1 School of Chinese Materia Medica, Beijing University of Chinese Medicine, Beijing, 102488, P. R. China
2 School of Chinese Medicine, Beijing University of Chinese Medicine, Beijing, 100029, P. R. China
†These authors contributed equally.
Abstract
Depression is closely linked to hypothalamus-pituitary-adrenal
axis hyperactivity. Honokiol, a biphenolic lignan compound obtained from the
traditional Chinese medicine Magnolia officinalis, can reduce the activity of the
hypothalamus-pituitary-adrenal axis and improve depression-like behavior caused
by hypothalamus-pituitary-adrenal axis hyperactivity. The current study
investigated the specific mechanism of action of this effect. A depression model
was established by repeated injections of corticosterone to study the
antidepressant-like effect of honokiol and its potential mechanism. Honokiol
prevented the elevated activity of the hypothalamus-pituitary-adrenal axis and
the depression-like behavior induced by corticosterone. Treatment with honokiol
resulted in greater glucocorticoid receptor mRNA expression, greater
glucocorticoid receptor-positive expression, and a greater ratio of
glucocorticoid receptor to the mineralocorticoid receptor in the hippocampus.
Moreover, honokiol treatment led to lower levels of interleukin-1
Keywords
- Honokiol
- antidepressant effect
- anti-inflammatory
- glucocorticoid receptor
- hypothalamus-pituitary-adrenal axis
Depression is a mood disorder that is manifested as anhedonia, despair, and pessimism, which has high morbidity, recurrence, and mortality rates (LeMoult and Gotlib, 2019). Many studies have reported that the hypothalamus-pituitary-adrenal (HPA) axis hyperactivity is closely linked to depression, indicated the need for it to be considered in studies of depression etiology (Galts et al., 2019; Karstens et al., 2019). Clinical studies have found that the HPA axis of many patients with depression are hyperactive and return to a normal state following antidepressant treatment (Høifødt et al., 2019). Also, depression causes an increase in the adrenal index and the volume of the pituitary and adrenal glands, and a decrease in the volume of the hippocampus (Gong et al., 2016). Related animal experiments also indicate that a high dose of glucocorticoids (GC) induces depression-like behavior in rodents (Raone et al., 2007).
As the regulatory center of the HPA axis, the hippocampus mainly maintains the
normal activity of the HPA axis through the adjustment of mineralocorticoid
receptor (MR) and the negative feedback of the glucocorticoid receptor (GR)
(Mifsud and Reul, 2018). Chronic stress may damage this process and cause HPA
axis hyperactivity. This HPA axis hyperactivity can also cause damage to the
hippocampus, inducing a maladaptive cycle. This may represent the key underlying
mechanism of depression (Kim et al., 2015). Also, depression is often
accompanied by inflammatory symptoms, notably increased levels of
interleukin-1
Honokiol (3,5-di-2-propenyl-1,1-biphenyl-2,4-diol), a biphenolic lignan compound, is the main bioactive component of Magnolia officinalis(Talarek et al., 2017). Previous research has demonstrated that honokiol may play an important role in improving depression-like behavior in rats, such as shortening the immobility time in the forced swimming test (FST) and tail suspension test (TST) (Zhang et al., 2019). Recent studies conclude that honokiol has antidepressant effects on chronic unpredictable mild stress exposed rats by regulating HPA axis activity (Li et al., 2020; Pitta et al., 2013). We further investigated the potential mechanism of honokiol in a rodent model of depression induced by repeated corticosterone (CORT) injections, which disrupts the HPA axis.
The animals used in the experiment were adult male Sprague-Dawley rats, all of
which were specific pathogen-free (SPF) grade. The animal license number is SCXK
(Beijing) 2014-0004, purchased from Beijing HuaFukang Biotechnology Co., Ltd.
Animals were kept under standard conditions of ambient temperature (20-22
The forty rats were randomly divided into four groups after acclimating for one week, with ten rats in each group. There was a control group, CORT group, honokiol-treated group (10 mg/kg honokiol + CORT) and a fluoxetine-treated group (10 mg/kg fluoxetine + CORT). We established the fluoxetine-treated group as the positive control group. Except for the control group, the other three groups of rats received subcutaneously CORT injection (40 mg/kg, suspended in saline containing 0.1% dimethyl sulfoxide and 0.1% Tween-80) once daily for 21 days. At the same time, an equal volume of vehicle was subcutaneously injected in the control group. Honokiol and fluoxetine were suspended in 0.5% sodium carboxymethyl cellulose (CMC-Na) 30 minutes before the injection of corticosterone. In the drug-treated groups, rats were intragastrically administrated with 10 ml/kg volume of honokiol or fluoxetine once a day. In the normal control group and the CORT groups, rats received 0.5% CMC-Na solution in equal volume.
