1 Department of Biomedical and Neuromotor Sciences, University of Bologna, 40126 Bologna, Italy
Abstract
The superior parietal lobule of the macaque monkey occupies the postero-medial part of the parietal lobe and plays a crucial role in the integration of different sources of information (from visual, motor and somatosensory brain regions) for the purpose of high-level cognitive functions, as perception for action. This region encompasses the intraparietal sulcus and the parieto-occipital sulcus and includes also the precuneate cortex in the mesial surface of the hemisphere. It hosts several areas extensively studied in the macaque: PE, PEip, PEci anteriorly and PEc, MIP, PGm and V6A posteriorly. Recently studies based on functional MRI have suggested putative human homologue of some of the areas of the macaque superior parietal lobule. Here we review the anatomical subdivision, the cortico-cortical and thalamo-cortical connections of the macaque superior parietal lobule compared with their functional properties and the homology with human organization in physiological and lesioned situations. The knowledge of this part of the macaque brain could help in understanding pathological conditions that in humans affect the normal behaviour of arm-reaching actions and can inspire brain computer interfaces performing in more accurate ways the sensorimotor transformations needed to interact with the surrounding environment.
Keywords
- Sensory information
- Multisensory
- Memory
- Attention
- Cerebral cortex
- Neural networks
- Connectivity
The associative areas of the cerebral cortex are involved in “cognitive functions”, and this is known since long time, as reported in didactic books of neuroscience [1]. Cognitive functions are mental processes that enable us to perform a variety of tasks. The parietal cortex contributes to active roles in the processes of receiving, choosing, transforming, storing, processing and retrieving information, necessary to interact with the world around us. The high complexity of these tasks implies that the associative areas receive and integrate information from multiple sources of the cortical and subcortical structures of the brain.
The majority of the knowledge of the cerebral processes we manage derives from studies performed in the non-human primates. The superior parietal lobule (SPL) of the macaque monkey is an associative cortex and belongs to the parietal neuronal network that associates information coming from motor and premotor, visual and somatosensory cortices. This information is involved in aspects of attention and visuospatial perception, and is found to be at the base of planning and controlling the execution of reaching and grasping movements [2, 3, 4, 5]. The SPL region encompasses the intraparietal sulcus and the parieto-occipital sulcus, as caudal part of the parietal lobe in both humans and non-human primates and includes the precuneate cortex in the mesial surface of the hemisphere. In the macaque, it hosts several areas, cyto- and myelo-architectonically defined, that are PE, PEip, PEci anteriorly and PEc, MIP, PGm and V6A posteriorly (Fig. 1A). In human, cytoarchitectonic investigations recently subdivided the SPL in a mosaic of seven distinct areas 5L, 5M and 5Ci anteriorly, 7PC, 7A, 7P and 7M posteriorly (Fig. 1B). In addition, functional MRI studies have been shown that for some of the macaque cortical areas (V6A, PEc and PGm) a perfect parallel functional organization exists in the human SPL [2, 6, 7, 8, 9, 10, 11, 12].
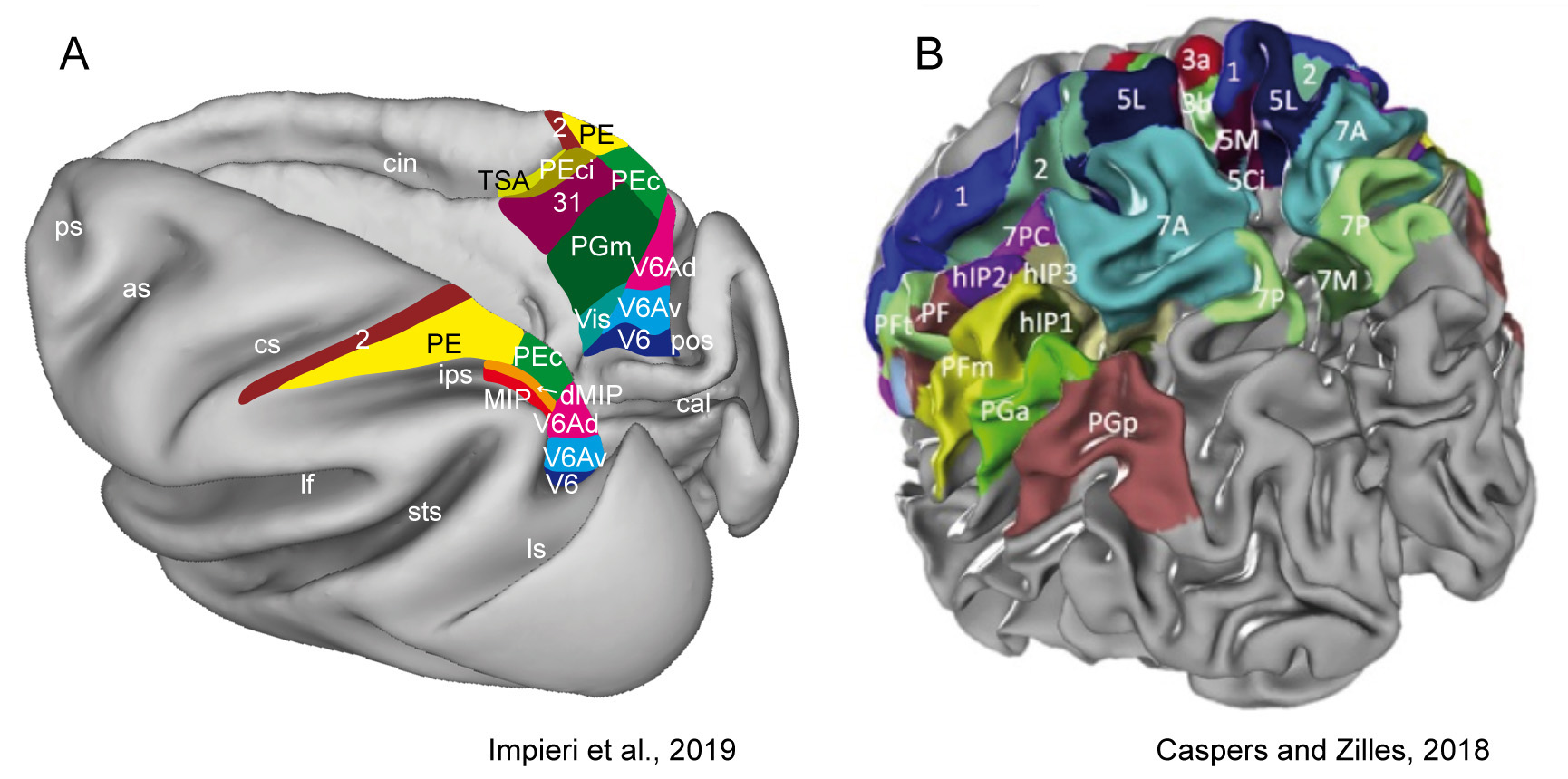 Fig. 1.
Fig. 1.Superior parietal lobule areas.
(A): Macaca fascicularis brain, reconstructed using Caret software
(http://brainvis.wustl.edu/wiki/index.php/Caret:
Download). On the left the dorsal view of left hemisphere and on the right the
medial view of right hemisphere showing in different colours the location and
extent of SPL areas. Adapted from [13].
