychiatric disorders such as depression, anxiety, schizophrenia, and cocaine addiction. In schizophrenia, neuroinflammation may also perturb the integrative process of synaptic and VT signals in glutamate synapses by altering kynurenines in the mammalian brain [213]. As we pointed out, VT has fundamental regulating roles in both presynaptic and postsynaptic processes, and the extracellular space could work as an analog communication pathway in the brain [203].
The dorsal raphe nucleus (DRN) is a major source of neuromodulators in the CNS and is the largest of the serotonergic nuclei, containing approximately one-third of all 5-HT neurons in the brain [214]. From the DRN, serotonergic neurons innervate the mPFC and perform top–down control of information processes mainly by nonsynaptic volume transmission [215, 216]. The release of 5-HT in dorsal raphe neurons is achieved from vesicles in the soma, dendrites, and/or axonal varicosities, which is independent of synapses [217]. 5-HT release can be produced by neuron depolarization, by the stimulation of L-type calcium channels, by the activation of glutamatergic receptors, and/or by the activation of 5-HT2 receptors. This induced serotonin release is characterized by slow kinetics and large diffusion, i.e., volume transmission. This VT may ultimately affect the rate of discharge of serotonergic neurons and their tonic activity. In addition, serotonin cell bodies in the Raphe nucleus are surrounded by free serotonin [218, 219].
There is increasing experimental evidence that most transmitters and peptides can be released extrasynaptically [220, 221]. Synaptic and extrasynaptic exocytoses coexist in the same neurons. Extrasynaptic communication can integrate the activities of neurons, glia and blood vessels. Transmitters and peptides that are released extrasynaptically from the soma, dendrites, and axon can modulate the responses (i.e., the neuronal electrical activities) of the whole neuronal network from seconds to days. However, monoaminergic neurotransmission (dopamine, serotonin, and norepinephrine) is predominantly realized by nonsynaptic volume transmission in the CNS [203, 221]. In addition, Pérez de la Mora et al. [222] suggested that neurotransmitters within the amygdala could regulate fear learning and memories through effects on receptor mosaics in the fear circuits through wiring and VT mechanisms.
Both wake and sleep electroencephalograms (EEGs) can be biomarkers of depression and antidepressant treatments. Disturbed sleep and the sleep-wake cycle are essentially associated with various psychological disorders, including depression [223, 224, 225]. The major typical sleep EEG biomarkers of MDD are reduced rapid eye movement (REM) sleep onset latency, increased REM sleep time and increased density of REMs during REM sleep, reduced sleep efficiency, and reduced total sleep time of slow wave sleep (SWS, the deepest stage of nonrapid eye movement (NREM) sleep) [226].
There are various hypotheses that try to explain the effect of antidepressant drugs on sleep [227]. Antidepressants represent a broad class of medications that have different effects on sleep, i.e., some antidepressants can alleviate sleep problems, others can cause sleep disorders that affect the outcome of treatment [228]. In depressed patients and healthy volunteers, most antidepressants suppress REM sleep, but REM suppression is not an absolute requirement for antidepressant effects [229, 230]. In particular, SSRIs are potent inhibitors of REM sleep when administered acutely, sub-chronically, or chronically [231].
One of the special features of REM sleep is the activation of limbic and paralimbic structures (amygdala, hippocampus, and anterior cingulate cortex). It seems that the amygdala and the hippocampus are among the most active brain regions during REM sleep [232, 233]. The processing of neutral episodic memories relies on the hippocampus and adjacent structures, and emotional episodic memories receive a special boost from the amygdala that can modulate hippocampal activity and thus affect the development of emotional memory.