Based on our previous studies and related published reports, we chose established doses of CORT, honokiol, and fluoxetine (Gregus et al., 2005; Wang et al., 2017, 2018). In our previous study (Wang et al., 2017), we used three dosages (2.5, 5, and 10 mg/kg) to study the antidepressant effects of honokiol and its mechanism. Our results indicate that honokiol can improve the depression-like behavior of mice and has a specific antidepressant effect. We found that high-dose honokiol (10 mg/kg) had the most significant effect. Based on this, we conduct the current experiment. Behavioral tests were completed after the last dose on day 21. The rats were given 1% pentobarbital intraperitoneally for anesthesia after the behavioral tests, and then blood samples were collected. After collecting the blood, we removed the brain of each rat and stored it on ice for analysis.
The flow of experimental design is shown in (Fig. 1). The figure clearly describes the experimental design, including grouping, drug administration, behavioral testing, and biochemical parameters detected.
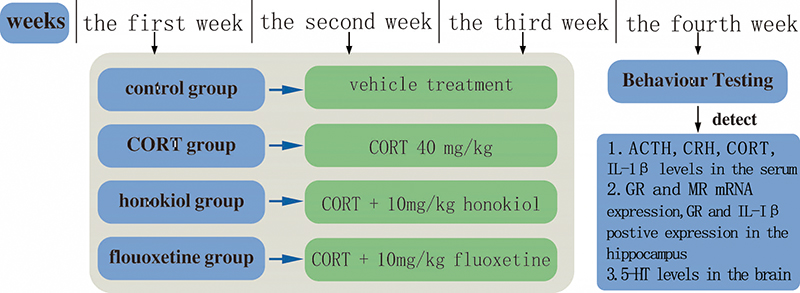 Fig. 1.
Fig. 1.A detailed schedule of the experimental design. The CORT group, honokiol-treated group, and fluoxetine-treated group received a subcutaneous injection of CORT every day for 21 days. The control group was injected with an equal volume of vehicle subcutaneously. In the honokiol-treated group, rats were intragastrically administrated with honokiol once a day. In the fluoxetine-treated group, rats were intragastrically administrated with fluoxetine. The behavioral tests were after the last dose on day 21. The rats were then sacrificed to evaluate the relevant biochemical parameters. Abbreviations: ACTH, adrenocorticotropic hormone; CRH, corticotrophin-releasing hormone; CORT, corticosterone; GR, glucocorticoid receptor; MR, mineralocorticoid receptor; IL-1β, interleukin-1β; 5-HT, 5-hydroxytryptamine.
Fluoxetine (Patheon, France), honokiol (Shanghai Titan Scientific Co., Ltd.,
Shanghai, P. R. China), corticosterone (Chengdu Chroma-Biotechnology Co., Ltd.,
Chengdu, P. R. China), interleukin-1
The sucrose preference test (SPT) is an effective method to measure anhedonia in rodents (Eagle et al., 2016). We modified the SPT based on the previous reference. In brief, we trained all rats to consume sucrose solution in the following ways before the formal test: all rats were given a bottle of 1% (w/v) sucrose water and a bottle of drinking water to acclimate them to the choice of two bottles in the test. 24 hours before the start of the formal test, rats were deprived of water and food. Subsequently, 100 mL 1% sucrose solution and 100 mL drinking water were placed in each cage, and individual rats could enter the cage freely. 3 hours later, the consumption levels of the sucrose solution and water were recorded, respectively. The sucrose preference was calculated as follows: the sucrose preference rate (%) = sucrose consumption/(sucrose consumption + water consumption).
The open-field test (OFT) is a method of measuring the general locomotor and
exploratory behaviors of rats (Ramos and Mormède, 1997). Following the
previously used protocols, a quiet, light-stable environment was selected, and
the rats were placed in a four-sided black wooden box with dimensions of 100 cm
On the 22nd day, blood samples were collected by the abdominal aortic method at
9:00-11:00 a.m., cooled on ice as soon as possible, and centrifuged at 4
Following the behavioral testing, blood samples were collected by an abdominal
aortic method, immediately cooled on ice, and centrifuged at 4
The rats were decapitated, and their brains were quickly taken out and placed on
ice. Then the cerebral cortex and hippocampus were dissected. After separation,
the cerebral cortex was put in a centrifuge tube containing 4
The hippocampus of five rats from each group was dissected from the brains.
Based on the Hipure Total RNA mini kit instructions, the total RNA was extracted.
The ultraviolet spectrophotometer (UV-2000, Unico, Shanghai, P. R. China) was
used to detect the RNA concentration. We used the RevertAid first-strand cDNA
synthesis kit to synthesize cDNA. According to the manufacturers’ instructions,
we used the cDNA and the SYBR PCR master mix to conduct the reverse transcription
on the T100 Thermal Cycler PCR machine (Bio-Rad, USA). Amplification and
quantitative detection were performed in a Real-Time PCR machine (Bio-Rad, USA).