(B): Human brain showing a modern parcellation of the parietal cortex based on
the JuBrain Cytoarchitectonic Atlas (https://julich-brain-atlas.de/). Left dorsocaudal view of
both hemispheres showing in different colours the location and extent of SPL
areas. Reprinted with permission from [6] published in Elsevier.
as, arcuate sulcus; cal, calcarine sulcus; cin, cingulate sulcus; cs, central
sulcus; ips, intraparietal sulcus; lf, lateral fissure; ls, lunate sulcus; pos,
parieto-occipital sulcus; ps, principal sulcus; sts, superior temporal sulcus. D,
dorsal; P, posterior.
The SPL of both humans and non-human primates is then an intricate system whose areas show anatomical and functional properties that underlie different functional roles. We here review the anatomical subdivision, the cortico-cortical and the thalamo-cortical connection and the functional characteristic of the macaque SPL in order to examine how it processes distinct sensory information to guide actions. State-of-the-art studies of the human SPL organization will be discussed by emphasizing the similarities to structure and circuits of the non-human primates directly accessible through experimental investigation. The knowledge of the areas of SPL in the macaque brain could help to understand pathological conditions that in humans affect the normal behaviour of arm-reaching actions. Based on the proposed similarities, we finally indicate new perspectives for the translational approaches in the field of neural prostheses, which allow brain computer interfaces to perform the sensorimotor transformations needed to interact more accurately with the surrounding environment.
The study of cortical and subcortical connections is crucial to describe the neuronal networks in which SPL areas are involved, and their functional role. The following sections summarize current knowledge on the connectivity of this brain region with other cortical and subcortical structures to provide a short survey of the principal functional features of the macaque SPL areas.
The caudalmost part of the SPL, on the anterior bank of the parieto-occipital sulcus, hosts area V6A [14, 15, 16] that is characterized by a cytoarchitectonic pattern typical of the parietal areas [17] and has been divided in ventral (V6Av) and dorsal (V6Ad) subsectors [18]. V6A occupies about two thirds of the anterior bank of the POs and extends to the caudalmost part of the precuneate cortex on the mesial surface of the hemisphere [15]. Overall, the extrastriate areas of the occipital lobe (V2, V3, V4, MST, V6) and the visual and visuomotor areas of the posterior parietal cortex (e.g. AIP and LIP) are the source of visual information to V6A. V6A receives also somatosensory and somatomotor inputs from PEc and MIP [19, 20] and is connected reciprocally to the dorsal premotor area F7 and with the sector of premotor area F2, that represents the upper limb. This is in line with the reaching/grasping activity and the primarily upper limb representation in V6A [21, 22, 23]. Specifically, V6Av has connections mostly with extrastriate visual areas while V6Ad with parietal and frontal areas (Fig. 2A top) [18, 24]. The flow of visual information from V6Av to V6Ad can be useful to control the movements of the upper limbs [18].
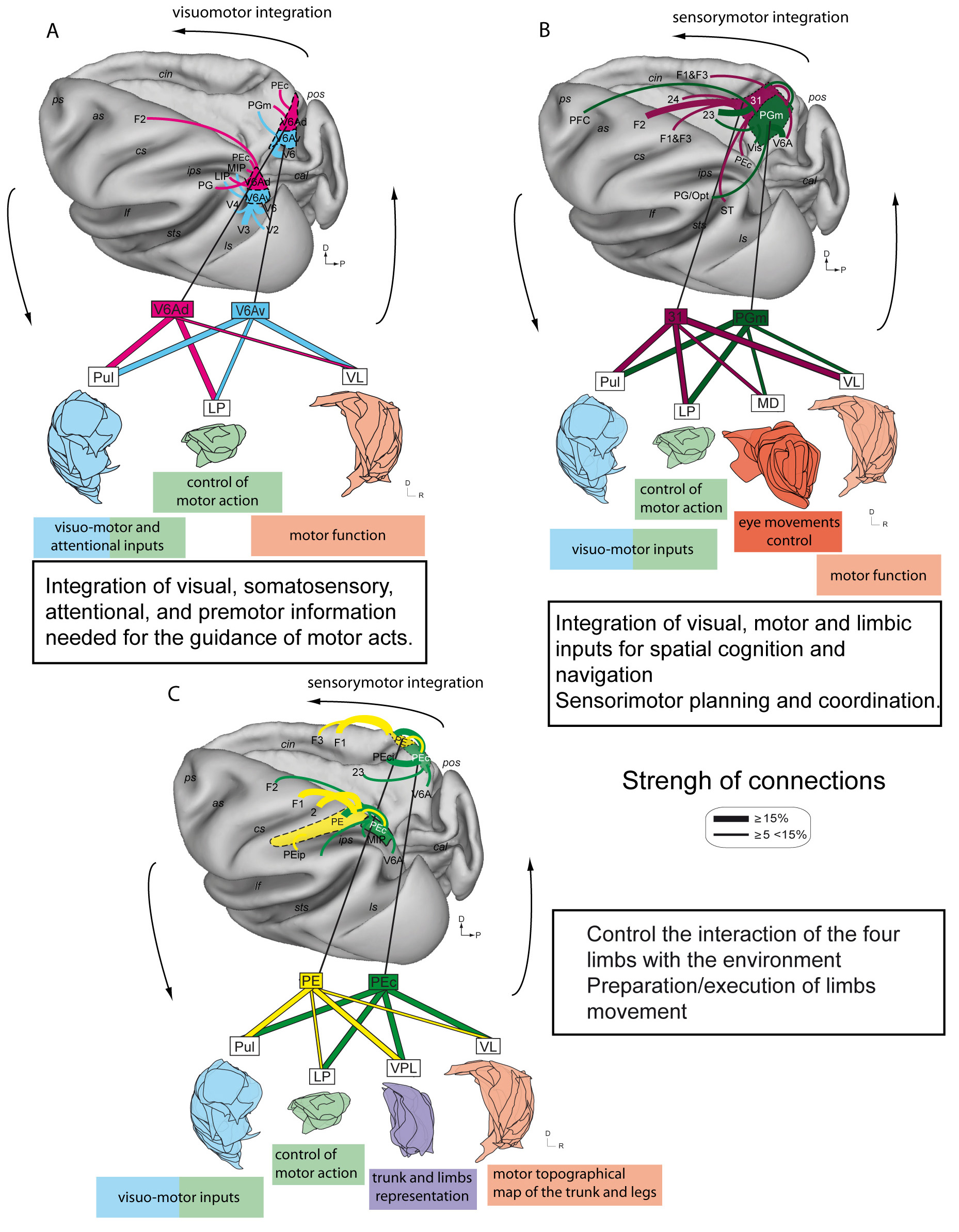 Fig. 2.
Fig. 2.Cortico-cortical and Thalamo-cortical circuits
involving SPL areas.
Representation of the main cortico-cortical and thalamo-cortical connections of
areas V6Av, V6Ad, PE, PEc, PGm, 31, shown in pairs.
