1 IRCCS Centro Neurolesi “Bonino-Pulejo” S.S. 113, Contrada Casazza, 98124 Messina, Italy
2 Department of Biomedical and Biotechnological Science, University of Catania, Via S. Sofia, 98, 95123 Catania CT., Italy
3 tudio di Riabilitazione Nutrizionale e Cognitiva, Via Sant’Agostino, 14, 98121, Messina, Italy
4 OU Policlinico G. Martino, Via Consolare Valeria, 1, 98124 Messina, Italy
Abstract
The purpose of this commentary is to investigate the pathophysiological mechanisms underlying hypersexuality and its manifestation in neurological diseases through a meta-analysis. Studies were identified by searching on PubMed, Web of Science and Cochrane databases. All results of each database between 2014 and 2020 were evaluated for possible inclusion. After an accurate revision of complete manuscripts, forty articles satisfied the inclusion/exclusion criteria. Data from our meta-analysis indicated hypersexuality to be a frequent sexual disorder in patients with neurological disorders, especially neurodegenerative ones. Hypersexuality could negatively affect a patient’s management and outcomes. This commentary discusses studies that are often incomplete for evaluation measures or sample selection. In our opinion, it is necessary to consider hypersexuality with particular attention, so more extensive sample studies are needed to find the most appropriate treatment to improve the quality of life for both the patient and the caregiver.
Keywords
- Sexual addiction
- Hypersexuality
- Neurological disorder
- Pathophysiology
Hypersexuality (HS) refers to abnormally increased or extreme involvement in any sexual activity. HS consists of worries about intense and repetitive sexual behaviors, fantasies, and impulses. The patient has to show significant distress or difficulty in social and/or occupational functioning. Symptoms must persist for at least 6 months [1, 2, 3, 4]. Moreover, HS consists of sexual comments and extreme behavior caused by high libido and sexual disinhibition (such as masturbating and showing genitals). Patients may experience changes in sexual orientation, and often exhibit excessive hugging/kissing, delusions of marital infidelity; they may also perform acts towards others such as grabbing the genitals and/or breasts or chasing others for sexual reasons [5, 6, 7, 8, 9]. HS typically occurs in adolescence (with a mean age of onset is 18.7 years) and has a duration of approximately 12.3 years [10, 11, 12].
In recent years, some authors have considered HS as a behavioral addiction, but there has been intense debate on the appropriateness of this classification [5]. However, the World Health Organization has included the compulsive sexual behavior disorder into Impulse Control Disorders, as per the chapter on Mental and Behavioral Disorders of the Eleventh Revision of the World Health Organization’s International Classification of Diseases and Related Health Problems (ICD-11) [4]. According to this, the new diagnosis considers this disorder as “a persistent pattern of inability to control intense and repetitive sexual impulses, or urges, with repetitive sexual behavior for an extended period (greater than six months) that causes marked discomfort or impairment to be personal, family, social, educational, occupational or other important areas of functioning”. The diagnosis can be made in the presence of the following features: (i) involvement in repetitive sexual activities represents the fulcrum of the person’s life, which may neglect other important aspects, such as health, interests, and responsibilities; (ii) the person tries unsuccessfully to control or decrease repetitive sexual behavior; (iii) despite the negative consequences in one’s life (relationship failures, health difficulties), the person fails to reduce repetitive sexual behaviors; (iv) the person engages in repetitive sexual behavior even if he does not derive satisfaction from it. Therefore, diagnosis cannot be made only in the presence of increased sexual interest or behaviors, which are common in adolescents, unless there is also impaired control over sexual behavior, significant distress, and impaired daily functioning. In addition, the patient must not have physiological alterations resulting from alcohol or drug abuse or other general medical conditions [4, 5].
Disruption of the endocrine/metabolic response at the neural or vascular level can lead to HS. It results from multiple causes, such as drugs, diseases, or aging. The mechanics involved are complex and poorly understood. Indeed, sexual behavior is mediated by multiple interactions involving the somatic and autonomic nervous system at the central and peripheral level, allowing the processing of sexual stimuli, desire, arousal, and orgasm [13]. Thus, although HS is mainly a psychiatric symptom (and thus sometimes associated with schizophrenia and major depression), it may be caused potentially by all neurological disorders involving the brain pathways subtending human sexual behavior.
This is why HS might be not so uncommon (although often underreported) in several neurological disorders, compromising the quality of life and psychosocial well-being.
This commentary aims to investigate the pathophysiological mechanisms underlying HS and its manifestation in neurological diseases.
