1. Introduction
Consciousness is an unsolved scientific enigma. Diverse quantum models attempt
to explain consciousness mechanisms (Fisher, 2015; Hameroff and Penrose, 2014; Jibu and Yasue, 1995; Kumar et al., 2016; Poznanski et al., 2019).
Fisher (2015) first proposed the phosphorus-31 nuclear-spin entanglement
model. According to Fisher’s proposal, in bulk water, the survival time of
nuclear-spin entanglements for phosphorus-31 is too short (within one second) in
free phosphate ions due to proton attack. However, if the phosphorus-31 are the
constituents of Ca(PO), Posner molecules, its unique structure
protects phosphorus-31 nuclear-spin entanglements from proton attack. As a
result, the decoherent time lasts as long as 21 days or longer for the
entanglements (Swift et al., 2018). Long-lived quantum entanglement is
central in quantum consciousness processing (Swift et al., 2018). Thus,
Posner molecules play a crucial role in processing consciousness (Fisher, 2015; Swift et al., 2018).
However, though attractive, Fisher’s proposal is speculative (Fisher, 2015; Swift et al., 2018) and lacks experimental evidence to support or disprove it.
Fisher’s entire proposal cannot be tested experimentally, but a partial test is
possible because some verifiable predictions can be derived from the proposal
(Fisher, 2015). The testable predictions include varying calcium
concentrations and the existence of calcium isotope dependence on consciousness
(Fisher, 2015). Since Posner molecules are important in enabling
phosphorus-31 nuclear-spin quantum entanglement to mediate consciousness in the
brain, it is expected that varying calcium ion concentrations in the
extracellular fluid would affect Posner molecules formation and accordingly it
will influence the levels of consciousness (Fisher, 2015). So, an increase of
Ca concentration in the cerebrospinal fluid is expected to
increase the number of Posner molecules and increase the level of consciousness.
Conversely, a decreased Ca concentration is expected to lower the
level of consciousness.
Calcium isotope dependence is also expected in Fisher’s proposal (Fisher, 2015). Specifically, Ca would disturb and decohere the
phosphorus-31 nuclear-spin entanglements if Ca replaces the
Ca within the Posner molecules (Fisher, 2015). Therefore, an
increase of Ca concentration in the cerebrospinal fluid would lead
to a decrease in the animal’s consciousness level, opposite to the effect of
Ca.
We use the loss of righting reflex (LORR) ED for mice to sevoflurane to
measure consciousness level (Franks and Lieb, 1994; Franks, 2008; Li et al., 2018) to test the predictions mentioned above. LORR ED closely
correlates to the consciousness level; as described in our previous study, an
increased LORR ED indicates increased consciousness level and vice
versa (Li et al., 2018). By quantifying consciousness levels, we disprove
Fisher’s proposal because our findings oppose his predictions.
2. Materials and methods
2.1 Animals
C57BL/6 male mice (7 weeks old) were obtained from Beijing Vital River
Laboratory Animal Technology Co., Ltd. (SCXK [jing] 2016-0006; P. R. China). The
animals were kept in a 12:12 hour cycle of light/darkness with free access to
water and food at room temperature (24 1 C) for a week before
experiments. When experiments were performed, the animals were 8 weeks old. All
animal operations and experimental protocols conformed to the US National
Institutes of Health guide for laboratory animals’ care and use (NIH Publications
No. 8023, revised 1978). They were approved by the Institutional Animal Care and
Use Committee (approval No: S164) at Tongji Medical College, Huazhong University
of Science and Technology.
2.2 Reagents
Sevoflurane was obtained from Maruishi
Pharmaceutical Co., Ltd. (Osaka, Japan). Calcium-43 carbonate (CaCO)
was purchased from ISOFLEX USA (San Francisco, USA) with an abundance of 90%. As
the abundance of calcium-40 is 96.9% in nature (Wieser et al., 2004),
natural CaCl was regarded as CaCl in this study. CaCl,
NaCl, NaOH, and HCl were purchased from Sinopharm Chemical Reagent Co., Ltd.