(A) Top: Cortical connections of areas V6Av and V6Ad, Bottom: Reconstruction of
the thalamic nuclei that send afferences to areas V6Av and V6Ad obtained by
superimposing all sections scanned. For each area and thalamic nucleus only
projections encompassing more than 5% of the total connections of that area or
nucleus are represented. The thickness of connecting arrows and lines is
proportional to the strength of connections, averaged across cases with
injections in the same area (middle right) (data taken from [12, 18, 24, 43]). The
thalamic nuclei are highlighted with different colors: blue for Pul, green for
LP, pink for VL, purple for VPL and red for MD. Area V6A is shown in the two
cyto-sectors, ventral and dorsal (V6Av and V6Ad) to appreciate the differences in
connectional properties, even though, currently it is considered that the two
cyto-sectors of area V6A work as a single functional entity in the control of
reach-to-grasp movements, V6Av mainly managing the visual aspect and V6Ad the
somato-motor aspect of this control [44].
Abbreviations as in Fig. 1.
(B): Cortico-cortical connections and thalamo-cortical connectivity of areas PGm
and 31 are shown, same as in A (data taken from [28, 45]).
(C): Cortico-cortical connections and thalamo-cortical connectivity of areas PE
and PEc are shown, same as in A (data taken from [19, 42, 46]).
The precuneate cortex hosts also the mesial sectors of area V6A and PEc, as well as areas PGm and 31 [25, 26, 27, 28]. The majority of inputs to PGm and 31 come from V6A and from the dorsal portion of the posterior cingulate area 23 (23e), but some differences are present. Visuomotor areas are involved in the spatial cognition (including the lateral intraparietal cortex, inferior parietal lobule, and the putative visual areas in the ventral part of the precuneus) and are strongly connected with area PGm, whereas premotor and parietal sensorimotor areas are connected with area 31. Area 31 receives additional inputs from the anterior cingulate cortex (area 24), and PGm from retrosplenial areas, located in the mesial surface of the brain (Fig. 2B top). These findings suggest for PGm a crucial role in visuospatial cognition and navigation, and for area 31 in sensorimotor planning and coordination [28].
In addition to the above-mentioned areas, the mesial surface of the SPL hosts area PEci or SSA (Supplementary Somatosensory Area) hidden into the depth of the caudal tip of the cingulate sulcus [27, 29, 30]. Cortical afferences to area PEci come from frontal (areas 4, 6, 8, F2, and 46), sensory (areas 1, 2, and 3), superior parietal (areas PE, PEa, PEc, PO), inferior parietal (areas SII, paAc, PGop, PG, Tpt, and MST), insular, and medial (areas PGm, 31, TSA, 23a/b, 24a/b, F3 and F4) cortices [27]. Interestingly, PEci has many connectional similarities with PE, but not with PEc. Like PE, PEci receives afferents from motor and premotor cortical regions and does not receive visual inputs, while PEc is connected with somatomotor and visual areas [19, 31].
Moving laterally on the SPL, PEip rostrally and MIP caudally [32, 33, 34, 35] occupy the medial wall of intraparietal sulcus in the lateral portion of SPL. PEip shows connections with anterior parietal, primary motor, and ventral premotor areas, and seems to be part of the parietal field that contains large representations of the distal forelimb [36, 37]. Several studies on grasping objects [38, 39], also in the case of either movements highly stereotyped or in the absence of visual information [40, 41], were carried out precisely in the rostral parts of the posterior parietal cortex, near both banks of the intraparietal sulcus. MIP receives denser input from somatosensory areas, the primary motor cortex, and the medial motor fields, as well as from visual cortex in the ventral precuneate cortex and frontal regions associated with oculomotor guidance. These connections suggest that MIP is involved in sensorimotor behaviour, being directly involved in movement planning and execution [20].
Finally in the cortical convexity of the SPL, the areas PE rostrally and PEc caudally [25] have been described, and both extend also medially and laterally into the mesial surface of the hemisphere and the medial wall of the intraparietal sulcus, respectively. Areas PEc and PE share several cortical connections (Fig. 2C top). The cortical inputs to area PE come from regions of parietal and frontal cortices with somatosensory and motor characteristics, which support the proposed functional role for this area: to plan and guide reaching movements [42]. Area PEc is mainly connected with somatosensory and somato-motor areas of parietal and frontal lobes, and receives visual information from the visuo-motor area V6A, and in particular from its dorsal subdivision [19, 34]. This connectional pattern supports the involvement of area PEc in the process of locomotion and in coordination of movements in natural environments [19].
We provide hereafter an overview of the thalamo-cortical connections that have been recently described in detail (Fig. 2) [43, 45, 46] to gain insight and complete the neural circuitry of SPL areas and relative functional role.
Studies on the thalamo-cortical connections in macaques [47, 48, 49] and New World owl and squirrel monkeys [50] have demonstrated that posterior parietal areas are mainly connected with the lateral posterior (LP), ventral lateral (VL), and medial pulvinar (PuM) thalamic nuclei [51].
Recent studies [43] demonstrated that the major inputs of thalamic projections to area V6A as a whole come from the lateral posterior and medial pulvinar nuclei, whereas minor inputs come from the ventro-lateral nucleus (Fig. 2A bottom). The functional characteristics of area V6A [43] well agree with the role of the visuo-motor integration of lateral posterior and medial pulvinar nuclei [51], as well as the motor functions of the ventral lateral nucleus [52], and the eye-related activity of medial dorsal nucleus [53]. The consistency in the talamo-cortical pattern supports the view of an integrative role of visual, somatosensory, attentional, and premotor information needed for the guidance of motor acts [43].
Thalamic inputs to areas PEc and PE mostly originate from lateral posterior, medial pulvinar, ventral posterior lateral (VPL), and ventral lateral nuclei, all structures involved in visual, somatosensory, and/or sensorimotor processing (Fig. 2C bottom). VPL and VL nuclei, characterized by a precise somatotopy, show labelled neurons in regions likely to correspond to the trunk and limb representations (in particular the legs) [46]. The sensory-motor integrative nature of these posterior parietal areas PEc and PE is supported by the thalamic inputs above described [54, 55, 56, 57, 58, 59, 60], with the former area involved in the control of four limbs interaction with the environment [19], and the latter one involved in the preparation/execution of limb movement [55, 60, 61, 62, 63, 64, 65]. The corticothalamo-cortical circuits including PEc and PE also suggest playing a role in supporting some degree of motor automatism, crucial in locomotion.
The medial and lateral subdivisions of the pulvinar linked to association and visual function, transmit strong inputs to area PGm, whereas area 31 receives afferents from the oral division of the pulvinar (Fig. 2B bottom). Both areas also receive input from subdivisions of the lateral thalamus linked to motor function (VL and VA nuclei), and only PGm receives important limbic association afferents, mainly via the lateral dorsal nucleus. Therefore, PGm integrates information from visual association, motor and limbic regions of the thalamus by giving support at the hypothesized role in spatial cognition, including navigation [28], and in spatial learning and memory [66]. By comparison, dorsal precuneate area 31 is more involved in sensorimotor functions, being similar to adjacent areas of the dorsal parietal cortex [45]. To date, the talamo-cortical connections of the lateral part of the SPL are poorly understood.