There is a significant overlap between dreams and psychedelic states that suggests psychedelics induce transient dreamlike subjective experiences with long-term beneficial effects on mental and physical functioning [234]. In healthy subjects, LSD produced mental imagery similar to dreaming, mainly via activation of the 5-HT2A receptor, which was associated with loss of self-boundaries and cognitive control [235]. Experiments have revealed that serotonergic hallucinogens via 5-HT2AR activation can induce visual hallucinations [236, 237]. In addition, during sleep paralysis, hallucinatory experiences present the classic features of serotonergic hallucinations that are similar to subjective states produced by hallucinogenic drugs (LSD and psilocybin), i.e., they imply visual hallucinations, mystical experiences, and extreme fear reactions [238]. Kuypers [9] emphasizes that regarding the neurobiological mechanisms of psychedelics, studies have focused on the CNS, including the immune system and the neuroendocrine system. Kuypers [9] suggested that sleep and the microbiome play a key role in the effects of regular and low doses of psychedelics, respectively. In this context, the positive effects of psychedelics may have a “psychosomatic” influence.
Kometer et al. [237] conducted double-blind, placebo-controlled trials in which 50 healthy volunteers were given moderate doses of psilocybin while high-density EEG was recorded at eyes-open and eyes-closed resting states. It was found that the current source density of neuronal oscillations at 1.5–20 Hz was decreased within the anterior and posterior cingulate cortices and the parahippocampal areas. In addition, the intensity of psilocybin-induced mystical-type experience and insightfulness correlated with the lagged phase synchronization of delta oscillations (1.5–4 Hz) between the retrosplenial cortex, the parahippocampus, and the lateral orbitofrontal area. It seems that the incidence of mystical-type experiences is a predictive factor of long-term therapeutic benefit from psychedelics [239].
Because psychedelic drugs can induce a sleep-like state and mystical-type experiences, the exploration of similar neuromolecular processes in these states may contribute to a better understanding of the mechanism of action of psychedelic drugs.
Based on the brief review of relevant literature about serotonin, platelets, classic psychedelics, gut-brain axis, BBB, and volume transmission, and based on our previous article ideas [7, 8], here we present a hypothesis about the psychosomatic mechanisms of serotonergic psychedelics (see Fig. 1).
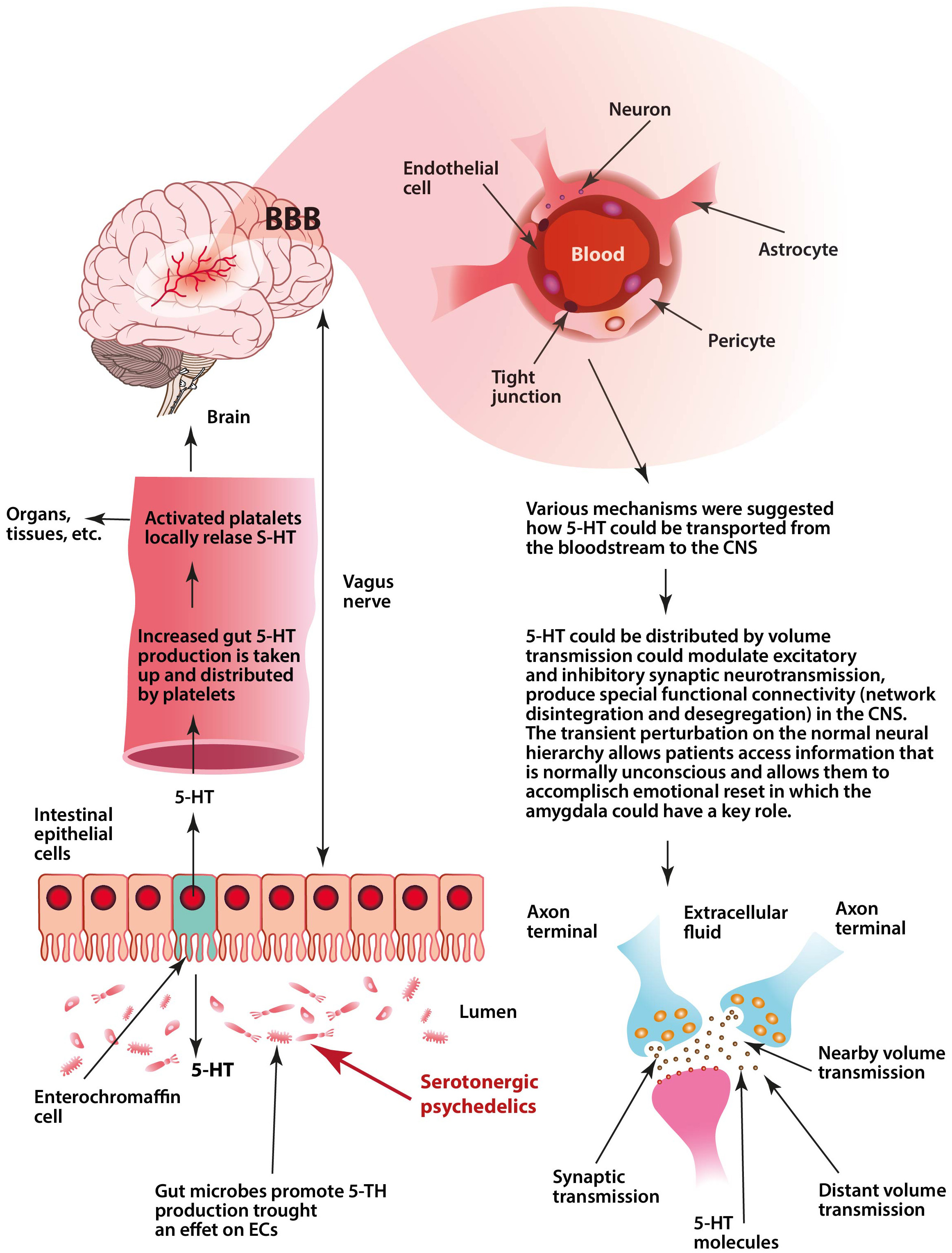 Fig. 1.