(Shanghai, P. R. China). A calcium chelator ethylene glycol-bis (2-aminoethyl
ether)-N,N,N’,N’-tetraacetic acid (EGTA) was purchased from
Beijing Solarbio Science & Technology Co., Ltd. (Beijing, P. R. China). 0.9%
sodium chloride solution was obtained from Baxter Healthcare Co., Ltd. (Shanghai,
P. R. China).
2.3 Reagent preparations
The calcium-43 isotope we obtained was calcium carbonate, an insoluble powder.
We had to transfer CaCO powder into CaCl solution
before use. CaCO powder 0.5 g was dissolved in 10 mL HCl
solution (1 mol/l) to transfer CaCO powder into CaCl
solution. The final concentration of Ca ion was 50 mmol/l, verified
by ABL800 FLEX blood gas analyzer (Radiometer
Medical ApS, Brønshøj, Denmark). EGTA 0.38 g was dissolved in NaOH
1 mol/l. The final concentration of EGTA was 7.5 mmol/l. All injection solutions
were prepared in the 0.9% NaCl solution, adjusted to an osmolality of 300
10 mOsm/l by NaCl or tri-distilled water and adjusted to pH values
7.35-7.45 by NaOH or HCl solution. The solutions were filtered through a
0.22-m filter (Millipore, Bedford, MA, USA) before use.
2.4 Intracerebroventricular
injections
Sixty C57BL/6 male mice (8 weeks old) were weighed and randomly divided into
four groups, with 15 mice in each group. The mice in the four groups received
intracerebroventricular (ICV) injections of 0.9% NaCl as control, EGTA 7.5
mmol/l, CaCl 50 mmol/l, and CaCl 50 mmol/l in a volume
of 2 L, respectively. The dosages of calcium chloride and EGTA injected
into mice’s lateral cerebral ventricle were determined as previously described
(Erickson et al., 1978; Liang et al., 2004).
Basal LORR ED for each mouse to sevoflurane was determined. A sterile
25-gauge stainless steel guide cannula (RWD Life Science Co., Ltd., Shenzhen, P.
R. China) was inserted into the lateral cerebral ventricle following a previously
described method (Li et al., 2015; Marsh et al., 1999) with slight
modifications. Briefly, under 2% sevoflurane anesthesia, the mouse was placed on
a stereotaxic frame (RWD Life Science Co., Ltd., Shenzhen, P. R. China). After
disinfecting the surgical area three times with 75% alcohol, a 1 cm midline
incision was made behind the eyes toward the cranium’s posterior. The skull was
exposed, and the periosteum was cleaned by wiping the skull with a sterile cotton
swab. A hole 0.5 mm in diameter was made, 0.3 mm posterior and 1.0 mm lateral to
the bregma using a dental drill. The guide cannula was inserted 3.0 mm below the
skull surface and fixed to the skull with dental cement. A matching stylet was
then inserted into the guide cannula to prevent infection and obstruction. After
1 day of recovery, the mouse was sedated with 1% sevoflurane. The stylet was
then removed, and a 30-gauge cannula was inserted into the guide cannula. Saline
or a test agent was contained in a 5-L Hamilton syringe. The syringe
connected to the cannula was fixed in a syringe pump (RWD Life Science Co., Ltd.,
Shenzhen, P. R. China). All agents were warmed to 37 C before infusion.
The ICV injection was performed automatically by the pump at a rate of 1
L/min. A total volume of 2 L was injected. After injection, the
30-gauge cannula was kept in place for 5 min. The cannula was then slowly pulled
out, and the stylet was inserted back into the guide cannula to prevent the
backflow of the injected solution. Sevoflurane was then discontinued, and the
mouse could recover. When the mouse could move freely, the LORR ED for the
animal to sevoflurane was determined.
