1 Department of Anesthesiology, the First Affiliated Hospital of Jinan University, 510630 Guangzhou, Guangdong, China
2 Department of Anesthesiology, Meizhou People’s Hospital (Huangtang Hospital), 514031 Meizhou, Guangdong, China
3 Medical Laboratory Center, Meizhou People’s Hospital (Huangtang Hospital), 514031 Meizhou, Guangdong, China
4 Department of Orthopedics, Meizhou People’s Hospital (Huangtang Hospital), 514031 Meizhou, Guangdong, China
Abstract
Collapsin response mediator protein 4 (CRMP4) is critical for neuronal development. However, whether CRMP4 could be SUMOylated and how the SUMOylation regulates the interaction with the L-type voltage-gated calcium channel (Cav1.2), neurite outgrowth, and thermal pain sensitivity remain to be elucidated. To determine the SUMOylation of CRMP4, Glutathione S-transferase (GST) - Small Ubiquitin-like Modifier 1 (-SUMO1), -SUMO2, and -SUMO3 proteins were purified for GST-pulldown. Immunofluorescence staining was performed to observe colocalization of CRMP4 and SUMOs. Co-immunoprecipitation (co-IP) was performed to assess the interaction between CRMP4 and SUMO2. GST-pulldown and co-IP were performed to verify the interaction between CRMP4 and Cav1.2. The impact of SUMOylation of CRMP4 on its interaction with Cav1.2 was determined. Then, the effect of CRMP4 SUMOylation on neurite outgrowth was observed. Whole-cell patch clamping revealed the effect of CRMP4 SUMOylation on Cav1.2 mediated calcium influx. Paw withdrawal latency was measured to assess the impact of CRMP4 SUMOylation on thermal pain sensitivity in rats. The data revealed that CRMP4 K374 is a potential site for SUMO modification. SUMO1, SUMO2, and SUMO3 can all interact with CRMP4. SUMO2 interacts with CRMP4, but not a variant of CRMP4 harboring a mutation of K374. CRMP4 and SUMO proteins colocalized in neurites, and CRMP4 deSUMOylation promoted neurite outgrowth. CRMP4 interacted with Cav1.2, and deSUMOylation of CRMP4 strengthened this interaction. CRMP4 promoted calcium influx via Cav1.2, and overexpression of CRMP4 significantly increased thermal pain sensitivity in rats, which CRMP4 deSUMOylation strengthened. In conclusion, these data demonstrate the SUMOylation of CRMP4, elucidate the impacts of SUMOylation on the interaction with Cav1.2 on neurite outgrowth and thermal pain sensitivity.
Keywords
- CRMP4
- SUMO protein
- SUMOylation
- L-type voltage-gated calcium channel
- Neurite outgrowth
- Thermal pain sensitivity
- Cytoskeletal dynamics
Neuropathic pain, a type of chronic pain caused by direct injury or dysfunction of the peripheral or central nervous system, manifests as spontaneous pain, hyperalgesia, allodynia, and paresthesia, along with other clinical features [1, 2]. Neuropathic pain is one of the most common clinical causes of chronic pain [3]. According to a survey by the Institute of Medicine of the National Academy of Sciences, at least 116 million adults in the United States suffer from chronic pain; neuropathic pain accounts for 17.9% of the total, corresponding to 3.5% [4]. The causes of neuropathic pain are diverse, and the pathogenesis is complex, with no effective clinical treatment [5, 6]. Several lines of evidence indicate that central sensitization, a condition of the nervous system with the development and maintenance of chronic pain, results from elevated synaptic transmission [7, 8]. Increases in chemical synapse transmission occurring simultaneously are closely related to the formation of neuropathic pain [9]. Calcium channels are important for neural transmission in CNS synapses and have been identified as key targets for synaptic signal transduction and treatment of chronic pain [10, 11]. In particular, L-type voltage-gated calcium channel (Cav1.2) controls short- and long-term sensitization to pain [12, 13]. However, it remains to be determined how calcium channels, especially Cav1.2, are regulated.