The RT-PCR protocol was as follows: initial denaturation for 10 min at 95
MR F: 5’ CAAGGAGCCATCGGTGAACT 3’
MR R: 5’ AGAGGAGTTGGCTGTTCGTG 3’ (341bp);
GR F: 5’ GGCTTCTGTCTAGAATATGCCTG 3’
GR R: 5’ GATTTGTCCCCATTATATAGCCTT 3’ (156bp);
The 2
The other five rats per group were perfused with icy phosphate buffered-saline
(PBS) transcardially, and their brains were fixed in 4% paraformaldehyde at 4
Data were expressed as mean
SPT is one of the most commonly used behavioral tests for depression. We applied
the SPT to assess the antidepressant effect of honokiol on corticosterone-induced
rats exhibiting depressive behavior. Compared with the control group, the sucrose
preference of the CORT group was significantly lower (P
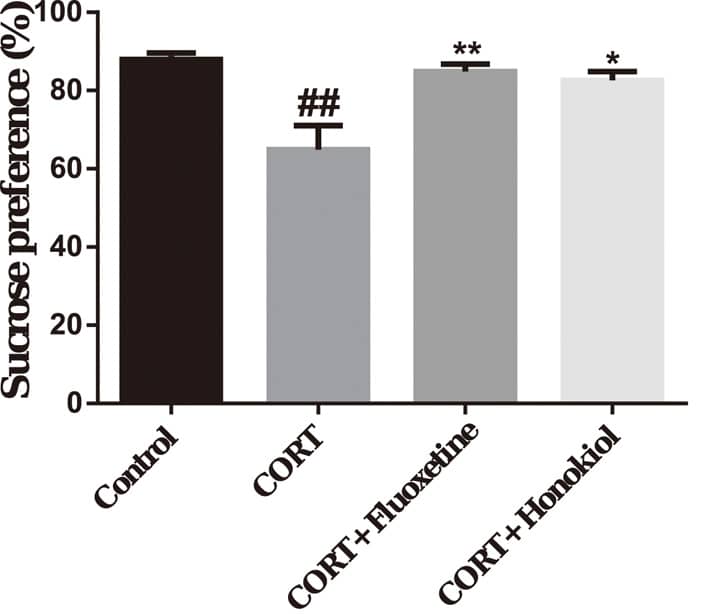 Fig. 2.
Fig. 2.The effects of honokiol on the sucrose
preference test. Honokiol can increase the sucrose preference rate of
corticosterone-induced depressive rats. Data are expressed as mean
OFT is another commonly used behavioral test for depressive behavior. Compared
with the control group, the horizontal movement score, vertical movement score,
and total movement score of the CORT group were significantly lower (P
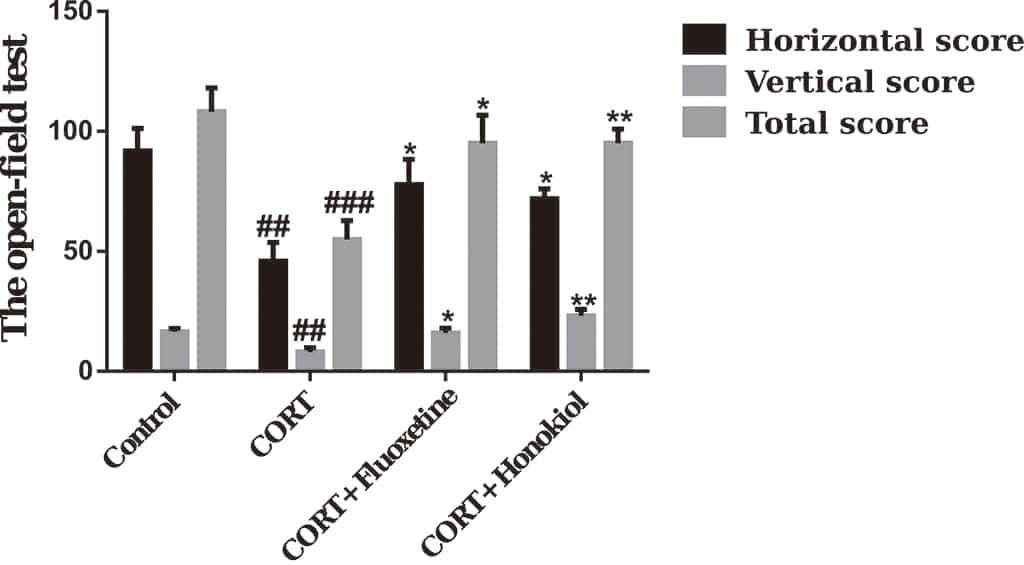 Fig. 3.
Fig. 3. The effects of honokiol on the open field test.