The pulvinar complex and the lateral posterior nucleus constitute the numerically predominant projections of the thalamus to the parietal lobe, as described in recent and former studies [43, 45, 46, 48, 67], and thalamic connections likely reflect parietal specializations in cognition and in the planning and control of motor actions [2, 68, 69, 70, 71, 72]. However, they are often considered as a unique functional unit as the failure to establish a clear border between the LP and the pulvinar nuclei [73, 74, 75]. The associative nucleus LP provides information on somatic and attentional stimuli useful in the control of movements [43, 76, 77]. The medial pulvinar is a multimodal associative nucleus: it has extensive connectivity with high-order cortical systems (but not with early sensory areas) [78], it forms the subcortical component of the attentional network of the brain [79, 80] and is involved in visuospatial processing and reward-based decision making. These connectional data encourage the hypothesis that the dorsal and caudomedial portion of SPL are primarily involved in the guidance of sensory-spatial behavior [45].
The knowledge of the connectivity above summarized, indicate that areas PE, PEip, and PEci are somatomotor areas non affected by visual signals. Conversely PEc, MIP, PGm, and V6A areas are influenced by both somatomotor and visuomotor signals. All these areas of the SPL form two pathways heavily interconnected and working in opposite direction; the visual pathway moves from the posterior to the anterior areas, while the somatosensory runs contrariwise, with an overlap in the intermediate areas, typically the sensory-motor areas. These two interconnected flows of visual and somatosensory information with an overlap in sensory-motor areas, are part of neuronal networks functional to program and control the execution of reaching and grasping movements [2, 3, 12] .
The proposed role of SPL areas based on cortical and subcortical connections is supported by many functional studies. In the last decades these functional studies have demonstrated that SPL is not only a structure deputed to the integration of diverse types of sensory information [54, 81, 82], particularly somatic and visual, but it also plays a role in the control of somatomotor, visuomotor, oculomotor activities, as well as in the guidance of visuospatial attention [3, 64, 83, 84, 85, 86, 87, 88, 89]. Neurons in the SPL are important in the perception and relation of the body to the external space during reaching, pointing, and grasping. In fact, arm movements performed by monkeys to reach and grasp objects in the peri-personal space, activate SPL neurons. Therefore, the areas of the SPL are implicated in processes related to arm movement planning, execution, and online control [2, 3, 21, 22, 23, 62, 83, 90, 91, 92, 93, 94, 95, 96, 97].
Recent structural and functional studies have evidenced a correspondence between the monkey and the human parietal cortex by proposing putative human homologues of some of the areas we have described above.
Under the structural point of view, the SPL in human is bounded anteriorly by the postcentral sulcus (PoCS), posteriorly by the lateral extension of the parieto-occipital sulcus, and laterally by the intraparietal sulcus, and involves the precuneus (PrC) located medially, but caudal of the posterior cingulate sulcus (CiS). Cytoarchitectonic analysis [7], probabilistic mapping of cytoarchitectonic areas [8] along with the investigation of the distribution of the receptors of the main neurotransmitter families [98, 99] have been recently used to characterize SPL, allowing to supersede Brodmann’s [100] original subdivision into areas 5 and 7. Brodmann area 5 (BA5) has been further subdivided in two mesial areas, 5Ci (cingulate parietal) and 5M (medial parietal), and one lateral area (5L) located in the rostral bank of the post-central sulcus. Area 5L and 5M would be equivalent of macaque’s area PE, while 5Ci would correspond to macaque area PEci [98, 99]. BA7 has been split into area 7PC, located in the posterior wall of the post-central sulcus, 7A and 7P in the anterior and posterior part of dorsal SPL, respectively, and 7M in the medial wall [98, 99] (Fig. 1B). The macaque equivalent of these areas would be areas MIP and/or PEip, PEc, V6A and PGm respectively [12, 89].
The onset and development of in vivo diffusion-weighted magnetic resonance imaging and fiber tracking algorithms have considerably advanced the structural connectivity in human brains. Diffusion tensor tractography (DTT) based on the diffusion tensor-MRI, locate and image the course of the neuronal fiber tract and its trajectory. The superior longitudinal fasciculus (SLF) is a large bundle of association fibers in the white matter of each cerebral hemisphere connecting the parietal, occipital and temporal lobes with ipsilateral frontal cortices, subdivided in five subcomponents [101]. The SLF eases the formation of a bidirectional neural network essential for core processes such as attention, memory, emotions and language [102, 103]. Studies in non-human primates have been largely used to trace long fiber pathways within the fronto-temporo-parietal region [104, 105, 106, 107, 108, 109, 110, 111, 112]. It has been demonstrated that connectivity of the SPL, medial SPL and precuneus in particular, follows primarily the course of the SLF I [6]. The SLF I originates in the superior and medial parietal cortex, that is located near the superior-lateral cingulum bundle on the coronal planes in each cerebral hemisphere. The SLF I extends from the superior parietal lobule along the cingulate gyrus in the superior parietal and frontal white matter (Brodmann area (BA) 7, 5, 4) rostrally to the superior frontal gyrus white matter, and terminates in the dorsal and medial cortex of the frontal lobe in the supplementary [101, 113] and premotor areas of the frontal lobe (BA 6, 8, and 9) [114].
The present understanding of human connectivity is comparable with neurotracer injections studies on non-human primates where it has been shown that all areas of SPL form a network with the areas of the frontal and premotor cortices, as we have described in section 2.
Moreover, the analysis of DTT images has indicated in the human brain a strong SPL interconnection with the posterior temporal lobe, called the SLF/AF TP-SPL, the temporo-parietal (TP) white matter connections of the superior longitudinal fasciculus/arcuate fasciculus (SLF/AF) connecting the posterior temporal lobe with the SPL. This connection has not yet been elucidated on the previous DTI or anatomical studies in non-human primates, and in human brain it may be linked to language development and may explain the higher cognitive and comprehensive language functions in humans compared with other primates.
The human parietal lobe is also largely connected with subcortical structures, especially with the thalamus. It has been demonstrated in vivo that posterior parietal areas target mostly a region within the thalamus corresponding to the lateral posterior nucleus and the anterior and lateral pulvinar [115, 116]. Medial SPL areas within the precuneus have prevalent connections with the dorsal pulvinar. These connections were clearly distinguishable from connections of the ventral pulvinar to early and higher visual areas [117, 118]. The human connections between thalamus and cortex are similar to those reported for non-human primates [43, 45, 46].