Collapsin response mediator protein 4 (CRMP4), one of five members of the CRMP family (CRMPs, CRMP 1–5), is a cytosolic phosphoprotein highly expressed in the CNS [14]. CRMPs regulate neuronal differentiation and axonal development by interacting with microtubule and actin cytoskeletons [15]. CRMP4 promotes F-actin assembly to regulate axonal growth and neurodegeneration [16]. CRMP4 and CRMP2 interact to coordinate cytoskeletal dynamics, regulating growth cone development and axon growth [17]. CRMP4 localizes in the synapses and regulates dendritic growth and maturation [14]. In mice, inhibition of CRMP4 induces phenotypes similar to those observed in human patients with autism [18], such as impaired social interaction, abnormal sensory sensitivity, changes in dendritic structure, and abnormal gene expression. Together, these observations indicate that CRMP4 plays an important role in synapses. CRMP2 regulation with calcium channels is involved in chronic pain [19, 20]; however, the role of CRMP4 in the central sensitization of chronic pain remains obscure, as does the underlying mechanism. Here, we applied the thermal pain sensitivity on rats as a method to reveal neuropathic pain [21, 22, 23].
In this research, we demonstrate that CRMP4 is post-translationally SUMOylated and that the deSUMOylated form of CRMP4 promotes the interaction with Cav1.2 to modulate calcium influx, modulates neurite outgrowth and regulates thermal pain sensitivity.
The CRMP4, Cav1.2 (a1c,
Cortical neurons were separated as previously reported [24]. Briefly, newborn
1-day-old Sprague-Dawley rats from the Animal Center of Jinan University were
sacrificed by CO
GFP-overexpressing neurons were immunostained with GFP antibody to boost the GFP signal and reveal the neurites to measure cortical neuron morphology. Images were captured in a blinded manner with an Axio Observer Z1 (#AXIO Observer Z1, Carl Zeiss, Germany). At least 60 neurons in each group were imaged. The total neurite length and branch tip number of each neuron were analyzed using ImageJ [26].
The procedure was performed as previously reported [27]. Briefly, cell lysates
were separated by 10% SDS-PAGE and transferred onto a PVDF membrane (Millipore,
Sigma, Burlington, MA, USA). The membrane was blocked at room temperature for 1 h
with 5% non-fat milk in TBS containing 0.1% Tween-20 and then incubated at 4
GST-CRMP4 and GST-Cav1.2 (a1c) plasmids were transformed into Top10F
For immunoprecipitation assays, neuronal extracts were prepared by
solubilization in cell lysis buffer (Beyotime Institute of Biotechnology) for 10
min at 4
Immunofluorescence assays were performed as previously described [28]. Briefly,
neurons cultured on coverslips were fixed in 4% paraformaldehyde for 40 min at 4
HEK293T cells cultured on coverslips were transfected with DsRed-Cav1.2
(
The viral vectors, including AAV-GFP, AAV-CRMP4-GFP and AAV-CRMP4-K374R-GFP
(serotypes 2, 1
Individual rats (male Sprague-Dawley rat, 180–200 g, 8 weeks of age) were placed in test chambers, supported by an elevated platform, with no bottom or cover on a wide-gauge wire mesh. After habituation for 30 min, mechanical pain thresholds were measured, and the 50% paw withdrawal threshold (PWT) was determined as described previously [29]. A heat/light source was positioned under the glass surface, pointing to the plantar surface of the hind paw. Thermal paw withdrawal latencies (PWLs) were measured using a plantar anesthesia tester (Boerni, Tianjin, China).