We identified articles through careful search in PubMed, Web of Science, and Cochrane databases. The search combined the following terms: “hypersexuality”; “nervous system”; “diseases”; “sexual behaviors”; “neurological disease”. All results of each database between 2010 and 2020 were evaluated (Fig. 1). First, we eliminated duplicates and evaluated the remaining articles based on title and abstract. The inclusion criteria were: (i) HS; (ii) neurological pathologies; (iii) publication in peer-reviewed journals; and (iv) English language. In addition, we have excluded: (i) conference proceedings, (ii) reviews, (iii) studies on animal samples and (iv) studies involving patients with “pure” psychiatric disorders. Overall, we identified 103 articles, from which we excluded 35 duplicates (Fig. 1). Subsequently, we identified 26 manuscripts that met the inclusion/exclusion criteria by evaluating the year and type of publication, participants, and purposes of the studies (Table 1, Ref. [6, 8, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34]).
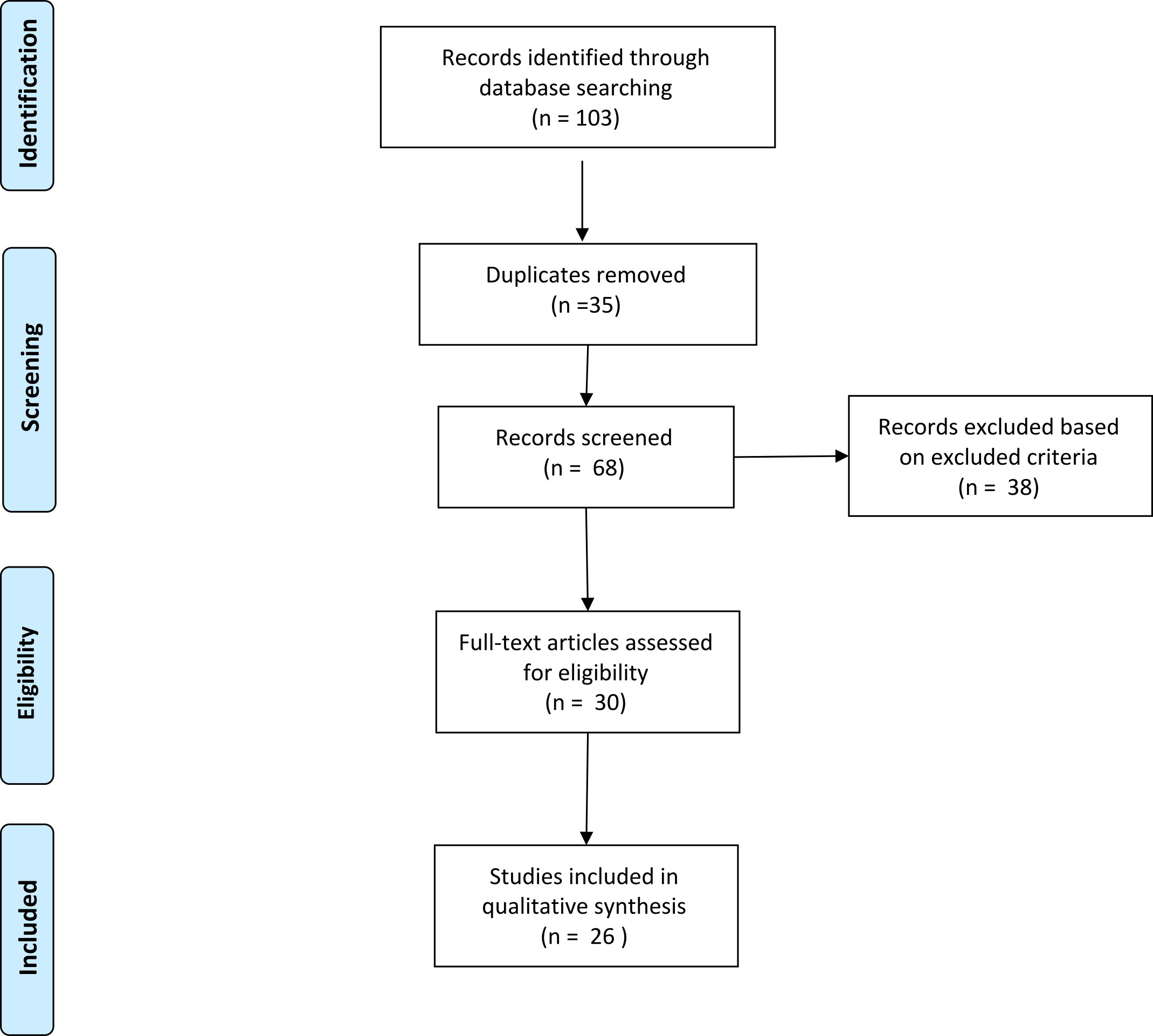 Fig. 1.
Fig. 1.The flow diagram of the screened/included studies.