2.5 Loss of righting reflex testing
To determine the LORR ED for mice to sevoflurane, animals were
individually placed in an isolated plastic mesh cage fixed in a 1-liter clear
plastic chamber. One side of the chamber was connected to the oxygen source and a
sevoflurane vaporizer (Aika, Ichikawa Shiseido, Tokyo, Japan). The other side was
connected to an infrared gas monitor (BeneView T5; Shenzhen Mindray Bio-Medical
Electronics, P. R. China) to measure sevoflurane, oxygen and CO
concentrations in real-time. The monitor was chosen because it can provide a
precision of 0.01% when sevoflurane concentration is more than 1%, whereas most
commercial monitors only provide a precision of 0.1% under the same condition.
The chamber was warmed (Li et al., 2018), and the chamber temperature was
monitored continuously and maintained at 36.0 0.2 C.
When the mouse was placed in the chamber, pure oxygen was supplied at a rate of
600 mL/min immediately. When the chamber’s oxygen concentration increased to
99%, sevoflurane gas mixed in pure oxygen was provided by the vaporizer. The
initial sevoflurane concentration was 1.00% in the chamber. After 15 minutes at
that concentration to equilibrate the mouse with sevoflurane gas, the chamber was
rotated 180 to place the mouse on its back in a V-shaped trough, and
its righting reflex was observed. LORR was defined as the supine mouse unable to
turn itself onto all 4 paws three times within 1 min (Kelz et al., 2008).
According to the mouse’s righting reflex, a stepwise increase or decrease of
0.10% sevoflurane in the chamber was given. Specifically, if the mouse’s
righting reflex disappeared, a decreased 0.10% sevoflurane concentration was
given; otherwise, an increased 0.10% sevoflurane concentration was supplied.
After 15 minutes of equilibration at each sevoflurane concentration, the mouse’s
righting reflex was observed again. LORR ED was the average value of the
two critical sevoflurane concentrations at which the mouse either lost or
returned its righting reflex (Fukagawa et al., 2014). During the
measurements, the actual concentrations of sevoflurane in the chamber were
continuously monitored and adjusted to guarantee that the difference between the
actual concentration of sevoflurane and the target concentration was at
0.01%. All righting reflexes were observed by a trained observer who was unaware
of drug administration. All determinations were made between 8 and 18 o’clock.
After measurements, the mice could recover for 1 week. After recovery, the mice
were weighed again. Since weight loss is an overall measure for possible neural
pathologic damages that inevitably affect animal feeding, the mice that did not
reach the weight a week before were excluded in this study as described
previously (Perrin et al., 2004). To confirm successful ICV injection, the
mice were then anesthetized with 3% sevoflurane, and 1 : 10 of Evans blue 2
L was injected through the guide cannula. The mice were then decapitated,
and the brain was removed to verify diffusion of the injected dye throughout the
ventricular system (Davisson et al., 1998). Mice without dyed ventricles were
regarded as unsuccessful ICV injections attempts and were excluded (Davisson et al., 1998).
2.6 Statistical analyses
The sample size was determined based on the literature (Hu et al., 2012). A
larger number of mice were enrolled in this study than the literature (Hu et al., 2012) to allow for possible exclusion of mice that did not pass stated
experiment requirements. Statistical analyses were performed using GraphPad Prism
software version 6.07 (GraphPad Software Inc., USA). LORR ED data were
presented as mean SD. Two-way repeated-measures analysis of variance
(ANOVA) was used to analyze the data, followed by the Bonferroni correction as
a post hoc test. P 0.05 (two-tailed) was designated as
statistically significant.