The experimental data are presented as means
Using bioinformatics tools (SUMPsp, SUMOplot, and GPS-SUMO), we found that
CRMP4K374 was a potential SUMOylation site. The GKMD sequence around CRMP4 K374
is consistent with the canonical
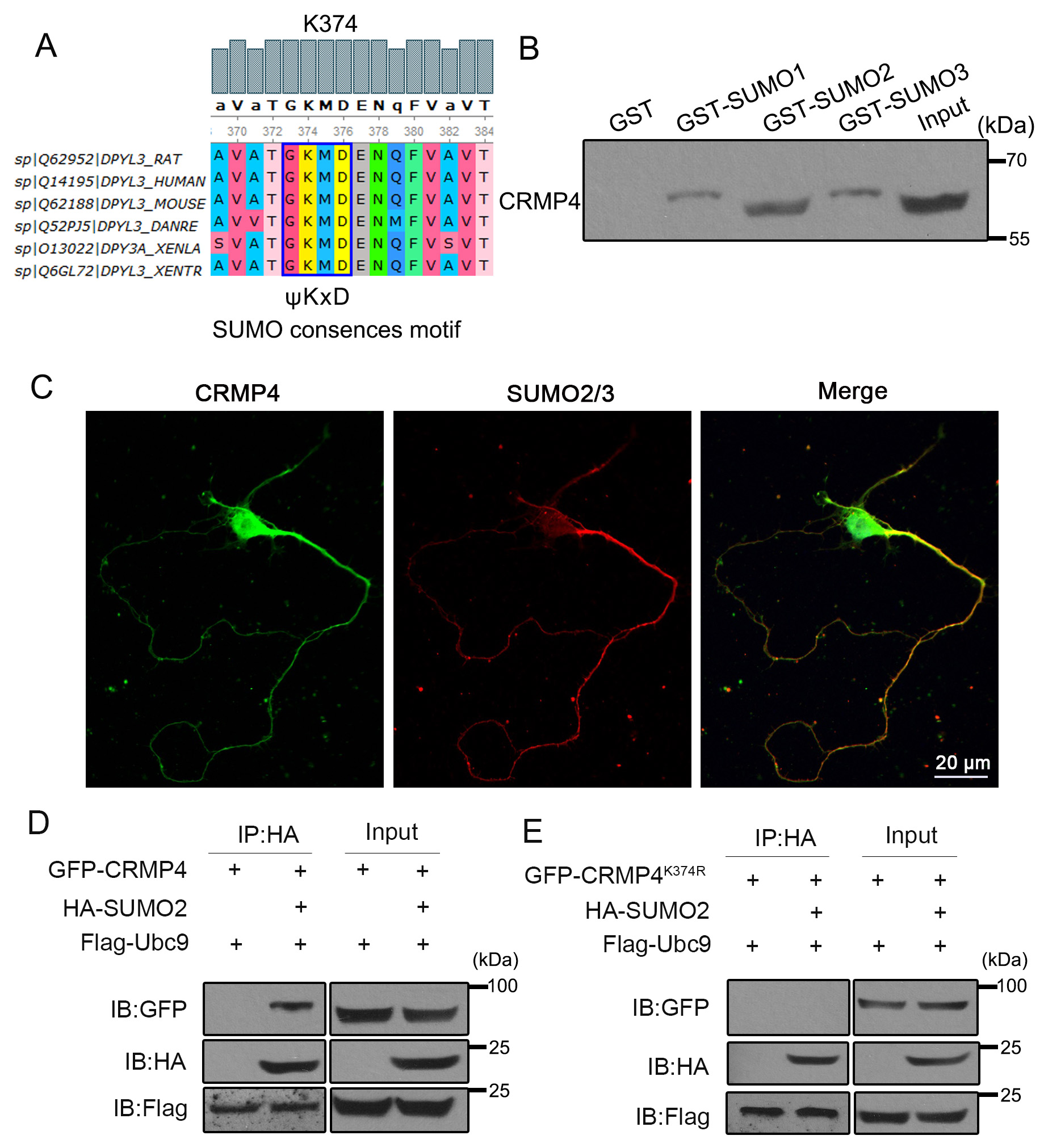 Fig. 1.
Fig. 1.CRMP4 can be SUMOylated at K374. (A) The amino acids around
K374 are conserved among species.