Honokiol can increase the horizontal movement score, vertical movement score, and
total movement score of corticosterone-induced depressive rats. Data are
expressed as mean
To measure the regulatory effects of honokiol on the HPA axis, the
concentrations of ACTH, CORT, and CRH in serum were determined by
radioimmunoassay. Compared with the control group, the serum levels of ACTH,
CORT, and CRH were significantly greater in the CORT group (CORT and CRH: P
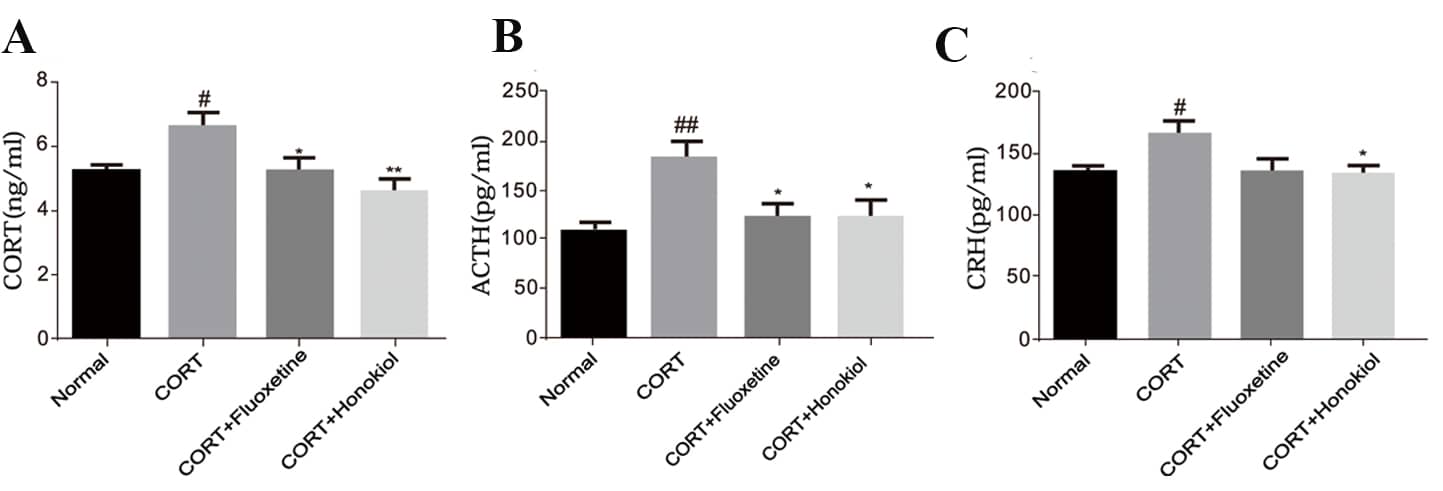 Fig. 4.
Fig. 4.The effects of honokiol on adrenocorticotropic hormone (ACTH), corticosterone (CORT), corticotrophin-releasing hormone (CRH) levels in serum. Honokiol can significantly reduce the levels of CORT, ACTH, and CRH in the serum. (A) levels of CORT in serum; (B) levels of ACTH in serum; (C) levels of CRH in serum. Data are expressed as mean ± SEM (n = 10). **P
The serum concentrations of IL-1