Fig. 1.Visualization of the key aspects of our hypothesis. Serotonergic psychedelics could affect gut microbes that produce a transient increase of serotonin (5-HT) by host enterochromaffin cells (ECs). This increased gut 5-HT production—which is taken up and distributed by platelets—may work as a hormone-like regulatory signal that could influence membrane structure (that modifies membrane signaling processes) and membrane permeability in the host organs and tissues and in the brain. Increased plasma 5-HT levels could enhance permeability of microvessel endothelial cells and the blood-brain barrier (BBB). Transiently increased permeability of the BBB allows for plasma 5-HT to enter the central nervous system (CNS) and be distributed by the volume transmission (VT). Then, this gut-derived 5-HT could transiently modulate excitatory and inhibitory neurotransmission, produce special network disintegration in the CNS mainly within the default mode network (DMN), and promote structural and functional neural plasticity. This transient perturbation of the normal neural hierarchy (reduced connectivity between the frontal cortex and the lower brain areas, i.e., inhibition of top-down control) allows patients access to suppressed fear information and perform an emotional reset, in which the amygdala may have a key function.
Diverse medications and environmental factors can perturb 5-HT biosynthesis in the gut that could cause dysbiosis, which impairs the serotonergic gut-brain axis and produces alterations in platelet-dependent signal processes, including changes in vascular permeability throughout the whole body as well as in the BBB [240, 241, 242].
We should consider that in the case under discussion, i.e., serotonergic psychedelics, we are not talking about dysbiosis, but a transient modulation of 5-HT biosynthesis by serotonergic psychedelics in the gut.
Serotonergic psychedelics could modulate the gut microbiota and excitatory and inhibitory neurons in the ENS. This may influence the microbiota that promotes 5-HT biosynthesis from ECs that supply 5-HT to the mucosa, lumen, and circulating platelets [18]. Recently, we raised the idea that a large part of 5-HT-produced by ECs that is taken up and distributed by platelets-may operate as a hormone-like regulatory signal for the whole body [7]. This platelet-mediated distribution of intestinal 5-HT is dependent on the intestine’s actual health condition and influences membrane permeability throughout our organization.
5-HT has a strong lipid membrane affinity, influences membrane structure properties that modify membrane signaling processes, and affects vascular permeability [28, 29, 30, 31, 32]. Consequently, the hormone-like regulatory signal by platelet 5-HT can modulate countless signal processes in the whole body. Nevertheless, here we focus on membrane permeability, particularly on BBB permeability.