To further explore the detailed mechanism of CRMP4 SUMOylation, we applied
interaction proteomics to identify the functional partners of CRMP4. This
approach revealed that the L-type calcium channel Cav1.2 interacted with CRMP4
(data no shown). Hence, we performed GST-pulldown and immunoprecipitation
experiments to confirm this potential interaction. Purified GST-CRMP4 or
GST-Cav1.2 protein was incubated with cortical neuronal lysates, and then the
pulldown sediments were subjected to western blotting. As shown in Fig. 2A–B,
Cav1.2 was detected in the GST-CRMP4 pulldown sediment and CRMP4 in the
GST-Cav1.2 pulldown sediment. Next, we subjected neuronal lysates to
co-immunoprecipitation experiments using CRMP4 and Cav1.2 antibodies. The results
confirmed that CRMP4 and Cav1.2 proteins were present in each other’s
immunoprecipitates (Fig. 2C–D). Together, these data indicate that CRMP4
interacts with Cav1.2. Next, we asked whether CRMP4 SUMOylation affects the
interaction between CRMP4 and Cav1.2. To this end, we transfected HEK293 cells
with V5-Cav1.2, GFP-CRMP4, or GFP-CRMP4
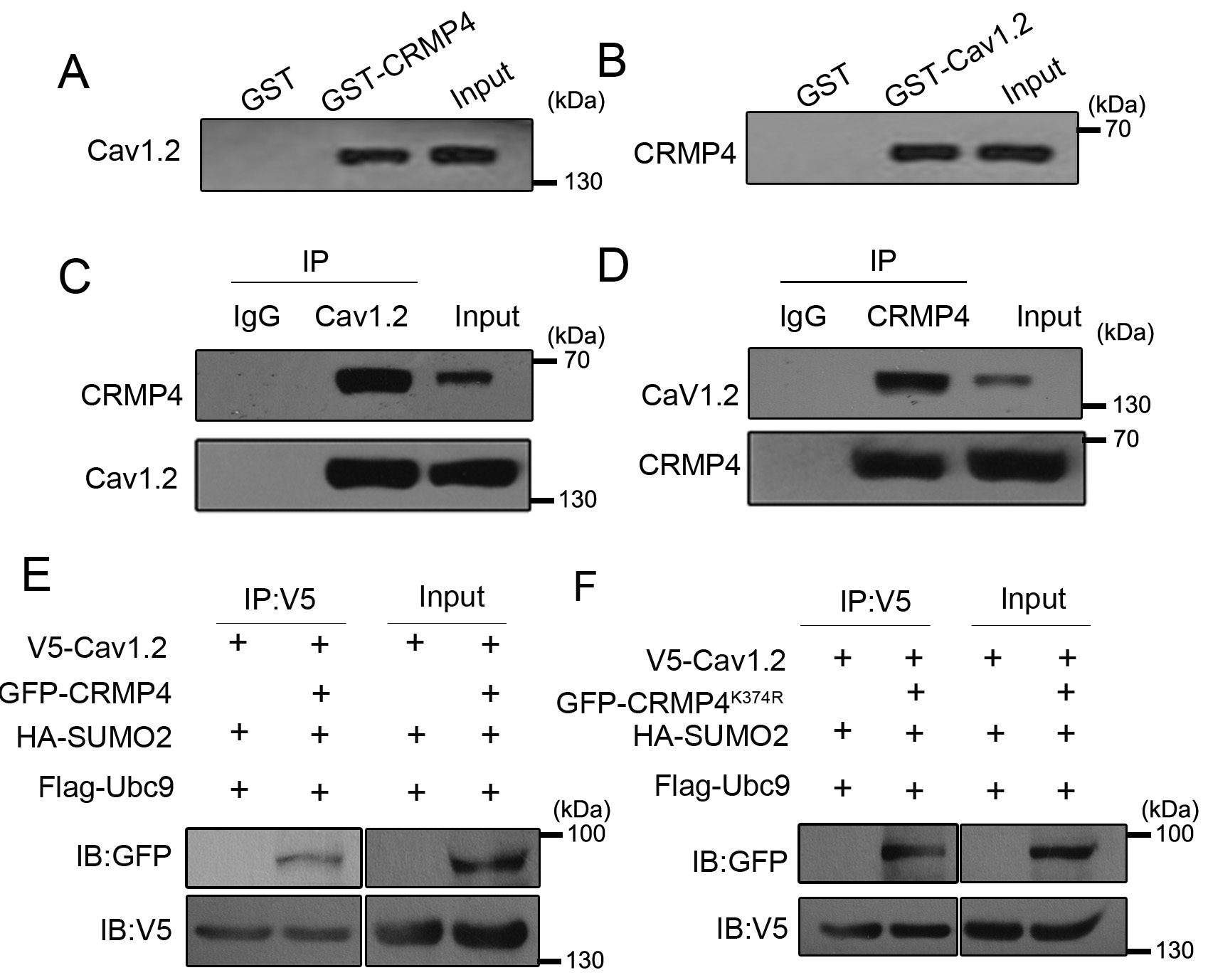 Fig. 2.