The content of 5-HT in the brain tissue of the CORT group was significantly
lower in comparison with the control group (P
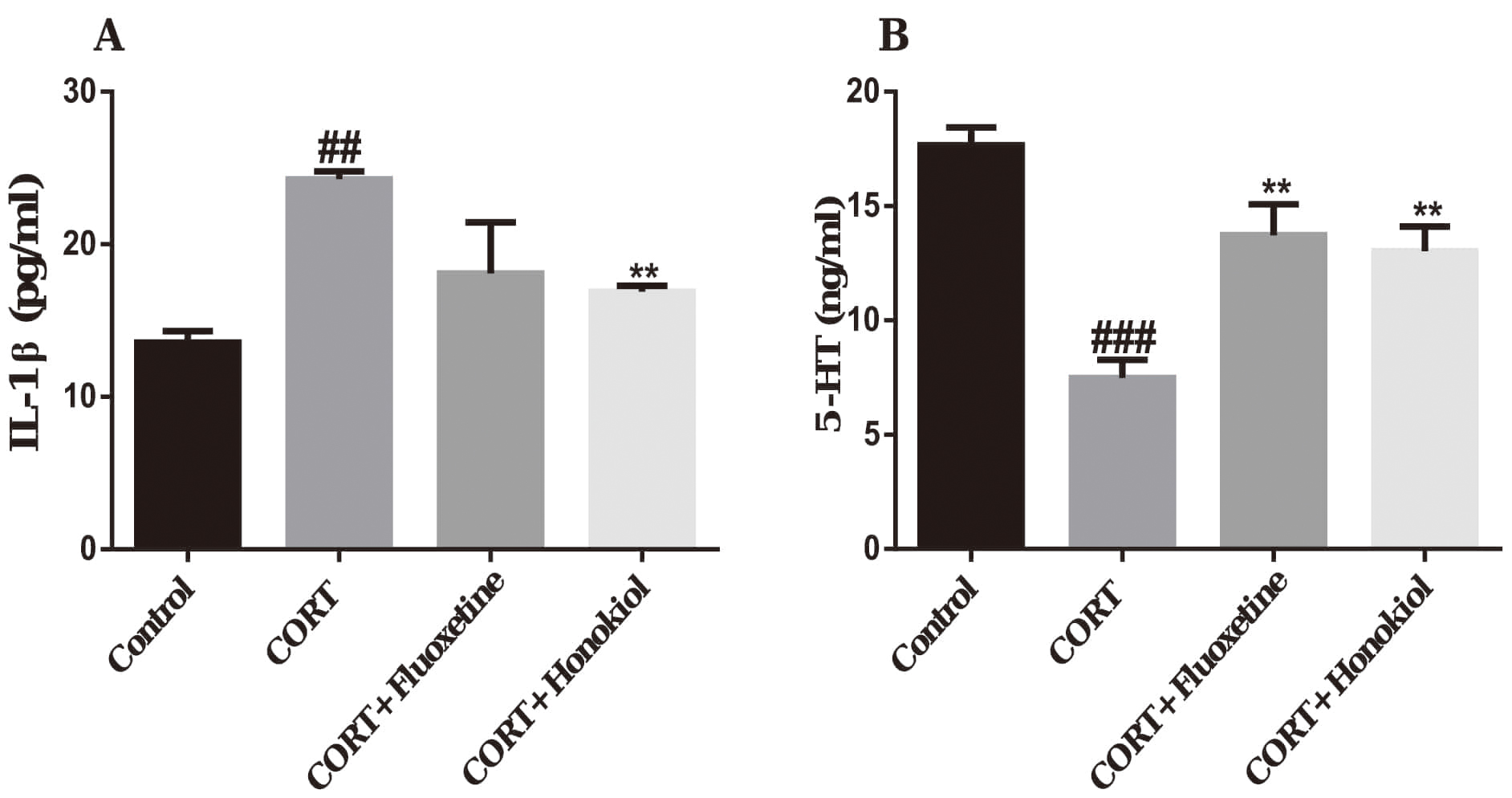 Fig. 5.
Fig. 5.The effects of honokiol on interleukin-1
Real-time quantitative PCR was used to detect the expression of GR and MR mRNA in the hippocampus to assess whether corticosterone-induced depression involved the involvement of GR and MR.
In the CORT group, the expression of GR mRNA in the hippocampus was
significantly lower than in the control group (P
The mRNA expression of MR in the hippocampus was significantly higher in the
CORT group compared with the control group (P
The GR/MR ratio in the CORT group rats was significantly lower than the control
group (P
The above results indicate that honokiol normalizes the HPA axis function in CORT treated rats by mediating the expression of MR and GR in the hippocampus.
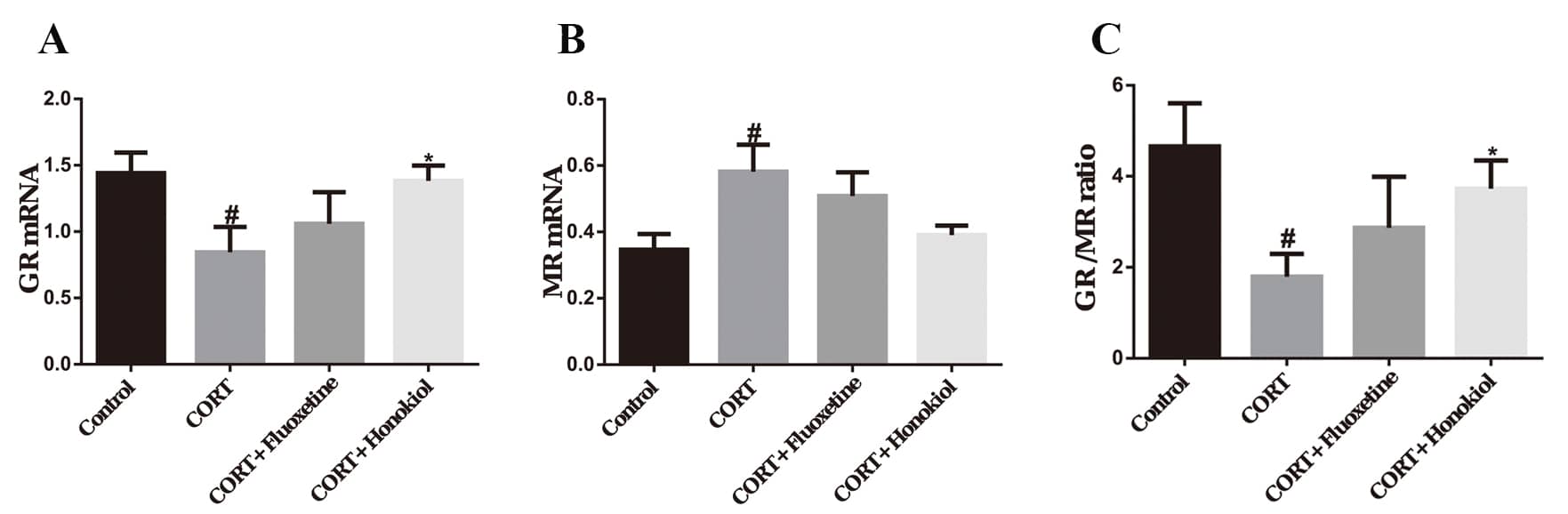 Fig. 6.