Despite all the aforementioned studies provide a comprehensive and multimodal
representation of the network architecture of parietal areas, the cortical
network and functional organization of each human SPL subregion remains largely
unknown. For this reason, many functional studies with fMRI techniques have been
carried out to identify putative human homologue of areas of macaque brain, in
the following we focus on those studies only referring to SPL areas. Sereno and
co-workers, using fMRI techniques, have identified a second somatosensory
homunculus in a region of the human SPL, located in the postcentral gyrus,
immediately posterior to the primary somatosensory cortex: they called it
Parietal Body Area (PBA) [119, 120]. PBA contains a rough topographical map of the
body resembling the one observed in the macaque area PE [36]. Another very recent
neuroimaging study demonstrated that the same areas described in the macaque SPL,
V6A, PEc, and PE can be found in the human medial SPL cortex, as shown in Fig. 3
where the brains of the two species are compared [10]. Human (h) V6A is located
in the posterior portion of SPL, anterior to the parieto-occipital sulcus, in a
cortical territory that includes the caudalmost portion of the precuneate cortex;
hV6A is anterior to hV6 [121] and posterior to hPEc. The latter occupies the
dorsalmost portion of the anterior precuneus, just behind to the dorsal tip of
the cingulate sulcus [122]; area hPE is anterior to hPEc and sits in the
dorsomedial portion of the post-central gyrus, just over the tip of the cingulate
sulcus [10]. In parallel with macaque studies, human V6A shows a clear
over-representation of the contralateral lower visual field and responds only to
visual stimulation of the far periphery (starting from 30
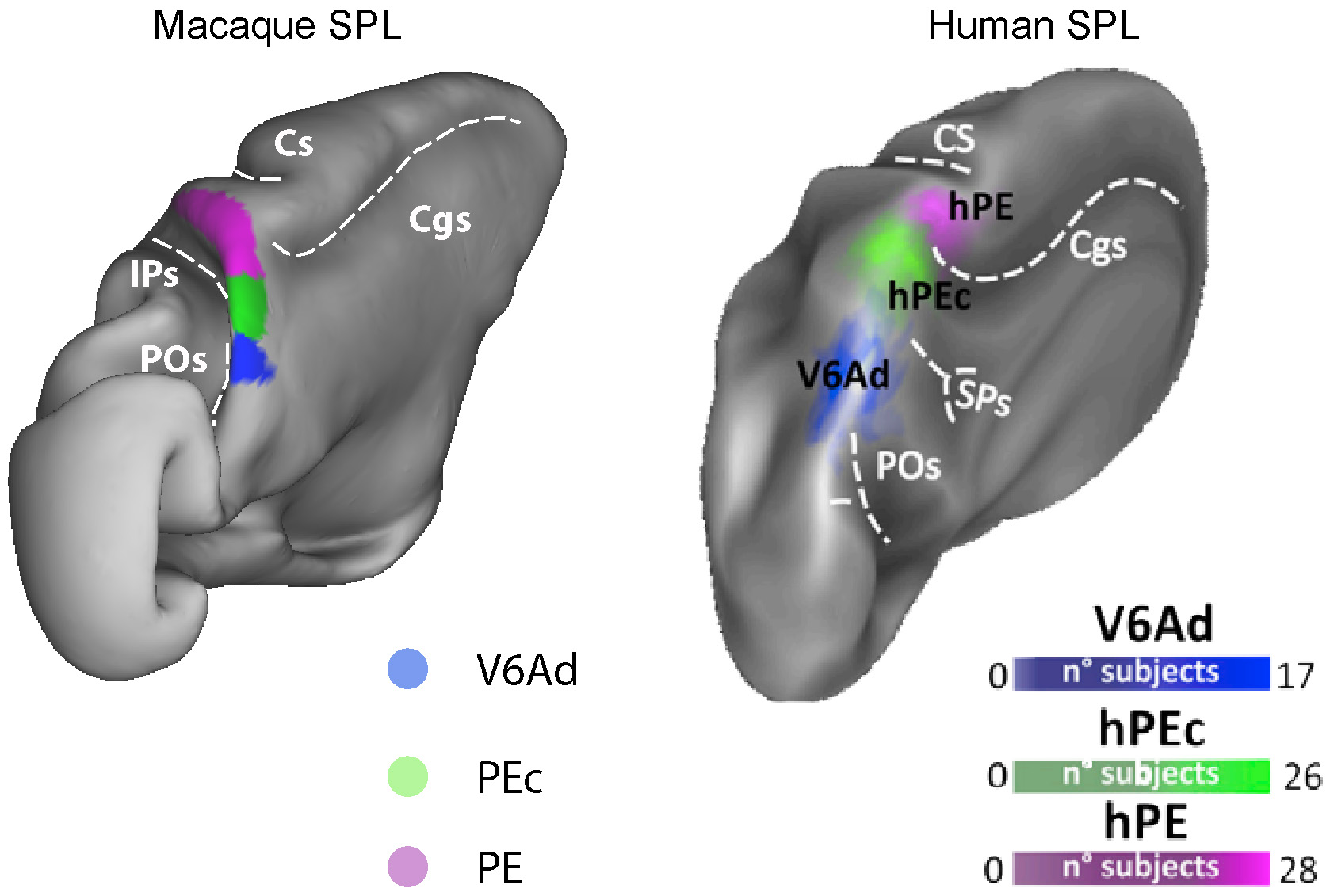 Fig. 3.
Fig. 3.Homologies between some SPL areas in macaque and human brain.
3D reconstructions of the postero-medial view of a left hemisphere of the
macaque (left) and of the human (right), not in scale.
Left: Extent and location of the cytoarchitectonically defined areas V6A, PEc
and PE. The borders were obtained overlapping retinotopic and functional
properties of single neurons and cytoarchitectonic features in monkeys
[14, 44, 136].
Right: Extent and location of hV6Ad, hPEc and hPE [10, 122]. Individual ROIs are
overlapped onto Conte69 brain atlas to form group probabilistic ROIs. The color
scale indicates the proportion of subjects whose ROI included that surface node,
thus the probability that the node is included in the ROI.
Dotted line indicates the fundus of main sulci:
Cgs, cingulate sulcus; CS, central sulcus;
IPs, intraparietal sulcus;
POs, parieto-occipital sulcus;
SPs, sub-parietal sulcus.
Modified with permission from [10] published in Elsevier.
In this section, we provide a revision of some neurological syndromes associated to lesions in both human and non-human SPL areas. The study of lesions in a specific brain area permits to understand in an indirect way its role in normal conditions starting from specific dysfunctions. In addition, it allows the understanding of the functional role of SPL areas to process distinct sensory and motor information to guide actions.
Parietal lesions result in severe impairments in the control of eye movements towards visual targets, in the guidance of reaching actions and of the grasping of nearby objects, as well as in the ability to direct attention to visual stimuli, in both human and non-human primates.
In humans, the Balint’s syndrome is a manifestation of behavioural impairments specifically associated with lesion in the SPL and the parieto-occipital junction. Balint’s syndrome includes difficulty in executing eye movements to capture visual targets (ocular apraxia), inability to perceive multiple objects simultaneously (simultanagnosia), and inability in reaching for visual targets (optic ataxia). These symptoms have a heavy impact onto the visuospatial skills, the visual scanning and the attentional mechanisms [137]. Simultanagnosia is the inability of the patient to perceive multiple items when visually displayed concurrently but maintaining the ability to recognize single objects. Simultanagnosia might result from an extreme form of competition between objects, which makes it difficult for attention to be detached from an object once it has been selected [138]. Patients have a narrowed spatial window of visual attention and only one object at a time can be seen [139]. As a result of this impairment, patients with simultanagnosia often fail to understand the overall meaning of a scene. After six months of rehabilitation, a sensory/motor evaluation ascertained that clinical neurological symptoms in patients with Balint’s syndrome did not disappear, despite observable functional and physical improvements. Further studies are required to adequately develop effective rehabilitation protocols and demonstrate the effectiveness of rehabilitation in treating patients with Balint’s syndrome [137].
