1 Cellular and Network Physiology Group, Neuroscience Research Center, Charité – Universitätsmedizin Berlin, 10117 Berlin, Germany
2 Institute of Integrative Neuroanatomy, Charité – Universitätsmedizin Berlin, 10117 Berlin, Germany
3 Department of Neurosurgery, Lucerne Cantonal Hospital, 6000 Lucerne, Switzerland
4 Institute for Diagnostics of Epilepsy, Epilepsy-Center Berlin-Brandenburg, 10365 Berlin, Germany
5 Department of Neurology with Experimental Neurology, Charité – Universitätsmedizin Berlin, 10117 Berlin, Germany
Academic Editors: Ayataka Fujimoto, Tohru Okanishi and Rafael Franco
Abstract
Surgery is an essential treatment option for patients with drug-resistant epilepsy. While most epilepsy patients worldwide live in low- and middle-income countries (LMIC), most of these countries do not have epilepsy surgery, and those that do have surgical epilepsy services lack capacities. The rapidly growing population in LMIC further widens the gap between the number of patients who can potentially benefit from surgery and those who can actually receive it. This makes the initiation of new surgical epilepsy centers in those countries an urgent issue. Epilepsy surgery is feasible in LMIC, even in resource-poor settings, but lack of local expertise is a major obstacle to the introduction of new surgical services. Importantly, expertise deficits can be compensated by collaborating with a well-established epilepsy center for knowledge transfer, skill building and mentoring. Such projects need to be organized in a multidisciplinary team, should focus on the given circumstances, and should use technologies and personnel that are reasonably available and can function sustainably. Local cultural factors and improvement of patients’ quality of life are further spotlights reflected by an increasing number of studies. As a general outline for a new surgical epilepsy program in LMIC, it is recommended to initially focus on patients with mesial temporal lobe epilepsy due to hippocampal sclerosis or other well defined pathologies. These constellations have an excellent surgical outcome in terms of seizure control, can be diagnosed by non-invasive methods, and can be reliably identified even under low-resource conditions. Moreover, surgery can be performed with a highly standardized approach and at reasonable costs, and the vast majority of patients will benefit from surgical intervention. The range of services can then be gradually expanded, depending on growing expertise, local needs, prospects and constraints. Although the introduction of surgical epilepsy services in LMIC can face several challenges, none of them should be a permanent barrier for further establishments.
Keywords
- drug-resistant epilepsy
- knowledge transfer
- mesial temporal lobe epilepsy
- presurgical evaluation
- postsurgical care
Antiseizure medication (ASM) fails to control seizure activity in about 30% of epilepsy patients (drug-resistant epilepsy, DRE [1]). While the development of more than 20 new ASM over the past three decades has not had a substantial impact on the rate of DRE [2, 3], surgical intervention is an essential treatment option for patients with medically refractory focal-onset seizures [2, 4]. Consequently, great efforts are being made to expand the pool of surgically treatable DRE patients through the development of more complex diagnostics (including e.g., positron emission tomography, PET, or single-photon emission computed tomography, SPECT), focused surgery with reduced tissue damage (like stereotactic laser ablation), advanced investigative technologies (like intracranial EEG-recordings with subdural or depth electrodes) or non-destructive extra- and intracranial neurostimulation [5, 6]. However, the global volume of epileptic disorders is not evenly distributed [7], with 80% of affected people living in low- and middle-income countries (LMIC [8]). This is an important issue as LMIC may not only lack the ability to perform complex diagnostic and surgical scenarios, but the option of epilepsy surgery may not be available at all, may only be available to a small proportion of patients who can afford surgery abroad, or may only be performed in a small number of centers that are insufficient to meet all the demand. Increasing efforts are being made at the international level to address this problem, such as the ILAE task force on ‘Epilepsy Surgery in Low Resource Settings’ of the ILAE Surgical Therapies Commission (https://www.ilae.org/about-ilae/committees-task-forces-and-advisory-commissions/epilepsy-surgery-in-low-resource-settings), but the gap is still wide. A recent review identified epilepsy surgery programs in only 22% of the LMIC [9]. In the ASEAN countries, for example, epilepsy surgery is underutilized due to a lack of surgical epilepsy centers and appropriately trained personal [10]. A recent survey for Africa [11] found that epilepsy surgery is performed in only 8 out of 51 countries, with only one country reporting invasive presurgical diagnostics. As a consequence, clear-cut cases with excellent outcome prognosis, such as patients with mesial temporal lobe epilepsy (MTLE) with hippocampal sclerosis (HS), accumulate in LMIC [12, 13, 14], even though the establishment of low-cost epilepsy surgery programs is feasible in countries with resource-poor settings [15, 16]. Furthermore, with a rapidly growing population in LMIC, the gap between the number of patients who could benefit from surgical intervention and those who can actually receive it is widening yearly [11], making the initiation of surgical epilepsy services for those countries a timely issue. While there appears to be broad consensus on the basis for establishing such programs, as outlined in several early publications [17, 18, 19, 20, 21, 22, 23, 24], subsequent reports have emphasized additional aspects, substantiated former claims and set out new spotlights. It is our aim to briefly summarize the established basics and highlight new building blocks and key aspects in light of recent publications. This endeavor was triggered by our own experience [25, 26]. In this regard, it is also a personal view that draws on the growing literature to discuss barriers and challenges, in order to propose a framework for building of such a surgical program. With this approach, we hope to stimulate and facilitate further efforts to establish new epilepsy surgical services in LMIC, which are essential to reduce the surgical treatment gap worldwide.