Fig. 2.CRMP4 interacts with Cav1.2. (A,B) Neuronal lysates were incubated with GST or GST-CRMP4 and GST-Cav1.2 for GST-pulldown assays, and the pulldown sediments were subjected to western blotting. (C,D) Neuronal lysates were incubated with IgG or Cav1.2/CRMP4 antibodies for immunoprecipitation assay, and the sediments were subjected to western blotting analysis. (E,F) 293T cells were transfected with the indicated plasmids; 24 h later, cells were harvested for immunoprecipitation with V5 antibody and western blotting with GFP or V5 antibody. N = 3 for all experiments.
CRMP4 promotes neurite growth in cultured hippocampal neurons [14, 30]. However,
the role of CRMP4 SUMOylation remains to be explored. To further evaluate the
effect of CRMP SUMOylation on neurite growth, we transfected cortical neurons
with GFP control, GFP-CRMP4, and GFP-CRMP4
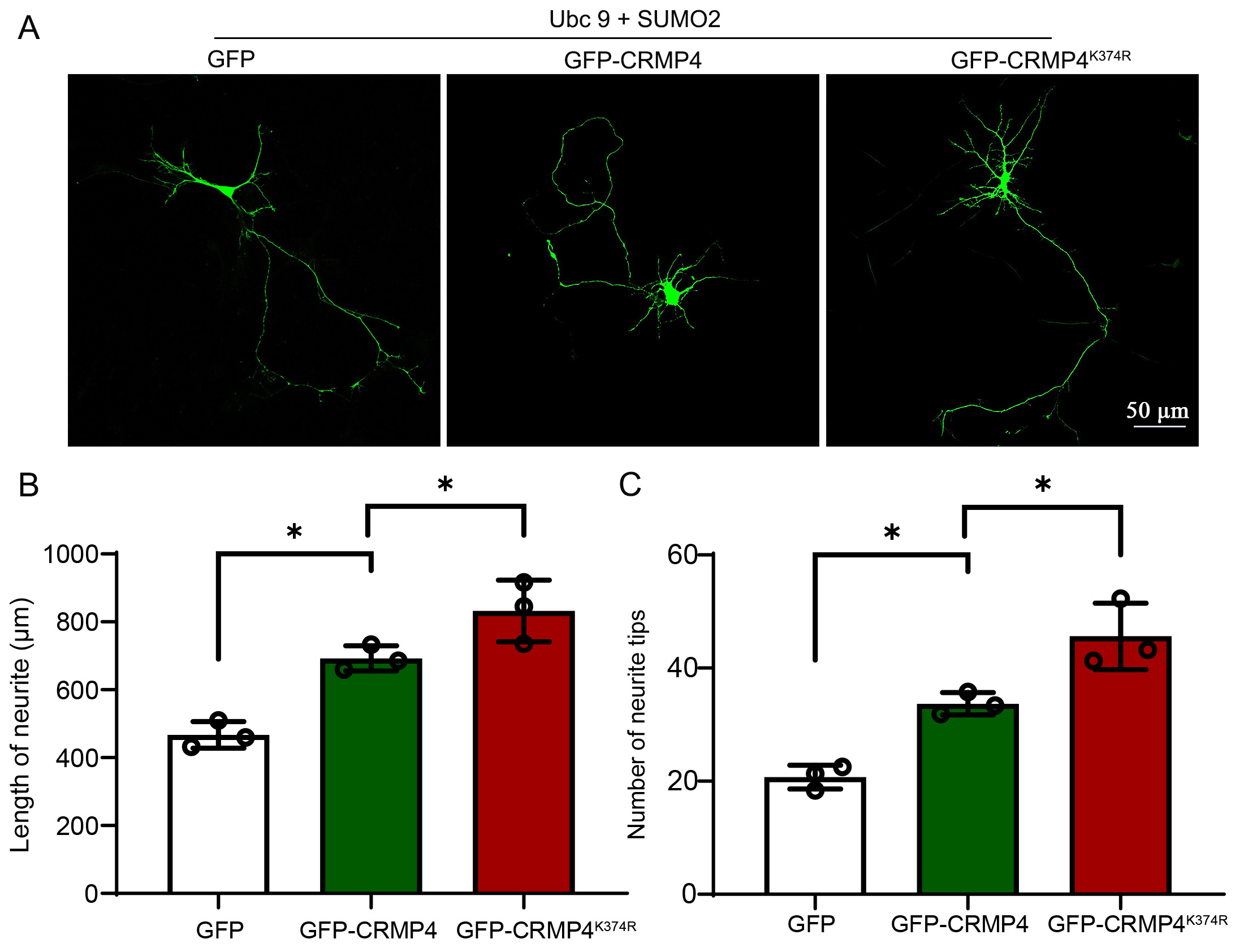 Fig. 3.
Fig. 3.The deSUMOylated form of CRMP4 promotes neurite growth. (A)
DIV3 Cultured neurons were transfected with plasmids encoding Ubc9, SUMO2, GFP,
GFP-CRMP4, or GFP-CRMP4K374R, then harvested DIV5 neuronal morphological
analysis. Representative images of neurons are shown in (A), and length per