Changes in sexual desire and behavior are widespread following a brain injury [35, 36, 37, 38] due to the dysregulation of specific networks regarding the dopaminergic circuits, including the mesolimbic and mesocortical ones. The mesolimbic pathway is responsible for reward learning during the mesocortical for executive decision-making. The dysregulation of these circuits leads to the clinical manifestation of impulsive and compulsive behaviors. The mesocorticolimbic dopaminergic network links to key cortical and subcortical regions, especially the prefrontal cortex (PFC), ventral striatum, ventral tegmental area (VTA), and amygdala [39]. Also, the nucleus accumbens (NAc) plays a crucial role in learning reinforcement (Fig. 2). In addition, the mesocortical pathway is essential for executive functions, as the prefrontal cortex projects to the ventral striatum [40]. Impulsive Control Disorder behaviors emerge after exposure to specific rewards, which become compulsive [41]. During the first instance of a reward, an “unexpected” activation of the ventral striatum elicits a strong emotional response and increased ventral striatal dopamine [42]. After this action is repeated, the behavior becomes a “habit” and may be associated with craving [39]. At the first time, this behavior probably localizes at the ventral striatum; later, behaviors are reinforced by the dorsal striatum [43]. Hence, injuries to the frontal and temporal lobe lead to sexual problems, including HS, more frequently than lesions involving the back of the brain [44].
| Study | Study’s design | Patients | Major findings |
| Grandgenevre et al., 2015 [6]. | Observational study | 6 PD patients | The interview administered to the partners helped them to improve verbal communication and, therefore, to be more informative concerning sexual behavior changes in connection with the treatment and its management.The authors have shown that HS in PD is greatly underestimated. |
| Voon et al., 2006 [8]. | Controlled clinical trial | 297 PD patients | The authors showed that the lifetime prevalence of these behaviors was 6.1% and increased to 13.7% in patients on DA. |
| Cilia et al., 2011 [14]. | Observational study | 30 PD patients, 15 healthy controls | In this analysis, gambling severity is associated with dysfunction of the network involving the ventrolateral and ventromedial prefrontal cortex anterior and posterior cingulate cortex, insula, and striatum. PD patients reported a disconnection between the anterior cingulate cortex and striatum. |
| van Eimeren et al., 2010 [15]. | Comparative study | 7 PD patients, 7 controls | The authors observed that DA significantly increased activity in some areas (lateral orbitofrontal cortex, rostral cingulate zone, amygdala, external pallidum) in controls, while PD patients showed a significant DA-induced reduction of activity. |
| Biundo et al., 2011 [16]. | Observational study | 59 PD patients, 22 healthy controls | The results suggest that PD is characterized by an overall loss of gray matter in pre-frontal regions. |
| Bentivoglio et al., 2013 [17]. | Observational study | 17 PD with ICD, 17 PD without ICD | Although this study did not show a significant difference between PD patients with ICDs as compared with patients without ICDs, some preliminary evidence was detected suggesting a trend toward a worse performance of the PD-ICD group on some neuropsychological tasks which are sensitive to the frontal lobe dysfunction and ventral fronto-striatal dysfunction. |
| Voon et al., 2009 [18]. | Clinical trial | 14 PD patients with ICD, 16 PD controls without ICD | The authors demonstrate a pharmacological manipulation of risk taking in a susceptible population of subjects with ICD. Compared to Parkinson’s disease controls, patients with ICD demonstrate an overall bias towards riskier choices associated with lower correlation between risk and neural activity in the orbitofrontal cortex and anterior cingulate. |
| Antonini et al., 2016 [19]. | Meta-analysis | 786 PD patients | In this analysis of 786 PD patients treated with rotigotine, the frequency of ICD was 9.0%, with a specific incidence timeline observed. Once ICD behaviours are present rotigotine dose reduction may be considered. |
| Djamshidian et al., 2012 [20]. | Observational study | 26 PD patients with ICD, 27 PD patients without ICD | The authors found that all patients with PD made more impulsive and irrational choices than the control group. The results also suggest that intact cortical processing and less distractibility in PD patients without ICD may protect them from developing behavioral addictions. |
| Sharma et al., 2015 [21]. | Observational cross-sectional study | 299 PD patients | The authors observed that a high proportion of DA use (81.6%) among our PD patients might be responsible for the development of ICD. The role of genetic factors that might increase the risk of developing ICD in this population needs further exploration. |