Success is the best advocate for acceptance of a new neurosurgical program [20], especially because many people have concerns about brain surgery [27, 28]. Importantly, the outcome perspective of epilepsy surgery correlates with pathological findings and causes of the disease [29, 30], making patient selection a decisive factor for the expected success rate. This may put pressure on the selection of ideal candidates who are expected to have a postsurgical seizure-free outcome (Engel class I; seizure-free with or without residual auras). In addition, newly established centers in LMIC usually operate with limitations in terms of locally available technology, experience and expertise. The burden, however, is offset for the launching phase of a new surgical epilepsy program in LMIC by the unanimous recommendation to initially confine surgical candidates to patients with drug-resistant MTLE with HS [17, 19, 20, 23], or other well defined pathologies, such as glioma. Temporal lobe epilepsy (TLE) is the most common type of surgically treated focal epilepsy in the adult population [31, 32], and MTLE-HS is the best understood form in terms of risk factors, clinical seizure characteristics, and EEG findings [23]. It can be diagnosed in most cases with noninvasive procedures and minimal technical requirements [19, 23, 24], the diagnostic workup is clearly outlined [33], the temporal lobe is relatively easy to operate on [32], and the well-established standard anterior temporal lobectomy (ATL) can be used as a surgical procedure [34], preferentially on the non-dominant hemisphere for the introductory phase. Taking such considerations into account, a suitable standard procedure should first be established, which can then be gradually expanded to more complex and challenging diagnostic and surgical scenarios, hand in hand with increasing expertise [19, 20]. In the following outlines, we will thus focus on DRE with MTLE-HS to illustrate common limitations for the establishment of a new surgical program, even though other obvious radiographic lesions such as glioneuronal tumors or cavernous malformations are also not challenging to diagnose and could be well addressed within a resource-limited setting.
MTLE-HS is not only the prototype of a surgically remediable epileptic syndrome [33], but with more than 80% of patients achieving remission also the one with the best seizure outcome [35, 36, 37]. For DRE MTLE patients, surgery is in fact the most effective treatment, it is safe and superior to prolonged drug therapy [38, 39, 40]. In LMIC, surgery is also superior to drug therapy, which is also true for DRE in children as demonstrated by Dwivedi, et al. [41]. Notably, seizure-free rates in pediatric patients is equal to or even higher than in adult patients [42, 43], making surgical treatment a promising approach in selected pediatric DRE cases. However, because pediatric and adult epilepsy surgery are markedly different in several aspects [43], we focus here on the adult population, in particular on patients with MTLE-HS. Importantly, while surgery itself is the culminating point, the entire process of DRE surgical intervention is based on three pillars: presurgical diagnostics, the surgical intervention itself and postsurgical long-term follow-up care (Fig. 1). These parts are sequentially organized, focus on distinct personnel and include different timescales to be considered.