Fig. 6.The effects of honokiol on mRNA expression of
hippocampal GR and MR. (A) mRNA expression of GR in the hippocampus; (B) mRNA
expression of MR in the hippocampus; (C) The GR/MR ratio in the hippocampus. Data
are expressed as mean
Immunohistochemistry was used to assess the expression of GR and IL-1
The expression of IL-1
These results indicate that honokiol can effectively inhibit the GR reduction caused by corticosterone administration. Also, honokiol has a significant anti-inflammatory effect.
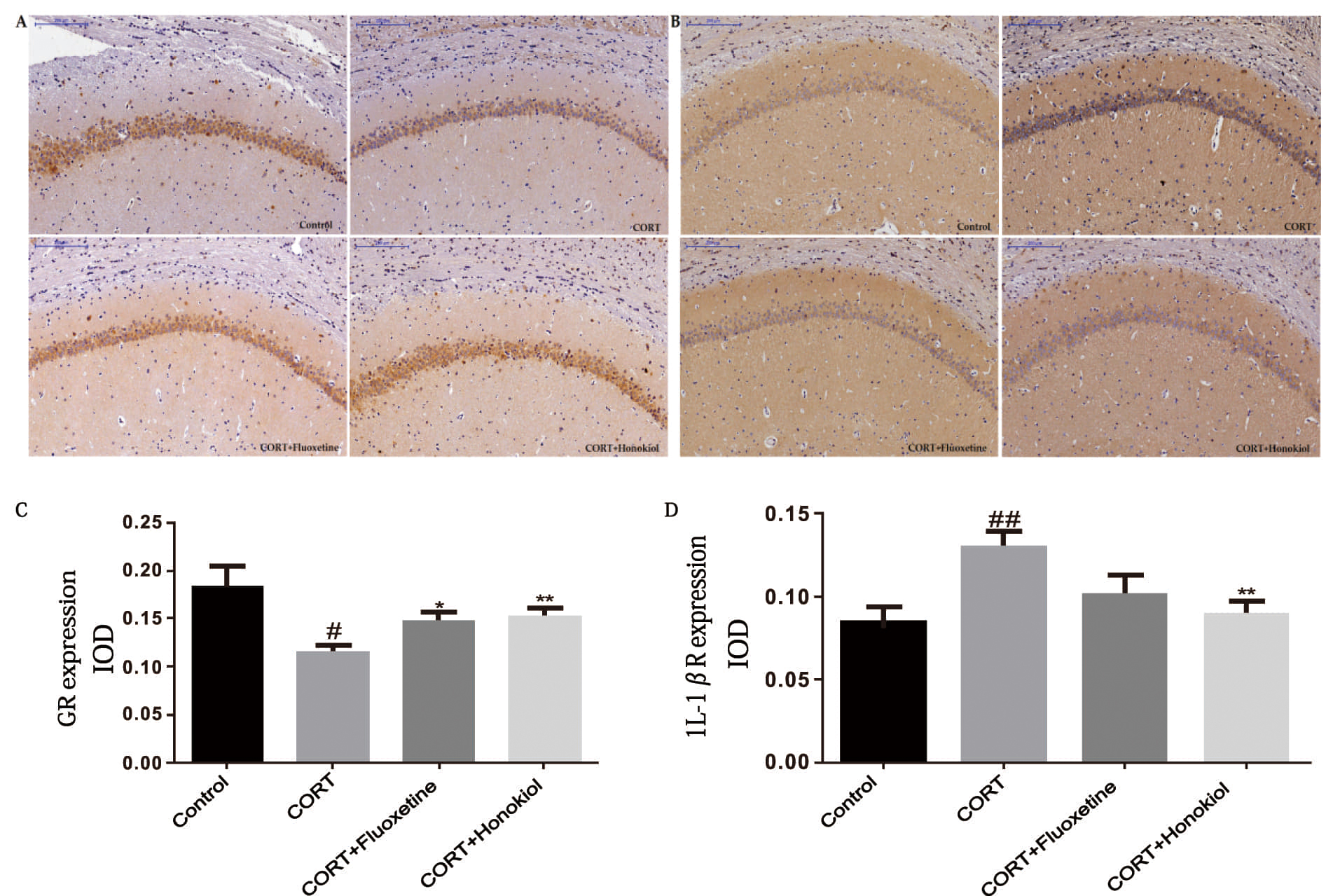 Fig. 7.