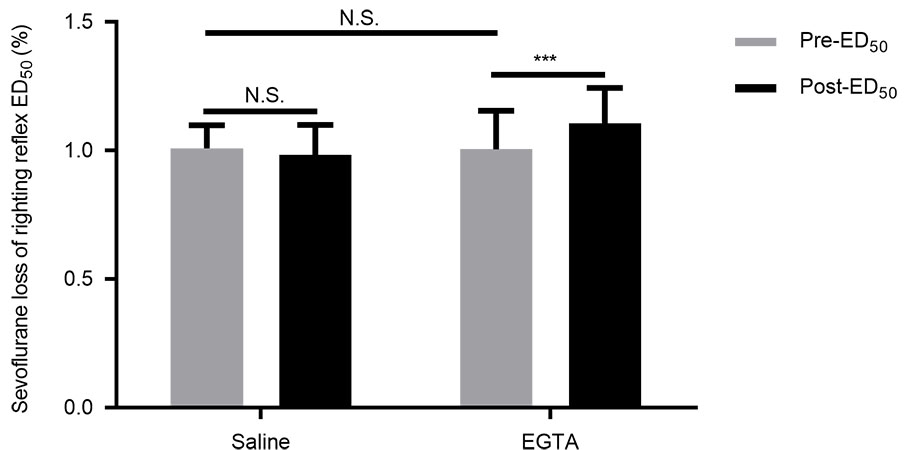 Fig. 1.
Fig. 1.
Effects of normal saline and EGTA on righting reflex
ED for mice to sevoflurane. Pre-ED denotes the basal LORR
ED. Post-ED presents LORR ED after lateral ventricle
administration of normal or EGTA. The difference in the values of the
pre-ED between the two groups is not significant. The value of the
pre-ED is comparable to the post-ED in the saline group. The
post-ED value is significantly higher than the pre-ED in the EGTA
group. Data are shown as mean (SD), n = 12 in the saline group and n = 13 in the
EGTA group. ***P 0.001; N.S. = not significantly different (two-way
repeated-measures ANOVA followed by the Bonferroni correction as a post
hoc test).
3. Results
To investigate the effects of calcium concentrations and calcium isotopes on
sevoflurane-induced LORR ED, sixty C57BL/6 male mice were included. No
mice were excluded due to weight loss. Three mice in the saline group and two in
each of the other three groups were excluded due to unsuccessful ICV injection.
Finally, data of the LORR ED of 51 mice, 12 in the saline group and 13 in
each of the other three groups were taken into statistical analysis. Two-way
repeated-measures ANOVA was used to analyze the data, with one main factor for
time and the other for the group. Analyses revealed that regardless of the
treatment condition, LORR ED values between the first and the second
measurements had critical differences (F = 35.47, P
0.0001). Not surprisingly, the effects of group type on LORR ED values
were significant (F = 7.77, P = 0.0003) regardless of the
measurement condition. The Bonferroni post hoc multiple-comparison test analyzed
the effect of a group within each level of time and the effect of time within
each group level. A strong time group interaction was found (F = 34.32, P 0.0001).
3.1 Effects of saline and EGTA on LORR ED
The basal value of LORR ED was determined for the 12 mice to sevoflurane
in the saline group, before the ventricular cannulation, was at 1.01
0.09% (95% CI, 0.95% to 1.07%). The value of LORR ED for the same mice
to sevoflurane, determined after ICV injection of saline was at 2 L, was
0.98 0.12% (95% CI, 0.91% to 1.06%). The two values were highly
comparable (P 0.9999, Fig. 1). Since the mice were determined
twice for their LORR ED to sevoflurane, there existed a possibility that
the residual sevoflurane of the first determination might influence the second
one. However, one day was allowed for the animals to recover between the two
determinations. This interval is regarded as adequate for the animals to exhale
out the sevoflurane completely. The lateral ventricular catheterization itself
might also influence the second LORR ED. The highly comparable LORR
ED values before and after saline injection of the mice in the control
group ruled out these possibilities, indicating that the two determinations were
independent. Therefore, LORR ED values of the mice after ICV injections of
EGTA, CaCl and CaCl in the other three groups were
respectively independent on their first determinations.
To examine the effect of decreased calcium ion concentration in the
cerebrospinal fluid on a consciousness level, EGTA, a calcium chelator, was
directly injected into the mice’s lateral ventricle. The basal LORR ED for
the 13 mice to sevoflurane in the EGTA group was 1.00 0.15% (95% CI,
0.91% to 1.09%). The LORR ED for the mice to sevoflurane after EGTA ICV
injection was 1.10 0.14% (95% CI, 1.02% to 1.19%), significantly
higher than the basal value (P 0.001, Fig. 1).