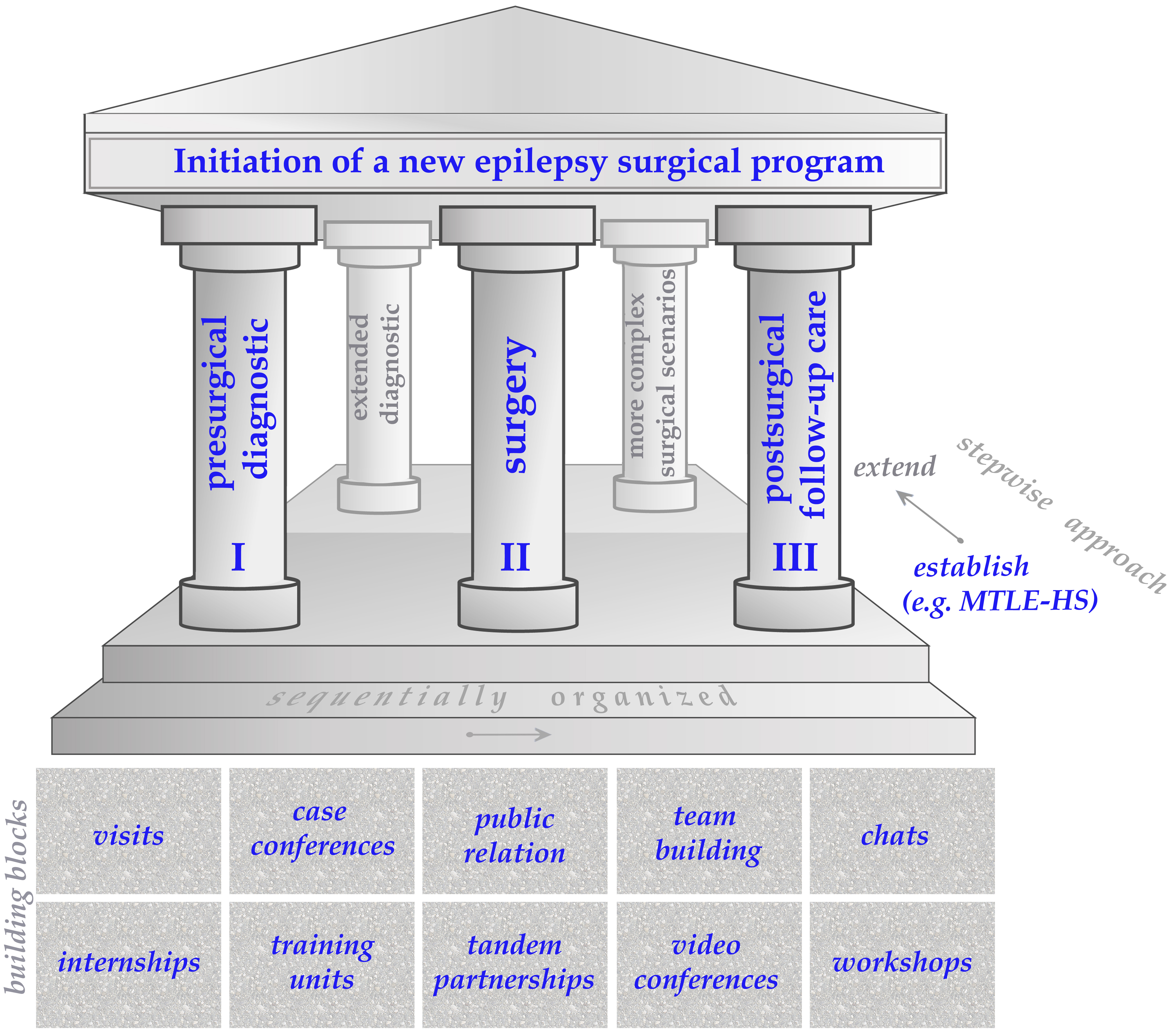 Fig. 1.
Fig. 1.Pillars of an epilepsy surgery program and useful building blocks.
Epilepsy surgery is an effective and safe treatment option for a subset but not all DRE patients [44], therefore adequate presurgical examination is essential for the application of surgical intervention [45]. Importantly, patients who have been referred with the diagnosis of DRE require a systematic and comprehensive assessment. First of all, the nature of seizures, former medication and the claim for refractoriness has to be critically reviewed and second the potential for surgical candidacy has to be evaluated [19, 20]. The primary aim of presurgical diagnostics is then to determine the epileptogenic zone and its relationship to eloquent brain areas (lateralize and localize [46]) in order to propose eligible candidates without compromising the patient’s safety [20]. This process depends on an adequate level of basic local infrastructure (technology dependent) as well as a comprehensive epilepsy care organization were those candidates can be subjected by a multidisciplinary team (competence dependent [47]). However, surgical epilepsy centers in LMIC will lack the full range of state-of-the-art technologies that is available in high-income countries (HIC [47]), and it is essential to identify the candidates that are appropriate for the existing medical infrastructure [23]. Classical MTLE-HS patients can be reliable selected by a noninvasive protocol comprising history taking, interictal EEG or preferable ictal video-EEG monitoring (VEM), magnetic resonance imaging (MRI) or computed tomography (CT) and neuropsychological assessment [19, 48]. The radiological detection of a single, unilateral and well-circumscribed lesion that is potentially epileptogenic is an extremely important finding. If such a lesion is surgically accessible, concordant to the clinical history and EEG evaluation, it is most likely the cause for seizure, and surgery can be recommended [17, 23].