Unilateral or bilateral lesions in regions of the SPL can also produce a distinct syndrome, optic ataxia [140, 141, 142, 143, 144] characterized by errors in reaching, particularly toward objects in the visual periphery [145], as documented in Fig. 4. Patients suffering from optic ataxia after lesions of the SPL, the parieto-occipital junction, or the intraparietal sulcus, due to stroke or surgery circumscribed to unilateral right or left hemisphere [143, 146] experience difficulty in making visually guided arm movements towards an object in the visual environment (Fig. 4). The impairment is severe when the target is located peripherally in the visual field, diminishes when the target lies in the parafoveal region, and it is negligible when the target is foveated by the patient. A worsening in the performance occurs in the use of the hand or in the reaching into the visual field both contralateral to the lesion [144, 147, 148]. Patients with optic ataxia also manifest deficits in regulating hand orientation [140] and shaping [149, 150] during visual guided grasping that may arise from a failure to transform retinal information into appropriate spatial coordinates for action [151]. The failure is due to the fact that the brain damage involves the human cortical territory of area V6A (roughly corresponding to the purple area shown in the 3D image of the bottom of Fig. 4) [3, 9, 15, 94].
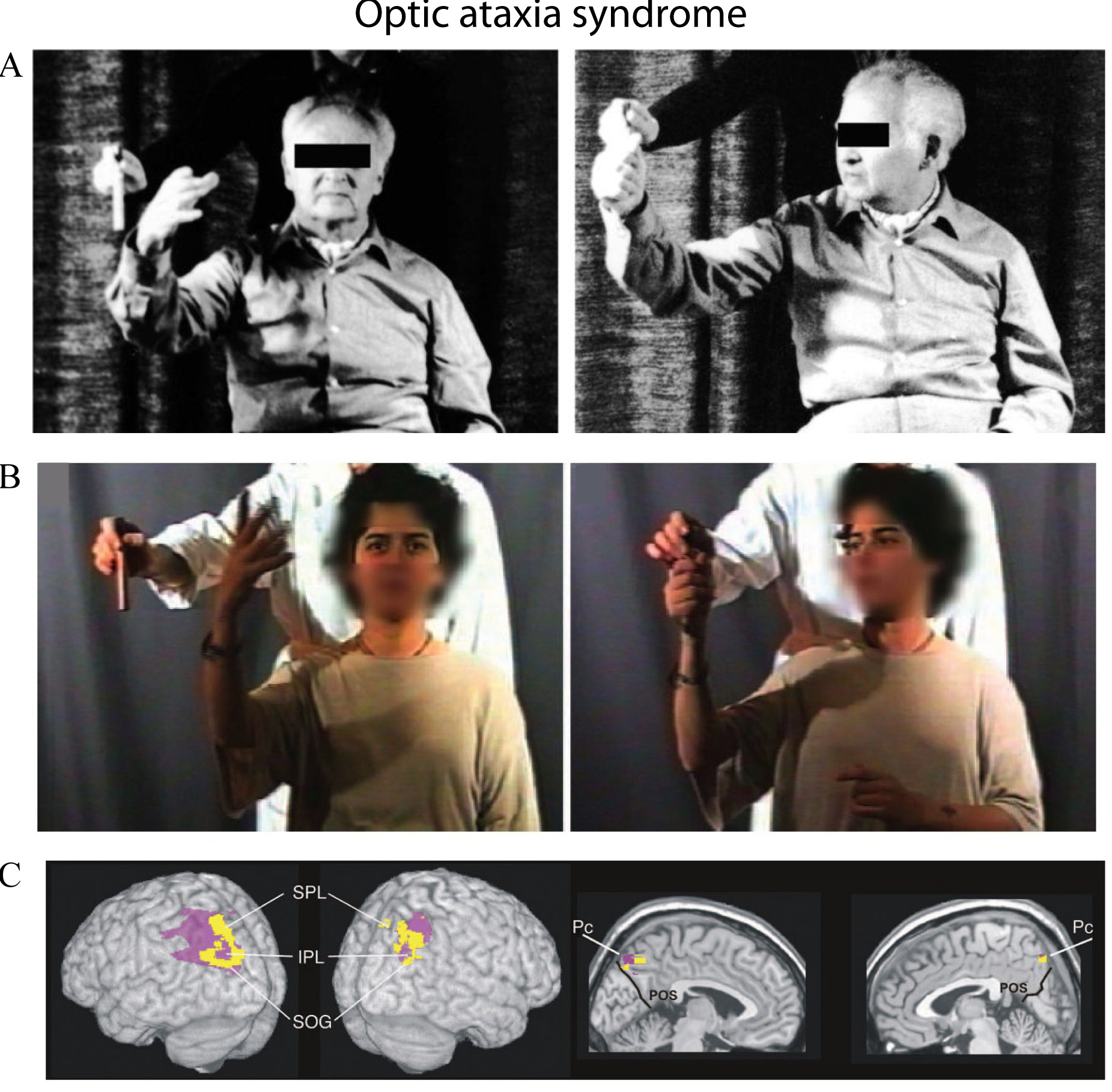 Fig. 4.
Fig. 4.Reaching for a target in two exemplary patients with
optic ataxia.
(A-B): Left: patients showed misreaching for a target in peripheral vision, when
they had to fixate the camera lens in front of them. Right: the patients showed
normal reaching under foveal vision when they had to orient eyes and head towards
the object while reaching for it. Typically, such ataxic reaches were shown most
frequently with the contralesional hand in contralesional space. (C): Location of
the centre of the lesion that causes optic ataxia, in lateral and mesial views.
Subtracted superimposed lesions of 10 left and 6 right optic ataxia patients
minus 2 brain-damaged control groups, matched either for associated symptoms
(pink area) or for lesion location, primarily involving the parietal lobe (yellow
area).
IPL, inferior parietal lobule; Pc, precuneus; SOG, superior occipital gyrus; SPL, superior parietal lobule. The parieto-occipital sulcus (POS) is shown in
black on the medial views. Adapted with permission from [143, 160] published in Oxford University Press and in SAGE journals..
Similarly, in monkeys, the evidence that brain lesions restricted to area V6A produce misreaching and misgrasping during visually guided movements [152] supports the same hypothesis. In particular, V6A lesions produce underestimation of the position of reach/grasp targets, increase the reach and grasp times, with abnormality in conforming and orienting the hand towards the targets. This, overall, results in an inability of correctly reaching and grasping a visual target, especially with the arm contralateral to the lesion. The functional characteristics of V6A cells are congruent with the misreaching and misgrasping observed after V6A lesion [3].
Enhancement in both reaction- and movement-times necessary to adjust the trajectory towards a target, while monkeys executed reaches and saccades to visual targets and the target position changed unexpectedly, has been showed in a study in the monkey superior parietal area 5 (PE/PEc). In this study this region of the brain was reversibly inactivated after bilateral muscimol injection [153] and the authors interpreted these findings as a deficit in the online control similar to that observed in optic ataxia patients.
Lesions in the human SPL may cause also tactile agnosia, an inability to identify familiar objects manually [154] or tactile apraxia, characterized by an isolated disturbance of hand movements for use of and interaction with an object [155]. This evidence highlights relevance of the areas of SPL in perception of the objects, spatial attention and in encoding of target location for the online control of arm movements.