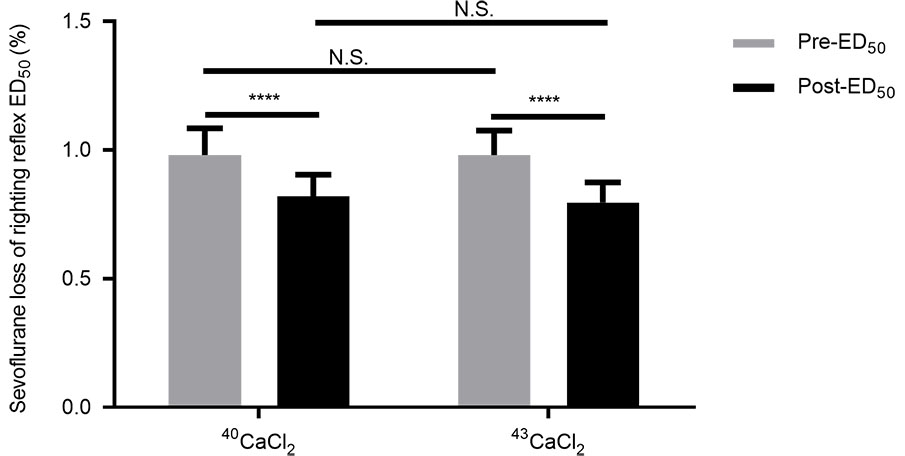 Fig. 2.
Fig. 2.
Effects of CaCl and CaCl
on righting reflex ED for mice to sevoflurane. Pre-ED denotes the
basal LORR ED. Post-ED presents LORR ED after lateral
ventricle administration of CaCl or CaCl. The
difference in the values of the pre-ED between the two groups is not
significant. The post-ED value is significantly lower than the
pre-ED in the CaCl group or in the CaCl group.
The post-ED values of the two groups are not significantly different. Data
are shown as mean (SD), n = 13 mice per group. ****P 0.0001; N.S. =
not significantly different (two-way repeated-measures ANOVA followed by the
Bonferroni correction as a post hoc test).
3.2 Effects of calcium-40 chloride and calcium-43 chloride on LORR
ED
To examine the prediction of calcium isotope dependence on a consciousness
level, the mice in the CaCl and
CaCl groups received
ICV injections of calcium-40 chloride and calcium-43 chloride, respectively.
Calcium-40 injection resulted in a significant decrease in LORR ED for the
mice to sevoflurane as compared with the basal value (0.82 0.09%, 95%
CI, 0.77% to 0.87% vs. 0.98 0.10%, 95% CI, 0.92% to 1.04%,
P 0.0001, Fig. 2). Similar change was observed after calcium-43
chloride injection (0.80 0.08%, 95% CI, 0.75% to 0.84% vs. 0.98
0.09%, 95% CI, 0.92% to 1.04%, P 0.0001, Fig. 2).
Further analysis showed that equimolar ICV injections of calcium-40 and
calcium-43 had no difference in LORR ED values (P 0.9999,
Fig. 2), indicating no calcium isotope dependence.
4. Discussion
Several quantum models based on quantum dynamics have been proposed with
attempts to demystify the mechanisms of consciousness. For example, the electric
spin entangled neural microtubules (the “Orch OR” theory) (Hameroff and Penrose, 2014), phosphorus-31 nuclear spin entanglements (Fisher, 2015),
photon-spin interface (Kumar et al., 2016), and unpaired electron-spin
activating nuclear spin-based mind-pixels (Hu and Wu, 2004), all set upon
theoretical speculations or simulations. Hence, debates on consciousness models
persist because there is no direct experimental evidence to confirm or reject any
theory of consciousness (Hameroff and Penrose, 2014), primarily due to a lack of
measures to probe consciousness. Anesthesia is a reversible unconscious state
induced by general anesthetics. General anesthetics are relatively selective for
consciousness while they spare many non-conscious brain activities (Craddock et al., 2015; Hameroff, 2006), anesthesia and consciousness may share the same
mechanism, thus making general anesthetics natural probes for consciousness
(Craddock et al., 2015; Hameroff, 2006).