Detection rates for radiographic lesions have increased over time with the improvement of MRI technology [6], but it is worth noting that patients who underwent surgery for MTLE up to 1990 were evaluated with 1980s technology (mostly 16–32 channel VEM recording system, facilities to perform Wada tests and 0.5–1.0 T MRI machines or even earlier with CT imaging [19, 49, 50]). Those patients already had very good outcome results [33, 51]. In a resource-limited setting MRI might be out of scope and CT could be a cheaper and available alternative [15]. Radiological examination would be done early in the evaluation process to identify a putative local lesion indicative for a positive outcome prognosis [23]. Subsequent VEM is also a cost-intensive part in the whole setting [33], and some publications highlight the possibility to recommend surgery in selected cases of consistent unilateral temporal interictal epileptiform abnormalities without VEM [52, 53, 54, 55, 56], but the underlying decision-making process strongly relies on experienced epileptologists. Not using VEM should therefore not be the first choice outside a well-established epilepsy center with a high level of expertise [52], but might be implemented if technological resources are lacking [57]. VEM can further provide a crucial piece of information to verify the diagnosis, characterize ictal behavior and aiding to localize the epileptogenic zone [17], which makes it highly desirable if affordable. In addition, a comprehensive neuropsychological evaluation is helpful in assessing potential neurocognitive risk factors [6], and a good history taking is another key for the selection [17]. Overall, realistic presurgical protocols have to be developed based on locally available technology and personnel to assess surgical candidacy [15, 19, 23]. Notably, a surgical epilepsy center with a rudimentary setting based on noninvasive procedures can already help a large proportion of DRE patients (approx. 50%–60% [19]). Such a center depends on the availability of well-trained personnel, in particular the epileptologist, rather than on the need for sophisticated state-of-the-art equipment [19, 20]. In line with this, the upscaling of surgically capable epilepsy centers in China, for example, is not restricting by monetary funding, but by the quest for qualified personnel [32]. A good initial mentoring program with a well-established epilepsy center is thus highly recommended. Such cooperation is further helpful in reconciling the desire for widespread use of surgical epilepsy interventions with the need for careful assessment and to act in this potential area of conflict [15, 19, 58].
Resective surgery is the first choice for treating focal DRE when a single seizure focus can be well localized and surgery can be performed safely without causing intolerable neurologic deficits [46]. The optimal outcome of surgical intervention is a complete cessation of seizures without any postsurgical deficits [17, 59]. The accurate demarcation of both seizure foci and eloquent cortices is essential for this purpose [23, 60]. During the surgical procedure, the epileptogenic tissue (epileptogenic zone [61]), and not only the lesion needs to be removed in order to achieve seizure freedom. Importantly, the epileptogenic zone is a theoretical construct defined by the minimum amount of tissue that has to be removed to gain seizure freedom [33], but the exact zone cannot be characterized reliably by presurgical investigations [59, 61]. Therefore, a compromise between the extent of surgically removed tissue and the reduction of potential impairment is required [59]. Interestingly, health-related quality of life worsens after surgery if persistent seizures are accompanied by memory decline, but the latter alone doesn’t result in a negative assessment [62]. For MTLE-HS patients, a standard ATL is not only a well-established surgical routine, but also a well-established compromise between seizure control and none or at most minimal neuropsychological and neurological impairments. An alternative surgical procedure is selective amygdalohippocampectomy [63]. Eventually, the neurosurgeon should use the method which is the standard of care in this center and which he feels most comfortable with. In the end, a highly localized epileptic focus predicts the best surgical outcome and not the surgical procedure applied [64].
Complications are commonly transient or minor, major complications are rare, and the surgical seizure outcome in LMIC is comparable to that of HIC [9]. The development of a primary standard routine, taken from the well-established mentoring center and adapted to the local conditions, is a valuable tool to implement and improve local surgical care of patients. But the local circumstances and equipment can be quite distinct from what is normally used by the mentoring experts, which may require creative solutions [65]. Working together can also be hampered by language barriers, local customs, unexpected routines, or unfamiliar interactions. Successful mentoring thus also depends on a motivated surgical team in order to overcome such obstacles and focus primarily on the surgical implementation. To make epilepsy surgery an established practice, it is further recommended that surgical procedures should be performed on a regular base rather than conducted as single events [9, 20, 58].
It is the responsibility of the neurosurgeon, within the context of the multidisciplinary team, to decide whether surgery is feasible and advisable [66]. Therefore, the surgeon must adequately understand and interpret the diagnostic data to identify as accurately as possible the epileptogenic zone where surgery has to be carried out. Consequently, the role of the neurosurgeon is not limited to the surgical procedure itself. He is also an integral part of the multidisciplinary team to decide on surgical interventions, especially when it comes to predicting the likelihood of success or failure of a particular procedure for a given candidate [46, 59, 66]. Thus, the participation of the neurosurgeon in multidisciplinary case conferences is mandatory. To fulfill this role, the surgeon should not only be trained in epilepsy-related surgical techniques, but also be tought knowledge of seizure semiology and the corresponding radiological findings [46].
A surgeon is also essential if invasive diagnostics like intracranial EEG are required to clarify the seizure onset zone [6, 58], but the surgical requirements in terms of equipment and competency must be met for any chosen procedure [15, 23]. Stereotactic EEG recording through intracranial electrodes is an increasingly common procedure in HIC [67], but this invasive technique requires robotic or stereotactic system assistance and an appropriate epilepsy monitoring unit. Consequently, it is quite demanding in terms of equipment, expertise, and diagnostic resources, which might be a major barrier to adoption in resource-limited settings. Such expensive state-of-the-art equipment may not be feasible for local surgical epilepsy centers in LMIC, so cost-effective methods are crucial [15] and low-cost options should be used first. For example, further diagnostics to clarify a suspected bilateral epileptic focus can be carried out with subdural or depth EEG recordings, but also with foramen ovale electrode recordings [26]. This technique is less demanding, minimally invasive, and less expensive compared with subdural or depth EEG recordings, but also provides an effective way to assess lateralization of the seizure focus in MTLE patients [68].