It is not surprising, considering the above recalled thalamo-cortical connections, that lesion in the dorsal pulvinar in both human and non-human primates leads to a constellation of deficits involving the selection of eye and hand movement targets and coordination of visually guided manual reaching and grasping [156, 157] very similar to that observed after V6A lesion. In macaque monkeys, reversible inactivation of the dorsal pulvinar with muscimol injection resulted in a spatial neglect syndrome accompanied by visuo-motor deficits including optic ataxia during visually guided limb movements [156]. In humans, the patient with a lesion centred on the medial portion of the dorsal pulvinar exhibited: reach and grasp difficulties, problems to form a precision grip, reach deficits expressed in decreased accuracy, delayed initiation and prolonged movement durations. Reach deficits were similar in foveal and extrafoveal viewing conditions and in both visual hemifields but were stronger with the right hand [157]. These findings highlight the importance of the dorsal aspect of the pulvinar nucleus as a critical hub for spatial attention, visual cognition and selection of visually guided actions, according to the thalamo-cortical network described in section 2.
The comparison of the lesions in SPL in the two species of primates is not trivial and creates greater difficulties in making comparisons than relying on the other techniques described in this review. Brain lesions in fact differ in the two species for at least two aspects: the extent of the cortical lesion and the duration of the functional recovery. The extent of human lesions is caused by pathological situations that often involve large territories, often including many different areas and largely diverse from one patient to the other. On the contrary, lesions in non-human primates are performed with the intent of elucidating the role of a single area, or portions of areas, so they are limited in extent and very precisely located in particular districts. Any comparison among species must consider this important difference. Regarding recovery time, in the monkey, muscimol creates lesions of few hours, and the deficits after surgical lesions often disappear in a few days (3-4 days) [152]. This fast recovery can be caused by the limited extent of the lesion, but also for other reasons, like the faster development of monkey brains and their greater plasticity [158]. Furthermore, areas distant from the core of the lesion can play an important role during the early post-recovery period, whereas the perilesional areas contribute to functional recovery especially during the late post-recovery period [159]. For this reason, the lesion in the monkey can likely have a shorter recovery time as being more limited in its spatial extend. On the contrary, in human the lesions can involve large parts of the brain and the variability between individuals can be high, by delaying or preventing a full recovery of the patients. Despite differences in the two models, the pathological effects of the SPL lesions described in this chapter are comparable. Therefore, the proposed homology between human and monkey SPL could help to understand the role of this part of the brain tapping from animal studies to find out possible therapeutic applications for human health.
The homology between human and monkey SPL, discussed in previous sections, could find applications also in the field of neural prostheses and brain-machine interfaces (BMIs). BMIs are designed to circumvent lesions and repair human cognitive or sensory-motor functions [161].
In the past decade, in order to guide robotic limbs in monkeys and humans, the majority of studies has investigated the signals to reconstruct reach trajectories from primary motor and premotor cortex [162, 163, 164, 165]. Today the new field of investigation has moved towards the posterior parietal cortex in humans and monkeys as a good source of different kinds of signals, such as those related to movement intentions and goals. Information about reach-endpoints [166, 167], trajectories [168], and grips [169] are encoded from the monkey posterior parietal cortex. Translated to humans, these studies produced comparable and promising results [170], together with reliable hand shape decoding [170]. Among the posterior parietal areas, studies by Filippini and colleagues [171, 172, 173] have shown that grasping and reaching signals and also multiple task- and intention-related variables could be decoded simultaneously from population activity in area V6A. Area V6A is a good source of information on the guidance of grasping movement with or without visual information and on the control of the interaction between hands and object. Information that can be decoded and used in preparation and execution of movement of prehension [171] and for the offline decoding of reach goals located at different depths and directions, mimicking everyday life conditions [172]. Filippini used two different tasks, one in which the monkeys fixated the goal of reaching movement, which is the most common physiological condition [174, 175] and another where gaze and reaching targets did no coincide. In both conditions, the accuracy of decoding was optimal from early target fixation to the end of the reaching task and supports a strong spatial tuning in V6A reach-related activity when gaze is dissociated from the reach target position [176]. The results suggest that V6A could be a source for BMIs both when the patient can move his or her eyes to the reaching target and also in the absence of ocular motility [172]. From V6A population activity, it is also possible to decode simultaneously information regarding the target position, the required movement type and the time interval as the task progresses [173]. A metric estimation of the target positions has been derived via maximum likelihood estimation that when compared with the Naïve Bayes classifier, allowed obtaining a classification of task type. Finally, by using a Bayesian decoder, the posterior probabilities of free, delay and movement states could be obtained [173]. Therefore, neurons in V6A encode several types of information such as spatial position, intention for a specific motor response and progress of the task in the same population. Additionally, they support the idea that neurons are not simply tuned to a single feature, but they encode several task-relevant variables at the same time [177]. In terms of invasiveness and accuracy, decoding multiple parameters from the same area could be advantageous for both BMI and neuroprosthetic applications [171, 177] by allowing a single electrode array to decode multiple action scenarios (Fig. 5).
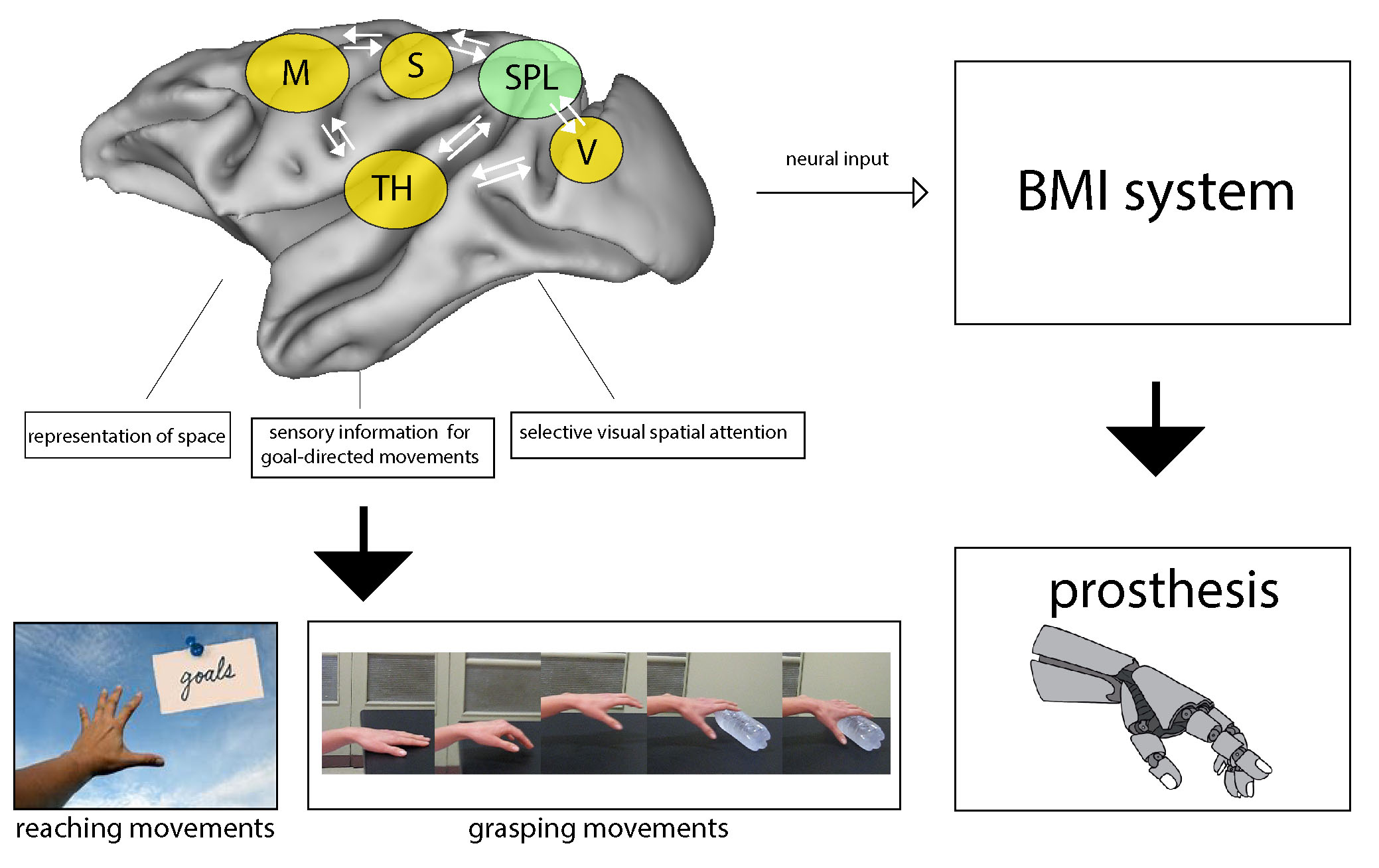 Fig. 5.