In this study, LORR ED for mice to sevoflurane was used as a measure to
quantify consciousness levels in mice. Thereby, we tested the predictions derived
from the proposal that phosphorus-31 nuclear-spin entanglements protected by
Posner molecules may mediate consciousness, first proposed by Fisher in 2015
(Fisher, 2015). It is necessary to give a brief introduction to the proposal
first. Nine Ca and six PO ions form a Posner molecule,
Ca(PO). Two calcium-phosphate “structural clusters” with atomic
constituents Ca(PO), subsequently named “Posner clusters”, were
first found by Posner and Betts (Posner and Betts, 1975) while they were
examining the X-ray crystal structure of bone mineral-hydroxyapatite. The term
“Posner cluster” is expanded to “Posner molecule,” which has been assumed to be
central in consciousness due to its unique chemical structure (Fisher, 2015).
Posner molecules are assumed to protect the phosphorus-31 nuclear-spin
entanglements located within those molecules (Fisher, 2015). The
phosphorus-31 nuclear-spin entanglements within the Posner molecules, for
encoding information, remain coherent for times of a day, 21 days, or possibly
much longer (Swift et al., 2018; Weingarten et al., 2016). This would allow
Posner molecules to function as “neural qubits” to transmit quantum information
to the spatially separated neurons in the brain (Fisher, 2017; Weingarten et al., 2016). In addition to the phosphorus-31 nuclear-spin entanglements within
the molecules, their nuclear spins are further entangled among Posner molecules
once two bound Posner molecules start rotating about one another (Weingarten et al., 2016). Once at rest, Posner molecule pairs are susceptible to proton
attack and therefore are melted and release their Ca ions into the
neuronal cytoplasm (Weingarten et al., 2016). Because the Posner molecules in
the spatially separated neurons are entangled, when disentangled, all the
molecules are melted simultaneously and subsequently release a swarm of Ca
ions (considering nine Ca ions in each Posner molecule) in the neurons.
This might modulate intraneuronal Ca levels and thus make multiple neurons
spike simultaneously. The simultaneous spiking of spatially separated neurons is
considered a mechanism of consciousness (Weingarten et al., 2016).
If the Ca ion concentration in the cerebrospinal fluid is reduced, in
line with Fisher’s proposal, a decrease in the number of Posner molecules in the
cerebrospinal fluid may occur, and the consciousness level may be lowered
(Fisher, 2015). For the same reason, an elevation in the Ca
concentration in the cerebrospinal fluid may increase consciousness level.
However, our findings in mice contradict these predictions. EGTA reduces
Ca concentration in the cerebrospinal fluid after ICV injection (Liang et al., 2004). Contrary to the prediction that the consciousness level of the
mice would be reduced after ICV injection of EGTA, instead, an increased
consciousness level of the mice, as shown by an increased LORR ED value
for the mice to sevoflurane in comparison to the basal value, was found after the
animals received ICV injection of EGTA in this study. A significantly decreased
consciousness level of the mice was found after ICV injection of
CaCl in this study, again in contradiction to Fisher’s prediction.
Unlike calcium-40 with zero nuclear spin, calcium-43 nuclei have 7/2 nuclear
spins. In Posner molecules, if calcium-43 ions replace the calcium-40 ions,
especially the central ones, the decoherent time for the phosphorus-31
nuclear-spin entanglements is expected to be shortened and thus, the
consciousness level of the animals is expected to reduce - i.e., calcium
isotope dependence on consciousness is expected (Fisher, 2015). Indeed, we
found the consciousness level of the mice was reduced after ICV injections of
CaCl. However, further analysis showed no difference in LORR
ED between ICV injections of CaCl and CaCl
(P 0.9999). The identical effects of CaCl and
CaCl on the LORR ED indicate no calcium isotope dependence on
consciousness.