Patients hope to gain seizure remission by surgical intervention. However, for some patients, surgery might not be effective or even worsen the situation (Engel class III or IV [58, 69]). Reasons could be a wrong localization or an incomplete resection of the epileptogenic zone. In both cases, reoperation may be beneficial for the patient [70]. In rare cases, patients might possess an occult epileptogenic area with a higher threshold for seizure activity that is activated after the resection of the primary epileptogenic region [69]. Hidden bitemporal onset zones do not exhibit particular clinical semiology or preoperative discordant information and are therefore not seen on initial diagnostic studies. Those patients are not eligible for a second surgical resection, but neurostimulation might be an option [70]. In the event of surgical failure, patients should be reexamined to determine the cause of failure and to clarify further options [71]. While multiple seizures within the first postsurgical year are in general associated with a poor long-term prognosis [72], it should be noted that some patients with persistent seizures after surgery may achieve long-term remission without changes in ASM or further surgical intervention (so called running-down phenomenon [29, 73]).
In a prospective longitudinal cohort follow-up study over a period of up to 18 years, the risk of recurrences for surgical MTLE-HS patients was 22% in the first 24 months, followed by a further 1.4% annually [74]. This finding depicts a dualistic dynamic of acquired epilepsy and demonstrates that MTLE-HS patients, in addition to surgical failures, remain at significant risk for seizure recurrence after surgical intervention, even if they have been seizure-free for more than 5 years [29, 75]. Hence, the overall outcome evaluation is also a factor of time [57, 74, 75, 76]. In the aforementioned study, 62% of MTLE-HS patients remained completely seizure-free 18 years after surgery [74], and in another 26-year follow-up study, 57% of patients with temporal resection remained seizure-free during the observation period [76]. Importantly, epilepsy and in particular acquired MTLE is a network disease, where the process of epileptogenesis turns the neural network into a pathological condition. Those alterations go beyond the epileptic focus [77, 78], and the brain remains prone to pathological synchronization. Accordingly, late recurrences often behave like new-onset epilepsy [72]. However, a second focus is not common in humans and late recurrences usually do not presage the return of frequent uncontrolled seizures, but are rather rare events in most patients. Half of the patients with recurrences who are seizure-free initially (for at least one year after surgery) have one or fewer seizures per year [79] and half of the patients with late recurrences (being seizure-free for at least five years after surgery) subsequently enter remission [75]. Importantly, a change in ASM might result in seizure occurrence [80, 81, 82, 83, 84] and tapering or omission of ASM could cause postsurgical seizures [29]. MTLE-HS is special in the sense that surgery might turn a DRE in a drug-controlled one [85]. Hence, precaution should be taken in the reduction or a switch of ASM and drug discontinuation should not be done directly after surgery. On the other hand, continous use of ASM does not prevent seizure recurrences [77, 80, 86] and vice versa discontinuation of medication after one [87] or two years [88] is not associated with an increased relapse rate. Furthermore, most patients who experience seizures after drug-discontinuation become seizure-free with continued medication [83]. Taking together, ASM should be continued after surgery, but there is no favorable time to eventually start tapering or finally withdraw ASM [89]. The decision to stay with, reduce or discontinue ASM depends, at least in part, on patient preference [75], but patients should be fully informed of the steps taken and well monitored and counseled [90]. Notably, seizure freedom is the main factor for a positive assessment of the quality of life (QoL) [91, 92, 93, 94, 95] and by far the most important predictor for improvement in QoL after surgery [96], whereas continued medication is unrelated to this dimension [97].
In developing countries, epilepsy surgery offers the opportunity to reduce stigma and improve QoL [92, 97, 98, 99, 100, 101, 102], also with a positive impact on patients with low resources and precarious social circumstances [103]. Sustained and meaningful improvements in QoL, as patients hope for from surgical interventions, are mainly achieved within the first two years after surgery [91, 97]. Unfortunately, half of the relapses also occur during this period. Therefore, in addition to direct postoperative care, patients should receive appropriate counseling during this two years after surgery, if possible (pillar III, Fig. 1).