neuron (B) and the number of tips per neuron (C) were measured in each group.
Each group was enrolled in over 30 neurons. N = 3; Scale bar, 50
The interaction between CRMP4 with Cav1.2 suggests that CRMP4 is involved in
calcium influx and neuronal signal transmission. To confirm this, we transfected
HEK293 cells with a plasmid encoding GFP, GFP-CRMP4, or GFP-CRMP4
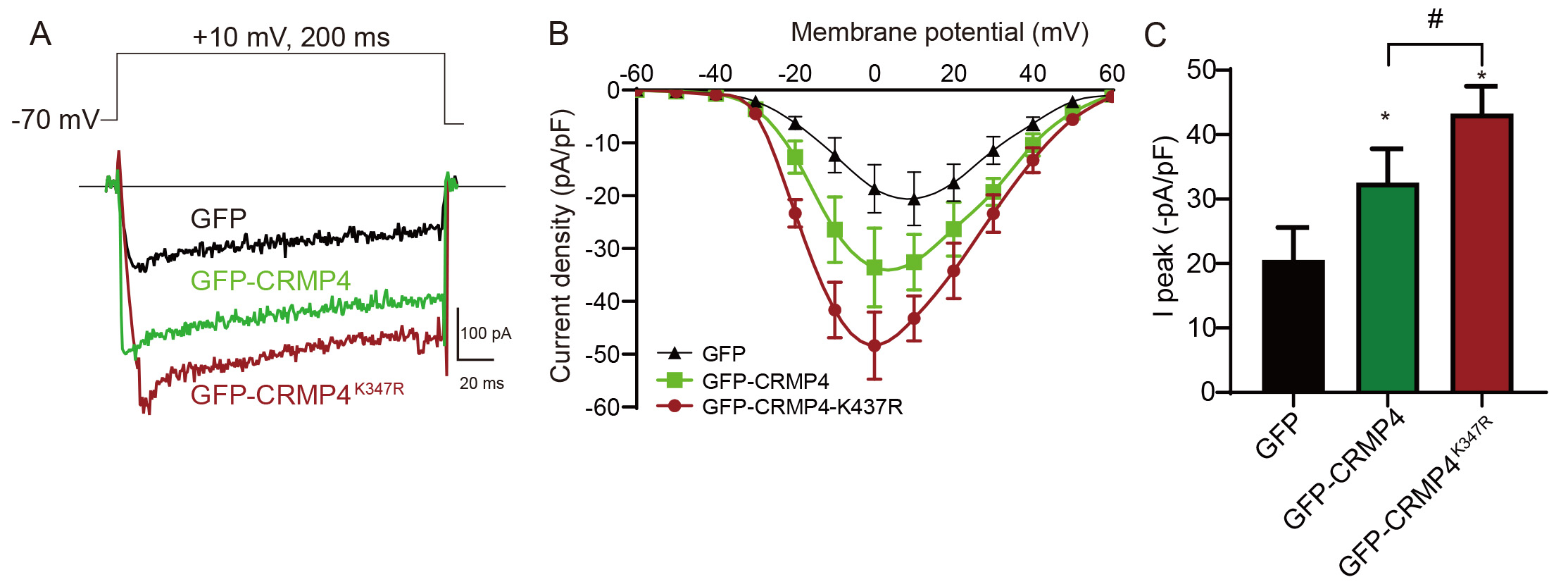 Fig. 4.
Fig. 4.CRMP4 SUMOylation regulates calcium influx via Cav1.2. (A) 293T
cells were transfected with Cav1.2 (
Because Cav1.2 is involved in pain signaling [10], we asked whether CRMP4 and
SUMOylation regulate pain sensation. To determine this, we used a rat model of
thermal pain sensitivity. CRMP4-overexpressing AAV (AAV-CRMP4 and
AAV-CRMP4
 Fig. 5.
Fig. 5.CRMP4 SUMOylation regulates thermal pain sensitivity in rats.
AAV viruses of Control, CRMP4, or CRMP4
In this study, we demonstrated that CRMP4 could be SUMOylated at K374. In addition, CRMP4 interacts with Cav1.2 to regulate calcium current density, which SUMOylation of CRMP4 could enhance. The deSUMOylated form of CRMP4 promotes neurite outgrowth. Furthermore, CRMP4 increases thermal pain sensitivity and SUMOylation of CRMP4 influences the effects. Our findings collectively reveal a new mechanism by which CRMP4 SUMOylation regulates its interaction with Cav1.2, neurite growth, and signal transmission during pain sensitivity.
Recent work showing that SUMOylation regulates CRMP2 has been revealed. K374 of
CRMP2 can be SUMOylated [31]. This modification changes the interaction of CRMP2
with calcium and sodium channels, promoting the Na
In general, nerve damage leads to persistent neuropathic pain via changes in
primary sensory neurons and central nervous system pain pathways [40]. During
these processes, voltage-gated calcium channels play important roles in
upregulating sensory neuronal excitability and promoting the transmission of pain
signals [41]. CRMPs often interact with voltage-gated calcium channels (VGCCs).
The Ca
In summary, we found that SUMOylation of CRMP4 affects its interaction with Cav1.2, neurite outgrowth and thermal pain sensitivity (Fig. 6). These findings may provide new insights into CRMP-related regulation of neuronal development and reveal novel therapeutic targets for the clinical treatment of chronic pain.