Fig. 7.The effects of honokiol on the expression of GR and
IL-1
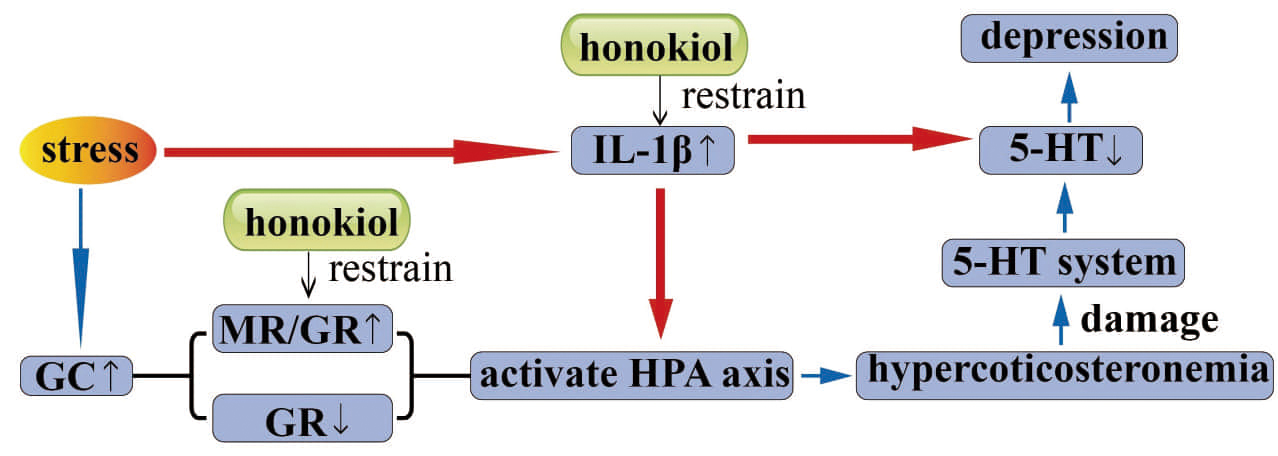 Fig. 8.
Fig. 8.Graphical abstract. Honokiol regulates HPA axis activity by restoring the GR-mediated negative feedback mechanism and regulating the balance between GR and MR, thereby exerting an antidepressant effect. The antidepressant effect is also related to the suppression of inflammatory responses. CORT, corticosterone; GC, glucocorticoids; MR, mineralocorticoid receptor; GR, glucocorticoid receptor; HPA, hypothalamus-pituitary-adrenal axis; IL-1β, interleukin-1β; 5-HT, 5-hydroxytryptamine.
The rodent CORT induced model of depression is suitable for exploring the molecular mechanisms of depression related to HPA axis dysfunction and can simulate depression caused by exposure to stress in humans (Gourley et al., 2013; Thibaut, 2019). The sucrose preference and exploratory activity significantly decreased after the rats received repeated corticosterone injections. These results indicate the successful application of the CORT induced depression model. Additionally, sucrose preference and exploratory activity increased after treatment with honokiol, demonstrating honokiol has antidepressant-like effects on depressed rats. Honokiol can ameliorate depression-like effects of chronic CORT, similar to the widely used antidepressant, fluoxetine.
HPA axis dysfunction is a critical etiological factor in depression. The current data support these findings, as the corticosterone group exhibits HPA axis hyperactivity and depressive-like behavior compared with the control group. The levels of CORT, ACTH, and CRH in the serum of depressed rats caused by repeated corticosterone injections were higher than those of the control group but were lower after treatment with honokiol. CORT, ACTH, and CRH are all hormones essential to the regulation of the HPA axis. The HPA axis can maintain a steady-state of the internal environment through the negative feedback mechanism (van Bodegom et al., 2017). When the body is under intense stress, the HPA axis is stimulated, and neurons in the paraventricular nucleus of the hypothalamus synthesize and secrete CRH. CRH is delivered to the anterior pituitary by the portal vein system of the pituitary tract and stimulates ACTH release by adrenocorticotropic hormone cells. ACTH reaches the adrenal gland through systemic circulation and promotes the release of GC from the adrenal gland (cortisol in primates, corticosterone in rodents) (Ulrich-Lai and Herman, 2009; Watson and Mackin, 2006). Our results indicate that the HPA axis activity of depressed rats is increased, and honokiol is likely to improve depression-like behavior by reducing levels of CORT, ACTH, and CRH. Abnormal elevations of CORT, ACTH, and CRH can induce depressive symptoms and signs (Sun et al., 2020). This may be due to an impairment of learning and memory caused by the increase of CRH and ACTH, as excessive exposure to CORT can cause learning deficits and emotional instability.