Although there is generally a marked improvement in QoL after epilepsy surgery, this is of course not a guaranteed effect. To achieve meaningful improvement in QoL from a patient’s perspective, the patient must acknowledge a change as important for his or her life. In this way, QoL is a useful method to assess patient’s satisfaction with the treatment [97], but the assessment of QoL is highly dependent on the focused dimension [96]. Seizure freedom is just one dimension, another are comorbidities that can exacerbate the burden of epilepsy and may need to be treated in parallel. In developing countries, parasitic and infectious diseases are the most common comorbidities [104]. Neurocysticercosis for example is the major cause of acquired epilepsy in tropical low-income countries [105]. However, psychiatric comorbidities are also a common phenomenon in refractory focal epilepsy (e.g., 40% of patients [106]), especially depression and anxiety disorders [107]. For example, in an epidemiological survey in Canadian communities [108], these two disorders were found in 30% of epilepsy patients, and comparable or higher rates are assumed in LMIC [104]. It is thus beneficial if possible to include a psychiatrist in the assessment team [109], and psychiatric disorders as well as other comorbidities should be evaluated as part of the presurgical diagnostic process whenever possible [110]. Patients hope to gain remission of disease burden through surgical epilepsy intervention, but patients who undergo surgical resection are prone to psychiatric disorders in the postsurgical period, either as an exacerbation of a known psychiatric diagnosis or the development of a new disorder. These postsurgical psychiatric disorders can significantly affect the QoL and overshadow a positive outcome of surgical seizure treatment [92, 111]. Surgical intervention may also have a positive impact on existing depression and anxiety in some cases [112], but unrealistic expectations have a negative impact on patient’s QoL rating [96]. It should be recognized that surgical intervention is intended to relieve or reduce seizures and not to treat possible comorbidities, which may persist afterwards and should be treated separately. Patients should develop realistic expectations in order to adequately assess the benefits of surgery, and thus it is particularly important to appropriately explain outcome perspectives to patients and their families. Although seizure freedom is the patient’s primary concern and an important predictor of improvement in QoL, a reduction in seizure frequency could already be judged positively. It is worth to mention that although patient selection is a decisive factor for the final success in terms of seizure-free rates, a medical project does not aim to achieve optimal statistic performance, but to help patients in the best possible way by balancing outcome perspectives with risk factors and patient expectations. In the end, surgery is beneficial for the vast majority of drug-resistant MTLE patients [91] and it is generally associated with an improvement in QoL that then lasts for decades [76, 96].
Surgical epilepsy services are based on a sequentially organized process that focuses on different personnel and tasks. Therefore, the first step is an assessment of the current situation, whereby a broad kick-off event can be helpful [25]. Once suitable partners are found who are motivated to participate in the project, knowledge transfer, training and internships can be organized through different building blocks that could function as separate modules (Fig. 1). In this whole process, it is natural to start with the presurgical evaluation and the quest for eligible surgical candidates, accompanied by surgical training and followed by the long-term follow-up care.
Presurgical evaluation and epilepsy surgery requires training and experience that is unlikely to be present in developing countries [15, 45]. For a sustainable approach, well-trained people are essential to train future specialists on site [19]. Thus, if a surgical epilepsy program should have a lasting impact, in addition to building on locally available technology, training of local professionals is critical, and the project has to create appropriate educational opportunities [23]. This could include visits by established experts to local facilities, accompanied by local training units [26], to provide knowledge transfer, advice and consultation. It is a valuable tool to initiate direct partnerships between individual professionals and to carry out profession-specific training and knowledge transfer. On the other hand, local workshops, joined internships and training sessions in larger groups are fruitful to disseminate knowledge and initiate team building. Both parts can be integrated into focused events, depending on the availability of resources and opportunities. In the end, the surgical decision-making involves a multidisciplinary team, making team building an important element [20, 58]. When it comes to the surgical intervention itself, a tandem approach has proven effective, with the experienced surgeon initially acting as primary surgeon to advise the local surgical team and care for the establishment of appropriate routines, followed by the newly introduced neurosurgeon who then performs surgery under the supervision of the experienced colleague [15, 21, 26, 58, 113].
In general, time abroad can be spent in a single block (e.g., Boling W, et al.[15]) or in multiple units (e.g., Dugladze, et al. [26]), but time is a valuable resource, in particular for clinicians beneficially participating in a mentoring program. Hence, the essential time for a stay abroad should be carefully considered. Notably, many activities can be planned and implemented through volunteerism, but financial support, e.g., from governmental programs or developmental aid, is needed at least to cover travel expenses. It is also helpful that information technology can link the center in a LMIC with the partner organization’s experts, e.g., for data analysis, treatment recommendations, and patient selection [15, 114]. This is well in line with the increasing global trend, fostered by pandemic situation such as a Covid-19 crisis, to take advantage of videoconferencing, electronic communications, and virtual case conferencing. Data transmission is ubiquitous nowadays, it can be exploited for mentoring or even be established as an independent building block [114]. Electronic communication allows experienced colleagues to participate remotely in joined case conferences, discuss diagnostic findings, or advise on the management of ASM [15, 26, 114, 115]. Initial personal contact is highly valued in the introductory phase and in initiating tandem partnerships, but workflows in presurgical diagnostics and postsurgical care (pillar I and III in Fig. 1) could also be organized via electronic communication channels. However, from our own experience, internships, joined local case conferences and workshops are generally appreciated [26]. Regardless of the choice of building blocks, the appropriate workflow must be organized in interdisciplinary cooperation.