| Hibbard et al., 2000 [22]. | Observational study | 322 mild to severe TBI patients, 264 controls | Individuals with TBI reported more physiological, physical and body image difficulties than controls. Age at onset and severity of injury were negatively related to reports of sexual difficulties in individuals with TBI. |
| Gaudet et al., 2001 [23]. | Observational study | 50 mild to moderate TBI patients, 56 controls | Control group has more positive feelings concerning cognition, ego adaptability and sexuality than TBI participants. |
| Sandel et al., 1996 [24]. | Observational study | 52 patients with severe TBI | Significant reduction in orgasm, desire/drive; frontal lobe and right hemisphere lesions were related to higher sexual satisfaction. |
| Crowe et al. 1999 [25]. | Observational study | 14 TBI patients, 14 controls | 59% of the sample indicated decrease in sexual function overall and the TBI group was less able to form images on sexual themes than controls. |
| Kreuter et al., 1998 [26]. | Observational study | 92 patients TBI | Patients report decreased or absent ability to achieve an erection and decreased frequency of: ejaculation, ability to experience an orgasm, sexual desire and intercourse. |
| Ponsford et al., 2003 [27]. | Observational study | 208 moderate to severe TBI patients, 150 controls | The authors observed the changes in sexual behavior, affect, self-esteem, and relationship quality 1–5 years post-injury. It’clear a decreased frequency of engaging in sexual activities, diminished sex drive, lower self-esteem, higher levels of depression, and decreased communication levels compared to controls. |
| Wick et al., 2005 [28]. | Observational study | 435 AD patients | 30% of people with dementia may manifest behavioral disinhibition, including sexual disinhibition. Several causal relationships have been hypothesized, with frontal lobe deterioration, but also lobar lesions or trauma, seizure activity, cerebellar dysfunction, and drugs have been implicated as well. |
| Mendez et al., 2013 [29]. | Comparative study | 58 AD patients, 47 patients with behavioral variant frontotemporal dementia (bvFTD) | HS occurred in 6 (13%) bvFTD patients compared to none of the AD patients. All had general disinhibition, poor impulse control, and actively sought sexual stimulation. They had widened sexual interests and experienced sexual arousal from previously unexciting stimuli. These patients with dementia illustrate varying degrees of increased sexual desire. The authors conclude that bvFTD is uniquely associated with hypersexuality; it is more than just cognitive impairment with frontal disinhibition but also involves alterations in sexual drive, possibly from right anterior temporal- limbic involvement in this disease. |
| Ahmed et al., 2015 [30]. | Observational study | 49 AD patients | BvFTD patients show hyposexual behavior including decreased affection, initiation, and response to advances by partners, and decreased frequency of sexual relations, compared to AD and to SD patients. These symptoms are a feature of bvFTD.The underlying pathophysiology of these changes likely reflects structural and functional changes in frontoinsular and limbic regions including the hypothalamus. |
| Alagiakrishnan et al., 2005 [31]. | Retrospective cross sectional study | 41 AD patients | In this study, sexually inappropriate behavior was seen in all stages of dementia, more commonly associated with subjects of vascular dementia. |
| Baird et al., 2002 [32]. | Observational study | 7 patients after unilateral temporal lobe resection | The results suggest that hypersexuality following unilateral temporal lobe resection is more likely to occur in the presence of psychosocial difficulties and contralateral temporal lobe abnormalities. Neurophysiological processes associated with seizure cessation alone do not appear to account for postoperative hypersexuality. |
| Shukla et al., 1979 [33]. | Comparative study | 70 patients | 70% of temporal lobe epileptics were found to be hyposexual. They had a global loss of performance and interest in the sexual sphere and showed no concern over it. Only one case in the group of temporal lobe epilepsy was hypersexual. |
| Blumer et al., 1970 [34]. | Observational study | 50 temporal lobe epileptics | The author documents the occurrence of hypersexual behavior in 50 temporal lobe epileptics, 42 of whom had a unilateral temporal lobectomy. Twenty-nine (58%) were clearly hyposexual before surgery; seven (14%) had distinct episodes of hypersexuality. In six patients the hypersexual episodes followed the abrupt cessation of temporal lobe seizure activity following either temporal lobectomy or seizures. These findings document the role of the temporal limbic structures in the regulation of sexual arousal. |
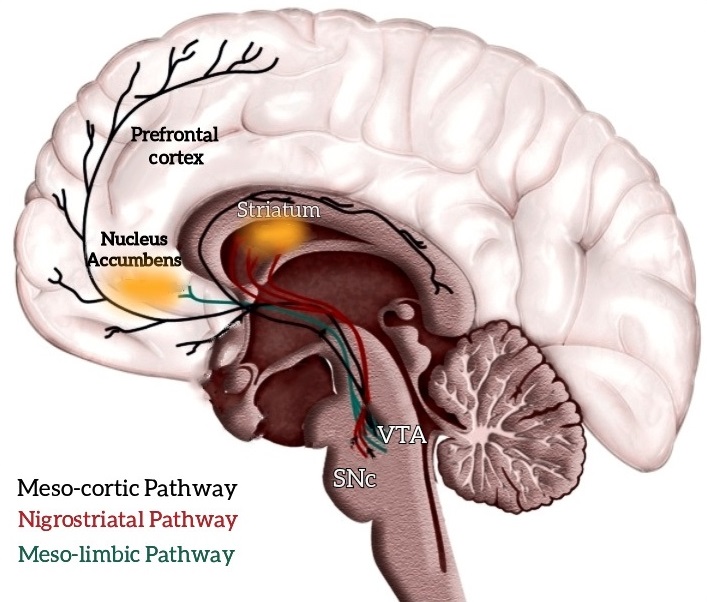 Fig. 2.