Our findings are consistent with previous studies (Erickson et al., 1978; Harris, 1979). ICV injection of CaCl increases ethanol-induced sleeping
time, whereas EGTA decreases sleeping time in mice, as reported previously
(Erickson et al., 1978; Harris, 1979). The inhibitory effect of calcium was
also found in in vitro studies, as an increase in extracellular
Ca ion concentration suppressed neuronal excitability, while a decrease in
extracellular Ca concentration excited neurons (Lu et al., 2010; Ma et al., 2012; Wang et al., 2004). Although exploring the mechanisms of varying
calcium concentrations on neural excitability is beyond the scope of this study,
it is necessary to point out that varying calcium ion concentrations affect
Posner molecules the most and, therefore, affect consciousness more than other
possible protein- or ion-involved mechanisms, as Posner molecules are crucial for
consciousness, superseding any other potential mechanisms according to
the proposal (Fisher, 2015). Therefore, our study is vital to test the
predictions derived from Fisher’s proposal (Fisher, 2015) if it overshadows
other possible mechanisms.
Consciousness is a challenging problem (Miller, 2005), both in philosophy
and in science. Philosophically, consciousness belongs only to humans -
consciousness is a feeling of oneness. Scientifically, however, it seems
unjustified to claim that consciousness is uniquely specific to humans
(Trewavas and Baluška, 2011). For living organisms, even a little bit of
consciousness confers sentience and awareness of environment. Since consciousness is a
vital function of living organisms, it merits scientific study. However,
scientific study on consciousness has methodological problems - consciousness is
difficult to define and therefore measure. Fortunately, we have inhaled
anesthetics at hand. The potency of inhaled anesthetics can be quantified
(Koblin et al., 1998). Therefore, consciousness can be quantified by using
inhaled anesthetics in an alternative way.
Our previous study found that the nuclear spin property of xenon partly
antagonizing its anesthetic property is why the less anesthetic potency of
xenon than xenon isotopes with zero nuclear spin (Li et al., 2018).
The existence of isotopic dependence of xenon anesthesia suggests a possible
nuclear-spin mechanism of consciousness in the brain. Therefore, inhaled
anesthetics can be applied to investigate nuclear-spin involving the mechanism of
consciousness. In Fisher’s theory (Fisher, 2015), Posner molecules can be
considered the nuclear-spin property of xenon. In our work, sevoflurane
was the anesthetic property of xenon. The use of sevoflurane to detect
Posner molecules’ effect on consciousness is reasonable since Posner molecules
partially antagonizable effect is predicted to reduce the anesthetic potency of
sevoflurane. Although we disprove Posner molecules in consciousness, it does not
mean that nuclear spin is not crucial for consciousness. Nuclear spin may also be
implied in sleep. The similarities between sleep and general anesthesia exist
(Kelz et al., 2008), as sleep is a natural while general anesthesia is a
drug-induced unconscious state.
5. Conclusions
In conclusion, our findings refute the predictions that phosphorus-31
nuclear-spin entanglements within Posner molecules play a central role in
consciousness.
Author contributions
R. C, N. L., H. Q., and R. Z. performed the experiments. N. L. and R. C.
analyzed the data and wrote the paper, which all authors read. S. Z. conceived
the study, designed the experiments, supervised the overall project. All authors
contributed to editorial changes in the manuscript. All authors read and approved
the final manuscript.
Ethics approval and consent to participate
All animal operations and experimental protocols conformed to the US National
Institutes of Health guide for laboratory animals’ care and use (NIH Publications
No. 8023, revised 1978). They were approved by the Institutional Animal Care and
Use Committee (approval No: S164) at Tongji Medical College, Huazhong University
of Science and Technology.
Acknowledgment
We thank Prof. Ping Yin, Department of Statistics, Tongji Medical College, at
Huazhong University of Science and Technology for statistical assistance. This
work was supported by a grant from the National Natural Science Foundation of
China (grant 81670068) to S. Z.
Conflict of Interest
The authors declare no conflict of interest.
 Fig. 1.
Fig. 1. Fig. 2.
Fig. 2.