The integration of various building blocks is one side, the stepwise approach from a basic routine to more challenging scenarios the other (Fig. 1). Proper selection of appropriate diagnostics and their accurate interpretation at each stage is required to ensure the balance between outcome perspective and risk factors [110]. This path must be followed according to local constraints in terms of available technology, increasing expertise, patient needs, and economic constraints. The evolution of a center from a stepwise perspective does not imply continuous improvement in surgical outcomes, as basic surgical epilepsy centers achieve comparable outcomes to well-established ones [9, 21, 22, 24, 45, 48, 116, 117]. Rather, the evolution implies the types of patients that can be treated and the different procedures that have been performed over the years [19]. Experienced and well-established centers can usually offer a broad range of diagnostic and interventional tools, whereas newly established centers should initially defer more difficult-to-treat patients to a later date as experiences develops, or refer them to a better equipped and/or more qualified surgical epilepsy center [20]. Such an evolution is nicely illustrated in a recent report from the neurosurgical center in Semarang, Indonesia [58]. The surgical program started with MTLE patients with seizure-related lesion, under the direction of mentoring universities from Japan, but invasive recordings and surgery in patients without MRI confirmed lesions were initially unfeasible. Subsequently, the spectrum was expanded with the introduction of subdural electrode recordings, long-term EEG and PET examination for difficult cases. Selective amygdalohippocampectomy and the treatment of extratemporal lobe epilepsy were introduced [58] and the center was enabled to perform surgery for radiologically normal appearing temporal lobe epilepsy patients [118]. The evolution of a center in this sense means that more and more complex investigative and surgical procedures are added [19, 116], while a major challenge for such a development often lies in the paucity or resources [32, 118].
A single epilepsy center may already have a large impact in small countries such as Georgia [26], but for large countries such as India or China, a multi-tiered system of epilepsy centers with different levels of expertise is advisable [16, 19, 32, 116]. At the most basic level, specially trained physicians would treat and care for patients with epilepsy according to standard protocols, simple centers could perform temporal lobectomy according to standardized protocols for diagnosis, assessment, and surgery, while comprehensive epilepsy surgery centers are needed for further diagnostics and more complex surgical scenarios. Partnerships with regional epilepsy centers are an appropriate means to improve local access to surgical care through specialized centers [119], and appropriate training of rural physicians or medical personnel is essential to identify referral candidates. For this reason, integration of all levels of epilepsy care is needed to close the surgical treatment gap in the long term [120]. It is noteworthy that even if epilepsy surgery already exists in an LMIC, surgical centers are most likely located in the major cities [9] and are not sufficient to cover all the needs [10, 116, 121, 122]. Thus, on the one hand, capacity building is an important issue, and on the other hand, there is a parallel need to raise awareness of surgical options in order to extend the impact to rural areas.
Teamwork is essential for the road to success [123] and it is crucial that all stakeholders, including patients and their families, are appropriately integrated [66]. At best, patients should receive comprehensive advice [124], and surgical epilepsy interventions should be introduced as a treatment option in the case of DRE right from the beginning of a newly diagnosed epilepsy [27, 39]. Patients who have achieved seizure freedom and/or experienced a meaningful improvement in their QoL through epilepsy surgery are the best advocates for a surgical program to publicize the effort. However, as pointed out by Sylaja & Radhakrishnan [20], one patient satisfied with the outcome of epilepsy surgery may inform five other epilepsy patients, but one dissatisfied patient will spread its adversity to 50 others. Realistic concepts are valuable tools for avoiding exaggerated or false expectations. Misconceptions and concerns are also found in HIC for both, physicians [28, 125, 126, 127, 128] and patients [28, 124, 129, 130], adding to the surgical treatment gap [44, 131] and making epilepsy surgery to one of the most underutilized interventions of all proven effective therapies [2, 27]. Cultural beliefs and local attitudes must also be taken into account [132]. People with epilepsy may suffer serious disadvantages due to misunderstanding or stigmatization, e.g., association of epilepsy with mental disease, insanity or as divine punishment [133, 134, 135, 136, 137, 138]. Stigma in the form of prevailing local beliefs associated with negative stereotypes may be quite resilient and might have a greater negative impact than the disease itself [139]. In addition, patients may have significant decision-making conflicts or fear of brain surgery [140], and rather than a promising therapeutic approach, it may still be viewed a last resort by professionals [40]. Therefore, to achieve a lasting impact, it is also important to work with the local public and professionals, educating and informing them about epilepsy and, in particular, about the possibilities and potential of surgical interventions [20]. Public relations can thus be a valuable tool for raising awareness and promoting acceptance of epilepsy surgery [26, 141], including one’s own project work, which can be developed to a separate building block.