Lipid rafts may also play an important function in serotonergic psychedelic mechanisms. Increased plasma 5-HT levels induced by serotonergic psychedelics may modulate the permeability and fluidity of lipid rafts in plasma membrane subregions that participate dynamically in cell signaling and molecular trafficking operations in various cell membranes like platelets and the BBB [243, 244, 245, 246].
Several studies have found that increased plasma 5-HT levels can increase the permeability of microvessel endothelial cells as well as the BBB [118, 119, 128, 129, 247]. 5-HT injected intravenously in rats produces an increase in 5-HT and 5-HIAA (5-Hydroxyindoleacetic acid) in the brain [120].
Platelets are the key regulators of plasma 5-HT concentration [45, 248]. The regulation of the SERT activity on platelets has a key role to stabilize the concentration of plasma 5HT. It has been proposed that 5-HT regulates its own plasma concentration by modulating the uptake properties of platelet SERT, and that SERT does not regulate plasma 5-HT levels [249]. Since SERT is present in the BBB, it was proposed that 5-HT may enter into the extracellular fluid of the CNS [123].
Serotonergic psychedelics could increase the circulating concentration of PAF. Increases in 5-HT in the gut and, as a result, the increased level of 5-HT carried by platelets in the blood can act as an inflammatory signal [250, 251]. This could increase the circulating concentration of PAF, so PAF can also contribute to inflammation-mediated tissue injury, including BBB breakdown [116].
Volume transmission is a general mode of intercellular communication that takes place in the extracellular fluid and in the cerebrospinal fluid of the brain [201, 203, 210]. In the CNS, serotonergic transmission is mainly achieved by a non-synaptic VT mechanism [252]. Neuropsychoactive drugs act rather as VT signals [253].
We hypothesize (see Fig. 1) that the increased level of 5-HT—induced by serotonergic psychedelics in the gut and carried by platelets—produces a transient increase in permeability in microvessel endothelial cells within the BBB. This lets 5-HT enter into the extracellular fluid of the CNS and may act via volume transmission (diffusion mechanism). As a result, serotonin has a wide range of effects in the CNS that can modulate both excitatory and inhibitory neurotransmission, in which the DMN and amygdala may have key roles.
Our hypothesis may provide a possible mechanism for how psychedelics could create a transient disintegration in the normal neural network by reducing connectivity between the frontal cortex and the lower brain areas and promoting structural and functional neural plasticity, which allows patients access to suppressed fear information and perform an emotional reset [169, 170, 171]. Finally, it seems that there are important differences between the therapeutic mechanisms by which SSRIs mitigate negative emotions and those by psilocybin, since the latter allows patients to process traumatic memories and stressful experiences [184].
The limitations of this new complex hypothesis are that many parts are difficult to substantiate with scientific literature and that findings from cited animal and cell research might not translate to the complex physiology of humans. For example, the role of volumetric transport in brain processes is not yet known, and research has been neglected to this day, although it is one of the major and widespread forms of intercellular communication in the CNS. Monoamine neurotransmission, such as dopamine, norepinephrine, and serotonin, basically performs VT that could modulate glutamate and GABA neurons in the mammalian CNS. Furthermore, 5-HT is essentially produced in the gut, which is distributed by platelets and affects the regulation of the whole body and the brain, and the existing literature suggests that 5-HT may enter the brain via the BBB and could affect brain processes. Is it possible that a portion of brain serotonin comes from the gut? Studies have also suggested that BBB permeability can be regulated continuously and extensively, allowing types of molecules to enter the CNS that we would not believe.
One may argue that such enhancement of BBB permeability by 5-HT that is derived from ECs and conveyed by platelets may cause neuronal toxicity through the passage of peripheral cytokines and toxic substances into the brain. However, it is possible that 5-HT and other various molecules can selectively enter the brain through as yet unknown mechanisms.
IB developed the concept. NCsN, PB and IB contributed to data collection, analysis and interpretation of results, and manuscript preparation.
Not applicable.
Not applicable.
This research was supported by Charles University project Cooperatio SVV.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