Fig. 2.The main neurological pathways and areas involved in hypersexuality. The mesolimbic pathway is responsible for reward learning during the mesocortical for executive decision making. The mesocorticolimbic dopaminergic network connects to key cortical and subcortical regions, most notably the prefrontal cortex (PFC), ventral striatum, ventral tegmental area (VTA), and amygdala. The nucleus accumbens (NAc) plays a crucial role in reinforcing learning. Dysregulation of these circuits leads to the clinical manifestation of impulsive and compulsive behaviors, including HS.
Kleine-Levin syndrome (KLS) is a rare neurological disorder with an unknown etiology [45]. KLS consists of episodes of relapsing/remitting hypersomnia, accompanied by cognitive and behavioral symptoms, such as anxiety, depression or compulsive/impulsive disorder (53% of cases); eating disorders (66% of cases); and HS (53% of cases), alternating with asymptomatic periods [46]. Kleine (1925) and Levine (1936) were the first to describe KLS by identifying a triad of symptoms in some patients, such as hypersomnia, “morbid” hunger and HS [47]. Other studies noted that KLS was distinct from the more common narcolepsy. They found that the triad of symptoms (hypersomnia, hyperphagia, and HS) only occurred in half of KLS patients, so all three symptoms were not needed to make the diagnosis [48]. The disease mainly affects male adolescents (85% of cases), while it is rare to onset before 12 or after 30 [49]. In a systematic review of 186 patients with KLS, Arnulf et al. [50] observed that 43% of subjects (65 participants), primarily males, had HS symptoms during the episodes, especially in the pre-pubescent phase [50]. Another work carried out by the same authors [51] showed that in males, HS symptoms included increased/over masturbation, exhibitionism, obscene language, and sexual behaviors (i.e., the aggression of female subjects, and in rare cases, males). Additionally, the authors demonstrated that 14 patients had normal testosterone levels, luteinizing hormone, and follicle-stimulating hormone [51]. Regarding the therapy of KLS, specific treatments are not available; generally, antipsychotics, mood stabilizers, and stimulants are used, as they also affect HS [47].
Kluver-Bucy Syndrome (KBS) is a neurological disorder caused by bilateral impairment of the temporal lobes, primarily involving the hippocampus and amygdala [52, 53, 54]. In adults, symptoms include hyperorality, eating disorder, visual agnosia, hypermetamorphosis, amnesia, and HS [55]. The prevalence of KBS is poorly understood, as it is described in case series or reports and often linked to other diseases, including stroke [56, 57], traumatic brain injury [58], Alzheimer’s disease [59], and herpes simplex encephalitis [60]. Although KBS is diagnosed by clinical observation [61], there are no specific treatments, but usually, a therapy aimed at managing symptoms (i.e., antidepressant drugs, mood stabilizers, and antipsychotics) is used [62].
Parkinson’s disease (PD) is a neurodegenerative disorder caused by dopamine deficiency in the basal ganglia. It has an incidence that varies according to age: 1–2% of cases in the general population over 60 years, and 3–5% over 85 years [63]. PD usually presents with four typical motor symptoms: bradykinesia, tremor, stiffness, and postural instability. However, non-motor disorders may also emerge due to the nigral system (such as apathy, cognitive impairment, and dysphoria) or extranigral (such as HS, sleep disorders, psychosis and mood disorders) [14, 15, 16, 17, 64]. The etiopathogenesis of non-motor disorders differs according to symptoms. Some symptoms are directly related to PD (such as mood disorders), others are due to iatrogenic effects, such as psychosis and impulse control disorders, including HS. Indeed, in a case-control study, Voon et al. [18] showed that 7.2% of PD patients treated with dopaminergic therapy were affected by HS. Furthermore, a meta-analysis of 786 PD patients treated with rotigotine has shown a prevalence of 9% of HS cases [19]. The close relationship between impulse control disorders (including HS) and the use of dopamine receptor agonists was also demonstrated by a retrospective study [65]. The authors found an association between HS, male sex and young-onset of PD. Other authors have shown that in the etiology of some behavioral disorders in PD, including HS, multiple intrinsic and extrinsic risk factors must be considered, including drug treatment, cognitive and psychiatric symptoms and personality traits [20, 21]. In any case, the relationship between PD and HS remains unclear, so new studies are needed to understand their pathophysiology and identify new, safer, and more effective treatments [64].