The mRNA expression of GR in the hippocampus of depressed rats was lower than controls but returned to control levels after treatment with honokiol. These results indicate that honokiol can reduce HPA axis activity by regulating GC and GR mediated negative feedback to exert antidepressant effects in depressed rats. The hippocampus is a key regulatory center of HPA axis activity and the stress response, which can mediate the recovery from hyperactive HPA axis activity resulting from chronic stress (Belleau et al., 2019). There are two steroid hormone receptors in the hippocampus, GR and MR. MR has a strong affinity for GC, and its expression in the central nervous system is mainly concentrated in the marginal area. It maintains the basal level of cortisol in circulation when the body is in a normal state (Micale and Drago, 2018). GR has a much weaker affinity for GC than MR. GR contributes to the circadian secretion of cortisol and the regulation of stress-related cortisol secretion (Odeon et al., 2017). The hippocampus maintains HPA axis activity stability through the regulation of MR and the negative feedback mechanism mediated by GR and GC. The body exhibits HPA axis hyperactivity and secretes large amounts of glucocorticoids when it is subjected to chronic stress.
Furthermore, glucocorticoids will be maintained at high levels during chronic stress exposure. The high concentration of glucocorticoids repeatedly stimulates GRs, inducing GR dysfunction, with deficits in the negative feedback mechanism. The result is ongoing HPA axis hyperactivity and damage to the hippocampus, inducing a maladaptive cycle (Bellavance and Rivest, 2014; Gądek-Michalska et al., 2013). This process is considered to be the most substantial physiological and biochemical basis of stress-induced depression. In general, GR-mediated negative feedback regulation plays a vital role in maintaining the typical activity of the HPA axis. Repeated corticosterone injections can reduce GR mRNA expression in the hippocampus in rats, accompanied by HPA axis hyperactivity and depression-like behavior. However, these adverse effects of CORT are successfully treated with honokiol. This indicates that the antidepressant effects of honokiol are related to the restoration of GR mediated negative feedback.
The results also demonstrated that the ratio of GR/MR in depressed rats was significantly lower, which was ameliorated with honokiol. These results suggest that honokiol can restore HPA axis activity by regulating the balance between GR and MR, and exhibits an antidepressant-like effect in depressed rats. The ratio of GR/MR plays a vital role in neuronal excitement, stress response, and adaptability to stress, and the pathogenesis of depression. The regulatory functions of the two types of receptors are closely interrelated (Pariante and Miller, 2001). Studies have shown that defects in MR can cause a more rapid corticosterone response and produce a more pronounced GR-mediated effect.
Moreover, the imbalance of the GR/MR ratio stimulates hippocampal neurons to transition to a vulnerable state where they are prone to stress-related diseases, including depression (de Kloet et al., 2018; Lucassen et al., 2001). An imbalance in GR and MR may result in damage to the hippocampus. The hippocampus itself has an inhibitory effect on the HPA axis activity, and this may be a factor in increased HPA axis activity. Repeated injections of corticosterone can cause a decrease in the GR/MR ratio in rats, accompanied by depression-like behaviors. After treatment with honokiol treatment prevents the development of this pathological state. Therefore, the mechanism of the antidepressant effect of honokiol may be related to regulating the balance between GR and MR.
Damaged to the GR negative feedback mechanism and GR/MR imbalance will result in excessive activation of the HPA axis, resulting in increased CORT, which is the final metabolite of the HPA axis, leading to hypercortisonemia. There is a key functional interaction between the HPA axis and the 5-HT system. Elevated CORT levels can damage the 5-HT neural pathway of the raphe nucleus-hippocampus system, resulting in an array of depression symptoms such as depression, inattention, memory loss, and insomnia (Pérez-Ortiz et al., 2013; Snyder et al., 2011).
The antidepressant effect of honokiol is likely to be related to its
anti-inflammatory effect. Repeated corticosterone injections can increase
IL-1
The potential mechanisms by which honokiol exerts antidepressant-like effects in the CORT induced rat model of depression are shown in (Fig. 8).
In conclusion, the results reveal that honokiol has significant antidepressant-like effects on corticosterone induced depressive rats. The mechanism of these effects may be that honokiol regulates HPA axis activity by restoring the GR-mediated negative feedback mechanism and regulating the balance between GR and MR. This antidepressant-like effect may also be related to the suppression of inflammatory responses.
The Experimental Animal Ethics Committee of the Academic Committee of Beijing University of Chinese Medicine approved all the steps involving animals in the experiment (project identification code: BUCM-4-2019021502-1012).
This project was supported by the National Natural Science Foundation of China (No. 81373584).
The authors declare no competing interests.