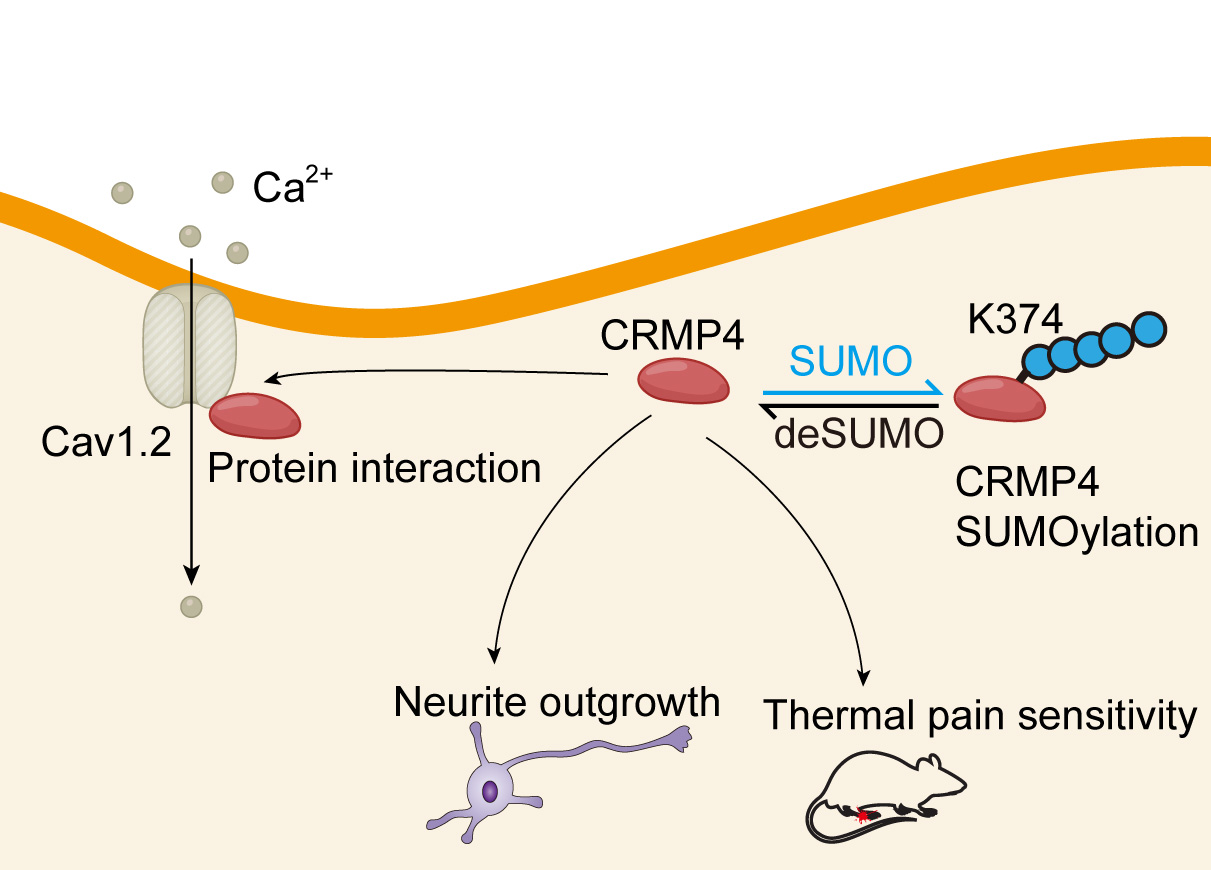 Fig. 6.
Fig. 6.CRMP4 could be SUMOylated, and SUMOylation affects its interaction with Cav1.2, neurite outgrowth and thermal pain sensitivity.
CBD3, calcium-binding domain 3; CRMP, collapsin response mediator protein; DIV,
days in vitro; GSK-3
YL conceived and designed the experiments; SL performed the experiments; MP and HL analyzed the data; JC and YJ contributed reagents and materials; SL and YL wrote the paper.
All animal procedures were approved by the Institutional Animal Care and Use Committee at Jinan University (2018122302) and were performed in accordance with the guidelines of the Institutional Animal Care and Use Committee of the National Institutes of Health.
Not applicable.
This work was supported by the Major Technological Breakthrough Project of Meizhou People’s Hospital (No. 2019-74), the Scientific Research and Cultivation Project of Meizhou People’s Hospital (No. PY-C2019023), and the Science and Technology Project of Meizhou City (No. 2018B018).
The authors declare no conflict of interest.
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.