Traumatic Brain Injury (TBI) can cause multiple cognitive, physiological, and psychosocial changes in patients, negatively impacting the patient’s life, and sexual functioning [22, 44]. In particular, HS has been found to have a significantly higher incidence in individuals with TBI than in the general population [23, 66]. Patients with TBI complain of HS disorders in 36–54% of cases. The most common symptoms are: excessive increase or decrease in sexual desire/impulse or activity, difficulty reaching orgasm, inappropriate sexual behavior, other secondary symptoms to the sexual dysfunction, such as irritability, dysphoria or disinhibition [24, 25, 26, 67, 68, 69, 70]. Sexual symptoms manifest based on the brain area involved in TBI; in particular, damage at the anterior brain regions (hypothalamus-pituitary axis) causes desire disorders and male erectile dysfunction, which are the prevalent sexual symptoms following TBI. However, following TBI, lesions generate more frequent hyposexuality rather than HS. On the other hand, damage to the hypothalamus, amygdala, temporal and frontal lobes can elicit HS, such as inappropriate attention to others (including masturbation or obscene language), sexual exhibitionism and sexually deviant criminal activities (rape and pedophilia). In particular, specific injuries to the frontal lobes can lead to HS without other typical symptoms, such as loss of initiative or intellectual disturbances [44, 71, 72].
Sander et al. [73, 74] found sexual problems,
including HS, in around 20% of patients with TBI after 1 year of injury. The
authors identified injury severity, demographic characteristics, motor and
cognitive independence, emotional functioning and social participation as
predictors of sexual abnormalities. These results are confirmed by Ponsford’s
study [27], which highlights other predictive factors for sexual symptoms and HS,
including lower self-esteem, depressive symptoms, reduced autonomy, older age,
psychological and interpersonal factors, especially in men. Althof [75] grouped these latter factors into 4 categories: insufficient stimulation, high
frequency of masturbation, inner conflict and desire disorder masked by
ejaculatory dysfunction. Furthermore, Goldin et al. [76] found a
correlation between fatigue and sexual desire/activity disorders. In particular,
women with TBI were less interested in sex and reported less importance of sex
than men, who had higher rates of HS. Finally, the work and economic situation carry a high relevance in sexual disorders [77].
Adults with higher annual incomes (
Dementia is a significant cognitive deterioration that interferes with autonomy in daily functioning and includes various symptoms, such as memory deficits and abnormalities in other cognitive domains (communication, language, attention, visual perception, learning, verbal fluency). The most common form of dementia is Alzheimer’s Disease (70% of cases) [79]. Besides cognitive symptoms, patients with dementia often present with behavioral and psychological dysfunctions, such as anxiety, aggressivity, mood disorders, illusions, sleep disorders, sexual symptoms, such as HS. These symptoms have been classified in different clusters [80]: the psychosis cluster (delusion and hallucination); the emotional cluster (anxiety, depression, eating and sleeping, apathy), and the hyperactive cluster (aggressive and criminal behavior, disinhibition, aberrant motor behavior, abnormal eating behavior, and HS) [80, 81, 82, 83]. During the disease course, 90% of patients showed one or more behavioral and psychological dysfunctions [84], increasing the caregivers’ emotional and physical burden and the whole family system [85].
Among the various sexual disorders, HS is the most frequent in patients with dementia. However, sexuality is still taboo in this population as it is believed that older people are not interested in sexual activities. This leads to a misinterpretation of some normal sexual behaviors considered pathological, especially among older adults with cognitive decline living in nursing homes [86]. The prevalence of HS in patients with dementia varies between 2% and 30%, in both men and women, as demonstrated by Burns et al. [87]. Lindau et al. [88] noted that in a large sample of older people, 73% of people aged between 57 and 64 and 53% of people between 65 and 74 reported being sexually active. Healthcare workers are reluctant to talk about sexuality with older patients, and, as a result, patients are reluctant to express sexual problems so that HS prevalence might be underestimated [89]. Burns et al. [87] found that sexual disinhibition has the exact incidence in men and women, with men exhibiting sexual behaviors mainly physically, while women in a verbal mode. Alterations of the frontal and temporal lobe lead to HS, given that the frontal lobe hinders the inhibition mechanism of sexual behavior. In contrast, temporal lesions compromise intellectual understanding and the decision making of sexual desire [28, 62, 90, 91, 92]. In addition, frontotemporal dementia is the pathology in which the highest percentage of HS is found, compared to other forms of dementia [29]. What is more, in frontotemporal dementia there is also a high incidence of hyposexual behaviors, such as reduced affection, poor initiation and response to the partner and decreased frequency of sexual intercourse, compared to Alzheimer’s and semantic dementia [30]. In contrast, in a retrospective cross-sectional study, Alagiaskrishnan et al. [31] showed a higher rate of sexually inappropriate symptoms in patients with vascular dementia involving only a small sample.
Healthcare professionals working with demented patients should be aware of sexual dysfunctions and inappropriate behaviors in this aged population to better manage this complex neurodegenerative disease.