Fig. 5.The Superior Parietal lobule of primates: a
sensory-motor hub for interaction with the environment useful for improving human
health. Simplified scheme showing a network of areas of the macaque brain that are
reciprocally connected with the areas of SPL and the thalamus, involved in the
guidance of arm-reaching and reach-to-grasp movements. The SPL acts as a hub for
exchange of sensory and motor-related information crucial for guiding arm actions
towards targets in the environment and for adjusting hand shape to objects so to
grasp them. The knowledge about this network in the macaque can help to conceive
brain machine interfaces able to use neural signals from human SPL to guide
prosthesis in tetraplegic patients.
Abbreviations: M, motor areas; S, somatic areas; SPL, superior parietal lobule areas; TH, thalamus; V, visual areas.
We have summarized the anatomical subdivision, the cortical and thalamic connections of the macaque SPL, we have compared their functional properties and summarized the organization of the human SPL in physiological and injuried situations. Despite the methodological differences in the study of macaque and human brains, the results obtained over the years show independent and convergent evidence that the human and macaque brain as a whole are similar and similarly wired. In fact, starting from the consolidated models in the monkey and from the large amount of data collected over decades, and with the help of new high spatial resolution techniques (e.g., fMRI, diffusion tensor imaging, tractography) experimental models in human and non-human primates show a high level of comparability. This strengthens and underlines the importance of looking for homologies, as in the cases previously discussed, where the proposed homology with the monkey brain has guided the study of the organization of the human SPL.
The rich set of inputs reaching the SPL merge to serve different goals, involving the representation of space, grasping actions, and the use of the sensory information for goal-directed movements. Areas of the SPL elaborate also higher-level cognitive functions, like selective visual spatial attention. All these properties are building neuronal networks useful in the sensory-cognitive interplay, essential in the correct execution of reaching and grasping movements (Fig. 5). Moreover, the parallel between macaque and human SPL described here supports the view of an analogous functional organization, with a somatosensory-dominated anterior sector and a bimodal, visual-somatosensory caudal part, both contributing to vision-for-action and spatial functions. The suggested interspecies homology could address forthcoming research in humans and monkeys aimed to improve the knowledge about brain structure, function and disease, useful for clinical applications and novel therapies of nervous system diseases, or brain-machine interface technologies.
Neuronal circuitry of the areas of the SPL as described above, mediate sensory-motor transformations, encode the spatial coordinates for reaching goals, hand position and movements, and other spatial variables. The new challenges for the next decades of neuroscience and neuro-engineering research will be how to encode and better implement the different kinds of information processed by the SPL areas in brain-machine interfaces to control both goal-directed movements and cognitive functions and how to decode efficiently the neural signals to be used in order to move neural prostheses with the aim of regaining motor function in patients with motor disabilities.
Macaque cortical areas:
1, Brodmann area 1; 2, Brodmann area 2; 3, Brodmann area 3; 4, Brodmann area 4; 6, Brodmann area 6; 8, Brodmann area 8; 23e, Area 23, external subdivision; 23a/b, Area 23 external and internal subdivision; 24a/b, Area 24 external and internal subdivision; 31, Area 31; 46, Brodmann area 46; AIP, Anterior intraparietal area; F2, Frontal area 2; F3, Frontal area 3; F4, Frontal area 4; F7, Frontal area 7; LIP, Lateral intraparietal area; MIP, Medial intraparietal area; MST, Medial superior temporal area; PE, Area PE; PEa, Area PE anterior; PEc, Area PE caudal; PEci, Area PE cingulate portion; PEip, Area PE intraparietal portion; PG, Area PG; PGm, Area PG mesial; PGop, Area PG opercular; PO, Parieto occipital area; SII, Secondary somatosensory cortex; SPL, Superior parietal lobule; SSA, Supplementary Somatosensory Area; Tpt, Temporoparietal transition area; TSA, Transitional sensory area; V2, Area V2; V3, Area V3; V4, Area V4; V6, Area V6; V6Ad, Area V6A, dorsal portion; V6Av, Area V6A, ventral portion.
Human cortical areas:
5Ci, Area 5, cingulate parietal; 5L, Area 5, lateral; 5M, Area 5, medial parietal; 7A, Area 7, anterior; 7M, Area 7, mesial; 7P, Area 7, posterior; 7PC, Area 7, postero-caudal; BA5, Brodmann area 5; BA7, Brodmann area 7; hPE, Human PE; hPEc, Human PEc; hV6, Human V6; hV6A, Human V6A; PBA, Parietal Body Area.
Thalamic nuclei:
LP, Lateral posterior; PuM, Medial pulvinar; VA, Ventral anterior; VL, Ventral lateral; VPL, Ventral posterior lateral.
Others:
BMIs, Brain-machine interfaces; DTT, Diffusion tensor tractography; fMRI, Functional magnetic resonance imaging; MRI, Magnetic resonance imaging; SLF, Superior longitudinal fasciculus; SLF/AF, Superior longitudinal fasciculus/arcuate fasciculus; TP, Temporo-parietal white matter connections.
LP wrote the manuscript. LP, MG and PF contributed to editorial changes in the manuscript.
LP, MG and PF read and approved the final manuscript.
This article does not contain any new, previously unpublished study with human participants or animals performed by the authors.
The manuscript benefited by the comments and suggestions of the editors and three anonymous reviewers that greatly improved this work.
This work was supported by grants from Ministero dell’Università e della Ricerca (2017KZNZLN), and Fondazione Cassa di Risparmio di Bologna (Bando Internazionalizzazione), Italy, and partially supported by the European Commission funded project, “HumanE AI Network - HumanE-AI-Net”, Grant. Agreement n. 952026, H2020-MSCA-734227-PLATYPUS, H2020-EIC-FETPROACT-2019-951910-MAIA. The support is gratefully acknowledged.
The authors declare that they have no conflict of interest.