Epilepsy imposes significant economic costs [142], greatly affects the people’s ability to work and contributes to social and psychological burden [143, 144, 145, 146]. A seizure-free person could be better employed and generally achieves an improvement in QoL [9, 20, 48, 85]. DRE is further associated with high morbidity and mortality rates [2]. Surgery is a cost-efficient treatment for DRE in terms of long-term direct, indirect and intangible costs [45, 147, 148, 149], making it an essential component of a comprehensive therapeutic approach to epilepsy [17, 150]. It should be noted that the median epilepsy prevalence and incidence in LMIC may be almost twice that in HIC [151], with even higher rates for the lowest-income countries (e.g., incidence may be 10-fold higher than in HIC [11]) and proportional numbers of drug-resistant variants. While the number of MTLE-HS surgeries in European epilepsy centers has decreased in recent decades thanks to ongoing efforts including therapy and surgery [42, 152, 153, 154], MTLE-HS patients still accumulate in LMIC without surgical epilepsy services. Importantly, epilepsy surgery in developing countries is not only feasible but also cost-effective [17, 21, 48, 155]. The cost per surgery in LMIC is usually just a fraction of the comparable costs in HIC [9, 10, 16, 21, 22, 156], but without qualified local personal or appropriate centers, such potential economic benefits could not be realized and exploited.
LMIC might have an unevenly distributed healthcare system and the cost for epilepsy surgery may not be covered by the public health sector or private health insurances [47]. As a consequence, patients have to bear the cost out of their own pocket, which may be unaffordable for most of them [20, 47, 48, 156]. However, the lower prize in LMIC will expand the group of patients who can afford surgical intervention. The option for a complete local service (pillar I-III, Fig. 1) is also more favorable from a medical point of view than surgery abroad, as patients there could face high daily costs and corresponding pressure to return to their resident country as soon as possible, even if this is not advisable. In addition, long-term aftercare should be organized locally. Otherwise, it will be missing or once again involve travel expenses and possibly high medical costs abroad. Therefore, it is valuable for a country to initiate its own surgical services. A successful establishment could also be a key argument for a change in policy so that the healthcare system or government funding might bear the costs in the future. It is further important to understand the long-term costs associated with different treatment modalities in order to economically evaluate the impact of surgical interventions [45]. However, the balance between the high initial cost of presurgical evaluation and surgical intervention and future economic benefits is difficult to estimate a priori in individual cases. It is not a short-term effect and strongly depends on the specific situation, outcome, and improvement in QoL [147, 149, 157]. Economic constraints can be an obstacle to epilepsy surgery, but the individual decision to undergo surgery, if financially feasible, is not primarily determined by the economic perspective but by the desire to overcome the disease burden [140]. Importantly, at the macroeconomic level, the expansion and public funding of epilepsy treatments, including surgical interventions, has been shown to be an overall cost-effective national strategy that reduces the financial burden of the disease in national economic terms [158].
Epilepsy is a common and shattering neurological disorder for which DRE patients have little hope of achieving adequate seizure control with medication alone. This is a devastated prospect and a heavy burden not only on their QoL, but also on the healthcare system. Epilepsy surgery is an effective and safe treatment option for a subset of DRE patients, particular those with MTLE-HS, which are straightforward to identify, even in resource-poor settings. While the number of MTLE-HS surgeries is decreasing in HIC, these patients are still accumulating in LMIC, in particular in those countries without surgical epilepsy services. Epilepsy surgery in LMIC is feasible, but it must rely on technology and expertise that are reasonably available and can function sustainably. It is important to emphasize that despite possible resource constraints, basic surgical epilepsy facilities rely primarily on well-trained personnel, which unfortunately are often lacking, rather than the need for sophisticated cutting-edge technology. To compensate for deficits in expertise, a good mentoring program is highly recommended. The necessary knowledge transfer can be organized with various building blocks by creating educational opportunities, while electronic communication channels can reduce the necessary travel time for the experts involved. Exaggerated expectations are a negative factor in the patient’s evaluation of surgical success. Therefore, stakeholder engagement, educational work and public relations work are suitable means to reduce existing knowledge gaps, dispel misconceptions, and raise awareness of surgical epilepsy intervention. Although the feasibility of epilepsy surgery in LMIC has been documented by numerous reports, in the face of insufficient capacity and a rapidly growing population, it remains an urgent issue and an essential step to reduce the burden of DRE worldwide.
PB, US, MH, TG and TD wrote the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript.
Not applicable.
Not applicable.
This work was supported by the German Federal Ministry of Economic Cooperation and Development (BMZ) and the German Society of International Cooperation (GIZ) (Grants Number: GIZ: 17.2170.3.-004.04 and GIZ: 17.2170.3.-002.08, for TG and TD) and by the grant German Research Foundation (DFG; GL 254/8-1 for TG).
The authors declare no conflict of interest.
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