Temporal Lobe Epilepsy (TLE) is a chronic neurological disease characterized by diurnal and/or nocturnal epileptic seizures that can be silent, partial or secondarily generalized with tonic-clonic seizures. Symptoms depend on the specific regions of the temporal lobe affected by the epileptic waves and the type and frequency of the epileptic seizures. In addition, TLE is associated with changes in sexual behavior, such as interictal hyposexuality or HS. The former consists of reducing sexual activity, with possible erectile dysfunction or anorgasmia [32, 33], whereas the latter causes sexual impulses accompanied by orgasm and genital sensations. In addition, some authors have highlighted other sexual symptoms in TLE, such as exhibitionism, masturbation, and sexual automatism [93]. Furthermore, it has been observed that the surgical removal of the epileptogenic temporal lobe can cause changes in sexual behavior [94]. In particular, after lobectomy, between 28–81% of patients have noted an improvement in sex drive and periods of HS [95]. Blumer et al. [34, 96] have proposed three mechanisms to account for postoperative changes in sexual behavior regarding hypersexuality after temporal lobectomy.
Firstly, before surgery, the constant epileptic discharges from the limbic system inhibit normal function, leading to preoperative hyposexuality. Neurophysiological changes following surgery release the inhibitory effects, producing hypersexuality. This statement derives from a previous work evaluating the preoperative and postoperative sexual behavior of 21 patients with TLE who underwent unilateral temporal lobectomy for epilepsy. More than half of these patients were hyposexual before surgery. If surgery was successful, there was a marked improvement in sexual arousal and responses. However, in two patients, postoperative hypersexuality terminated with recurrence of seizures. According to the authors, an increase in neural activity of the limbic areas of the temporal lobes produces a suppression of sexual behavior. In contrast, the presence of hypoactivity of the same areas can cause sexual arousal and reactivity [34, 96].
Secondly, HS can be considered a Kluver-Bucy feature. In a case report, Anson and Kuhlman [97] described a patient with Kluver-Bucy syndrome in the post-stroke phase. The authors noted that the patient’s syndrome occurred transiently due to bilateral temporal lobectomy. In addition, it has been observed that the change in sexual behavior can be associated with TLE and lobectomy. In particular, hyposexual patients following even unilateral lobectomy of the temporal lobe tend to experience an increased libido and HS. However, aberrant sexual behavior usually occurs due to bilateral injury, rarely due to unilateral injury. For example, Blumer et al. [96] observed only 2 cases out of 42 patients. In addition, another case found that it is doubtful whether the patient could have transient Kluver-Bucy syndrome after a unilateral temporal lobectomy, with chronic behavioral syndrome not related to the post-stroke period. Finally, other authors have reported sexual alterations due to a stroke involving the temporal lobe and unrelated to surgery intervention, possibly caused by transient bilateral temporal lobe dysfunction following seizures.
Thirdly, HS can be a behavioral feature of the postoperative period. Wilson et al. [98] observed that the post-operative phase could be complex and turbulent after temporal lobectomy. In particular, psychosocial difficulties associated with epilepsy may persist, or new difficulties and sexual symptoms may arise due to stress in returning to daily life. These new behavioral difficulties can manifest in different stages of post-operative recovery. In particular, the authors highlighted that behavioral symptoms, such as HS, became evident following the reintegration of patients into their life contexts, with part-time or full-time activities, as patients felt the weight of their own and others’ expectations in their daily performance. In any case, the nature of sexual changes following temporal lobectomy is poorly documented.
In his survey, Wilson described seven cases of TLE with striking postoperative sexual changes. Postoperatively, a clear correlation between the presence of seizure activity and hyposexuality became apparent. The absence of sexual response persisted in three patients despite the improvement in their seizure condition. Instead, four patients who presented reduced or absent seizure conditions had intense sexual responsiveness and improved sexual desire. In two patients, postoperative HS ended with seizure recurrence [98].
In summary, sexual changes appear to be related to the medial temporal lobes’ activity, due to the presence or absence of intrinsic seizure discharges which confirms the importance of these areas for regulating sexual functions [93, 98].
Neurological patients can experience multiple physiological, cognitive, and psychosocial changes, which negatively affect various areas of their daily functioning, including sexual activity [99]. Sexuality is a fundamental topic of a person’s life affecting interpersonal relationships, psychological well-being, and quality of life. Neurobehavioral symptoms concerning HS are widespread in patients with neurological disorders, and such symptoms can lead to high levels of distress to caregivers and patients [66]. Data from our review demonstrates that HS is a frequent sexual symptom in patients with neurological disorders, especially in neurodegenerative ones. We have found that HS manifests in multiple forms from the data available, such as increased libido, masturbation in public, sexual harassment, or changes in sexual orientation. Then, HS in the neurological population is mainly expressed in verbal and/or physical acts with a sexual meaning and intention [100].
Moreover, our review pointed out that this behavior could negatively affect a patient’s management and outcomes. Consequently, in our opinion, it is necessary to correctly assess HS by using the proper diagnostic tools when dealing with patients affected by neurological disorders. Indeed, this review revealed that studies are often incomplete for either assessment measures or sample selection. Furthermore, in most studies, the psychometric properties of the instruments had not been verified, resulting in difficulties in obtaining reliable results. In fact, the research is based on the patient’s communication of symptoms and/or questions implemented by the clinician, without following standardized scales, such as the Hypersexual Behavior Inventory [101] or the Hypersexual Behavior Consequences Scale [102].
We believe that an accurate diagnostic work-out is fundamental in the presence of HS. Therefore, every patient with symptoms indicating HS should be submitted to a detailed general, psychiatric, sexual and medical history, and general and neurological examinations with neuroimaging. In addition, an individual’s experiences, roles, and sexual attitudes, as well as substance abuse and other compulsive behaviors, should be pointed out. However, this counseling requires high soft skills since the embarrassment or shame of the patient and the therapist can create communication difficulties that do not allow the fine collection of important information; therefore, healthcare professionals need to be adequately trained to consider sexual functioning [13]. Furthermore, the diagnostic process should allow integration between the information and other salient aspects of the patient, such as his/her personality, relational capacity and past and/or present illness. Finally, based on the diagnostic information and the results obtained from the standardized psychosexual tools, the objectives of the treatment must be defined.
More accurate sample selection modalities are required to obtain valid data concerning the sampling bias in HS research. In particular, we had difficulties finding out/reviewing studies on specific diseases, such as Kleine Levin syndrome and TLE, in which only clinical case series are present. This could be due to the lack of a proper diagnosis of the neurological disorder or the overlooked compulsive symptom. Moreover, no studies have been published on other frequent neurological diseases, such as stroke and multiple sclerosis. As both the diseases may disrupt the neural pathways involved in sexual behavior, prevalence of HS may be underestimated in this patient population.
It is noteworthy that the sample in the different studies is mainly composed of males (with a few females), and this aspect should be better investigated for clarity. This could be due to a higher incidence of the disorder in males. As observed in some studies [10, 11, 12], or preconceptions, like sexuality, especially in females, is still considered a taboo and often poorly investigated. In fact, in some studies (mainly focusing on dementia), the manifestation of HS seems similar in both genders [87, 88, 89]. Hence, prospective studies with larger samples, which also consider gender differences and standardized measures, are needed to make the results reliable and generalizable for the neurological population.
Finally, since HS is associated with the activity of the mesocorticolimbic pathway, it would be interesting to investigate the manifestation of the disorder in diseases directly related to this function of the circuit, such as schizophrenia and bipolar disorder and other significant psychiatric disorders. Indeed, psychiatric disorders have been excluded from the revision as we decided to focus on neurological pathologies.
Besides diagnosis, another hot topic is the treatment of HS in such patients. To control HS, the treatment aims to improve sexual self-control, reduce problematic sexual behavior and adverse consequences, such as damage to oneself or others, and reduce anxiety and impairment of personal health. Currently, data indicate that a combination of pharmacological and non-pharmacological treatment is the best approach to manage HS. In patients affected by neurological disorders, as the pathways underpinning sexual behavior are abnormal (especially in neurodegenerative diseases), the use of specific and selective drugs should be recommended. In particular, like other impulsive/compulsive disorders with an organic cause, the mainstay pharmacological treatments include selective serotonin reuptake inhibitors (concerning citalopram, up to 60 mg/daily) and or dopamine-agonists. However, specific guidelines in HS due to neurological illness are still lacking. Thus, further research is needed to identify adequate treatments to improve patients’ management and reduce their caregivers’ burden.
HS, Hypersexuality; ICD-11, World Health Organization’s International Classification of Diseases and Related Health Problems; KBS, Kluver-Bucy Syndrome; KLS, Kleine-Levin syndrome; NAc, the nucleus accumbens; PD, Parkinson’s disease; PFC, prefrontal cortex; TBI, Traumatic Brain Injury; TLE, Temporal Lobe Epilepsy; VTA, ventral tegmental area.
These should be presented as follows: DL, MGM, AM, RSC and DM designed the research study. DL, MGM, DM, AA performed the research. DL, MGM, AA, AM and RC wrote the manuscript. All authors contributed to editorial changes in the manuscript. Finally, all authors read and approved the final manuscript.
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The Institutional Review Board of IRCCS Centro Neurolesi Bonino Pulejo (Messina, Italy) approved the study.
We thank two/three anonymous reviewers for their excellent criticism of the article.
This study received no external funding.
Given his role as the Editorial Board Member of JIN, Prof. Rocco Salvatore Calabrò had no involvement in the peer-review of this article and has no access to information regarding its peer-review.


